A Novel Hybrid Drug Delivery System for Treatment of Aortic Aneurysms
Abstract
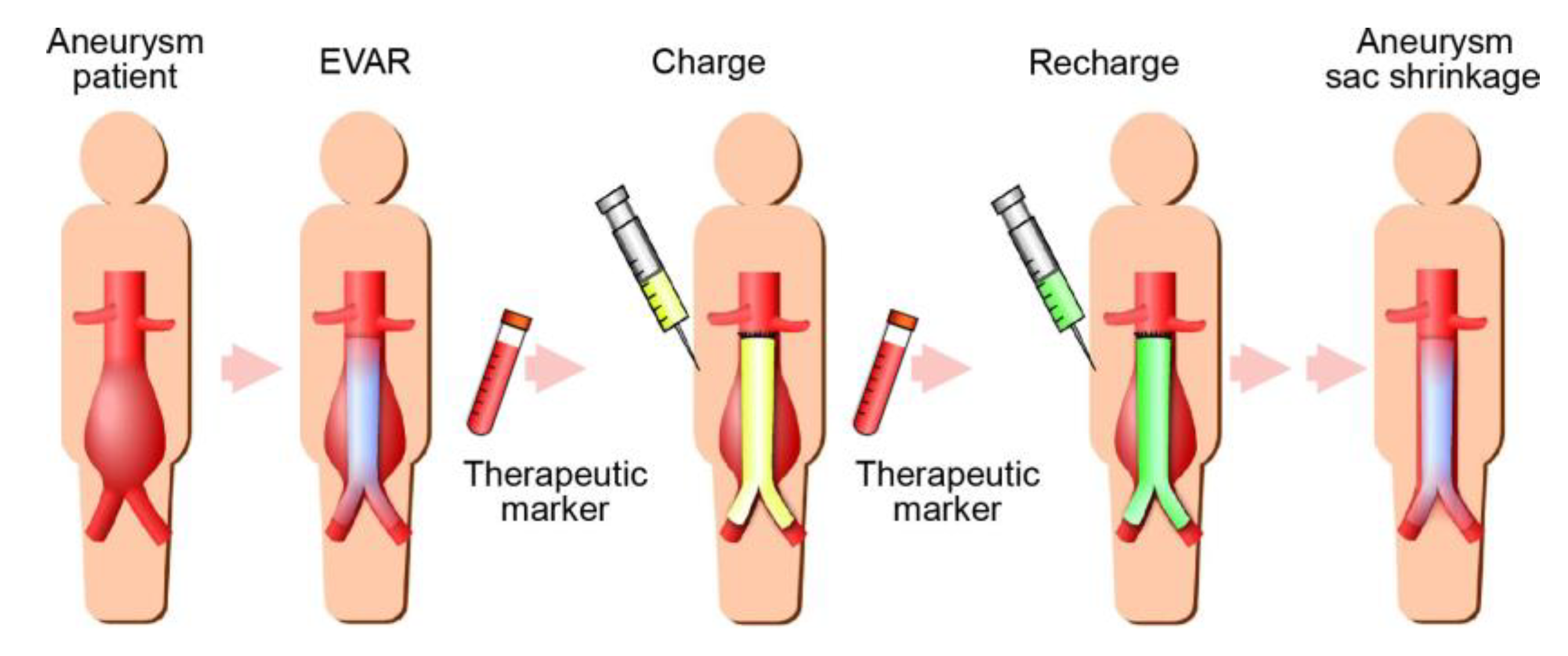
:1. Introduction
2. Results and Discussion
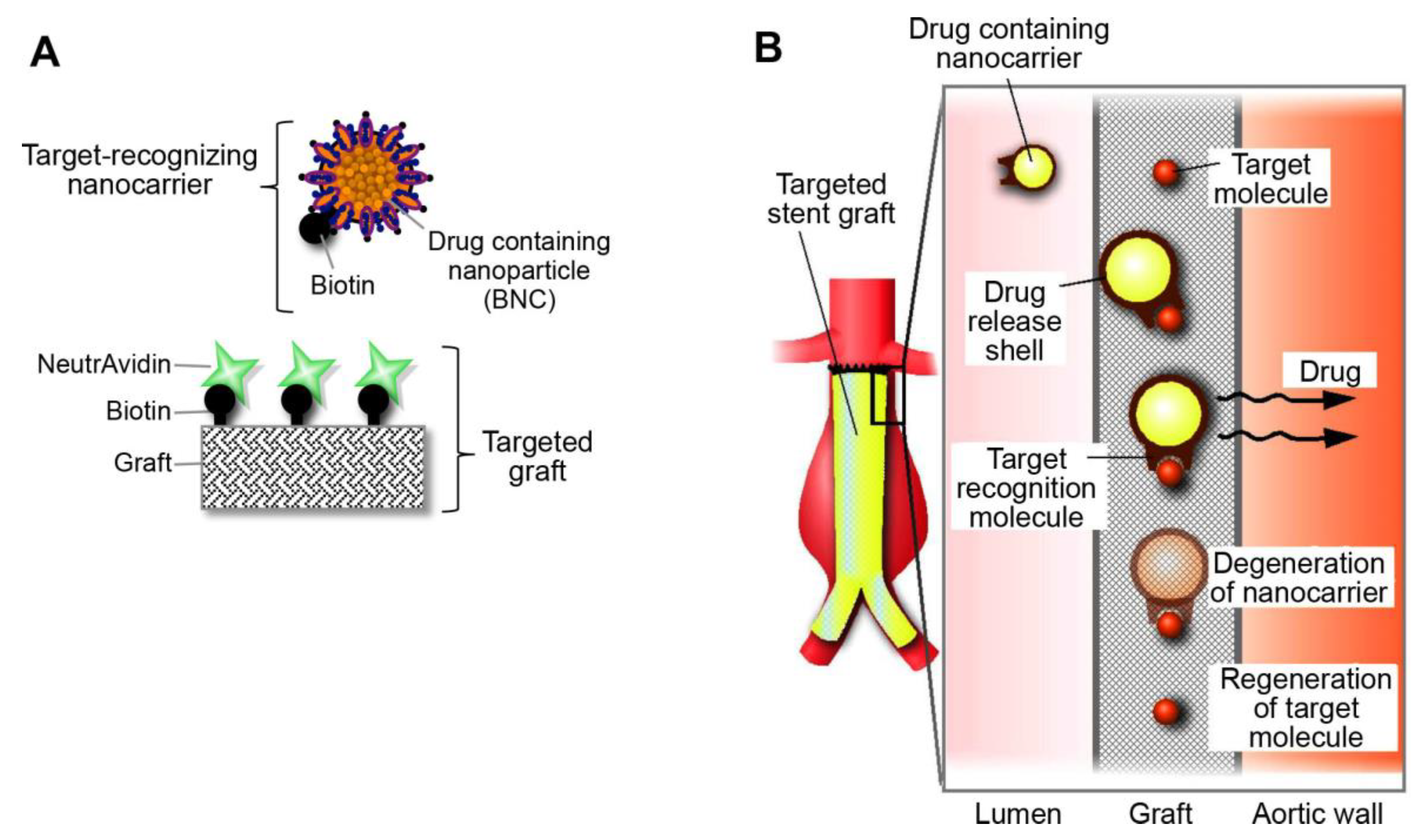
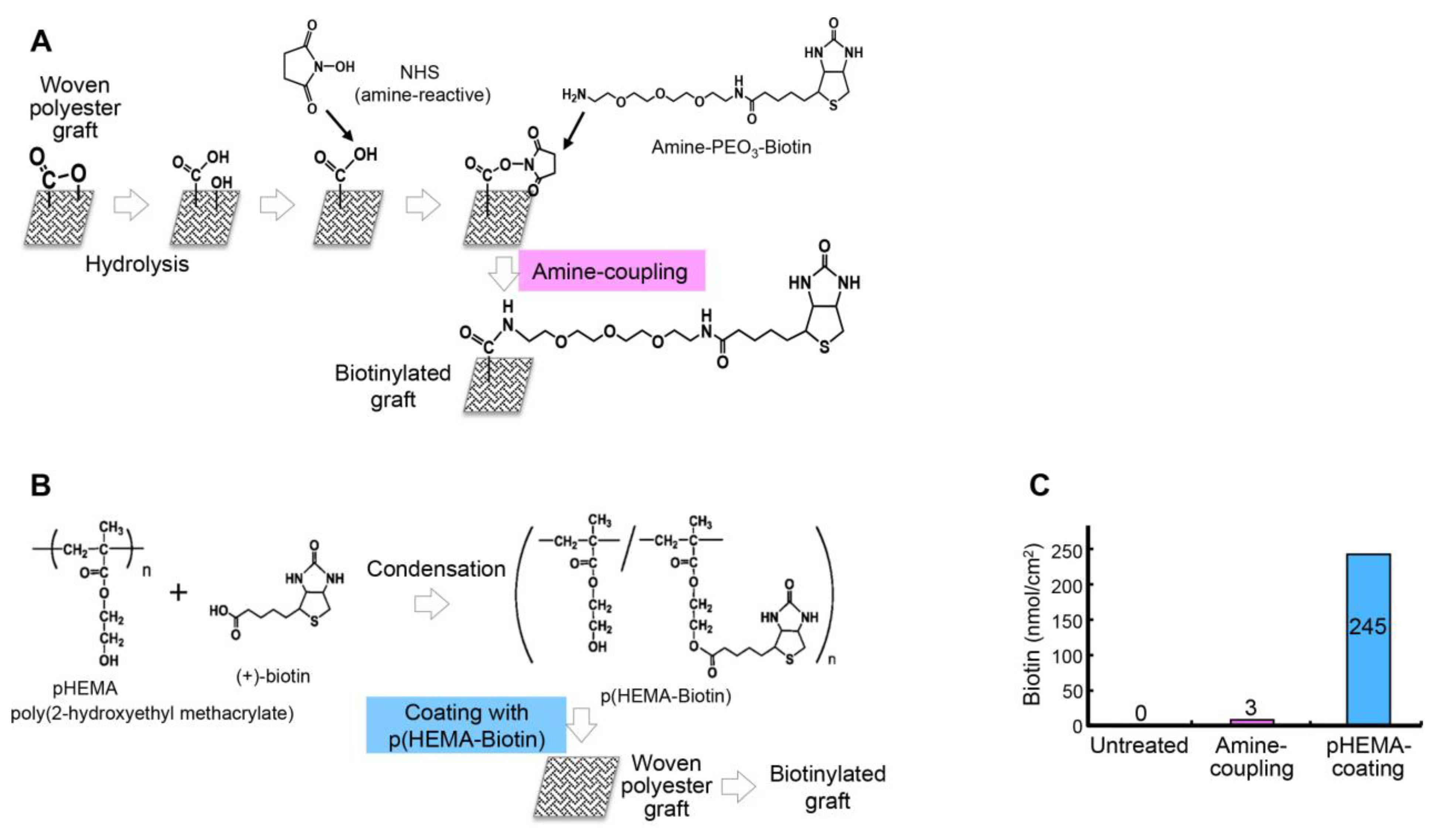
2.1. Development of the Targeted Graft
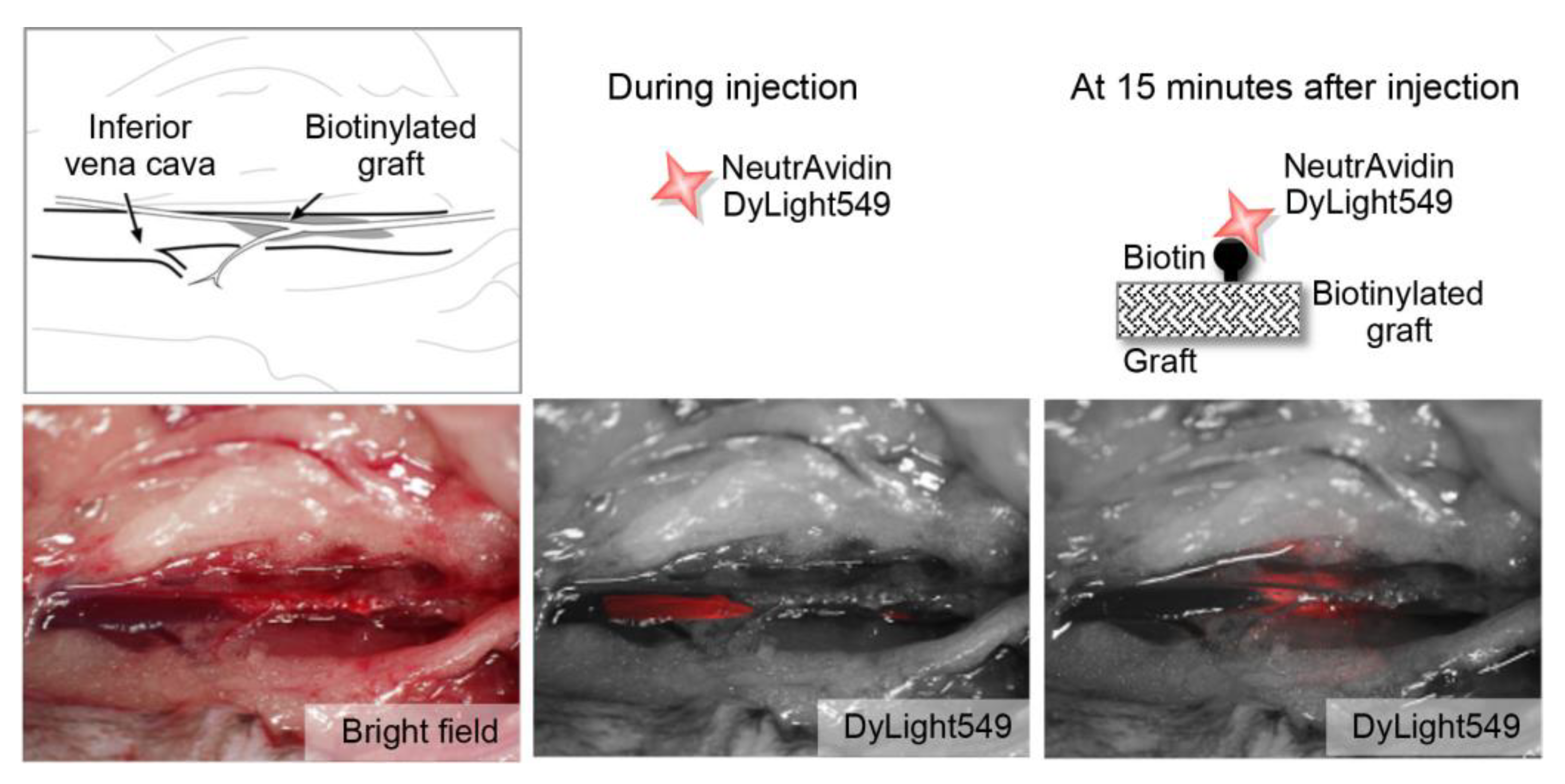
2.2. Feasibility of Using the Targeted Graft in Mouse Blood Vessels
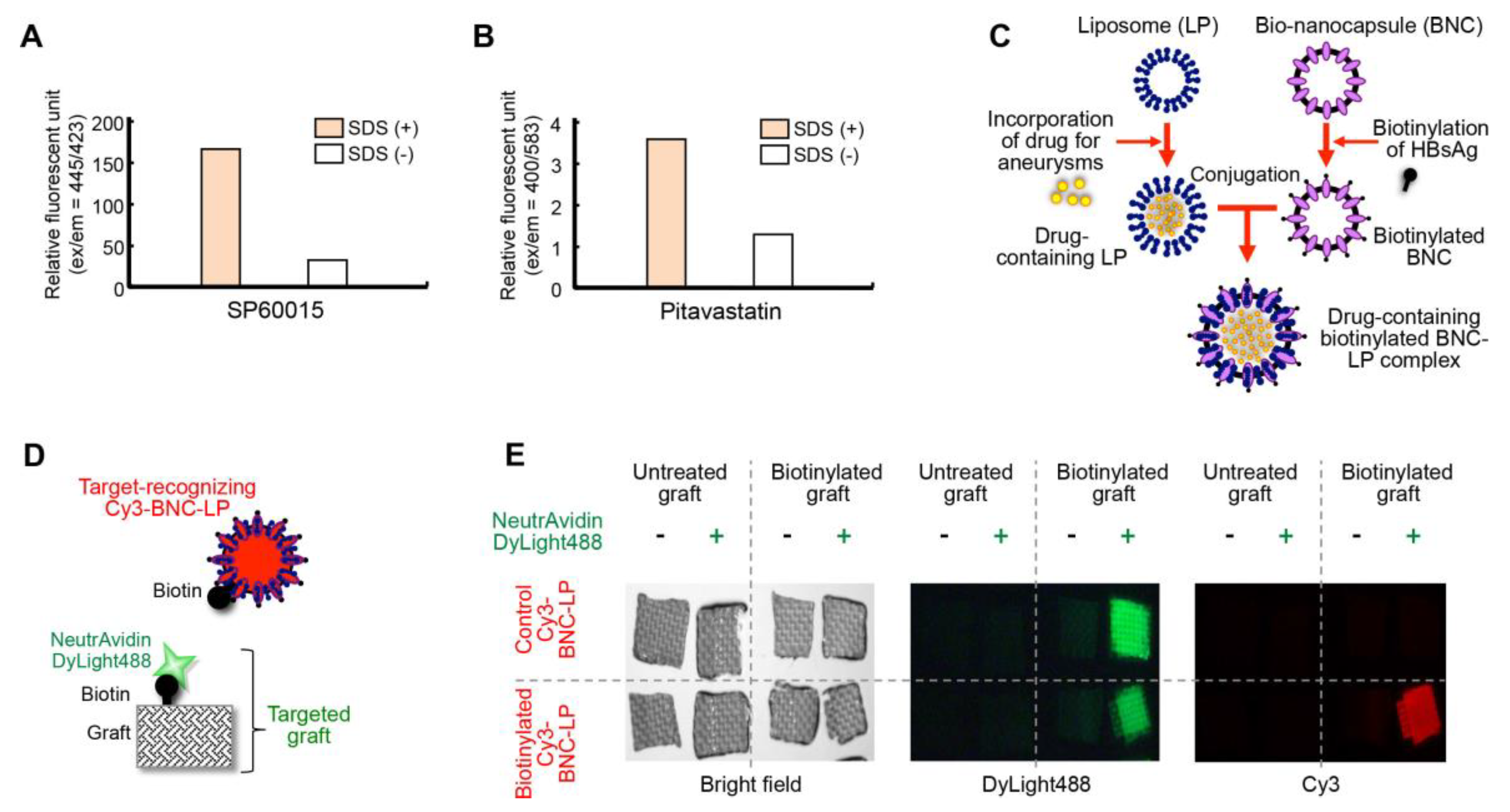
2.3. Development of the Bio-Nanocapsule-Liposome Complex
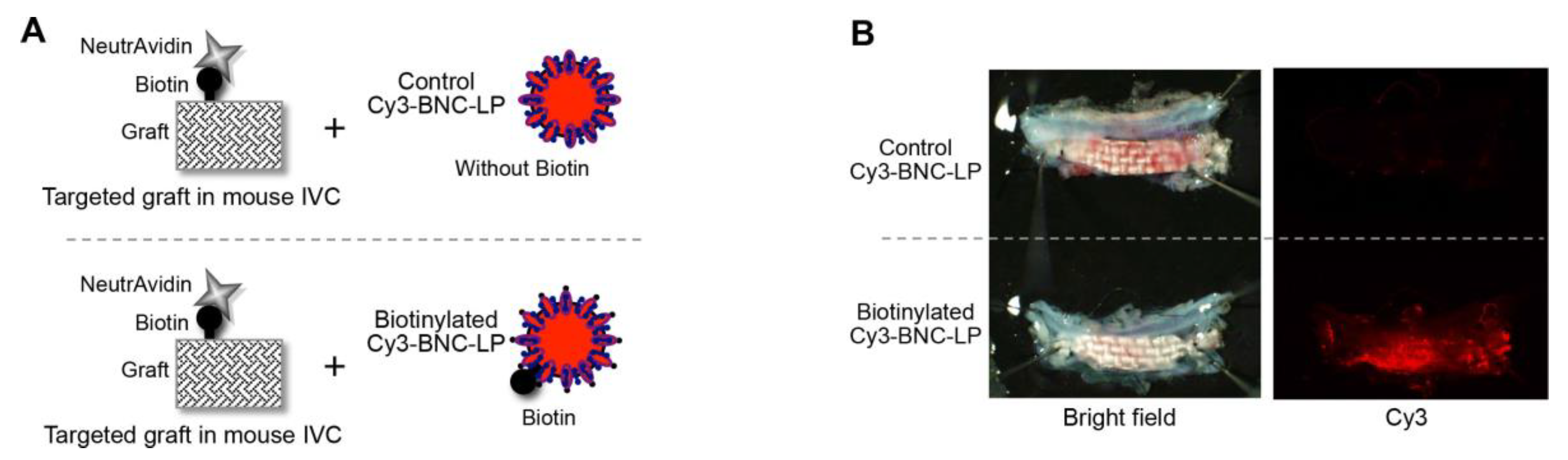
2.4. Accumulation of the Target-Recognizing BNC-LP Complex on the Targeted Graft in Mouse Blood Vessels
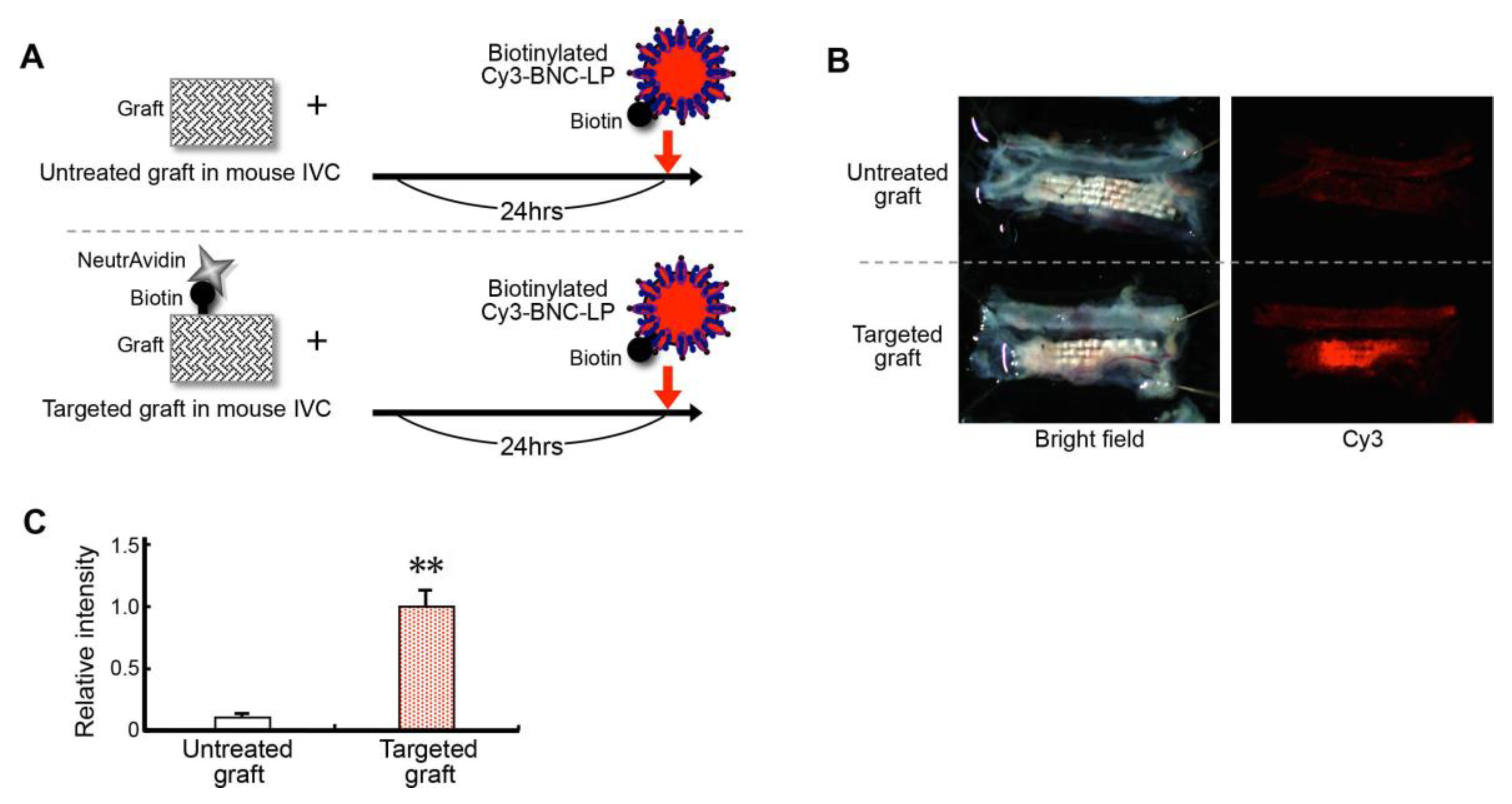
2.5. Efficiency of Loading (Charging) the Targeted Graft with the Target-Recognizing BNC-LP Complex In Vivo
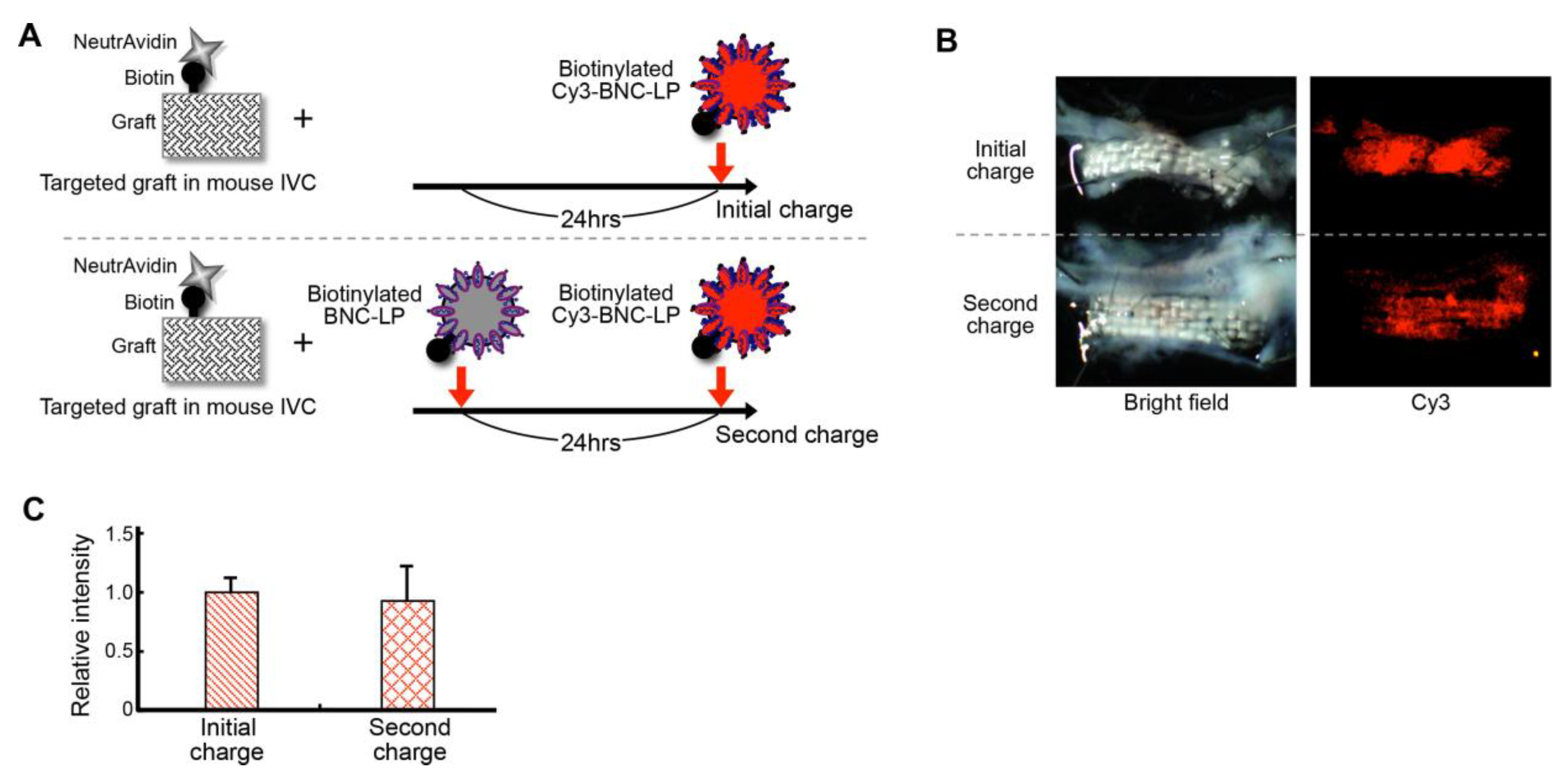
2.6. Efficiency of Recharging the Targeted Graft with the Target-Recognizing BNC-LP Complex In Vivo
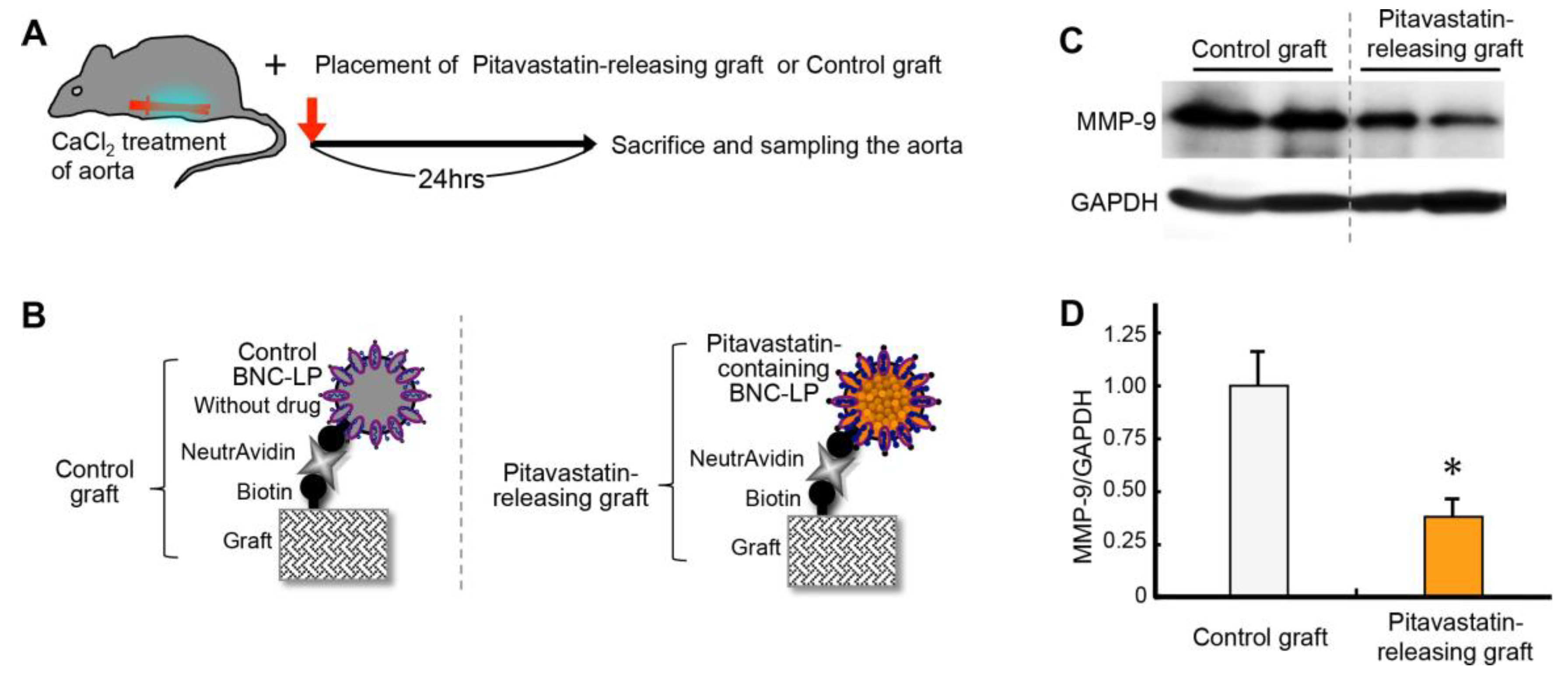
2.7. Effect of Releasing Drug from the Graft Charged with BNC-LP Complexes
2.8. Summary of the Results
2.9. Clinical Implications and Future Directions
3. Materials and Methods
3.1. Biotinylation of a Graft Surface and In Vitro Detection of Biotin
3.2. Verification of the In Vivo Interaction Between Biotin and Neutravidin
3.3. Drug Encapsulation in Liposomes
3.4. Preparation of the Biotinylated BNC-LP Complex
3.5. Binding Assay In Vitro
3.6. Binding Assay In Vivo
3.7. Assay for Charging the Targeted Graft with the Target-Recognizing BNC-LP Complex In Vivo
3.8. Assay for Recharging the Targeted Graft with the Target-Recognizing BNC-LP Complex In Vivo
3.9. Assay for Releasing Drug from the Graft Charged with BNC-LP Complex In Vivo
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAA | Abdominal aortic aneurysm |
| BNC | Bio-nanocapsule |
| BNC-LP EDC EVAR GAPDH HEPES JNK MMP NHS PBS pHEMA RDDS SDS TBS-T | Bio-nanocapsule-liposome 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride Endovascular aneurysm repair Glyceraldehyde 3-phosphate dehydrogenase 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid c-Jun N-terminal kinase Matrix metalloproteinase N-hydroxysuccinimide Phosphate buffered saline poly (2-hydroxyethyl methacrylate) Rechargeable drug delivery system Sodium dodecyl sulfate Tris-buffered saline with Tween20 |
References
- Sakalihasan, N.; Michel, J.B.; Katsargyris, A.; Kuivaniemi, H.; Defraigne, J.O.; Nchimi, A.; Powell, J.T.; Yoshimura, K.; Hultgren, R. Abdominal aortic aneurysms. Nat. Rev. Dis. Primers 2018, 4, 34. [Google Scholar] [CrossRef]
- Erbel, R.; Aboyans, V.; Boileau, C.; Bossone, E.; Bartolomeo, R.D.; Eggebrecht, H.; Evangelista, A.; Falk, V.; Frank, H.; Gaemperli, O.; et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. Eur. Heart J. 2014, 35, 2873–2926. [Google Scholar]
- Yoshimura, K.; Aoki, H. Recent advances in pharmacotherapy development for abdominal aortic aneurysm. Int. J. Vasc. Med. 2012, 2012, 648167. [Google Scholar] [CrossRef]
- Golledge, J. Abdominal aortic aneurysm: Update on pathogenesis and medical treatments. Nat. Rev. Cardiol. 2019, 16, 225–242. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Aoki, H.; Ikeda, Y.; Furutani, A.; Hamano, K.; Matsuzaki, M. Development of pharmacological therapy for abdominal aortic aneurysms based on animal studies. In Aortic Aneurysms, New Insights into an Old Problem; Sakalihasan, N., Ed.; Édition de I’Université de Liège: Liège, Belgium, 2008; pp. 453–476. [Google Scholar]
- Yoshimura, K.; Nagasawa, A.; Kudo, J.; Onoda, M.; Morikage, N.; Furutani, A.; Aoki, H.; Hamano, K. Inhibitory effect of statins on inflammation-related pathways in human abdominal aortic aneurysm tissue. Int. J. Mol. Sci. 2015, 16, 11213–11228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schouten, O.; van Laanen, J.H.; Boersma, E.; Vidakovic, R.; Feringa, H.H.; Dunkelgrun, M.; Bax, J.J.; Koning, J.; van Urk, H.; Poldermans, D. Statins are associated with a reduced infrarenal abdominal aortic aneurysm growth. Eur. J. Vasc. Endovasc. Surg. 2006, 32, 21–26. [Google Scholar] [CrossRef] [Green Version]
- Karrowni, W.; Dughman, S.; Hajj, G.P.; Miller, F.J., Jr. Statin therapy reduces growth of abdominal aortic aneurysms. J. Investig. Med. 2011, 59, 1239–1243. [Google Scholar] [CrossRef] [PubMed]
- Schlosser, F.J.; Tangelder, M.J.; Verhagen, H.J.; van der Heijden, G.J.; Muhs, B.E.; van der Graaf, Y.; Moll, F.L. Growth predictors and prognosis of small abdominal aortic aneurysms. J. Vasc. Surg. 2008, 47, 1127–1133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sukhija, R.; Aronow, W.S.; Sandhu, R.; Kakar, P.; Babu, S. Mortality and size of abdominal aortic aneurysm at long-term follow-up of patients not treated surgically and treated with and without statins. Am. J. Cardiol. 2006, 97, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Aoki, H.; Ikeda, Y.; Fujii, K.; Akiyama, N.; Furutani, A.; Hoshii, Y.; Tanaka, N.; Ricci, R.; Ishihara, T.; et al. Regression of abdominal aortic aneurysm by inhibition of c-Jun N-terminal kinase. Nat. Med. 2005, 11, 1330–1338. [Google Scholar] [CrossRef]
- Yoshimura, K.; Aoki, H.; Ikeda, Y.; Furutani, A.; Hamano, K.; Matsuzaki, M. Regression of abdominal aortic aneurysm by inhibition of c-Jun N-terminal kinase in mice. Ann. N. Y. Acad. Sci. 2006, 1085, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Morikage, N.; Nishino-Fujimoto, S.; Furutani, A.; Shirasawa, B.; Hamano, K. Current status and perspectives on pharmacologic therapy for abdominal aortic aneurysm. Curr. Drug Targets 2018, 19, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, N.J.; Wu, W.W.; Schermerhorn, M.L. Open and endovascular management of aortic aneurysms. Circ. Res. 2019, 124, 647–661. [Google Scholar] [CrossRef] [PubMed]
- Lim, G.B. Aortic disease: Endovascular vs. open AAA repair. Nat. Rev. Cardiol. 2015, 12, 500. [Google Scholar] [CrossRef] [PubMed]
- Hackmann, A.E.; Rubin, B.G.; Sanchez, L.A.; Geraghty, P.A.; Thompson, R.W.; Curci, J.A. A randomized, placebo-controlled trial of doxycycline after endoluminal aneurysm repair. J. Vasc. Surg. 2008, 48, 519–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, J.; Matsuzaki, T.; Tatematsu, K.; Okajima, T.; Tanizawa, K.; Kuroda, S. Bio-nanocapsule conjugated with liposomes for in vivo pinpoint delivery of various materials. J. Control. Release 2008, 126, 255–264. [Google Scholar] [CrossRef]
- Somiya, M.; Kuroda, S. Development of a virus-mimicking nanocarrier for drug delivery systems: The bio-nanocapsule. Adv. Drug Deliv. Rev. 2015, 95, 77–89. [Google Scholar] [CrossRef]
- Somiya, M.; Sasaki, Y.; Matsuzaki, T.; Liu, Q.; Iijima, M.; Yoshimoto, N.; Niimi, T.; Maturana, A.D.; Kuroda, S. Intracellular trafficking of bio-nanocapsule-liposome complex: Identification of fusogenic activity in the pre-S1 region of hepatitis B virus surface antigen L protein. J. Control. Release 2015, 212, 10–18. [Google Scholar] [CrossRef]
- Yamada, T.; Iwasaki, Y.; Tada, H.; Iwabuki, H.; Chuah, M.K.; VandenDriessche, T.; Fukuda, H.; Kondo, A.; Ueda, M.; Seno, M.; et al. Nanoparticles for the delivery of genes and drugs to human hepatocytes. Nat. Biotechnol. 2003, 21, 885–890. [Google Scholar] [CrossRef]
- Iijima, M.; Matsuzaki, T.; Kadoya, H.; Hatahira, S.; Hiramatsu, S.; Jung, G.; Tanizawa, K.; Kuroda, S. Bionanocapsule-based enzyme-antibody conjugates for enzyme-linked immunosorbent assay. Anal. Biochem. 2010, 396, 257–261. [Google Scholar] [CrossRef]
- Iijima, M.; Kuroda, S. Scaffolds for oriented and close-packed immobilization of immunoglobulins. Biosens. Bioelectron. 2017, 89, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Golledge, J.; Norman, P.E.; Murphy, M.P.; Dalman, R.L. Challenges and opportunities in limiting abdominal aortic aneurysm growth. J. Vasc. Surg. 2017, 65, 225–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nosoudi, N.; Nahar-Gohad, P.; Sinha, A.; Chowdhury, A.; Gerard, P.; Carsten, C.G.; Gray, B.H.; Vyavahare, N.R. Prevention of abdominal aortic aneurysm progression by targeted inhibition of matrix metalloproteinase activity with batimastat-loaded nanoparticles. Circ. Res. 2015, 117, e80–e89. [Google Scholar] [CrossRef] [PubMed]
- Nosoudi, N.; Chowdhury, A.; Siclari, S.; Parasaram, V.; Karamched, S.; Vyavahare, N. Systemic delivery of nanoparticles loaded with pentagalloyl glucose protects elastic lamina and prevents abdominal aortic aneurysm in rats. J. Cardiovasc. Transl. Res. 2016, 9, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Shirasu, T.; Koyama, H.; Miura, Y.; Hoshina, K.; Kataoka, K.; Watanabe, T. Nanoparticles effectively target rapamycin delivery to sites of experimental aortic aneurysm in rats. PLoS ONE 2016, 11, e0157813. [Google Scholar] [CrossRef] [Green Version]
- Camardo, A.; Seshadri, D.; Broekelmann, T.; Mecham, R.; Ramamurthi, A. Multifunctional, JNK-inhibiting nanotherapeutics for augmented elastic matrix regenerative repair in aortic aneurysms. Drug Deliv. Transl. Res. 2018, 8, 964–984. [Google Scholar] [CrossRef]
- Cheng, J.; Zhang, R.; Li, C.; Tao, H.; Dou, Y.; Wang, Y.; Hu, H.; Zhang, J. A targeting nanotherapy for abdominal aortic aneurysms. J. Am. Coll. Cardiol. 2018, 72, 2591–2605. [Google Scholar] [CrossRef]
- Yamawaki-Ogata, A.; Hashizume, R.; Satake, M.; Kaneko, H.; Mizutani, S.; Moritan, T.; Ueda, Y.; Narita, Y. A doxycycline loaded, controlled-release, biodegradable fiber for the treatment of aortic aneurysms. Biomaterials 2010, 31, 9554–9564. [Google Scholar] [CrossRef]
- Tanaka, A.; Hasegawa, T.; Morimoto, K.; Bao, W.; Yu, J.; Okita, Y.; Tabata, Y.; Okada, K. Controlled release of ascorbic acid from gelatin hydrogel attenuates abdominal aortic aneurysm formation in rat experimental abdominal aortic aneurysm model. J. Vasc. Surg. 2014, 60, 749–758. [Google Scholar] [CrossRef] [Green Version]
- Yu, M.; Dong, A.; Chen, C.; Xu, S.; Cao, Y.; Liu, S.; Zhang, Q.; Qi, R. Thermosensitive hydrogel containing doxycycline exerts inhibitory effects on abdominal aortic aneurysm induced by pancreatic elastase in mice. Adv. Healthc. Mater. 2017, 6, 1700671. [Google Scholar] [CrossRef]
- Yoshimura, K.; Aoki, H.; Ikeda, Y.; Furutani, A.; Hamano, K.; Matsuzaki, M. Identification of c-Jun N-terminal kinase as a therapeutic target for abdominal aortic aneurysm. Ann. N. Y. Acad. Sci. 2006, 1085, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Iijima, M.; Kadoya, H.; Hatahira, S.; Hiramatsu, S.; Jung, G.; Martin, A.; Quinn, J.; Jung, J.; Jeong, S.Y.; Choi, E.K.; et al. Nanocapsules incorporating IgG Fc-binding domain derived from Staphylococcus aureus protein A for displaying IgGs on immunosensor chips. Biomaterials 2011, 32, 1455–1464. [Google Scholar] [CrossRef] [PubMed]
- Iijima, M.; Yoshimoto, N.; Niimi, T.; Maturana, A.D.; Kuroda, S. Nanocapsule-based probe for evaluating the orientation of antibodies immobilized on a solid phase. Analyst 2013, 138, 3470–3477. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Iijima, M.; Yoshimoto, N.; Sasaki, M.; Niimi, T.; Tatematsu, K.; Jeong, S.Y.; Choi, E.K.; Tanizawa, K.; Kuroda, S. Efficient and rapid purification of drug- and gene-carrying bio-nanocapsules, hepatitis B virus surface antigen L particles, from Saccharomyces cerevisiae. Protein Expr. Purif. 2011, 78, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Iijima, M.; Matsuzaki, T.; Yoshimoto, N.; Niimi, T.; Tanizawa, K.; Kuroda, S. Fluorophore-labeled nanocapsules displaying IgG Fc-binding domains for the simultaneous detection of multiple antigens. Biomaterials 2011, 32, 9011–9020. [Google Scholar] [CrossRef]
- Harada, T.; Yoshimura, K.; Yamashita, O.; Ueda, K.; Morikage, N.; Sawada, Y.; Hamano, K. Focal adhesion kinase promotes the progression of aortic aneurysm by modulating macrophage behavior. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 156–165. [Google Scholar] [CrossRef] [Green Version]
- Ueda, K.; Yoshimura, K.; Yamashita, O.; Harada, T.; Morikage, N.; Hamano, K. Possible dual role of decorin in abdominal aortic aneurysm. PLoS ONE 2015, 10, e0120689. [Google Scholar] [CrossRef]
- Onoda, M.; Yoshimura, K.; Aoki, H.; Ikeda, Y.; Morikage, N.; Furutani, A.; Matsuzaki, M.; Hamano, K. Lysyl oxidase resolves inflammation by reducing monocyte chemoattractant protein-1 in abdominal aortic aneurysm. Atherosclerosis 2010, 208, 366–369. [Google Scholar] [CrossRef]
- Yamashita, O.; Yoshimura, K.; Nagasawa, A.; Ueda, K.; Morikage, N.; Ikeda, Y.; Hamano, K. Periostin links mechanical strain to inflammation in abdominal aortic aneurysm. PLoS ONE 2013, 8, e79753. [Google Scholar] [CrossRef]
- Nagasawa, A.; Yoshimura, K.; Suzuki, R.; Mikamo, A.; Yamashita, O.; Ikeda, Y.; Tsuchida, M.; Hamano, K. Important role of the angiotensin II pathway in producing matrix metalloproteinase-9 in human thoracic aortic aneurysms. J. Surg. Res. 2013, 183, 472–477. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshimura, K.; Aoki, H.; Teruyama, C.; Iijima, M.; Tsutsumi, H.; Kuroda, S.; Hamano, K. A Novel Hybrid Drug Delivery System for Treatment of Aortic Aneurysms. Int. J. Mol. Sci. 2020, 21, 5538. https://doi.org/10.3390/ijms21155538
Yoshimura K, Aoki H, Teruyama C, Iijima M, Tsutsumi H, Kuroda S, Hamano K. A Novel Hybrid Drug Delivery System for Treatment of Aortic Aneurysms. International Journal of Molecular Sciences. 2020; 21(15):5538. https://doi.org/10.3390/ijms21155538
Chicago/Turabian StyleYoshimura, Koichi, Hiroki Aoki, Chie Teruyama, Masumi Iijima, Hiromori Tsutsumi, Shun’ichi Kuroda, and Kimikazu Hamano. 2020. "A Novel Hybrid Drug Delivery System for Treatment of Aortic Aneurysms" International Journal of Molecular Sciences 21, no. 15: 5538. https://doi.org/10.3390/ijms21155538
APA StyleYoshimura, K., Aoki, H., Teruyama, C., Iijima, M., Tsutsumi, H., Kuroda, S., & Hamano, K. (2020). A Novel Hybrid Drug Delivery System for Treatment of Aortic Aneurysms. International Journal of Molecular Sciences, 21(15), 5538. https://doi.org/10.3390/ijms21155538





