Characterization of p53 Family Homologs in Evolutionary Remote Branches of Holozoa
Abstract
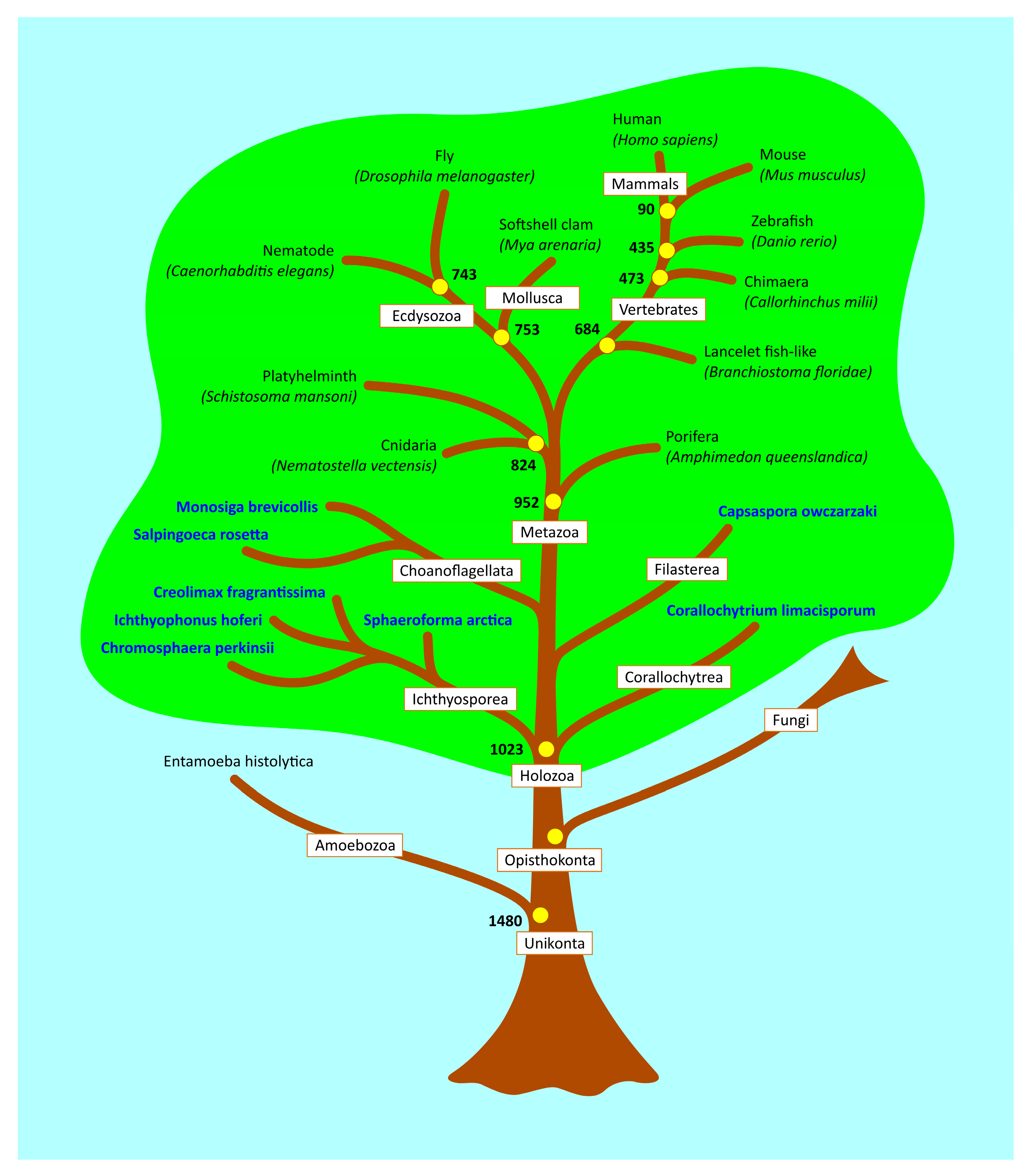
1. Introduction
2. Results
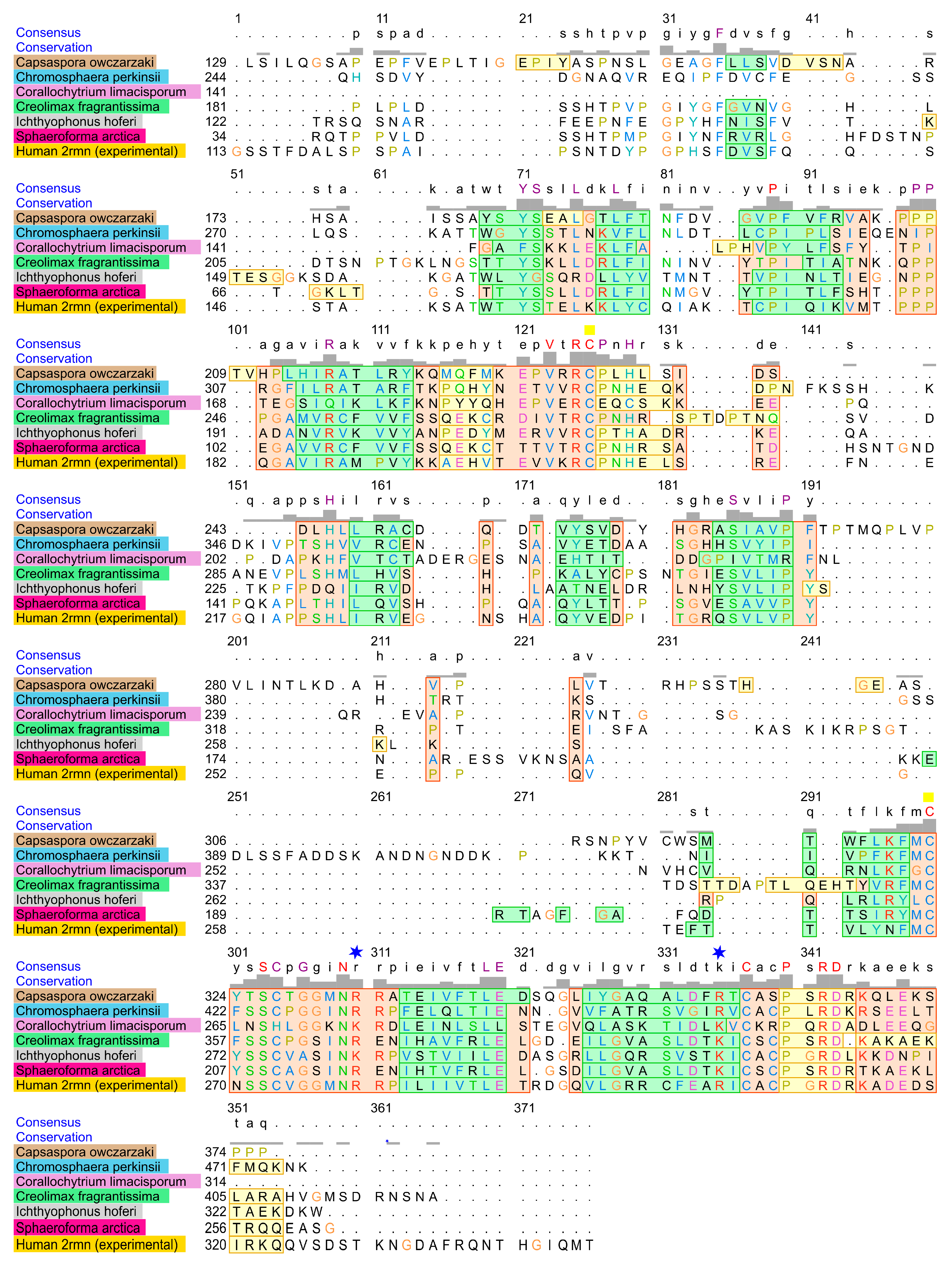
2.1. Homology of the Sequences
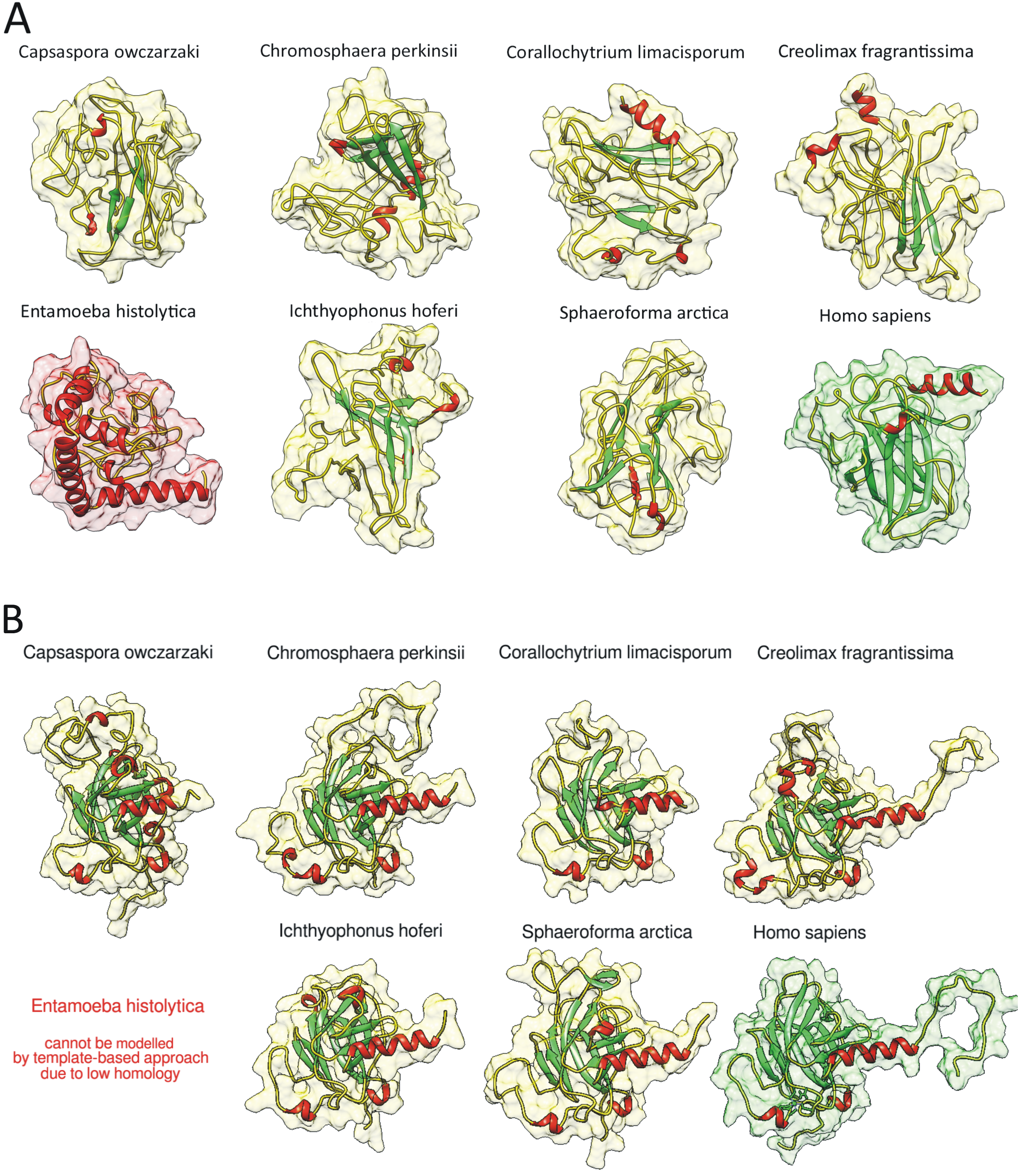
2.2. Homology of Predicted Structures
3. Discussion
4. Materials and Methods
4.1. P53 Family Homolog Search
4.2. Validation of p53 Homologs (Reciprocal Search)
4.3. Isoelectric Point Predictions
4.4. Domain Searches and Visualization
4.5. Nuclear Localization Signal Predictions
4.6. Estimation of Evolutionary Age
4.7. Validation of Entamoeba Histolytica Putative p53 Homolog
4.8. Alignment of DBD from the Most Distant Human p53 Homologs
4.9. p53 Protein Tree
4.10. Modelling of 3D Protein Structures
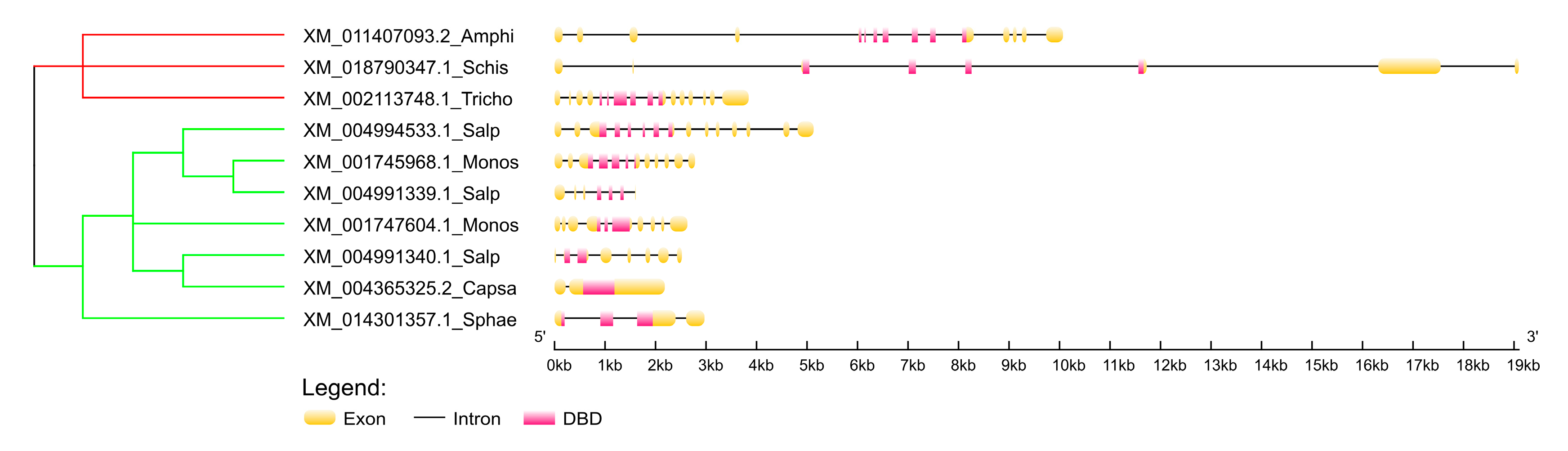
4.11. Comparison of Exon-Intron Composition of the Selected p53 Homologs
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hollstein, M.; Sidransky, D.; Vogelstein, B.; Harris, C. P53 Mutations in Human Cancers. Science 1991, 253, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Goh, A.M.; Coffill, C.R.; Lane, D.P. The role of mutant p53 in human cancer. J. Pathol. 2011, 223, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Stiewe, T.; Haran, T.E. How mutations shape p53 interactions with the genome to promote tumorigenesis and drug resistance. Drug Resist. Update 2018, 38, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Joerger, A.C.; Fersht, A.R. The p53 pathway: Origins, inactivation in cancer, and emerging therapeutic approaches. Annu. Rev. Biochem. 2016, 85, 375–404. [Google Scholar] [CrossRef] [PubMed]
- Strano, S.; Rossi, M.; Fontemaggi, G.; Munarriz, E.; Soddu, S.; Sacchi, A.; Blandino, G. From p63 to p53 across p73. FEBS Lett. 2001, 490, 163–170. [Google Scholar] [CrossRef]
- Belyi, V.A.; Levine, A.J. One billion years of p53/p63/p73 evolution. Proc. Natl. Acad. Sci. USA 2009, 106, 17609–17610. [Google Scholar] [CrossRef]
- Beckerman, R.; Prives, C. Transcriptional Regulation by P53. Cold Spring Harb. Perspect. Biol. 2010, 2, 1–18. [Google Scholar] [CrossRef]
- Brazda, V.; Coufal, J. Recognition of Local DNA Structures by p53 Protein. Int. J. Mol. Sci. 2017, 18, 375. [Google Scholar] [CrossRef]
- Vyas, P.; Beno, I.; Xi, Z.; Stein, Y.; Golovenko, D.; Kessler, N.; Rotter, V.; Shakked, Z.; Haran, T.E. Diverse p53/DNA binding modes expand the repertoire of p53 response elements. Proc. Natl. Acad. Sci. USA 2017, 114, 10624–10629. [Google Scholar] [CrossRef]
- Brázda, V.; Fojta, M. The Rich World of p53 DNA Binding Targets: The Role of DNA Structure. Int. J. Mol. Sci. 2019, 20, 5605. [Google Scholar] [CrossRef]
- El-Deiry, W.S.; Kern, S.E.; Pietenpol, J.A.; Kinzler, K.W.; Vogelstein, B. Definition of a consensus binding site for p53. Nat. Genet. 1992, 1, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Tebaldi, T.; Zaccara, S.; Alessandrini, F.; Bisio, A.; Ciribilli, Y.; Inga, A. Whole-genome cartography of p53 response elements ranked on transactivation potential. BMC Genom. 2015, 16, 464. [Google Scholar] [CrossRef] [PubMed]
- Brázda, V.; Čechová, J.; Battistin, M.; Coufal, J.; Jagelská, E.B.; Raimondi, I.; Inga, A. The structure formed by inverted repeats in p53 response elements determines the transactivation activity of p53 protein. Biochem. Biophys. Res. Commun. 2017, 483, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Coufal, J.; Jagelská, E.B.; Liao, J.C.; Brázda, V. Preferential binding of p53 tumor suppressor to p21 promoter sites that contain inverted repeats capable of forming cruciform structure. Biochem. Biophys. Res. Commun. 2013, 441, 83–88. [Google Scholar] [CrossRef]
- Dötsch, V.; Bernassola, F.; Coutandin, D.; Candi, E.; Melino, G. p63 and p73, the Ancestors of p53. Cold Spring Harb. Perspect. Biol. 2010, 2. [Google Scholar] [CrossRef]
- Arrowsmith, C.H. Structure and function in the p53 family. Cell Death Differ. 1999, 6, 1169–1173. [Google Scholar] [CrossRef]
- Joerger, A.C.; Rajagopalan, S.; Natan, E.; Veprintsev, D.B.; Robinson, C.V.; Fersht, A.R. Structural evolution of p53, p63, and p73: Implication for heterotetramer formation. Proc. Natl. Acad. Sci. USA 2009, 106, 17705–17710. [Google Scholar] [CrossRef]
- Pavletich, N.P.; Chambers, K.A.; Pabo, C.O. The DNA-binding domain of p53 contains the four conserved regions and the major mutation hot spots. Genes Dev. 1993, 7, 2556–2564. [Google Scholar] [CrossRef]
- Xue, B.; Brown, C.J.; Dunker, A.K.; Uversky, V.N. Intrinsically disordered regions of p53 family are highly diversified in evolution. BBA Proteins Proteom. 2013, 1834, 725–738. [Google Scholar] [CrossRef]
- Joerger, A.C.; Wilcken, R.; Andreeva, A. Tracing the Evolution of the p53 Tetramerization Domain. Structure 2014, 22, 1301–1310. [Google Scholar] [CrossRef]
- Yang, A.; Kaghad, M.; Caput, D.; McKeon, F. On the shoulders of giants: p63, p73 and the rise of p53. Trends Genet. 2002, 18, 90–95. [Google Scholar] [CrossRef]
- Dos Santos, H.G.; Nunez-Castilla, J.; Siltberg-Liberles, J. Functional diversification after gene duplication: Paralog specific regions of structural disorder and phosphorylation in p53, p63, and p73. PLoS ONE 2016, 11, e0151961. [Google Scholar] [CrossRef] [PubMed]
- Laptenko, O.; Shiff, I.; Freed-Pastor, W.; Zupnick, A.; Mattia, M.; Freulich, E.; Shamir, I.; Kadouri, N.; Kahan, T.; Manfredi, J. The p53 C terminus controls site-specific DNA binding and promotes structural changes within the central DNA binding domain. Mol. Cell 2015, 57, 1034–1046. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Roeder, R.G. Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell 1997, 90, 595–606. [Google Scholar] [CrossRef]
- Pospísilová, S.; Brázda, V.; Kucharíková, K.; Luciani, M.G.; Hupp, T.R.; Skládal, P.; Palecek, E.; Vojtesek, B. Activation of the DNA-binding ability of latent p53 protein by protein kinase C is abolished by protein kinase CK2. Biochem. J. 2004, 378, 939–947. [Google Scholar] [CrossRef]
- Ou, Y.-H.; Chung, P.-H.; Sun, T.-P.; Shieh, S.-Y. p53 C-terminal phosphorylation by CHK1 and CHK2 participates in the regulation of DNA-damage-induced C-terminal acetylation. Mol. Biol. Cell 2005, 16, 1684–1695. [Google Scholar] [CrossRef]
- Muller, P.; Chan, J.M.; Simoncik, O.; Fojta, M.; Lane, D.P.; Hupp, T.; Vojtesek, B. Evidence for allosteric effects on p53 oligomerization induced by phosphorylation. Protein Sci. 2018, 27, 523–530. [Google Scholar] [CrossRef]
- Brázda, V.; Paleĉek, J.; Pospísilová, S.; Vojtêsek, B.; Paleĉek, E. Specific modulation of p53 binding to consensus sequence within supercoiled DNA by monoclonal antibodies. Biochem. Biophys. Res. Commun. 2000, 267, 934–939. [Google Scholar] [CrossRef]
- Weinberg, R.L.; Freund, S.M.V.; Veprintsev, D.B.; Bycroft, M.; Fersht, A.R. Regulation of DNA binding of p53 by its C-terminal domain. J. Mol. Biol. 2004, 342, 801–811. [Google Scholar] [CrossRef]
- Lu, W.-J.; Amatruda, J.F.; Abrams, J.M. p53 ancestry: Gazing through an evolutionary lens. Nat. Rev. Cancer 2009, 9, 758–762. [Google Scholar] [CrossRef]
- Belyi, V.A.; Ak, P.; Markert, E.; Wang, H.; Hu, W.; Puzio-Kuter, A.; Levine, A.J. The origins and evolution of the p53 family of genes. Cold Spring Harb. Perspect. Biol. 2010, 2, a001198. [Google Scholar] [CrossRef] [PubMed]
- Nedelcu, A.M.; Tan, C. Early diversification and complex evolutionary history of the p53 tumor suppressor gene family. Dev. Genes Evol. 2007, 217, 801–806. [Google Scholar] [CrossRef] [PubMed]
- King, N.; Westbrook, M.J.; Young, S.L.; Kuo, A.; Abedin, M.; Chapman, J.; Fairclough, S.; Hellsten, U.; Isogai, Y.; Letunic, I.; et al. The genome of the choanoflagellate Monosiga brevicollis and the origin of metazoans. Nature 2008, 451, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Sebé-Pedrós, A.; de Mendoza, A.; Lang, B.F.; Degnan, B.M.; Ruiz-Trillo, I. Unexpected repertoire of metazoan transcription factors in the unicellular holozoan Capsaspora owczarzaki. Mol. Biol. Evol. 2011, 28, 1241–1254. [Google Scholar] [CrossRef] [PubMed]
- de Mendoza, A.; Sebé-Pedrós, A.; Šestak, M.S.; Matejčić, M.; Torruella, G.; Domazet-Lošo, T.; Ruiz-Trillo, I. Transcription factor evolution in eukaryotes and the assembly of the regulatory toolkit in multicellular lineages. Proc. Natl. Acad. Sci. USA 2013, 110, E4858–E4866. [Google Scholar] [CrossRef] [PubMed]
- Sebé-Pedrós, A.; Peña, M.I.; Capella-Gutiérrez, S.; Antó, M.; Gabaldón, T.; Ruiz-Trillo, I.; Sabidó, E. High-throughput proteomics reveals the unicellular roots of animal phosphosignaling and cell differentiation. Dev. Cell 2016, 39, 186–197. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Grau-Bove, X.; Torruella, G.; Donachie, S.; Suga, H.; Leonard, G.; Richards, T.A.; Ruiz-Trillo, I. Dynamics of genomic innovation in the unicellular ancestry of animals. eLife 2017, 6, e26036. [Google Scholar] [CrossRef]
- de Mendoza, A.; Suga, H.; Permanyer, J.; Irimia, M.; Ruiz-Trillo, I. Complex transcriptional regulation and independent evolution of fungal-like traits in a relative of animals. eLife 2015, 4, e08904. [Google Scholar] [CrossRef]
- Potter, S.C.; Luciani, A.; Eddy, S.R.; Park, Y.; Lopez, R.; Finn, R.D. HMMER web server: 2018 update. Nucleic Acids Res. 2018, 46, W200–W204. [Google Scholar] [CrossRef]
- Chang, F.-L.; Lai, M.-D. Various forms of mutant p53 confer sensitivity to cisplatin and doxorubicin in bladder cancer cells. J. Urol. 2001, 166, 304–310. [Google Scholar] [CrossRef]
- Olivier, M.; Hollstein, M.; Hainaut, P. TP53 Mutations in Human Cancers: Origins, Consequences, and Clinical Use. Cold Spring Harb. Perspect. Biol. 2010, 2, a001008. [Google Scholar] [CrossRef] [PubMed]
- Eldar, A.; Rozenberg, H.; Diskin-Posner, Y.; Rohs, R.; Shakked, Z. Structural studies of p53 inactivation by DNA-contact mutations and its rescue by suppressor mutations via alternative protein–DNA interactions. Nucleic Acids Res. 2013, 41, 8748–8759. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.S.; Trovão, F.; Andrade Pinheiro, B.; Freire, F.; Gomes, S.; Oliveira, C.; Domingues, L.; Romão, M.J.; Saraiva, L.; Carvalho, A.L. The Crystal Structure of the R280K Mutant of Human p53 Explains the Loss of DNA Binding. Int. J. Mol. Sci. 2018, 19, 1184. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, D.; Pati, S.; Zambetti, G.; Chu, S.; Teresky, A.K.; Moore, M.; Finlay, C.; Levine, A.J. Gain of function mutations in p53. Nat. Genet. 1993, 4, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Zhang, Y. Ab initio protein structure assembly using continuous structure fragments and optimized knowledge-based force field. Proteins Struct. Funct. Bioinform. 2012, 80, 1715–1735. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Zhang, Y. Toward optimal fragment generations for ab initio protein structure assembly. Proteins Struct. Funct. Bioinform. 2013, 81, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Lion, M.; Raimondi, I.; Donati, S.; Jousson, O.; Ciribilli, Y.; Inga, A. Evolution of p53 Transactivation Specificity through the Lens of a Yeast-Based Functional Assay. PLoS ONE 2015, 10, e0116177. [Google Scholar] [CrossRef]
- Jordan, A.; Reichard, P. Ribonucleotide Reductases. Annu. Rev. Biochem. 1998, 67, 71–98. [Google Scholar] [CrossRef] [PubMed]
- Åberg, E.; Saccoccia, F.; Grabherr, M.; Ore, W.Y.J.; Jemth, P.; Hultqvist, G. Evolution of the p53-MDM2 pathway. BMC Evol. Biol. 2017, 17, 177. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, L.; Orozco, E.; Rodriguez, M.A.; Garcia-Rivera, G.; Sanchez, T.; Garcia, E.; Gariglio, P. Ehp53, an Entamoeba histolytica protein, ancestor of the mammalian tumour suppressor p53. Microbiology 2003, 149, 885–893. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Biscotti, M.A.; Barucca, M.; Carducci, F.; Forconi, M.; Canapa, A. The p53 gene family in vertebrates: Evolutionary considerations. J. Exp. Zool. Part B 2019, 332, 171–178. [Google Scholar] [CrossRef]
- Aravind, L.; Anantharaman, V.; Iyer, L.M. Evolutionary connections between bacterial and eukaryotic signaling systems: A genomic perspective. Curr. Opin. Microbiol. 2003, 6, 490–497. [Google Scholar] [CrossRef]
- Okonechnikov, K.; Golosova, O.; Fursov, M.; Team, U. Unipro UGENE: A unified bioinformatics toolkit. Bioinformatics 2012, 28, 1166–1167. [Google Scholar] [CrossRef]
- Kozlowski, L.P. IPC—Isoelectric Point Calculator. Biol. Direct 2016, 11, 55. [Google Scholar] [CrossRef]
- Marchler-Bauer, A.; Derbyshire, M.K.; Gonzales, N.R.; Lu, S.; Chitsaz, F.; Geer, L.Y.; Geer, R.C.; He, J.; Gwadz, M.; Hurwitz, D.I.; et al. CDD: NCBI’s conserved domain database. Nucleic Acids Res. 2015, 43, D222–D226. [Google Scholar] [CrossRef]
- Kosugi, S.; Hasebe, M.; Tomita, M.; Yanagawa, H. Systematic identification of cell cycle-dependent yeast nucleocytoplasmic shuttling proteins by prediction of composite motifs. Proc. Natl. Acad. Sci. USA 2009, 106, 10171–10176. [Google Scholar] [CrossRef]
- Hedges, S.B.; Marin, J.; Suleski, M.; Paymer, M.; Kumar, S. Tree of life reveals clock-like speciation and diversification. Mol. Biol. Evol. 2015, 32, 835–845. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Suleski, M.; Hedges, S.B. TimeTree: A resource for timelines, timetrees, and divergence times. Mol. Biol. Evol. 2017, 34, 1812–1819. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. 20 years of the SMART protein domain annotation resource. Nucleic Acids Res. 2018, 46, D493–D496. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- de Anta, J.M.; Jassem, E.; Rosell, R.; Martínez-Roca, M.; Jassem, J.; Martínez-López, E.; Monzó, M.; Sánchez-Hernández, J.J.; Moreno, I.; Sánchez-Céspedes, M. TP53 mutational pattern in Spanish and Polish non-small cell lung cancer patients: Null mutations are associated with poor prognosis. Oncogene 1997, 15, 2951. [Google Scholar] [CrossRef] [PubMed]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef] [PubMed]
- Enthart, A.; Klein, C.; Dehner, A.; Coles, M.; Gemmecker, G.; Kessler, H.; Hagn, F. Solution structure and binding specificity of the p63 DNA binding domain. Sci. Rep. 2016, 6, 26707. [Google Scholar] [CrossRef] [PubMed]
- Meng, E.C.; Pettersen, E.F.; Couch, G.S.; Huang, C.C.; Ferrin, T.E. Tools for integrated sequence-structure analysis with UCSF Chimera. BMC Bioinform. 2006, 7, 339. [Google Scholar]
- Hu, B.; Jin, J.; Guo, A.-Y.; Zhang, H.; Luo, J.; Gao, G. GSDS 2.0: An upgraded gene feature visualization server. Bioinformatics 2015, 31, 1296–1297. [Google Scholar] [CrossRef]
- Dereeper, A.; Guignon, V.; Blanc, G.; Audic, S.; Buffet, S.; Chevenet, F.; Dufayard, J.-F.; Guindon, S.; Lefort, V.; Lescot, M. Phylogeny. fr: Robust phylogenetic analysis for the non-specialist. Nucleic Acids Res. 2008, 36, W465–W469. [Google Scholar] [CrossRef]
- Notredame, C.; Higgins, D.G.; Heringa, J. T-Coffee: A novel method for fast and accurate multiple sequence alignment. J. Mol. Biol. 2000, 302, 205–217. [Google Scholar] [CrossRef]
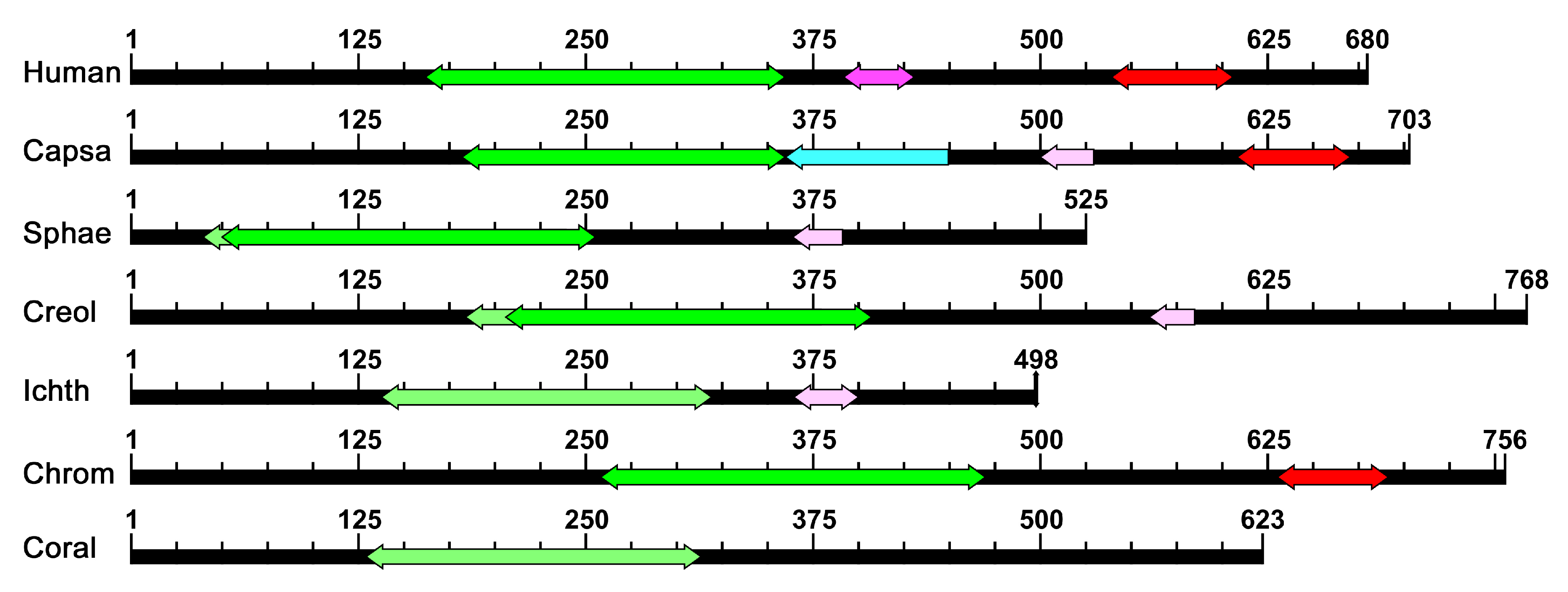
| Organism Name | ID of p53 Homolog | PFAM Domains | NLS | |||
|---|---|---|---|---|---|---|
| Protein Length (aa) | TAD | DBD | TET | SAM | ||
| Predicted pI | ||||||
| Monosiga brevicollis (class Choanoflagellate) | XP_001746020.1 | − | + | − | − | + |
| 523 | ||||||
| 6.68 | ||||||
| XP_001747656.1 | − | + | − | − | + | |
| 571 | ||||||
| 5.89 | ||||||
| Salpingoeca rosetta (class Choanoflagellate) | XP_004991396.1 | − | + | − | − | − |
| 170 | ||||||
| 5.55 | ||||||
| XP_004991397.1 | − | + | − | + | + | |
| 351 | ||||||
| 5.74 | ||||||
| XP_004994590.1 | − | + | + | + | − | |
| 613 | ||||||
| 6.72 | ||||||
| Capsaspora owczarzaki (class Filasterea) | XP_004365382.2 | − | + | + | + | |
| 703 | ||||||
| 7.05 | ||||||
| Sphaeroforma arctica (class Ichthyosporea) | XP_014156832.1 | − | + | + | − | − |
| 525 | ||||||
| 9.57 | ||||||
| Creolimax fragrantissima (class Ichthyosporea) | CFRG4869T1 | − | + | + | − | − |
| 768 | ||||||
| 8.72 | ||||||
| Ichthyophonus hoferi (class Ichthyosporea) | Ihof_evm3s137 | − | + | + | − | + |
| 498 | ||||||
| 7.75 | ||||||
| Chromosphaera perkinsii (class Ichthyosporea) | Nk52_evm78s1737 | − | + | − | + | + |
| 756 | ||||||
| 6.29 | ||||||
| Corallochytrium limacisporum (Corallochytrea) | Clim_evm153s157 | − | + | − | − | + |
| 623 | ||||||
| 5.50 | ||||||
| Human p53 Position | Amino Acid | Conservation in All Homologs | Function |
|---|---|---|---|
| 142 | P | 100% | Unknown |
| 173 | V | 100% | V173L hotspot [41] |
| 175 | R | 100% | R175H hotspot [42] |
| 176 | C | 100% | Zn2+ binding [43] |
| 238 | C | 100% | Zn2+ binding [43] |
| 241 | S | 100% | S241F hotspot [42] |
| 247 | N | 100% | N247I hotspot [41] |
| 248 | R | +charged aa | R248W hotspot [42] |
| 273 | R | +charged aa | R273H hotspot [42] |
| 275 | C | 100% | Unknown |
| 278 | P | 100% | Unknown |
| 280 | R | 100% | R280K hotspot [44] |
| 281 | D | 100% | D281G hotspot [45] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartas, M.; Brázda, V.; Červeň, J.; Pečinka, P. Characterization of p53 Family Homologs in Evolutionary Remote Branches of Holozoa. Int. J. Mol. Sci. 2020, 21, 6. https://doi.org/10.3390/ijms21010006
Bartas M, Brázda V, Červeň J, Pečinka P. Characterization of p53 Family Homologs in Evolutionary Remote Branches of Holozoa. International Journal of Molecular Sciences. 2020; 21(1):6. https://doi.org/10.3390/ijms21010006
Chicago/Turabian StyleBartas, Martin, Václav Brázda, Jiří Červeň, and Petr Pečinka. 2020. "Characterization of p53 Family Homologs in Evolutionary Remote Branches of Holozoa" International Journal of Molecular Sciences 21, no. 1: 6. https://doi.org/10.3390/ijms21010006
APA StyleBartas, M., Brázda, V., Červeň, J., & Pečinka, P. (2020). Characterization of p53 Family Homologs in Evolutionary Remote Branches of Holozoa. International Journal of Molecular Sciences, 21(1), 6. https://doi.org/10.3390/ijms21010006








