Insights on the Proteases Involved in Barley and Wheat Grain Germination
Abstract
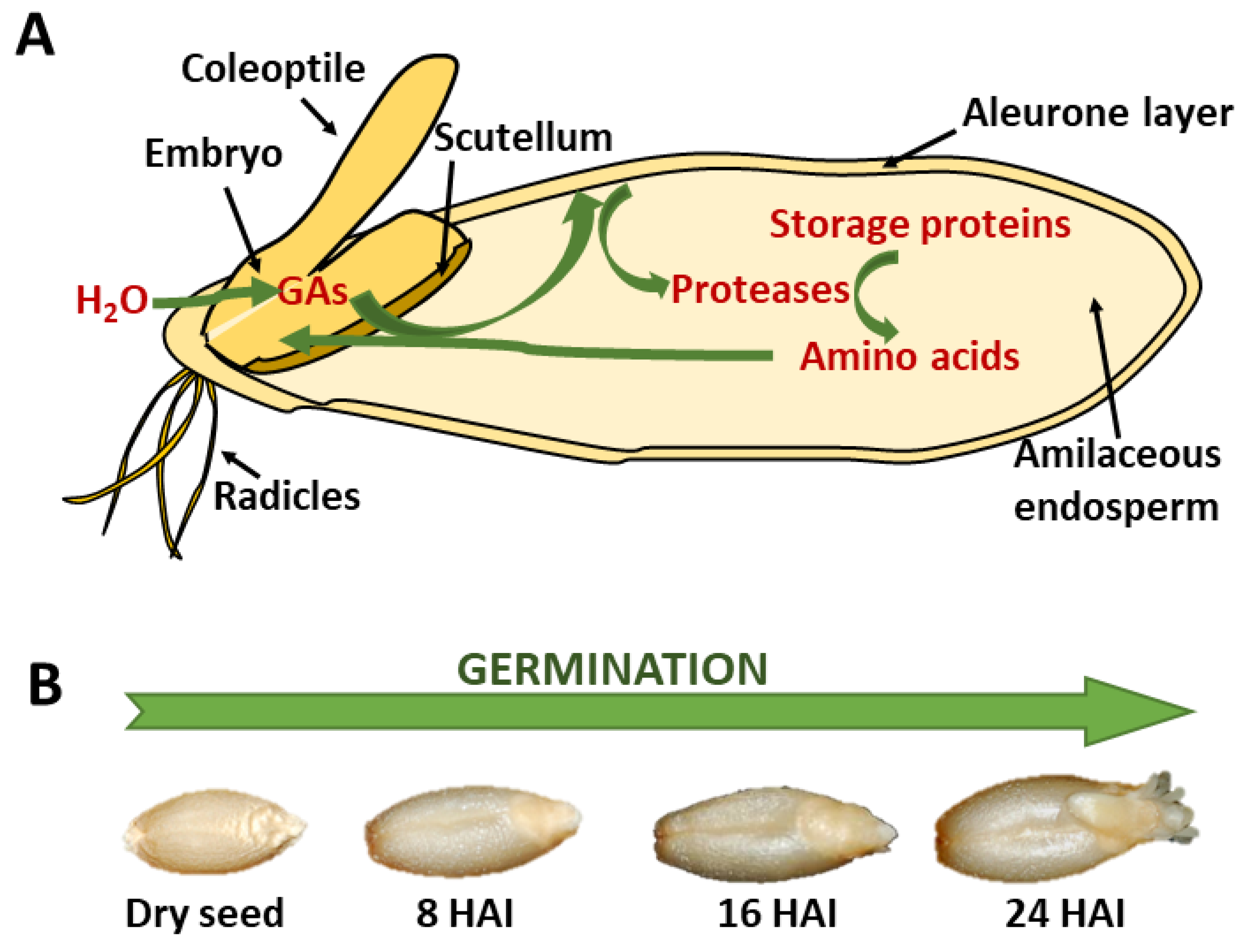
1. Introduction
2. Mobilization of Stored Proteins During the Germination of Barley and Wheat
3. Investigation of Proteases in the Germination of Barley and Wheat before High-Throughput Technologies
4. Functional Genomic-Based Advances in the Identification of Proteases in the Germination of Barley and Wheat Grains
5. Proteomic-Based Advances on Proteases in the Germination of Barley and Wheat Grains
6. Conclusions and Future Perspectives
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Grudkowska, M.; Zagdańska, B. Multifunctional role of plant cysteine proteinases. Acta Biochim. Pol. 2004, 51, 609–624. [Google Scholar] [PubMed]
- Szewińska, J.; Simińska, J.; Bielawski, W. The roles of cysteine proteases and phytocystatins in development and germination of cereal seeds. J. Plant Physiol. 2016, 207, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Han, C.; Deng, X.; Liu, D.; Liu, N.; Yan, Y. Integrated physiology and proteome analysis of embryo and endosperm highlights complex metabolic networks involved in seed germination in wheat (Triticum aestivum L.). J. Plant Physiol. 2018, 229, 63–76. [Google Scholar] [CrossRef]
- Tan-Wilson, A.L.; Wilson, K.A. Mobilization of seed protein reserves. Physiol. Plant 2012, 145, 140–153. [Google Scholar] [CrossRef]
- Fischer, J.; Becker, C.; Hillmer, S.; Horstmann, C.; Neubohn, B.; Schlereth, A.; Senyuk, V.; Shutov, A.; Müntz, K. The families of papain- and legumain-like cysteine proteinases from embryonic axes and cotyledons of Vicia seeds: Developmental patterns, intracellular localization and functions in globulin proteolysis. Plant Mol. Biol. 2000, 43, 83–101. [Google Scholar] [CrossRef]
- Schlereth, A.; Becker, C.; Horstmann, C.; Tiedemann, J.; Müntz, K. Comparison of globulin mobilization and cysteine proteinases in embryonic axes and cotyledons during germination and seedling growth of vetch (Vicia sativa L.). J. Exp. Bot. 2000, 51, 1423–1433. [Google Scholar] [PubMed]
- Toyooka, K.; Okamoto, T.; Minamikawa, T. Mass transport of proform of a KDEL-tailed cysteine proteinase (SH-EP) to protein storage vacuoles by endoplasmic reticulum-derived vesicle is involved in protein mobilization in germinating seeds. J. Cell Biol. 2000, 148, 453–464. [Google Scholar] [CrossRef]
- Zhang, N.; Jones, B.L. Characterization of germinated barley endoproteolytic enzymes by two dimensional gel electrophoresis. J. Cereal Sci. 1995, 21, 145–153. [Google Scholar] [CrossRef]
- Watanabe, H.; Abe, K.; Emori, Y.; Hosoyama, H.; Arai, S. Molecular cloning and gibberellin-induced expression of multiple cysteine proteinases of rice seeds (oryzains). J. Biol. Chem. 1991, 266, 16897–16902. [Google Scholar] [PubMed]
- Prabucka, B.; Bielawski, W. Purification and partial characteristic of a major gliadin-degrading cysteine endopeptidase from germinating triticale seeds. Acta Physiol. Plant. 2004, 26, 383. [Google Scholar] [CrossRef]
- Prabucka, B.; Drzymała, A.; Grabowska, A. Molecular cloning and expression analysis of the main gliadin-degrading cysteine endopeptidase EP8 from triticale. J. Cereal Sci. 2013, 58, 284–289. [Google Scholar] [CrossRef]
- González-Calle, V.; Iglesias-Fernández, R.; Carbonero, P.; Barrero-Sicilia, C. The BdGAMYB protein from Brachypodium distachyon interacts with BdDOF24 and regulates transcription of the BdCathB gene upon seed germination. Planta 2014, 240, 539–552. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, S.; Liu, S.; Jiang, L.; Chen, L.; Ren, Y.; Han, X.; Liu, F.; Ji, S.; Liu, X.; et al. The vacuolar processing enzyme OsVPE1 is required for efficient glutelin processing in rice. Plant J. 2009, 58, 606–617. [Google Scholar] [CrossRef]
- Kato, H.; Sutoh, K.; Minamikawa, T. Identification, cDNA cloning and possible roles of seed-specific rice asparaginyl endopeptidase, REP-2. Planta 2003, 217, 676–685. [Google Scholar] [CrossRef] [PubMed]
- Washio, K.; Ishikawa, K. Organ-specific and hormone-dependent expression of genes for serine carboxypeptidases during development and following germination of rice grains. Plant Physiol. 1994, 105, 1275–1280. [Google Scholar] [CrossRef]
- Li, Z.; Tang, L.; Qiu, J.; Zhang, W.; Wang, Y.; Tong, X.; Wei, X.; Hou, Y.; Zhang, J. Serine carboxypeptidase 46 Regulates Grain Filling and Seed Germination in Rice (Oryza sativa L.). PLoS ONE 2016, 11, e0159737. [Google Scholar] [CrossRef]
- Drzymała, A.; Bielawski, W. Isolation and characterization of carboxypeptidase III from germinating triticale grains. Acta Biochim. Biophys. Sin. 2009, 41, 69–78. [Google Scholar] [CrossRef][Green Version]
- Drzymała, A.; Prabucka, B.; Bielawski, W. Carboxypeptidase I from triticale grains and the hydrolysis of salt-soluble fractions of storage proteins. Plant Physiol. Biochem. 2012, 58, 195–204. [Google Scholar] [CrossRef]
- Shewry, P.R.; Halford, N.G. Cereal seed storage proteins: Structures, properties and role in grain utilization. J. Exp. Bot. 2002, 53, 947–958. [Google Scholar] [CrossRef]
- Shewry, P.R.; Napier, J.A.; Tatham, A.S. Seed storage proteins: Structures and biosynthesis. Plant Cell 1995, 7, 945–956. [Google Scholar] [CrossRef] [PubMed]
- Finch-Savage, W.E.; Leubner-Metzger, G. Seed dormancy and the control of germination. New Phytol. 2006, 171, 501–523. [Google Scholar] [CrossRef] [PubMed]
- Bewley, J.D. Seed Germination and Dormancy. Plant Cell 1997, 9, 1055–1066. [Google Scholar] [CrossRef]
- Dominguez, F.; Cejudo, F.J. Characterization of the Endoproteases Appearing during Wheat Grain Development. Plant Physiol. 1996, 112, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Poulle, M.; Jones, B.L. A Proteinase from Germinating Barley: I. Purification and Some Physical Properties of a 30 kD Cysteine Endoproteinase from Green Malt. Plant Physiol. 1988, 88, 1454–1460. [Google Scholar] [CrossRef] [PubMed]
- Koehler, S.M.; Ho, T.H. A major gibberellic Acid-induced barley aleurone cysteine proteinase which digests hordein: Purification and characterization. Plant Physiol. 1990, 94, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Koehler, S.; Ho, T.H. Purification and characterization of gibberellic Acid-induced cysteine endoproteases in barley aleurone layers. Plant Physiol 1988, 87, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Mikkonen, A.; Porali, I.; Cercos, M.; Ho, T.H. A major cysteine proteinase, EPB, in germinating barley seeds: Structure of two intronless genes and regulation of expression. Plant Mol. Biol. 1996, 31, 239–254. [Google Scholar] [CrossRef]
- Rogers, J.C.; Dean, D.; Heck, G.R. Aleurain: A barley thiol protease closely related to mammalian cathepsin H. Proc. Natl. Acad. Sci. USA 1985, 82, 6512–6516. [Google Scholar] [CrossRef]
- Martinez, M.; Rubio-Somoza, I.; Carbonero, P.; Diaz, I. A cathepsin B-like cysteine protease gene from Hordeum vulgare (gene CatB) induced by GA in aleurone cells is under circadian control in leaves. J. Exp. Bot. 2003, 54, 951–959. [Google Scholar] [CrossRef]
- Cejudo, F.J.; Murphy, G.; Chinoy, C.; Baulcombe, D.C. A gibberellin-regulated gene from wheat with sequence homology to cathepsin B of mammalian cells. Plant J. 1992, 2, 937–948. [Google Scholar]
- Jivotovskaya, A.V.; Horstmann, C.; Vaintraub, I.A. Detection of the isoenzymes of wheat grain proteinase A. Phytochemistry 1997, 45, 1549–1553. [Google Scholar] [CrossRef]
- Sutoh, K.; Kato, H.; Minamikawa, T. Identification and possible roles of three types of endopeptidase from germinated wheat seeds. J. Biochem. 1999, 126, 700–707. [Google Scholar]
- Tsuji, A.; Tsuji, M.; Takami, H.; Nakamura, S.; Matsuda, Y. Molecular cloning and expression analysis of novel wheat cysteine protease. Biochim. Biophys. Acta 2004, 1670, 84–89. [Google Scholar] [CrossRef]
- Kiyosaki, T.; Matsumoto, I.; Asakura, T.; Funaki, J.; Kuroda, M.; Misaka, T.; Arai, S.; Abe, K. Gliadain, a gibberellin-inducible cysteine proteinase occurring in germinating seeds of wheat, Triticum aestivum L., specifically digests gliadin and is regulated by intrinsic cystatins. FEBS J. 2007, 274, 1908–1917. [Google Scholar] [CrossRef]
- Dal Degan, F.; Rocher, A.; Cameron-Mills, V.; von Wettstein, D. The expression of serine carboxypeptidases during maturation and germination of the barley grain. Proc. Natl. Acad. Sci. USA 1994, 91, 8209–8213. [Google Scholar] [CrossRef]
- Dominguez, F.; Cejudo, F.J. Patterns of starchy endosperm acidification and protease gene expression in wheat grains following germination. Plant Physiol. 1999, 119, 81–88. [Google Scholar] [CrossRef]
- Domínguez, F.; González, M.C.; Cejudo, F.J. A germination-related gene encoding a serine carboxypeptidase is expressed during the differentiation of the vascular tissue in wheat grains and seedlings. Planta 2002, 215, 727–734. [Google Scholar] [CrossRef]
- Törmäkangas, K.; Runeberg-Roos, P.; Ostman, A.; Tilgmann, C.; Sarkkinen, P.; Kervinen, J.; Mikola, L.; Kalkkinen, N. Aspartic proteinase from barley seeds is related to animal cathepsin D. Adv. Exp. Med. Biol. 1991, 306, 355–359. [Google Scholar]
- Tamura, T.; Terauchi, K.; Kiyosaki, T.; Asakura, T.; Funaki, J.; Matsumoto, I.; Misaka, T.; Abe, K. Differential expression of wheat aspartic proteinases, WAP1 and WAP2, in germinating and maturing seeds. J. Plant Physiol. 2007, 164, 470–477. [Google Scholar] [CrossRef]
- Close, T.J.; Wanamaker, S.I.; Caldo, R.A.; Turner, S.M.; Ashlock, D.A.; Dickerson, J.A.; Wing, R.A.; Muehlbauer, G.J.; Kleinhofs, A.; Wise, R.P. A new resource for cereal genomics: 22K barley GeneChip comes of age. Plant Physiol. 2004, 134, 960–968. [Google Scholar] [CrossRef]
- Daneri-Castro, S.N.; Svensson, B.; Roberts, T.H. Barley germination: Spatio-temporal considerations for designing and interpreting ‘omics’ experiments. J. Cereal Sci. 2016, 70, 29–37. [Google Scholar] [CrossRef]
- Sreenivasulu, N.; Usadel, B.; Winter, A.; Radchuk, V.; Scholz, U.; Stein, N.; Weschke, W.; Strickert, M.; Close, T.J.; Stitt, M.; et al. Barley grain maturation and germination: Metabolic pathway and regulatory network commonalities and differences highlighted by new MapMan/PageMan profiling tools. Plant Physiol. 2008, 146, 1738–1758. [Google Scholar] [CrossRef]
- Mayer, K.F.; Waugh, R.; Brown, J.W.; Schulman, A.; Langridge, P.; Platzer, M.; Fincher, G.B.; Muehlbauer, G.J.; Sato, K.; Close, T.J.; et al. A physical, genetic and functional sequence assembly of the barley genome. Nature 2012, 491, 711–716. [Google Scholar] [CrossRef]
- Martinez, M.; Diaz, I. The origin and evolution of plant cystatins and their target cysteine proteinases indicate a complex functional relationship. BMC Evol. Biol. 2008, 8, 198. [Google Scholar] [CrossRef]
- Diaz-Mendoza, M.; Velasco-Arroyo, B.; Gonzalez-Melendi, P.; Martinez, M.; Diaz, I. C1A cysteine protease-cystatin interactions in leaf senescence. J. Exp. Bot. 2014, 65, 3825–3833. [Google Scholar] [CrossRef]
- Martinez, M.; Cambra, I.; Carrillo, L.; Diaz-Mendoza, M.; Diaz, I. Characterization of the Entire Cystatin Gene Family in Barley and Their Target Cathepsin L-Like Cysteine-Proteases, Partners in the Hordein Mobilization during Seed Germination. Plant Physiol. 2009, 151, 1531–1545. [Google Scholar] [CrossRef]
- Cambra, I.; Martinez, M.; Dader, B.; Gonzalez-Melendi, P.; Gandullo, J.; Santamaria, M.E.; Diaz, I. A cathepsin F-like peptidase involved in barley grain protein mobilization, HvPap-1, is modulated by its own propeptide and by cystatins. J. Exp. Bot. 2012, 63, 4615–4629. [Google Scholar] [CrossRef]
- Radchuk, V.; Weier, D.; Radchuk, R.; Weschke, W.; Weber, H. Development of maternal seed tissue in barley is mediated by regulated cell expansion and cell disintegration and coordinated with endosperm growth. J. Exp. Bot. 2011, 62, 1217–1227. [Google Scholar] [CrossRef]
- Julian, I.; Gandullo, J.; Santos-Silva, L.K.; Diaz, I.; Martinez, M. Phylogenetically distant barley legumains have a role in both seed and vegetative tissues. J. Exp. Bot. 2013, 64, 2929–2941. [Google Scholar] [CrossRef]
- Cambra, I.; Garcia, F.J.; Martinez, M. Clan CD of cysteine peptidases as an example of evolutionary divergences in related protein families across plant clades. Gene 2010, 449, 59–69. [Google Scholar] [CrossRef][Green Version]
- Betts, N.S.; Berkowitz, O.; Liu, R.; Collins, H.M.; Skadhauge, B.; Dockter, C.; Burton, R.A.; Whelan, J.; Fincher, G.B. Isolation of tissues and preservation of RNA from intact, germinated barley grain. Plant J. 2017, 91, 754–765. [Google Scholar] [CrossRef] [PubMed]
- Galotta, M.F.; Pugliese, P.; Gutiérrez-Boem, F.H.; Veliz, C.G.; Criado, M.V.; Caputo, C.; Echeverria, M.; Roberts, I.N. Subtilase activity and gene expression during germination and seedling growth in barley. Plant Physiol. Biochem. 2019, 139, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zhen, S.; Wang, S.; Wang, Y.; Cao, H.; Zhang, Y.; Li, J.; Yan, Y. Comparative transcriptome analysis of wheat embryo and endosperm responses to ABA and H2O2 stresses during seed germination. BMC Genom. 2016, 17, 97. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Guo, G.; Lv, D.; Hu, Y.; Li, J.; Li, X.; Yan, Y. Transcriptome analysis during seed germination of elite Chinese bread wheat cultivar Jimai 20. BMC Plant Biol. 2014, 14, 20. [Google Scholar] [CrossRef] [PubMed]
- The International Wheat Genome Sequencing Consortium (IWGSC). A chromosome-based draft sequence of the hexaploid bread wheat (Triticum aestivum) genome. Science 2014, 345, 1251788. [Google Scholar] [CrossRef] [PubMed]
- Appels, R.; Eversole, K.; Feuillet, C.; Keller, B.; Rogers, J.; Stein, N.; Pozniak, C.J.; Choulet, F.; Distelfeld, A.; Poland, J.; et al. Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 2018, 361, aar7191. [Google Scholar] [CrossRef]
- Ramírez-González, R.H.; Borrill, P.; Lang, D.; Harrington, S.A.; Brinton, J.; Venturini, L.; Davey, M.; Jacobs, J.; van Ex, F.; Pasha, A.; et al. The transcriptional landscape of polyploid wheat. Science 2018, 361, aar6089. [Google Scholar] [CrossRef]
- Finnie, C.; Svensson, B. Barley seed proteomics from spots to structures. J. Proteom. 2009, 72, 315–324. [Google Scholar] [CrossRef]
- Finnie, C.; Andersen, B.; Shahpiri, A.; Svensson, B. Proteomes of the barley aleurone layer: A model system for plant signalling and protein secretion. Proteomics 2011, 11, 1595–1605. [Google Scholar] [CrossRef]
- Dong, K.; Zhen, S.; Cheng, Z.; Cao, H.; Ge, P.; Yan, Y. Proteomic Analysis Reveals Key Proteins and Phosphoproteins upon Seed Germination of Wheat (Triticum aestivum L.). Front. Plant Sci. 2015, 6, 1017. [Google Scholar] [CrossRef]
- He, M.; Zhu, C.; Dong, K.; Zhang, T.; Cheng, Z.; Li, J.; Yan, Y. Comparative proteome analysis of embryo and endosperm reveals central differential expression proteins involved in wheat seed germination. BMC Plant Biol. 2015, 15, 97. [Google Scholar] [CrossRef]
- Nadaud, I.; Tasleem-Tahir, A.; Chateigner-Boutin, A.L.; Chambon, C.; Viala, D.; Branlard, G. Proteome evolution of wheat (Triticum aestivum L.) aleurone layer at fifteen stages of grain development. J. Proteom. 2015, 123, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Mahalingam, R. Temporal Analyses of Barley Malting Stages Using Shotgun Proteomics. Proteomics 2018, 18, e1800025. [Google Scholar] [CrossRef]
- Rosenkilde, A.L.; Dionisio, G.; Holm, P.B.; Brinch-Pedersen, H. Production of barley endoprotease B2 in Pichia pastoris and its proteolytic activity against native and recombinant hordeins. Phytochemistry 2014, 97, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Mendoza, M.; Dominguez-Figueroa, J.D.; Velasco-Arroyo, B.; Cambra, I.; Gonzalez-Melendi, P.; Lopez-Gonzalvez, A.; Garcia, A.; Hensel, G.; Kumlehn, J.; Diaz, I.; et al. HvPap-1 C1A Protease and HvCPI-2 Cystatin Contribute to Barley Grain Filling and Germination. Plant Physiol. 2016, 170, 2511–2524. [Google Scholar] [CrossRef] [PubMed]
- Bethune, M.T.; Khosla, C. Oral enzyme therapy for celiac sprue. Methods Enzymol. 2012, 502, 241–271. [Google Scholar] [CrossRef] [PubMed]
- Scherf, K.A.; Wieser, H.; Koehler, P. Novel approaches for enzymatic gluten degradation to create high-quality gluten-free products. Food Res. Int. 2018, 110, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, G.; Koehler, P.; Wieser, H. Rapid degradation of gliadin peptides toxic for coeliac disease patients by proteases from germinating cereals. J. Cereal Sci. 2006, 44, 368–371. [Google Scholar] [CrossRef]
- Kiyosaki, T.; Asakura, T.; Matsumoto, I.; Tamura, T.; Terauchi, K.; Funaki, J.; Kuroda, M.; Misaka, T.; Abe, K. Wheat cysteine proteases triticain alpha, beta and gamma exhibit mutually distinct responses to gibberellin in germinating seeds. J. Plant Physiol. 2009, 166, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Savvateeva, L.V.; Gorokhovets, N.V.; Makarov, V.A.; Serebryakova, M.V.; Solovyev, A.G.; Morozov, S.Y.; Reddy, V.P.; Zernii, E.Y.; Zamyatnin, A.A.; Aliev, G. Glutenase and collagenase activities of wheat cysteine protease Triticain-α: Feasibility for enzymatic therapy assays. Int. J. Biochem. Cell Biol. 2015, 62, 115–124. [Google Scholar] [CrossRef]
- Cravatt, B.F.; Wright, A.T.; Kozarich, J.W. Activity-based protein profiling: From enzyme chemistry to proteomic chemistry. Annu. Rev. Biochem. 2008, 77, 383–414. [Google Scholar] [CrossRef]
- Lu, H.; Chandrasekar, B.; Oeljeklaus, J.; Misas-Villamil, J.C.; Wang, Z.; Shindo, T.; Bogyo, M.; Kaiser, M.; van der Hoorn, R.A. Subfamily-Specific Fluorescent Probes for Cysteine Proteases Display Dynamic Protease Activities during Seed Germination. Plant Physiol. 2015, 168, 1462–1475. [Google Scholar] [CrossRef] [PubMed]
- Daneri-Castro, S.N.; Chandrasekar, B.; Grosse-Holz, F.M.; van der Hoorn, R.A.; Roberts, T.H. Activity-based protein profiling of hydrolytic enzymes induced by gibberellic acid in isolated aleurone layers of malting barley. FEBS Lett. 2016, 590, 2956–2962. [Google Scholar] [CrossRef] [PubMed]
- Kok, Y.J.; Ye, L.; Muller, J.; Ow, D.S.; Bi, X. Brewing with malted barley or raw barley: What makes the difference in the processes? Appl. Microbiol. Biotechnol. 2019, 103, 1059–1067. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-León, S.; Gil-Humanes, J.; Ozuna, C.V.; Giménez, M.J.; Sousa, C.; Voytas, D.F.; Barro, F. Low-gluten, nontransgenic wheat engineered with CRISPR/Cas9. Plant Biotechnol. J. 2018, 16, 902–910. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diaz-Mendoza, M.; Diaz, I.; Martinez, M. Insights on the Proteases Involved in Barley and Wheat Grain Germination. Int. J. Mol. Sci. 2019, 20, 2087. https://doi.org/10.3390/ijms20092087
Diaz-Mendoza M, Diaz I, Martinez M. Insights on the Proteases Involved in Barley and Wheat Grain Germination. International Journal of Molecular Sciences. 2019; 20(9):2087. https://doi.org/10.3390/ijms20092087
Chicago/Turabian StyleDiaz-Mendoza, Mercedes, Isabel Diaz, and Manuel Martinez. 2019. "Insights on the Proteases Involved in Barley and Wheat Grain Germination" International Journal of Molecular Sciences 20, no. 9: 2087. https://doi.org/10.3390/ijms20092087
APA StyleDiaz-Mendoza, M., Diaz, I., & Martinez, M. (2019). Insights on the Proteases Involved in Barley and Wheat Grain Germination. International Journal of Molecular Sciences, 20(9), 2087. https://doi.org/10.3390/ijms20092087






