Safety Assessment of Compounds after In Vitro Metabolic Conversion Using Zebrafish Eleuthero Embryos
Abstract
1. Introduction
2. Results
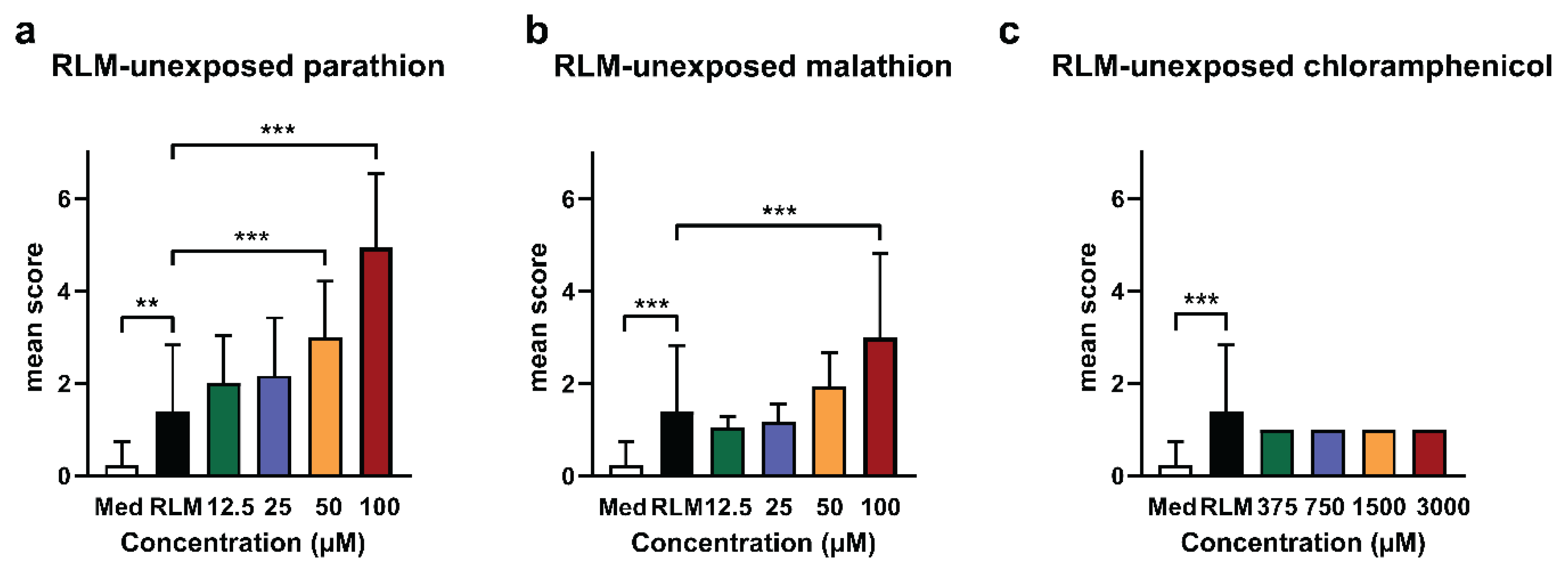
2.1. Adverse Effects of Test Compounds Unexposed to RLM in Zebrafish Eleuthero Embryos
2.2. Quantitative Determination of Recovery Yield and Metabolic Conversion of Test Compounds after RLM-Exposure
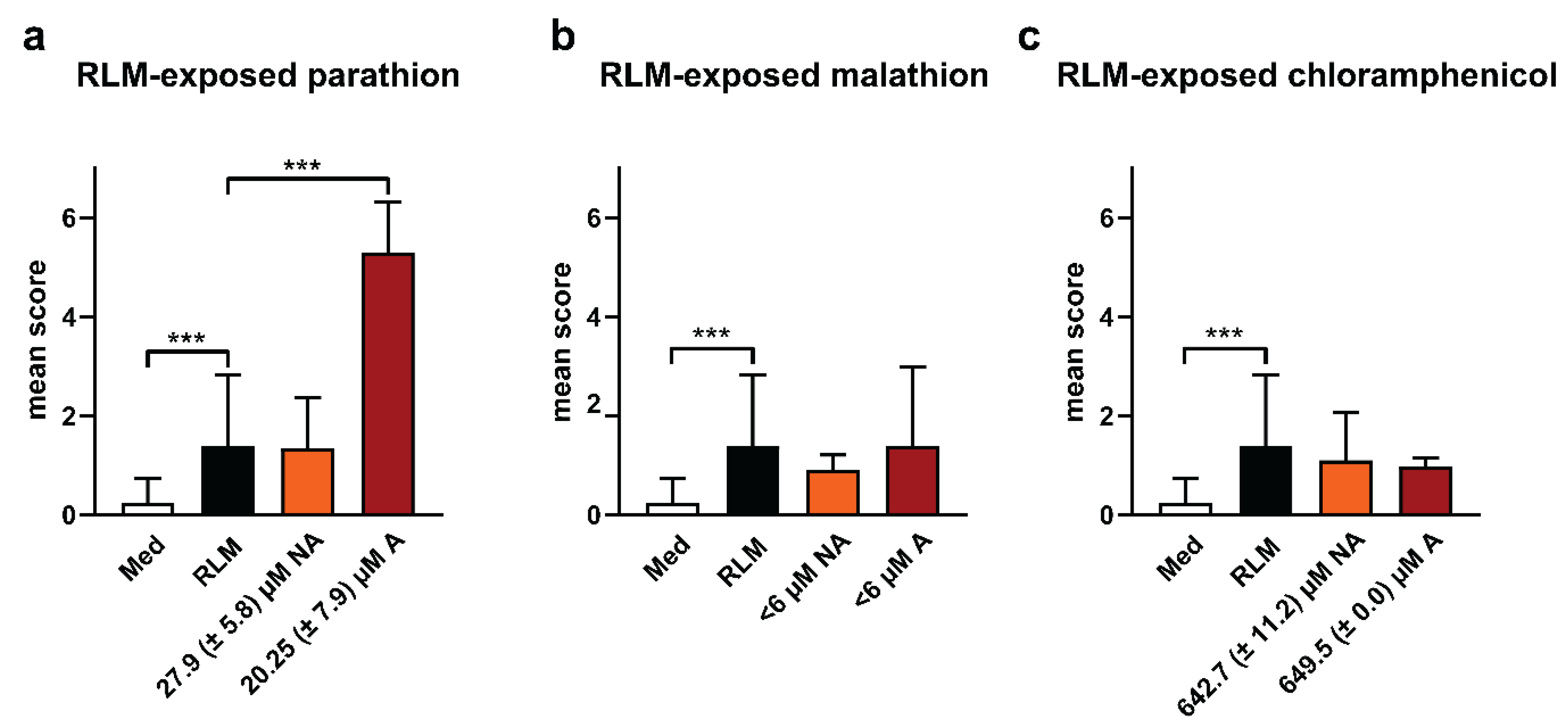
2.3. Adverse Effects of Test Compounds Exposed to RLM in Zebrafish Eleuthero Embryos
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Fish Eleuthero Embryo Tests
4.3. Determination of Sub-Lethal Toxicity and Lethality of Test Compounds Exposed or Unexposed to RLM in Zebrafish Eleuthero Embryos
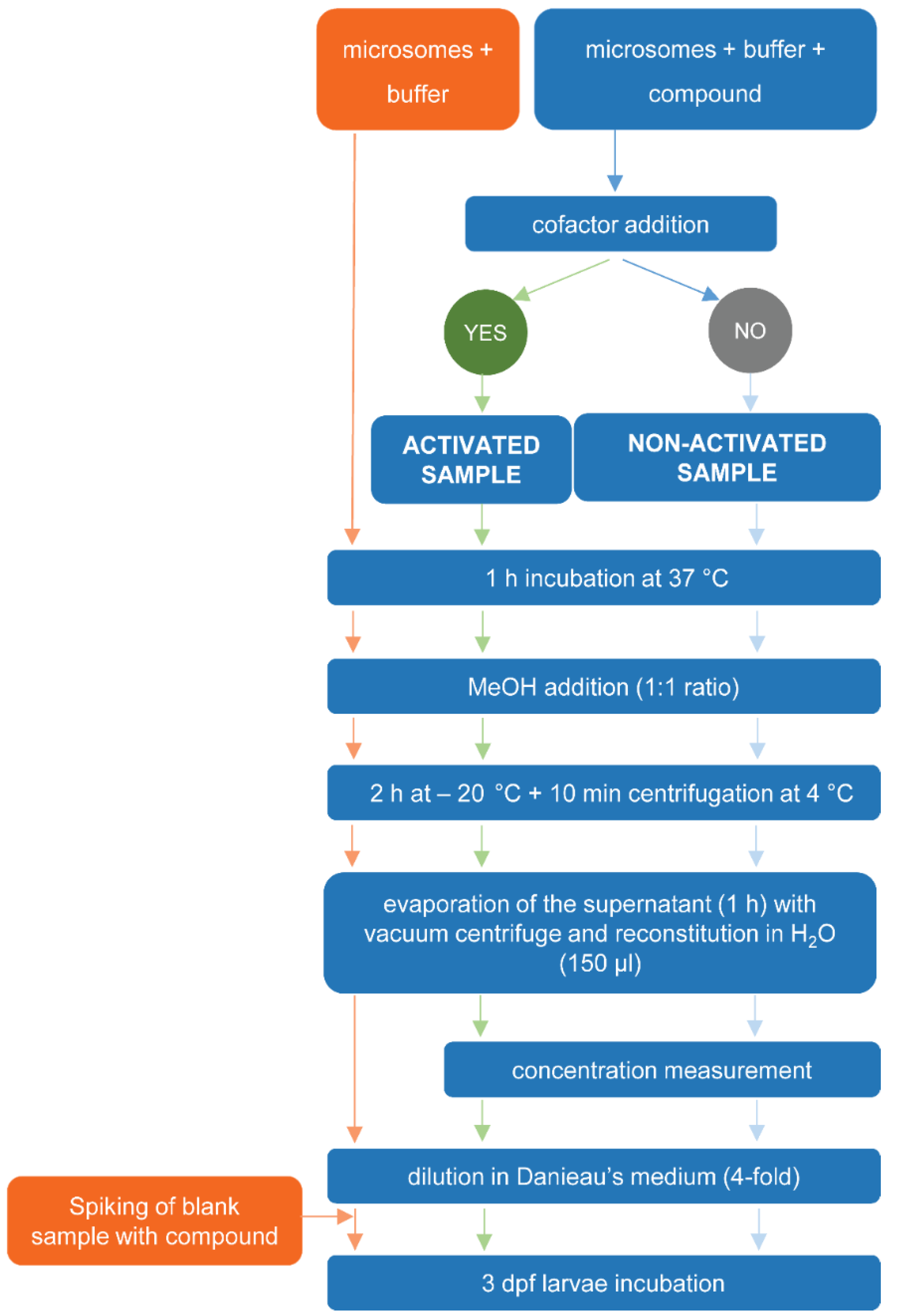
4.3.1. Exposure of Test Compound or Blank Samples to RLM
4.3.2. Preparation of Sample Extracts
4.3.3. Quantitative Determination of Recovery Yield and Metabolic Conversion of Test Compounds Exposed to RLM
4.3.4. Biological Assay Using Zebrafish Eleuthero Embryos
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| RLM | Rat liver microsomes |
| UHPLC-UV-MS | Ultra high performance liquid chromatography-Ultraviolet-Mass |
| dpf | Days post fertilization |
| hpf | Hours post fertilization |
| mDarT assay | Danio rerio teratogenic assay with metabolic activation |
| MIB | Microsome incubation buffer |
| NADPH | Reduced β-nicotinamide adenine dinucleotide 2′-phosphate |
| G6P | Glucose-6-phosphate |
| MMC | Monocarboxylic acid derivatives of malathion |
References
- Guengerich, F.P. Mechanisms of Drug Toxicity and Relevance to Pharmaceutical Development. Drug Metab. Pharmacokinet. 2011, 26, 3–14. [Google Scholar] [CrossRef]
- Onakpoya, I.J.; Heneghan, C.J.; Aronson, J.K. Post-marketing withdrawal of 462 medicinal products because of adverse drug reactions: a systematic review of the world literature. BMC Med. 2016, 14. [Google Scholar] [CrossRef]
- Kari, G.; Rodeck, U.; Dicker, A.P. Zebrafish: an emerging model system for human disease and drug discovery. Clin. Pharmacol. Ther. 2007, 82, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Cornet, C.; Calzolari, S.; Miñana-Prieto, R.; Dyballa, S.; van Doornmalen, E.; Rutjes, H.; Savy, T.; D’Amico, D.; Terriente, J. ZeGlobalTox: An Innovative Approach to Address Organ Drug Toxicity Using Zebrafish. Int. J. Mol. Sci. 2017, 18, 864. [Google Scholar] [CrossRef]
- Olson, H.; Betton, G.; Robinson, D.; Thomas, K.; Monro, A.; Kolaja, G.; Lilly, P.; Sanders, J.; Sipes, G.; Bracken, W.; et al. Concordance of the Toxicity of Pharmaceuticals in Humans and in Animals. Regul. Toxicol. Pharmacol. 2000, 32, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.; Mesens, N.; Steemans, M.; Xu, J.J.; Aleo, M.D. Comparisons between in vitro whole cell imaging and in vivo zebrafish-based approaches for identifying potential human hepatotoxicants earlier in pharmaceutical development. Drug Metab. Rev. 2012, 44, 127–140. [Google Scholar] [CrossRef]
- Rubinstein, A.L. Zebrafish assays for drug toxicity screening. Expert Opin. Drug Metab. Toxicol. 2006, 2, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Barros, T.P.; Alderton, W.K.; Reynolds, H.M.; Roach, A.G.; Berghmans, S. Zebrafish: an emerging technology for in vivo pharmacological assessment to identify potential safety liabilities in early drug discovery. Br. J. Pharmacol. 2008, 154, 1400–1413. [Google Scholar] [CrossRef]
- Li, C.; Luo, L.; Awerman, J.; McGrath, P. Whole Zebrafish Cytochrome P450 Assay for Assessing Drug Metabolism and Safety. In Zebrafish; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2011; pp. 103–115. ISBN 978-1-118-10213-8. [Google Scholar]
- Eimon, P.M.; Rubinstein, A.L. The use of in vivo zebrafish assays in drug toxicity screening. Expert Opin. Drug Metab. Toxicol. 2009, 5, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Selderslaghs, I.W.T.; Blust, R.; Witters, H.E. Feasibility study of the zebrafish assay as an alternative method to screen for developmental toxicity and embryotoxicity using a training set of 27 compounds. Reprod. Toxicol. 2012, 33, 142–154. [Google Scholar] [CrossRef]
- Van den Bulck, K.; Hill, A.; Mesens, N.; Diekman, H.; De Schaepdrijver, L.; Lammens, L. Zebrafish developmental toxicity assay: A fishy solution to reproductive toxicity screening, or just a red herring? Reprod. Toxicol. 2011, 32, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Halder, M.; Léonard, M.; Iguchi, T.; Oris, J.T.; Ryder, K.; Belanger, S.E.; Braunbeck, T.A.; Embry, M.R.; Whale, G.; Norberg-King, T.; et al. Regulatory aspects on the use of fish embryos in environmental toxicology. Integr. Environ. Assess. Manag. 2010, 6, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef]
- Garcia, G.R.; Noyes, P.D.; Tanguay, R.L. Advancements in zebrafish applications for 21st century toxicology. Pharmacol. Ther. 2016, 161, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Verstraelen, S.; Peers, B.; Maho, W.; Hollanders, K.; Remy, S.; Berckmans, P.; Covaci, A.; Witters, H. Phenotypic and biomarker evaluation of zebrafish larvae as an alternative model to predict mammalian hepatotoxicity. J. Appl. Toxicol. 2016, 36, 1194–1206. [Google Scholar] [CrossRef] [PubMed]
- King, A. Researchers Find Their Nemo. Cell 2009, 139, 843–846. [Google Scholar] [CrossRef]
- Alderton, W.; Berghmans, S.; Butler, P.; Chassaing, H.; Fleming, A.; Golder, Z.; Richards, F.; Gardner, I. Accumulation and metabolism of drugs and CYP probe substrates in zebrafish larvae. Xenobiotica 2010, 40, 547–557. [Google Scholar] [CrossRef]
- Busquet, F.; Nagel, R.; von Landenberg, F.; Mueller, S.O.; Huebler, N.; Broschard, T.H. Development of a new screening assay to identify proteratogenic substances using zebrafish danio rerio embryo combined with an exogenous mammalian metabolic activation system (mDarT). Toxicol. Sci. 2008, 104, 177–188. [Google Scholar] [CrossRef]
- Weigt, S.; Huebler, N.; Braunbeck, T.; von Landenberg, F.; Broschard, T.H. Zebrafish teratogenicity test with metabolic activation (mDarT): effects of phase I activation of acetaminophen on zebrafish Danio rerio embryos. Toxicology 2010, 275, 36–49. [Google Scholar] [CrossRef]
- Mattsson, A.; Ullerås, E.; Patring, J.; Oskarsson, A. Albendazole causes stage-dependent developmental toxicity and is deactivated by a mammalian metabolization system in a modified zebrafish embryotoxicity test. Reprod. Toxicol. 2012, 34, 31–42. [Google Scholar] [CrossRef]
- Nguyen, X.-B.; Kislyuk, S.; Pham, D.-H.; Kecskés, A.; Maes, J.; Cabooter, D.; Annaert, P.; De Witte, P.; Ny, A. Cell Imaging Counting as a Novel Ex Vivo Approach for Investigating Drug-Induced Hepatotoxicity in Zebrafish Larvae. Int. J. Mol. Sci. 2017, 18, 356. [Google Scholar] [CrossRef]
- Mesens, N.; Crawford, A.D.; Menke, A.; Hung, P.D.; Van Goethem, F.; Nuyts, R.; Hansen, E.; Wolterbeek, A.; Van Gompel, J.; De Witte, P.; et al. Are zebrafish larvae suitable for assessing the hepatotoxicity potential of drug candidates? J. Appl. Toxicol. 2015, 35, 1017–1029. [Google Scholar] [CrossRef] [PubMed]
- Pham, D.-H.; De Roo, B.; Nguyen, X.-B.; Vervaele, M.; Kecskés, A.; Ny, A.; Copmans, D.; Vriens, H.; Locquet, J.-P.; Hoet, P.; et al. Use of Zebrafish Larvae as a Multi-Endpoint Platform to Characterize the Toxicity Profile of Silica Nanoparticles. Sci. Rep. 2016, 6, 37145. [Google Scholar] [CrossRef]
- Cano-Nicolau, J.; Vaillant, C.; Pellegrini, E.; Charlier, T.D.; Kah, O.; Coumailleau, P. Estrogenic Effects of Several BPA Analogs in the Developing Zebrafish Brain. Front. Neurosci. 2016, 10. [Google Scholar] [CrossRef]
- Vong, L.B.; Kobayashi, M.; Nagasaki, Y. Evaluation of the Toxicity and Antioxidant Activity of Redox Nanoparticles in Zebrafish (Danio rerio) Embryos. Mol. Pharm. 2016, 13, 3091–3097. [Google Scholar] [CrossRef]
- Butler, A.M.; Murray, M. Biotransformation of Parathion in Human Liver: Participation of CYP3A4 and its Inactivation during Microsomal Parathion Oxidation. J. Pharmacol. Exp. Ther. 1997, 280, 966–973. [Google Scholar]
- Albores, A.; Ortega-Mantilla, G.; Sierra-Santoyo, A.; Cebrián, M.E.; Muñoz-Sánchez, J.L.; Calderón-Salinas, J.V.; Manno, M. Cytochrome P450 2B (CYP2B)-mediated activation of methyl-parathion in rat brain extracts. Toxicol. Lett. 2001, 124, 1–10. [Google Scholar] [CrossRef]
- Bouchard, M.; Gosselin, N.H.; Brunet, R.C.; Samuel, O.; Dumoulin, M.-J.; Carrier, G. A Toxicokinetic Model of Malathion and Its Metabolites as a Tool to Assess Human Exposure and Risk through Measurements of Urinary Biomarkers. Toxicol. Sci. 2003, 73, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Buratti, F.M.; D’Aniello, A.; Volpe, M.T.; Meneguz, A.; Testai, E. Malathion Bioactivation in the Human Liver: The Contribution of Different Cytochrome P450 Isoforms. Drug Metab. Dispos. 2005, 33, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Howe, D.; Leduc, B.; Kerr, S.; Williams, D.A. Identification and characterization of two chloramphenicol glucuronides from the in vitro glucuronidation of chloramphenicol in human liver microsomes. Xenobiotica 2007, 37, 954–971. [Google Scholar] [CrossRef] [PubMed]
- Skidmore, J.F. Resistance to zinc sulphate of the zebrafish (Brachydanio rerio Hamilton-Buchanan) at different phases of its life history. Ann. Appl. Biol. 1965, 56, 47–53. [Google Scholar] [CrossRef]
- Driessen, M.; Kienhuis, A.S.; Pennings, J.L.A.; Pronk, T.E.; van de Brandhof, E.-J.; Roodbergen, M.; Spaink, H.P.; van de Water, B.; van der Ven, L.T.M. Exploring the zebrafish embryo as an alternative model for the evaluation of liver toxicity by histopathology and expression profiling. Arch. Toxicol. 2013, 87, 807–823. [Google Scholar] [CrossRef]
- Milan, D.J.; Peterson, T.A.; Ruskin, J.N.; Peterson, R.T.; MacRae, C.A. Drugs that induce repolarization abnormalities cause bradycardia in zebrafish. Circulation 2003, 107, 1355–1358. [Google Scholar] [CrossRef]
- Goldstone, J.V.; McArthur, A.G.; Kubota, A.; Zanette, J.; Parente, T.; Jönsson, M.E.; Nelson, D.R.; Stegeman, J.J. Identification and developmental expression of the full complement of Cytochrome P450 genes in Zebrafish. BMC Genomics 2010, 11, 643. [Google Scholar] [CrossRef]
- Verbueken, E.; Bars, C.; Ball, J.S.; Periz-Stanacev, J.; Marei, W.F.A.; Tochwin, A.; Gabriëls, I.J.; Michiels, E.D.G.; Stinckens, E.; Vergauwen, L.; et al. From mRNA Expression of Drug Disposition Genes to In Vivo Assessment of CYP-Mediated Biotransformation during Zebrafish Embryonic and Larval Development. Int. J. Mol. Sci. 2018, 19, 3976. [Google Scholar] [CrossRef] [PubMed]
- Parathion. Available online: http://pmep.cce.cornell.edu/profiles/extoxnet/metiram-propoxur/parathion-ext.html (accessed on 30 January 2019).
- Ellison, C.A.; Tian, Y.; Knaak, J.B.; Kostyniak, P.J.; Olson, J.R. Human Hepatic Cytochrome P450-Specific Metabolism of the Organophosphorus Pesticides Methyl Parathion and Diazinon. Drug Metab. Dispos. 2012, 40, 1–5. [Google Scholar] [CrossRef]
- Zuber, R.; Anzenbacherová, E.; Anzenbacher, P. Cytochromes P450 and experimental models of drug metabolism. J. Cell. Mol. Med. 2002, 6, 189–198. [Google Scholar] [CrossRef]
- Kislyuk, S.; Kroonen, J.; Adams, E.; Augustijns, P.; de Witte, P.; Cabooter, D. Development of a sensitive and quantitative UHPLC-MS/MS method to study the whole-body uptake of pharmaceuticals in zebrafish. Talanta 2017, 174, 780–788. [Google Scholar] [CrossRef]
- John, S.; Kale, M.; Rathore, N.; Bhatnagar, D. Protective effect of vitamin E in dimethoate and malathion induced oxidative stress in rat erythrocytes. J. Nutr. Biochem. 2001, 12, 500–504. [Google Scholar] [CrossRef]
- Martignoni, M.; Groothuis, G.M.M.; Kanter, R. de Species differences between mouse, rat, dog, monkey and human CYP-mediated drug metabolism, inhibition and induction. Expert Opin. Drug Metab. Toxicol. 2006, 2, 875–894. [Google Scholar] [CrossRef]
- Eriksson, E.; Auffarth, K.; Eilersen, A.-M.; Henze, M.; Ledin, A. Household chemicals and personal care products as sources for xenobiotic organic compounds in grey wastewater. Water SA 2003, 29. [Google Scholar] [CrossRef]
- Shapiro, H.; Micucci, S. Pesticide use for West Nile virus. CMAJ 2003, 168, 1427–1430. [Google Scholar]
- Palmer, D.L.; Despopoulos, A.; Rael, E.D. Induction of Chloramphenicol Metabolism by Phenobarbital. Antimicrob Agents Chemother 1972, 1, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Korsgaard Christensen, L.; Skovsted, L. INHIBITION OF DRUG METABOLISM BY CHLORAMPHENICOL. The Lancet 1969, 294, 1397–1399. [Google Scholar] [CrossRef]
- Pohl, L.R.; Krishna, G. Study of the mechanism of metabolic activation of chloramphenicol by rat liver microsomes. Biochem. Pharmacol. 1978, 27, 335–341. [Google Scholar] [CrossRef]
- Nicolaï, J.; Bruyn, T.D.; Veldhoven, P.P.V.; Keemink, J.; Augustijns, P.; Annaert, P. Verapamil hepatic clearance in four preclinical rat models: towards activity-based scaling. Biopharm. Drug Dispos. 2015, 36, 462–480. [Google Scholar] [CrossRef] [PubMed]

| Adverse Effect | Description |
|---|---|
| Bad development (BD) | Truncated body/ length and abnormal body shape (Figure 4a) |
| Curved body (CB) | (Figure 4b) |
| Impaired motility (IM) | No touch response or reduced touch response |
| Swim bladder defects (SBD) | Absent/undeveloped swim bladder (Figure 4a,c) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giusti, A.; Nguyen, X.-B.; Kislyuk, S.; Mignot, M.; Ranieri, C.; Nicolaï, J.; Oorts, M.; Wu, X.; Annaert, P.; De Croze, N.; et al. Safety Assessment of Compounds after In Vitro Metabolic Conversion Using Zebrafish Eleuthero Embryos. Int. J. Mol. Sci. 2019, 20, 1712. https://doi.org/10.3390/ijms20071712
Giusti A, Nguyen X-B, Kislyuk S, Mignot M, Ranieri C, Nicolaï J, Oorts M, Wu X, Annaert P, De Croze N, et al. Safety Assessment of Compounds after In Vitro Metabolic Conversion Using Zebrafish Eleuthero Embryos. International Journal of Molecular Sciences. 2019; 20(7):1712. https://doi.org/10.3390/ijms20071712
Chicago/Turabian StyleGiusti, Arianna, Xuan-Bac Nguyen, Stanislav Kislyuk, Mélanie Mignot, Cecilia Ranieri, Johan Nicolaï, Marlies Oorts, Xiao Wu, Pieter Annaert, Noémie De Croze, and et al. 2019. "Safety Assessment of Compounds after In Vitro Metabolic Conversion Using Zebrafish Eleuthero Embryos" International Journal of Molecular Sciences 20, no. 7: 1712. https://doi.org/10.3390/ijms20071712
APA StyleGiusti, A., Nguyen, X.-B., Kislyuk, S., Mignot, M., Ranieri, C., Nicolaï, J., Oorts, M., Wu, X., Annaert, P., De Croze, N., Léonard, M., Ny, A., Cabooter, D., & de Witte, P. (2019). Safety Assessment of Compounds after In Vitro Metabolic Conversion Using Zebrafish Eleuthero Embryos. International Journal of Molecular Sciences, 20(7), 1712. https://doi.org/10.3390/ijms20071712





