Lack of Cathepsin D in the Renal Proximal Tubular Cells Resulted in Increased Sensitivity against Renal Ischemia/Reperfusion Injury
Abstract
1. Introduction
2. Results
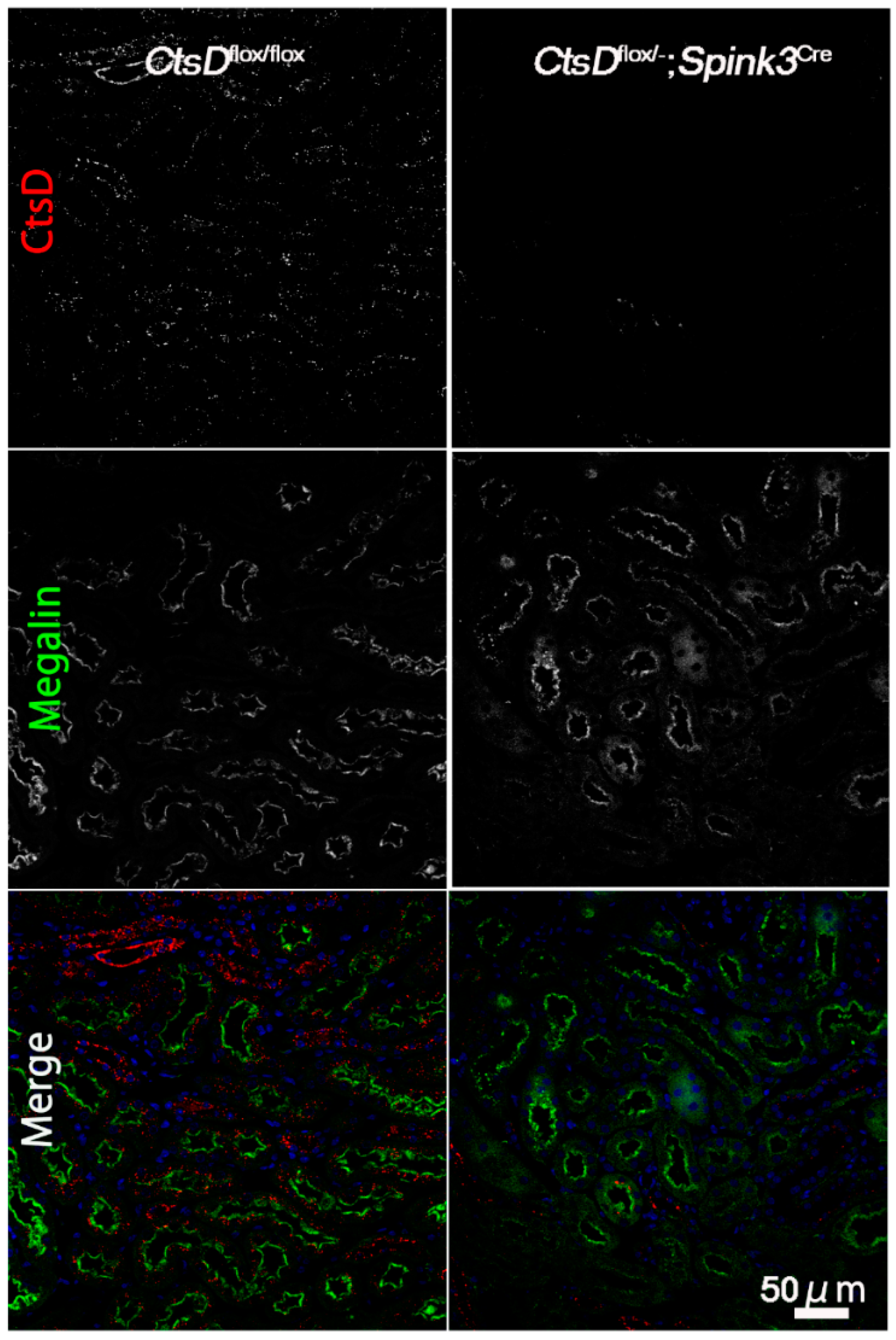
2.1. Cathepsin D-Deficiency Caused an Increase of LC3-Positive Signals in Renal Proximal Tubular Epithelial Cells
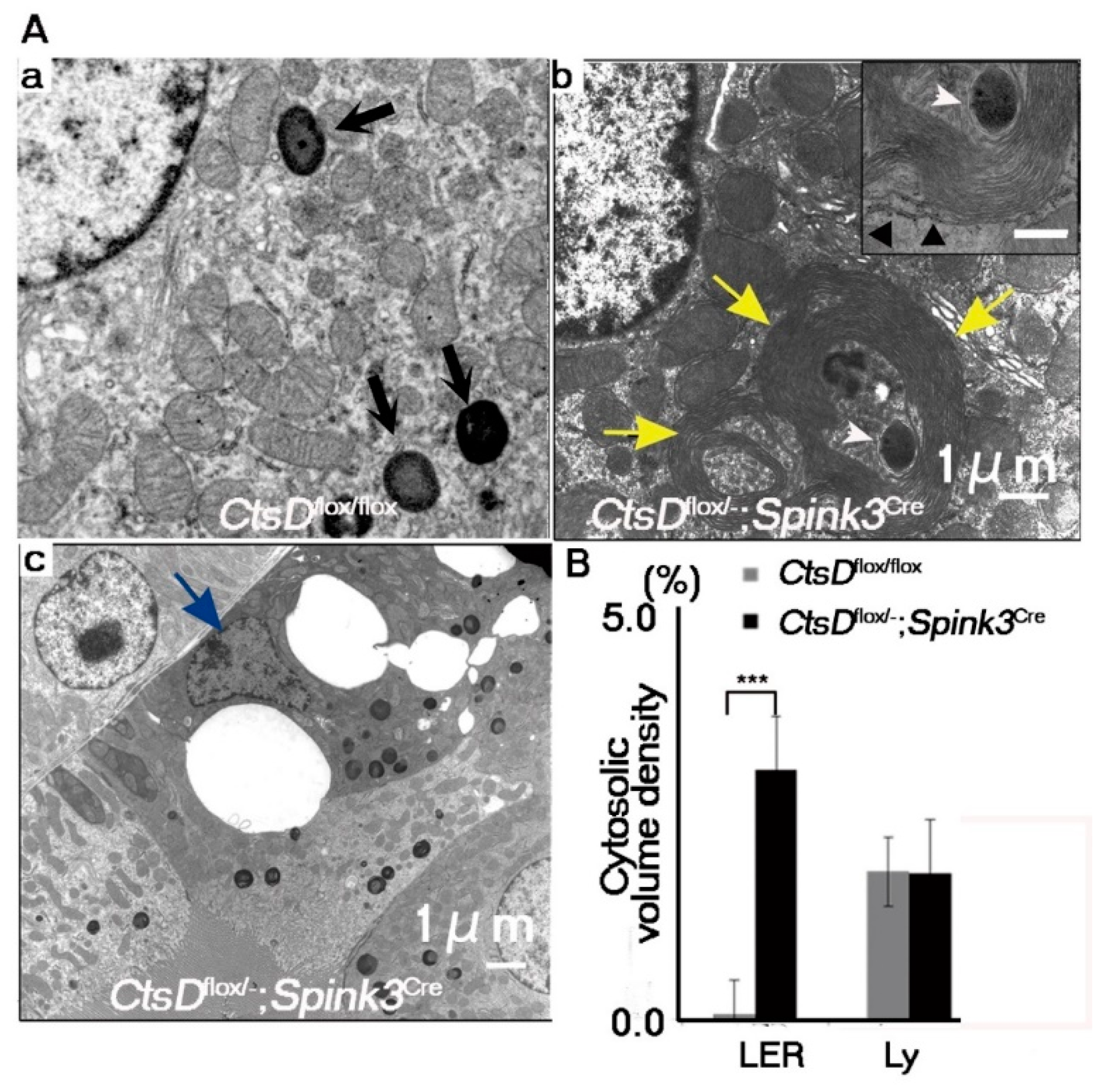
2.2. Cathepsin D-Deficiency Causes an Accumulation of Abnormal Autophagic Structures in Renal Proximal Tubular Epithelial Cells
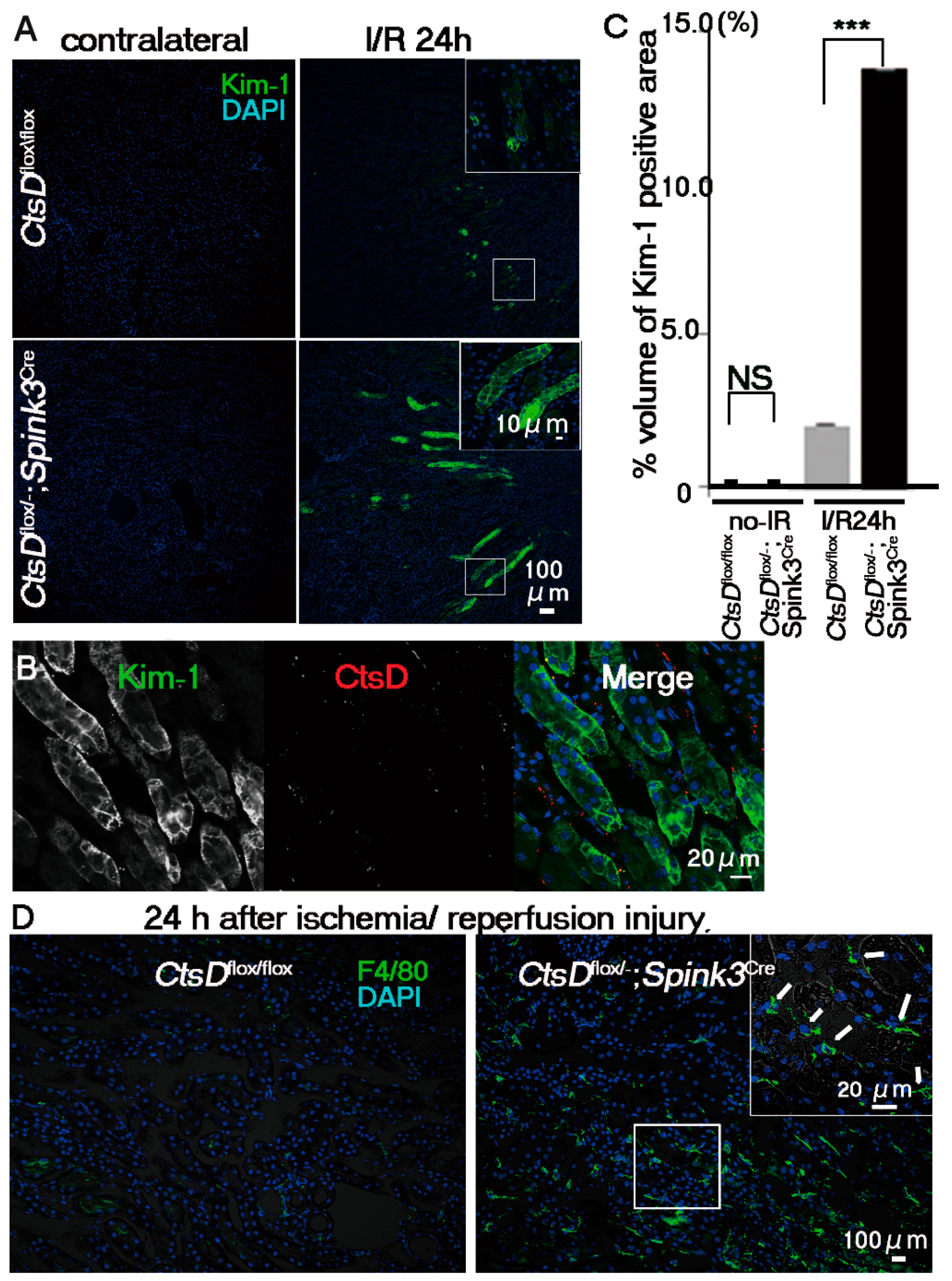
2.3. Cathepsin D-Deficiency Increased the Sensitivity against Renal Ischemia/Reperfusion Injury in Renal Proximal Tubular Epithelial Cells of the CtsDflox/−; Spink3Cre Mouse
3. Discussion
4. Materials and Methods
4.1. Animal Model
4.2. Antibodies
4.3. Fixation and Embedding for Light and Electron Microscopy
4.4. Immunohistochemistry
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| LC3 | Microtubule-associated protein light chain 3 |
| Kim-1 | Kidney injury molecule 1 |
References
- Dean, R.T. Lysosomes and protein degradation. Ciba Found Symp. 1979, 75, 139–149. [Google Scholar]
- Jones, E.W.; Zubenko, G.S.; Parker, R.R. PEP4 gene function is required for expression of several vacuolar hydrolases in Saccharomyces cerevisiae. Genetics 1982, 102, 665–677. [Google Scholar] [PubMed]
- Saftig, P.; Hetman, M.; Schmahl, W.; Weber, K.; Heine, L.; Mossmann, H.; Köster, A.; Hess, B.; Evers, M.; von Figura, K. Mice deficient for the lysosomal proteinase cathepsin D exhibit progressive atrophy of the intestinal mucosa and profound destruction of lymphoid cells. EMBO J. 1995, 14, 3599–3608. [Google Scholar] [CrossRef]
- Koike, M.; Nakanishi, H.; Saftig, P.; Ezaki, J.; Isahara, K.; Ohsawa, Y.; Schulz-Schaeffer, W.; Watanabe, T.; Waguri, S.; Kametaka, S.; et al. Cathepsin D deficiency induces lysosomal storage with ceroid lipofuscin in mouse CNS neurons. J. Neurosci. 2000, 20, 6898–6906. [Google Scholar] [CrossRef]
- Nakanishi, H.; Zhang, J.; Koike, M.; Nishioku, T.; Okamoto, Y.; Kominami, E.; von Figura, K.; Peters, C.; Yamamoto, K.; Saftig, P.; et al. Involvement of nitric oxide released from microglia-macrophages in pathological changes of cathepsin D-deficient mice. J. Neurosci. 2001, 21, 7526–7533. [Google Scholar] [CrossRef] [PubMed]
- Koike, M.; Shibata, M.; Ohsawa, Y.; Nakanishi, H.; Koga, T.; Kametaka, S.; Waguri, S.; Momoi, T.; Kominami, E.; Peters, C.; et al. Involvement of two different cell death pathways in retinal atrophy of cathepsin D-deficient mice. Mol. Cell. Neurosci. 2003, 22, 146–161. [Google Scholar] [CrossRef]
- Siintola, E.; Partanen, S.; Strömme, P.; Haapanen, A.; Haltia, M.; Maehlen, J.; Lehesjoki, A.E.; Tyynelä, J. Cathepsin D deficiency underlies congenital human neuronal ceroid-lipofuscinosis. Brain 2006, 129, 1438–1445. [Google Scholar] [CrossRef] [PubMed]
- Koike, M.; Shibata, M.; Waguri, S.; Yoshimura, K.; Tanida, I.; Kominami, E.; Gotow, T.; Peters, C.; von Figura, K.; Mizushima, N.; et al. Participation of autophagy in storage of lysosomes in neurons from mouse models of neuronal ceroid-lipofuscinoses (Batten disease). Am. J. Pathol. 2005, 167, 1713–1728. [Google Scholar] [CrossRef]
- Havasi, A.; Borkan, S.C. Apoptosis and acute kidney injury. Kidney Int. 2011, 80, 29–40. [Google Scholar] [CrossRef]
- Bonventre, J.V.; Weinberg, J.M. Recent advances in the pathophysiology of ischemic acute renal failure. J. Am. Soc. Nephrol. 2003, 14, 2199–2210. [Google Scholar] [CrossRef]
- Bonventre, J.V.; Yang, L. Cellular pathophysiology of ischemic acute kidney injury. J. Clin. Investig. 2011, 121, 4210–4221. [Google Scholar] [CrossRef] [PubMed]
- Bonegio, R.; Lieberthal, W. Role of apoptosis in the pathogenesis of acute renal failure. Curr. Opin. Nephrol. Hypertens. 2002, 11, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Lieberthal, W.; Rennke, H.G.; Sandock, K.M.; Valeri, C.R.; Levinsky, N.G. Ischemia in the isolated erythrocyte-perfused rat kidney. Protective effect of hypothermia. Kidney Blood Press. Res. 1988, 11, 60–69. [Google Scholar] [CrossRef]
- Lameire, N.; Van Biesen, W.; Vanholder, R. Acute renal failure. Lancet 2005, 365, 417–430. [Google Scholar] [CrossRef]
- Cocchiaro, P.; Fox, C.; Tregidgo, N.W.; Howarth, R.; Wood, K.M.; Situmorang, G.R.; Pavone, L.M.; Sheerin, N.S.; Moles, A. Lysosomal protease cathepsin D; a new driver of apoptosis during acute kidney injury. Sci. Rep. 2016, 6, 27112. [Google Scholar] [CrossRef] [PubMed]
- Fox, C.; Cocchiaro, P.; Oakley, F.; Howarth, R.; Callaghan, K.; Leslie, J.; Luli, S.; Wood, K.M.; Genovese, F.; Sheerin, N.S.; et al. Inhibition of lysosomal protease cathepsin D reduces renal fibrosis in murine chronic kidney disease. Sci. Rep. 2016, 6, 20101. [Google Scholar] [CrossRef] [PubMed]
- Sakata, K.; Ohmuraya, M.; Araki, K.; Suzuki, C.; Ida, S.; Hashimoto, D.; Wang, J.; Uchiyama, Y.; Baba, H.; Yamamura, K. Generation and analysis of serine protease inhibitor kazal type 3-cre driver mice. Exp. Anim. 2014, 63, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Klionsky, D.J.; Abdelmohsen, K.; Abe, A.; Abedin, M.J.; Abeliovich, H.; Acevedo Arozena, A.; Adachi, H.; Adams, C.M.; Adams, P.D.; Adeli, K.; et al. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy 2016, 12. [Google Scholar] [CrossRef] [PubMed]
- Koike, M.; Shibata, M.; Sunabori, T.; Yamaguchi, J.; Sakimura, K.; Komatsu, M.; Tanaka, K.; Uchiyama, Y. Purkinje Cells Are More Vulnerable to the Specific Depletion of Cathepsin D Than to That of Atg7. Am. J. Pathol. 2017, 187, 1586–1600. [Google Scholar] [CrossRef]
- Uchiyama, Y.; Watanabe, M. A morphometric study of developing pancreatic acinar cells of rats during prenatal life. Cell Tissue Res. 1984, 237, 117–122. [Google Scholar] [CrossRef]
- Han, W.K.; Bailly, V.; Abichandani, R.; Thadhani, R.; Bonventre, J.V. Kidney Injury Molecule-1 (KIM-1): A novel biomarker for human renal proximal tubule injury. Kidney Int. 2002, 62, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Pavlock, G.S.; Southard, J.H.; Starling, J.R.; Belzer, F.O. Lysosomal enzyme release in hypothermically perfused dog kidneys. Cryobiology 1984, 21, 521–528. [Google Scholar] [CrossRef]
- Olbricht, C.J.; Cannon, J.K.; Tisher, C.C. Cathepsin B and L in nephron segments of rats with puromycin aminonucleoside nephrosis. Kidney Int. 1987, 32, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Kominami, E.; Tsukahara, T.; Bando, Y.; Katunuma, N. Distribution of cathepsins B and H in rat tissues and peripheral blood cells. J. Biochem. 1985, 98, 87–93. [Google Scholar] [CrossRef]
- Wang, J.; Ohmuraya, M.; Hirota, M.; Baba, H.; Zhao, G.; Takeya, M.; Araki, K.; Yamamura, K. Expression pattern of serine protease inhibitor kazal type 3 (Spink3) during mouse embryonic development. Histochem. Cell Biol. 2008, 130, 387–397. [Google Scholar] [CrossRef]
- Araki, K.; Araki, M.; Yamamura, K. Site-directed integration of the cre gene mediated by Cre recombinase using a combination of mutant lox sites. Nucleic Acids Res. 2002, 30, e103. [Google Scholar] [CrossRef]
- Mehanna, S.; Suzuki, C.; Shibata, M.; Sunabori, T.; Imanaka, T.; Araki, K.; Yamamura, K.; Uchiyama, Y.; Ohmuraya, M. Cathepsin D in pancreatic acinar cells is implicated in cathepsin B and L degradation, but not in autophagic activity. Biochem. Biophys. Res. Commun. 2016, 469, 405–411. [Google Scholar] [CrossRef]
- Basile, D.P.; Anderson, M.D.; Sutton, T.A. Pathophysiology of acute kidney injury. Compr. Physiol. 2012, 2, 1303–1353. [Google Scholar]
- Wei, Q.; Dong, Z. Mouse model of ischemic acute kidney injury: Technical notes and tricks. Am. J. Physiol. Ren. Physiol. 2012, 303, F1487–F1494. [Google Scholar] [CrossRef]
- Hochegger, K.; Koppelstaetter, C.; Tagwerker, A.; Huber, J.M.; Heininger, D.; Mayer, G.; Rosenkranz, A.R. p21 and mTERT are novel markers for determining different ischemic time periods in renal ischemia-reperfusion injury. Am. J. Physiol. Ren. Physiol. 2007, 292, F762–F768. [Google Scholar] [CrossRef]
- Lu, Z.; Dono, K.; Gotoh, K.; Shibata, M.; Koike, M.; Marubashi, S.; Miyamoto, A.; Takeda, Y.; Nagano, H.; Umeshita, K.; et al. Participation of autophagy in the degeneration process of rat hepatocytes after transplantation following prolonged cold preservation. Arch. Histol. Cytol. 2005, 68, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Kominami, E.; Ezaki, J.; Muno, D.; Ishido, K.; Ueno, T.; Wolfe, L.S. Specific storage of subunit c of mitochondrial ATP synthase in lysosomes of neuronal ceroid lipofuscinosis (Batten’s disease). J. Biochem. 1992, 111, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat. Genet. 1999, 21, 70–71. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, C.; Tanida, I.; Ohmuraya, M.; Oliva Trejo, J.A.; Kakuta, S.; Sunabori, T.; Uchiyama, Y. Lack of Cathepsin D in the Renal Proximal Tubular Cells Resulted in Increased Sensitivity against Renal Ischemia/Reperfusion Injury. Int. J. Mol. Sci. 2019, 20, 1711. https://doi.org/10.3390/ijms20071711
Suzuki C, Tanida I, Ohmuraya M, Oliva Trejo JA, Kakuta S, Sunabori T, Uchiyama Y. Lack of Cathepsin D in the Renal Proximal Tubular Cells Resulted in Increased Sensitivity against Renal Ischemia/Reperfusion Injury. International Journal of Molecular Sciences. 2019; 20(7):1711. https://doi.org/10.3390/ijms20071711
Chicago/Turabian StyleSuzuki, Chigure, Isei Tanida, Masaki Ohmuraya, Juan Alejandro Oliva Trejo, Soichiro Kakuta, Takehiko Sunabori, and Yasuo Uchiyama. 2019. "Lack of Cathepsin D in the Renal Proximal Tubular Cells Resulted in Increased Sensitivity against Renal Ischemia/Reperfusion Injury" International Journal of Molecular Sciences 20, no. 7: 1711. https://doi.org/10.3390/ijms20071711
APA StyleSuzuki, C., Tanida, I., Ohmuraya, M., Oliva Trejo, J. A., Kakuta, S., Sunabori, T., & Uchiyama, Y. (2019). Lack of Cathepsin D in the Renal Proximal Tubular Cells Resulted in Increased Sensitivity against Renal Ischemia/Reperfusion Injury. International Journal of Molecular Sciences, 20(7), 1711. https://doi.org/10.3390/ijms20071711



