Adverse Effects of Proton Pump Inhibitors—Evidence and Plausibility
Abstract
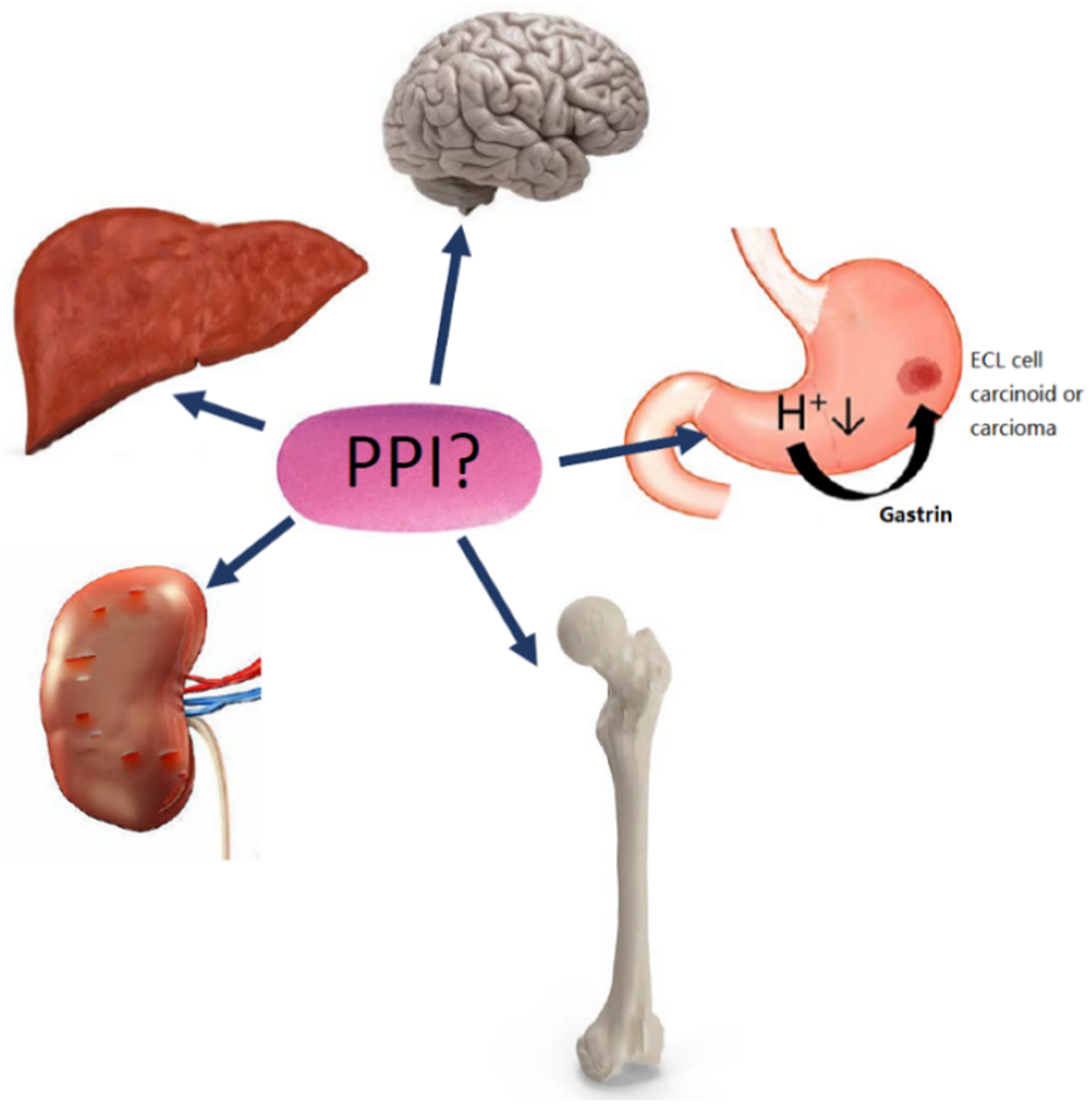
1. Introduction
2. Results and Discussion
2.1. Gastric Neoplasia
2.2. Renal Disease
2.3. Fracture Risk
2.4. Dementia
2.5. Liver Disease
2.6. Micronutrient Deficiency
2.6.1. Vitamin B12
2.6.2. Calcium
2.6.3. Iron
2.6.4. Magnesium
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AIN | Acute interstitial nephritis |
| BMD | Bone mineral density |
| CCKB | Cholecystokinin B |
| CKD | Chronic kidney disease |
| CTX | carboxy-terminal collagen crosslinks |
| ECL | Enterochromaffin-like |
| eGFR | Estimated glomerular filtration rate |
| GERD | Gastroesophageal reflux disease |
| GI | Gastrointestinal |
| H1RA | Histamine 1 receptor antagonist |
| H2RA | Histamine 2 receptor antagonist |
| HE | Hepatic encephalopathy |
| H. pylori | Helicobacter pylori |
| HR | Hazard ratio |
| NET | Neuroendocrine tumour |
| NSAID | Non-steroid anti-inflammatory drug |
| OR | Odds ratio |
| P1NP | procollagen type 1 N-terminal propeptide |
| PCoA | Principal coordinate analysis |
| PPI | Proton pump inhibitor |
| RR | Relative risk |
| SBP | Spontaneous bacterial peritonitis |
References
- Cederberg, C.; Rohss, K.; Lundborg, P.; Olbe, L. Effect of once daily intravenous and oral omeprazole on 24-hour intragastric acidity in healthy subjects. Scand. J. Gastroenterol. 1993, 28, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Wilder-Smith, C.; Halter, F.; Ernst, T.; Gennoni, M.; Zeyen, B.; Varga, L.; Roehmel, J.J.; Merki, H.S. Loss of acid suppression during dosing with H2-receptor antagonists. Aliment. Pharmacol. Ther. 1990, 4 (Suppl. 1), 15–27. [Google Scholar] [PubMed]
- Pounder, R.E.; Sharma, B.K.; Walt, R.P. Twenty-four hour intragastric acidity during treatment with oral omeprazole. Scand. J. Gastroenterol. 1986, 21 (Suppl. 118), 108–117. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, A.H. Phylogeny of the cholecystokinin/gastrin family. Front. Neuroendocrinol. 1998, 19, 73–99. [Google Scholar] [CrossRef] [PubMed]
- De la Coba Ortiz, C.; Arguelles Arias, F.; Martin de Argila de Prados, C.; Judez Gutierrez, J.; Linares Rodriguez, A.; Ortega Alonso, A.; Rodriguez de Santiago, E.; Rodriguez-Tellez, M.; Vera Mendoza, M.I.; Aguilera Castro, L.; et al. Proton-pump inhibitors adverse effects: A review of the evidence and position statement by the Sociedad Espanola de Patologia Digestiva. Rev. Esp. Enferm. Dig. 2016, 108, 207–224. [Google Scholar] [CrossRef]
- Pottegard, A.; Broe, A.; Hallas, J.; de Muckadell, O.B.; Lassen, A.T.; Lodrup, A.B. Use of proton-pump inhibitors among adults: A Danish nationwide drug utilization study. Ther. Adv. Gastroenterol. 2016, 9, 671–678. [Google Scholar] [CrossRef]
- Zink, D.A.; Pohlman, M.; Barnes, M.; Cannon, M.E. Long-term use of acid suppression started inappropriately during hospitalization. Aliment. Pharmacol. Ther. 2005, 21, 1203–1209. [Google Scholar] [CrossRef]
- Eid, S.M.; Boueiz, A.; Paranji, S.; Mativo, C.; Landis, R.; Abougergi, M.S. Patterns and predictors of proton pump inhibitor overuse among academic and non-academic hospitalists. Intern. Med. 2010, 49, 2561–2568. [Google Scholar] [CrossRef]
- Ahrens, D.; Behrens, G.; Himmel, W.; Kochen, M.M.; Chenot, J.F. Appropriateness of proton pump inhibitor recommendations at hospital discharge and continuation in primary care. Int. J. Clin. Pract. 2012, 66, 767–773. [Google Scholar] [CrossRef]
- Prewett, E.J.; Hudson, M.; Nwokolo, C.U.; Sawyerr, A.M.; Pounder, R.E. Nocturnal intragastric acidity during and after a period of dosing with either ranitidine or omeprazole. Gastroenterology 1991, 100, 873–877. [Google Scholar] [CrossRef]
- Jansen, J.B.; Klinkenberg-Knol, E.C.; Meuwissen, S.G.; De Bruijne, J.W.; Festen, H.P.; Snel, P.; Luckers, A.E.; Biemond, I.; Lamers, C.B. Effect of long-term treatment with omeprazole on serum gastrin and serum group A and C pepsinogens in patients with reflux esophagitis. Gastroenterology 1990, 99, 621–628. [Google Scholar] [CrossRef]
- Tang, H.L.; Li, Y.; Hu, Y.F.; Xie, H.G.; Zhai, S.D. Effects of CYP2C19 loss-of-function variants on the eradication of H. pylori infection in patients treated with proton pump inhibitor-based triple therapy regimens: A meta-analysis of randomized clinical trials. PLoS ONE 2013, 8, e62162. [Google Scholar] [CrossRef] [PubMed]
- Klotz, U.; Schwab, M.; Treiber, G. CYP2C19 polymorphism and proton pump inhibitors. Basic Clin. Pharmacol. Toxicol. 2004, 95, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Waldum, H.L.; Brenna, E.; Martinsen, T.C. Safety of proton pump inhibitors. Aliment. Pharmacol. Ther. 2000, 14, 1537–1538. [Google Scholar] [CrossRef] [PubMed]
- Zamcheck, N.; Grable, E.; Ley, A.; Normal, L. Occurrence of gastric cancer among patients with pernicious anemia at the Boston City Hospital. N. Engl. J. Med. 1955, 252, 1103–1110. [Google Scholar] [CrossRef]
- Havu, N. Enterochromaffin-like cell carcinoids of gastric mucosa in rats after life-long inhibition of gastric secretion. Digestion 1986, 35 (Suppl. 1), 42–55. [Google Scholar] [CrossRef]
- Poynter, D.; Selway, S.A.; Papworth, S.A.; Riches, S.R. Changes in the gastric mucosa of the mouse associated with long lasting unsurmountable histamine H2 blockade. Gut 1986, 27, 1338–1346. [Google Scholar] [CrossRef]
- Havu, N.; Mattsson, H.; Ekman, L.; Carlsson, E. Enterochromaffin-like cell carcinoids in the rat gastric mucosa following long-term administration of ranitidine. Digestion 1990, 45, 189–195. [Google Scholar] [CrossRef]
- Mattsson, H.; Havu, N.; Brautigam, J.; Carlsson, K.; Lundell, L.; Carlsson, E. Partial gastric corpectomy results in hypergastrinemia and development of gastric enterochromaffinlike-cell carcinoids in the rat. Gastroenterology 1991, 100, 311–319. [Google Scholar] [CrossRef]
- Fossmark, R.; Martinsen, T.C.; Bakkelund, K.E.; Kawase, S.; Torp, S.H.; Waldum, H.L. Hypergastrinaemia induced by partial corpectomy results in development of enterochromaffin-like cell carcinoma in male Japanese cotton rats. Scand. J. Gastroenterol. 2004, 39, 919–926. [Google Scholar] [CrossRef]
- Spencer, A.J.; Barbolt, T.A.; Henry, D.C.; Eason, C.T.; Sauerschell, R.J.; Bonner, F.W. Gastric morphological changes including carcinoid tumors in animals treated with a potent hypolipidemic agent, ciprofibrate. Toxicol. Pathol. 1989, 17, 7–15. [Google Scholar] [CrossRef]
- Martinsen, T.C.; Nesjan, N.; Ronning, K.; Sandvik, A.K.; Waldum, H.L. The peroxisome-proliferator ciprofibrate induces hypergastrinemia without raising gastric pH. Carcinogenesis 1996, 17, 2153–2155. [Google Scholar] [CrossRef]
- Martinsen, T.C.; Bakke, I.; Chen, D.; Sandvik, A.K.; Zahlsen, K.; Aamo, T.; Waldum, H.L. Ciprofibrate stimulates the gastrin-producing cell by acting luminally on antral PPAR-alpha. Am. J. Physiol. Gastrointest. Liver Physiol. 2005, 289, G1052–G1060. [Google Scholar] [CrossRef]
- Wang, T.C.; Dangler, C.A.; Chen, D.; Goldenring, J.R.; Koh, T.; Raychowdhury, R.; Coffey, R.J.; Ito, S.; Varro, A.; Dockray, G.J.; et al. Synergistic interaction between hypergastrinemia and Helicobacter infection in a mouse model of gastric cancer. Gastroenterology 2000, 118, 36–47. [Google Scholar] [CrossRef]
- Oettele, A.G. Spontaneous carcinoma of the glandular stomach in Rattus (mastomys) natalensis, an African rodent. Br. J. Cancer 1957, 11, 415–433. [Google Scholar] [CrossRef]
- Oettele, A.G. Spontaneous carcinoma of the glandular stomach in a laboratory stock of Rattus (Mastomys) natalensis. S. Afr. J. Med. Sci. 1955, 20, 36. [Google Scholar]
- Schaffer, K.; McBride, E.W.; Beinborn, M.; Kopin, A.S. Interspecies polymorphisms confer constitutive activity to the Mastomys cholecystokinin-B/gastrin receptor. J. Biol. Chem. 1998, 273, 28779–28784. [Google Scholar] [CrossRef]
- Nilsson, O.; Wangberg, B.; Johansson, L.; Theodorsson, E.; Dahlstrom, A.; Modlin, I.M.; Ahlman, H. Rapid induction of enterochromaffinlike cell tumors by histamine2-receptor blockade. Am. J. Pathol. 1993, 142, 1173–1185. [Google Scholar]
- Kidd, M.; Siddique, Z.L.; Drozdov, I.; Gustafsson, B.I.; Camp, R.L.; Black, J.W.; Boyce, M.; Modlin, I.M. The CCK(2) receptor antagonist, YF476, inhibits Mastomys ECL cell hyperplasia and gastric carcinoid tumor development. Regul. Pept. 2010, 162, 52–60. [Google Scholar] [CrossRef]
- Fossmark, R.; Martinsen, T.C.; Bakkelund, K.E.; Kawase, S.; Waldum, H.L. ECL-cell derived gastric cancer in male cotton rats dosed with the H2-blocker loxtidine. Cancer Res. 2004, 64, 3687–3693. [Google Scholar] [CrossRef]
- Martinsen, T.C.; Kawase, S.; Hakanson, R.; Torp, S.H.; Fossmark, R.; Qvigstad, G.; Sandvik, A.K.; Waldum, H.L. Spontaneous ECL cell carcinomas in cotton rats: Natural course and prevention by a gastrin receptor antagonist. Carcinogenesis 2003, 24, 1887–1896. [Google Scholar] [CrossRef]
- Fossmark, R.; Qvigstad, G.; Martinsen, T.C.; Hauso, O.; Waldum, H.L. Animal models to study the role of long-term hypergastrinemia in gastric carcinogenesis. J. Biomed. Biotechnol. 2011, 2011, 975479. [Google Scholar] [CrossRef]
- Waldum, H.L.; Sordal, O.F.; Mjones, P.G. The Enterochromaffin-like [ECL] Cell-Central in Gastric Physiology and Pathology. Int. J. Mol. Sci. 2019, 20, 2444. [Google Scholar] [CrossRef]
- Brenna, E.; Waldum, H.L. Trophic effect of gastrin on the enterochromaffin like cells of the rat stomach: Establishment of a dose response relationship. Gut 1992, 33, 1303–1306. [Google Scholar] [CrossRef]
- Sjoblom, S.M.; Sipponen, P.; Karonen, S.L.; Jarvinen, H.J. Mucosal argyrophil endocrine cells in pernicious anaemia and upper gastrointestinal carcinoid tumours. J. Clin. Pathol. 1989, 42, 371–377. [Google Scholar] [CrossRef]
- Sjöblom, S.M.; Sipponen, P.; Karonen, S.L.; Jarvinen, H.J. Argyrophilic cell hyperplasia and carcinoid tumours in oxyntic mucosa of the stomach. Dependence on duration of pernicious aniaemia. Eur. J. Gastroenterol. Hepatol. 1991, 31, 153–157. [Google Scholar]
- Waldum, H.L.; Fossmark, R. Proton pump inhibitors and gastric cancer: A long expected side effect finally reported also in man. Gut 2017. [Google Scholar] [CrossRef]
- Cavalcoli, F.; Zilli, A.; Conte, D.; Ciafardini, C.; Massironi, S. Gastric neuroendocrine neoplasms and proton pump inhibitors: Fact or coincidence? Scand. J. Gastroenterol. 2015, 50, 1397–1403. [Google Scholar] [CrossRef]
- Nandy, N.; Hanson, J.A.; Strickland, R.G.; McCarthy, D.M. Solitary Gastric Carcinoid Tumor Associated with Long-Term Use of Omeprazole: A Case Report and Review of the Literature. Dig. Dis. Sci. 2015, 61, 708–712. [Google Scholar] [CrossRef]
- Lahner, E.; Pilozzi, E.; Esposito, G.; Galli, G.; Annibale, B. Gastric carcinoid in the absence of atrophic body gastritis and with low Ki67 index: A clinical challenge. Scand. J. Gastroenterol. 2014, 49, 506–510. [Google Scholar] [CrossRef]
- Jianu, C.S.; Fossmark, R.; Viset, T.; Qvigstad, G.; Sordal, O.; Marvik, R.; Waldum, H.L. Gastric carcinoids after long-term use of a proton pump inhibitor. Aliment. Pharmacol. Ther. 2012, 36, 644–649. [Google Scholar] [CrossRef]
- Calvete, O.; Reyes, J.; Zuniga, S.; Paumard-Hernandez, B.; Fernandez, V.; Bujanda, L.; Rodriguez-Pinilla, M.S.; Palacios, J.; Heine-Suner, D.; Banka, S.; et al. Exome sequencing identifies ATP4A gene as responsible of an atypical familial type I gastric neuroendocrine tumour. Hum. Mol. Genet. 2015, 24, 2914–2922. [Google Scholar] [CrossRef]
- Fossmark, R.; Calvete, O.; Mjones, P.; Benitez, J.; Waldum, H.L. ECL-cell carcinoids and carcinoma in patients homozygous for an inactivating mutation in the gastric H K ATPase alpha subunit. APMIS 2016. [Google Scholar] [CrossRef]
- Elsborg, L.; Mosbech, J. Pernicious anaemia as a risk factor in gastric cancer. Acta Med. Scand. 1979, 206, 315–318. [Google Scholar] [CrossRef]
- Sjöblom, S.M.; Sipponen, P.; Miettinen, M.; Karonen, S.L.; Jrvinen, H.J. Gastroscopic screening for gastric carcinoids and carcinoma in pernicious anemia. Endoscopy 1988, 20, 52–56. [Google Scholar] [CrossRef]
- Brusselaers, N.; Wahlin, K.; Engstrand, L.; Lagergren, J. Maintenance therapy with proton pump inhibitors and risk of gastric cancer: A nationwide population-based cohort study in Sweden. BMJ Open 2017, 7, e017739. [Google Scholar] [CrossRef]
- Brusselaers, N.; Lagergren, J.; Engstrand, L. Duration of use of proton pump inhibitors and the risk of gastric and oesophageal cancer. Cancer Epidemiol. 2019, 62, 101585. [Google Scholar] [CrossRef]
- Niikura, R.; Hayakawa, Y.; Hirata, Y.; Yamada, A.; Fujishiro, M.; Koike, K. Long-term proton pump inhibitor use is a risk factor of gastric cancer after treatment for Helicobacter pylori: A retrospective cohort analysis. Gut 2017. [Google Scholar] [CrossRef]
- Cheung, K.S.; Chan, E.W.; Wong, A.Y.S.; Chen, L.; Wong, I.C.K.; Leung, W.K. Long-term proton pump inhibitors and risk of gastric cancer development after treatment for Helicobacter pylori: A population-based study. Gut 2018, 67, 28–35. [Google Scholar] [CrossRef]
- Murphy, G.; Abnet, C.C.; Choo-Wosoba, H.; Vogtmann, E.; Weinstein, S.J.; Taylor, P.R.; Mannisto, S.; Albanes, D.; Dawsey, S.M.; Rehfeld, J.F.; et al. Serum gastrin and cholecystokinin are associated with subsequent development of gastric cancer in a prospective cohort of Finnish smokers. Int. J. Epidemiol. 2017, 46, 914–923. [Google Scholar] [CrossRef]
- Fossmark, R.; Sagatun, L.; Nordrum, I.S.; Sandvik, A.K.; Waldum, H.L. Hypergastrinemia is associated with adenocarcinomas in the gastric corpus and shorter patient survival. APMIS 2015, 123, 509–514. [Google Scholar] [CrossRef]
- Kuipers, E.J.; Lundell, L.; Klinkenberg-Knol, E.C.; Havu, N.; Festen, H.P.; Liedman, B.; Lamers, C.B.; Jansen, J.B.; Dalenback, J.; Snel, P.; et al. Atrophic gastritis and Helicobacter pylori infection in patients with reflux esophagitis treated with omeprazole or fundoplication. N. Engl. J. Med. 1996, 334, 1018–1022. [Google Scholar] [CrossRef]
- Niikura, R.; Hayakawa, Y.; Hirata, Y.; Ogura, K.; Fujishiro, M.; Yamada, A.; Ushiku, T.; Konishi, M.; Fukayama, M.; Koike, K. The Reduction in Gastric Atrophy after Helicobacter pylori Eradication Is Reduced by Treatment with Inhibitors of Gastric Acid Secretion. Int. J. Mol. Sci. 2019, 20, 1913. [Google Scholar] [CrossRef]
- Moayyedi, P.; Eikelboom, J.W.; Bosch, J.; Connolly, S.J.; Dyal, L.; Shestakovska, O.; Leong, D.; Anand, S.S.; Stork, S.; Branch, K.R.H.; et al. Safety of Proton Pump Inhibitors Based on a Large, Multi-year, Randomized Trial of Patients Receiving Rivaroxaban or Aspirin. Gastroenterology 2019, 157, 682–691. [Google Scholar] [CrossRef]
- El-Zimaity, H.M.; Jackson, F.W.; Graham, D.Y. Fundic gland polyps developing during omeprazole therapy. Am. J. Gastroenterol. 1997, 92, 1858–1860. [Google Scholar]
- Jalving, M.; Koornstra, J.J.; Wesseling, J.; Boezen, H.M.; De Jong, S.; Kleibeuker, J.H. Increased risk of fundic gland polyps during long-term proton pump inhibitor therapy. Aliment. Pharmacol. Ther. 2006, 24, 1341–1348. [Google Scholar] [CrossRef]
- Takahari, K.; Haruma, K.; Ohtani, H.; Kiyoto, S.; Watanabe, A.; Kamada, T.; Manabe, N.; Hatano, Y. Proton Pump Inhibitor Induction of Gastric Cobblestone-like Lesions in the Stomach. Intern. Med. 2017, 56, 2699–2703. [Google Scholar] [CrossRef]
- Ruffenach, S.J.; Siskind, M.S.; Lien, Y.H. Acute interstitial nephritis due to omeprazole. Am. J. Med. 1992, 93, 472–473. [Google Scholar] [CrossRef]
- Geevasinga, N.; Coleman, P.L.; Webster, A.C.; Roger, S.D. Proton pump inhibitors and acute interstitial nephritis. Clin. Gastroenterol. Hepatol. 2006, 4, 597–604. [Google Scholar] [CrossRef]
- Moledina, D.G.; Perazella, M.A. PPIs and kidney disease: From AIN to CKD. J. Nephrol. 2016, 29, 611–616. [Google Scholar] [CrossRef]
- Xie, Y.; Bowe, B.; Li, T.; Xian, H.; Balasubramanian, S.; Al-Aly, Z. Proton Pump Inhibitors and Risk of Incident CKD and Progression to ESRD. J. Am. Soc. Nephrol. 2016, 27, 3153–3163. [Google Scholar] [CrossRef]
- Lazarus, B.; Chen, Y.; Wilson, F.P.; Sang, Y.; Chang, A.R.; Coresh, J.; Grams, M.E. Proton Pump Inhibitor Use and the Risk of Chronic Kidney Disease. JAMA Intern. Med. 2016, 176, 238–246. [Google Scholar] [CrossRef]
- Ness-Jensen, E.; Fossmark, R. Adverse Effects of Proton Pump Inhibitors in Chronic Kidney Disease. JAMA Intern. Med. 2016, 176, 868. [Google Scholar] [CrossRef]
- Froissart, M.; Borensztein, P.; Houillier, P.; Leviel, F.; Poggioli, J.; Marty, E.; Bichara, M.; Paillard, M. Plasma membrane Na(+)-H+ antiporter and H(+)-ATPase in the medullary thick ascending limb of rat kidney. Am. J. Physiol. 1992, 262, C963–C970. [Google Scholar] [CrossRef]
- Sachs, G.; Wallmark, B. Biological basis of omeprazole therapy. J. Gastroenterol. Hepatol. 1989, 4 (Suppl. 2), 7–18. [Google Scholar]
- Howden, C.W.; Reid, J.L. Omeprazole, a gastric ‘proton pump inhibitor’: Lack of effect on renal handling of electrolytes and urinary acidification. Eur. J. Clin. Pharmacol. 1984, 26, 639–640. [Google Scholar] [CrossRef]
- Muriithi, A.K.; Leung, N.; Valeri, A.M.; Cornell, L.D.; Sethi, S.; Fidler, M.E.; Nasr, S.H. Biopsy-proven acute interstitial nephritis, 1993-2011: A case series. Am. J. Kidney Dis. 2014, 64, 558–566. [Google Scholar] [CrossRef]
- Yang, Y.X.; Lewis, J.D.; Epstein, S.; Metz, D.C. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA 2006, 296, 2947–2953. [Google Scholar] [CrossRef]
- Targownik, L.E.; Lix, L.M.; Metge, C.J.; Prior, H.J.; Leung, S.; Leslie, W.D. Use of proton pump inhibitors and risk of osteoporosis-related fractures. CMAJ 2008, 179, 319–326. [Google Scholar] [CrossRef]
- Vestergaard, P.; Rejnmark, L.; Mosekilde, L. Proton pump inhibitors, histamine H2 receptor antagonists, and other antacid medications and the risk of fracture. Calcif. Tissue Int. 2006, 79, 76–83. [Google Scholar] [CrossRef]
- Poly, T.N.; Islam, M.M.; Yang, H.C.; Wu, C.C.; Li, Y.J. Proton pump inhibitors and risk of hip fracture: A meta-analysis of observational studies. Osteoporos. Int. 2019, 30, 103–114. [Google Scholar] [CrossRef]
- Liu, J.; Li, X.; Fan, L.; Yang, J.; Wang, J.; Sun, J.; Wang, Z. Proton pump inhibitors therapy and risk of bone diseases: An update meta-analysis. Life Sci. 2019, 218, 213–223. [Google Scholar] [CrossRef]
- Targownik, L.E.; Lix, L.M.; Leung, S.; Leslie, W.D. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology 2010, 138, 896–904. [Google Scholar] [CrossRef]
- Targownik, L.E.; Goertzen, A.L.; Luo, Y.; Leslie, W.D. Long-Term Proton Pump Inhibitor Use Is Not Associated With Changes in Bone Strength and Structure. Am. J. Gastroenterol. 2017, 112, 95–101. [Google Scholar] [CrossRef]
- Thaler, H.W.; Sterke, C.S.; van der Cammen, T.J. Association of Proton Pump Inhibitor Use with Recurrent Falls and Risk of Fractures in Older Women: A Study of Medication Use in Older Fallers. J. Nutr. Health Aging 2016, 20, 77–81. [Google Scholar] [CrossRef]
- Hansen, K.E.; Nieves, J.W.; Nudurupati, S.; Metz, D.C.; Perez, M.C. Dexlansoprazole and Esomeprazole Do Not Affect Bone Homeostasis in Healthy Postmenopausal Women. Gastroenterology 2019, 156, 926–934. [Google Scholar] [CrossRef]
- Cui, G.L.; Syversen, U.; Zhao, C.M.; Chen, D.; Waldum, H.L. Long-term omeprazole treatment suppresses body weight gain and bone mineralization in young male rats. Scand. J. Gastroenterol. 2001, 36, 1011–1015. [Google Scholar]
- Schinke, T.; Schilling, A.F.; Baranowsky, A.; Seitz, S.; Marshall, R.P.; Linn, T.; Blaeker, M.; Huebner, A.K.; Schulz, A.; Simon, R.; et al. Impaired gastric acidification negatively affects calcium homeostasis and bone mass. Nat. Med. 2009, 15, 674–681. [Google Scholar] [CrossRef]
- Krause, M.; Keller, J.; Beil, B.; van Driel, I.; Zustin, J.; Barvencik, F.; Schinke, T.; Amling, M. Calcium gluconate supplementation is effective to balance calcium homeostasis in patients with gastrectomy. Osteoporos. Int. 2014. [Google Scholar] [CrossRef]
- Fossmark, R.; Stunes, A.K.; Petzold, C.; Waldum, H.L.; Rubert, M.; Lian, A.M.; Reseland, J.E.; Syversen, U. Decreased bone mineral density and reduced bone quality in H(+) /K(+) ATPase beta-subunit deficient mice. J. Cell. Biochem. 2012, 113, 141–147. [Google Scholar] [CrossRef]
- Aasarod, K.M.; Ramezanzadehkoldeh, M.; Shabestari, M.; Mosti, M.P.; Stunes, A.K.; Reseland, J.E.; Beisvag, V.; Eriksen, E.F.; Sandvik, A.K.; Erben, R.G.; et al. Skeletal effects of a gastrin receptor antagonist in H+/K+ATPase beta subunit KO mice. J. Endocrinol. 2016, 230, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Goerss, J.B.; Kim, C.H.; Atkinson, E.J.; Eastell, R.; O’Fallon, W.M.; Melton, L.J., 3rd. Risk of fractures in patients with pernicious anemia. J. Bone Miner. Res. 1992, 7, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Merriman, N.A.; Putt, M.E.; Metz, D.C.; Yang, Y.X. Hip fracture risk in patients with a diagnosis of pernicious anemia. Gastroenterology 2010, 138, 1330–1337. [Google Scholar] [CrossRef] [PubMed]
- Aasarod, K.M.; Mosti, M.P.; Stunes, A.K.; Reseland, J.E.; Basso, T.; Syversen, U.; Fossmark, R. Impaired skeletal health in patients with chronic atrophic gastritis. Scand. J. Gastroenterol. 2016, 51, 774–781. [Google Scholar] [CrossRef]
- Recker, R.R. Calcium absorption and achlorhydria. N. Engl. J. Med. 1985, 313, 70–73. [Google Scholar] [CrossRef]
- Hansen, K.E.; Jones, A.N.; Lindstrom, M.J.; Davis, L.A.; Ziegler, T.E.; Penniston, K.L.; Alvig, A.L.; Shafer, M.M. Do proton pump inhibitors decrease calcium absorption? J. Bone Miner. Res. 2010, 25, 2786–2795. [Google Scholar] [CrossRef]
- Wright, M.J.; Sullivan, R.R.; Gaffney-Stomberg, E.; Caseria, D.M.; O’Brien, K.O.; Proctor, D.D.; Simpson, C.A.; Kerstetter, J.E.; Insogna, K.L. Inhibiting gastric acid production does not affect intestinal calcium absorption in young, healthy individuals: A randomized, crossover, controlled clinical trial. J. Bone Miner. Res. 2010, 25, 2205–2211. [Google Scholar] [CrossRef]
- Tuukkanen, J.; Vaananen, H.K. Omeprazole, a specific inhibitor of H+-K+-ATPase, inhibits bone resorption in vitro. Calcif. Tissue Int. 1986, 38, 123–125. [Google Scholar] [CrossRef]
- Waldum, H.L.; Sandvik, A.K.; Brenna, E.; Petersen, H. Gastrin-histamine sequence in the regulation of gastric acid secretion. Gut 1991, 32, 698–701. [Google Scholar] [CrossRef]
- Barete, S.; Assous, N.; de Gennes, C.; Grandpeix, C.; Feger, F.; Palmerini, F.; Dubreuil, P.; Arock, M.; Roux, C.; Launay, J.M.; et al. Systemic mastocytosis and bone involvement in a cohort of 75 patients. Ann. Rheum. Dis. 2010, 69, 1838–1841. [Google Scholar] [CrossRef]
- Fitzpatrick, L.A.; Buzas, E.; Gagne, T.J.; Nagy, A.; Horvath, C.; Ferencz, V.; Mester, A.; Kari, B.; Ruan, M.; Falus, A.; et al. Targeted deletion of histidine decarboxylase gene in mice increases bone formation and protects against ovariectomy-induced bone loss. Proc. Natl. Acad. Sci. USA 2003, 100, 6027–6032. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsen, B.; Vestergaard, P. Proton pump inhibitor use and fracture risk—Effect modification by histamine H1 receptor blockade. Observational case-control study using National Prescription Data. Bone 2013, 57, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Aasarod, K.M.; Stunes, A.K.; Mosti, M.P.; Ramezanzadehkoldeh, M.; Viggaklev, B.I.; Reseland, J.E.; Skallerud, B.H.; Fossmark, R.; Syversen, U. Effects of the Histamine 1 Receptor Antagonist Cetirizine on the Osteoporotic Phenotype in H/K ATPase Beta Subunit KO Mice. J. Cell. Biochem. 2016, 117, 2089–2096. [Google Scholar] [CrossRef] [PubMed]
- Gomm, W.; von Holt, K.; Thome, F.; Broich, K.; Maier, W.; Fink, A.; Doblhammer, G.; Haenisch, B. Association of Proton Pump Inhibitors With Risk of Dementia: A Pharmacoepidemiological Claims Data Analysis. JAMA Neurol. 2016, 73, 410–416. [Google Scholar] [CrossRef]
- Haenisch, B.; von Holt, K.; Wiese, B.; Prokein, J.; Lange, C.; Ernst, A.; Brettschneider, C.; Konig, H.H.; Werle, J.; Weyerer, S.; et al. Risk of dementia in elderly patients with the use of proton pump inhibitors. Eur. Arch. Psychiatry Clin. Neurosci. 2015, 265, 419–428. [Google Scholar] [CrossRef]
- Welu, J.; Metzger, J.; Bebensee, S.; Ahrendt, A.; Vasek, M. Proton Pump Inhibitor Use and Risk of Dementia in the Veteran Population. Fed. Pract. 2019, 36, S27–S31. [Google Scholar]
- Badiola, N.; Alcalde, V.; Pujol, A.; Munter, L.M.; Multhaup, G.; Lleo, A.; Coma, M.; Soler-Lopez, M.; Aloy, P. The proton-pump inhibitor lansoprazole enhances amyloid beta production. PLoS ONE 2013, 8, e58837. [Google Scholar] [CrossRef]
- Bloom, G.S. Amyloid-beta and tau: The trigger and bullet in Alzheimer disease pathogenesis. JAMA Neurol. 2014, 71, 505–508. [Google Scholar] [CrossRef]
- Yin, R.H.; Tan, L.; Jiang, T.; Yu, J.T. Prion-like Mechanisms in Alzheimer’s Disease. Curr. Alzheimer Res. 2014, 11, 755–764. [Google Scholar] [CrossRef]
- Martinsen, T.C.; Taylor, D.M.; Johnsen, R.; Waldum, H.L. Gastric acidity protects mice against prion infection? Scand. J. Gastroenterol. 2002, 37, 497–500. [Google Scholar] [CrossRef]
- Martinsen, T.C.; Benestad, S.L.; Moldal, T.; Waldum, H.L. Inhibitors of gastric acid secretion increase the risk of prion infection in mice. Scand. J. Gastroenterol. 2011, 46, 1418–1422. [Google Scholar] [CrossRef] [PubMed]
- Park, S.K.; Nam, J.H.; Lee, H.; Chung, H.; Lee, E.K.; Shin, J.Y. Beyond uncertainty: Negative findings for the association between the use of proton pump inhibitors and risk of dementia. J. Gastroenterol. Hepatol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Taipale, H.; Tolppanen, A.M.; Tiihonen, M.; Tanskanen, A.; Tiihonen, J.; Hartikainen, S. No Association Between Proton Pump Inhibitor Use and Risk of Alzheimer’s Disease. Am. J. Gastroenterol. 2017, 112, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.F.; Chen, M.H.; Wang, Y.P.; Chu, C.J.; Huang, Y.H.; Lin, H.C.; Hou, M.C.; Lee, F.Y.; Su, T.P.; Lu, C.L. Proton Pump Inhibitors Increase Risk for Hepatic Encephalopathy in Patients With Cirrhosis in A Population Study. Gastroenterology 2017, 152, 134–141. [Google Scholar] [CrossRef]
- Fasullo, M.; Rau, P.; Liu, D.Q.; Holzwanger, E.; Mathew, J.P.; Guilarte-Walker, Y.; Szabo, G. Proton pump inhibitors increase the severity of hepatic encephalopathy in cirrhotic patients. World J. Hepatol. 2019, 11, 522–530. [Google Scholar] [CrossRef]
- Ma, Y.J.; Cao, Z.X.; Li, Y.; Feng, S.Y. Proton pump inhibitor use increases hepatic encephalopathy risk: A systematic review and meta-analysis. World J. Gastroenterol. 2019, 25, 2675–2682. [Google Scholar] [CrossRef]
- Tantai, X.X.; Yang, L.B.; Wei, Z.C.; Xiao, C.L.; Chen, L.R.; Wang, J.H.; Liu, N. Association of proton pump inhibitors with risk of hepatic encephalopathy in advanced liver disease: A meta-analysis. World J. Gastroenterol. 2019, 25, 2683–2698. [Google Scholar] [CrossRef]
- Ghosh, G.; Jesudian, A.B. Small Intestinal Bacterial Overgrowth in Patients With Cirrhosis. J. Clin. Exp. Hepatol. 2019, 9, 257–267. [Google Scholar] [CrossRef]
- Jackson, M.A.; Goodrich, J.K.; Maxan, M.E.; Freedberg, D.E.; Abrams, J.A.; Poole, A.C.; Sutter, J.L.; Welter, D.; Ley, R.E.; Bell, J.T.; et al. Proton pump inhibitors alter the composition of the gut microbiota. Gut 2016, 65, 749–756. [Google Scholar] [CrossRef]
- Clooney, A.G.; Bernstein, C.N.; Leslie, W.D.; Vagianos, K.; Sargent, M.; Laserna-Mendieta, E.J.; Claesson, M.J.; Targownik, L.E. A comparison of the gut microbiome between long-term users and non-users of proton pump inhibitors. Aliment. Pharmacol. Ther. 2016, 43, 974–984. [Google Scholar] [CrossRef]
- Min, Y.W.; Lim, K.S.; Min, B.H.; Gwak, G.Y.; Paik, Y.H.; Choi, M.S.; Lee, J.H.; Kim, J.J.; Koh, K.C.; Paik, S.W.; et al. Proton pump inhibitor use significantly increases the risk of spontaneous bacterial peritonitis in 1965 patients with cirrhosis and ascites: A propensity score matched cohort study. Aliment. Pharmacol. Ther. 2014, 40, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, J.S.; Zadvornova, Y.; Heuman, D.M.; Hafeezullah, M.; Hoffmann, R.G.; Sanyal, A.J.; Saeian, K. Association of proton pump inhibitor therapy with spontaneous bacterial peritonitis in cirrhotic patients with ascites. Am. J. Gastroenterol. 2009, 104, 1130–1134. [Google Scholar] [CrossRef]
- Llorente, C.; Jepsen, P.; Inamine, T.; Wang, L.; Bluemel, S.; Wang, H.J.; Loomba, R.; Bajaj, J.S.; Schubert, M.L.; Sikaroodi, M.; et al. Gastric acid suppression promotes alcoholic liver disease by inducing overgrowth of intestinal Enterococcus. Nat. Commun. 2017, 8, 837. [Google Scholar] [CrossRef]
- Horvath, A.; Rainer, F.; Bashir, M.; Leber, B.; Schmerboeck, B.; Klymiuk, I.; Groselj-Strele, A.; Durdevic, M.; Freedberg, D.E.; Abrams, J.A.; et al. Biomarkers for oralization during long-term proton pump inhibitor therapy predict survival in cirrhosis. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tran, K.T.; McMenamin, U.C.; Hicks, B.; Murchie, P.; Thrift, A.P.; Coleman, H.G.; Iversen, L.; Johnston, B.T.; Lee, A.J.; Cardwell, C.R. Proton pump inhibitor and histamine-2 receptor antagonist use and risk of liver cancer in two population-based studies. Aliment. Pharmacol. Ther. 2018, 48, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Thorens, J.; Froehlich, F.; Schwizer, W.; Saraga, E.; Bille, J.; Gyr, K.; Duroux, P.; Nicolet, M.; Pignatelli, B.; Blum, A.L.; et al. Bacterial overgrowth during treatment with omeprazole compared with cimetidine: A prospective randomised double blind study. Gut 1996, 39, 54–59. [Google Scholar] [CrossRef]
- Llorente, C.; Schnabl, B. The gut microbiota and liver disease. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 275–284. [Google Scholar] [CrossRef]
- Kummen, M.; Hov, J.R. The gut microbial influence on cholestatic liver disease. Liver Int. 2019, 39, 1186–1196. [Google Scholar] [CrossRef]
- Schenk, B.E.; Festen, H.P.; Kuipers, E.J.; Klinkenberg-Knol, E.C.; Meuwissen, S.G. Effect of short- and long-term treatment with omeprazole on the absorption and serum levels of cobalamin. Aliment. Pharmacol. Ther. 1996, 10, 541–545. [Google Scholar] [CrossRef]
- Lam, J.R.; Schneider, J.L.; Zhao, W.; Corley, D.A. Proton pump inhibitor and histamine 2 receptor antagonist use and vitamin B12 deficiency. JAMA 2013, 310, 2435–2442. [Google Scholar] [CrossRef]
- Den Elzen, W.P.; Groeneveld, Y.; de Ruijter, W.; Souverijn, J.H.; le Cessie, S.; Assendelft, W.J.; Gussekloo, J. Long-term use of proton pump inhibitors and vitamin B12 status in elderly individuals. Aliment. Pharmacol. Ther. 2008, 27, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Attwood, S.E.; Ell, C.; Galmiche, J.P.; Fiocca, R.; Hatlebakk, J.G.; Hasselgren, B.; Langstrom, G.; Jahreskog, M.; Eklund, S.; Lind, T.; et al. Long-term safety of proton pump inhibitor therapy assessed under controlled, randomised clinical trial conditions: Data from the SOPRAN and LOTUS studies. Aliment. Pharmacol. Ther. 2015, 41, 1162–1174. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, M.B.; Madden, D.M.; Murray, A.M.; Heaney, R.P.; Kerzner, L.J. Effects of proton pump inhibitors on calcium carbonate absorption in women: A randomized crossover trial. Am. J. Med. 2005, 118, 778–781. [Google Scholar] [CrossRef]
- Serfaty-Lacrosniere, C.; Wood, R.J.; Voytko, D.; Saltzman, J.R.; Pedrosa, M.; Sepe, T.E.; Russell, R.R. Hypochlorhydria from short-term omeprazole treatment does not inhibit intestinal absorption of calcium, phosphorus, magnesium or zinc from food in humans. J. Am. Coll. Nutr. 1995, 14, 364–368. [Google Scholar] [CrossRef]
- Ajmera, A.V.; Shastri, G.S.; Gajera, M.J.; Judge, T.A. Suboptimal response to ferrous sulfate in iron-deficient patients taking omeprazole. Am. J. Ther. 2012, 19, 185–189. [Google Scholar] [CrossRef]
- Van Aerts, R.M.; van Deursen, C.T.; Koek, G.H. Proton Pump Inhibitors Reduce the Frequency of Phlebotomy in Patients With Hereditary Hemochromatosis. Clin. Gastroenterol. Hepatol. 2016, 14, 147–152. [Google Scholar] [CrossRef]
- Hutchinson, C.; Geissler, C.A.; Powell, J.J.; Bomford, A. Proton pump inhibitors suppress absorption of dietary non-haem iron in hereditary haemochromatosis. Gut 2007, 56, 1291–1295. [Google Scholar] [CrossRef] [PubMed]
- Cavalcoli, F.; Zilli, A.; Conte, D.; Massironi, S. Micronutrient deficiencies in patients with chronic atrophic autoimmune gastritis: A review. World J. Gastroenterol. 2017, 23, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.R.; Schneider, J.L.; Quesenberry, C.P.; Corley, D.A. Proton Pump Inhibitor and Histamine-2 Receptor Antagonist Use and Iron Deficiency. Gastroenterology 2017, 152, 821–829. [Google Scholar] [CrossRef]
- Epstein, M.; McGrath, S.; Law, F. Proton-pump inhibitors and hypomagnesemic hypoparathyroidism. N. Engl. J. Med. 2006, 355, 1834–1836. [Google Scholar] [CrossRef]
- Danziger, J.; William, J.H.; Scott, D.J.; Lee, J.; Lehman, L.W.; Mark, R.G.; Howell, M.D.; Celi, L.A.; Mukamal, K.J. Proton-pump inhibitor use is associated with low serum magnesium concentrations. Kidney Int. 2013, 83, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Gan, L.; Mei, Z. Does the use of proton pump inhibitors increase the risk of hypomagnesemia: An updated systematic review and meta-analysis. Medicine 2019, 98, e15011. [Google Scholar] [CrossRef] [PubMed]
- Cheungpasitporn, W.; Thongprayoon, C.; Kittanamongkolchai, W.; Srivali, N.; Edmonds, P.J.; Ungprasert, P.; O’Corragain, O.A.; Korpaisarn, S.; Erickson, S.B. Proton pump inhibitors linked to hypomagnesemia: A systematic review and meta-analysis of observational studies. Ren. Fail. 2015, 37, 1237–1241. [Google Scholar] [CrossRef] [PubMed]
- Hess, M.W.; Hoenderop, J.G.; Bindels, R.J.; Drenth, J.P. Systematic review: Hypomagnesaemia induced by proton pump inhibition. Aliment. Pharmacol. Ther. 2012, 36, 405–413. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fossmark, R.; Martinsen, T.C.; Waldum, H.L. Adverse Effects of Proton Pump Inhibitors—Evidence and Plausibility. Int. J. Mol. Sci. 2019, 20, 5203. https://doi.org/10.3390/ijms20205203
Fossmark R, Martinsen TC, Waldum HL. Adverse Effects of Proton Pump Inhibitors—Evidence and Plausibility. International Journal of Molecular Sciences. 2019; 20(20):5203. https://doi.org/10.3390/ijms20205203
Chicago/Turabian StyleFossmark, Reidar, Tom C. Martinsen, and Helge L. Waldum. 2019. "Adverse Effects of Proton Pump Inhibitors—Evidence and Plausibility" International Journal of Molecular Sciences 20, no. 20: 5203. https://doi.org/10.3390/ijms20205203
APA StyleFossmark, R., Martinsen, T. C., & Waldum, H. L. (2019). Adverse Effects of Proton Pump Inhibitors—Evidence and Plausibility. International Journal of Molecular Sciences, 20(20), 5203. https://doi.org/10.3390/ijms20205203





