Silencing of Odorant-Binding Protein Gene OBP3 Using RNA Interference Reduced Virus Transmission of Tomato Chlorosis Virus
Abstract
1. Introduction
2. Results
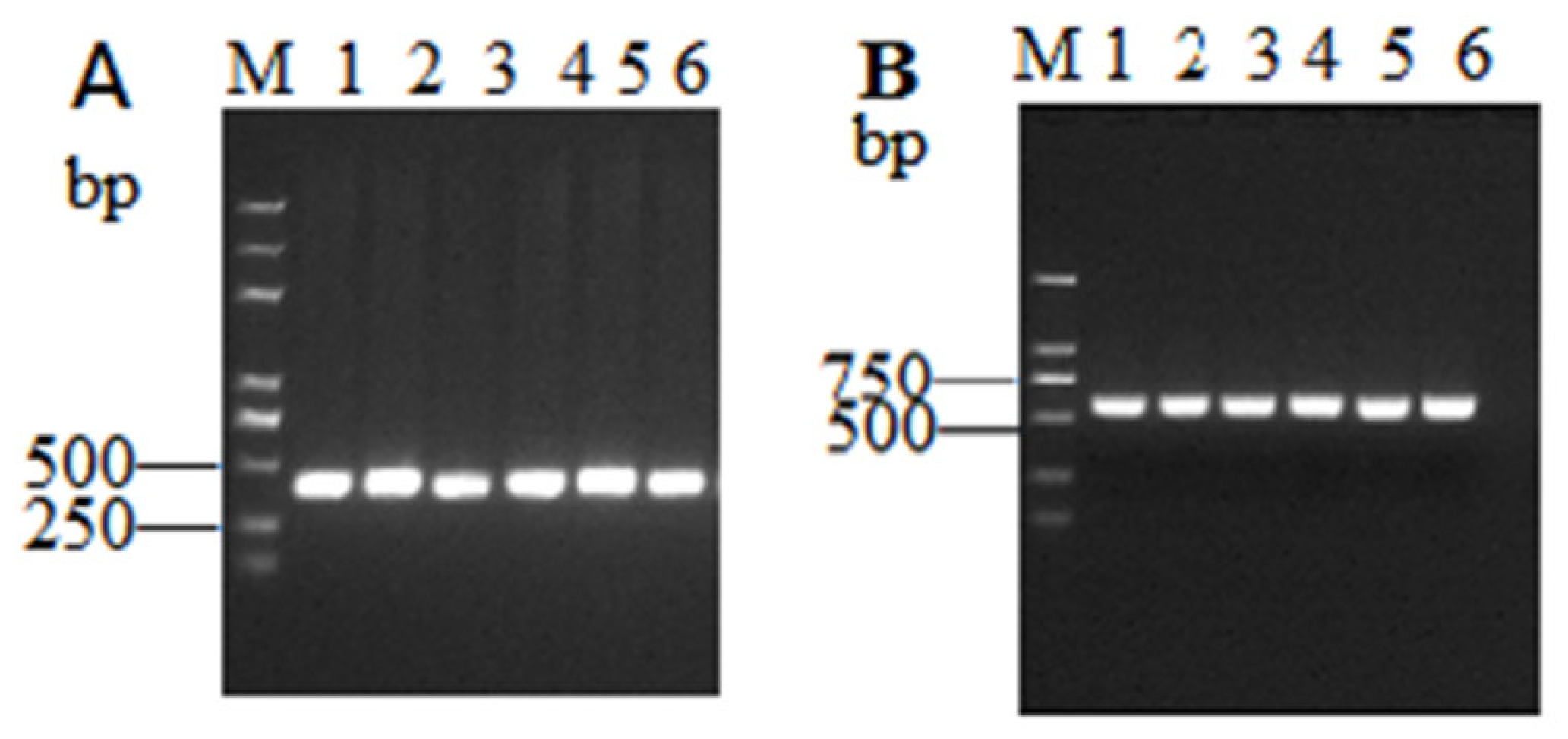
2.1. Detection of ToCV
2.2. Analysis of OBPs Expression in B. tabaci MED
2.3. dsRNA Synthesis
2.4. Expression of OBPs in B. tabaci MED in Response to dsRNA Treatment
2.5. Preference of MED on ToCV-infected vs Healthy Tomato Plants
2.6. ToCV Transmission Rate
3. Discussion
4. Materials and Methods
4.1. Plants and ToCV
4.2. B. tabaci MED Population
4.3. Analysis of OBPs Expression in B. tabaci MED
4.4. dsRNA Synthesis
4.5. Expression of OBPs in B. tabaci MED in Response to dsRNA Treatment
4.6. Preference of MED on ToCV-Infected vs Healthy Tomato Plants
4.7. ToCV Transmission Rate
4.8. Data Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Wisler, G.C.; Li, R.H.; Liu, H.Y.; Lowry, D.S.; Duffus, J.E. Tomato chlorosis virus: A new whitefly-transmitted, phloem-limited, bipartite closterovirus of tomato. Phytopathology 1998, 88, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.B.; Tang, X.; Zhang, X.; Zhang, D.Y.; Li, F.; Yan, F.; Zhang, Y.J.; Zhou, X.G.; Liu, Y. Transmission efficiency, preference and behavior of Bemisia tabaci MEAM1 and MED under the influence of Tomato chlorosis virus. Front. Plant Sci. 2017, 8, 2271. [Google Scholar] [CrossRef] [PubMed]
- Wintermantel, W.M.; Wisler, G.C. Vector specificity, host range, and genetic diversity of Tomato chlorosis virus. Plant Dis. 2006, 90, 814–819. [Google Scholar] [CrossRef]
- Elena, G.; Jesús, N.C.; Enrique, M. Resistance to Tomato chlorosis virus in wild tomato species that impair virus accumulation and disease symptom expression. Plant Dis. 2010, 100, 582–592. [Google Scholar]
- Wei, K.K.; Li, J.; Ding, T.B.; Chu, D. Research progress on distribution, identification method of Tomato chlorosis virus (ToCV)and whitefly transmission characteristics. Chin. Vegetables 2018, 19–24. [Google Scholar]
- Velasco, L.; Simón, B.; Janssen, D.; Cenis, J.L. Incidences and progression of Tomato chlorosis virus disease and Tomato yellow leaf curl virus disease in tomato under different greenhouse covers in southeast Spain. Ann. Appl. Biol. 2008, 153, 335–344. [Google Scholar] [CrossRef]
- Orfanidou, C.G.; Pappi, P.G.; Efthimiou, K.E.; Katis, N.I.; Maliogka, V.I. Transmission of Tomato chlorosis virus (ToCV) by Bemisia tabaci biotype Q and evaluation of four weed species as viral sources. Plant Dis. 2016, 100, 2043–2049. [Google Scholar] [CrossRef]
- Rao, Q.; Luo, C.; Zhang, H.; Guo, X.; Devine, G.J. Distribution and dynamics of Bemisia tabaci invasive biotypes in central China. Bull. Entomol. Res. 2011, 101, 81–88. [Google Scholar] [CrossRef]
- Pan, H.P.; Chu, D.; Ge, D.Q.; Wang, S.L.; Wu, Q.J.; Xie, W.; Jiao, X.G.; Liu, B.M.; Yang, X.; Yang, N.N.; et al. Further spread of and domination by Bemisia tabaci (Hemiptera: Aleyrodidae) biotype Q on field crops in China. J. Econ. Entomol. 2011, 104, 978–985. [Google Scholar] [CrossRef]
- Czosnek, H.; Ghanim, M. Back to basics: Are begomoviruses whitefly pathogens. J. Integr. Agr. 2012, 11, 225–234. [Google Scholar] [CrossRef]
- Davis, T.S.; Horton, D.R.; Munyaneza, J.E.; Landolt, P.J. Experimental infection of plants with an herbivore-associated bacterial endosymbiont influences herbivore host selection behavior. PLoS ONE 2012, 7, e49330. [Google Scholar] [CrossRef] [PubMed]
- McMenemy, L.S.; Hartley, S.E.; MacFarlane, S.A.; Karley, A.J.; Shepherd, T.; Johnson, S.N. Raspberry viruses manipulate the behaviour of their insect vectors. Entomol. Exp. Appl. 2012, 144, 56–68. [Google Scholar] [CrossRef]
- Fereres, A.; Penaflor, M.F.; Favaro, C.F.; Azevedo, K.E.; Landi, C.H.; Maluta, N.K.; Bento, J.M.; Lopes, J.R. Tomato infection by whitefly-transmitted circulative and non-circulative viruses induce contrasting changes in plant volatiles and vector behaviour. Viruses 2016, 8, 225. [Google Scholar] [CrossRef] [PubMed]
- Pichersky, E.; Gershenzon, J. The formation and function of plant volatiles: Perfumes for pollinator attraction and defense. Curr. Opin. Plant Bio. 2002, 5, 237–243. [Google Scholar] [CrossRef]
- Pichersky, E. Biosynthesis of plant volatiles: nature’s diversity and ingenuity. Science 2006, 311, 808–811. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, X.M.; Li, F.Q.; Wu, F.; Li, H.L.; Luo, C. Cloning and prokaryotic expression of odorant binding protein OBP8 in MED cryptic species Bemisia tabaci and the binding characteristics with plant volatiles. J.Plant Prot. 2016, 43, 32–39. [Google Scholar]
- Ko, H.J.; Park, T.H. Enhancement of odorant detection sensitivity by the expression of odorant-binding protein. Biosens. Bioelectron. 2008, 23, 1017–1023. [Google Scholar] [CrossRef]
- Swanson, J.A.I.; Torto, B.; Kells, S.A.; Mesce, K.A.; Tumlinson, J.H.; Spivak, M. Odorants that induce hygienic behavior in honeybees: Identification of volatile compounds in chalkbrood-infected honeybee larvae. J. Chem. Ecol. 2009, 35, 1108–1116. [Google Scholar] [CrossRef]
- Payne, E.B.T.L.; Birch, M.C.; Kennedy, A.C.E.J. Mechanisms in insect olfaction. Mech. Insect Olf. 1986, 201–208. [Google Scholar]
- Swarup, S.; Morozova, T.V.; Sridhar, S.; Nokes, M.; Anholt, R.R.H. Modulation of feeding behavior by odorant-binding proteins in Drosophila melanogaster. Chem. Senses 2014, 39, 125–132. [Google Scholar] [CrossRef]
- Campanini, E.B.; Brito, R.A.D. Molecular evolution of odorant-binding proteins gene family in two closely related Anastrepha fruit flies. BMC Evol. Biol. 2016, 16, 198. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, H.L.; Zhou, Y.X.; Zhao, L.; Zhang, L.Y.; Ni, C.X.; Shang, H.W. cDNA cloning, tissue expression and ligand binding characteristics of odorant-binding protein 2 from the oriental fruit fly, Bactrocera dorsalis (Diptera: Tephritidae). Acta Entomol. Sinica 2013, 56, 612–621. [Google Scholar]
- Guo, W.; Ren, D.; Zhao, L.F.; Jiang, F.; Song, J.; Wang, X.H.; Kang, L. Identification of odorant-binding proteins (OBPs) and functional analysis of phase-related OBPs in the migratory locust. Fron. Physiol. 2018, 9, 984. [Google Scholar] [CrossRef] [PubMed]
- Sun, X. Host location and functional analysis of odorant binding protein in Cnaphalocrocis medinalis. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2015. [Google Scholar]
- Dicke, M. Plants talk, but are they deaf? Trends Plant Sci. 2003, 8, 403–405. [Google Scholar] [CrossRef]
- Dorokhov, Y.L.; Komarova, T.V.; Sheshukova, E.V. Chapter 13 - Volatile organic compounds and plant virus–host interaction. Plant Virus–host Interaction 2014, 5, 241–262. [Google Scholar]
- Penuelas, J.; Llusia, J. Plant VOC emissions: Making use of the unavoidable. Trends Ecol. Evol. 2004, 19, 402–404. [Google Scholar] [CrossRef] [PubMed]
- Bruce, T.J.; Wadhams, L.J.; Woodcock, C.M. Insect host location: A volatile situation. Trends Plant Sci. 2005, 10, 269–274. [Google Scholar] [CrossRef]
- Filella, I.; Bosch, J.; Llusia, J.; Seco, R.; Penuelas, J. The role of frass and cocoon volatiles in host location by Monodontomerus aeneus, a parasitoid of Megachilid solitary bees. Environ. Entomol. 2011, 40, 126–131. [Google Scholar] [CrossRef]
- Pelosi, P.; Iovinella, I.; Felicioli, A.; Dani, F.R. Soluble proteins of chemical communication: An overview across arthropods. Front. Physiol. 2014, 5, 320. [Google Scholar] [CrossRef]
- Wang, L.F.; Hu, K.; He, H.L.; Ding, W.B.; Li, Y.Z. Southern rice black-streaked dwarf virus-induced volatiles from rice plants and behavioral responses of adult Sogatella furcifera(Hemiptera:Delphacidae) to the components of these volatiles. Acta Entomol. Sinica 2017, 60, 412–420. [Google Scholar]
- Shi, X.B.; Chen, G.; Pan, H.P.; Xie, W.; Wu, Q.; Wang, S.L.; Liu, Y.; Zhou, X.G.; Zhang, Y.J. Plants pre-infested with viruliferous MED/Q cryptic species promotes subsequent Bemisia tabaci infestation. Front. Microbiol. 2018, 9, 1404. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, S.; Michalakis, Y.; Munster, M.; Blanc, S. Plant feeding by insect vectors can affect life cycle, population genetics and evolution of plant viruses. Funct. Ecol. 2013, 27, 610–622. [Google Scholar] [CrossRef]
- Mauck, K.E.; De Moraes, C.M.; Mescher, M.C. Deceptive chemical signals induced by a plant virus attract insect vectors to inferior hosts. P. Natl. Acad. Sci. USA 2010, 107, 3600–3605. [Google Scholar] [CrossRef] [PubMed]
- JiménezMartínez, E.S.; BosquePérez, N.A.; Berger, P.H.; Zemetra, R.S. Life history of the bird cherry-oat aphid, Rhopalosiphum padi (Homoptera: Aphididae), on transgenic and untransformed wheat challenged with Barley yellow dwarf virus. J. Econ. Entomol. 2004, 97, 203–212. [Google Scholar] [CrossRef]
- Zhou, J.; Kan, Y.; Antoniw, J.; Pickett, J.; Field, L. Genome and EST analyses and expression of a gene family with putative functions in insect chemoreception. Chem. Senses 2006, 31, 453–465. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.; Guidolin, A.; Syed, Z.; Cornel, A.; Leal, W. Knockdown of a mosquito odorant-binding protein involved in the sensitive detection of oviposition attractants. J. Chem. Ecol. 2010, 36, 245–248. [Google Scholar] [CrossRef]
- Rebijith, K.; Asokan, R.; Hande, H.; Kumar, N.; Krishna, V.; Vinutha, J.; Bakthavatsalam, N. Interference of odorant-binding protein 2 (OBP2) of the cotton aphid, Aphis gossypii (Glover), resulted in altered electrophysiological responses. Appl. Biochem. Biotech. 2016, 178, 251–266. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Zhu, X.Q.; Gu, S.H.; Zhou, Y.L.; Wang, S.Y.; Zhang, Y.J.; Guo, Y.Y. Silencing of odorant binding protein gene AlinOBP4 by RNAi induces declining electrophysiological responses of Adelphocoris lineolatus to six semiochemicals. Insect Sci. 2017, 24, 789–797. [Google Scholar] [CrossRef]
- Wang, R.; Li, F.; Zhang, W.; Zhang, X.; Qu, C.; Tetreau, G.; Sun, L.; Luo, C.; Zhou, J. Identification and expression profile analysis of odorant binding protein and chemosensory protein genes in Bemisia tabaci MED by head transcriptome. PLoS ONE 2017, 12, e0171739. [Google Scholar] [CrossRef]
- Yang, X.; Xie, W.; Li, R.M.; Zhou, X.M.; Wang, S.L.; Wu, Q.J.; Yang, N.N.; Xia, J.X.; Yang, Z.Z.; Guo, L.T.; et al. RNA interference-mediated knockdown of the hydroxyacid-oxoacid transhydrogenase gene decreases thiamethoxam resistance in adults of the whitefly Bemisia tabaci. Sci. Rep. 2017, 7, 41201. [Google Scholar] [CrossRef]
- Upadhyay, S.K.; Chandrashekar, K.; Thakur, N.; Verma, P.C.; Borgio, J.F.; Singh, P.K.; Tuli, R. RNA interference for the control of whiteflies (Bemisia tabaci) by oral route. J. Biosciences 2011, 36, 153–161. [Google Scholar] [CrossRef] [PubMed]







| Primer Name | Primer Sequence (5′–3′) | Purpose |
|---|---|---|
| ToCV-6 | AAACTGCCTGCATGAAAAGTCTC | ToCV RT-PCR |
| ToCV-5 | GGTTTGGATTTTGGTACTACATTCAGT | |
| tocvhsp70-1 | TGTCGAAAGTACCGCCACC | ToCV RT-PCR |
| tocvhsp70-2 | GCTTCCGAAACTCCGTCTTG | |
| Cl-J-2195 | TTGATTTTTTGGTCATCCAGAAGT | Bemisia tabaci biotype |
| R-BQ-2819 | CTGAATATCGRCGAGGCATTCC |
| Primer Name | Primer Sequence (5′–3′) |
|---|---|
| BtabOBP1-F BtabOBP1-R | AAGTGCTTGACGGATTATTAC GCATCATATTATCGCAGTGT |
| BtabOBP2-F BtabOBP2-R | CTCTTATTGGTCTATTTCTCGTT CTTCTTCTTCTGGCATTGG |
| BtabOBP3-F BtabOBP3-R | CTATCTCGGTTCAGTTCCA TGTCTTTCCACTCGCTAT |
| BtabOBP4-F BtabOBP4-R | GTTTCTTGGAGTGCGTTTA TCATCATCATCAGCCTCTT |
| BtabOBP5-F BtabOBP5-R | AAGTAAAGGCTGTGGATGA CGAGTAATAGTTGTTGTCTTGA |
| BtabOBP6-F BtabOBP6-R | GTAGCAATACAGGTGGAGA ATGACACTCTTGACATTAGC |
| BtabOBP7-F BtabOBP7-R | TCGAATCAGATGCAGAGGGTG TATCCGGGGGACTCATTCCA |
| BtabOBP8-F BtabOBP8-R | TGATGGCGTGTCTTATGA CTGAGGTTGAGTGCTGTA |
| BtabActin-F BtabActin-R | TCTTCCAGCCATCCTTCTTG CGGTGATTTCCTTCTGCATT |
| BtabEF-1α-F BtabEF-1α-R | TAGCCTTGTGCCAATTTCCG CCTTCAGCATTACCGTCC |
| Primer Name | Sequence (5′–3′) |
|---|---|
| dsOBP3-R | ATTCTCTAGAAGCTTAATACGACTCACTATAGGGATTGAACCAGCCAAGCTCCC |
| dsOBP3-F | ATTCTCTAGAAGCTTAATACGACTCACTATAGGGATGATGGATCTCAAAGCTATTTTGCTC |
| F-086 | TAATACGACTCACTATAGGGTTCAGTGGAGAGGGTGAAGGT |
| R-612 | TAATACGACTCACTATAGGGTGTGTGGACAGGTAATGGTTG |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, X.-B.; Wang, X.-Z.; Zhang, D.-Y.; Zhang, Z.-H.; Zhang, Z.; Cheng, J.; Zheng, L.-M.; Zhou, X.-G.; Tan, X.-Q.; Liu, Y. Silencing of Odorant-Binding Protein Gene OBP3 Using RNA Interference Reduced Virus Transmission of Tomato Chlorosis Virus. Int. J. Mol. Sci. 2019, 20, 4969. https://doi.org/10.3390/ijms20204969
Shi X-B, Wang X-Z, Zhang D-Y, Zhang Z-H, Zhang Z, Cheng J, Zheng L-M, Zhou X-G, Tan X-Q, Liu Y. Silencing of Odorant-Binding Protein Gene OBP3 Using RNA Interference Reduced Virus Transmission of Tomato Chlorosis Virus. International Journal of Molecular Sciences. 2019; 20(20):4969. https://doi.org/10.3390/ijms20204969
Chicago/Turabian StyleShi, Xiao-Bin, Xue-Zhong Wang, De-Yong Zhang, Zhan-Hong Zhang, Zhuo Zhang, Ju’E Cheng, Li-Min Zheng, Xu-Guo Zhou, Xin-Qiu Tan, and Yong Liu. 2019. "Silencing of Odorant-Binding Protein Gene OBP3 Using RNA Interference Reduced Virus Transmission of Tomato Chlorosis Virus" International Journal of Molecular Sciences 20, no. 20: 4969. https://doi.org/10.3390/ijms20204969
APA StyleShi, X.-B., Wang, X.-Z., Zhang, D.-Y., Zhang, Z.-H., Zhang, Z., Cheng, J., Zheng, L.-M., Zhou, X.-G., Tan, X.-Q., & Liu, Y. (2019). Silencing of Odorant-Binding Protein Gene OBP3 Using RNA Interference Reduced Virus Transmission of Tomato Chlorosis Virus. International Journal of Molecular Sciences, 20(20), 4969. https://doi.org/10.3390/ijms20204969








