Antibiofilm Activity of Polyamide 11 Modified with Thermally Stable Polymeric Biocide Polyhexamethylene Guanidine 2-Naphtalenesulfonate
Abstract
1. Introduction
2. Results and Discussion
2.1. FTIR Analysis of PA-11/PHMG-NS Films
2.2. Surface Properties of PA-11/PHMG-NS Films
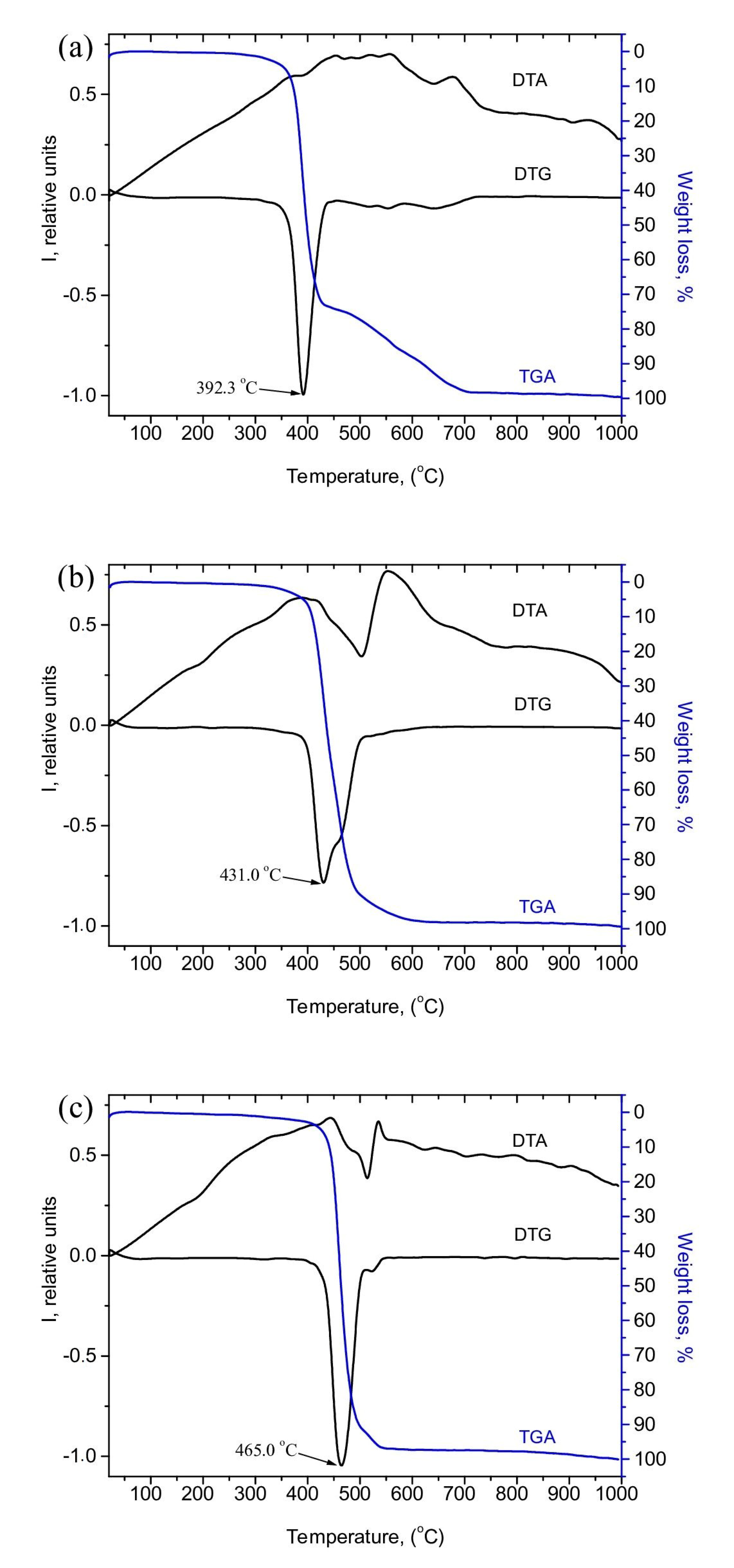
2.3. Mechanical and Thermal Properties of PA-11/PHMG-NS Composites
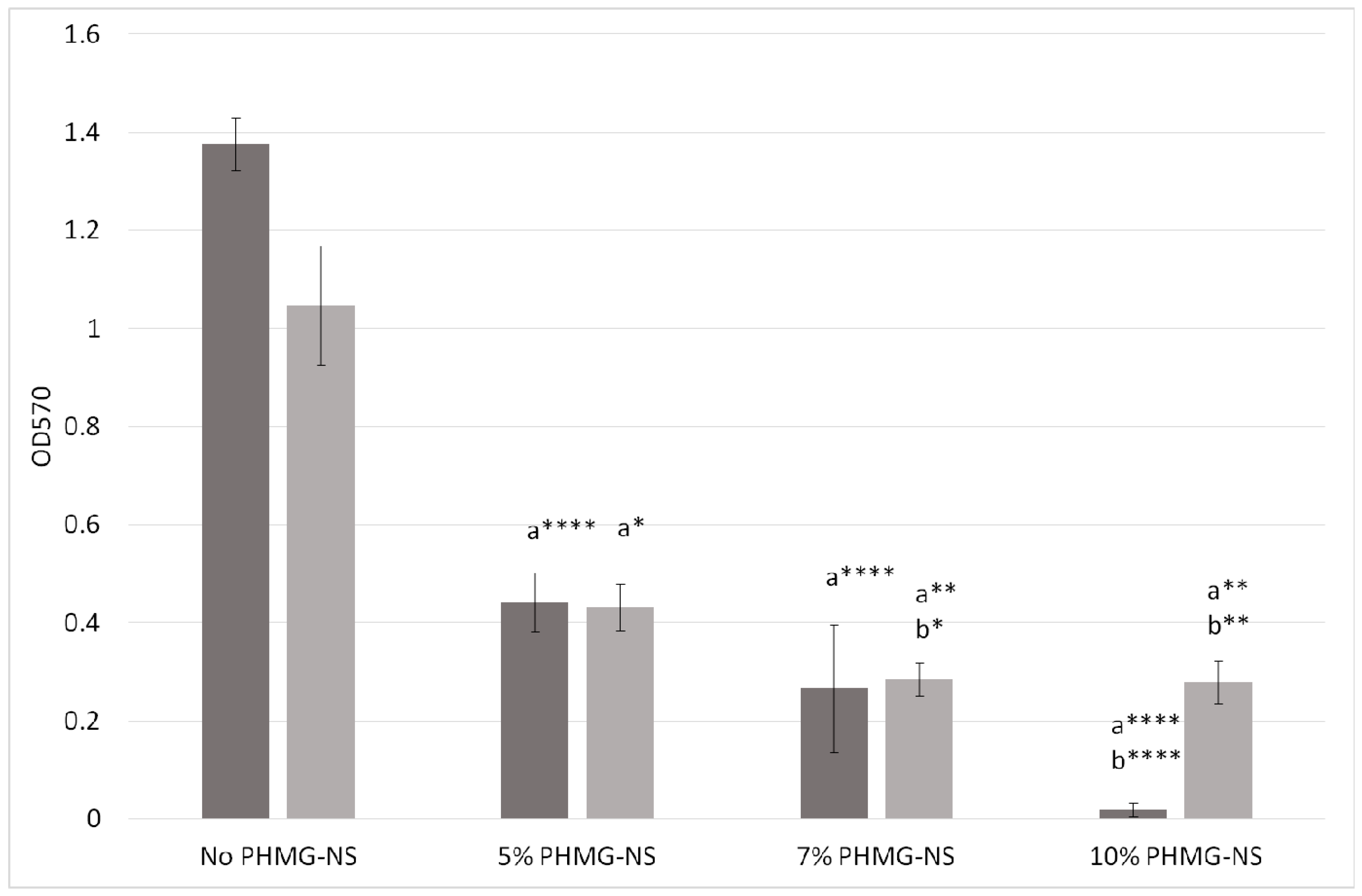
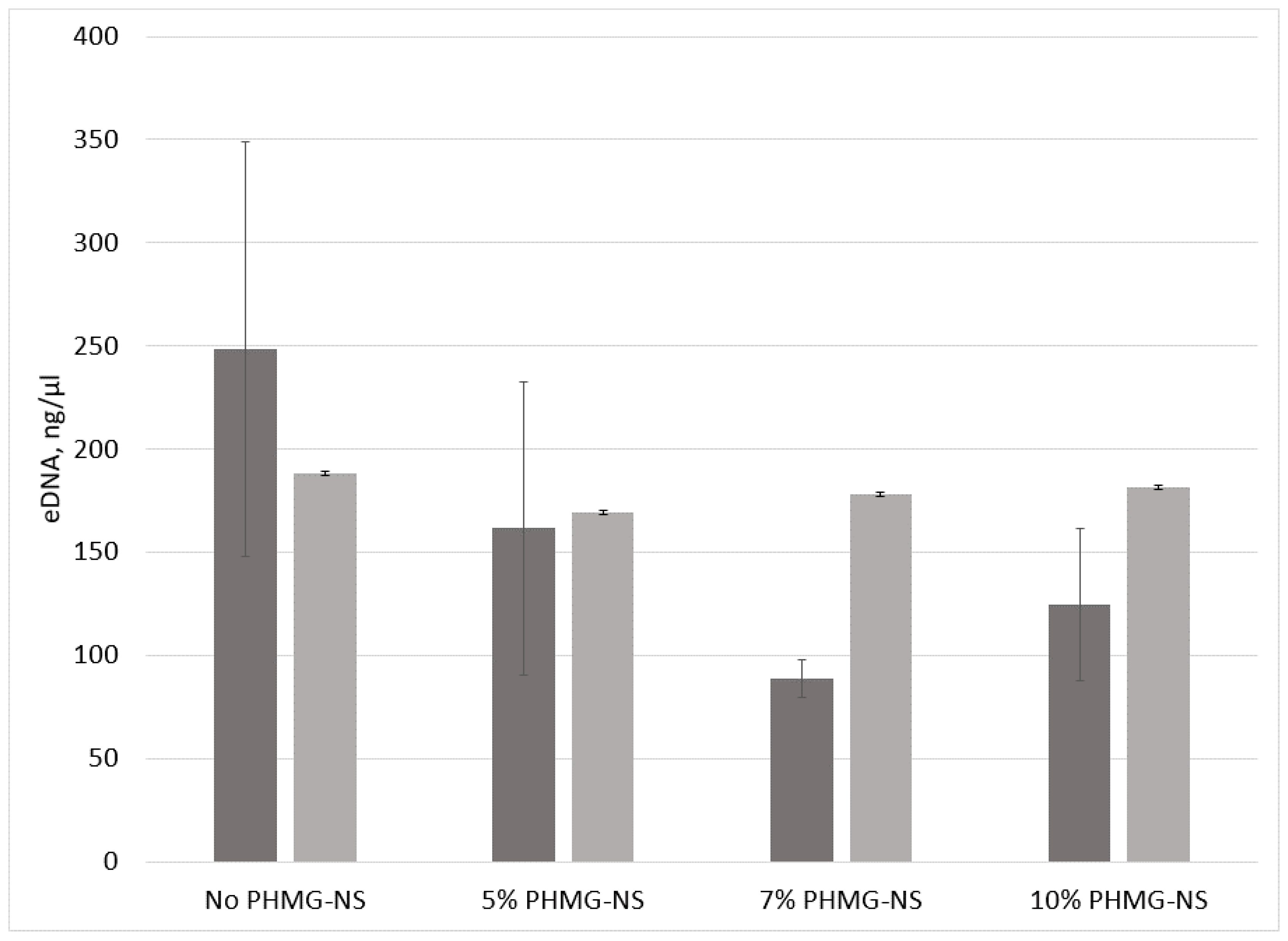
2.4. Antibiofilm Efficacy of PA-11/PHMG-NS Films
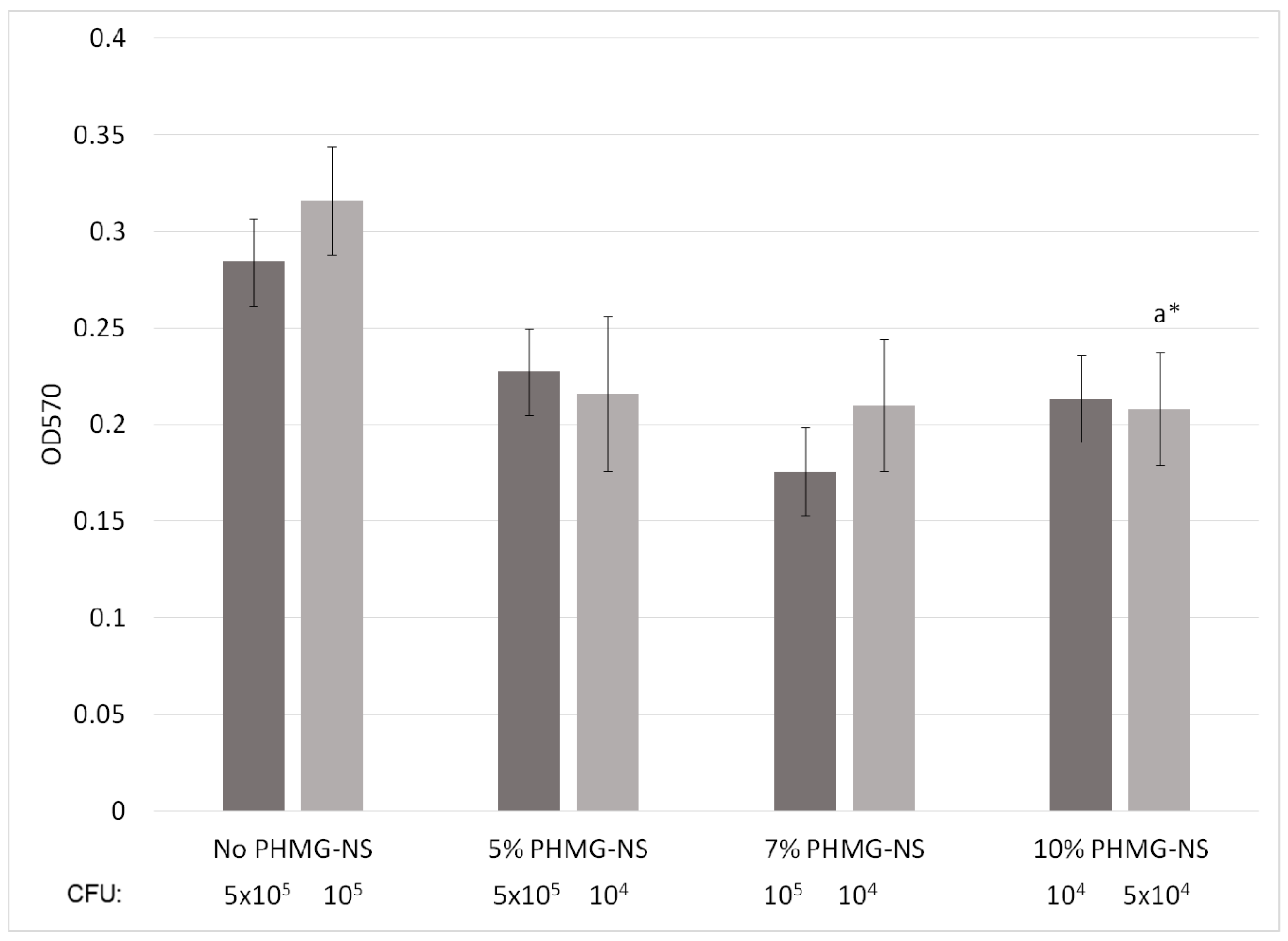
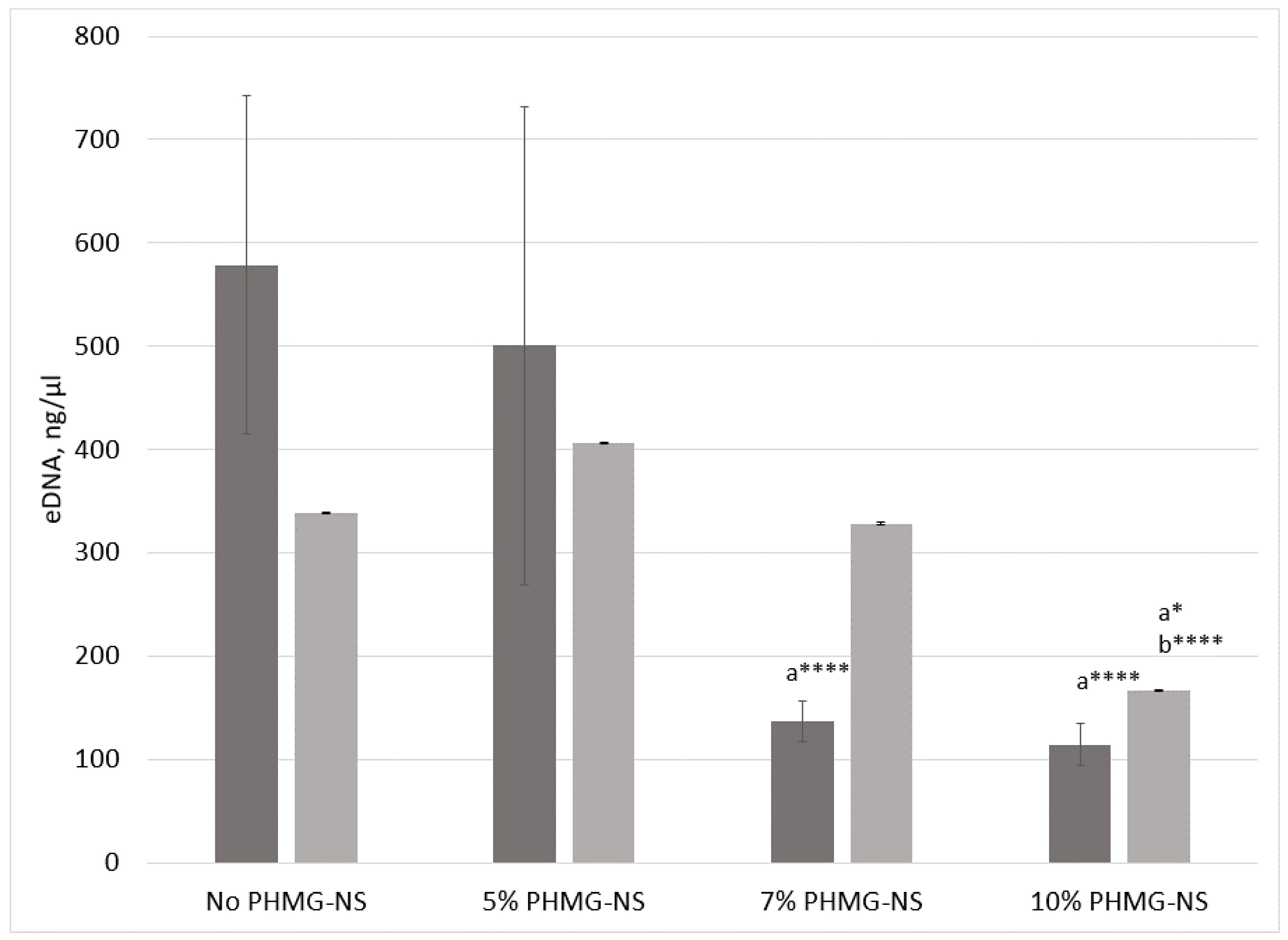
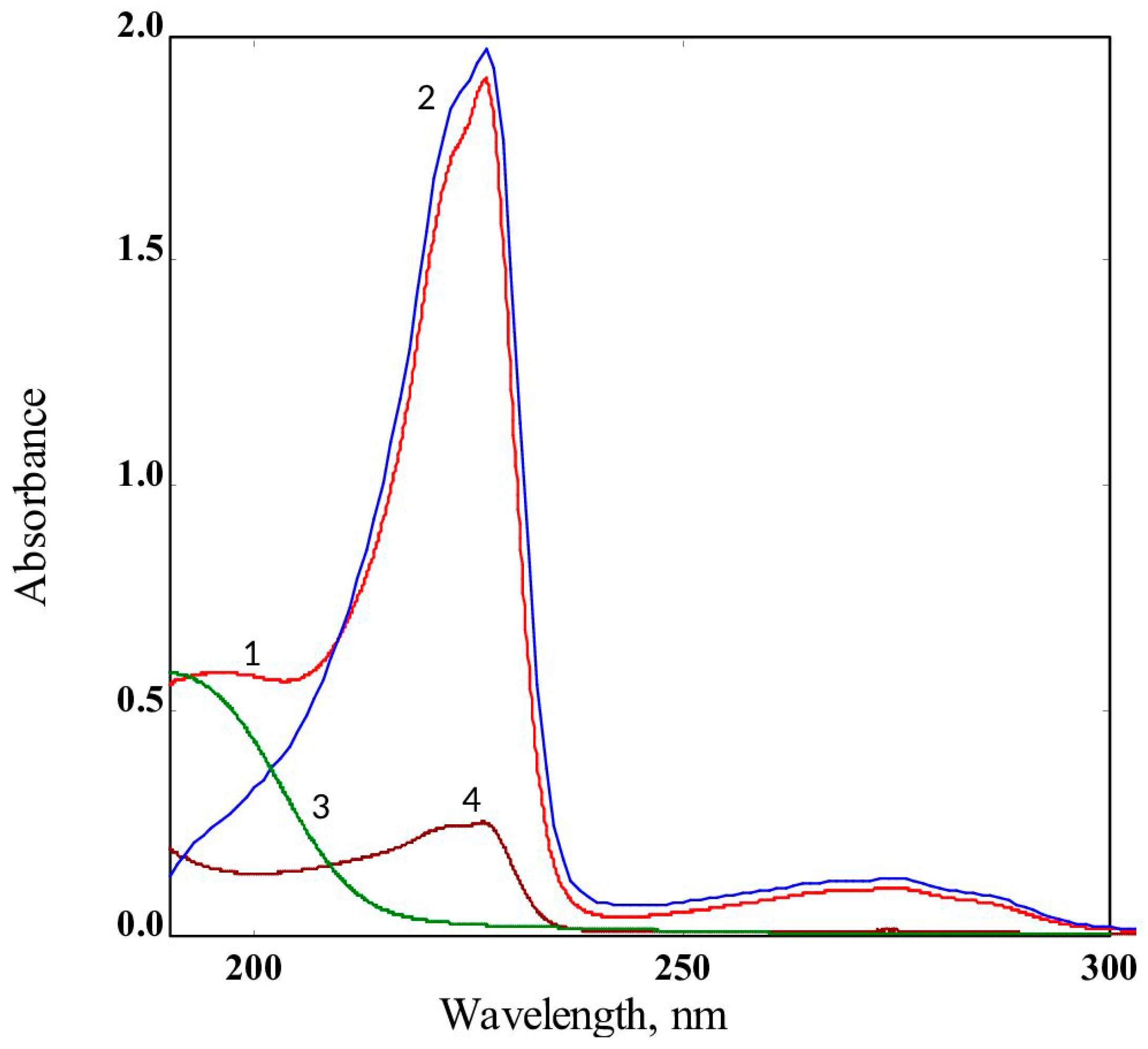
2.5. Leaching Resistance of PHMG-NS from PA-11 Films
3. Materials and Methods
3.1. Materials
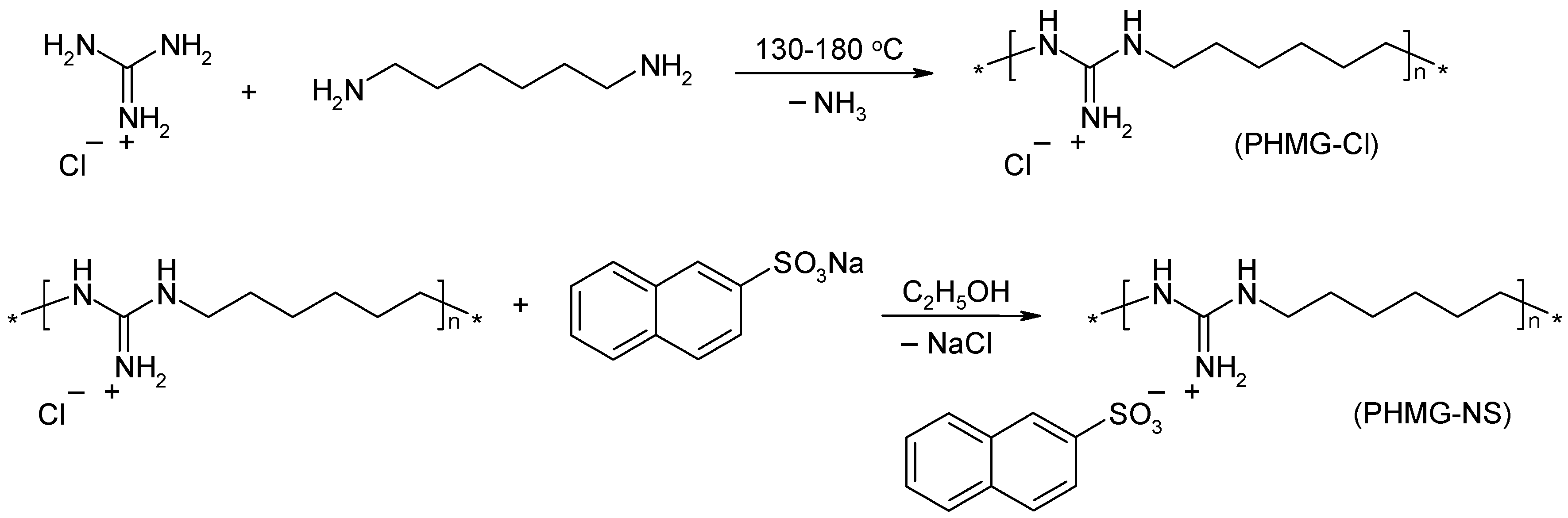
3.2. Synthesis of Polymeric Biocide PHMG-NS
3.3. Preparation of PA-11/PHMG-NS Composite Films
3.4. Characterisation of PA-11/PHMG-NS Composite
3.5. Biocide Release
3.6. Biofilm Assay
3.7. Bacteria Toxicity Assay
3.8. eDNA Assay
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| PA-11 | Polyamide 11 |
| PHMG-Cl | Polyhexamethylene guanidine hydrochloride |
| PHMG-NS | Polyhexamethylene guanidine 2-naphtalenesulfonate |
| LB | Luria Broth |
| MTT | Methylthiazolyldiphenyltetrazolium bromide |
| eDNA | Extracellular DNA |
| TE buffer | Tris-EDTA buffer |
| CFU | colony forming units |
References
- RILSAN® PA11: Created from a Renewable Source (Product Data Sheet); Arkema: Puteaux, France, 2005.
- Fine Powders: A Durable Coating for Durable Products (Product Data Sheet); Arkema: Colombes, France, 2010.
- Klun, U.; Friedrich, Z.; Kržan, A. Polyamide 6 fibre degradation by a lignolytic fungus. Polym. Degrad. Stab. 2003, 79, 99–104. [Google Scholar] [CrossRef]
- Tomita, K.; Ikeda, N.; Ueno, A. Isolation and characterization of a thermophilic bacterium, Geobacillus thermocatenulatus, degrading nylon 12 and nylon 66. Biotechnol. Lett. 2003, 25, 1743–1746. [Google Scholar] [CrossRef] [PubMed]
- Chonde Sonal, G.; Chonde Sachin, G.; Raut, P.D. Studies on Degradation of synthetic polymer Nylon 6 and Nylon 6, 6 by Pseudomonas aeruginosa NCIM 2242. IJETCAS 2013, 4, 362–369. [Google Scholar] [CrossRef]
- Kaali, P.; Strömberg, E.; Karlsson, S. Prevention of biofilm assotiated infections and degradation of polymeric materials used in biomedical applications. In Biomedical Engineering, Trends in Material Science; Laskovski, A.N., Ed.; In Tech: Rijeka, Slovenia, 2011; Chapter 22; pp. 513–541. [Google Scholar]
- Moshynets, O.V.; Spiers, A.J. Viewing biofilms within the larger context of bacterial aggregations. In Microbial Biofilms—Importance and Applications; Dhanasekaran, D., Thajuddin, N., Eds.; InTech: Rijeka, Croatia, 2016; pp. 3–22. ISBN 978-953-2436-8. [Google Scholar]
- Koza, A.; Kusmierska, A.; McLauglin, K.; Moshynets, O.; Spiers, A.J. Adaptive radiation of P. fluorescens SBW25 in experimental microcosms provides an understanding of the evolutionary ecology and molecular biology of A-L interface biofilm-formation. FEMS Microbiol. Lett. 2017, 364. [Google Scholar] [CrossRef]
- McLaughlin, K.; Folorunso, A.O.; Deeni, Y.Y.; Foster, D.; Gorbatiuk, O.; Hapca, S.M.; Immoor, C.; Koza, A.; Mohammed, I.U.; Moshynets, O.; et al. Biofilm formation and cellulose expression by Bordetella avium 197N, the causative agent of bordetellosis in birds and an opportunistic respiratory pathogen in humans. Res. Microbiol. 2017, 168, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival mechanisms of clinically relevant microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef] [PubMed]
- Nichols, D. Biocides in Plastics. Rapra Review Reports; Rapra Technology: Shrewsbury, UK, 2005; Volume 15, Report 180. [Google Scholar]
- Rabin, N.; Zheng, Y.; Opoku-Temeng, C.; Du, Y.; Bonsu, E.; Sintim, H.O. Agents that inhibits bacterial biofilm formation. Future Med. Chem. 2015, 7, 647–671. [Google Scholar] [CrossRef] [PubMed]
- Vitiello, G.; Pezzella, A.; Zanfardino, A.; Varcamonti, M.; Silvestri, B.; Costantini, A.; Branda, F.; Luciani, G. Titania as driving agent for DHICA polymerization: A novel strategy for the design of bioinspired antimicrobial nanomaterials. J. Mater. Chem. B 2015, 3, 2808–2815. [Google Scholar] [CrossRef]
- Vitiello, G.; Pezzella, A.; Zanfardino, A.; Varcamonti, M.; Silvestri, B.; Giudicianni, P.; Costantini, A.; Varcamonti, M.; Branda, F.; Luciani, G. Antimicrobial activity of eumelanin-based hybrids: The role of TiO2 in modulating the structure and biological performance. Mater. Sci. Eng. C 2017, 75, 454–462. [Google Scholar] [CrossRef]
- Vitiello, G.; Silvestri, B.; Luciani, G. Learning from nature: Bioinspired strategies towards antimicrobial nanostructured systems. Curr. Top. Med. Chem. 2018, 18, 22–41. [Google Scholar] [CrossRef] [PubMed]
- Kuratsuji, T.; Shimizu, H. Polyamide Based Antibacterial Powder Paint Composition. U.S. Patent 20030171452, 11 September 2003. [Google Scholar]
- Lapeyre, A.; Ganset, C. Polyamide-Based Powder and Its Use for Obtaining an Antibacterial Coating. U.S. Patent 8303970, 4 August 2005. [Google Scholar]
- Williams, R.L.; Doherty, P.J.; Vince, D.J.; Grashoff, G.J.; Williams, D.F. The biocompatibility of silver. Crit. Rev. Biocompat. 1989, 5, 221–223. [Google Scholar]
- Kumar, R.; Münstedt, H. Silver ion release from antimicrobial polyamide/silver composites. Biomaterials 2005, 26, 2081–2088. [Google Scholar] [CrossRef] [PubMed]
- Damm, C.; Münstedt, H.; Rösch, A. Long-term antimicrobial polyamide 6/silver-nanocomposites. J. Mater. Sci. 2007, 42, 6067–6073. [Google Scholar] [CrossRef]
- Damm, C.; Münstedt, H.; Rösch, A. The antimicrobial efficacy of polyamide 6/silver-nano- and microcomposites. Mater. Chem. Phys. 2008, 108, 61–66. [Google Scholar] [CrossRef]
- Thokala, N.; Kealey, C.; Kennedy, J.; Brady, D.B.; Farrell, J.B. Characterization of polyamide 11/copper antimicrobial composites for medical device applications. Mater. Sci. Eng. C 2017, 78, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P.; Moore, L.E. Cationic antiseptics: Diversity of action under a common epithet. J. Appl. Microbiol. 2005, 99, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Ribeiro, A.M.; de Melo Carrasco, L.D. Cationic antimicrobial polymers and their assemblies. Int. J. Mol. Sci. 2013, 14, 9906–9946. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Jiang, J.M.; Chen, Y.M. Synthesis and antimicrobial activity of polymeric guanidine and biguanidine salts. Polymer 1999, 40, 6189–6198. [Google Scholar] [CrossRef]
- Oulè, M.K.; Azinwi, R.; Bernier, A.M.; Kablan, T.; Maupertuis, A.M.; Mauler, S.; Koffi- Nevry, R.; Dembèlè, K.; Forbes, L.; Diop, L. Polyhexamethylene guanidine hydrochloride-based disinfectant: A novel tool to fight meticillin-resistant Staphylococcus aureus and nosocomial infections. J. Med. Microbiol. 2008, 57, 1523–1528. [Google Scholar] [CrossRef]
- Qian, L.; Guan, Y.; He, B.; Xiao, H. Modified guanidine polymers: Synthesis and antimicrobial mechanism revealed by AFM. Polymer 2008, 49, 2471–2475. [Google Scholar] [CrossRef]
- Zhou, Z.; Wei, D.; Guan, Y.; Zheng, A.; Zhong, J.-J. Damage of Escherichia coli membrane by bactericidal agent polyhexamethylene guanidine hydrochloride: Micrographic evidences. J. Appl. Microbiol. 2010, 108, 898–907. [Google Scholar] [CrossRef]
- Zhou, Z.; Wei, D.; Guan, Y.; Zheng, A.; Zhong, J.-J. Extensive in vitro activity of guanidine hydrochloride polymer analogs against antibiotics-resistant clinically isolated strains. Mater. Sci. Eng. 2011, 31, 1836–1843. [Google Scholar] [CrossRef]
- Zhou, Z.; Zheng, A.; Zhong, J. Interactions of biocidal guanidine hydrochloride polymer analogs with model membranes: A comparative biophysical study. Acta Biochim. Biophys. Sin. 2011, 43, 729–737. [Google Scholar] [CrossRef]
- Choi, H.; Kim, K.-J.; Lee, D.J. Antifungal activity of the cationic antimicrobial polymer-polyhexamethylene guanidine hydrochloride and its mode of action. Fungal Biol. 2017, 121, 53–60. [Google Scholar] [CrossRef]
- Han, J.-S.; Lim, K.-M.; Park, S.-J.; Song, W.-S. Polyhexamethyleneguanidine Phosphate Powder, Method of Making the Same and Antimicrobial Resin Containing the Same. Eur. Patent 1 110 948, 17 May 2001. [Google Scholar]
- Protasov, A.; Bardeau, J.-F.; Morozovskaya, I.; Boretska, M.; Cherniavska, T.; Petrus, L.; Tarasyuk, O.; Metelytsia, L.; Kopernyk, I.; Kalashnikova, L.; et al. New promising antifouling agent based on polymeric biocide polyhexamethylene guanidine molybdate. J. Environ. Toxicol. 2016, 36, 2543–2551. [Google Scholar] [CrossRef]
- Swiontek Brzezinska, M.; Walczak, M.; Jankiewizs, U.; Pejchalová, M. Antimicrobial activity of polyhexamethylene guanidine derivatives introduced into polycaprolactone. J. Polym. Environ. 2018, 26, 589–595. [Google Scholar] [CrossRef]
- Walczak, M.; Richert, A.; Burkowska-But, A. The effect of polyhexamethylene guanidine hydrochloride (PHMG) derivatives introduced into polylactide (PLA) on the activity of bacterial enzymes. J. Ind. Microbiol. Biotechnol. 2014, 41, 1719–1724. [Google Scholar] [CrossRef]
- Rogalskyy, S.; Bardeau, J.-F.; Tarasyuk, O.; Fatyeyeva, K. Fabrication of new antifungal polyamide-12 material. Polym. Int. 2012, 61, 686–691. [Google Scholar] [CrossRef]
- Rogalsky, S.; Bardeau, J.-F.; Wu, H.; Lyoshina, L.; Bulko, O.; Tarasyuk, O.; Makhno, S.; Cherniavska, T.; Kyselov, Y.; Koo, J.H. Structural, thermal and antibacterial properties of polyamide 11/polymeric biocide polyhexamethylene guanidine dodecylbenzenesulfonate composites. J. Mater. Sci. 2016, 51, 7716–7730. [Google Scholar] [CrossRef]
- Ghamrawi, S.; Bouchara, J.-P.; Tarasyuk, O.; Rogalsky, S.; Lyoshina, L.; Bulko, O.; Bardeau, J.-F. Promising silicones modified with cationic biocides for the development of antimicrobial medical devices. Mater. Sci. Eng. C 2017, 75, 969–979. [Google Scholar] [CrossRef]
- Nigmatullin, R.; Gao, F.; Konovalova, V. Permanent, non-leaching antimicrobial polyamide nanocomposites based on organoclays modified with a cationic polymer. Macromol. Mater. Eng. 2009, 294, 795–805. [Google Scholar] [CrossRef]
- Li, G.; Shen, J. A study of pyridinium-type functional polymers. IV. Behavioral features of the antibacterial activity of insoluble pyridinium-type polymers. J. Appl. Polym. Sci. 2000, 78, 676–684. [Google Scholar] [CrossRef]
- Desai, D.G.; Liao, K.S.; Cevallos, M.E.; Trautner, B.W. Silver or nitrofurazone impregnation of urinary catheters has a minimal effect on uropathogen adherence. J. Urol. 2010, 184, 2565–2571. [Google Scholar] [CrossRef]
- Ghatak, P.D.; Mathew-Steiner, S.S.; Pandey, P.; Roy, S.; Se, C.K. A surfactant polymer dressing potentiates antimicrobial efficacy in biofilm disruption. Sci. Rep. 2018, 8, 873. [Google Scholar] [CrossRef]
- Shao, Q.; Fan, Y.; Yang, L.; Gao, Y.Q. Counterion effects on the denaturing activity of guanidinium cation to protein. J. Chem. Theory Comput. 2012, 8, 4364–4373. [Google Scholar] [CrossRef]
- Jansen, B.; Peters, G. Modern strategies in the prevention of polymer-associated infections. J. Hosp. Infect. 1991, 19, 83–88. [Google Scholar] [CrossRef]
- An, Y.H.; Friedman, R.J. Concise review of mechanisms of bacterial adhesion to biomaterial surfaces. J. Biomed. Mater. Res. 1998, 43, 338–348. [Google Scholar] [CrossRef]
- Kügler, R.; Bouloussa, O.; Rondelez, F. Evidence of a charge-density threshold for optimum efficiency of biocidal cationic surfaces. Microbiology 2005, 151, 1341–1348. [Google Scholar] [CrossRef]
- Reisner, A.; Haagensen, J.A.J.; Schembri, M.A.; Zechner, E.L.; Molin, S. Development and maturation of Escherichia coli K-12 biofilms. Mol. Microbiol. 2003, 48, 933–946. [Google Scholar] [CrossRef]
- Avila-Novoa, M.G.; Iniguez-Moreno, M.; Solis-Velazquez, O.A.; Gonzalez-Gomez, J.P.; Guerrero-Medina, P.J.; Gutierrez-Lomeli, M. Biofilm formation by Staphyloccus aureus isolated from food contact surface in the dairy industry of Jalisco, Mexico. J. Food Qual. 2018. [Google Scholar] [CrossRef]
- Zhou, Z.; Wei, D.; Lu, Y. Polyhexamethylene guanidine hydrochloride bactericidal advantages over chlorhexidine bigluconate against ESKAPE bacteria. Biotechnol. Appl. Biochem. 2014. [Google Scholar] [CrossRef]
- Moshynets, O.; Koza, A.; Dello Sterpaio, P.; Kordium, V.; Spiers, A.J. Up-dating the Cholodny method using PET films to sample microbial communities in soil. Biopolym. Cell 2011, 27, 199–205. [Google Scholar] [CrossRef]
- Siedenbiedel, F.; Tiller, J.C. Antimicrobial polymers in solution and on surfaces: Overview and functional principles. Polymers 2012, 4, 46–71. [Google Scholar] [CrossRef]
- Wang, H.; Chen, H.; Wang, F.; Wei, D.; Wang, X. An improved 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) reduction assay for evaluating the viability of Escherichia coli cells. J. Microbiol. Methods 2010, 82, 330–333. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef]
- Allesen-Holm, M.; Barken, K.B.; Yang, L.; Klausen, M.; Webb, J.S.; Kjelleberg, S.; Molin, S.; Givskov, M.; Tolker-Neilsen, T. A characterization of DNA release in Pseudomonas aeruginosa cultures and biofilms. Mol. Microbiol. 2006, 59, 1114–1128. [Google Scholar] [CrossRef]
- Thomas, V.C.; Hiromasa, Y.; Harma, N.; Thurlow, L.; Tomich, J.; Hancock, L.E. A fratricidal mechanism is responsible for eDNA release and contributes to biofilm development of Enterococcus faecalis. Mol. Micriol. 2009, 72, 1022–1036. [Google Scholar] [CrossRef]
- Montanaro, L.; Poggi, A.; Visai, L.; Ravaioli, S.; Campoccia, D.; Speziale, P.; Arciola, C.R. Extracellular DNA in biofilms. Int. J. Artif. Organs 2011, 34, 824–831. [Google Scholar] [CrossRef]
- Christner, M.; Heinze, C.; Busch, M.; Franke, G.; Hentschke, M.; Duhring, S.B.; Buttner, H.; Kotasinska, M.; Wischnewski, V.; Kroll, G.; et al. sarA negatively regulats staphylococcus epidermidis biofilm formation by modulation expression of 1 MDa extracellular matrix binding protein and autolysis-dependent release of eDNA. Mol. Microbiol. 2012, 86, 394–410. [Google Scholar] [CrossRef]
- Wu, J.; Xi, C. Evaluation of different methods for extracting extracellular DNA from the biofilm matrix. Appl. Environ. Microbiol. 2009, 5390–5395. [Google Scholar] [CrossRef]
- Schwartz, K.; Ganesan, M.; Payne, D.E.; Solomon, M.J.; Boles, B.R. Extracellular DNA facilitates the formation of functional amyloids in Staphylococcus aureus biofilms. Mol. Microbiol. 2016, 99, 123–134. [Google Scholar] [CrossRef]
- Hannan, S.; Ready, D.; Jasni, A.S.; Rogers, M.; Pratten, J.; Roberts, A.P. Transfer of antibiotic resistance by transformation with eDNA within oral biofilms. FEMS Immunol. Med. Microbiol. 2010, 59, 345–349. [Google Scholar] [CrossRef]
- Itzek, A.; Zheng, L.; Chen, Z.; Merritt, J.; Kreth, J. Hydrogen peroxide-dependent DNA release and transfer of antibiotic resistance genes in Streptococcus gordonii. J. Bacteriol. 2011, 193, 6912–6922. [Google Scholar] [CrossRef]
- Wei, D.; Zhou, R.; Guan, Y.; Zheng, A.; Zhang, Y. Investigation on the reaction between polyhexamethylene guanidine hydrochloride oligomer and glycidyl methacrylate. J. Appl. Polym. Sci. 2013, 127, 666–674. [Google Scholar] [CrossRef]
- Lewis, K.; Klibanov, A.M. Surpassing nature: Rational design of sterile-surface materials. Trends Biotechnol. 2005, 23, 343–348. [Google Scholar] [CrossRef]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed.; Cold Spring Harbor Laboratory Press, Cold Spring Harbor: New York, NY, USA, 1989; Volume 3, ap. B.11 and B.23. [Google Scholar]


| Sample | Contact Angle (Degree) | Surface Charge Density (C/cm2) |
|---|---|---|
| PA-11 | 80 ± 2 | (0.47 ± 0.02) × 10−10 |
| PA-11/PHMH-NS (5%) | 70 ± 2 | (1.2 ± 0.1) × 10−10 |
| PA-11/PHMH-NS (7%) | 68 ± 2 | (6.5 ± 0.3) × 10−10 |
| PA-11/PHMH-NS (10%) | 68 ± 2 | (27 ± 1) × 10−10 |
| Sample | Tensile Strength, MPa | Elongation at Break, % |
|---|---|---|
| PA-11 | 45 ± 2 | 24 ± 4 |
| PA-11/PHMG-NS (5%) | 45 ± 1 | 25 ± 5 |
| PA-11/PHMG-NS (7%) | 43 ± 2 | 15 ± 3 |
| PA-11/PHMG-NS (10%) | 27 ± 2 | 6 ± 2 |
| Sample | TΔm = 5%, °C | TΔm = 10%, °C | TΔm = 20%, °C | TΔm = 50%, °C |
|---|---|---|---|---|
| PHMG-NS | 357 | 372 | 381 | 398 |
| PA-11 | 425 | 441 | 450 | 463 |
| PA-11/PHMG-NS (5%) | 413 | 434 | 445 | 456 |
| PA-11/PHMG-NS (7%) | 405 | 430 | 442 | 449 |
| PA-11/PHMG-NS (10%) | 391 | 408 | 420 | 443 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moshynets, O.; Bardeau, J.-F.; Tarasyuk, O.; Makhno, S.; Cherniavska, T.; Dzhuzha, O.; Potters, G.; Rogalsky, S. Antibiofilm Activity of Polyamide 11 Modified with Thermally Stable Polymeric Biocide Polyhexamethylene Guanidine 2-Naphtalenesulfonate. Int. J. Mol. Sci. 2019, 20, 348. https://doi.org/10.3390/ijms20020348
Moshynets O, Bardeau J-F, Tarasyuk O, Makhno S, Cherniavska T, Dzhuzha O, Potters G, Rogalsky S. Antibiofilm Activity of Polyamide 11 Modified with Thermally Stable Polymeric Biocide Polyhexamethylene Guanidine 2-Naphtalenesulfonate. International Journal of Molecular Sciences. 2019; 20(2):348. https://doi.org/10.3390/ijms20020348
Chicago/Turabian StyleMoshynets, Olena, Jean-François Bardeau, Oksana Tarasyuk, Stanislav Makhno, Tetiana Cherniavska, Oleg Dzhuzha, Geert Potters, and Sergiy Rogalsky. 2019. "Antibiofilm Activity of Polyamide 11 Modified with Thermally Stable Polymeric Biocide Polyhexamethylene Guanidine 2-Naphtalenesulfonate" International Journal of Molecular Sciences 20, no. 2: 348. https://doi.org/10.3390/ijms20020348
APA StyleMoshynets, O., Bardeau, J.-F., Tarasyuk, O., Makhno, S., Cherniavska, T., Dzhuzha, O., Potters, G., & Rogalsky, S. (2019). Antibiofilm Activity of Polyamide 11 Modified with Thermally Stable Polymeric Biocide Polyhexamethylene Guanidine 2-Naphtalenesulfonate. International Journal of Molecular Sciences, 20(2), 348. https://doi.org/10.3390/ijms20020348





