1. Introduction
Recently, metal–air batteries have attracted high interest of researchers and industry as post lithium–ion technology. Among all metal–air batteries, aqueous zinc–air battery is a relatively established technology (known to the scientific community since the late nineteenth century) with high potential to be used in future energy requirements [
1,
2,
3]. This type of battery has a very high theoretical energy density of 1086 Wh·kg
−1 (including oxygen), five times higher than that of existing lithium–ion batteries [
4]. Moreover, their production cost is estimated to be very low (~10
$·kW
−1·h
−1) in comparison to lithium–ion batteries. Technically and economically, the zinc–air battery is deemed to be the most feasible alternative for lithium–ion batteries in various applications. Despite the early start and great potential, the development of zinc–air batteries has been limited by issues related to separator, electrolyte, metal electrode, and air catalyst [
5].
Despite being an essential part of a zinc–air battery, the separator has not received its deserved attention compared to other components of the battery. The primary function of the separator is to prevent physical contact between the anode and cathode, while providing high ionic conductivity and selectivity to facilitate hydroxide ion transport with the aim of completing the battery circuit during its operation. An ideal separator must be chemically stable in contact with highly alkaline environment and electrochemically stable within a wide working potential window. Also it must be able to prevent zincate (Zn(OH)
42−) crossover and short-circuit due to the formation of zinc dendrites during battery charging. Besides, high electrical resistance and ionic conductivity is highly desirable [
6].
Practically, the separator for zinc–air batteries is usually a porous polypropylene membrane such as Celgard
® with the porosity of 10–20 µm. The porosity of these membranes results in their high ionic conductivity, which is crucial for a separator material. Nonetheless, due to the high porosity, beside hydroxide ions, zincate ions, which are produced through the oxidation of zinc during the battery discharge, also pass through the membrane from the anode to the cathode [
7]. At the cathode side, because of asymmetric water evaporation-induced electrolyte deficiency, zincate ions are precipitated as zinc oxide (ZnO) on the catalyst surface, forming a resistive layer to ion/electron conduction which in turn, leads to higher polarization of air electrode and capacity loss of zinc–air batteries [
8].
Even though there have been several attempts to develop a separator membrane with desired characteristics to be used in zinc–air batteries, more efforts are required in this field. Dewi et al. used a polyelectrolyte containing a sulfonium cation as a zinc–air battery separator. Even though they incorrectly assumed zinc ions in the electrolyte to be in the form of a cation (Zn
2+), the separator was highly effective in preventing zinc ions crossover to the cathode side, leading to the capacity increase by more than six times compared to polypropylene-based Celgard
® separator [
7]. More recently, electrospun polyetherimide (PEI) nanofibers impregnated with polyvinyl alcohol (PVA) has been used as a highly efficient separator for rechargeable zinc–air battery. In the prepared permselective membrane, PEI, which is known for its high chemical resistance in the alkaline environment, provides good mechanical stability. On the other hand, the pores of the electrospun nanofibers filled with PVA electrolyte offers high ionic conductivity. In such a design, zincate crossover decreased dramatically due to the bulky size of the zincate ions, making it difficult to pass through the separator compared to hydroxide ions [
8]. In another study, similar impregnated nanofibrous mat concept with electrospun PVA/polyacrylic acid (PAA) nanofibers along with the impregnation solution of Nafion bearing pendant sulfonate groups was used. In this case, the nanofibers acted as the ionic conductive pathways for hydroxide ions, and the impregnation solution played the role of an anion repelling component, preventing bulky zincate ions transport from the anode to the cathode side [
9].
Basically, the ionic conductivity of polymeric materials is determined by two factors: the ionic mobility and the ion exchange capacity (IEC). To increase hydroxide conductivity, improving IEC seems to be an easier way than improving hydroxide mobility. However, increasing IEC always results in excessive water uptake, which is a negative side effect, leading to severe swelling or even dissolution at higher temperatures. Moreover, increasing IEC usually leads to an increased zincate crossover. Therefore, a better and more efficient approach to expanding the hydroxide conductivity and selectivity is to enhance hydroxide mobility while keeping the IEC at a medium level. Such a growth in the hydroxide conductivity could be realized by reforming the ionic channels in polymer structure through hydrophilic/hydrophobic microphase separation. In this approach, a cation which is usually quaternary ammonium (–NR
3+), is covalently bound to a hydrophobic polymer backbone and the anion (hydroxide) is dissociated in aqueous phase [
10]. Several polymeric materials have been used to develop this type of separator membrane such as poly(arylene ether)s, poly-(phenylene)s, poly(ether imide)s, poly(styrene)s, poly(olefin)s, and poly(phenylene oxide)s [
11].
Recently, there has been high interest in poly (phenylene oxide) (PPO) as the polymer backbone. PPO exhibits low-cost; commercial availability; high thermal, mechanical, and chemical stabilities; and facile postfunctionalization [
12]. Li et al. designed and synthesized comb-shaped quaternized copolymer using PPO as the base polymer. The synthesis was carried out using Menshutkin reaction with N,N-dimethyl-1-hexadecylamine (DMHDA), and subsequent hydroxide exchange. In this process, DMHDA was attached to the benzylic position of PPO through covalent bonding from nitrogen atom, forming a quaternary ammonium group directly attached to the polymeric backbone with a pendant long hydrocarbon chain. The polymer exhibited phase-separated morphology with enhanced ionic conductivity and alkaline stability, making it a potential material as hydroxide exchange membrane for alkaline fuel cells [
13]. In another study, Yang et al. prepared various quaternized PPO-based anion-exchange membranes using a series of saturated heterocyclic compounds including 1-methylpyrrolidine (MPy), 1-ethylpyrrolidine (EPy), 1-butylpyrrolidine (BPy), 1-methylpiperidine (MPrD), 1-ethylpiperidine (EPrD), and N-methylmorpholine (NMM). Also, the comparison of their physicochemical characteristics with TMA-quaternized PPO as the benchmark polymer was performed. The quaternized membranes showed different ionic conductivity and alkaline stability, depending on the quaternization agent used. For example, the polymer containing 1-methylpyrolidine exhibited ionic conductivity of 27 mS·cm
−1 at 80 °C and excellent alkaline stability, keeping 87% of its original conductivity after being soaked in 1 M KOH at 80 °C for 500 h [
14].
Although this strategy has already been used in fuel cell technology, its application in zinc–air batteries, where an alkaline stable hydroxide exchange membrane is required as well, is very rare. The main focus of this study is to synthesize PPO-based microphase separated hydroxide exchange membrane through the attachment of three various quaternary ammonium molecules to the main polymeric backbone with the aim of developing ionic channels for selective transfer of hydroxide ions. Even though the main aim of developing new separators with acceptable ionic conductivity and low zincate crossover is for overcoming challenges associated with rechargeable zinc–air batteries, this phase of our study includes separators characterization and their application in a primary zinc–air battery as an indication of their applicability in secondary batteries. The separators are characterized using physicochemical and electrochemical characterization tools to investigate their properties, including their ionic conductivity and zincate diffusion coefficient. Then, they are implemented in a primary zinc–air battery to study their influence on battery performance. The ionic conductivity, along with suppressed zincate crossover, makes this new separator an excellent candidate to be used in primary and secondary zinc–air batteries. In the next phase, the application of the separators in rechargeable zinc–air batteries will be investigated.
3. Materials and Methods
3.1. Materials
Poly(2,6-dimethyl-1,4-phenylene oxide) powder (PPO), 1-methylpyrrolidine (≥98%), and manganese (IV) oxide (5 μm, 99.99%) were supplied by Sigma-Aldrich (St. Louis, MO, USA). N-bromosuccinimide (NBS) for synthesis, benzoyl peroxide (BPO, with 25% H2O), and chlorobenzene were purchased from Merck Millipore. 1-methyl-2-pyrrolidinone (NMP), ethanol, methanol, and toluene all with Grade AR were purchased from QRëC (New Zealand). N,N-dimethylformamide (DMF) (LOBA Chemie, Grade AR, Mumbai, India), trimethylamine (TCI, ca. 25% in methanol), 1-methylimidazole (TCI, ≥99%), chloroform (BDH Chemicals, UK), and KOH plates (Kemaus, Australia) were used as received without further purification. Nickel (Ni) foam as cathode current collector with a purity of 99.97%, 100 pores per inch (PPI), and 1 mm thick was purchased from Qijing Trading Co., Ltd. (Wenzhou, China). Carbon black (Vulcan® XC-72, Cabot Corporation, Boston, MA, USA), BP-2000 (BLACK PEARLS® 2000, Cabot Corporation), and zinc plate (0.1 mm thick 99.99%, Shandong Yr Electronic Co., Ltd., Shandong, China) were used as received. Poly (styrene-co-butadiene) (Sigma-Aldrich, butadiene 4 wt%) was used to prepare binder for the cathode. Poly(vinyl acetate) (PVAc) from TOA Paint Public Co., Ltd. (Samut Prakarn, Thailand) and No. 4 Whatman filter paper (Sigma-Aldrich) were used to prepare the benchmark separator.
3.2. Separator Membrane Synthesis
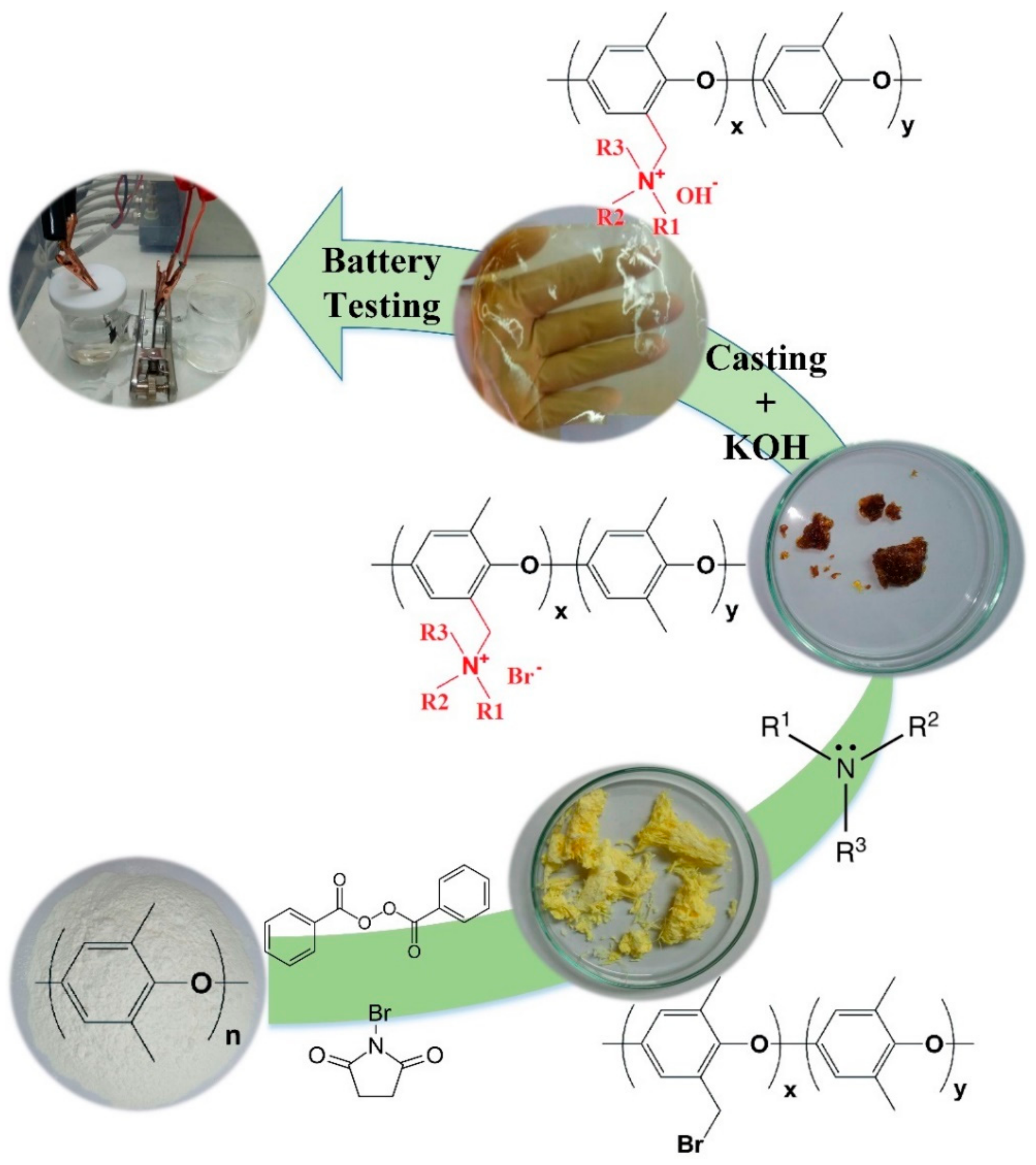
PPO-based phase-separated hydroxide exchange separator membranes were synthesized using a process described previously [
19]. The first stage of separator preparation was bromination of PPO at the benzylic position. A certain amount of PPO was dissolved in chlorobenzene at 50 °C under nitrogen atmosphere to prepare 5 wt/
v% solution. Then, the temperature was increased to 80 °C, NBS (with PPO:NBS weight ratio of 1:0.8) and BPO (with NBS:BPO weight ratio of 1:0.05) were added to the mixture, and the reaction continued for 4 h under nitrogen atmosphere. The solution was then cooled down to room temperature and poured into an excessive amount of methanol for product precipitation. A pale-yellow fiber-like product was separated and then dissolved in chloroform for purification. The solution was precipitated again in an excessive amount of ethanol, filtered and dried in the vacuum oven at 60 °C overnight.
The next stage was to introduce quaternary ammonium functional group. Brominated PPO (BrPPO) was dissolved in NMP with the concentration of 7.5 wt/
v%, and then an excessive amount of the quaternization agent (trimethylamine, 1-methylpyrrolidine, and 1-methylimidazole) was added to the solution. The reaction continued for 48 h in room temperature, and then the product was precipitated using toluene, washed a few times and then dried in a vacuum oven at 65 °C for 24 h [
14].
In the next stage, a certain amount of quaternized BrPPO (Q-BrPPO) was dissolved in DMF at room temperature to prepare 30 wt/
v% casting solution. The membrane was fabricated by casting the prepared solution onto a clean surface. Finally, the prepared cast films were soaked in degassed KOH, 7 M solution for 72 h to exchange the bromine ions in the polymer structure with hydroxide. Fresh hydroxide exchange solutions were used every 24 h to make sure of complete ion exchange [
15].
To evaluate the performance of the synthesized separator membranes, a benchmark membrane using Whatman filter paper was also prepared. For this purpose, both sides of No.4 Whatman filter paper was coated with a 24 wt% PVAc solution and dried in an oven at 60 °C for 15 min.
3.3. Structural/Physicochemical Characterization
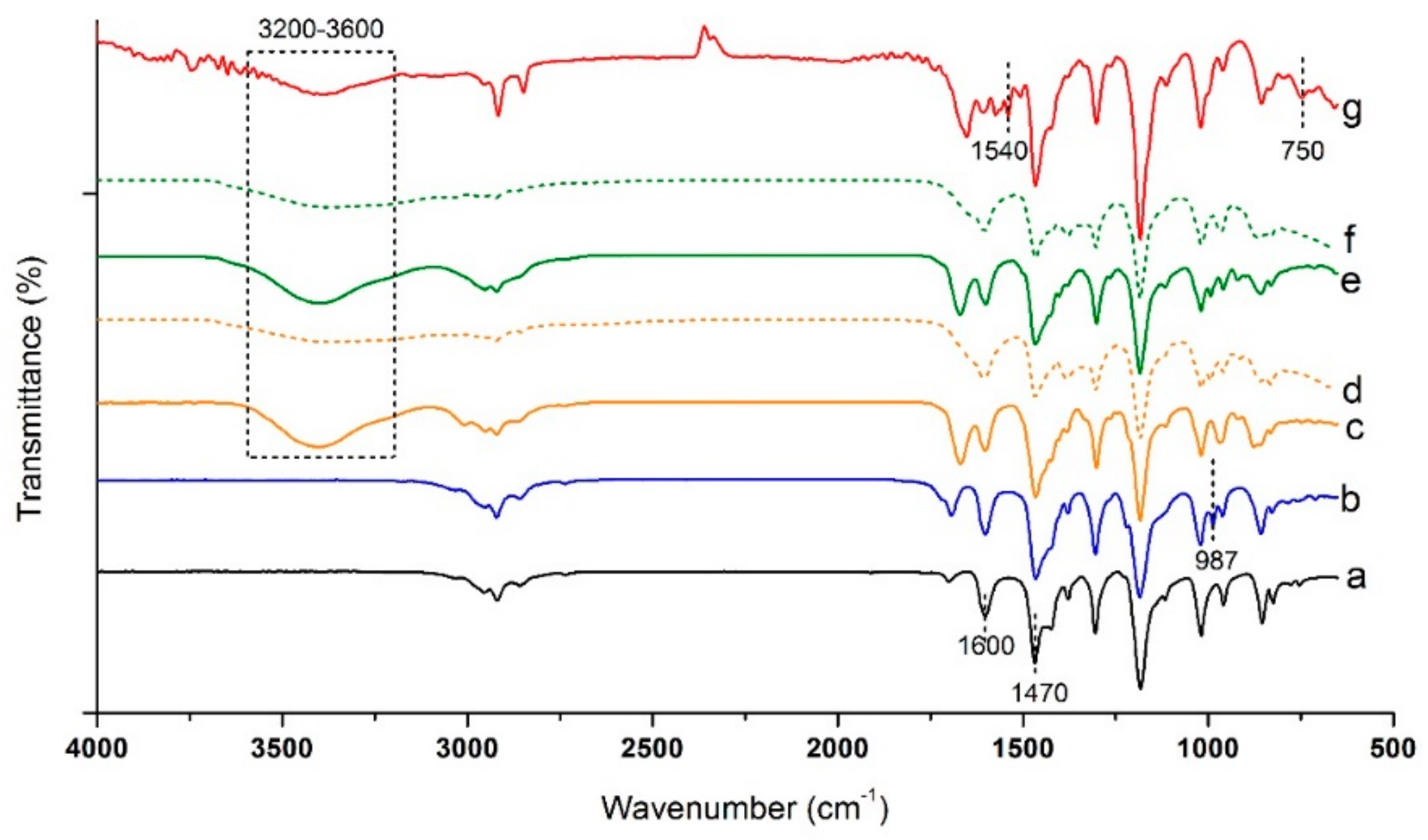
The 1HNMR spectrum of BrPPO was obtained on a Bruker, Avance III HD 500 MHz using deuterated trichloromethane (CDCl3) as a solvent to determine the bromination degree of PPO. A Perkin Elmer, Spectrum One Fourier-transform infrared (FTIR) spectrometer in the frequency range of 4000 to 600 cm−1 was carried out to study the chemical changes in the molecular structure during various synthesis stages. Thermogravimetric analysis was performed using SDT-Q600 TGA instrument in the temperature range of 30 to 600 °C at a heating rate of 10 °C min−1 to evaluate the thermal stability of the samples.
To measure water/electrolyte uptake capacity of the membranes, they were soaked in water/KOH, 7 M solution for 24 h and the weight differences before and after soaking were used for the measurements using Equation 1 [
8].
where W
wet and W
dry are the weights of the membranes after and before soaking in water/electrolyte, respectively. Similarly, dimensional changes of the membranes induced by water/electrolyte uptake were calculated by Equations 2 and 3 [
8].
3.4. Electrochemical Characterization
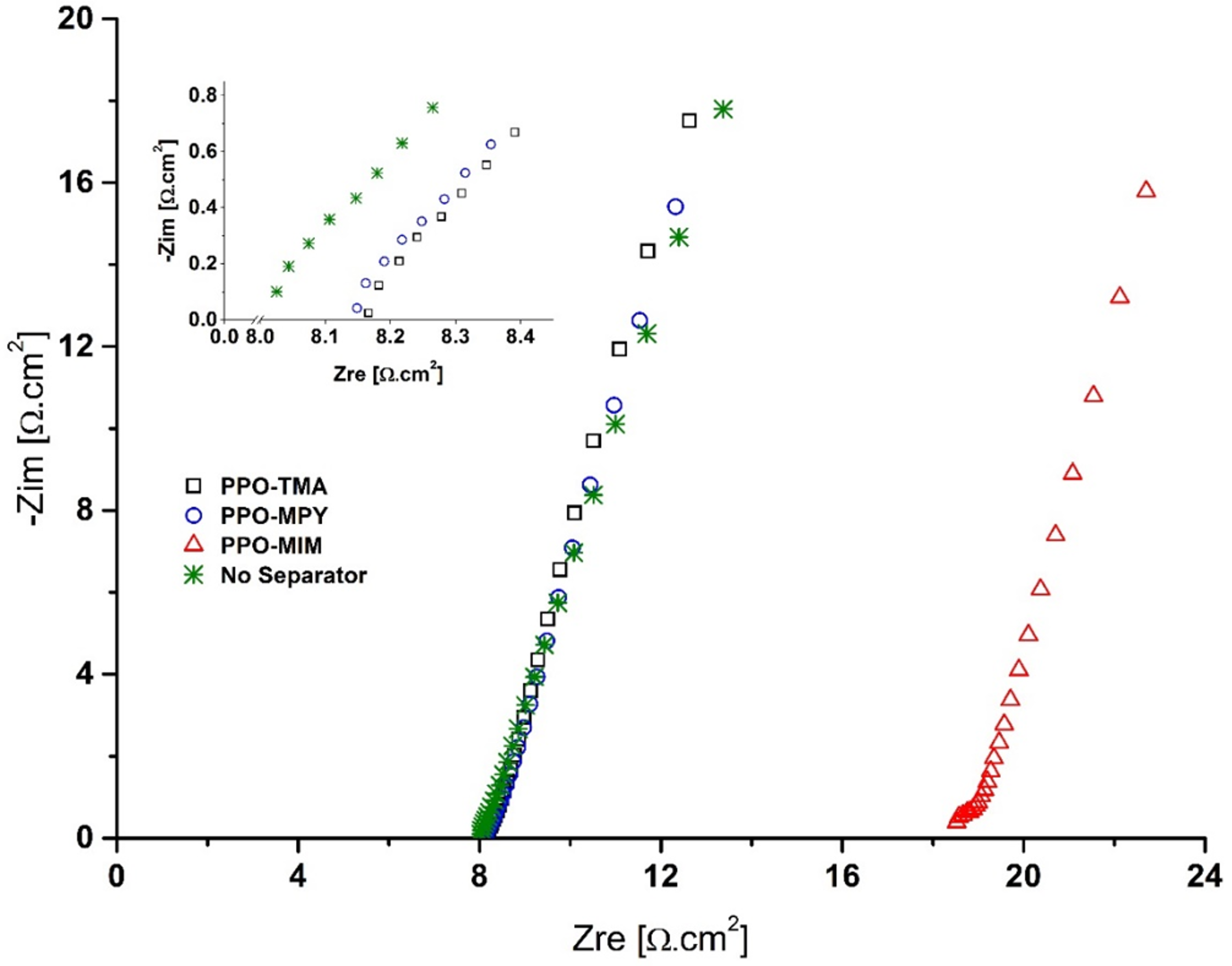
The ionic conductivity of the prepared membranes was measured using a potentiostat/galvanostat with impedance measurement unit (AMETEK, PAR VersaSTAT 3A) in the frequency range of 1 Hz–100 kHz with the excitation voltage of 10 mV
RMS at room temperature. For this measurement, a diffusion cell with two chambers was used. EIS measurement with and without the membrane placed between two chambers containing KOH, 7 M was performed, and the difference in the bulk resistance (
Rb) of the two measurements was used to calculate the ionic conductivity of the membranes using Equation 4 [
25].
σ is the ionic conductivity (S/cm),
Rb is the bulk resistance (Ω), and
l and
A are thickness (cm) and area (cm
2) of the membrane, respectively.
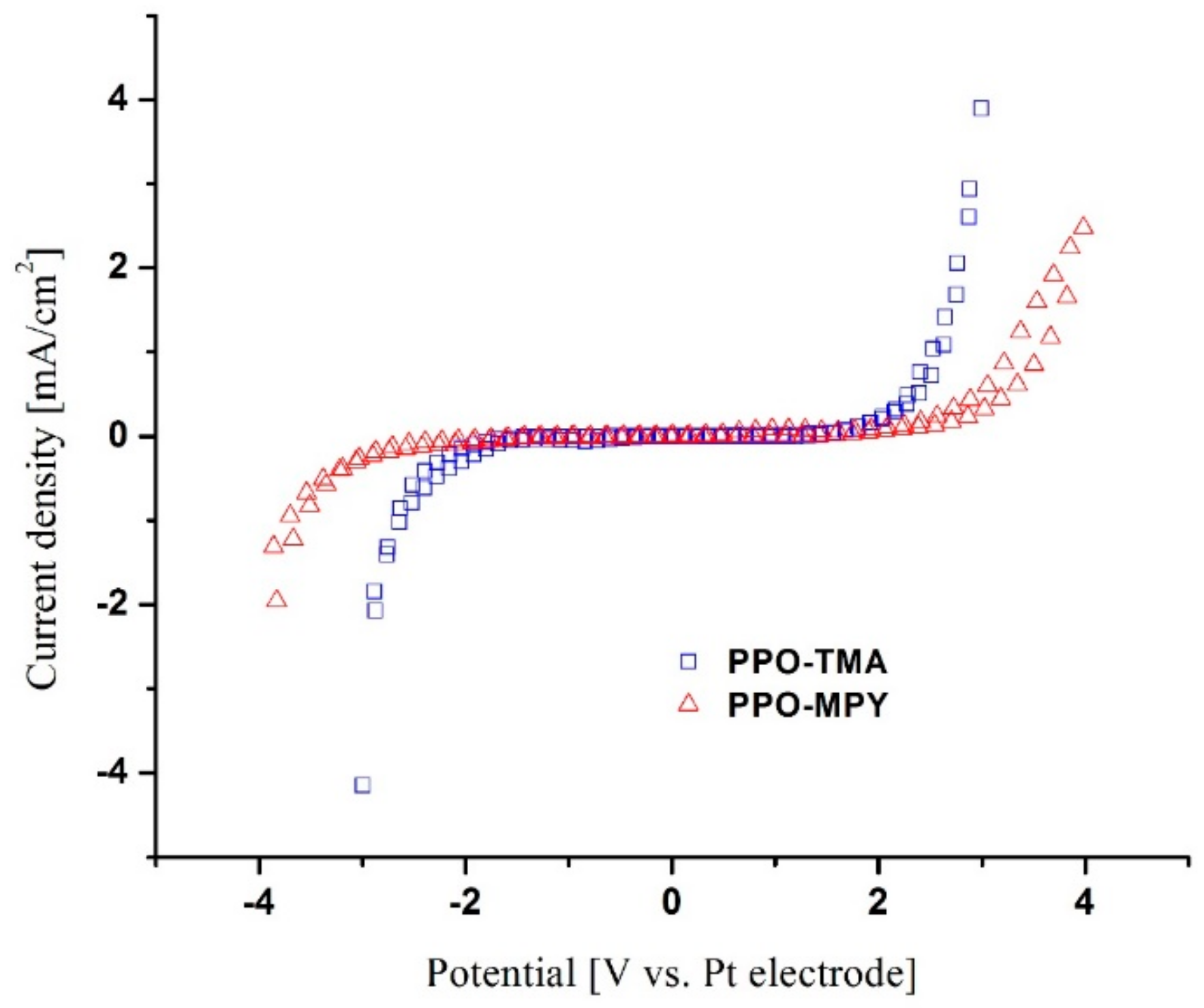
Cyclic voltammetry (CV) was carried out using an AMETEK, PAR VersaSTAT 3A potentiostat/galvanostat to evaluate electrochemical stability window of the membranes. Two-electrode configuration tests using 1 × 1 cm2 platinum (Pt) working and counter electrodes were carried out for CV with a scan rate of 0.05 mV/s.
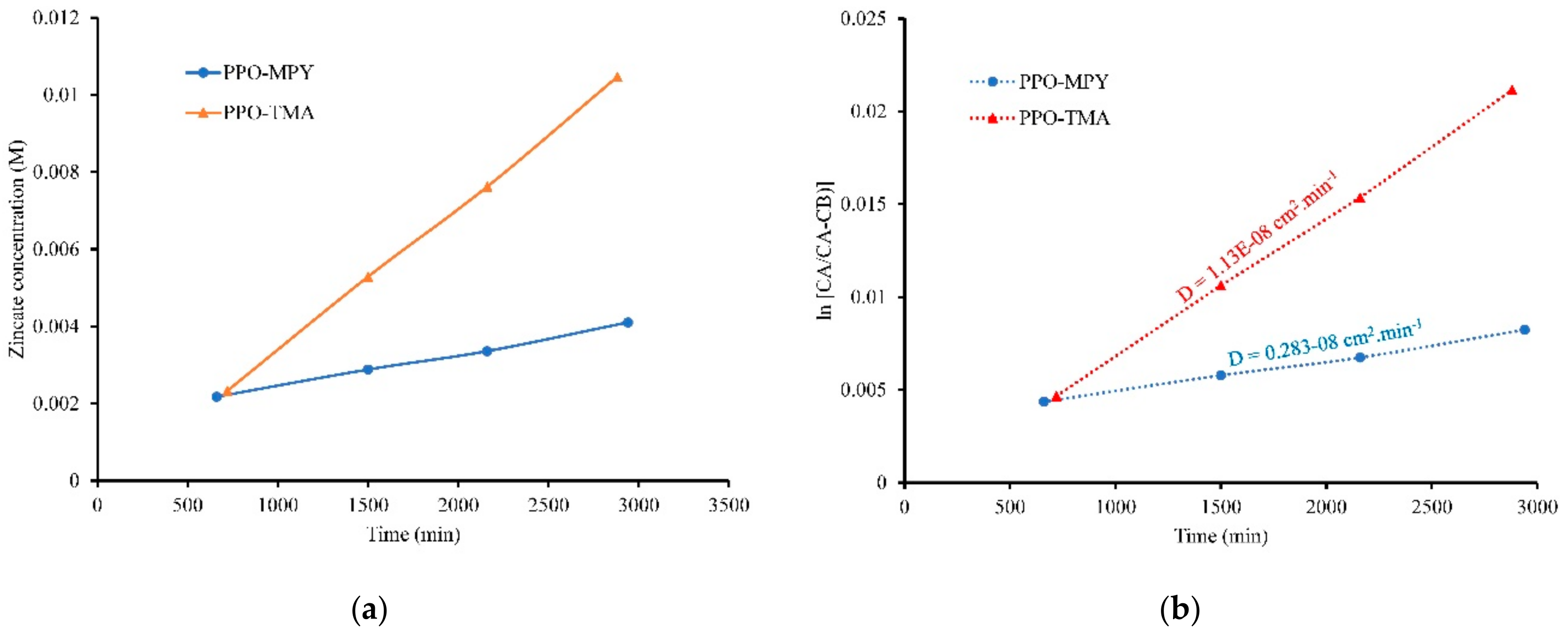
To study zincate ion (Zn(OH)
42-) crossover characteristics of the membranes, a kind of diffusion cell with two chambers was used [
9]. The left chamber contained 50 mL of KOH, 7 M solution plus 0.5 M dissolved ZnO in the form of zincate ions while the right chamber contained only 50 mL of KOH, 7 M solution (
Figure 11). The separator membrane was placed between two chambers. The chambers were stirred continuously to prevent concentration polarization. The concentration of zinc ions in the right chamber was measured in a predetermined time interval (12–14 h) using inductively coupled plasma optical emission spectroscopy (ICP-OES) to obtain time-dependent concentration variation graph. Moreover, the diffusion coefficient of zincate ions across the separator membranes was calculated from the experimental data using Equation 5 [
9].
D is the diffusion coefficient of zincate ions across the separator membrane (cm2/min), t is the time (min), VB is the solution volume in the right chamber (deficiency chamber), A is the effective surface area (cm2) of the separator, L is the thickness (cm) of the separator, and CA and CB are the concentration of zincate ions (mol/L) in the left and right chambers, respectively.
3.5. Discharge Performance
A homemade zinc–air cell was used to evaluate the performance of the separator membranes. In this cell, the separator was in direct contact with the cathode, and the anode was a 1 × 1 cm
2 pure zinc plate immersed in 40 mL of KOH, 7 M solution (
Figure 12). To prepare the cathode, a Ni-foam was used as the current collector and gas diffusion layer. One side of the foam was coated with a mixture of BP-2000 (30%) and PTFE (70%), dispersed in ethanol and pressed using a hot-press at 350 °C for 15 min (air diffusion side). For preparing the catalyst side of the foam, a mixture of MnO
2 (1.2 g: 30%), BP-2000 (1.4 g: 35%) and VXC-72 (1.4 g: 35%) was used. The mixture was stirred in 35 mL toluene for 2 h, and then 5 mL of 7.5 wt% poly(styrene-co-butadiene) solution in toluene (as a binder) was added and stirred for another 2 h. The final mixture was coated onto the Ni foam and pressed using a manual hot-press at 150 °C for 10 min. The size of the circular cathode used in the battery tests was 15 mm in the diameter.
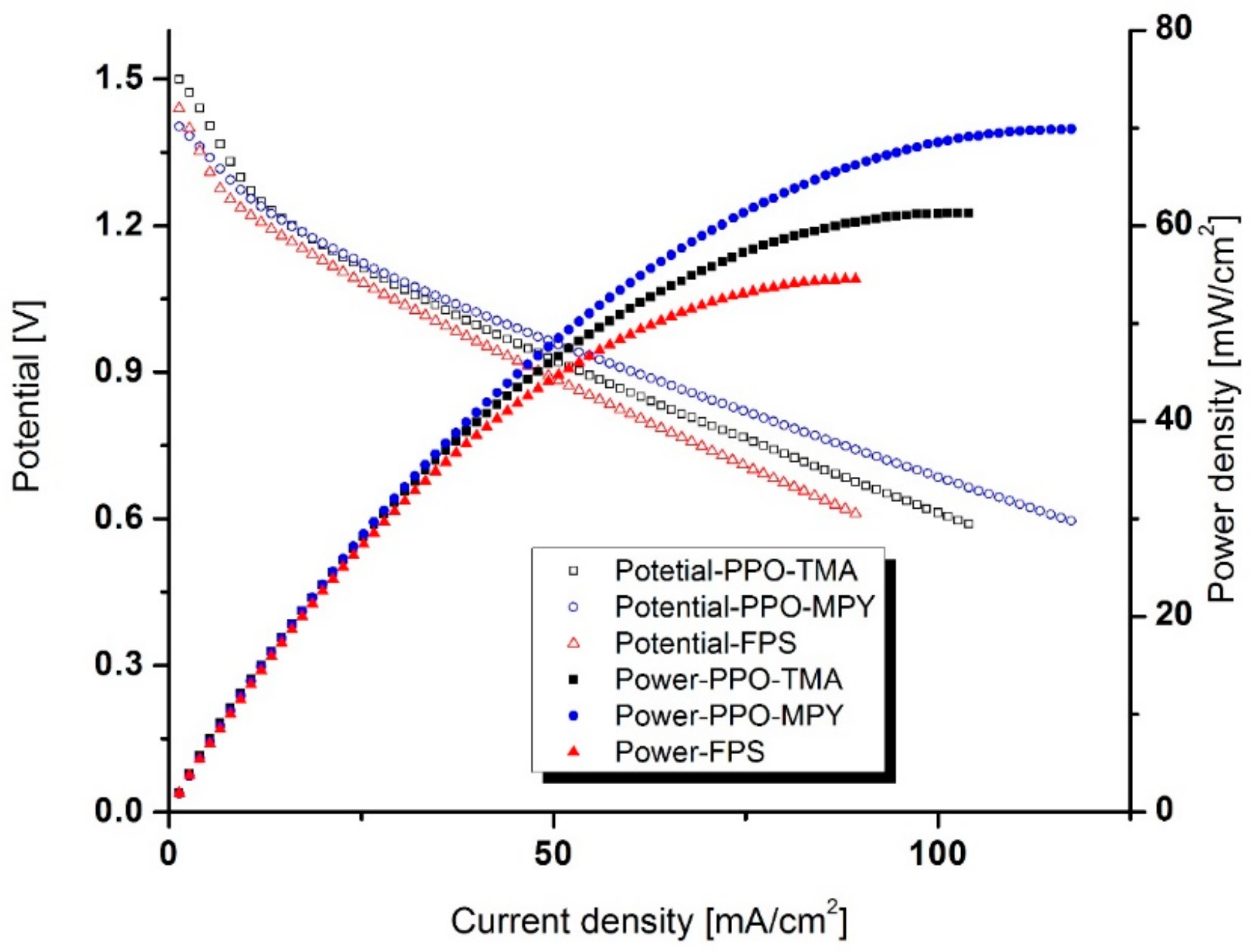
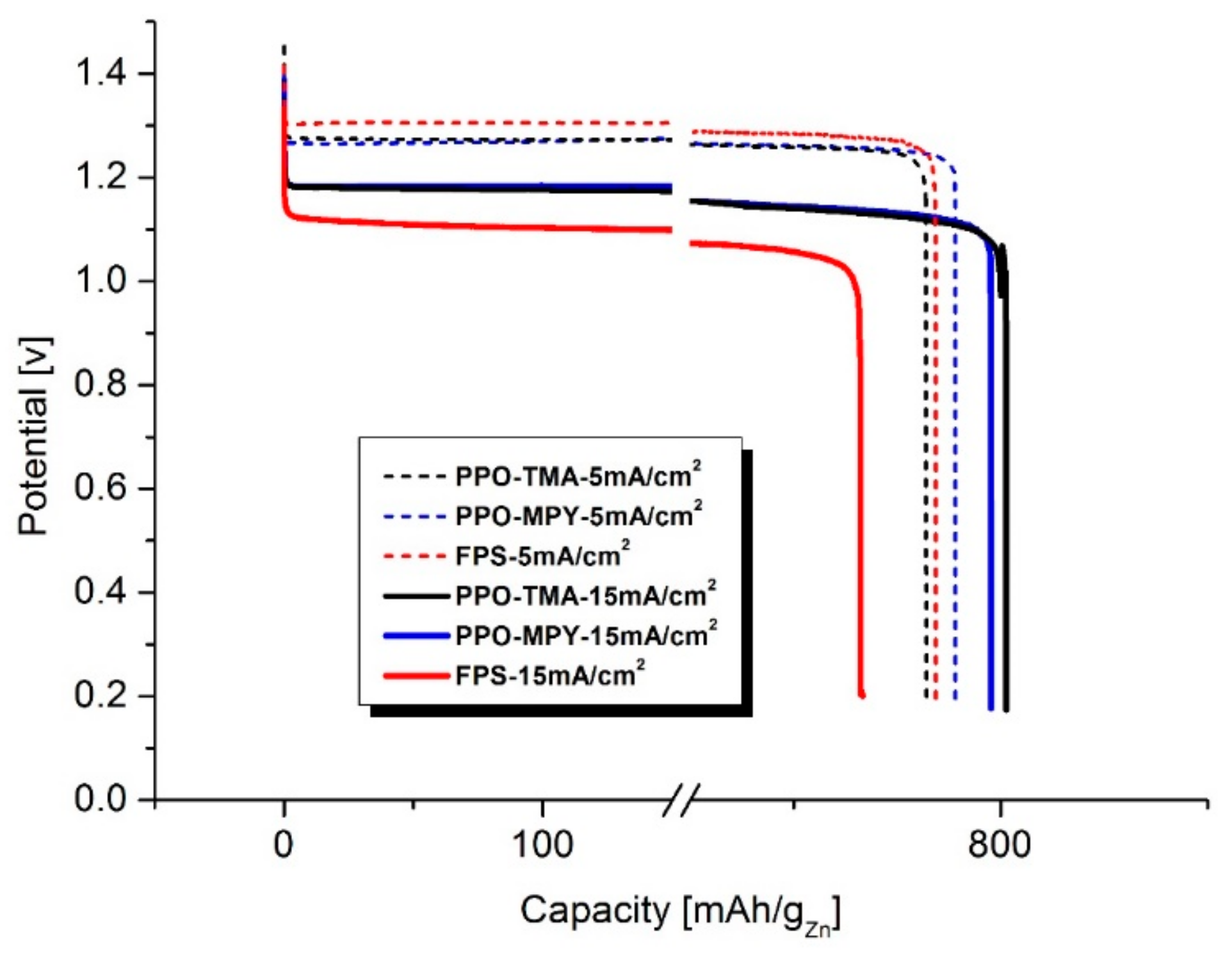
Discharge performance was measured using a Battery Testing System (NEWARE, Shenzhen, China) at room temperature. The cell was discharged at a constant discharge current in the range of 2.5 to 15 mA/cm2. For all experiments, the cut-off voltage was 0.9 V.