Yeast Sup35 Prion Structure: Two Types, Four Parts, Many Variants
Abstract
1. Introduction
2. Results
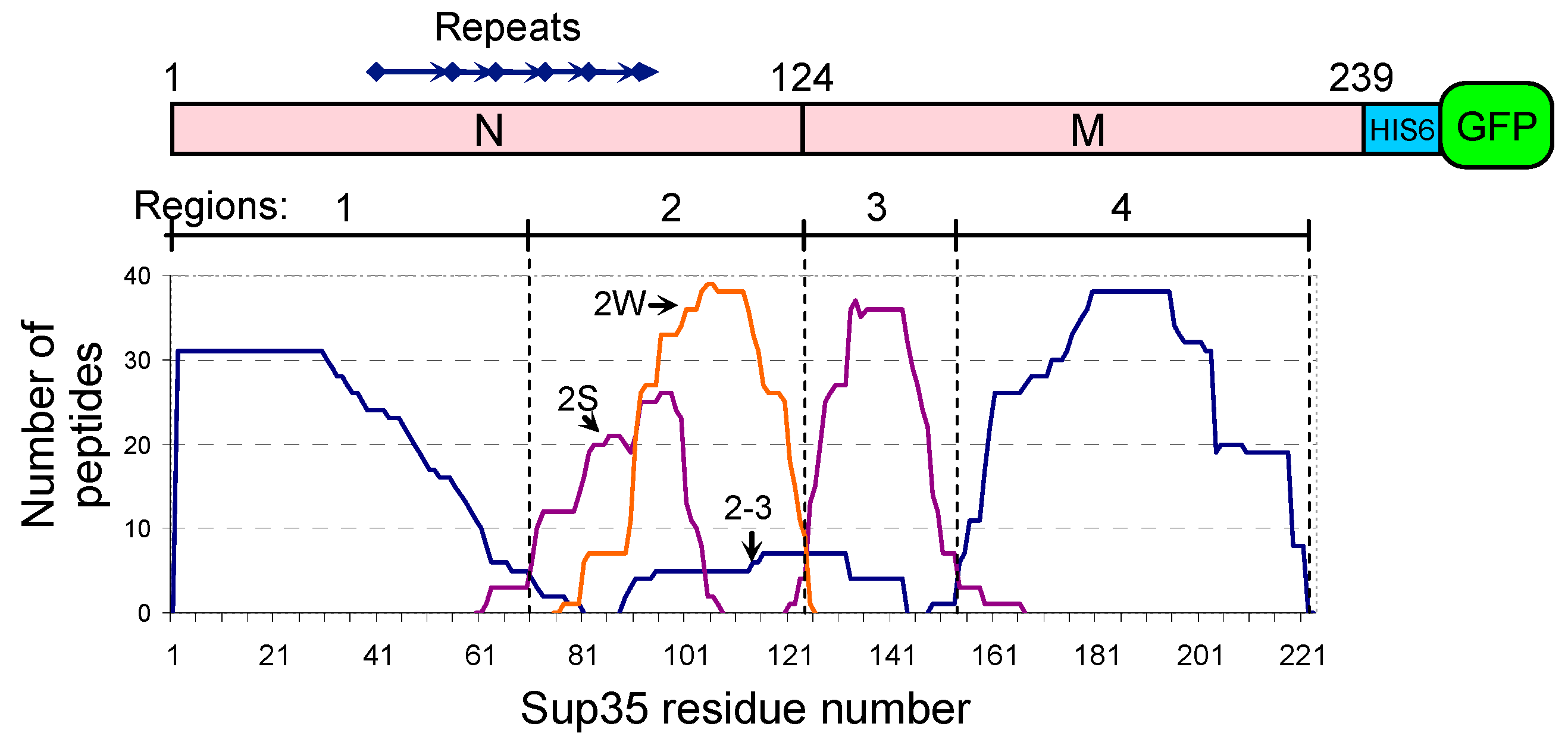
2.1. General Structure of Sup35 Prion
2.2. C-Terminal Core of the Rnq1 Prion
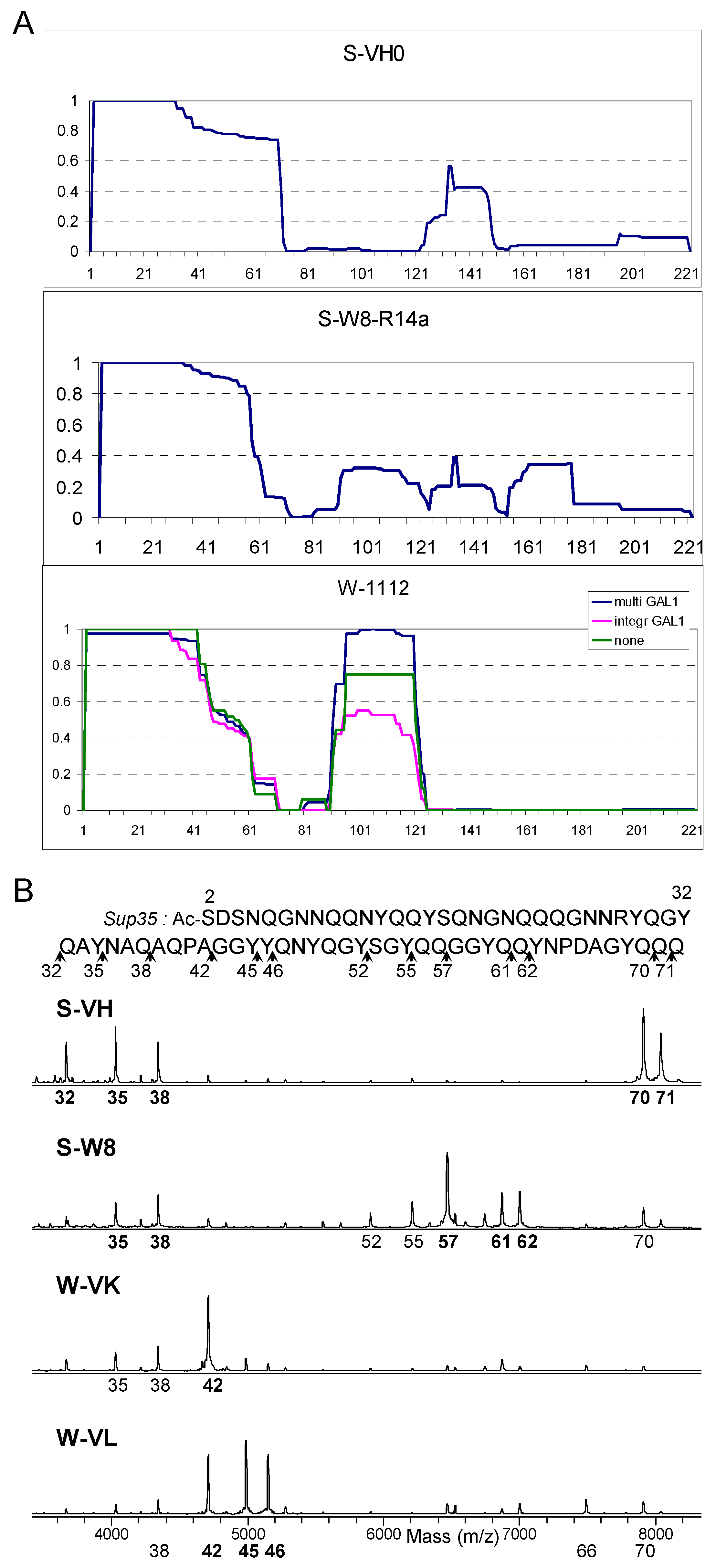
2.3. Weak and Strong [PSI+] Differ in Core 1 Structure
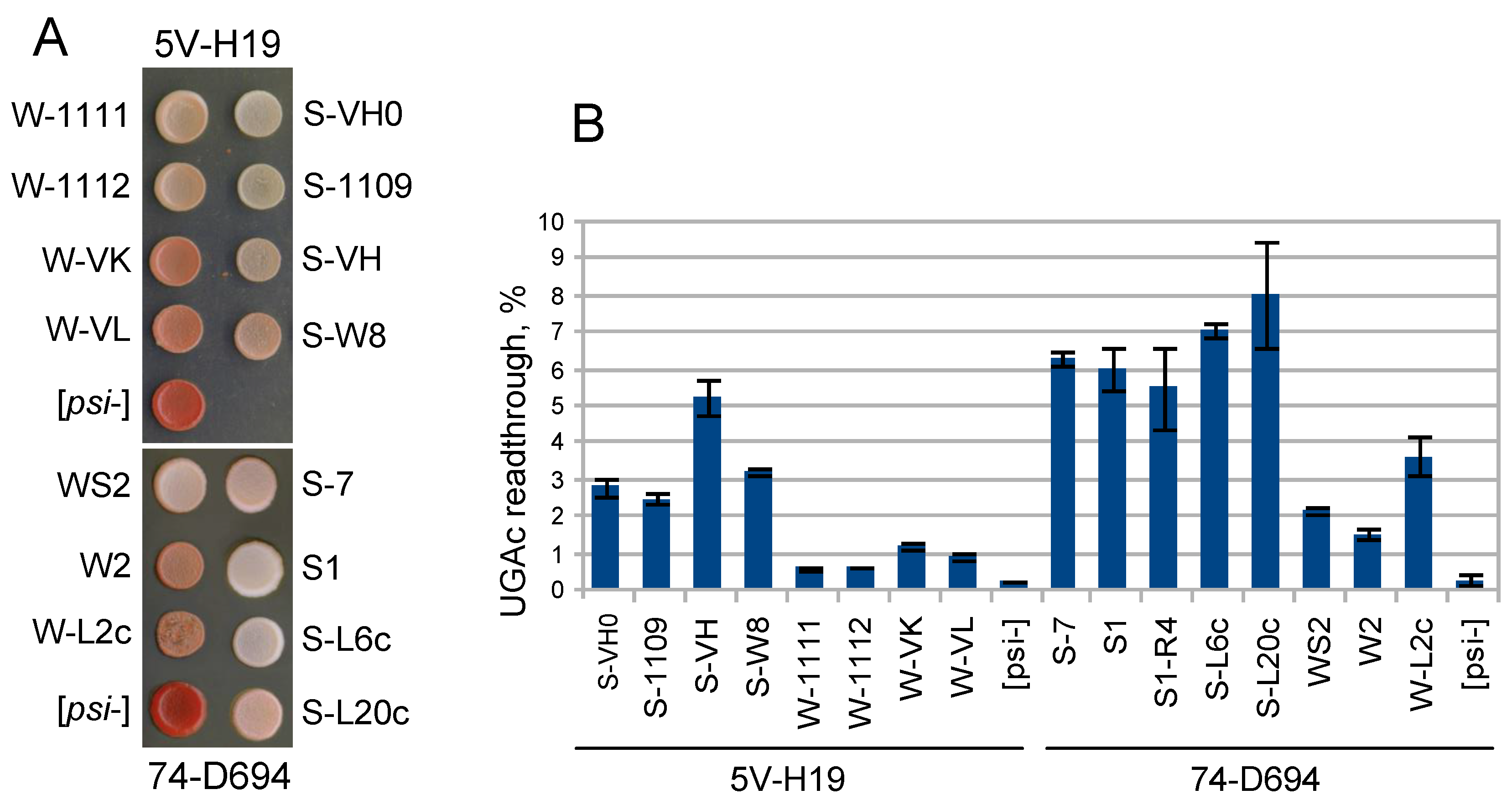
2.4. Additional Phenotypical Assays Distinguishing Weak and Strong [PSI+] Types
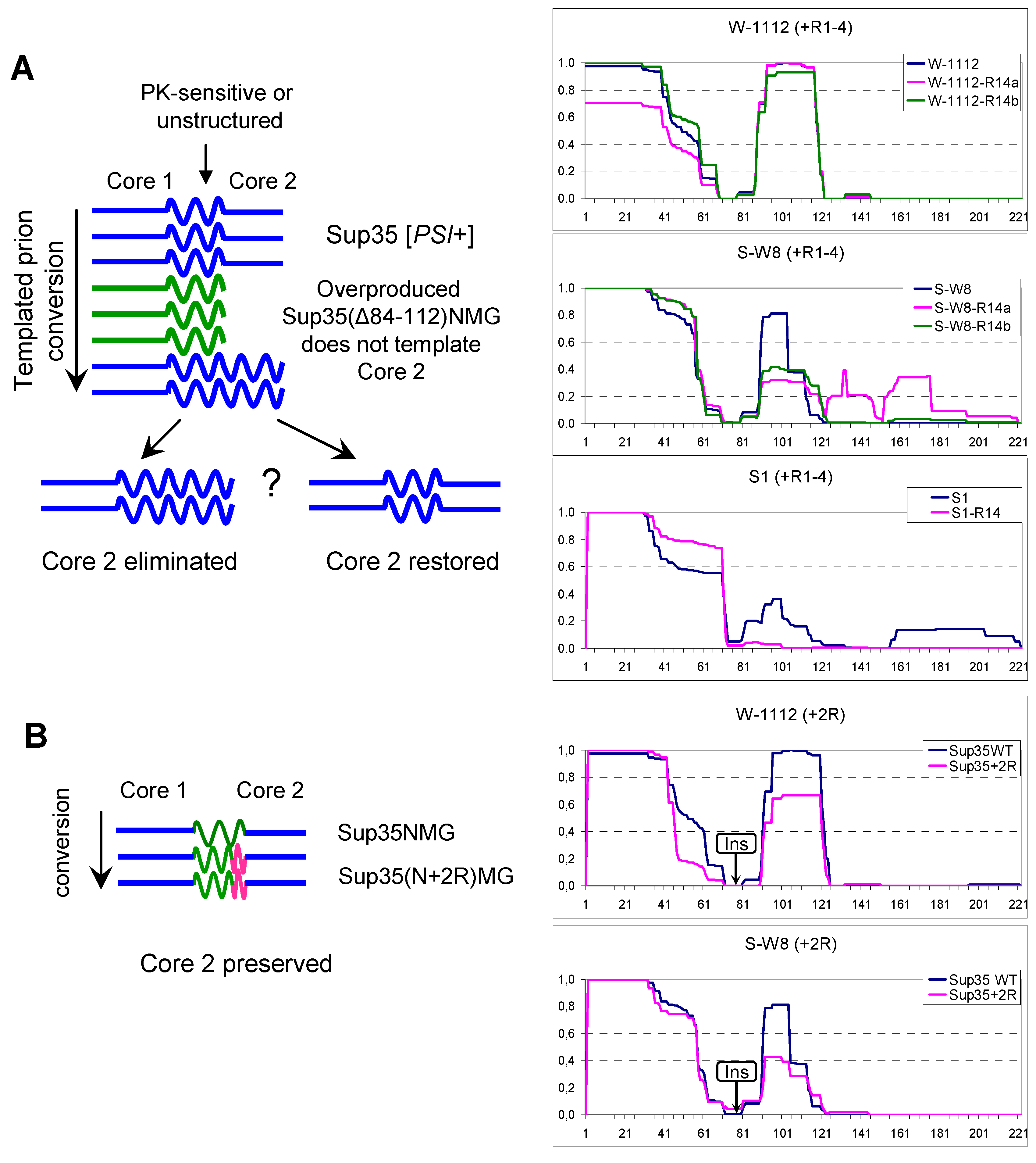
2.5. Manipulations with Core 2
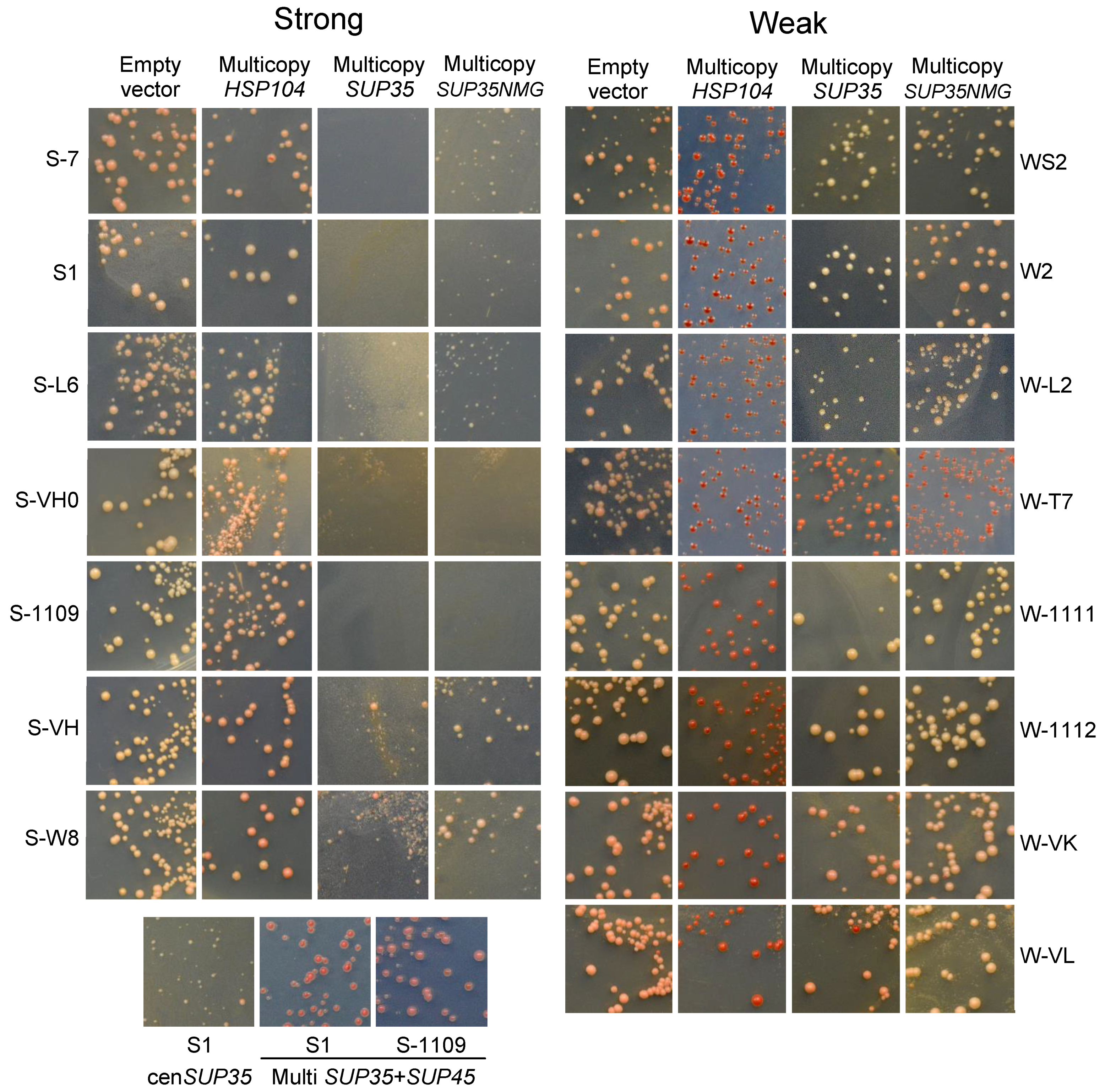
2.6. Instability and Heterogeneity of [PSI+] Variants
2.7. Involvement of the PK-Protected Regions in Intermolecular Structure
3. Discussion
3.1. General Properties of the Sup35 Prion Core
3.2. Two Types of [PSI+]
3.3. Comparison of the Sup35 Fibrils Obtained in Vitro and in Vivo
3.4. Problems in Discrimination of [PSI+] Variants
3.5. Details of Sup35 Prion Structure
4. Materials and Methods
4.1. Yeast Strains and Media
4.2. Selection and Characterization of [PSI+] Isolates
4.3. [PSI+] Variants Obtained in Nonstandard Ways
4.4. Plasmids
4.5. Prion Isolation From Yeast
4.6. PK Digestion and Mass Spectrometry
4.7. Nonsense Readthrough Measurement
4.8. PK Resistance Index Calculation
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PK | Proteinase K |
| MS | Mass spectral |
| MALDI-TOF | Matrix-assisted Laser Desorption/Ionization Time of Flight |
References
- Wickner, R.B.; Edskes, H.K.; Kryndushkin, D.; McGlinchey, R.; Bateman, D.; Kelly, A. Prion diseases of yeast: amyloid structure and biology. Semin. Cell Dev. Biol. 2011, 22, 469–475. [Google Scholar] [CrossRef]
- Shewmaker, F.; Wickner, R.B.; Tycko, R. Amyloid of the prion domain of Sup35p has an in-register parallel beta-sheet structure. Proc. Natl. Acad. Sci. USA 2006, 103, 19754–19759. [Google Scholar] [CrossRef]
- Paushkin, S.V.; Kushnirov, V.V.; Smirnov, V.N.; Ter-Avanesyan, M.D. Propagation of the yeast prion-like [psi+] determinant is mediated by oligomerization of the SUP35-encoded polypeptide chain release factor. EMBO J. 1996, 15, 3127–3134. [Google Scholar] [CrossRef] [PubMed]
- Pezza, J.A.; Villali, J.; Sindi, S.S.; Serio, T.R. Amyloid-associated activity contributes to the severity and toxicity of a prion phenotype. Nat. Commun. 2014, 5, 4384. [Google Scholar] [CrossRef] [PubMed]
- Ter-Avanesyan, M.D.; Dagkesamanskaya, A.R.; Kushnirov, V.V.; Smirnov, V.N. The SUP35 omnipotent suppressor gene is involved in the maintenance of the non-Mendelian determinant [psi+] in the yeast Saccharomyces cerevisiae. Genetics 1994, 137, 671–676. [Google Scholar] [PubMed]
- Ter-Avanesyan, M.D.; Kushnirov, V.V.; Dagkesamanskaya, A.R.; Didichenko, S.A.; Chernoff, Y.O.; Inge-Vechtomov, S.G.; Smirnov, V.N. Deletion analysis of the SUP35 gene of the yeast Saccharomyces cerevisiae reveals two non-overlapping functional regions in the encoded protein. Mol. Microbiol. 1993, 7, 683–692. [Google Scholar] [CrossRef] [PubMed]
- Helsen, C.W.; Glover, J.R. Insight into molecular basis of curing of [PSI +] prion by overexpression of 104-kDa heat shock protein (Hsp104). J. Biol. Chem. 2012, 287, 542–556. [Google Scholar] [CrossRef]
- Franzmann, T.M.; Jahnel, M.; Pozniakovsky, A.; Mahamid, J.; Holehouse, A.S.; Nüske, E.; Richter, D.; Baumeister, W.; Grill, S.W.; Pappu, R.V.; et al. Phase separation of a yeast prion protein promotes cellular fitness. Science 2018, 359. [Google Scholar] [CrossRef]
- Derkatch, I.L.; Chernoff, Y.O.; Kushnirov, V.V.; Inge-Vechtomov, S.G.; Liebman, S.W. Genesis and variability of [PSI] prion factors in Saccharomyces cerevisiae. Genetics 1996, 144, 1375–1386. [Google Scholar]
- Kushnirov, V.V.; Vishnevskaya, A.B.; Alexandrov, I.M.; Ter-Avanesyan, M.D. Prion and nonprion amyloids: a comparison inspired by the yeast Sup35 protein. Prion 2007, 1, 179–184. [Google Scholar] [CrossRef]
- Kushnirov, V.V.; Ter-Avanesyan, M.D. Structure and replication of yeast prions. Cell 1998, 94, 13–16. [Google Scholar] [CrossRef]
- Kryndushkin, D.S.; Alexandrov, I.M.; Ter-Avanesyan, M.D.; Kushnirov, V.V. Yeast [PSI+] prion aggregates are formed by small Sup35 polymers fragmented by Hsp104. J. Biol. Chem. 2003, 278, 49636–49643. [Google Scholar] [CrossRef]
- Shkundina, I.S.; Kushnirov, V.V.; Tuite, M.F.; Ter-Avanesyan, M.D. The role of the N-terminal oligopeptide repeats of the yeast Sup35 prion protein in propagation and transmission of prion variants. Genetics 2006, 172, 827–835. [Google Scholar] [CrossRef]
- Kushnirov, V.V.; Kryndushkin, D.S.; Boguta, M.; Smirnov, V.N.; Ter-Avanesyan, M.D. Chaperones that cure yeast artificial [PSI+] and their prion-specific effects. Curr. Biol. 2000, 10, 1443–1446. [Google Scholar] [CrossRef]
- Chang, H.-Y.; Lin, J.-Y.; Lee, H.-C.; Wang, H.-L.; King, C.-Y. Strain-specific sequences required for yeast [PSI+] prion propagation. Proc. Natl. Acad. Sci. USA 2008, 105, 13345–13350. [Google Scholar] [CrossRef]
- Huang, Y.W.; Chang, Y.C.; Diaz-Avalos, R.; King, C.Y. W8, a new Sup35 prion strain, transmits distinctive information with a conserved assembly scheme. Prion 2015, 9, 207–227. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kochneva-pervukhova, N.V.; Chechenova, M.B.; Valouev, I.A.; Kushnirov, V.V.; Smirnov, V.N.; Ter-avanesyan, M.D. [PSI+] prion generation in yeast: characterization of the ‘strain’ difference. Yeast 2001, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, R.; Lindquist, S.L. Structural insights into a yeast prion illuminate nucleation and strain diversity. Nature 2005, 435, 765–772. [Google Scholar] [CrossRef]
- Gorkovskiy, A.; Thurber, K.R.; Tycko, R.; Wickner, R.B. Locating folds of the in-register parallel -sheet of the Sup35p prion domain infectious amyloid. Proc. Natl. Acad. Sci. USA 2014, 111, E4615–E4622. [Google Scholar] [CrossRef]
- Toyama, B.H.; Kelly, M.J.S.; Gross, J.D.; Weissman, J.S. The structural basis of yeast prion strain variants. Nature 2007, 449, 233–237. [Google Scholar] [CrossRef]
- Tanaka, M.; Chien, P.; Naber, N.; Cooke, R.; Weissman, J.S. Conformational variations in an infectious protein determine prion strain differences. Nature 2004, 428, 323–328. [Google Scholar] [CrossRef]
- Ohhashi, Y.; Yamaguchi, Y.; Kurahashi, H.; Kamatari, Y.O.; Sugiyama, S.; Uluca, B.; Piechatzek, T.; Komi, Y.; Shida, T.; Müller, H.; et al. Molecular basis for diversification of yeast prion strain conformation. Proc. Natl. Acad. Sci. USA 2018, 115, 2389–2394. [Google Scholar] [CrossRef]
- Keil, B. Specificity of Proteolysis; Springer Berlin Heidelberg: Berlin, Heidelberg, 1992; ISBN 978-3-642-48382-0. [Google Scholar]
- Polevoda, B.; Sherman, F. N-terminal acetyltransferases and sequence requirements for N-terminal acetylation of eukaryotic proteins. J. Mol. Biol. 2003, 325, 595–622. [Google Scholar] [CrossRef]
- Vishveshwara, N.; Bradley, M.E.; Liebman, S.W. Sequestration of essential proteins causes prion associated toxicity in yeast. Mol. Microbiol. 2009, 73, 1101–1114. [Google Scholar] [CrossRef]
- Zhao, X.; Rodriguez, R.; Silberman, R.E.; Ahearn, J.M.; Saidha, S.; Cummins, K.C.; Eisenberg, E.; Greene, L.E. Heat shock protein 104 (Hsp104)-mediated curing of [PSI+] yeast prions depends on both [PSI+] conformation and the properties of the Hsp104 homologs. J. Biol. Chem. 2017, 292, 8630–8641. [Google Scholar] [CrossRef]
- Stansfield, I.; Jones, K.M.; Kushnirov, V.V.; Dagkesamanskaya, A.R.; Poznyakovski, A.I.; Paushkin, S.V.; Nierras, C.R.; Cox, B.S.; Ter-Avanesyan, M.D.; Tuite, M.F. The products of the SUP45 (eRF1) and SUP35 genes interact to mediate translation termination in Saccharomyces cerevisiae. EMBO J. 1995, 14, 4365–4373. [Google Scholar] [CrossRef]
- Parham, S.N.; Resende, C.G.; Tuite, M.F. Oligopeptide repeats in the yeast protein Sup35p stabilize intermolecular prion interactions. EMBO J. 2001, 20, 2111–2119. [Google Scholar] [CrossRef]
- Valouev, I.A.; Kushnirov, V.V.; Ter-Avanesyan, M.D. Yeast polypeptide chain release factors eRF1 and eRF3 are involved in cytoskeleton organization and cell cycle regulation. Cell Motil. Cytoskelet. 2002, 52, 161–173. [Google Scholar] [CrossRef]
- Sant’Anna, R.; Fernández, M.R.; Batlle, C.; Navarro, S.; de Groot, N.S.; Serpell, L.; Ventura, S. Characterization of Amyloid Cores in Prion Domains. Sci. Rep. 2016, 6, 34274. [Google Scholar] [CrossRef]
- Bateman, D.A.; Wickner, R.B. The [PSI+] Prion Exists as a Dynamic Cloud of Variants. PLoS Genet. 2013, 9, 1–13. [Google Scholar] [CrossRef]
- Ghosh, R.; Dong, J.; Wall, J.; Frederick, K.K. Amyloid fibrils embodying distinctive yeast prion phenotypes exhibit diverse morphologies. FEMS Yeast Res. 2018, 18. [Google Scholar] [CrossRef]
- McGlinchey, R.P.; Kryndushkin, D.; Wickner, R.B. Suicidal [PSI+] is a lethal yeast prion. Proc. Natl. Acad. Sci. USA 2011, 108, 5337–5341. [Google Scholar] [CrossRef]
- Chan, P.H.W.; Lee, L.; Kim, E.; Hui, T.; Stoynov, N.; Nassar, R.; Moksa, M.; Cameron, D.M.; Hirst, M.; Gsponer, J.; et al. The [PSI+] yeast prion does not wildly affect proteome composition whereas selective pressure exerted on [PSI+] cells can promote aneuploidy. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef]
- Nizhnikov, A.A.; Ryzhova, T.A.; Volkov, K.V.; Zadorsky, S.P.; Sopova, J.V.; Inge-Vechtomov, S.G.; Galkin, A.P. Interaction of Prions Causes Heritable Traits in Saccharomyces cerevisiae. PLoS Genet. 2016, 12, e1006504. [Google Scholar] [CrossRef]
- Mathur, V.; Hong, J.Y.; Liebman, S.W. Ssa1 overexpression and [PIN(+)] variants cure [PSI(+)] by dilution of aggregates. J. Mol. Biol. 2009, 390, 155–167. [Google Scholar] [CrossRef][Green Version]
- Astor, M.T.; Kamiya, E.; Sporn, Z.A.; Berger, S.E.; Hines, J.K. Variant-specific and reciprocal Hsp40 functions in Hsp104-mediated prion elimination. Mol. Microbiol. 2018. [Google Scholar] [CrossRef]
- Yu, C.-I.; King, C.-Y. Forms and abundance of chaperone proteins influence yeast prion variant competition. Mol. Microbiol. 2019, 111, 798–810. [Google Scholar] [CrossRef]
- Stein, K.C.; True, H.L. Structural variants of yeast prions show conformer-specific requirements for chaperone activity. Mol. Microbiol. 2014, 93, 1156–1171. [Google Scholar] [CrossRef]
- Killian, A.N.; Miller, S.C.; Hines, J.K. Impact of Amyloid Polymorphism on Prion-Chaperone Interactions in Yeast. Viruses 2019, 11. [Google Scholar] [CrossRef]
- Wickner, R.B.; Son, M.; Edskes, H.K. Prion Variants of Yeast are Numerous, Mutable, and Segregate on Growth, Affecting Prion Pathogenesis, Transmission Barriers, and Sensitivity to Anti-Prion Systems. Viruses 2019, 11. [Google Scholar] [CrossRef]
- Huang, Y.-W.; King, C.-Y. (Institute of Molecular Biology, Academia Sinica, Taipei, Taiwan). Personal communication, 2018.
- Tanaka, M.; Collins, S.R.; Toyama, B.H.; Weissman, J.S. The physical basis of how prion conformations determine strain phenotypes. Nature 2006, 442, 585–589. [Google Scholar] [CrossRef]
- Depace, A.H.; Santoso, A.; Hillner, P.; Weissman, J.S. A Critical Role for Amino-Terminal Glutamine / Asparagine Repeats in the Formation and Propagation of a Yeast Prion. Cell 1998, 93, 1241–1252. [Google Scholar] [CrossRef]
- Kajava, A.V.; Baxa, U.; Wickner, R.B.; Steven, A.C. A model for Ure2p prion filaments and other amyloids: The parallel superpleated beta-structure. Proc. Natl. Acad. Sci. USA 2004, 101, 7885–7890. [Google Scholar] [CrossRef]
- Vázquez-Fernández, E.; Alonso, J.; Pastrana, M.A.; Ramos, A.; Stitz, L.; Vidal, E.; Dynin, I.; Petsch, B.; Silva, C.J.; Requena, J.R. Structural organization of mammalian prions as probed by limited proteolysis. PLoS ONE 2012, 7, e50111. [Google Scholar] [CrossRef]
- Alexandrov, A.I.; Polyanskaya, A.B.; Serpionov, G.V.; Ter-Avanesyan, M.D.; Kushnirov, V.V. The Effects of Amino Acid Composition of Glutamine-Rich Domains on Amyloid Formation and Fragmentation. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Derkatch, I.L.; Bradley, M.E.; Zhou, P.; Liebman, S.W. The PNM2 mutation in the prion protein domain of SUP35 has distinct effects on different variants of the [PSI+] prion in yeast. Curr. Genet. 1999, 35, 59–67. [Google Scholar] [CrossRef]
- King, C.Y. Supporting the structural basis of prion strains: Induction and identification of [PSI] variants. J. Mol. Biol. 2001, 307, 1247–1260. [Google Scholar] [CrossRef]
- Bailleul, P.A.; Newnam, G.P.; Steenbergen, J.N.; Chernoff, Y.O. Genetic Study of Interactions Between the Cytoskeletal Assembly Protein Sla1. Genetics 1999, 153, 81–94. [Google Scholar]
- Bradley, M.E.; Edskes, H.K.; Hong, J.Y.; Wickner, R.B.; Liebman, S.W. Interactions among prions and prion “strains” in yeast. Proc. Natl. Acad. Sci. USA 2002, 99 Suppl. 4, 16392–16399. [Google Scholar] [CrossRef]
- Dagkesamanskaya, A.R.; Ter-Avanesyan, M.D. Interaction of the yeast omnipotent suppressors SUP1(SUP45) and SUP2(SUP35) with non-mendelian factors. Genetics 1991, 128, 513–520. [Google Scholar]
- Gari, E.; Piedrafita, L.; Aldea, M.; Herrero, E. A set of vectors with a tetracycline-regulatable promoter system for modulating gene expression inSaccharomyces cerevisiae. Yeast 1997, 13, 837–848. [Google Scholar] [CrossRef]
- Kushnirov, V.V.; Ter-Avanesyan, M.D.; Didichenko, S.A.; Smirnov, V.N.; Chernoff, Y.O.; Derkach, I.L.; Novikova, O.N.; Inge-Vechtomov, S.G.; Neistat, M.A.; Tolstorukov, I.I. Divergence and conservation of SUP2 (SUP35) gene of yeast Pichia pinus and Saccharomyces cerevisiae. Yeast 1990, 6, 461–472. [Google Scholar] [CrossRef]
- Urakov, V.N.; Mitkevich, O.V.; Safenkova, I.V.; Ter-Avanesyan, M.D. Ribosome-bound Pub1 modulates stop codon decoding during translation termination in yeast. FEBS J. 2017, 284, 1914–1930. [Google Scholar] [CrossRef]
- Keeling, K.M.; Lanier, J.; Du, M.; Salas-Marco, J.; Gao, L.; Kaenjak-Angeletti, A.; Bedwell, D.M. Leaky termination at premature stop codons antagonizes nonsense-mediated mRNA decay in S. cerevisiae. RNA 2004, 10, 691–703. [Google Scholar] [CrossRef]
| Weak [PSI+] | Strong [PSI+] | ||||
|---|---|---|---|---|---|
| Name | Core 2 | Core 3 | Name | Core 2 | Core 3 |
| WS2 | 96,101–120 * | - | S-7 | 91–104 | - |
| W2 | - | - | S-7-3 | 81–100 * | 125–148 |
| W3 | 91–120 | - | S-7-4 | 82,92–100,113 * | - |
| W-L2 | (91,96,101)–123 | 133–153* | S1 | 82–100,92–113 | - |
| W-T7 | 81–100,92–120 | 133–149 | S1-R4 | 72,82–100 * | - |
| W-Pb | 91–121 | - | S-L4 | 72,82–100 * | - |
| W-R4b | (92,96)–(104,113,121) | 133–148 | S-L6 | - | - |
| W-HT2889 | 91–122 | - | S-L20 | 82–100 | 91–144*† |
| W-1111 | 91,96–120 | - | S-Pb | 92–115 | - |
| W-1112 | 91–121 | - | S-R4b | 91–113 | - |
| W-1112-R14a, R14b | 91–121 | - | S-Cl1 | 82,92–100,113 * | - |
| W-VK | 96–(113,120) | - | S-Cl2 | - | - |
| W-VL | - | - | S-VH0 | - | 125–148 |
| S-1109 | 82–100 * | - | |||
| Amyloid | S-VH | 82–100 | - | ||
| Δ2-30 | 81–113 | - | S-W8 | 91–(104,113) | - |
| S-W8-R14a | 91–121 | (125,133)–148 | |||
| S-W8-R14b | 91–121 | - | |||
| Variant | Original Name/Comment | Source/Reference |
|---|---|---|
| Yeast Strain 74-D694 | ||
| [psi−][PIN+] | OT60, parent for nontoxic [PSI+] obtained in this work | Y. Chernoff [50] |
| [psi−][PIN+]very high | Parent for the lethal and toxic isolates | S. Liebman [51] |
| WS2 | This work | |
| W2 | This work | |
| W3 | This work | |
| W-L2 | This work | |
| W-Pb | This work | |
| W-T7 | This work | |
| W-R4 | This work | |
| S-7 | 7-74-D694, also known as OT56* | Y. Chernoff [50] |
| S1 | This work | |
| S1-R4 | This work | |
| S-L4 | This work | |
| S-L6 | This work | |
| S-L20 | This work | |
| S-Pb | This work | |
| S-R4b | This work | |
| S-Cl1 | This work | |
| S-Cl2 | This work | |
| W-HT2889 | Heather True collection #2889 | H. True |
| Yeast Strain 5V-H19 | ||
| S-VH0† | Our collection; [PSI+] originates from Albert Hinnen strain AH216 | [52] |
| S-1109 | Our collection #1109 | |
| W-1111 | Our collection #1111 | |
| W-1112 | Our collection #1112 | |
| S-VH† | VH | C.Y. King [15] |
| S-W8 | W8 | C.Y. King [16] |
| W-VK | VK | C.Y. King [15] |
| W-VL | VL | C.Y. King [15] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dergalev, A.A.; Alexandrov, A.I.; Ivannikov, R.I.; Ter-Avanesyan, M.D.; Kushnirov, V.V. Yeast Sup35 Prion Structure: Two Types, Four Parts, Many Variants. Int. J. Mol. Sci. 2019, 20, 2633. https://doi.org/10.3390/ijms20112633
Dergalev AA, Alexandrov AI, Ivannikov RI, Ter-Avanesyan MD, Kushnirov VV. Yeast Sup35 Prion Structure: Two Types, Four Parts, Many Variants. International Journal of Molecular Sciences. 2019; 20(11):2633. https://doi.org/10.3390/ijms20112633
Chicago/Turabian StyleDergalev, Alexander A., Alexander I. Alexandrov, Roman I. Ivannikov, Michael D. Ter-Avanesyan, and Vitaly V. Kushnirov. 2019. "Yeast Sup35 Prion Structure: Two Types, Four Parts, Many Variants" International Journal of Molecular Sciences 20, no. 11: 2633. https://doi.org/10.3390/ijms20112633
APA StyleDergalev, A. A., Alexandrov, A. I., Ivannikov, R. I., Ter-Avanesyan, M. D., & Kushnirov, V. V. (2019). Yeast Sup35 Prion Structure: Two Types, Four Parts, Many Variants. International Journal of Molecular Sciences, 20(11), 2633. https://doi.org/10.3390/ijms20112633







