“Fifty Shades” of Black and Red or How Carboxyl Groups Fine Tune Eumelanin and Pheomelanin Properties
Abstract
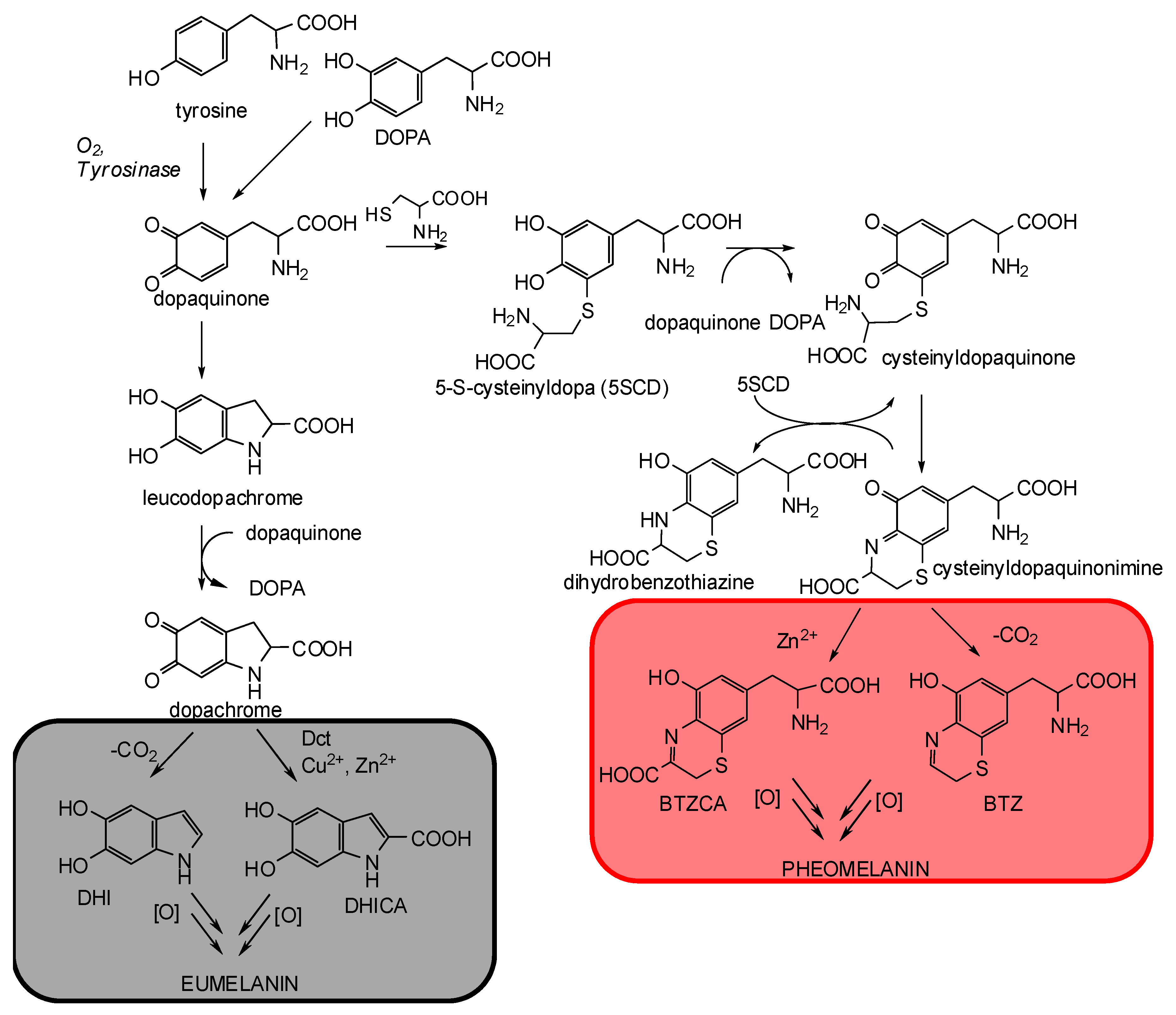
:1. The Control Mechanisms of Carboxyl Groups in Melanogenesis
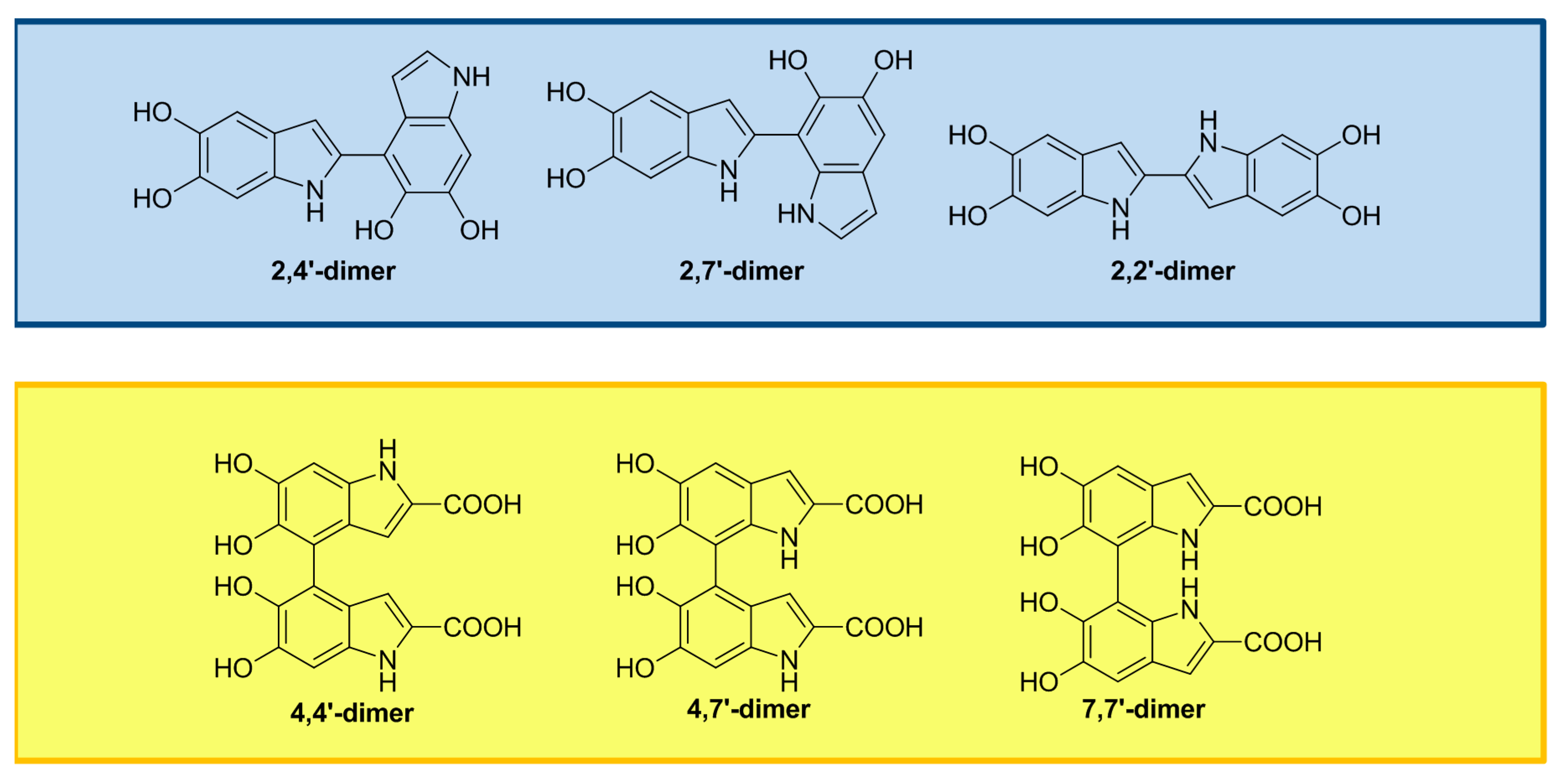
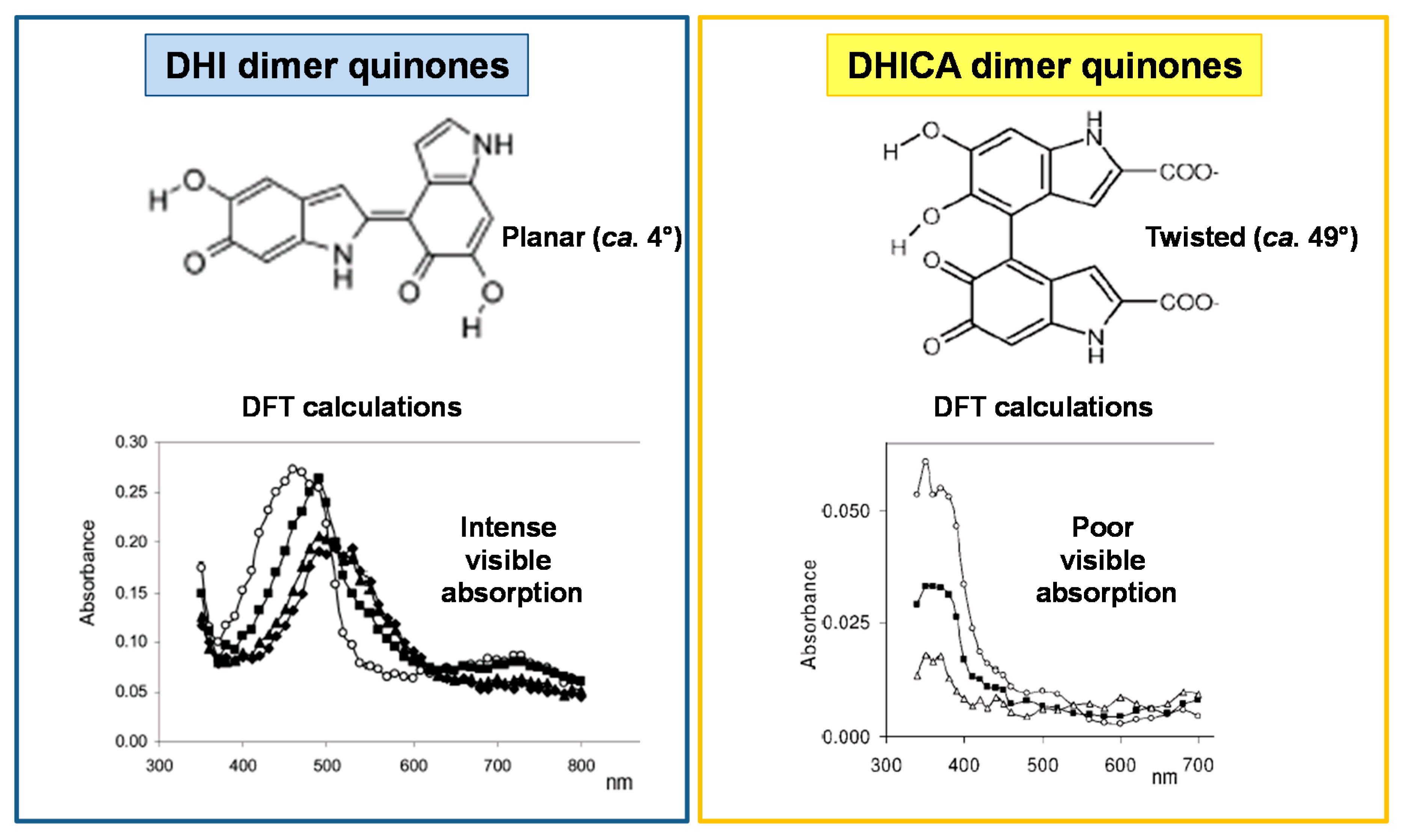
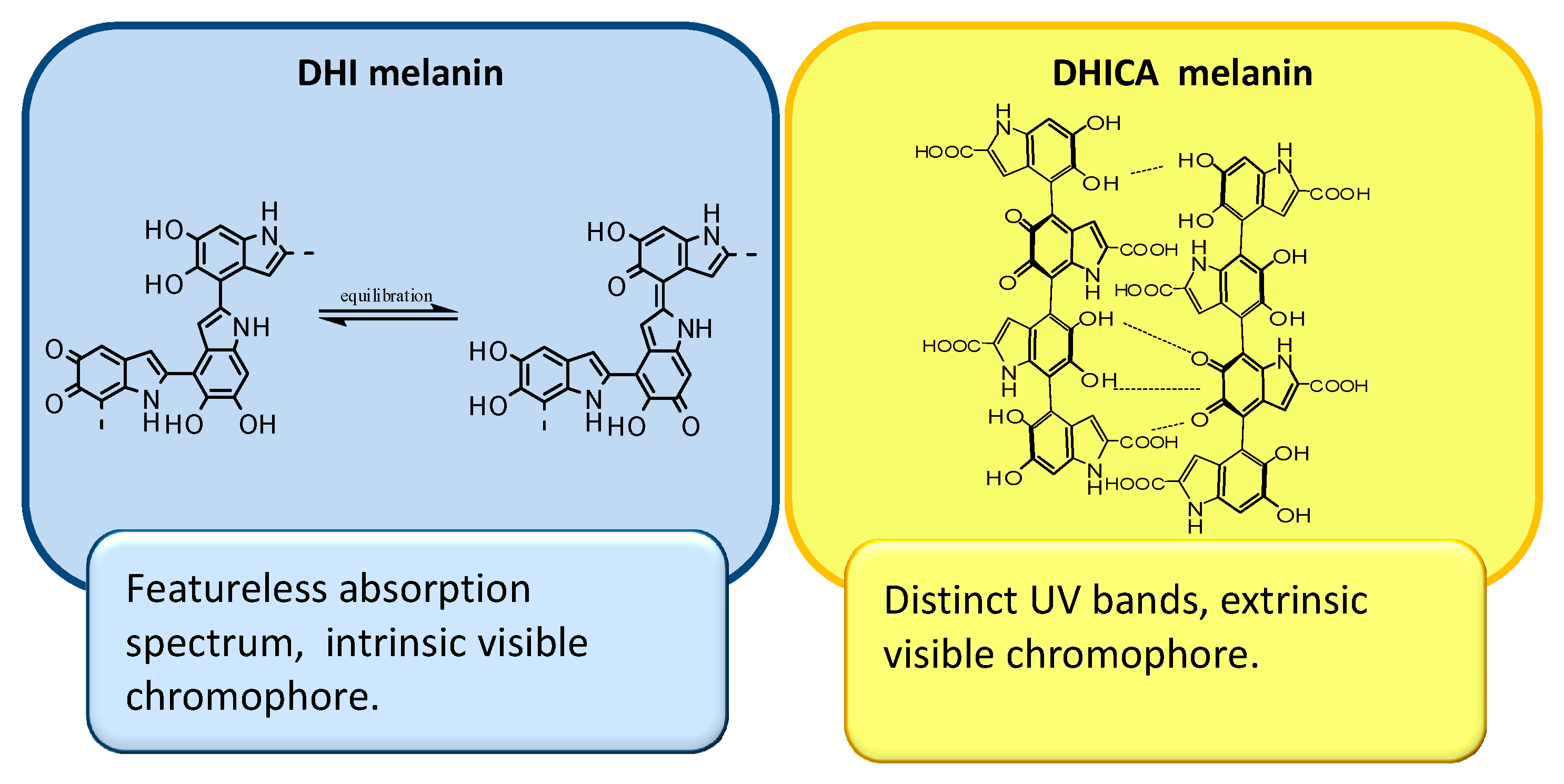
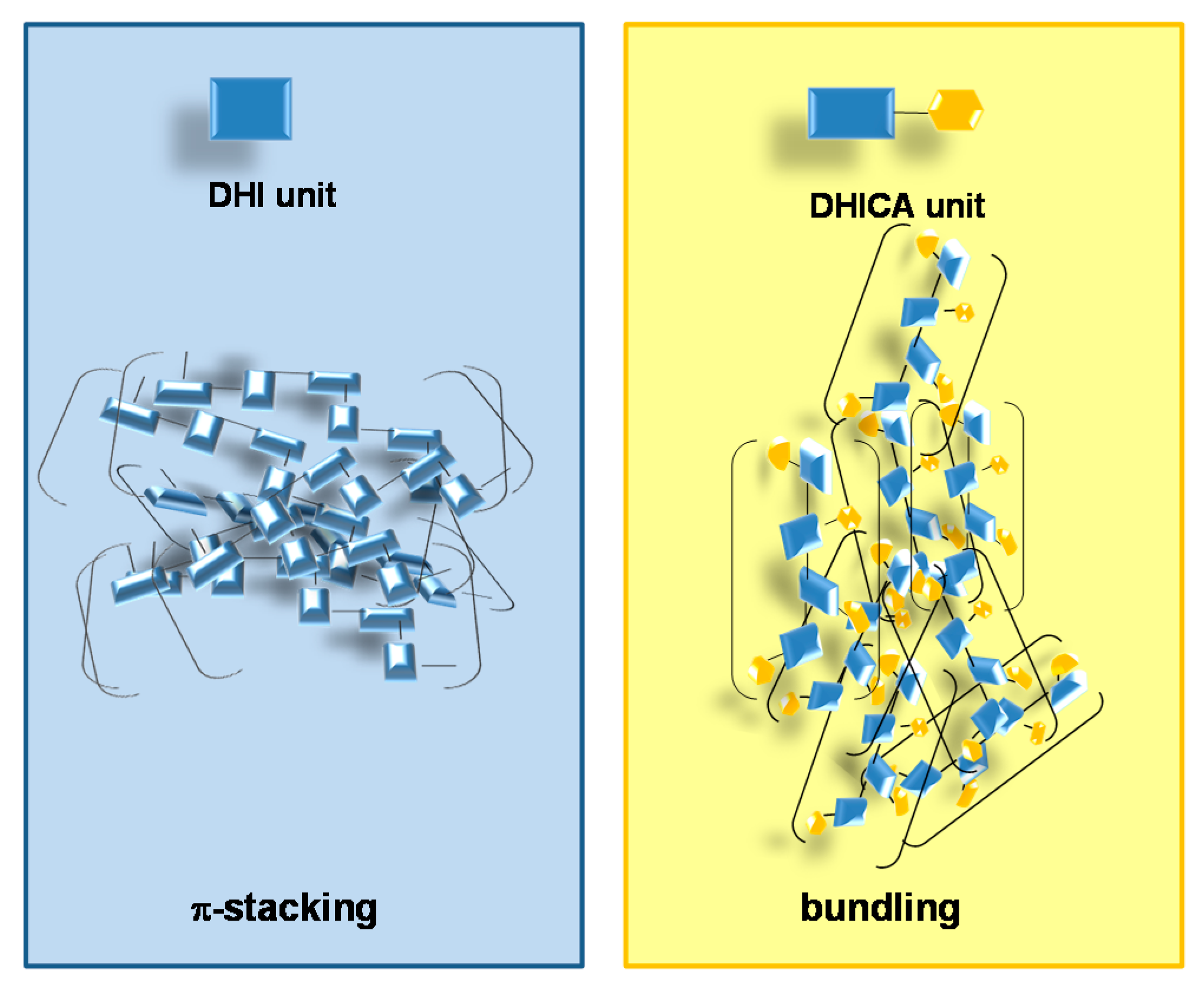
2. The Impact of Carboxylated Units on Eumelanin Structure and Properties
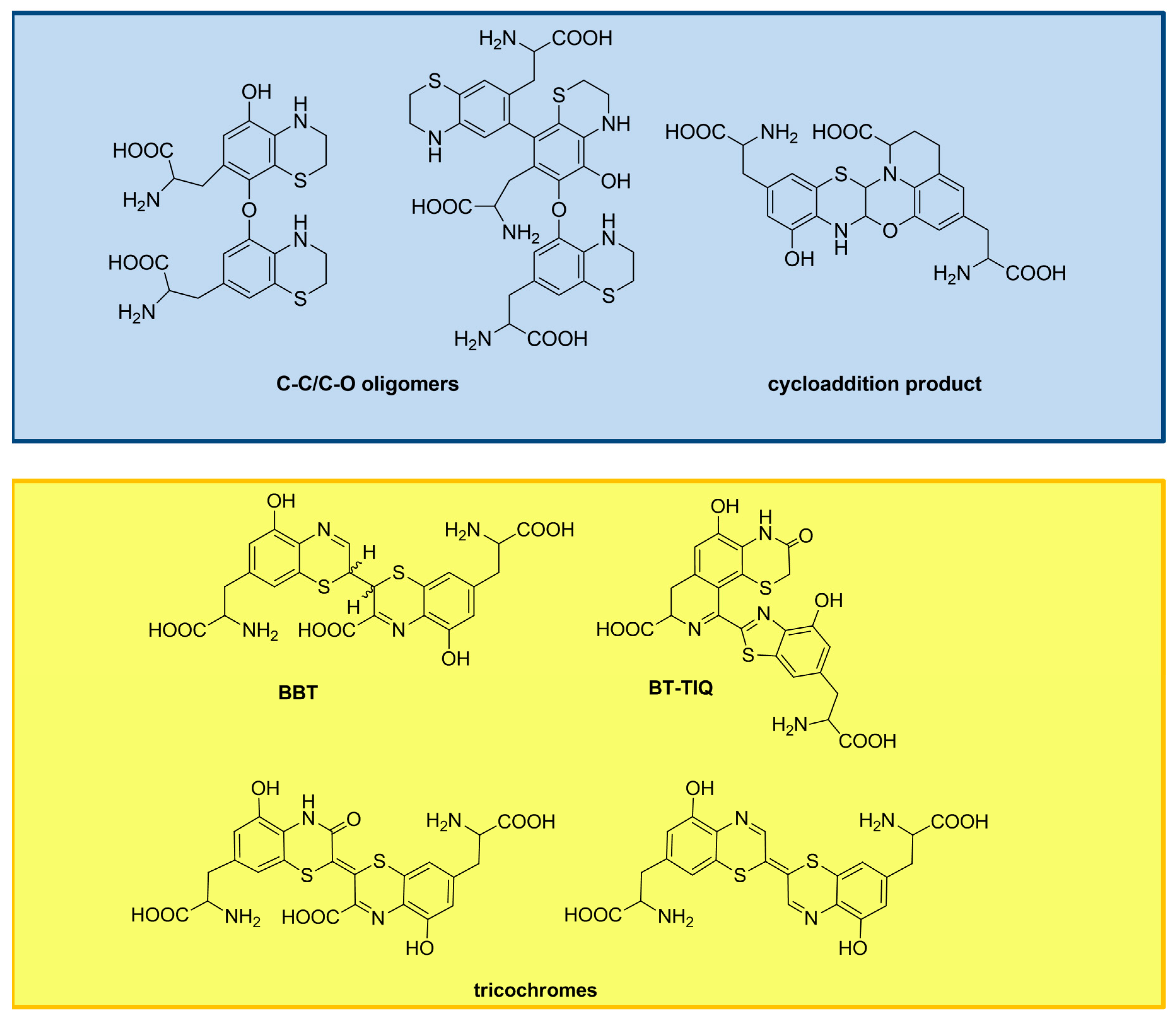
3. The Impact of Carboxylated Units on Pheomelanin Structure and Properties
4. Conclusions
Conflicts of Interest
Abbreviations
| DHI | 5,6-dihydroxyindole |
| DHICA | 5,6-dihydroxyindole-2-carboxylic acid |
| BTZ | 1,4-benzothiazine |
| BTZCA | 1,4-benzothiazine carboxylic acid |
| UVA | ultraviolet A |
| ROS | reactive oxygen species |
| MC1R | melanocortin-1-receptor |
| α-MSH | α-melanocyte stimulating hormone |
| ASIP | Agouti signal protein |
| cAMP | cyclic adenosine 3’,5’-monophosphate |
| Tyrp1 | tyrosinase-related protein 1 |
| Dct | dopachrome tautomerase |
| 5SCD | 5-S-cysteinyldopa |
| 2SCD | 2-S-cysteinyldopa |
| PVA | polyvinyl alcohol |
| EPR | electron paramagnetic resonance |
| DPPH | 1,1-diphenyl-2-picrylhydrazyl |
| ABTS | 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) |
| NO | nitric oxide |
| BBT | 2,2’-bibenzothiazines |
| BT-TIQ | benzothiazolylthiazinodihydroisoquinoline |
| GSH | glutathione |
References
- Prota, G. The chemistry of melanins and melanogenesis. In Fortschritte der Chemie Organischer Naturstoffe/Progress in the Chemistry of Organic Natural Products; Herz, W., Kirby, G.W., Moore, R.E., Steglich, W., Tamm, C., Eds.; Springer-Verlag: Wien, Austria, 1995; Volume 64, pp. 93–148. [Google Scholar]
- Simon, J.D.; Peles, D.; Wakamatsu, K.; Ito, S. Current challenges in understanding melanogenesis: Bridging chemistry, biological control, morphology, and function. Pigment Cell Melanoma Res. 2009, 22, 563–579. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.D.; Peles, D.N. The red and the black. Acc. Chem. Res. 2010, 43, 1452–1460. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Wakamatsu, K.; d’Ischia, M.; Napolitano, A.; Pezzella, A. Structure of melanins. In Melanins and Melanosomes; Borovanský, J., Riley, P.A., Eds.; Wiley-VCH: Weinheim, Germany, 2011; pp. 167–185. [Google Scholar]
- Corani, A.; Huijser, A.; Gustavsson, T.; Markovitsi, D.; Malmqvist, P.Å.; Pezzella, A.; d’Ischia, M.; Sundström, V. Superior photoprotective motifs and mechanisms in eumelanins uncovered. J. Am. Chem. Soc. 2014, 136, 11626–11635. [Google Scholar] [CrossRef] [PubMed]
- Olsen, C.M.; Carroll, H.J.; Whiteman, D.C. Estimating the attributable fraction for melanoma: A meta-analysis of pigmentary characteristics and freckling. Int. J. Cancer 2010, 127, 2430–2445. [Google Scholar] [CrossRef] [PubMed]
- Sarna, T.; Menon, I.A.; Sealy, R.C. Photosensitization of melanins: A comparative study. Photochem. Photobiol. 1985, 42, 529–532. [Google Scholar] [CrossRef] [PubMed]
- Le Pape, E.; Wakamatsu, K.; Ito, S.; Wolber, R.; Hearing, V.J. Regulation of eumelanin/pheomelanin synthesis and visible pigmentation in melanocytes by ligands of the melanocortin 1 receptor. Pigment Cell Melanoma Res. 2008, 21, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Candille, S.I.; Kaelin, C.B.; Cattanach, B.M.; Yu, B.; Thompson, D.A.; Nix, M.A.; Kerns, J.A.; Schmutz, S.M.; Millhauser, G.L.; Barsh, G.S. A β-defensin mutationcauses black coat color in domestic dogs. Science 2007, 318, 1418–1423. [Google Scholar] [CrossRef] [PubMed]
- Dessinioti, C.; Antoniou, C.; Katsambas, A.; Stratigos, A.J. Melanocortin 1 receptor variants: Functional role and pigmentary associations. Photochem. Photobiol. 2011, 87, 978–987. [Google Scholar] [CrossRef] [PubMed]
- García-Borrón, J.C.; Abdel-Malek, Z.; Jiménez-Cervantes, C. MC1R, the cAMP pathway, and the response to solar UV: Extending the horizon beyond pigmentation. Pigment Cell Melanoma Res. 2014, 27, 699–720. [Google Scholar] [CrossRef] [PubMed]
- Olivares, C.; Solano, F. New insights into the active site structure and catalytic mechanism of tyrosinase and its related proteins. Pigment Cell Melanoma Res. 2009, 22, 750–760. [Google Scholar] [CrossRef] [PubMed]
- Ramsden, C.A.; Riley, P.A. Tyrosinase: The four oxidation states of the active site and their relevance to enzymatic activation, oxidation and inactivation. Bioorg. Med. Chem. 2014, 15, 2388–2395. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; IFPCS. The IFPCS presidential lecture: A chemist’s view of melanogenesis. Pigment Cell Res. 2003, 16, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Panzella, L.; Napolitano, A.; d’Ischia, M. Is DHICA the key to dopachrome tautomerase and melanocyte functions? Pigment Cell Melanoma Res. 2011, 24, 248–249. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, A.; d’Ischia, M.; Misuraca, G.; Prota, G. Effect of metal ions on the rearrangement of dopachrome. Biochim. Biophys. Acta 1987, 925, 203–209. [Google Scholar] [CrossRef]
- Palumbo, A.; d’Ischia, M.; Misuraca, G.; Prota, G.; Schultz, T.M. Structural modifications in biosynthetic melanins induced by metal ions. Biochim. Biophys. Acta 1988, 964, 193–199. [Google Scholar] [CrossRef]
- Napolitano, A.; Chioccara, F.; Prota, G. A re-examination of the zinc-catalysed rearrangement of dopachrome using immobilised tyrosinase. Gazz. Chim. Ital. 1985, 115, 357–359. [Google Scholar]
- Valverde, P.; Healy, E.; Jackson, I.; Rees, J.L.; Thody, A.J. Variants of the melanocyte−stimulating hormone receptor gene are associated with red hair and fair skin in humans. Nat. Genet. 1995, 11, 328–330. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; Panzella, L.; Leone, L.; d’Ischia, M. Red hair benzothiazines and benzothiazoles: Mutation-inspired chemistry in the quest for functionality. Acc. Chem. Res. 2013, 46, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Di Donato, P.; Napolitano, A. 1,4-Benzothiazines as key intermediates in the biosynthesis of red hair pigment pheomelanins. Pigment Cell Res. 2003, 16, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; Di Donato, P.; Prota, G.; Land, E.J. Transient quinonimines and 1,4-benzothiazines of pheomelanogenesis: New pulse radiolytic and spectrophotometric evidence. Free Radic. Biol. Med. 1999, 27, 521–528. [Google Scholar] [CrossRef]
- Napolitano, A.; Di Donato, P.; Prota, G. Zinc-catalyzed oxidation of 5-S-cysteinyldopa to 2,2’-bi(2H-1,4-benzothiazine): Tracking the biosynthetic pathway of trichochromes, the characteristic pigments of red hair. J. Org. Chem. 2001, 66, 6958–6966. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; De Lucia, M.; Panzella, L.; d’Ischia, M. The “Benzothiazine” chromophore of pheomelanins: A reassessment. Photochem. Photobiol. 2008, 84, 593–599. [Google Scholar] [CrossRef] [PubMed]
- D’Ischia, M.; Napolitano, A.; Pezzella, A.; Land, E.J.; Ramsden, C.A.; Riley, P.A. 5,6-Dihydroxyindoles and indole-5,6-diones. Adv. Heterocycl. Chem. 2005, 89, 1–63. [Google Scholar]
- D’Ischia, M.; Napolitano, A.; Pezzella, A. 5,6-Dihydroxyindole chemistry: Unexplored opportunities beyond eumelanin. Eur. J. Org. Chem. 2011, 28, 5501–5516. [Google Scholar] [CrossRef]
- Panzella, L.; Pezzella, A.; Napolitano, A.; d’Ischia, M. The first 5,6-dihydroxyindole tetramer by oxidation of 5,5’,6,6’-tetrahydroxy- 2,4’-biindolyl and an unexpected issue of positional reactivity en route to eumelanin-related polymers. Org. Lett. 2007, 72, 9225–9230. [Google Scholar] [CrossRef] [PubMed]
- Pezzella, A.; Panzella, L.; Natangelo, A.; Arzillo, M.; Napolitano, A.; d’Ischia, M. 5,6-dihydroxyindole tetramers with “anomalous” interunit bonding patterns by oxidative coupling of 5,5’,6,6’-tetrahydroxy-2,7’-biindolyl: Emerging complexities on the way toward an improved model of eumelanin buildup. J. Org. Chem. 2007, 72, 9225–9230. [Google Scholar] [CrossRef] [PubMed]
- Corradini, M.G.; Napolitano, A.; Prota, G. A biosynthetic approach to the structure of eumelanins. The isolation of oligomers from 5,6-dihydroxy-1-methylindole. Tetrahedron 1986, 42, 2083–2088. [Google Scholar] [CrossRef]
- Pezzella, A.; Napolitano, A.; d’Ischia, M.; Prota, G. Oxidative polymerisation of 5,6-dihydroxyindole-2-carboxylic acid to melanin: A new insight. Tetrahedron 1996, 52, 7913–7920. [Google Scholar] [CrossRef]
- Pezzella, A.; Vogna, D.; Prota, G. Atropoisomeric melanin intermediates by oxidation of the melanogenic precursor 5,6-dihydroxyindole-2-carboxylic acid under biomimetic conditions. Tetrahedron 2002, 58, 3681–3687. [Google Scholar] [CrossRef]
- Pezzella, A.; Vogna, D.; Prota, G. Synthesis of optically active tetrameric melanin intermediates by oxidation of the melanogenic precursor 5,6-dihydroxyindole-2-carboxylic acid under biomimetic conditions. Tetrahedron Asymmetry 2003, 14, 1133–1140. [Google Scholar] [CrossRef]
- Pezzella, A.; Panzella, L.; Crescenzi, O.; Napolitano, A.; Navaratnam, S.; Edge, R.; Land, E.J.; Barone, V.; d’Ischia, M. Short-lived quinonoid species from 5,6-dihydroxyindole dimers en route to eumelanin polymers: Integrated chemical, pulse radiolytic, and quantum mechanical investigation. J. Am. Chem. Soc. 2006, 128, 15490–15498. [Google Scholar] [CrossRef] [PubMed]
- Pezzella, A.; Panzella, L.; Crescenzi, O.; Napolitano, A.; Navaratnam, S.; Edge, R.; Land, E.J.; Barone, V.; d’Ischia, M. Lack of visible chromophore development in the pulse radiolysis oxidation of 5,6-dihydroxyindole-2-carboxylic acid oligomers: DFT investigation and implications for eumelanin absorption properties. J. Org. Chem. 2009, 74, 3727–3734. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Chuang, C.; Cao, J.; Ball, V.; Ruch, D.; Buehler, M.J. Excitonic effects from geometric order and disorder explain broadband optical absorption in eumelanin. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Panzella, L.; Gentile, G.; D’Errico, G.; Della Vecchia, N.F.; Errico, M.E.; Napolitano, A.; Carfagna, C.; d’Ischia, M. Atypical structural and π-electron features in the melanin polymer from the major human melanogen underpin superior free radical scavenger properties. Angew. Chem. Int. Ed. 2013, 52, 12684–12687. [Google Scholar] [CrossRef] [PubMed]
- Pezzella, A.; Iadonisi, A.; Valerio, S.; Panzella, L.; Napolitano, A.; Adinolfi, M.; d’Ischia, M. Disentangling eumelanin “black chromophore”: Visible absorption changes as signatures of oxidation state- and aggregation-dependent dynamic interactions in a model water-soluble 5,6-dihydroxyindole polymer. J. Am. Chem. Soc. 2009, 131, 15270–15275. [Google Scholar] [CrossRef] [PubMed]
- Ascione, L.; Pezzella, A.; Ambrogi, V.; Carfagna, C.; d’Ischia, M. Intermolecular π-electron perturbations generate extrinsic visible contributions to eumelanin black chromophore in model polymers with interrupted interring conjugation. Photochem. Photobiol. 2013, 89, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Arzillo, M.; Mangiapia, G.; Pezzella, A.; Heenan, R.K.; Radulescu, A.; Paduano, L.; d’Ischia, M. Eumelanin buildup on the nanoscale: Aggregate growth/assembly and visible absorption development in biomimetic 5,6-dihydroxyindole polymerization. Biomacromolecules 2012, 13, 2379–2390. [Google Scholar] [CrossRef] [PubMed]
- Meredith, P.; Sarna, T. The physical and chemical properties of eumelanin. Pigment Cell Res. 2006, 19, 572–594. [Google Scholar] [CrossRef] [PubMed]
- Mostert, A.B.; Hanson, G.R.; Sarna, T.; Gentle, I.R.; Powell, B.J.; Meredith, P. Hydration-controlled X-band EPR spectroscopy: A tool for unravelling the complexities of the solid-state free radical in eumelanin. J. Phys. Chem. B 2013, 117, 4965–4972. [Google Scholar] [CrossRef] [PubMed]
- Rienecker, S.B.; Mostert, A.B.; Schenk, G.; Hanson, G.R.; Meredith, P. Heavy water as a probe of the free radical nature and electrical conductivity of melanin. J. Phys. Chem. B 2015, 119, 14994–15000. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Liu, X.M.; Dai, X.; Zhou, Q.; Lei, T.C.; Beermann, F.; Wakamatsu, K.; Xu, S.Z. Regulation of DHICA-mediated antioxidation by dopachrome tautomerase: Implication for skin photoprotection against UVA radiation. Free Radic. Biol. Med. 2010, 48, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; Memoli, S.; Crescenzi, O.; Prota, G. Oxidative polymerization of the pheomelanin precursor 5-hydroxy-1,4-benzothiazinylalanine: A new hint to the pigment structure. J. Org. Chem. 1996, 61, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Costantini, C.; Crescenzi, O.; Prota, G.; Palumbo, A. New intermediates of phaeomelanogenesis in vitro beyond the 1,4-benzothiazine stage. Tetrahedron 1990, 46, 6831–6838. [Google Scholar] [CrossRef]
- Thomson, R.H. The pigments of reddish hair and feathers. Angew. Chem. Int. Ed. 1974, 13, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Greco, G.; Panzella, L.; Verotta, L.; d’Ischia, M.; Napolitano, A. Uncovering the structure of human red hair pheomelanin: Benzothiazolylthiazinodihydroisoquinolines as key building blocks. J. Nat. Prod. 2011, 74, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Greco, G.; Panzella, L.; Napolitano, A.; d’Ischia, M. The fundamental building blocks of red human hair pheomelanin are isoquinoline-containing dimers. Pigment Cell Melanoma Res. 2012, 25, 110–112. [Google Scholar] [CrossRef] [PubMed]
- Thureau, P.; Ziarelli, F.; Thévand, A.; Martin, R.W.; Farmer, P.J.; Viel, S.; Mollica, G. Probing the motional behavior of eumelanin and pheomelanin with solid-state NMR spectroscopy: New insights into the pigment properties. Chem. Eur. J. 2012, 18, 10689–10700. [Google Scholar] [CrossRef] [PubMed]
- Chedekel, M.R.; Smith, S.K.; Post, P.W.; Pokora, A.; Vessel, D.L. Photodestruction of pheomelanin: Role of oxygen. Proc. Natl. Acad. Sci. USA 1978, 75, 5395–5399. [Google Scholar] [CrossRef] [PubMed]
- Felix, C.C.; Hyde, J.S.; Sarna, T.; Sealy, R.C. Melanin photoreactions in aerated media: Electron spin resonance evidence for production of superoxide and hydrogen peroxide. Biochem. Biophys. Res. Commun. 1978, 84, 335–341. [Google Scholar] [CrossRef]
- Panzella, L.; Szewczyk, G.; d’Ischia, M.; Napolitano, A.; Sarna, T. Zinc-induced structural effects enhance oxygen consumption and superoxide generation in synthetic pheomelanins on UVA/visible light irradiation. Photochem. Photobiol. 2010, 86, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Rouzaud, F.; Kadekaro, A.L.; Abdel-Malek, Z.A.; Hearing, V.J. MC1R and the response of melanocytes to ultraviolet radiation. Mutat. Res. 2005, 571, 133–152. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, S.; Zhang, W.; Wakamatsu, K.; Ito, S.; Hearing, V.J.; Kraemer, K.H.; Brash, D.E. Melanin acts as a potent UVB photosensitizer to cause an atypical mode of cell death in murine skin. Proc. Natl. Acad. Sci. USA 2004, 101, 15076–15081. [Google Scholar] [CrossRef] [PubMed]
- Wenczl, E.; Van der Schans, G.P.; Roza, L.; Kolb, R.M.; Timmerman, A.J.; Smit, N.P.; Pavel, S.; Schothorst, A.A. (Pheo)melanin photosensitizes UVA-induced DNA damage in cultured human melanocytes. J. Investig. Dermatol. 1998, 111, 678–682. [Google Scholar] [CrossRef] [PubMed]
- Bennett, D.C. Ultraviolet wavebands and melanoma initiation. Pigment Cell Melanoma Res. 2008, 21, 520–524. [Google Scholar] [CrossRef] [PubMed]
- Ye, T.; Simon, J.D. Ultrafast spectroscopic study of pheomelanin: Implications on the mechanism of superoxide anion formation. J. Phys. Chem. B 2002, 106, 6133–6135. [Google Scholar] [CrossRef]
- Ye, T.; Hong, L.; Garguilo, J.; Pawlak, A.; Edwards, G.S.; Nemanich, R.J.; Sarna, T.; Simon, J.D. Photoionization thresholds of melanins obtained from free electron laser-photoelectron emission microscopy, femtosecond transient absorption spectroscopy and electron paramagnetic resonance measurements of oxygen photoconsumption. Photochem. Photobiol. 2006, 82, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Chedekel, M.R.; Agin, P.P.; Sayre, R.M. Photochemistry of pheomelanin: Action spectrum for superoxide production. Photochem. Photobiol. 1980, 31, 553–555. [Google Scholar] [CrossRef]
- Korytowski, W.; Kalyanaraman, B.; Menon, I.A.; Sarna, T.; Sealy, R.C. Reaction of superoxide anions with melanins: Electron spin resonance and spin trapping studies. Biochim. Biophys. Acta 1986, 882, 145–153. [Google Scholar] [CrossRef]
- Ou-Yang, H.; Stamatas, G.; Kollias, N. Spectral responses of melanin to ultraviolet A irradiation. J. Investig. Dermatol. 2004, 122, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Ye, T.; Simon, J.D. The action spectrum for generation of the primary intermediate revealed by ultrafast absorption spectroscopy studies of pheomelanin. Photochem. Photobiol. 2003, 77, 41–45. [Google Scholar] [CrossRef]
- Ye, T.; Pawlak, A.; Sarna, T.; Simon, J.D. Different molecular constituents in pheomelanin are responsible for emission, transient absorption and oxygen photoconsumption. Photochem. Photobiol. 2008, 84, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Ye, T.; Lamb, L.E.; Wakamatsu, K.; Ito, S.; Simon, J.D. Ultrafast absorption and photothermal studies of decarboxytrichochrome C in solution. Photochem. Photobiol. Sci. 2003, 2, 821–823. [Google Scholar] [CrossRef] [PubMed]
- Mitra, D.; Luo, X.; Morgan, A.; Wang, J.; Hoang, M.P.; Lo, J.; Guerrero, C.R.; Lennerz, J.K.; Mihm, M.C.; Wargo, J.A.; et al. An ultraviolet-radiation-independent pathway to melanoma carcinogenesis in the red hair/fair skin background. Nature 2012, 491, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Wendt, J.; Rauscher, S.; Burgstaller-Muehlbacher, S.; Fae, I.; Fischer, G.; Phehamberger, H.; Okamoto, I. Human determinants and the role of melanocortin 1 receptor variants in melanoma risk independent of UV radiation exposure. JAMA Dermatol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Premi, S.; Wallisch, S.; Mano, C.M.; Weiner, A.B.; Bacchiocchi, A.; Wakamatsu, K.; Bechara, E.J.; Halaban, R.; Douki, T.; Brash, D.E. Photochemistry. Chemiexcitation of melanin derivatives induces DNA photoproducts long after UV exposure. Science 2015, 347, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Panzella, L.; Leone, L.; Greco, G.; Vitiello, G.; D’Errico, G.; Napolitano, A.; d’Ischia, M. Red human hair pheomelanin is a potent pro-oxidant mediating UV-independent contributory mechanisms of melanomagenesis. Pigment Cell Melanoma Res. 2014, 27, 244–252. [Google Scholar] [CrossRef] [PubMed]
- D’Ischia, M.; Wakamatsu, K.; Napolitano, A.; Briganti, S.; Garcia-Borron, J.C.; Kovacs, D.; Meredith, P.; Pezzella, A.; Picardo, M.; Sarna, T.; et al. Melanins and melanogenesis: Methods, standards, protocols. Pigment Cell Melanoma Res. 2013, 26, 616–633. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Panzella, L.; Micillo, R.; Bentley, W.E.; Napolitano, A.; Payne, G.F. Reverse engineering applied to red human hair pheomelanin reveals redox-buffering as a pro-oxidant mechanism. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; Panzella, L.; Monfrecola, G.; d’Ischia, M. Pheomelanin-induced oxidative stress: Bright and dark chemistry bridging red hair phenotype and melanoma. Pigment Cell Melanoma Res. 2014, 27, 721–733. [Google Scholar] [CrossRef] [PubMed]
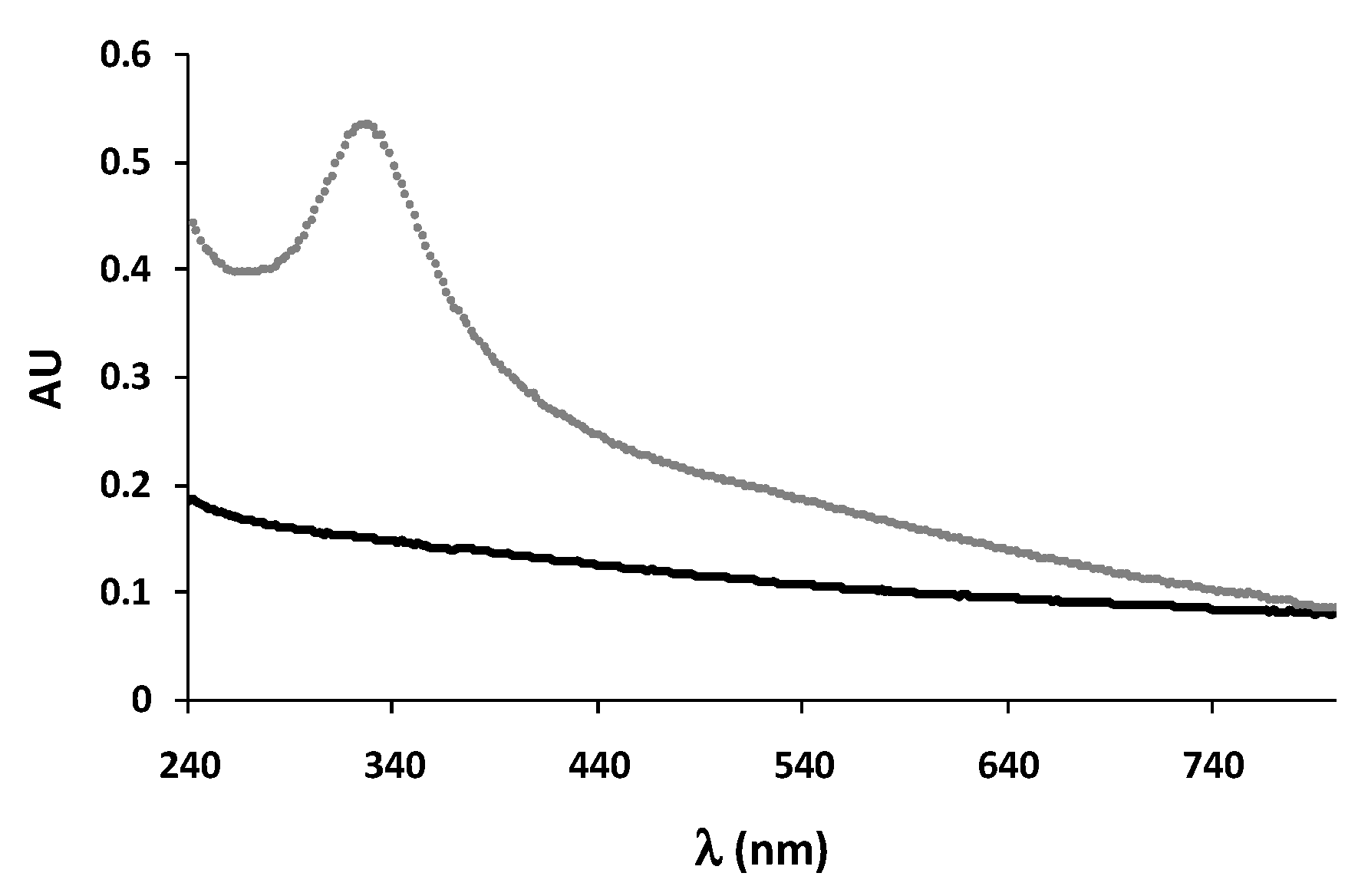

| Melanin | DPPH (%) | ABTS (%) | NO (%) |
|---|---|---|---|
| DHICA melanin | 100 ± 4 | 46 ± 2 | 64 ± 3 |
| DHI melanin | 29 ± 1 | 4 ± 1 | 26 ± 1 |
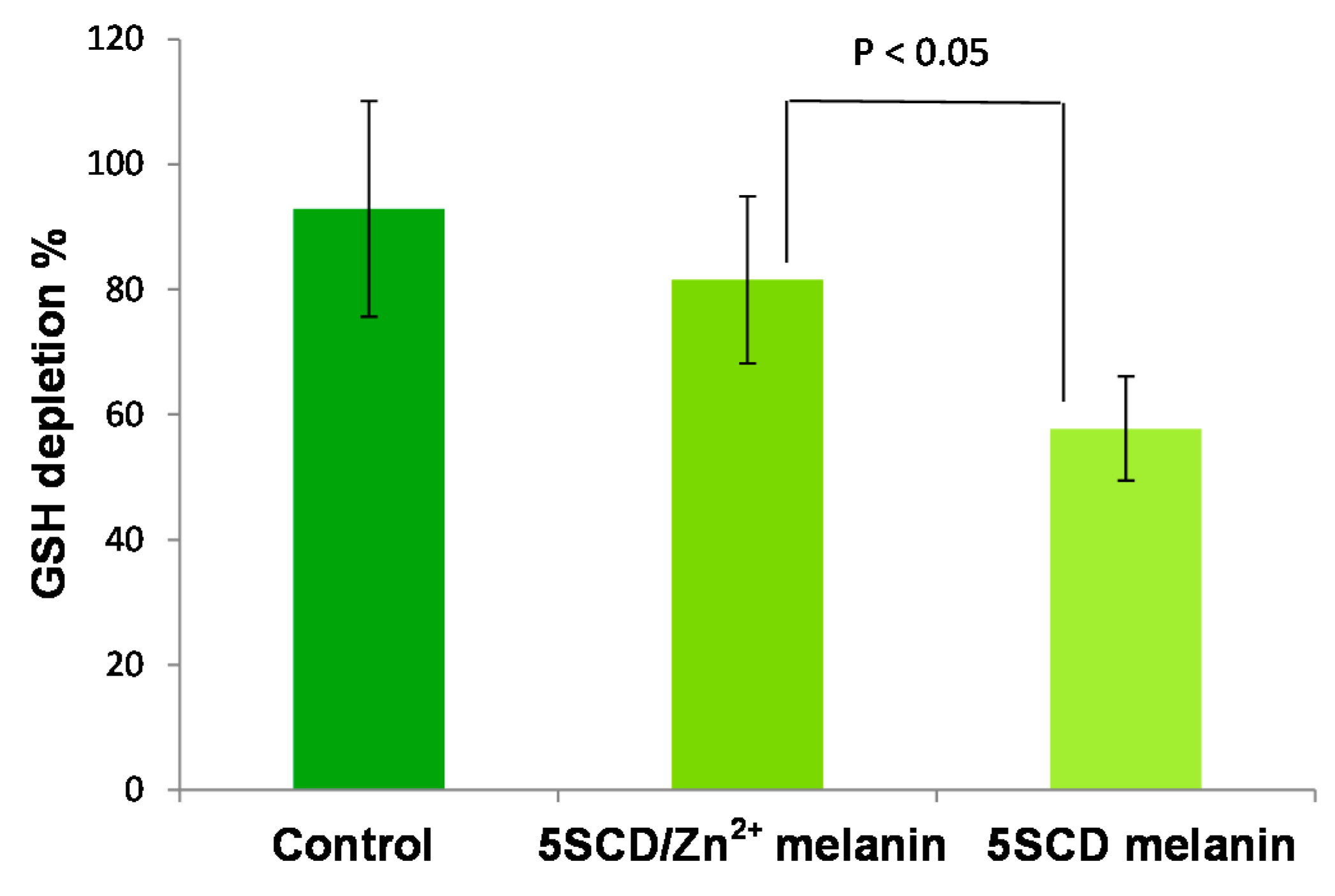
| Melanin | UVA-Induced Oxygen Consumption Rate (mM/min) | Blue Light-Induced Oxygen Consumption Rate (mM/min) |
|---|---|---|
| 5SCD/Zn2+ melanin | 0.12 ± 0.02 | 0.13 ± 0.02 |
| 5SCD melanin | 0.032 ± 0.005 | 0.024 ± 0.004 |
| Monomer Characteristics | DHI | DHICA (Carboxylated) | BTZ | BTZCA (Carboxylated) |
|---|---|---|---|---|
| Reactive sites | At least 4 | Usually 2 | At least 3 | Usually 1 |
| Oxidizability | High | Low | High | Low |
| Polymer properties | ||||
| Solubility | Nihil | Very slight 1 | Low 1 | Moderate 1 |
| EPR signal intensity | Strong | Weak | Weak | Intense |
| Visible absorption | Intense | Poor | Moderate | Intense |
| Photophysical properties | Efficient excited state decay | Highly efficient excited state decay | Poor photosensitizer | Strong photosensitizer |
| Chemical properties | Antioxidant | Highly potent antioxidant | Strong pro-oxidant | Weak pro-oxidant |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Micillo, R.; Panzella, L.; Koike, K.; Monfrecola, G.; Napolitano, A.; D’Ischia, M. “Fifty Shades” of Black and Red or How Carboxyl Groups Fine Tune Eumelanin and Pheomelanin Properties. Int. J. Mol. Sci. 2016, 17, 746. https://doi.org/10.3390/ijms17050746
Micillo R, Panzella L, Koike K, Monfrecola G, Napolitano A, D’Ischia M. “Fifty Shades” of Black and Red or How Carboxyl Groups Fine Tune Eumelanin and Pheomelanin Properties. International Journal of Molecular Sciences. 2016; 17(5):746. https://doi.org/10.3390/ijms17050746
Chicago/Turabian StyleMicillo, Raffaella, Lucia Panzella, Kenzo Koike, Giuseppe Monfrecola, Alessandra Napolitano, and Marco D’Ischia. 2016. "“Fifty Shades” of Black and Red or How Carboxyl Groups Fine Tune Eumelanin and Pheomelanin Properties" International Journal of Molecular Sciences 17, no. 5: 746. https://doi.org/10.3390/ijms17050746
APA StyleMicillo, R., Panzella, L., Koike, K., Monfrecola, G., Napolitano, A., & D’Ischia, M. (2016). “Fifty Shades” of Black and Red or How Carboxyl Groups Fine Tune Eumelanin and Pheomelanin Properties. International Journal of Molecular Sciences, 17(5), 746. https://doi.org/10.3390/ijms17050746











