Long-Term Follow-up of HPV Infection Using Urine and Cervical Quantitative HPV DNA Testing
Abstract
:1. Introduction
2. Results
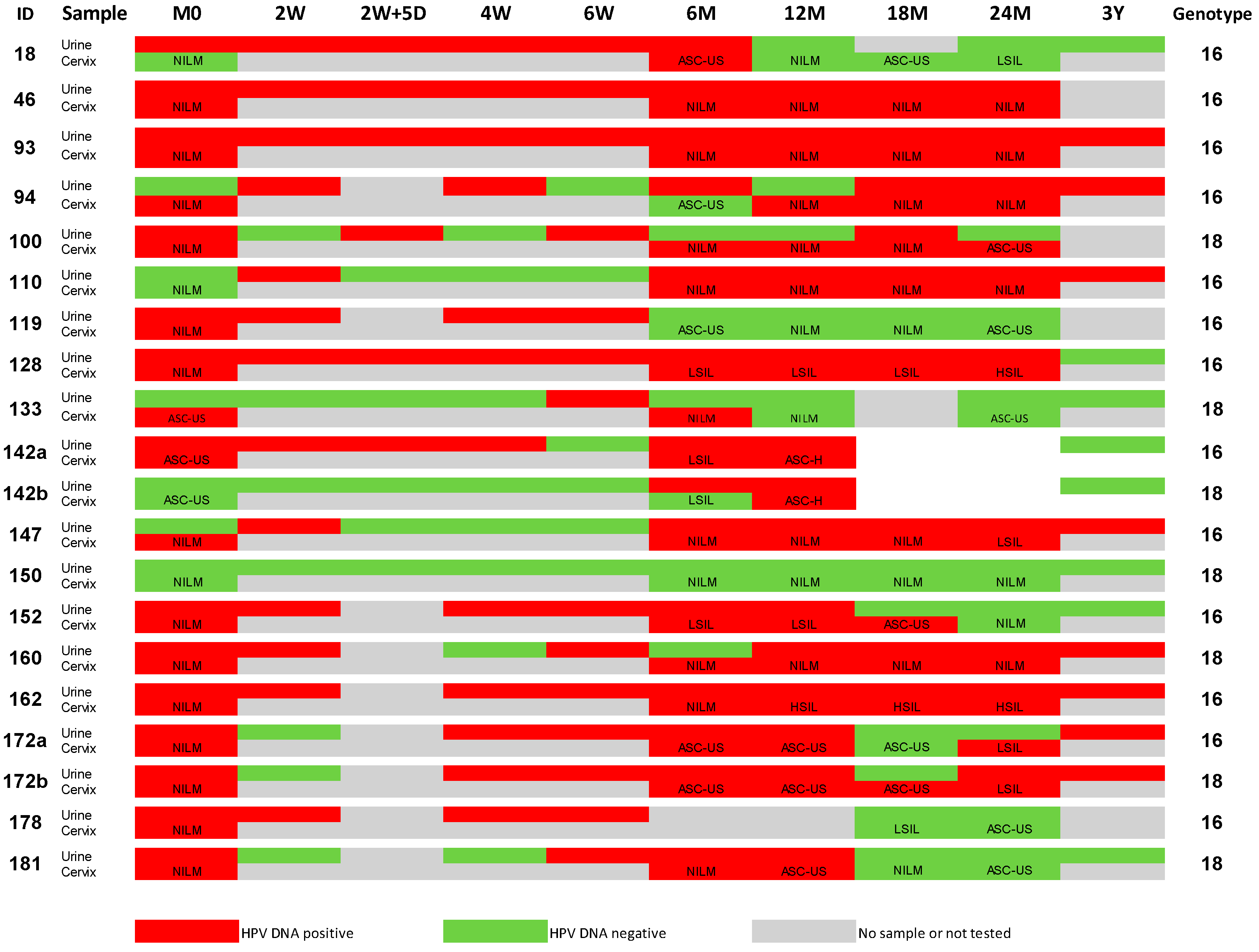
2.1. HPV DNA Results from Urine and Cervical Samples
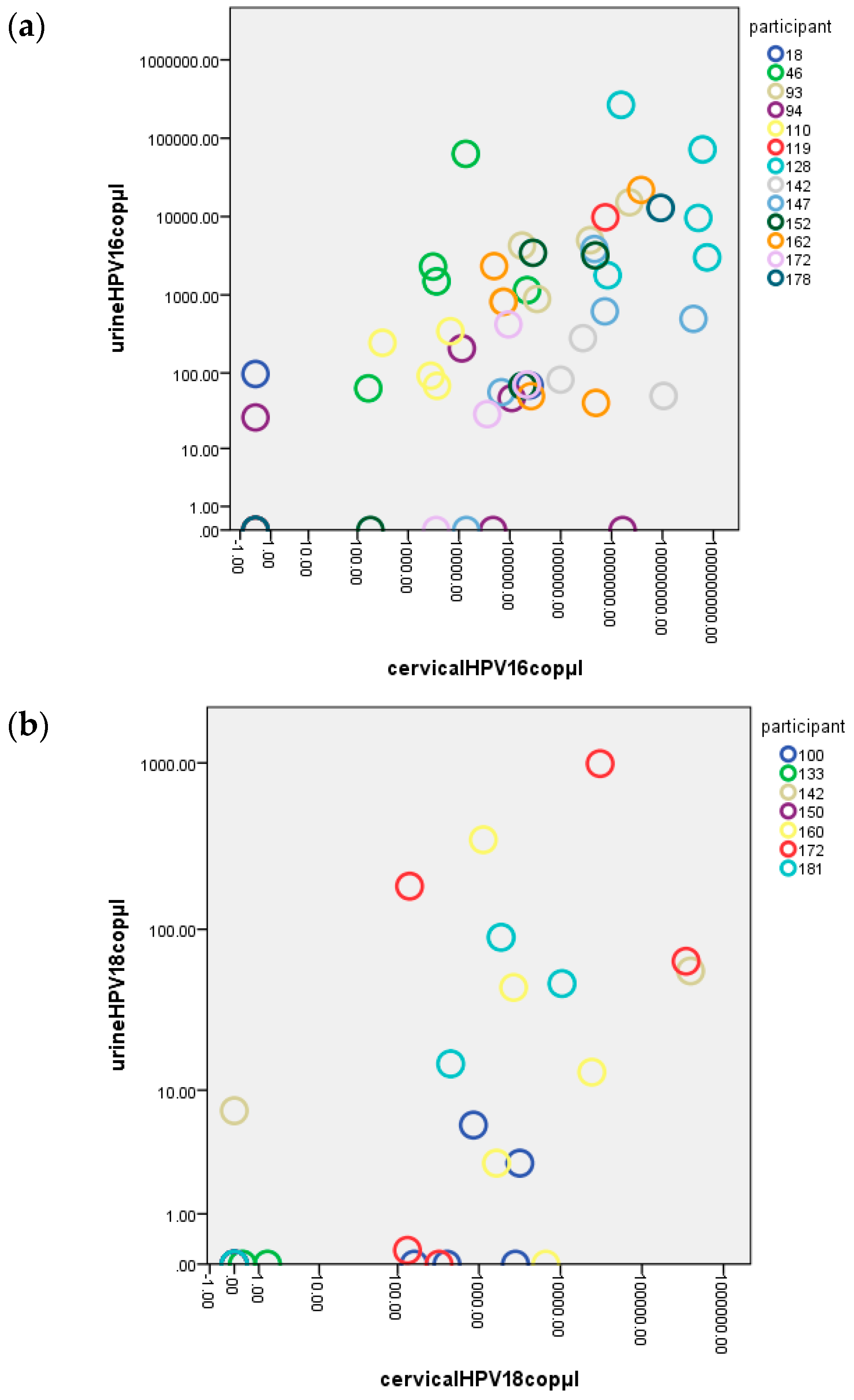
2.2. Correlation of HPV DNA Levels in Urine and Cervical Samples
2.3. Impact of Cytology on the Correlation of Urine versus Cervical
2.4. Results for Home-Collected Urine Samples
3. Discussion
3.1. Human DNA Detection in Urine
3.2. Detection of HPV DNA in Urine
3.3. Determination of the HPV DNA Viral Load in Urine
3.4. Advantages of Urine Sampling over Cervical Sampling
3.5. Limitations of the Study
4. Materials and Methods
4.1. Participants
4.2. Collection of Urine Samples
4.3. DNA Extraction from Urine Samples
4.4. Type-Specific Real-Time Quantitative PCR of DNA Extracted from Urine
4.5. Collection of Cervical Samples
4.6. DNA Extraction and PCR of Cervical Samples
4.7. Statistics and Software
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AGC | Atypical Glandular Cells |
| ASC-H | Atypical Squamous Cells cannot exclude High-grade Squamous Intraepithelial Lesions |
| ASC-US | Atypical Squamous Cells of Undetermined Significance |
| CIN | Cervical intraepithelial neoplasia |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| hDNA | Human DNA |
| HPV | Human Papillomavirus |
| hrHPV | High-Risk Human Papillomavirus |
| HSIL | High-Grade Squamous Intraepithelial Lesions |
| IARC | International Agency for Research on Cancer |
| LSIL | Low-Grade Squamous Intraepithelial Lesions |
| NILM | Negative for Intraepithelial Lesion or Malignancy |
| PAP | Papanicolaou |
| PCR | Polymerase Chain Reaction |
| qPCR | Quantitative PCR |
| WHO | World Health Organisation |
References
- Zur Hausen, H. Human papillomaviruses and their possible role in squamous cell carcinomas. Curr. Top. Microbiol. Immunol. 1977, 78, 1–30. [Google Scholar] [PubMed]
- Walboomers, J.M.; Jacobs, M.V.; Manos, M.M.; Bosch, F.X.; Kummer, J.A.; Shah, K.V.; Snijders, P.J.; Peto, J.; Meijer, C.J.; Munoz, N. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J. Pathol. 1999, 189, 12–19. [Google Scholar] [CrossRef]
- Joura, E.A.; Giuliano, A.R.; Iversen, O.-E.; Bouchard, C.; Mao, C.; Mehlsen, J.; Moreira, E.D.; Ngan, Y.; Petersen, L.K.; Lazcano-Ponce, E.; et al. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N. Engl. J. Med. 2015, 372, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Kjaer, S.K.; Sigurdsson, K.; Iversen, O.-E.; Hernandez-Avila, M.; Wheeler, C.M.; Perez, G.; Brown, D.R.; Koutsky, L.A.; Tay, E.H.; García, P.; et al. A pooled analysis of continued prophylactic efficacy of quadrivalent human papillomavirus (types 6/11/16/18) vaccine against high-grade cervical and external genital lesions. Cancer Prev. Res. 2009, 2, 868–878. [Google Scholar] [CrossRef] [PubMed]
- Paavonen, J.; Naud, P.; Salmeron, J.; Wheeler, C.M.; Chow, S.N.; Apter, D.; Kitchener, H.; Castellsague, X.; Teixeira, J.C.; Skinner, S.R.; et al. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): Final analysis of a double-blind, randomised study in young women. Lancet 2009, 374, 301–314. [Google Scholar] [CrossRef]
- The FUTURE II Study Group. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N. Engl. J. Med. 2007, 356, 1915–1927. [Google Scholar]
- IARC. Primay End-Points for Prophylactic HPV Vaccine Trials; IARC: Lyon, France, 2014; pp. 1–94. [Google Scholar]
- Brown, D.R.; Kjaer, S.K.; Sigurdsson, K.; Iversen, O.-E.; Hernandez-Avila, M.; Wheeler, C.M.; Perez, G.; Koutsky, L.A.; Tay, E.H.; Garcia, P.; et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in generally HPV-naive women aged 16–26 years. J. Infect. Dis. 2009, 199, 926–935. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, N.; Teixeira, J.; Roteli-Martins, C.M.; Naud, P.; De Borba, P.; Zahaf, T.; Sanchez, N.; Schuind, A. Sustained efficacy and immunogenicity of the HPV-16/18 AS04-adjuvanted vaccine up to 7.3 years in young adult women. Vaccine 2010, 28, 6247–6255. [Google Scholar] [CrossRef] [PubMed]
- Herrero, R.; Wacholder, S.; Rodríguez, A.C.; Solomon, D.; González, P.; Kreimer, A.R.; Porras, C.; Schussler, J.; Jiménez, S.; Sherman, M.E.; et al. Prevention of persistent human papillomavirus infection by an HPV16/18 vaccine: A community-based randomized clinical trial in Guanacaste, Costa Rica. Cancer Discov. 2011, 1, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Insinga, R.P.; Perez, G.; Wheeler, C.M.; Koutsky, L.A.; Garland, S.M.; Leodolter, S.; Joura, E.A.; Ferris, D.G.; Steben, M.; Brown, D.R.; et al. Incidence, duration, and reappearance of type-specific cervical human papillomavirus infections in young women. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1585–1594. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, N.; Manalastas Jr, R.; Pitisuttithum, P.; Tresukosol, D.; Monsonego, J.; Ault, K.; Clavel, C.; Luna, J.; Myers, E.; Hood, S.; et al. Safety, immunogenicity, and efficacy of quadrivalent human papillomavirus (types 6, 11, 16, 18) recombinant vaccine in women aged 24–45 years: A randomised, double-blind trial. Lancet 2009, 373, 1949–1957. [Google Scholar] [CrossRef]
- Villa, L.L.; Costa, R.L.R.; Petta, C.A.; Andrade, R.P.; Paavonen, J.; Iversen, O.E.; Olsson, S.E.; Hoye, J.; Steinwall, M.; Riis-Johannessen, G.; et al. High sustained efficacy of a prophylactic quadrivalent human papillomavirus types 6/11/16/18 L1 virus-like particle vaccine through 5 years of follow-up. Br. J. Cancer 2006, 95, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- Arbyn, M.; Verdoodt, F.; Snijders, P.J.; Verhoef, V.M.; Suonio, E.; Dillner, L.; Minozzi, S.; Bellisario, C.; Banzi, R.; Zhao, F.H.; et al. Accuracy of human papillomavirus testing on self-collected versus clinician-collected samples: A meta-analysis. Lancet Oncol. 2014, 15, 172–183. [Google Scholar] [CrossRef]
- Vorsters, A.; Micalessi, I.; Bilcke, J.; Ieven, M.; Bogers, J.; van Damme, P. Detection of human papillomavirus DNA in urine: A review of the literature. Eur. J. Clin. Microbiol. 2012, 31, 627–640. [Google Scholar] [CrossRef] [PubMed]
- Vorsters, A.; van Keer, S.; Biesmans, S.; Vankerckhoven, V.; de Koeijer, M.; Beyers, K.; Ieven, M.; van Damme, P. HPV DNA detection in urine: Effect of a first-void urine collection device and time of collection. In Proceedings of the 30th International Papillomavirus Conference, Lisbon, Portugal, 17–21 September 2015.
- Payan, C.; Ducancelle, A.; Aboubaker, M.H.; Caer, J.; Tapia, M.; Chauvin, A.; Peyronnet, D.; le Hen, E.; Arab, Z.; Legrand, M.-C.; et al. Human papillomavirus quantification in urine and cervical samples by using the Mx4000 and LightCycler general real-time PCR systems. J. Clin. Microbiol. 2007, 45, 897–901. [Google Scholar] [CrossRef] [PubMed]
- Ducancelle, A.; Legrand, M.C.; Pivert, A.; Veillon, P.; le Guillou-Guillemette, H.; de Brux, M.A.; Beby-Defaux, A.; Agius, G.; Hantz, S.; Alain, S.; et al. Interest of human papillomavirus DNA quantification and genotyping in paired cervical and urine samples to detect cervical lesions. Arch. Gynecol. Obstet. 2014, 290, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Vorsters, A.; van den Bergh, J.; Micalessi, I.; Biesmans, S.; Bogers, J.; Hens, A.; de Coster, I.; Ieven, M.; van Damme, P. Optimization of HPV DNA detection in urine by improving collection, storage, and extraction. Eur. J. Clin. Microbiol. 2014, 33, 2005–2015. [Google Scholar] [CrossRef] [PubMed]
- Combita Rojas, A.; Gheit, T.; González, P.; Puerto, D.; Montoya, L.; Vorsters, A.; Tommasino, M.; Hernandez, G.; Murillo, R.; Wiesner, C. Validation of HPV DNA detection and typing in urine samples from young women from Colombia. In Proceedings of the 30th International Papillomavirus Conference, Lisbon, Portugal, 17–21 September 2015.
- Cuschieri, K.; Nandwani, R.; McGough, P.; Cook, F.; Hogg, L.; Robertson, C.; Cubie, H. Urine testing as a surveillance tool to monitor the impact of HPV immunization programs. J. Med. Virol. 2011, 83, 1983–1987. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, S.; Umulisa, M.C.; Tshomo, U.; Gheit, T.; Baussano, I.; Tenet, V.; Tshokey, T.; Gatera, M.; Ngabo, F.; van Damme, P.; et al. Urine testing to monitor the impact of HPV vaccination in Bhutan and Rwanda. Int. J. Cancer 2016. [Google Scholar] [CrossRef] [PubMed]
- Constandinou-Williams, C.; Collins, S.I.; Roberts, S.; Young, L.S.; Woodman, C.B.; Murray, P.G. Is human papillomavirus viral load a clinically useful predictive marker? A longitudinal study. Cancer Epidemiol. Biomark. Prev. 2010, 19, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Depuydt, C.E.; Jonckheere, J.; Berth, M.; Salembier, G.M.; Vereecken, A.J.; Bogers, J.J. Serial type-specific human papillomavirus (HPV) load measurement allows differentiation between regressing cervical lesions and serial virion productive transient infections. Cancer Med. 2015, 4, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Doorbar, J.; Quint, W.; Banks, L.; Bravo, I.G.; Stoler, M.; Broker, T.R.; Stanley, M.A. The biology and life-cycle of human papillomaviruses. Vaccine 2012, 30, F55–F70. [Google Scholar] [CrossRef] [PubMed]
- Schlecht, N.F.; Trevisan, A.; Duarte-Franco, E.; Rohan, T.E.; Ferenczy, A.; Villa, L.L.; Franco, E.L. Viral load as a predictor of the risk of cervical intraepithelial neoplasia. Int. J. Cancer 2003, 103, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Vorsters, A.; van Damme, P.; Clifford, G. Urine testing for HPV: Rationale for using first void. BMJ 2014, 349, g6252. [Google Scholar] [CrossRef] [PubMed]
- Vorsters, A.; van Keer, S.; van Damme, P. The use of urine in the follow-up of HPV vaccine trials. Hum. Vaccines Immunother. 2015, 11, 350–352. [Google Scholar] [CrossRef] [PubMed]
- Sellors, J.W.; Lorincz, A.T.; Mahony, J.B.; Mielzynska, I.; Lytwyn, A.; Roth, P.; Howard, M.; Chong, S.; Daya, D.; Chapman, W.; et al. Comparison of self-collected vaginal, vulvar and urine samples with physician-collected cervical samples for human papillomavirus testing to detect high-grade squamous intraepithelial lesions. CMAJ 2000, 163, 513–518. [Google Scholar] [PubMed]
- Senkomago, V.; Des Marais, A.C.; Rahangdale, L.; Vibat, C.R.; Erlander, M.G.; Smith, J.S. Comparison of urine specimen collection times and testing fractions for the detection of high-risk human papillomavirus and high-grade cervical precancer. J. Clin. Virol. 2016, 74, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Van Damme, P. Phase I trial results with Procervix vaccine in HPV 16/18 infected women with normal cytology. In Proceedings of the EUROGIN 2013, Florence, Italy, 3–6 November 2013.
- Solomon, D.; Davey, D.; Kurman, R.; Moriarty, A.; O'Connor, D.; Prey, M.; Raab, S.; Sherman, M.; Wilbur, D.; Wright, T., Jr.; et al. The 2001 Bethesda System: Terminology for reporting results of cervical cytology. JAMA 2002, 287, 2114–2119. [Google Scholar] [CrossRef] [PubMed]
- Depuydt, C.E.; Benoy, I.H.; Bailleul, E.J.; Vandepitte, J.; Vereecken, A.J.; Bogers, J.J. Improved endocervical sampling and HPV viral load detection by Cervex-Brush® Combi. Cytopathology 2006, 17, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Micalessi, I.M.; Boulet, G.A.; Bogers, J.J.; Benoy, I.H.; Depuydt, C.E. High-throughput detection, genotyping and quantification of the human papillomavirus using real-time PCR. Clin. Chem. Lab. Med. 2012, 50, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Depuydt, C.E.; Benoy, I.H.; Beert, J.F.; Criel, A.M.; Bogers, J.J.; Arbyn, M. Clinical validation of a type-specific real-time quantitative human papillomavirus PCR against the performance of hybrid capture 2 for the purpose of cervical cancer screening. J. Clin. Microbiol. 2012, 50, 4073–4077. [Google Scholar] [CrossRef] [PubMed]
| Case Summaries | ||||||
|---|---|---|---|---|---|---|
| Sample Type and DNA Tested | Urine HPV16 | Cervix HPV16 | Urine HPV18 | Cervix HPV18 | Urine hDNA | Cervix hDNA |
| N (number of samples) * | 124 | 60 | 66 | 32 | 172 | 85 |
| Mean (copies/µL) ** | 7474 | 43,566,170 | 43 | 27,670 | 10,983 | 3081 |
| Median (copies/µL) ** | 140 | 74,826 | 0 | 364 | 5300 | 2018 |
| Minimum (copies/µL) ** | 0 | 0 | 0 | 0 | 15 | 44 |
| Maximum (copies/µL) ** | 267,000 | 752,829,409 | 989 | 394,818 | 59,800 | 19,559 |
| Cervical HPV16 | Total | |||
|---|---|---|---|---|
| Negative | Positive | |||
| Urine HPV16 | Negative | 11 | 5 | 16 |
| Positive | 2 | 42 | 44 | |
| Total | 13 | 47 | 60 | |
| Cervical HPV18 | Total | |||
|---|---|---|---|---|
| Negative | Positive | |||
| Urine HPV18 | Negative | 10 | 7 | 17 |
| Positive | 1 | 14 | 15 | |
| Total | 11 | 21 | 32 | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vorsters, A.; Van Keer, S.; Biesmans, S.; Hens, A.; De Coster, I.; Goossens, H.; Ieven, M.; Van Damme, P. Long-Term Follow-up of HPV Infection Using Urine and Cervical Quantitative HPV DNA Testing. Int. J. Mol. Sci. 2016, 17, 750. https://doi.org/10.3390/ijms17050750
Vorsters A, Van Keer S, Biesmans S, Hens A, De Coster I, Goossens H, Ieven M, Van Damme P. Long-Term Follow-up of HPV Infection Using Urine and Cervical Quantitative HPV DNA Testing. International Journal of Molecular Sciences. 2016; 17(5):750. https://doi.org/10.3390/ijms17050750
Chicago/Turabian StyleVorsters, Alex, Severien Van Keer, Samantha Biesmans, Annick Hens, Ilse De Coster, Herman Goossens, Margareta Ieven, and Pierre Van Damme. 2016. "Long-Term Follow-up of HPV Infection Using Urine and Cervical Quantitative HPV DNA Testing" International Journal of Molecular Sciences 17, no. 5: 750. https://doi.org/10.3390/ijms17050750
APA StyleVorsters, A., Van Keer, S., Biesmans, S., Hens, A., De Coster, I., Goossens, H., Ieven, M., & Van Damme, P. (2016). Long-Term Follow-up of HPV Infection Using Urine and Cervical Quantitative HPV DNA Testing. International Journal of Molecular Sciences, 17(5), 750. https://doi.org/10.3390/ijms17050750







