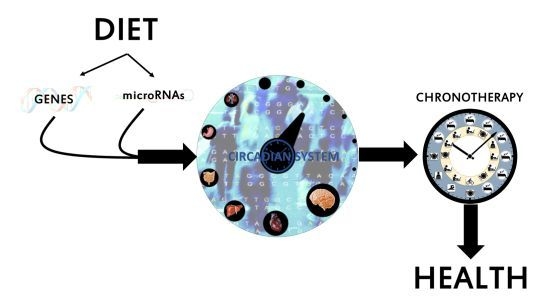
Nutrigenetics and Nutrimiromics of the Circadian System: The Time for Human Health
Abstract
:1. Introduction
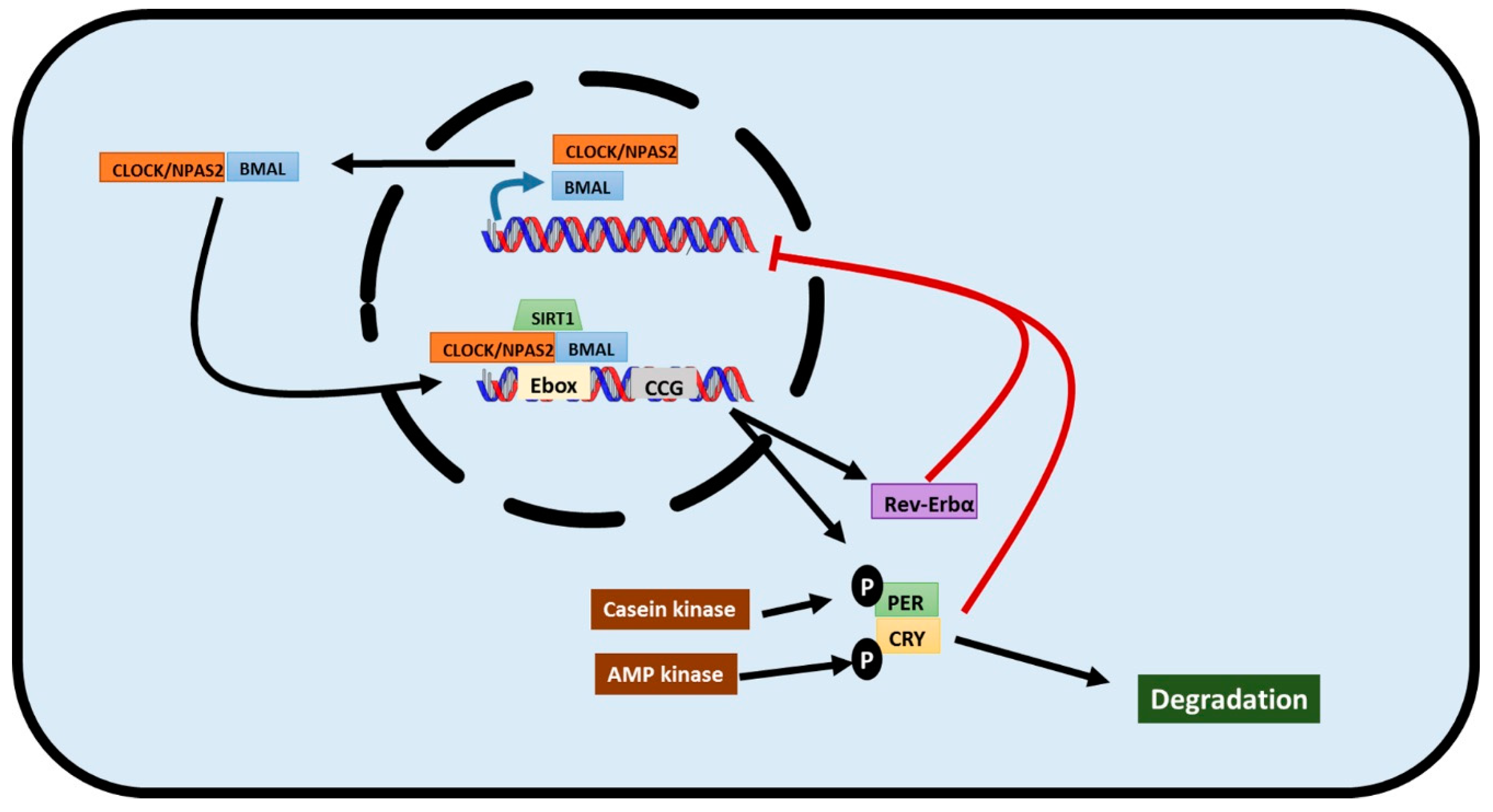
2. The Transcriptional/Translational Feedback Loop that Governs Circadian Rhythms
3. Peripheral Clocks, Time of Feeding and Metabolic Consequences of Chronodisruption
4. Gene Variants and Gene-Diet Interactions in Circadian-Related Genes Associated to Disease
4.1. Lessons from Animal Models of Genetic Chronodisruption
4.2. Gene Variants in Circadian-RelatedG Are Associated with Metabolic Disorders and Modulate the Individual Response to Diet: Human Population Studies
4.2.1. Variants in the CLOCK Gene
4.2.2. Variants in Other Circadian-Related Genes
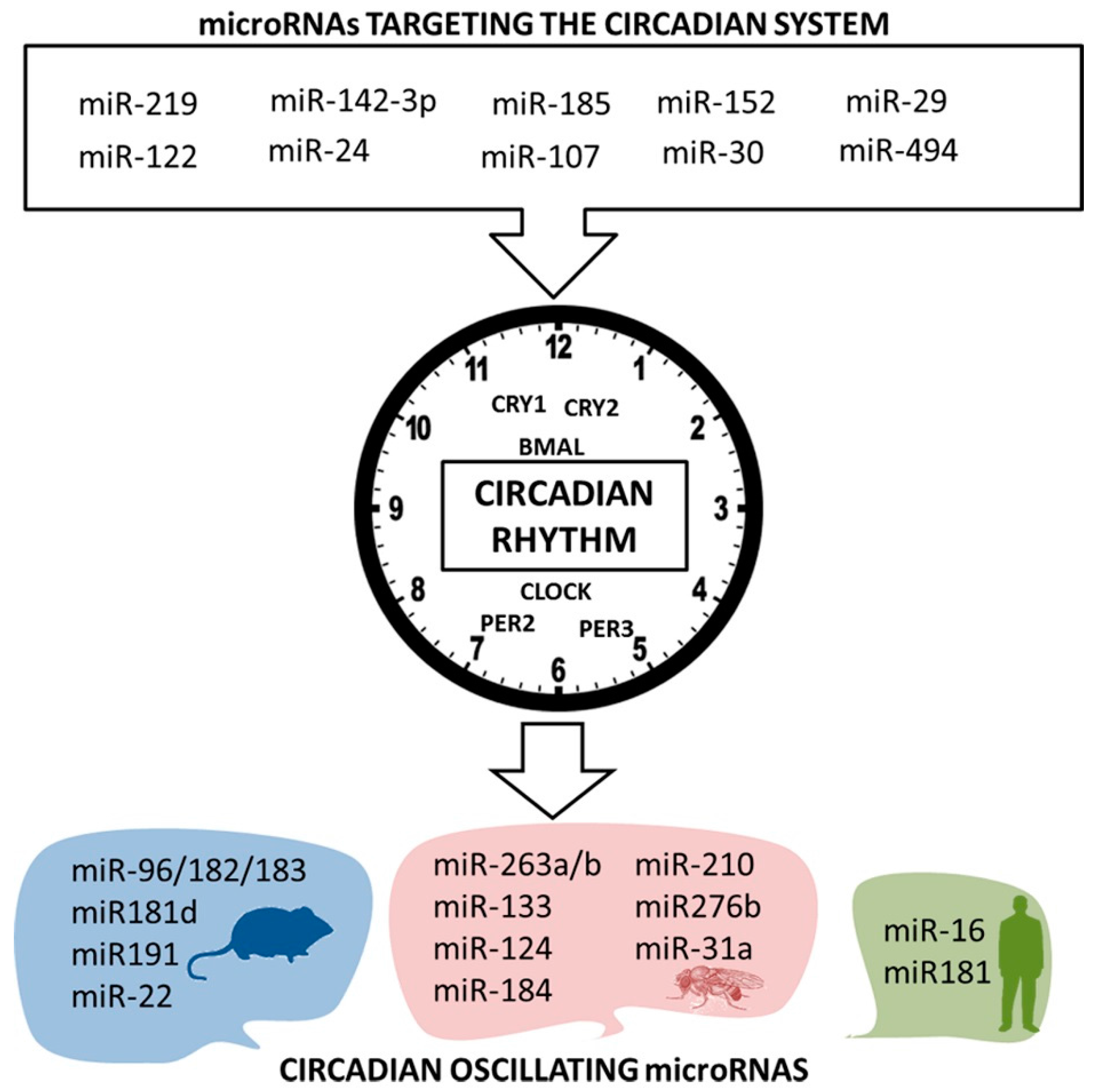
5. Nutrimiromics of the Circadian System
5.1. Circadian Expression of MicroRNAs
5.2. Micromanaging the Circadian Clock
6. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MetS | Metabolic Syndrome |
| T2DM | type 2 Diabetes Mellitus |
| CVD | Cardiovascular Disease |
| SCN | Suprachiasmatic Nucleus |
| CCG | clock controlled genes |
| PUFAs | polyunsaturated fatty acids |
| SNPs | single nucleotide polymorphisms |
| SFA | saturated fat intake |
| MUFA | monounsaturated fatty acids |
References
- Corbalan-Tutau, D.; Madrid, J.A.; Nicolas, F.; Garaulet, M. Daily profile in two circadian markers “melatonin and cortisol” and associations with metabolic syndrome components. Physiol. Behav. 2012, 123, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Ordovás, J.M. Chronobiology and Obesity; Springer: New York, NY, USA, 2012. [Google Scholar]
- Chen, L.; Yang, G. Recent advances in circadian rhythms in cardiovascular system. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Feillet, C.; van der Horst, G.T.; Levi, F.; Rand, D.A.; Delaunay, F. Coupling between the circadian clock and cell cycle oscillators: Implication for healthy cells and malignant growth. Front. Neurol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Corbalan-Tutau, M.D.; Gomez-Abellan, P.; Madrid, J.A.; Canteras, M.; Ordovas, J.M.; Garaulet, M. Toward a chronobiological characterization of obesity and metabolic syndrome in clinical practice. Clin. Nutr. 2015, 34, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Gomez-Abellan, P.; Alburquerque-Bejar, J.J.; Lee, Y.C.; Ordovas, J.M.; Scheer, F.A. Timing of food intake predicts weight loss effectiveness. Int. J. Obes. 2013, 37, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Bandin, C.; Martinez-Nicolas, A.; Ordovas, J.M.; Madrid, J.A.; Garaulet, M. Circadian rhythmicity as a predictor of weight-loss effectiveness. Int. J. Obes. 2014, 38, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Arble, D.M.; Bass, J.; Laposky, A.D.; Vitaterna, M.H.; Turek, F.W. Circadian timing of food intake contributes to weight gain. Obesity 2009, 17, 2100–2102. [Google Scholar] [CrossRef] [PubMed]
- Eckel-Mahan, K.L.; Patel, V.R.; Mohney, R.P.; Vignola, K.S.; Baldi, P.; Sassone-Corsi, P. Coordination of the transcriptome and metabolome by the circadian clock. Proc. Natl. Acad. Sci. USA 2012, 109, 5541–5546. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.R.; Eckel-Mahan, K.; Sassone-Corsi, P.; Baldi, P. CircadiOmics: Integrating circadian genomics, transcriptomics, proteomics and metabolomics. Nat. Meth. 2012, 9, 772–773. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Lahens, N.F.; Ballance, H.I.; Hughes, M.E.; Hogenesch, J.B. A circadian gene expression atlas in mammals: Implications for biology and medicine. Proc. Natl. Acad. Sci. USA 2014, 111, 16219–16224. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, J.S.; Hong, H.K.; Ko, C.H.; McDearmon, E.L. The genetics of mammalian circadian order and disorder: Implications for physiology and disease. Nat. Rev. Genet. 2008, 9, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Oosterman, J.E.; Kalsbeek, A.; la Fleur, S.E.; Belsham, D.D. Impact of nutrients on circadian rhythmicity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 308, R337–R350. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Madrid, J.A. Chronobiological aspects of nutrition, metabolic syndrome and obesity. Adv. Drug Deliv. Rev. 2010, 62, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.D. Physiological links between circadian rhythms, metabolism and nutrition. Exp. Physiol. 2014, 99, 1133–1137. [Google Scholar] [CrossRef] [PubMed]
- Merbitz-Zahradnik, T.; Wolf, E. How is the inner circadian clock controlled by interactive clock proteins?: Structural analysis of clock proteins elucidates their physiological role. FEBS Lett. 2015, 589, 1516–1529. [Google Scholar] [CrossRef] [PubMed]
- Versteeg, R.I.; Serlie, M.J.; Kalsbeek, A.; la Fleur, S.E. Serotonin, a possible intermediate between disturbed circadian rhythms and metabolic disease. Neuroscience 2015, 301, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Mukherji, A.; Kobiita, A.; Chambon, P. Shifting the feeding of mice to the rest phase creates metabolic alterations, which, on their own, shift the peripheral circadian clocks by 12 hours. Proc. Natl. Acad. Sci. USA 2015, 112, E6683–E6690. [Google Scholar] [CrossRef] [PubMed]
- Bass, J.; Takahashi, J.S. Circadian integration of metabolism and energetics. Science 2010, 330, 1349–1354. [Google Scholar] [CrossRef] [PubMed]
- Ferrell, J.M.; Chiang, J.Y. Circadian rhythms in liver metabolism and disease. Acta Pharm. Sin. B 2015, 5, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Lecarpentier, Y.; Claes, V.; Duthoit, G.; Hebert, J.L. Circadian rhythms, Wnt/β-catenin pathway and PPAR α/γ profiles in diseases with primary or secondary cardiac dysfunction. Front. Physiol. 2014, 5, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yang, G. PPARs integrate the mammalian clock and energy metabolism. PPAR Res. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Brewer, M.; Lange, D.; Baler, R.; Anzulovich, A. SREBP-1 as a transcriptional integrator of circadian and nutritional cues in the liver. J. Biol. Rhythms 2005, 20, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.M.; Udoh, U.S.; Young, M.E. Circadian regulation of metabolism. J. Endocrinol. 2014, 222, R75–R96. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Santos, C.; Gomez-Abellan, P.; Madrid, J.A.; Hernandez-Morante, J.J.; Lujan, J.A.; Ordovas, J.M.; Garaulet, M. Circadian rhythm of clock genes in human adipose explants. Obesity 2009, 17, 1481–1485. [Google Scholar] [CrossRef] [PubMed]
- Erren, T.C.; Reiter, R.J. Defining chronodisruption. J. Pineal Res. 2009, 46, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.H.; Jeong, J.U.; Jeon, M.J.; Sakong, J. The association between shift work and the metabolic syndrome in female workers. Ann. Occup. Environ. Med. 2013, 25. [Google Scholar] [CrossRef] [PubMed]
- Burgueno, A.; Gemma, C.; Gianotti, T.F.; Sookoian, S.; Pirola, C.J. Increased levels of resistin in rotating shift workers: A potential mediator of cardiovascular risk associated with circadian misalignment. Atherosclerosis 2010, 210, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Hermansson, J.; Gillander Gadin, K.; Karlsson, B.; Reuterwall, C.; Hallqvist, J.; Knutsson, A. Case fatality of myocardial infarction among shift workers. Int. Arch. Occup. Environ. Health 2015, 88, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Rutters, F.; Lemmens, S.G.; Adam, T.C.; Bremmer, M.A.; Elders, P.J.; Nijpels, G.; Dekker, J.M. Is social jetlag associated with an adverse endocrine, behavioral, and cardiovascular risk profile? J. Biol. Rhythms 2014, 29, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Alibhai, F.J.; Tsimakouridze, E.V.; Reitz, C.J.; Pyle, W.G.; Martino, T.A. Consequences of circadian and sleep disturbances for the cardiovascular system. Can. J. Cardiol. 2015, 31, 860–872. [Google Scholar] [CrossRef] [PubMed]
- Neil-Sztramko, S.E.; Pahwa, M.; Demers, P.A.; Gotay, C.C. Health-related interventions among night shift workers: A critical review of the literature. Scand. J. Work Environ. Health 2014, 40, 543–556. [Google Scholar] [CrossRef] [PubMed]
- Viitasalo, K.; Kuosma, E.; Laitinen, J.; Harma, M. Effects of shift rotation and the flexibility of a shift system on daytime alertness and cardiovascular risk factors. Scand. J. Work Environ. Health 2008, 34, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Reid, K.J.; Abbott, S.M. Jet lag and shift work disorder. Sleep Med. Clin. 2015, 10, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.J.; Purvis, T.E.; Mistretta, J.; Scheer, F.A. Effects of the internal circadian system and circadian misalignment on glucose tolerance in chronic shift workers. J. Clin. Endocrinol. Metab. 2016, jc20153924. [Google Scholar] [CrossRef] [PubMed]
- Dibner, C.; Gachon, F. Circadian dysfunction and obesity: Is leptin the missing link? Cell Metab. 2015, 22, 359–360. [Google Scholar] [CrossRef] [PubMed]
- Mukherji, A.; Kobiita, A.; Damara, M.; Misra, N.; Meziane, H.; Champy, M.F.; Chambon, P. Shifting eating to the circadian rest phase misaligns the peripheral clocks with the master scn clock and leads to a metabolic syndrome. Proc. Natl. Acad. Sci. USA 2015, 112, E6691–E6698. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Sun, L.; Zhuge, F.; Guo, X.; Zhao, Z.; Tang, R.; Chen, Q.; Chen, L.; Kato, H.; Fu, Z. Differential roles of breakfast and supper in rats of a daily three-meal schedule upon circadian regulation and physiology. Chronobiol. Int. 2011, 28, 890–903. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Yao, C.; Huang, L.; Mao, Y.; Zhang, W.; Jiang, J.; Fu, Z. Nutrients and circadian rhythms in mammals. J. Nutr. Sci. Vitaminol. 2015, 61, S89–S91. [Google Scholar] [CrossRef] [PubMed]
- Bandin, C.; Scheer, F.A.; Luque, A.J.; Avila-Gandia, V.; Zamora, S.; Madrid, J.A.; Gomez-Abellan, P.; Garaulet, M. Meal timing affects glucose tolerance, substrate oxidation and circadian-related variables: A randomized, crossover trial. Int. J. Obes. 2015, 39, 828–833. [Google Scholar] [CrossRef] [PubMed]
- Hunt, S.C.; Stone, S.; Xin, Y.; Scherer, C.A.; Magness, C.L.; Iadonato, S.P.; Hopkins, P.N.; Adams, T.D. Association of the FTO gene with BMI. Obesity 2008, 16, 902–904. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, A.; Sanna, S.; Chen, W.M.; Uda, M.; Albai, G.; Strait, J.; Najjar, S.; Nagaraja, R.; Orru, M.; Usala, G.; et al. Genome-wide association scan shows genetic variants in the FTO gene are associated with obesity-related traits. PLoS Genet. 2007, 3, e115. [Google Scholar] [CrossRef] [PubMed]
- Yeo, G.S. The role of the FTO (Fat Mass and Obesity Related) locus in regulating body size and composition. Mol. Cell. Endocrinol. 2014, 397, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Cauchi, S.; El Achhab, Y.; Choquet, H.; Dina, C.; Krempler, F.; Weitgasser, R.; Nejjari, C.; Patsch, W.; Chikri, M.; Meyre, D.; et al. TCF7L2 is reproducibly associated with type 2 diabetes in various ethnic groups: A global meta-analysis. J. Mol. Med. 2007, 85, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Voight, B.F.; Scott, L.J.; Steinthorsdottir, V.; Morris, A.P.; Dina, C.; Welch, R.P.; Zeggini, E.; Huth, C.; Aulchenko, Y.S.; Thorleifsson, G.; et al. Twelve type 2 diabetes susceptibility loci identified through large-scale association analysis. Nat. Genet. 2010, 42, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Saxena, R.; Elbers, C.C.; Guo, Y.; Peter, I.; Gaunt, T.R.; Mega, J.L.; Lanktree, M.B.; Tare, A.; Castillo, B.A.; Li, Y.R.; et al. Large-scale gene-centric meta-analysis across 39 studies identifies type 2 diabetes loci. Am. J. Hum. Genet. 2012, 90, 410–425. [Google Scholar] [CrossRef] [PubMed]
- Daimiel, L.; Vargas, T.; Ramirez de Molina, A. Nutritional genomics for the characterization of the effect of bioactive molecules in lipid metabolism and related pathways. Electrophoresis 2012, 33, 2266–2289. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidou, V.; Ruiz, L.A.; Ordovas, J.M. Personalized nutrition and cardiovascular disease prevention: From Framingham to PREDIMED. Adv. Nutr. 2014, 5, 368S–371S. [Google Scholar] [CrossRef] [PubMed]
- Luglio, H.F.; Sulistyoningrum, D.C.; Susilowati, R. The role of genes involved in lipolysis on weight loss program in overweight and obese individuals. J. Clin. Biochem. Nutr. 2015, 57, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Qi, L.; Sorli, J.V.; Godoy, D.; Portoles, O.; Coltell, O.; Greenberg, A.S.; Ordovas, J.M. Obese subjects carrying the 11482G>A polymorphism at the perilipin locus are resistant to weight loss after dietary energy restriction. J. Clin. Endocrinol. Metab. 2005, 90, 5121–5126. [Google Scholar] [CrossRef] [PubMed]
- Turek, F.W.; Joshu, C.; Kohsaka, A.; Lin, E.; Ivanova, G.; McDearmon, E.; Laposky, A.; Losee-Olson, S.; Easton, A.; Jensen, D.R.; et al. Obesity and metabolic syndrome in circadian clock mutant mice. Science 2005, 308, 1043–1045. [Google Scholar] [CrossRef] [PubMed]
- Marcheva, B.; Ramsey, K.M.; Buhr, E.D.; Kobayashi, Y.; Su, H.; Ko, C.H.; Ivanova, G.; Omura, C.; Mo, S.; Vitaterna, M.H.; et al. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature 2010, 466, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Rudic, R.D.; McNamara, P.; Curtis, A.M.; Boston, R.C.; Panda, S.; Hogenesch, J.B.; Fitzgerald, G.A. BMAL1 and CLOCK, two essential components of the circadian clock, are involved in glucose homeostasis. PLoS Biol. 2004, 2, e377. [Google Scholar] [CrossRef] [PubMed]
- Kettner, N.M.; Mayo, S.A.; Hua, J.; Lee, C.; Moore, D.D.; Fu, L. Circadian dysfunction induces leptin resistance in mice. Cell Metab. 2015, 22, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Oishi, K.; Ohkura, N.; Wakabayashi, M.; Shirai, H.; Sato, K.; Matsuda, J.; Atsumi, G.; Ishida, N. CLOCK is involved in obesity-induced disordered fibrinolysis in ob/ob mice by regulating PAI-1 gene expression. J. Thromb. Haemost. 2006, 4, 1774–1780. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Jiang, X.C.; Hussain, M.M. Impaired cholesterol metabolism and enhanced atherosclerosis in clock mutant mice. Circulation 2013, 128, 1758–1769. [Google Scholar] [CrossRef] [PubMed]
- Peliciari-Garcia, R.A.; Goel, M.; Aristorenas, J.A.; Shah, K.; He, L.; Yang, Q.; Shalev, A.; Bailey, S.M.; Prabhu, S.D.; Chatham, J.C.; et al. Altered myocardial metabolic adaptation to increased fatty acid availability in cardiomyocyte-specific CLOCK mutant mice. Biochim. Biophys. Acta 2015, 22. [Google Scholar] [CrossRef] [PubMed]
- Dallmann, R.; Touma, C.; Palme, R.; Albrecht, U.; Steinlechner, S. Impaired daily glucocorticoid rhythm in Per1 ( Brd ) mice. J. Comp. Physiol. A Neuroethol. Sens. Neural. Behav. Physiol. 2006, 192, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Liu, A.; Weidenhammer, A.; Cooksey, R.C.; McClain, D.; Kim, M.K.; Aguilera, G.; Abel, E.D.; Chung, J.H. The role of mPer2 clock gene in glucocorticoid and feeding rhythms. Endocrinology 2009, 150, 2153–2160. [Google Scholar] [CrossRef] [PubMed]
- Husse, J.; Hintze, S.C.; Eichele, G.; Lehnert, H.; Oster, H. Circadian clock genes Per1 and Per2 regulate the response of metabolism-associated transcripts to sleep disruption. PLoS ONE 2012, 7, e52983. [Google Scholar] [CrossRef] [PubMed]
- Le Martelot, G.; Claudel, T.; Gatfield, D.; Schaad, O.; Kornmann, B.; Lo Sasso, G.; Moschetta, A.; Schibler, U. REV-ERBα participates in circadian SREBP signaling and bile acid homeostasis. PLoS Biol. 2009, 7, e1000181. [Google Scholar] [CrossRef] [PubMed]
- Griebel, G.; Ravinet-Trillou, C.; Beeske, S.; Avenet, P.; Pichat, P. Mice deficient in cryptochrome 1 (cry1 (-/-)) exhibit resistance to obesity induced by a high-fat diet. Front. Endocrinol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.S.; Aslibekyan, S.; Scheer, F.A.; Smith, C.E.; Lamon-Fava, S.; Jacques, P.; Lai, C.Q.; Tucker, K.L.; Arnett, D.K.; Ordovas, J.M. Clock genes explain a large proportion of phenotypic variance in systolic blood pressure and this control is not modified by environmental temperature. Am. J. Hypertens. 2016, 29, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Corbalan, M.D.; Madrid, J.A.; Morales, E.; Baraza, J.C.; Lee, Y.C.; Ordovas, J.M. CLOCK gene is implicated in weight reduction in obese patients participating in a dietary programme based on the mediterranean diet. Int. J. Obes. 2010, 34, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Lee, Y.C.; Shen, J.; Parnell, L.D.; Arnett, D.K.; Tsai, M.Y.; Lai, C.Q.; Ordovas, J.M. Genetic variants in human CLOCK associate with total energy intake and cytokine sleep factors in overweight subjects (GOLDN population). Eur. J. Hum. Genet. 2010, 18, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Sanchez-Moreno, C.; Smith, C.E.; Lee, Y.C.; Nicolas, F.; Ordovas, J.M. Ghrelin, sleep reduction and evening preference: Relationships to CLOCK 3111 T/C SNP and weight loss. PLoS ONE 2011, 6, e17435. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Guimera, G.; Dashti, H.S.; Smith, C.E.; Sanchez-Carracedo, D.; Ordovas, J.M.; Garaulet, M. CLOCK 3111 T/C SNP interacts with emotional eating behavior for weight-loss in a Mediterranean population. PLoS ONE 2014, 9, e99152. [Google Scholar]
- Dashti, H.S.; Scheer, F.A.; Jacques, P.F.; Lamon-Fava, S.; Ordovas, J.M. Short sleep duration and dietary intake: Epidemiologic evidence, mechanisms, and health implications. Adv. Nutr. 2015, 6, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Bandin, C.; Martinez-Nicolas, A.; Ordovas, J.M.; Ros Lucas, J.A.; Castell, P.; Silvente, T.; Madrid, J.A.; Garaulet, M. Differences in circadian rhythmicity in CLOCK 3111T/C genetic variants in moderate obese women as assessed by thermometry, actimetry and body position. Int. J. Obes. 2013, 37, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Cong, W.N.; Golden, E.; Pantaleo, N.; White, C.M.; Maudsley, S.; Martin, B. Ghrelin receptor signaling: A promising therapeutic target for metabolic syndrome and cognitive dysfunction. CNS Neurol. Disord. Drug Targets 2010, 9, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Lee, Y.C.; Shen, J.; Parnell, L.D.; Arnett, D.K.; Tsai, M.Y.; Lai, C.Q.; Ordovas, J.M. CLOCK genetic variation and metabolic syndrome risk: Modulation by monounsaturated fatty acids. Am. J. Clin. Nutr. 2009, 90, 1466–1475. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rios, A.; Gomez-Delgado, F.J.; Garaulet, M.; Alcala-Diaz, J.F.; Delgado-Lista, F.J.; Marin, C.; Rangel-Zuniga, O.A.; Rodriguez-Cantalejo, F.; Gomez-Luna, P.; Ordovas, J.M.; et al. Beneficial effect of CLOCK gene polymorphism rs1801260 in combination with low-fat diet on insulin metabolism in the patients with metabolic syndrome. Chronobiol. Int. 2014, 31, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.S.; Tucker, K.L.; Smith, C.E.; Ma, Y.; Lee, Y.-C.; Ordovas, J.M. CLOCK 3111 T/C SNP interacts with saturated fatty acid intake to modulate plasma LDL-c Concentrations in the Boston-Puerto Rican Health Study. FASEB J. 2013, 27, 222–225. [Google Scholar]
- Uemura, H.; Katsuura-Kamano, S.; Yamaguchi, M.; Arisawa, K.; Hamajima, N.; Hishida, A.; Kawai, S.; Oze, I.; Shinchi, K.; Takashima, N.; et al. Variant of the CLOCK gene and related haplotypes are associated with the prevalence of type 2 diabetes in the Japanese population. J. Diabetes 2015. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Asensio, E.M.; Coltell, O.; Sorli, J.V.; Estruch, R.; Martinez-Gonzalez, M.A.; Salas-Salvado, J.; Castaner, O.; Aros, F.; Lapetra, J.; et al. CLOCK gene variation is associated with incidence of type-2 diabetes and cardiovascular diseases in type-2 diabetic subjects: Dietary modulation in the PREDIMED randomized trial. Cardiovasc. Diabetol. 2016, 15. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.S.; Smith, C.E.; Lee, Y.C.; Parnell, L.D.; Lai, C.Q.; Arnett, D.K.; Ordovas, J.M.; Garaulet, M. CRY1 circadian gene variant interacts with carbohydrate intake for insulin resistance in two independent populations: Mediterranean and north american. Chronobiol. Int. 2014, 31, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Li, H.; Qi, L.; Loos, R.J.; Qi, Q.; Lu, L.; Gan, W.; Lin, X. Variants in GLIS3 and CRY2 are associated with type 2 diabetes and impaired fasting glucose in Chinese Hans. PLoS ONE 2011, 6, e21464. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.S.; Follis, J.L.; Smith, C.E.; Tanaka, T.; Garaulet, M.; Gottlieb, D.J.; Hruby, A.; Jacques, P.F.; Kiefte-de Jong, J.C.; Lamon-Fava, S.; et al. Gene-environment interactions of circadian-related genes for cardiometabolic traits. Diabetes Care 2015, 38, 1456–1466. [Google Scholar] [CrossRef] [PubMed]
- Kovanen, L.; Donner, K.; Kaunisto, M.; Partonen, T. CRY1, CRY2 and PRKCDBP genetic variants in metabolic syndrome. Hypertens. Res. 2015, 38, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, A.; Ladenvall, C.; Ahluwalia, T.S.; Kravic, J.; Krus, U.; Taneera, J.; Isomaa, B.; Tuomi, T.; Renstrom, E.; Groop, L.; et al. Effects of common genetic variants associated with type 2 diabetes and glycemic traits on α- and β-cell function and insulin action in humans. Diabetes 2013, 62, 2978–2983. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, K.; Xu, M.; Qi, Q.; de Jonge, L.; Bray, G.A.; Sacks, F.; Qi, L. Variants in glucose- and circadian rhythm-related genes affect the response of energy expenditure to weight-loss diets: The POUNDS LOST Trial. Am. J. Clin. Nutr. 2014, 99, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.A.; Rees, S.D.; Hydrie, M.Z.; Shera, A.S.; Bellary, S.; O’Hare, J.P.; Kumar, S.; Taheri, S.; Basit, A.; Barnett, A.H. Circadian gene variants and susceptibility to type 2 diabetes: A pilot study. PLoS ONE 2012, 7, e32670. [Google Scholar] [CrossRef] [PubMed]
- Mariman, E.C.; Bouwman, F.G.; Aller, E.E.; van Baak, M.A.; Wang, P. Extreme obesity is associated with variation in genes related to the circadian rhythm of food intake and hypothalamic signaling. Physiol. Genom. 2015, 47, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Hamet, P.; Tremblay, J. Genetics of the sleep-wake cycle and its disorders. Metabolism 2006, 55, S7–S12. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Corbalan-Tutau, M.D.; Madrid, J.A.; Baraza, J.C.; Parnell, L.D.; Lee, Y.C.; Ordovas, J.M. PERIOD2 variants are associated with abdominal obesity, psycho-behavioral factors, and attrition in the dietary treatment of obesity. J. Am. Diet. Assoc. 2010, 110, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Woon, P.Y.; Kaisaki, P.J.; Braganca, J.; Bihoreau, M.T.; Levy, J.C.; Farrall, M.; Gauguier, D. Aryl hydrocarbon receptor nuclear translocator-like (BMAL1) is associated with susceptibility to hypertension and type 2 diabetes. Proc. Natl. Acad. Sci. USA 2007, 104, 14412–14417. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Smith, C.E.; Gomez-Abellan, P.; Ordovas-Montanes, M.; Lee, Y.C.; Parnell, L.D.; Arnett, D.K.; Ordovas, J.M. REV-ERB-α circadian gene variant associates with obesity in two independent populations: Mediterranean and north american. Mol. Nutr. Food Res. 2014, 58, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Ruano, E.G.; Canivell, S.; Vieira, E. REV-ERBα polymorphism is associated with obesity in the spanish obese male population. PLoS ONE 2014, 9, e104065. [Google Scholar] [CrossRef] [PubMed]
- Goumidi, L.; Grechez, A.; Dumont, J.; Cottel, D.; Kafatos, A.; Moreno, L.A.; Molnar, D.; Moschonis, G.; Gottrand, F.; Huybrechts, I.; et al. Impact of REV-ERBα gene polymorphisms on obesity phenotypes in adult and adolescent samples. Int. J. Obes. 2013, 37, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Bouatia-Naji, N.; Bonnefond, A.; Cavalcanti-Proenca, C.; Sparso, T.; Holmkvist, J.; Marchand, M.; Delplanque, J.; Lobbens, S.; Rocheleau, G.; Durand, E.; et al. A variant near MTNR1B is associated with increased fasting plasma glucose levels and type 2 diabetes risk. Nat. Genet. 2009, 41, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Lyssenko, V.; Nagorny, C.L.; Erdos, M.R.; Wierup, N.; Jonsson, A.; Spegel, P.; Bugliani, M.; Saxena, R.; Fex, M.; Pulizzi, N.; et al. Common variant in MTNR1B associated with increased risk of type 2 diabetes and impaired early insulin secretion. Nat. Genet. 2009, 41, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Prokopenko, I.; Langenberg, C.; Florez, J.C.; Saxena, R.; Soranzo, N.; Thorleifsson, G.; Loos, R.J.; Manning, A.K.; Jackson, A.U.; Aulchenko, Y.; et al. Variants in MTNR1B influence fasting glucose levels. Nat. Genet. 2009, 41, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Dalla Man, C.; Cobelli, C.; Groop, L.; Zhao, H.; Bale, A.E.; Shaw, M.; Duran, E.; Pierpont, B.; Caprio, S.; et al. A common variant in the MTNR1B gene is associated with increased risk of impaired fasting glucose (IFG) in youth with obesity. Obesity 2015, 23, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, L.; Zhao, J.; Cui, G.; Chen, C.; Ding, H.; Wang, D.W. Large scale meta-analyses of fasting plasma glucose raising variants in GCK, GCKR, MTNR1B and G6PC2 and their impacts on type 2 diabetes mellitus risk. PLoS ONE 2013, 8, e67665. [Google Scholar] [CrossRef] [PubMed]
- Been, L.F.; Hatfield, J.L.; Shankar, A.; Aston, C.E.; Ralhan, S.; Wander, G.S.; Mehra, N.K.; Singh, J.R.; Mulvihill, J.J.; Sanghera, D.K. A low frequency variant within the GWAS locus of MTNR1B affects fasting glucose concentrations: Genetic risk is modulated by obesity. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Pevet, P.; Challet, E. Melatonin: Both master clock output and internal time-giver in the circadian clocks network. J. Physiol. Paris 2011, 105, 170–182. [Google Scholar] [CrossRef] [PubMed]
- Simonis-Bik, A.M.; Nijpels, G.; van Haeften, T.W.; Houwing-Duistermaat, J.J.; Boomsma, D.I.; Reiling, E.; van Hove, E.C.; Diamant, M.; Kramer, M.H.; Heine, R.J.; et al. Gene variants in the novel type 2 diabetes loci CDC123/CAMK1D, THADA, ADAMTS9, BCL11A, and MTNR1B affect different aspects of pancreatic β-cell function. Diabetes 2010, 59, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Gaulton, K.J.; Ferreira, T.; Lee, Y.; Raimondo, A.; Magi, R.; Reschen, M.E.; Mahajan, A.; Locke, A.; William Rayner, N.; Robertson, N.; et al. Genetic fine mapping and genomic annotation defines causal mechanisms at type 2 diabetes susceptibility loci. Nat. Genet. 2015, 47, 1415–1425. [Google Scholar] [CrossRef] [PubMed]
- Powell, W.T.; LaSalle, J.M. Epigenetic mechanisms in diurnal cycles of metabolism and neurodevelopment. Hum. Mol. Genet. 2015, 24, R1–R9. [Google Scholar] [CrossRef] [PubMed]
- Winter, J.; Jung, S.; Keller, S.; Gregory, R.I.; Diederichs, S. Many roads to maturity: MicroRNA biogenesis pathways and their regulation. Nat. Cell Biol. 2009, 11, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Filipowicz, W.; Bhattacharyya, S.N.; Sonenberg, N. Mechanisms of post-transcriptional regulation by microRNAs: Are the answers in sight? Nat. Rev. Genet. 2008, 9, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Jeon, K.; Lee, J.T.; Kim, S.; Kim, V.N. MicroRNA maturation: Stepwise processing and subcellular localization. EMBO J. 2002, 21, 4663–4670. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Segura, L.; Perez-Andrade, M.; Miranda-Rios, J. The emerging role of microRNAs in the regulation of gene expression by nutrients. J. Nutrigenet. Nutrigenom. 2013, 6, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Arora, A.; Singh, S.; Bhatt, A.N.; Pandey, S.; Sandhir, R.; Dwarakanath, B.S. Interplay between metabolism and oncogenic process: Role of microRNAs. Transl. Oncogenom. 2015, 7, 11–27. [Google Scholar]
- Fernandez-Hernando, C.; Suarez, Y.; Rayner, K.J.; Moore, K.J. MicroRNAs in lipid metabolism. Curr. Opin. Lipidol. 2011, 22, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Hernando, C.; Ramirez, C.M.; Goedeke, L.; Suarez, Y. MicroRNAs in metabolic disease. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.J.; Rayner, K.J.; Suarez, Y.; Fernandez-Hernando, C. MicroRNAs and cholesterol metabolism. Trends Endocrinol. Metab. 2010, 21, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Trajkovski, M.; Hausser, J.; Soutschek, J.; Bhat, B.; Akin, A.; Zavolan, M.; Heim, M.H.; Stoffel, M. MicroRNAs 103 and 107 regulate insulin sensitivity. Nature 2011, 474, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Daimiel-Ruiz, L.; Klett-Mingo, M.; Konstantinidou, V.; Mico, V.; Aranda, J.F.; Garcia, B.; Martinez-Botas, J.; Davalos, A.; Fernandez-Hernando, C.; Ordovas, J.M. Dietary lipids modulate the expression of miR-107, a miRNA that regulates the circadian system. Mol. Nutr. Food Res. 2015, 59, 552–565. [Google Scholar] [CrossRef] [PubMed]
- Zampetaki, A.; Willeit, P.; Drozdov, I.; Kiechl, S.; Mayr, M. Profiling of circulating microRNAs: From single biomarkers to re-wired networks. Cardiovasc. Res. 2012, 93, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Witmer, P.D.; Lumayag, S.; Kovacs, B.; Valle, D. MicroRNA (miRNA) transcriptome of mouse retina and identification of a sensory organ-specific miRNA cluster. J. Biol. Chem. 2007, 282, 25053–25066. [Google Scholar] [CrossRef] [PubMed]
- Leung, W.K.; He, M.; Chan, A.W.; Law, P.T.; Wong, N. Wnt/β-Catenin activates miR-183/96/182 expression in hepatocellular carcinoma that promotes cell invasion. Cancer Lett. 2015, 362, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Sheng, C.; Huang, L.; Zhang, H.; Cheng, Z.; Zhu, Q. miR-183/-96/-182 cluster is up-regulated in most breast cancers and increases cell proliferation and migration. Breast Cancer Res. 2014, 16. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, C.; Aoyama, K.; Matsumura, N.; Kikuchi-Utsumi, K.; Watabe, M.; Nakaki, T. Rhythmic oscillations of the microRNA miR-96-5p play a neuroprotective role by indirectly regulating glutathione levels. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Na, Y.J.; Sung, J.H.; Lee, S.C.; Lee, Y.J.; Choi, Y.J.; Park, W.Y.; Shin, H.S.; Kim, J.H. Comprehensive analysis of microRNA-mRNA co-expression in circadian rhythm. Exp. Mol. Med. 2009, 41, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, N.; Kulshreshtha, R. miR-191: An emerging player in disease biology. Front. Genet. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ling, S.; Sun, W.; Liu, T.; Li, Y.; Zhong, G.; Zhao, D.; Zhang, P.; Song, J.; Jin, X.; et al. Circulating microRNAs correlated with the level of coronary artery calcification in symptomatic patients. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sundquist, J.; Zoller, B.; Memon, A.A.; Palmer, K.; Sundquist, K.; Bennet, L. Determination of 14 circulating microRNAs in Swedes and Iraqis with and without diabetes mellitus type 2. PLoS ONE 2014, 9, e86792. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Lee, J.E.; Padgett, R.W.; Edery, I. Circadian regulation of a limited set of conserved microRNAs in Drosophila. BMC Genom. 2008, 9. [Google Scholar] [CrossRef] [PubMed]
- Figueredo Dde, S.; Gitai, D.L.; Andrade, T.G. Daily variations in the expression of miR-16 and miR-181a in human leukocytes. Blood Cells Mol. Dis. 2015, 54, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, J.; Hoadley, K.; Kushwaha, D.; Ramakrishnan, V.; Li, S.; Kang, C.; You, Y.; Jiang, C.; Song, S.W.; et al. miR-181d: A predictive glioblastoma biomarker that downregulates mgmt expression. Neuro. Oncol. 2012, 14, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, R.; Loy, P.A.; Sisman, E.; Suyama, E.; Aza-Blanc, P.; Ingermanson, R.S.; Price, J.H.; McDonough, P.M. Identification of microRNAs that control lipid droplet formation and growth in hepatocytes via high-content screening. J. Biomol. Screen. 2010, 15, 798–805. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Celikbilek, M.; Baskol, M.; Taheri, S.; Deniz, K.; Dogan, S.; Zararsiz, G.; Gursoy, S.; Guven, K.; Ozbakir, O.; Dundar, M.; et al. Circulating microRNAs in patients with non-alcoholic fatty liver disease. World J. Hepatol. 2014, 6, 613–620. [Google Scholar] [PubMed]
- Hulsmans, M.; Sinnaeve, P.; Van der Schueren, B.; Mathieu, C.; Janssens, S.; Holvoet, P. Decreased miR-181a expression in monocytes of obese patients is associated with the occurrence of metabolic syndrome and coronary artery disease. J. Clin. Endocrinol. Metab. 2012, 97, E1213–E1218. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Papp, J.W.; Varlamova, O.; Dziema, H.; Russell, B.; Curfman, J.P.; Nakazawa, T.; Shimizu, K.; Okamura, H.; Impey, S.; et al. MicroRNA modulation of circadian-clock period and entrainment. Neuron 2007, 54, 813–829. [Google Scholar] [CrossRef] [PubMed]
- Gatfield, D.; Le Martelot, G.; Vejnar, C.E.; Gerlach, D.; Schaad, O.; Fleury-Olela, F.; Ruskeepaa, A.L.; Oresic, M.; Esau, C.C.; Zdobnov, E.M.; et al. Integration of microRNA miR-122 in hepatic circadian gene expression. Genes Dev. 2009, 23, 1313–1326. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; D’Alessandro, M.; Lee, C. miRNAs are required for generating a time delay critical for the circadian oscillator. Curr. Biol. 2013, 23, 1959–1968. [Google Scholar] [CrossRef] [PubMed]
- Du, N.H.; Arpat, A.B.; De Matos, M.; Gatfield, D. MicroRNAs shape circadian hepatic gene expression on a transcriptome-wide scale. eLife 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y. miR-24 in diabetes. Oncotarget 2015, 6, 16816–16817. [Google Scholar] [CrossRef] [PubMed]
- Deiuliis, J.A. MicroRNAs as regulators of metabolic disease: Pathophysiologic significance and emerging role as biomarkers and therapeutics. Int. J. Obes. 2016, 40, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Zampetaki, A.; Kiechl, S.; Drozdov, I.; Willeit, P.; Mayr, U.; Prokopi, M.; Mayr, A.; Weger, S.; Oberhollenzer, F.; Bonora, E.; et al. Plasma microRNA profiling reveals loss of endothelial miR-126 and other microRNAs in type 2 diabetes. Circ. Res. 2010, 107, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Soh, J.; Iqbal, J.; Queiroz, J.; Fernandez-Hernando, C.; Hussain, M.M. MicroRNA-30c reduces hyperlipidemia and atherosclerosis in mice by decreasing lipid synthesis and lipoprotein secretion. Nat. Med. 2013, 19, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Gil-Zamorano, J.; Martin, R.; Daimiel, L.; Richardson, K.; Giordano, E.; Nicod, N.; Garcia-Carrasco, B.; Soares, S.M.; Iglesias-Gutierrez, E.; Lasuncion, M.A.; et al. Docosahexaenoic acid modulates the enterocyte caco-2 cell expression of microRNAs involved in lipid metabolism. J. Nutr. 2014, 144, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zhang, J.; Du, Y.; Jia, X.; Yang, F.; Si, S.; Wang, L.; Hong, B. MicroRNA-185 modulates low density lipoprotein receptor expression as a key posttranscriptional regulator. Atherosclerosis 2015, 243, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Liu, W.; Pellicane, C.; Sahyoun, C.; Joseph, B.K.; Gallo-Ebert, C.; Donigan, M.; Pandya, D.; Giordano, C.; Bata, A.; et al. Identification of miR-185 as a regulator of de novo cholesterol biosynthesis and low density lipoprotein uptake. J. Lipid Res. 2014, 55, 226–238. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Jia, X.J.; Jiang, H.J.; Du, Y.; Yang, F.; Si, S.Y.; Hong, B. MicroRNAs 185, 96, and 223 repress selective high-density lipoprotein cholesterol uptake through posttranscriptional inhibition. Mol. Cell. Biol. 2013, 33, 1956–1964. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.C.; Zhan, X.R.; Li, X.Y.; Yu, J.J.; Liu, X.M. MicroRNA-185 regulates expression of lipid metabolism genes and improves insulin sensitivity in mice with non-alcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 17914–17923. [Google Scholar] [PubMed]
- Lee, K.H.; Kim, S.H.; Lee, H.R.; Kim, W.; Kim, D.Y.; Shin, J.C.; Yoo, S.H.; Kim, K.T. MicroRNA-185 oscillation controls circadian amplitude of mouse Cryptochrome 1 via translational regulation. Mol. Biol. Cell 2013, 24, 2248–2255. [Google Scholar] [CrossRef] [PubMed]
- Shende, V.R.; Kim, S.M.; Neuendorff, N.; Earnest, D.J. MicroRNAs function as cis- and trans-acting modulators of peripheral circadian clocks. FEBS Lett. 2014, 588, 3015–3022. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Cai, C.; Wang, X.; Liu, M.; Li, X.; Tang, H. MicroRNA-142-3p, a new regulator of RAC1, suppresses the migration and invasion of hepatocellular carcinoma cells. FEBS Lett. 2011, 585, 1322–1330. [Google Scholar] [CrossRef] [PubMed]
- Ohdaira, H.; Sekiguchi, M.; Miyata, K.; Yoshida, K. MicroRNA-494 suppresses cell proliferation and induces senescence in A549 lung cancer cells. Cell Prolif. 2012, 45, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.B.; Chen, X.; Ji, H.; Wu, T.; Lu, H.W.; Zhang, Y.; Li, H.; Li, Y.M. miR-494 is an independent prognostic factor and promotes cell migration and invasion in colorectal cancer by directly targeting PTEN. Int. J. Oncol. 2014, 45, 2486–2494. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.Y.; Yang, J.S.; Kim, B.Y.; Bae, I.H.; Han, Y.H. Ionizing radiation-inducible miR-494 promotes glioma cell invasion through EGFR stabilization by targeting p190B rhoGAP. Biochim. Biophys. Acta 2014, 1843, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.F.; Li, X.Q.; Hu, H.Y.; Li, Y.C.; Cai, Z.; Mei, X.S.; Yu, P.; Nie, L.P.; Zhang, W.; Yu, Z.D.; et al. Functional elucidation of miR-494 in the tumorigenesis of nasopharyngeal carcinoma. Tumour Biol. 2015, 36, 6679–6689. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhao, X.; Wang, L.; Zhang, S.; Cui, M.; He, J. miR-494 suppresses tumor growth of epithelial ovarian carcinoma by targeting IGF1R. Tumour Biol. 2015, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Finnerty, J.R.; Wang, W.X.; Hebert, S.S.; Wilfred, B.R.; Mao, G.; Nelson, P.T. The miR-15/107 group of microRNA genes: Evolutionary biology, cellular functions, and roles in human diseases. J. Mol. Biol. 2010, 402, 491–509. [Google Scholar] [CrossRef] [PubMed]
- Shende, V.R.; Goldrick, M.M.; Ramani, S.; Earnest, D.J. Expression and rhythmic modulation of circulating microRNAs targeting the clock gene Bmal1 in mice. PLoS ONE 2011, 6, e22586. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Zhang, P.; Zhou, L.; Yin, B.; Pan, H.; Peng, X. Clock-controlled miR-142-3p can target its activator, Bmal1. BMC Mol. Biol. 2012, 13. [Google Scholar] [CrossRef] [PubMed]
- Terman, M.; MaMahan, I. Chronotherapy: Resetting Your Inner Clock to Boost Mood, Alertness, and Quality Sleep; Penguin Group: 80 Strand, London WC2R 0RL, UK, 2012; pp. 93–128. [Google Scholar]
- Lam, R.W.; Levitt, A.J.; Levitan, R.D.; Michalak, E.E.; Cheung, A.H.; Morehouse, R.; Ramasubbu, R.; Yatham, L.N.; Tam, E.M. Efficacy of Bright Light Treatment, Fluoxetine, and the Combination in Patients With Nonseasonal Major Depressive Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2016, 73, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Van Maanen, A.; Meijer, A.M.; van der Heijden, K.B.; Oort, F.J. The effects of light therapy on sleep problems: A systematic review and meta-analysis. Sleep Med. Rev. 2015, 29, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Urrestarazu, E.; Iriarte, J. Clinical management of sleep disturbances in Alzheimer’s disease: Current and emerging strategies. Nat. Sci. Sleep 2016, 8, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Singh, H.; Ahmad, N.; Mishra, P.; Tiwari, A. The role of melatonin in diabetes: Therapeutic implications. Arch. Endocrinol. Metab. 2015, 59, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Corbalan-Tutau, M.D.; Madrid, J.A.; Garaulet, M. Timing and duration of sleep and meals in obese and normal weight women. Association with increase blood pressure. Appetite 2012, 59, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Rosselot, A.E.; Hong, C.I.; Moore, S.R. Rhythm and bugs: Circadian clocks, gut microbiota, and enteric infections. Curr. Opin. Gastroenterol. 2016, 32, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Paulose, J.K.; Wright, J.M.; Patel, A.G.; Cassone, V.M. Human Gut Bacteria Are Sensitive to Melatonin and Express Endogenous Circadian Rhythmicity. PLoS ONE 2016, 11, e0146643. [Google Scholar] [CrossRef] [PubMed]
| Gene | Nucleotide Polymorphisms (SNP) | Population | Association | Reference |
|---|---|---|---|---|
| Circadian locomotor output cycles kaput (CLOCK) | rs3749474 | 500 overweight/obese subjects from Spain (Caucasian) recruited into a weight-loss program | Carriers of the minor allele showed higher weight and body mass index (BMI) | [64] |
| 540 men and 560 women with overweight of the Genetics of Lipid Lowering Drugs and Diet Network population (GOLDN) population | Carriers of the minor allele showed higher energy intake | [65] | ||
| rs1801260 | 500 overweight/obese subjects from Spain (Caucasian) recruited into a weight-loss program | Carriers of the minor allele showed higher weight and BMI,shorter sleep and lower response to a weight-loss program | [64] | |
| 1495 overweight/obese subjects from Spain (Caucasian) recruited into a weight-loss program (82.5% females) | Carriers of the minor allele showed a lower response to a weight-loss program, especifically among those subjects characterized as ”emotional eaters”, had higher ghrelin plasma levels and showed an evening chronotype | [66,67] | ||
| 85 overweight caucasian women (43 women carrying the C allele and 42 women with the TT genotype) | Women carrying the C allele showed a tendency to the eveningness and less stable rhtyhms | [69] | ||
| 540 men and 560 women with overweight of the GOLDN population | Carriers of the minor allele showed higher waist circumference when saturated fatty acids (SFA) intake was ≥11.8% | [71] | ||
| 475 subjects with MetS from the CORDIOPREV study | Homozygote subjects for the major allele showed lower insulin levels, lower HOMA-IR (homesotatic model assesment-insulin resistance) and higher insulin sensitivity after 12 month of a low-fat diet | [72] | ||
| 772 participants of the Boston-Puerto Rican Health Study | Minor C allele was protective against high low-density lipoprotein (LDL)-cholesterol plasma levels in the group with high SFA intake | [73] | ||
| 2485 subjects enrolled in the baseline surveys of the Japan Multi-institutional Collaborative Cohort (J-MICC) Study | Minor C allele was associated with higher risk of prevalent diabetes, especially in lean subjects | [74] | ||
| rs4580704 | 540 men and 560 women overweight of the GOLDN population | Carriers of the minor allele showed lower weight, BMI, blood pressure, postpandrial triglyceridemia and risk of type 2 diabetes mellitus (T2DM) and hypertension. A gene-diet interaction exists with monounsaturated fatty acids (MUFA) intake and T2DM-related parameters | [71] | |
| 540 men and 560 women overweight of the GOLDN population | Carriers of the minor allele showed lower energy intake | [65] | ||
| 7098 subjects with high cardiovascular risk from the PREDIMED trial | The G allele was associated with lower weight, BMI, and waist circumference and with lower fasting glucose in non-diabetic subjects. The protective effect of the G allele againts T2DM was higher in the Mediterranean Diet intervention group | [75] | ||
| CRY1 | rs2287161 | 728 Mediterranean (81% women) and 820 North American (50.5% women) overweight subjects | Carriers of the minor C allele showed higher HOMA-IR when carbohydrate intake was >41.65% of total energy | [76] |
| rs12315175 | Meta-analysis using 3 cohorts composed by participants of different ethnic origin | This variant is associated with a lower risk of T2DM | [82] | |
| CRY2 | rs11605924 | 3210 unrelated Chinese Hans from Beijing and Shanghai | This SNP has been assocaited with lower fasting glucose levels and risk of T2DM | [77] |
| 4654 non-diabetic Finland subjects from the PPP-Botnia Study. | This variant is associated with a better β-cell function | [80] | ||
| 721 obese individuals following a weight-loss program with diets with different fat and protein composition from the POUND LOSS trial (Harvard School of Public Health and Brigham and Women’s Hospital in Boston, MA, USA) | The variant allele was associated with a lower respiratory quotient and higher resting metabolic rate | [81] | ||
| Meta-analysis including 28,190 participants of European descendent from CHARGE consortium | The previously reported association of this SNP and fasting glucose was not replicated in this meta-analysis | [78] | ||
| rs2292912 | Meta-analysis using 3 cohorts composed by participants of different ethnic origin | This variant is associated with a higher risk of T2DM | [82] | |
| PER2 | rs4663302 | 454 overweight/obese subjects from Spain (Caucasian) recruited into a weight-loss program | Homozygote carriers of the minor allele showed higher abdominal obesity and were prone to withdraw from a weight-reduction program | [84] |
| rs2304672 | 454 overweight/obese subjects from Spain (Caucasian) recruited into a weight-loss program | Carriers of the G allele showed lower waist-to-hip ratio but they also showed eating behaviors alterations | [85] | |
| rs7602358 | 3512 subjects with from the Pujabi cohort resident in the United Kingdom and Pakistan | The G allele was negatively associated with T2DM | [82] | |
| Brain and muscle ARNT-like protein 1 (BMAL1) | rs11022775 | 3512 subjects with from the Pujabi cohort resident in the United Kingdom and Pakistan | The T allele is associated with an increased risk of T2DM | [82] |
| rs6486121 | 1304 individuals from 424 British T2D families from the Diabetes in Families (DIF) study collection | Variant allele is nominally associated with hypertension | [86] | |
| rs7950226 | 1304 individuals from 424 British T2D families from the Diabetes in Families (DIF) study collection | Variant allele is nominally associated with T2DM | [86] | |
| REV-ERBα1 | rs2314339 | 1402 Mediterranean (82% women) and 810 North American (48.2% women) overweight subjects | Carriers of the minor allele showed lower waist circumference, BMI and abdominal adiposity. An interaction with MUFA intake was found for BMI | [87] |
| rs939347 | 1197 Spanish subjects, 779 of them obese | AA genotype was most frequent among men obese subjects | [88] | |
| rs2071427 | 3480 adolescents and adults from three independent population-based studies (MONICA, MONA-LISA and HELENA) | The minor T allele is associated with higher BMI in adults and adolescents | [89] | |
| MTNR1B | rs1387153 | GWAS data from 2151 nondiabetic French subjects | T allele was associated with higher fastin glucose levels and higher risk of developing hyperglycemia and T2DM | [90] |
| Meta-analysis including 28,190 participants of European descendent from CHARGE consortium | T allele was associated with higher fastin glucose | [78] | ||
| 2222 Asian subjects (1201 T2DM and 1021 Control) | There was no association between this SNP and fasting glucose levels | [95] | ||
| rs10830963 | GWAS conducted in 6 cohorts of European origin | The risk allele was associated with higher fasting glucose, lower insulin secretion and also showed a weak association with the risk of developing T2DM | [91] | |
| 10 GWAS involving a total of 36,610 individuals of European descent | G allele is associated with higher fasting glucose, lower β-cell function and the risk of T2DM | [92] | ||
| 346 Caucasians, 218 African-Americans, and 217 Hispanics obese children and adolescents | The MTNR1B rs10830963 variant was associated with higher fasting glucose levels and lower dynamic β-cell response in Caucasians and Hispanics | [93] | ||
| Meta-analysis including 28,190 participants of European descendent from CHARGE consortium | G allele is associated with higher fasting glucose and HOMA-IR | [78] | ||
| 721 obese individuals following a weight-loss program with diets with different fat and protein composition from the POUND LOSS trial (USA) | Carriers of the G allele showed higher respiratory quotient in relation with the dietary fat | [81] | ||
| A meta-analysis including 113,025 T2DM patients and 199,997 controls from 38 studies | There was an association between this SNP and T2DM risk in Caucasian but not Asian subjects | [94] | ||
| 2222 Asian subjects (1201 T2DM and 1021 Control) | There was no association between this SNP and fasting glucose levels | [95] | ||
| rs1374645 | 2222 Asian subjects (1201 T2DM and 1021 Control) | This SNP was associated with lower fasting glucose in subjects with low BMI | [95] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Micó, V.; Díez-Ricote, L.; Daimiel, L. Nutrigenetics and Nutrimiromics of the Circadian System: The Time for Human Health. Int. J. Mol. Sci. 2016, 17, 299. https://doi.org/10.3390/ijms17030299
Micó V, Díez-Ricote L, Daimiel L. Nutrigenetics and Nutrimiromics of the Circadian System: The Time for Human Health. International Journal of Molecular Sciences. 2016; 17(3):299. https://doi.org/10.3390/ijms17030299
Chicago/Turabian StyleMicó, Víctor, Laura Díez-Ricote, and Lidia Daimiel. 2016. "Nutrigenetics and Nutrimiromics of the Circadian System: The Time for Human Health" International Journal of Molecular Sciences 17, no. 3: 299. https://doi.org/10.3390/ijms17030299
APA StyleMicó, V., Díez-Ricote, L., & Daimiel, L. (2016). Nutrigenetics and Nutrimiromics of the Circadian System: The Time for Human Health. International Journal of Molecular Sciences, 17(3), 299. https://doi.org/10.3390/ijms17030299