Consumption of Bilberries Controls Gingival Inflammation
Abstract
:1. Introduction
2. Results and Discussion
| Individual | 250 g Bilberry | 500 g Bilberry | Placebo | SOC | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BL (%) | EP (%) | Ch (%) | BL (%) | EP (%) | Ch (%) | BL (%) | EP (%) | Ch (%) | BL (%) | EP (%) | Ch (%) | |
| 1 | 13 | 6 | 54 | 34 | 18 | 47 | 37 | 15 | 59 | 53 | 18 | 66 |
| 2 | 23 | 11 | 52 | 48 | 33 | 31 | 54 | 54 | 0 | 32 | 14 | 56 |
| 3 | 28 | 17 | 39 | 30 | 8 | 73 | 25 | 25 | 0 | 43 | 27 | 37 |
| 4 | 25 | 14 | 44 | 29 | 15 | 48 | 39 | 26 | 33 | 33 | 4 | 88 |
| 5 | 33 | 21 | 36 | 24 | 8 | 67 | 31 | 21 | 32 | 33 | 12 | 64 |
| 6 | 21 | 11 | 48 | 39 | 16 | 59 | 26 | 14 | 46 | 36 | 20 | 44 |
| 7 | 23 | 19 | 17 | 39 | 10 | 74 | 31 | 16 | 48 | 37 | 15 | 59 |
| 8 | 23 | 15 | 35 | 25 | 7 | 72 | 26 | 18 | 31 | 50 | 27 | 46 |
| mean | 25 | 15 | 41 | 34 | 14 | 59 | 34 | 24 | 31 | 40 | 17 | 58 |
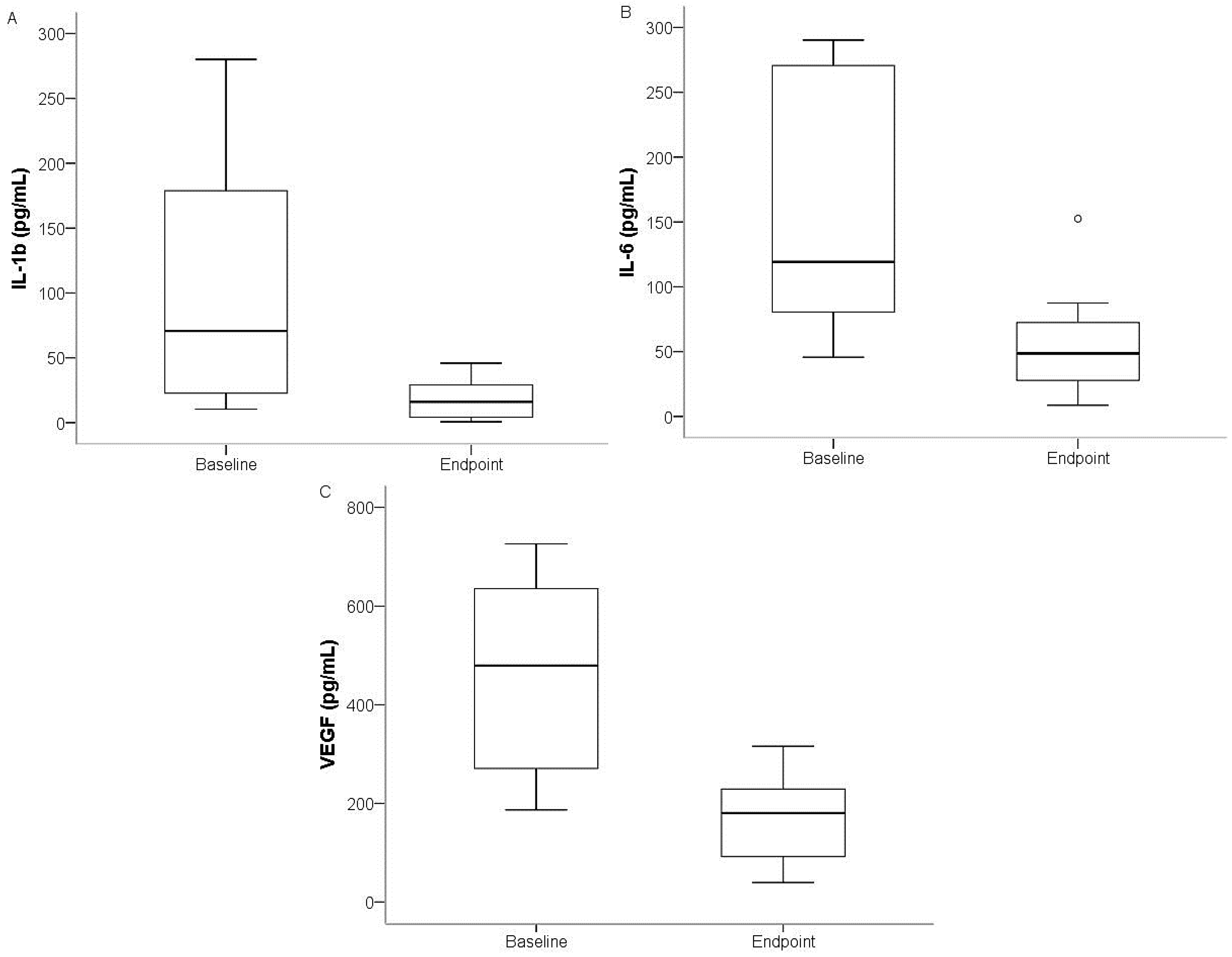
| Cytokine | Median pg/mL | 25% pg/mL | 75% pg/mL | Median pg/mL | 25% pg/mL | 75% pg/mL | Median Ratio | p Value |
|---|---|---|---|---|---|---|---|---|
| 250 g bilberry baseline | 250 g bilberry study end point | |||||||
| IL-1ra | 0.7 | 0.2 | 6.9 | 0.4 | 0.0 | 35.8 | 1.8 | 0.715 |
| IL-1β | 18.0 | 10.7 | 144.2 | 29.6 | 6.2 | 146.0 | 0.6 | 0.715 |
| IL-6 | 84.8 | 52.9 | 96.0 | 67.7 | 34.3 | 106.5 | 1.3 | 0.715 |
| IL-12 | 0.8 | 0.8 | 1.4 | 0.8 | 0.6 | 1.4 | 1.0 | 0.317 |
| IP-10 | 371.5 | 235.9 | 689.0 | 522.5 | 196.7 | 1392.2 | 0.7 | 0.735 |
| MIP-1α | 3.0 | 1.9 | 4.7 | 2.0 | 0.9 | 2.7 | 1.5 | 0.180 |
| PDGF | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 1.7 | 0.0 | 0.068 |
| VEGF | 303.7 | 228.9 | 575.3 | 266.8 | 224.9 | 483.1 | 1.1 | 0.263 |
| 500 g bilberry baseline | 500 g bilberry study end point | |||||||
| IL-1ra | 0.5 | 0.1 | 2.9 | 0.2 | 0.0 | 0.6 | 2.5 | 0.310 |
| IL-1β | 70.6 | 21.8 | 220.5 | 16.1 | 4.1 | 31.9 | 4.4 | 0.025 |
| IL-6 | 119.3 | 73.9 | 280.5 | 48.7 | 25.2 | 80.0 | 2.4 | 0.012 |
| IL-12 | 1.5 | 1.2 | 2.3 | 0.7 | 0.6 | 1.9 | 2.1 | 0.310 |
| IP-10 | 389.3 | 135.4 | 990.3 | 176.3 | 113.6 | 659.2 | 2.2 | 0.069 |
| MIP-1α | 3.2 | 1.8 | 7.1 | 1.1 | 0.8 | 2.7 | 2.9 | 0.107 |
| PDGF | 0.8 | 0.0 | 1.7 | 1.5 | 0.1 | 3.5 | 0.5 | 0.237 |
| VEGF | 479.6 | 250.6 | 680.5 | 180.6 | 87.0 | 238.6 | 2.7 | 0.017 |
| Placebo baseline | Placebo study end point | |||||||
| IL-1β | 70.9 | 21.8 | 196.5 | 30.5 | 23.4 | 88.1 | 2.3 | 0.674 |
| IL-6 | 1.5 | 0.3 | 2.2 | 0.6 | 0.3 | 1.6 | 2.5 | 0.889 |
| VEGF | 330.9 | 299.6 | 592.4 | 305.1 | 188.7 | 723.2 | 1.1 | 0.889 |
3. Experimental Section
3.1. Study Individuals
3.2. Bleeding on Probing
3.3. Sampling from Gingival Crevicular Fluid for Cytokine Analysis
3.4. Sampling from Blood for Cytokine Analysis
3.5. Determination of Cytokines Using Bio-Plex Assays
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Abrahamsson, K.H.; Koch, G.; Norderyd, O.; Romao, C.; Wennstrom, J.L. Periodontal conditions in a Swedish city population of adolescents: A cross-sectional study. Swed. Dent. J. 2006, 30, 25–34. [Google Scholar] [PubMed]
- Loe, H.; Silness, J. Periodontal disease in pregnancy. I. Prevalence and severity. Acta Odontol. Scand. 1963, 21, 533–551. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.; Tabeta, K.; Sugita, N.; Yoshie, H. Profiling biomarkers in gingival crevicular fluid using multiplex bead immunoassay. Arch. Oral Biol. 2013, 58, 724–730. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Tulipani, S.; Casoli, T.; di Stefano, G.; Gonzalez-Paramas, A.M.; Santos-Buelga, C.; Busco, F.; Quiles, J.L.; Cordero, M.D.; et al. One-month strawberry-rich anthocyanin supplementation ameliorates cardiovascular risk, oxidative stress markers and platelet activation in humans. J. Nutr. Biochem. 2014, 25, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Daniels, J.A.; Mulligan, C.; McCance, D.; Woodside, J.V.; Patterson, C.; Young, I.S.; McEneny, J. A randomised controlled trial of increasing fruit and vegetable intake and how this influences the carotenoid concentration and activities of PON-1 and LCAT in HDL from subjects with type 2 diabetes. Cardiovasc. Diabetol. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Prymont-Przyminska, A.; Zwolinska, A.; Sarniak, A.; Wlodarczyk, A.; Krol, M.; Nowak, M.; de Graft-Johnson, J.; Padula, G.; Bialasiewicz, P.; Markowski, J.; et al. Consumption of strawberries on a daily basis increases the non-urate 2,2-diphenyl-1-picryl-hydrazyl (DPPH) radical scavenging activity of fasting plasma in healthy subjects. J. Clin. Biochem. Nutr. 2014, 55, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Kolehmainen, M.; Mykkanen, O.; Kirjavainen, P.V.; Leppanen, T.; Moilanen, E.; Adriaens, M.; Laaksonen, D.E.; Hallikainen, M.; Puupponen-Pimia, R.; Pulkkinen, L.; et al. Bilberries reduce low-grade inflammation in individuals with features of metabolic syndrome. Mol. Nutr. Food Res. 2012, 56, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
- LaPlante, K.L.; Sarkisian, S.A.; Woodmansee, S.; Rowley, D.C.; Seeram, N.P. Effects of cranberry extracts on growth and biofilm production of Escherichia Coli and staphylococcus species. Phytother. Res. 2012, 26, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, S.; Imfeld, T.; Schicht, O.; Rath, C.; Persson, R.E.; Persson, G.R. The impact of the stone age diet on gingival conditions in the absence of oral hygiene. J. Periodontol. 2009, 80, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Chapple, I.L.; Milward, M.R.; Ling-Mountford, N.; Weston, P.; Carter, K.; Askey, K.; Dallal, G.E.; de Spirt, S.; Sies, H.; Patel, D.; et al. Adjunctive daily supplementation with encapsulated fruit, vegetable and berry juice powder concentrates and clinical periodontal outcomes: A double-blind RCT. J. Periodontol. 2012, 39, 62–72. [Google Scholar] [CrossRef]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Loe, H.; Silness, J. Tissue reactions to a new gingivectomy pack. Oral Surg. Oral Med. Oral Pathol. 1961, 14, 1305–1314. [Google Scholar] [CrossRef] [PubMed]
- Masada, M.P.; Persson, R.; Kenney, J.S.; Lee, S.W.; Page, R.C.; Allison, A.C. Measurement of interleukin-1 α and -1 β in gingival crevicular fluid: Implications for the pathogenesis of periodontal disease. J. Periodontal Res. 1990, 25, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Ericsson, J.S.; Abrahamsson, K.H.; Ostberg, A.L.; Hellstrom, M.K.; Jonsson, K.; Wennstrom, J.L. Periodontal health status in swedish adolescents: An epidemiological, cross-sectional study. Swed. Dent. J. 2009, 33, 131–139. [Google Scholar] [PubMed]
- Puupponen-Pimia, R.; Nohynek, L.; Alakomi, H.L.; Oksman-Caldentey, K.M. Bioactive berry compounds-novel tools against human pathogens. Appl. Microbiol. Biotechnol. 2005, 67, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Karlsen, A.; Paur, I.; Bohn, S.K.; Sakhi, A.K.; Borge, G.I.; Serafini, M.; Erlund, I.; Laake, P.; Tonstad, S.; Blomhoff, R. Bilberry juice modulates plasma concentration of NF-κb related inflammatory markers in subjects at increased risk of CVD. Eur. J. Nutr. 2010, 49, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Prapulla, D.V.; Sharma, A.; Sujatha, P.B. Gingival crevicular fluid and serum vascular endothelial growth factor: Their relationship in periodontal health, disease and after treatment. Cytokine 2011, 54, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Prapulla, D.V.; Sujatha, P.B.; Pradeep, A.R. Gingival crevicular fluid VEGF levels in periodontal health and disease. J. Periodontol. 2007, 78, 1783–1787. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Widén, C.; Coleman, M.; Critén, S.; Karlgren-Andersson, P.; Renvert, S.; Persson, G.R. Consumption of Bilberries Controls Gingival Inflammation. Int. J. Mol. Sci. 2015, 16, 10665-10673. https://doi.org/10.3390/ijms160510665
Widén C, Coleman M, Critén S, Karlgren-Andersson P, Renvert S, Persson GR. Consumption of Bilberries Controls Gingival Inflammation. International Journal of Molecular Sciences. 2015; 16(5):10665-10673. https://doi.org/10.3390/ijms160510665
Chicago/Turabian StyleWidén, Cecilia, Michael Coleman, Sladjana Critén, Pernilla Karlgren-Andersson, Stefan Renvert, and G. Rutger Persson. 2015. "Consumption of Bilberries Controls Gingival Inflammation" International Journal of Molecular Sciences 16, no. 5: 10665-10673. https://doi.org/10.3390/ijms160510665
APA StyleWidén, C., Coleman, M., Critén, S., Karlgren-Andersson, P., Renvert, S., & Persson, G. R. (2015). Consumption of Bilberries Controls Gingival Inflammation. International Journal of Molecular Sciences, 16(5), 10665-10673. https://doi.org/10.3390/ijms160510665





