Osmotic Dehydration of Apples in a Saccharose Solution Containing Fragrant Agrimony or Rosehip Extract
Abstract
1. Introduction
2. Results and Discussion
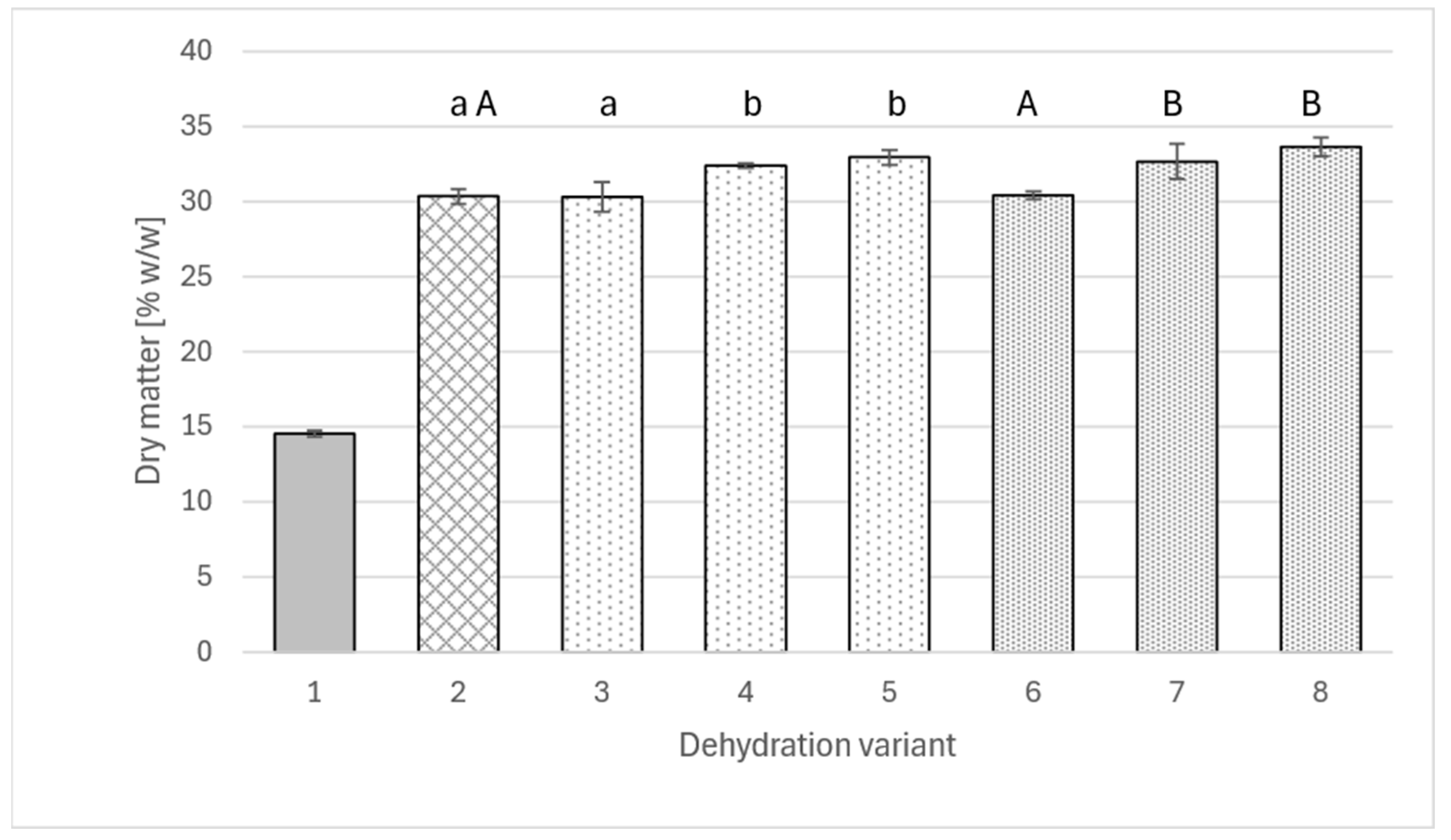
2.1. Dry Matter Content, Water Loss and Solids Gain
2.2. Polyphenol Composition of the Extracts
2.3. Transfer of Polyphenols During the Osmotic Dehydration of Apples
3. Materials and Methods
3.1. Materials
3.2. The Method of Obtaining Extracts
3.3. Osmotic Dehydration
3.4. Dry Matter Content
- m0—weight of sample before drying [g]
- mk—weight of sample after drying [g]
3.5. Water Loss and Solid Gain Calculation
- m0—weight of sample before osmotic dehydration [g]
- mk—weight of sample after osmotic dehydration [g]
- s0—solids content before osmotic dehydration [g of dry matter per g]
- sk—solids content after osmotic dehydration [g of dry matter per g]
3.6. Phenolic Extraction
3.7. Identification and Quantification Phenolic Compounds by UHPLC-DAD-MS
3.8. Flavanols: Proanthocyanidins and Free Catechins
3.9. Phenolic Compounds Spectrophotometric Method Folin-Ciocalteu
3.10. Experimental Design and Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Asghari, A.; Zongo, P.A.; Osse, E.F.; Aghajanzadeh, S.; Raghavan, V.; Khalloufi, S. Review of osmotic dehydration: Promising technologies for enhancing products’ attributes, opportunities, and challenges for the food industries. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13346. [Google Scholar] [CrossRef]
- Nambiar, S.S.; Basu, A.; Shetty, N.P.; Rastogi, N.K.; Prapulla, S.G. Infusion of fructooligosaccharide in Indian gooseberry (Emblica officinalis) fruit using osmotic treatment and its effect on the antioxidant activity of the fruit. J. Food Eng. 2016, 190, 139–146. [Google Scholar] [CrossRef]
- Wang, X.; Kapoor, R.; Feng, H. Exploring the effects of vacuum and ultrasound treatments on calcium fortification in osmotically dehydrated apple slices. LWT-Food Sci. Technol. 2023, 187, 115386. [Google Scholar] [CrossRef]
- Martínez-Sánchez, C.E.; Solis-Ramos, A.C.; Rodríguez-Miranda, J.; Juárez-Barrientos, J.M.; Ramírez-Rivera, E.J.; Ruiz-López, I.I.; Gómez-Aldapa, C.A.; Herman-Lara, E. Evaluation of ascorbic acid impregnation by ultrasound-assisted osmotic dehydration in plantain. J. Food Process. Preserv. 2022, 46, e16839. [Google Scholar] [CrossRef]
- Rana, A.; Samtiya, M.; Dhewa, T.; Mishra, V.; Aluko, R.E. Health benefits of polyphenols: A concise review. J. Food Biochem. 2022, 46, e14264. [Google Scholar] [CrossRef] [PubMed]
- Rathee, P.; Sehrawat, R.; Rathee, P.; Khatkar, A.; Akkol, E.K.; Khatkar, S.; Redhu, N.; Türkcanoglu, G.; Sobarzo-Sánchez, E. Polyphenols: Natural preservatives with promising applications in food, cosmetics and pharma industries; problems and toxicity associated with synthetic preservatives; impact of misleading advertisements; recent trends in preservation and legislation. Materials 2023, 16, 4793. [Google Scholar] [CrossRef]
- Adsare, S.R.; Bellary, A.N.; Sowbhagya, H.B.; Baskaran, R.; Prakash, M.; Rastogi, N.K. Osmotic treatment for the impregnation of anthocyanin in candies from Indian gooseberry (Emblica officinalis). J. Food Eng. 2016, 175, 24–32. [Google Scholar] [CrossRef]
- Channannavar, S.S.; Thippanna, K.S.; Naik, K.R.; Sabarad, A.I.; Yadachi, S.; Kareem, M.A. Studies on mass transfer kinetics of kokum based anthocyaninin fused osmo-dehydrated pineapple cubes. Biol. Forum—Int. J. 2022, 14, 818–823. [Google Scholar]
- Bellary, A.N.; Indiramma, A.R.; Prakash, M.; Baskaran, R.; Rastogi, N.K. Anthocyanin infused watermelon rind and its stability during storage. Innov. Food Sci. Emerg. Technol. 2016, 33, 554–562. [Google Scholar] [CrossRef]
- Hernández-Carranza, P.; Heredia-Soberanes, K.; Ruiz-López, I.I.; Ochoa-Velasco, C.E. Effect of impregnation-osmodehydration with Hibiscus sabdariffa extracts on the bioactive compounds and sensory acceptance of apple wedges: Fresh, convective dried, and stored. J. Food Process. Preserv. 2022, 46, e17110. [Google Scholar] [CrossRef]
- Cichowska, J.; Samborska, K.; Kowalska, H. Influence of chokeberry juice concentrate used as osmotic solution on the quality of differently dried apples during storage. Eur. Food Res. Technol. 2018, 244, 1773–1782. [Google Scholar] [CrossRef]
- Samborska, K.; Eliasson, L.; Marzec, A.; Kowalska, J.; Piotrowski, D.; Lenart, A.; Kowalska, H. The effect of adding berry fruit juice concentrates and by-product extract to sugar solution on osmotic dehydration and sensory properties of apples. J. Food Sci. Technol. 2019, 56, 1927–1938. [Google Scholar] [CrossRef] [PubMed]
- Castagnini, J.M.; Betoret, N.; Betoret, E.; Fito, P. Vacuum impregnation and air drying temperature effect on individual anthocyanins and antiradical capacity of blueberry juice included into an apple matrix. LWT Food Sci. Technol. 2015, 64, 1289–1296. [Google Scholar] [CrossRef]
- Medeiros, R.A.B.; Silva Júnior, E.V.; Silva, J.H.F.; Cunha Ferreira Neto, O.; Brandão, S.C.R.; Barros, Z.M.P.; Rocha, O.R.S.; Azoubel, P.M. Effect of different grape residues polyphenols impregnation techniques in mango. J. Food Eng. 2019, 262, 1–8. [Google Scholar] [CrossRef]
- Kopera, M.; Mitek, M. Effect of osmotic dehydration on polyphenols content in dried pears (Pyrus communis i Pyrus pyrifolia). Food. Sci. Technol. Qual. 2007, 5, 213–221. [Google Scholar]
- Yazidi, R.; Yeddes, W.; Rybak, K.; Witrowa-Rajchert, D.; Wannes, W.A.; Hammami, M.; Hessini, K.; Tounsi, M.S.; Nowacka, M. Osmotic dehydration of orange fruits in sucrose and prickly pear molasses solutions: Mass transfer and quality of dehydrated products. Pol. J. Food Nutr. Sci. 2024, 74, 340–349. [Google Scholar] [CrossRef]
- Bellary, A.N.; Sowbhagya, H.B.; Rastogi, N.K. Osmotic dehydration assisted impregnation of curcuminoids in coconut slices. J. Food Eng. 2011, 105, 453–459. [Google Scholar] [CrossRef]
- Kawiji, K.; Siahaan, A.H.J.; Manuhara, G.J.; Atmaka, W. Sucrose osmotic treatment effect on moisture, antioxidant, texture, and sensory properties of ginger extract-imersed coconut chips. J. Appl. Agric. Sci. Technol. 2024, 8, 144–158. [Google Scholar] [CrossRef]
- Kowalska, H.; Marzec, A.; Domian, E.; Masiarz, E.; Ciurzyńska, A.; Galus, S.; Małkiewicz, A.; Lenart, A.; Kowalska, J. Physical and sensory properties of Japanese quince chips obtained by osmotic dehydration in fruit juice concentrates and hybrid drying. Molecules 2020, 25, 5504. [Google Scholar] [CrossRef]
- Han, Y.; Sun, R. Blueberry Functional Food Pretreated by Pulse Electric Field and Air-Dried by Osmotic Dehydration and Preparation and Application Thereof. Chinese Patent CN112690420A. 23 April 2021. Available online: https://worldwide.espacenet.com/patent/search/family/075512762/publication/CN112690420A?q=CN112690420A (accessed on 18 September 2025).
- Galus, S.; Rybak, K.; Dadan, M.; Witrowa-Rajchert, D.; Nowacka, M. The effect of the use of unconventional solutions for osmotic dehydration on selected properties of fresh-cut oranges. Foods 2025, 14, 468. [Google Scholar] [CrossRef]
- Demir, N.; Alpaslan, M. Determination of impregnation parameters and volatile components in vacuum impregnated apricots. Heliyon 2024, 10, e28294. [Google Scholar] [CrossRef]
- Stavropoulou, N.A.; Giannakourou, M.C. Combined effect of bioactive compound enrichment using Rosa damascena distillation side streams and an optimized osmotic treatment on the stability of frozen oyster mushrooms. Appl. Sci. 2023, 13, 9734. [Google Scholar] [CrossRef]
- Çiçek, H.; Kaya, H.S.; Kilic, C.; Savas, M.; Ravichandran, S. Medical effects of products obtained from wild rose plant. J. Chem. Nutr. Biochem. 2022, 3, 59–64. [Google Scholar] [CrossRef]
- Klewicka, E.; Piekarska-Radzik, L.; Milala, J.; Klewicki, R.; Sójka, M.; Rosół, N.; Otlewska, A.; Matysiak, B. Antagonistic activity of lactic acid bacteria and Rosa rugosa Thunb. Pseudo-fruit extracts against Staphylococcus spp. strains. Appl. Sci. 2022, 12, 4005. [Google Scholar] [CrossRef]
- Milala, J.; Piekarska-Radzik, L.; Sójka, M.; Klewicki, R.; Matysiak, B.; Klewicka, E. Rosa spp. extracts as a factor that limits the growth of Staphylococcus spp. bacteria, a food contaminant. Molecules 2021, 26, 4590. [Google Scholar] [CrossRef] [PubMed]
- Piekarska-Radzik, L.; Milala, J.; Klewicki, R.; Sójka, M.; Żyżelewicz, D.; Matysiak, B.; Klewicka, E. Extract from Rosa spp. as a factor influencing the growth rate of coagulase-negative Staphylococcus strains. Molecules 2025, 30, 1443. [Google Scholar] [CrossRef] [PubMed]
- Cendrowski, A.; Kraśniewska, K.; Przybył, J.L.; Zielińska, A.; Kalisz, S. Antibacterial and antioxidant activity of extracts from rose fruits (Rosa rugosa). Molecules 2020, 25, 1365. [Google Scholar] [CrossRef]
- Granica, S.; Kluge, H.; Horn, G.; Matkowski, A.; Kiss, A.K. The phytochemical investigation of Agrimonia eupatoria L. and Agrimonia procera Wallr. as valid sources of Agrimoniae herba—The pharmacopoeial plant material. J. Pharm. Biomed. Anal. 2015, 114, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Karlińska, E.; Romanowska, B.; Kosmala, M. The aerial parts of Agrimonia procera Wallr. and Agrimonia eupatoria L. as a source of polyphenols, and especially agrimoniin and flavonoids. Molecules 2021, 26, 7706. [Google Scholar] [CrossRef]
- Kashchenko, N.; Olennikov, D.N. Phenolome of Asian agrimony tea (Agrimonia asiatica Juz., Rosaceae): LC-MS profile, α-glucosidase inhibitory potential and stability. Foods 2020, 9, 1348. [Google Scholar] [CrossRef]
- Kim, C.Y.; Yu, Q.M.; Kong, H.-J.; Lee, J.-Y.; Yang, K.-M.; Seo, J.-S. Antioxidant and anti-inflammatory activities of Agrimonia pilosa Ledeb. extract. Evid. Based Complement. Altern. Med. 2020, 2020, 8571207. [Google Scholar] [CrossRef] [PubMed]
- Santos, T.L.; Costa, G.; Pinto Ferreira, J.; Liberal, J.; Francisco, V.; Paranthos, A.; Crus, M.T.; Castelo-Branco, M.; Figueiredo, V.; Batista, M.T. Antioxidant, anti-inflammatory, and analgesic activities of Agrimonia eupatoria L. infusion. Evid. Based Complement. Altern. Med. 2017, 2017, 8309894. [Google Scholar] [CrossRef]
- Gräber, T.; Kluge, H.; Granica, S.; Horn, G.; Kalbitz, J.; Brandsch, C.; Breittenstein, A.; Brütting, C.; Stangl, G.I. Agrimonia procera exerts antimicrobial effects, modulates the expression of defensins and cytokines in colonocytes and increases the immune response in lipopolysaccharide-challenged piglets. BMC Vet. Res. 2018, 14, 346. [Google Scholar] [CrossRef]
- Muruzović, M.Z.; Mladenović, K.G.; Stefanović, O.D.; Vasić, S.M.; Comić, L.R. Extracts of Agrimonia eupatoria L. as sources of biologically active compounds and evaluation of their antioxidant, antimicrobial, and antibiofilm activities. J. Food Drug Anal. 2016, 24, 539–547. [Google Scholar] [CrossRef]
- Bator, P.; Razik, M.; Rozwadowska, P.; Ramian, J.; Rybak, J.; Magiera, B.; Magiera, K.; Grabowska-Szczurek, M.; Grabarczyk, A.; Razik, W. An apple a day keeps the doctor away?—A review of health benefits of apples. J. Educ. Health Sport 2024, 55, 73–86. [Google Scholar] [CrossRef]
- Lech, K.; Figiel, A.; Michalska, A.; Wojdyło, A.; Nowicka, P. The effect of selected fruit juice concentrates used as osmotic agents on the drying kinetics and chemical properties of vacuum-microwave drying of pumpkin. J. Food Qual. 2018, 2018, 7293932. [Google Scholar] [CrossRef]
- Rózek, A.; García-Pérez, J.V.; López, F.; Güell, C.; Ferrando, M. Infusion of grape phenolics into fruits and vegetables by osmotic treatment: Phenolic stability during air drying. J. Food Eng. 2010, 99, 142–150. [Google Scholar] [CrossRef]
- Karonen, M. Insights into polyphenol–lipid interactions: Chemical methods, molecular aspects and their effects on membrane structures. Plants 2022, 11, 1809. [Google Scholar] [CrossRef]
- Kowalska, H.; Trusinska, M.; Rybak, K.; Wiktor, A.; Witrowa-Rajchert, D.; Nowacka, M. Shaping the properties of osmo-dehydrated strawberries in fruit juice concentrates. Appl. Sci. 2023, 13, 2728. [Google Scholar] [CrossRef]
- Ramos-Morales, M.; Aguirre-García, M.; Cortés-Zavaleta, O.; Ruiz-Espinosa, H.; Estévez-Sánchez, K.H.; Ochoa-Velasco, C.E.; Ruiz-López, I.I. Improving the bioactive content in honeydew melon by impregnation with Hibiscus extract/sucrose solutions: A coupled mass transfer analysis. Food Bioprod. Process. 2024, 144, 1–12. [Google Scholar] [CrossRef]
- Okuda, T.; Yoshida, T.; Kuwahara, M.; Memon, M.; Shingu, T. Agrimoniin and potentillin, an ellagitannin dimer and monomer having an α-glucose core. J. Chem. Soc. 1982, 163–164. [Google Scholar] [CrossRef]
- Chen, P.; Chen, F.; Guo, Z.; Lei, J.; Zhou, B. Recent advancement in bioeffect, metabolism, stability, and delivery systems of apigenin, a natural flavonoid compound: Challenges and perspectives. Front. Nutr. 2023, 26, 1221227. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Lin, F.; Chen, Y.; Dou, Y.; Li, T.; Jin, X.; Song, J.; Wang, F. Luteolin-7-O-β-D-glucuronide ameliorates cerebral ischemic injury: Involvement of RIP3/MLKL signaling pathway. Molecules 2024, 29, 1665. [Google Scholar] [CrossRef]
- Seo, S.-W.; Choi, S.H.; Hong, J.-K.; Kim, K.M.; Kang, S.C.; Yoon, I.-S. Pharmacokinetics and extensive intestinal first-pass effects of apigenin and its active metabolite, apigenin-7-O-glucuronide, in rats. J. Pharm. Investig. 2024, 54, 467–481. [Google Scholar] [CrossRef]
- Kucner, A.; Klewicki, R.; Sójka, M. The influence of selected osmotic dehydration and pretreatment parameters on dry matter and polyphenol content in highbush blueberry (Vaccinium corymbosum L.) fruits. Food Bioprocess Technol. 2013, 6, 2031–2047. [Google Scholar] [CrossRef]
- Renard, C.M.G.C.; Baron, A.; Guyot, S.; Drilleau, J.F. Interactions between apple cell walls and native apple polyphenols: Quantification and some consequences. Int. J. Biol. Macromol. 2001, 29, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Marstrand, K.; Campbell-Tofte, J. The role of rose hip (Rosa canina L) powder in alleviating arthritis pain and inflammation—Part II animal and human studies. Bot. Targets Ther. 2016, 6, 59–73. [Google Scholar] [CrossRef]
- Masztalerz, K.; Lech, K.; Wojdyło, A.; Nowicka, P.; Michalska-Ciechanowska, A.; Figiel, A. The impact of the osmotic dehydration process and its parameters on the mass transfer and quality of dried apples. Dry. Technol. 2020, 39, 1074–1086. [Google Scholar] [CrossRef]
- Sójka, M.; Hejduk, A.; Piekarska-Radzik, L.; Ścieszka, S.; Grzelak-Błaszczyk, K.; Klewicka, E. Antilisterial activity of tannin rich preparations isolated from raspberry (Rubus idaeus L.) and strawberry (Fragaria X ananassa Duch.) fruit. Sci. Rep. 2025, 15, 10196. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]


| Compound | Concentration of Polyphenols (mg/100 g DM) |
|---|---|
| Agrimoniin | 3966 ± 116 |
| Sum of ellagitannins | 5217 ±157 |
| Ellagic acid | 169.3 ± 7.2 |
| Ellagic acid pentoside | 58.6 ± 2.3 |
| Apigenin 7-O-glucoside | 16.5 ± 0.1 |
| Apigenin 7-O-glucuronide | 1942 ± 45 |
| Luteolin 7-O-glucoside | 104.5 ± 4.1 |
| Luteolin 7-O-glucuronide | 313.9 ± 4.2 |
| Quercetin arabinoglycoside | 874.0 ±18.9 |
| Quercetin 3-O-ramnozyl-7-O-glucoside | 257.8 ± 4.4 |
| Quercetin 3-O-galactoside | 258.1 ± 12.6 |
| Kaempferol 3-O-rutoside | 83.6 ± 1.7 |
| Kaempferol 3-O-glucoside | 44.1 ± 0.7 |
| KpCG * | 212.3 ± 4.7 |
| Proanthocyanidins ** | 4784 ± 167 |
| Sum of flavanols | 5200 ± 169 |
| DP | 4.1 ± 0.1 |
| Total polyphenols HPLC *** | 14,773 ± 347 |
| Total polyphenols F-C **** | 12,260 ± 179 |
| Compound | Concentration of Polyphenols (mg/100 g DM) |
|---|---|
| Agrimoniin | 1763 ± 43 |
| Sum of ellagitannins | 3049 ± 82 |
| Ellagic acid | 162.9 ± 5.3 |
| Quercetin-3-O-galactoside | 18.5 ± 1.4 |
| Quercetin | 32.1 ± 1.6 |
| KpCG * | 68.7 ± 2.0 |
| Proanthocyanidins ** | 3733 ± 144 |
| Sum of flavanols | 3812 ± 142 |
| DP | 6.2 ± 0.1 |
| Total polyphenols HPLC *** | 7143 ± 97 |
| Total polyphenols F-C **** | 11,382 ± 415 |
| Total Polyphenol Content | ||
|---|---|---|
| Sample | mg GAE/100 g FM | mg GAE/100 g DM |
| Fresh apple | 108.5 ± 7.4 ab | 746.8 ± 59.1 c |
| OD apples in 50% sucrose | 132.0 ± 6.6 b | 435.5 ± 23.3 a |
| OD apples in 50% sucrose + 2 g/L PPs RH | 156.9 ± 9.4 c | 517.8 ± 31.9 b |
| OD apples in 50% sucrose + 4 g/L PPs RH | 224.4 ± 9.0 d | 693.5 ± 28.4 c |
| OD apples in 50% sucrose + 6 g/L PPs RH | 272.5 ± 9.7 f | 827.1 ± 22.4 d |
| OD apples in 50% sucrose + 2 g/L PPs AP | 161.5 ± 12.6 c | 530.7 ± 39.7 b |
| OD apples in 50% sucrose + 4 g/L PPs AP | 241.9 ± 6.2 e | 741.2 ± 40.0 c |
| OD apples in 50% sucrose + 6 g/L PPs AP | 277.7 ± 8.0 f | 825.0 ± 18.0 d |
| Compound | Fresh Apple | OD Apples in 50% Sucrose |
|---|---|---|
| mg/100 g DM | ||
| Chlorogenic acid | 53.6 ± 8.5 a | 29.0 ± 9.6 b |
| p-Coumaric acid | 6.7 ± 0.2 a | 3.7 ± 0.3 b |
| Quercetin rhamnoside | 1.6 ± 0.2 a | 1.1 ± 0.2 b |
| Proanthocyanidins | 295.5 ± 28.6 | 264.1 ± 16.3 |
| Sum of flavanols | 355.4 ± 25.2 a | 313.7 ± 19.7 b |
| Total polyphenols HPLC | 417.3 ± 16.4 a | 347.5 ± 28.1 b |
| Compound | Concentrations of Polyphenols in OD Apples in 50% Sucrose + | ||
|---|---|---|---|
| 2 g/L PPs AP | 4 g/L PPs AP | 6 g/L PPs AP | |
| mg/100 g DM | |||
| Chlorogenic acid * | 23.7 ± 0.8 a | 20.5 ± 0.4 b | 14.7 ± 2.2 c |
| p-Coumaric acid * | 3.0 ± 0.1 a | 2.6 ± 0.3 b | 2.6 ± 0.2 b |
| Quercetin rhamnoside * | 0.64 ± 0,05 a | 0.52 ± 0.10 b | 0.38 ± 0.04 c |
| Proanthocyanidins ** | 282.8 ± 16.2 b | 387.7 ± 26.6 a | 437.3 ± 55.1 a |
| Sum of flavanols | 324.0 ± 17.0 b | 431.4 ± 28.2 a | 470.5 ± 38.8 a |
| Agrimoniin | 67.5 ± 0.5 c | 126.7 ± 2.6 b | 195.8 ± 5.2 a |
| Sum of ETs | 108.2 ± 1.1 c | 179.8 ± 1.9 b | 266.9 ± 7.5 a |
| Ellagic acid | 6.0 ± 0.1 c | 8.0 ± 0.1 b | 10,9 ± 0.30 a |
| Ellagic acid pentoside | 3.6 ± 0.1 b | 3.7 ± 0.2 b | 4.1 ± 0.1 a |
| Apigenin 7-O-glucoside | 1.4 ± 0.1 | 1.3 ± 0.1 | 1.3 ± 0.08 |
| Apigenin 7-O-glucuronide | 25.3 ± 0.4 c | 54.2 ± 1.9 b | 86.1 ± 2.3 a |
| Luteolin 7-O-glucoside | 1.4 ± 0.1 c | 2.9 ± 0.1 b | 4.5 ± 0.3 a |
| Luteolin 7-O-glucuronide | 6.6 ± 0.1 c | 10.5 ± 0.2 b | 15.1 ± 0.4 a |
| Quercetin arabinoglycoside | 12.3 ± 0.2 c | 25.1 ± 0.8 b | 39.6 ± 1.2 a |
| Quercetin 3-O-rhamnosyl-7-O-glucoside | 4.2 ± 0.1 c | 7.4 ± 0.4 b | 11.5 ± 0.2 a |
| Quercetin 3-O-galactoside | 4.5 ± 0.1 c | 7.7 ± 0.3 b | 11.5 ± 0.6 a |
| Kaempferol 3-O-rutoside | 1.4 ± 0.1 c | 2.5± 0.1 b | 3.7 ± 0.1 a |
| Kaempferol 3-O-glucoside | 0.89 ± 0.05 c | 1.7 ± 0.1 b | 2.6 ± 0.1 a |
| KpCG *** | 4.9 ± 0.1 c | 8.3 ± 0.1 b | 11.8 ± 0.4 a |
| Total polyphenols HPLC | 531.7 ± 17.7 c | 768.1 ± 25.1 b | 958.5 ± 26.4 a |
| Compound | Concentrations of Polyphenols in OD Apples in 50% Sucrose + | ||
|---|---|---|---|
| 2 g/L PPs RH | 4 g/L PPs RH | 6 g/L PPs RH | |
| mg/100 g DM | |||
| Chlorogenic acid * | 22.4 ± 0.9 a | 15.3 ± 0.5 b | 16.1 ± 0.8 b |
| p-Coumaric acid * | 2.9 ± 0.1 a | 2.5 ± 0.0 b | 2.8 ± 0.1 a |
| Quercetin rhamnoside * | 0.75 ± 0.10 a | 0.50 ± 0.02 b | 0.53 ± 0.0 b |
| Proanthocyanidins ** | 262.2 ± 17.6 b | 270.8 ± 26.3 b | 320.3 ± 2.4 a |
| Sum of flavanols | 296.0 ± 19.4 b | 300.8 ± 26.0 b | 352.4 ± 2.3 a |
| Agrimoniin | 32.0 ± 0.9 c | 57.2 ± 1.5 b | 81.3 ± 2.6 a |
| Sum of ETs | 82.1 ± 2.2 c | 122.7 ± 1.5 b | 169.8 ± 4.3 a |
| Ellagic acid | 5.5 ± 0.1 c | 7.3 ± 0.1 b | 9.7 ± 0.4 a |
| Quercetin | 1.7 ± 0.0 c | 2.1 ± 0.0 b | 2.8 ± 0.1 a |
| KpCG *** | 1.8 ± 0.1 c | 2.7 ± 0.1 b | 3.6 ± 0.3 a |
| Total polyphenols | 413.1 ± 15.2 c | 454.1 ± 26.8 b | 557.6 ± 6.3 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karlińska, E.; Milala, J.; Kosmala, M.; Klewicki, R. Osmotic Dehydration of Apples in a Saccharose Solution Containing Fragrant Agrimony or Rosehip Extract. Molecules 2025, 30, 4708. https://doi.org/10.3390/molecules30244708
Karlińska E, Milala J, Kosmala M, Klewicki R. Osmotic Dehydration of Apples in a Saccharose Solution Containing Fragrant Agrimony or Rosehip Extract. Molecules. 2025; 30(24):4708. https://doi.org/10.3390/molecules30244708
Chicago/Turabian StyleKarlińska, Elżbieta, Joanna Milala, Monika Kosmala, and Robert Klewicki. 2025. "Osmotic Dehydration of Apples in a Saccharose Solution Containing Fragrant Agrimony or Rosehip Extract" Molecules 30, no. 24: 4708. https://doi.org/10.3390/molecules30244708
APA StyleKarlińska, E., Milala, J., Kosmala, M., & Klewicki, R. (2025). Osmotic Dehydration of Apples in a Saccharose Solution Containing Fragrant Agrimony or Rosehip Extract. Molecules, 30(24), 4708. https://doi.org/10.3390/molecules30244708







