Structural Modulation of Musky Octopus Proteins by pH and Ultrasound: From Aggregates to Protein–Quercetin Emulsion Stabilisers
Abstract
1. Introduction
2. Results and Discussion
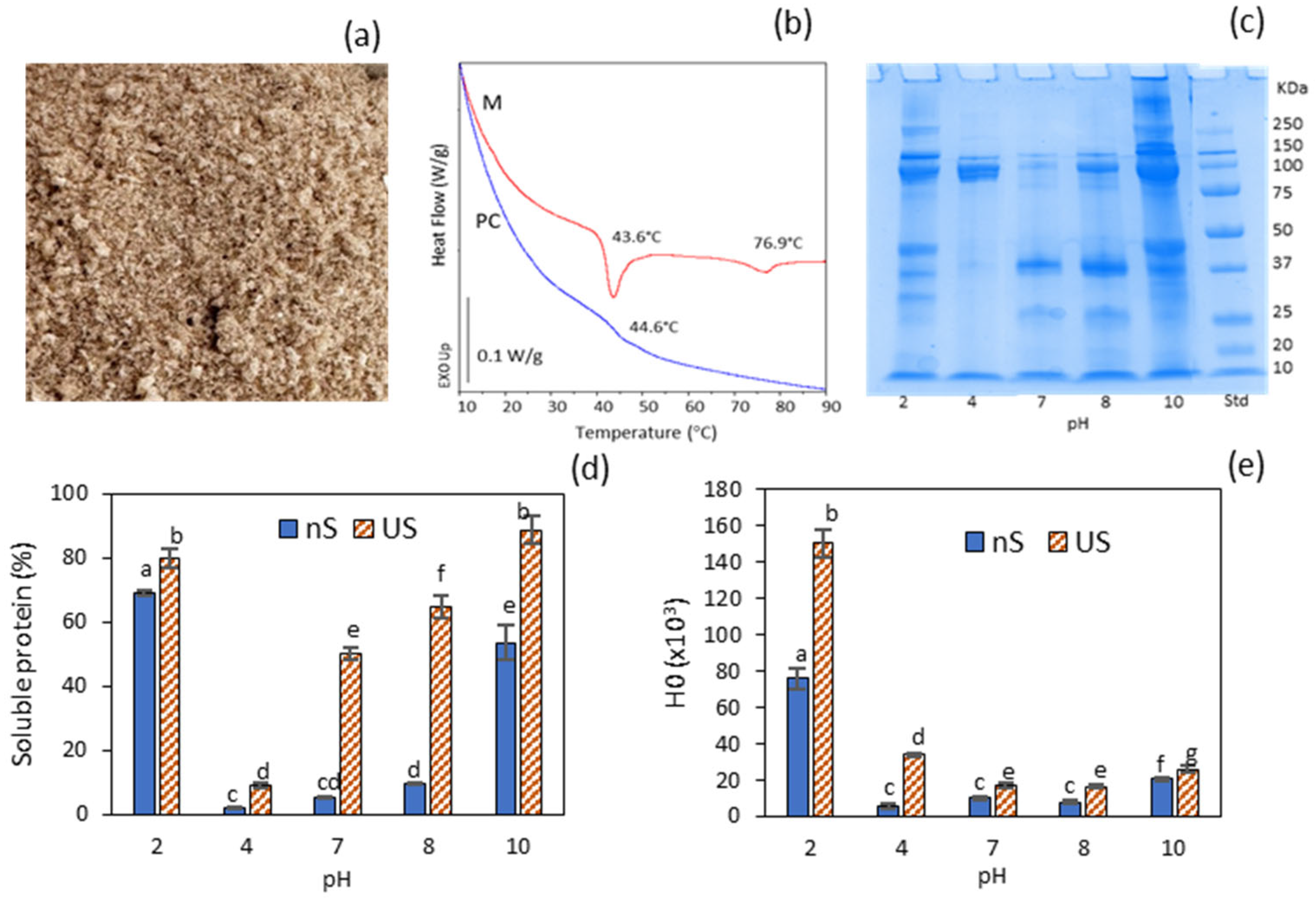
2.1. Properties of the Protein Concentrate
2.2. Water Solubility and Effect of Ultrasonication
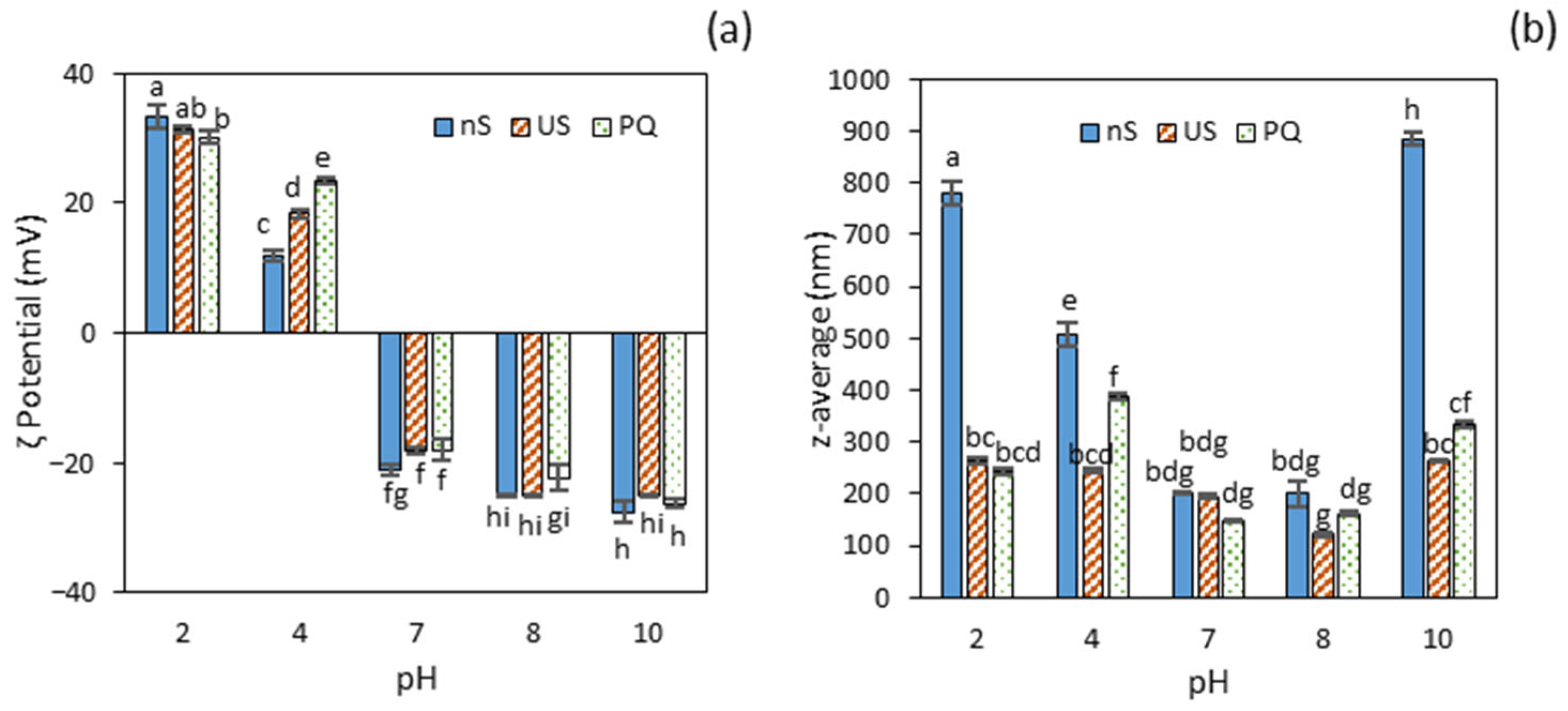
2.3. Effect of Ultrasonication
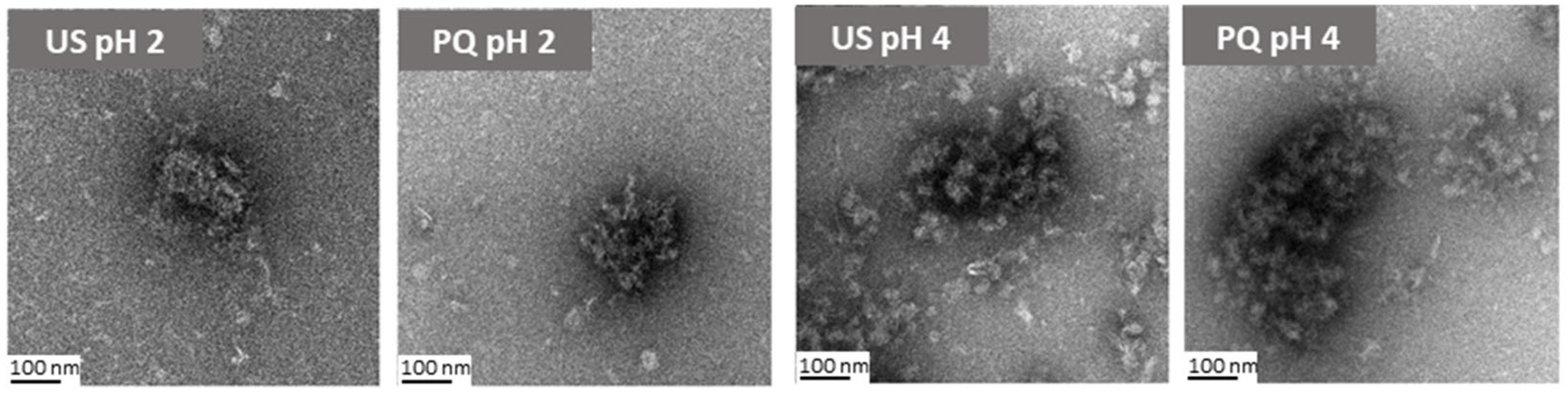
2.4. Effect of Quercetin Addition
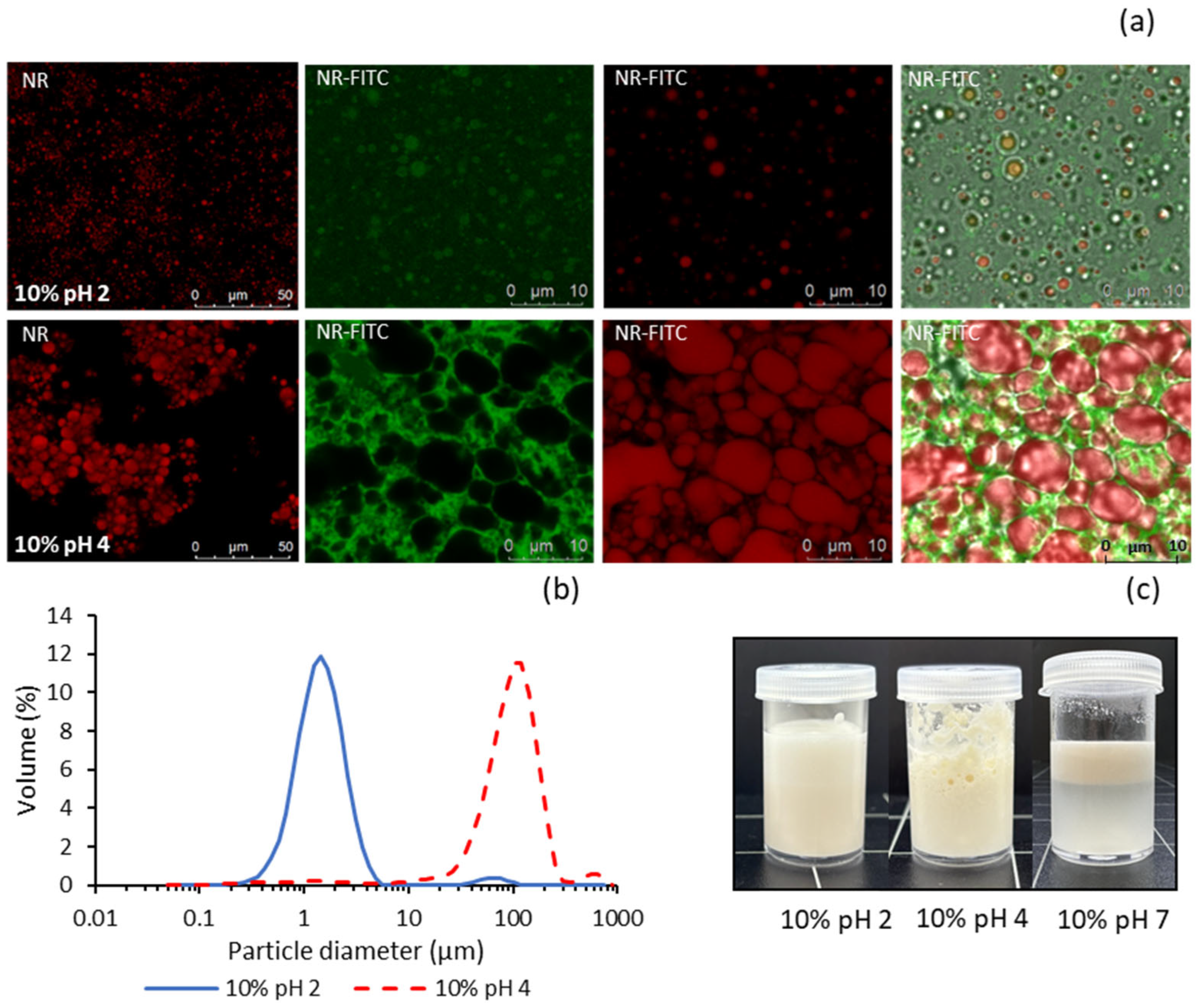
2.5. Oil-in-Water (O/W) Emulsions
3. Materials and Methods
3.1. Materials
3.2. Preparation of Protein Concentrate (PC)
3.3. Proximate Analysis and Amino Acid Composition of PC
3.4. Differential Scanning Calorimetry of PC
3.5. pH Modification and Ultrasonication (US) of PC
3.6. Protein Solubility, Electrophoretic Profile (SDS-PAGE), and Surface Hydrophobicity
3.7. Complexation of Quercetin with US-Treated PC
3.8. Preparation of Oil-in-Water Emulsions
3.9. Characterisation of Particle Size and ζ Potential
3.10. Transmission Electron Microscopy
3.11. Rheological Properties
3.12. Confocal Laser Scanning Microscopy (CLSM)
3.13. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Papadopoulou, S.N.A.; Adamantidi, T.; Kranas, D.; Cholidis, P.; Anastasiadou, C.; Tsoupras, A. Valorization of bioactives from marine animal by-products for health-promoting, biofunctional cosmetics. Mar. Drugs 2025, 23, 299. [Google Scholar] [CrossRef]
- Pascual-Silva, C.; Alemán, A.; Montero, M.P.; Gómez-Guillén, M.C. Storage stability and antioxidant properties of liposomes loaded with sea cucumber and musky octopus hydrolysates. Food Bioproc. Technol. 2025, 18, 4826–4842. [Google Scholar] [CrossRef]
- Ruiz-Capillas, C.; Moral, A.; Morales, J.; Montero, P. Characterization and Functionality of Frozen Muscle Protein in Volador (Illex coindetii), Pota (Todaropsis eblanae), and Octopus (Eledone cirrhosa). J. Food Sci. 2003, 68, 2164–2168. [Google Scholar] [CrossRef]
- Rahaman, A.; Kumari, A.; Zeng, X.A.; Korin, A.; Khalifa, I.; Maqsood, S. Processing of marine-derived by-products and their applications: A review. Waste Biomass Valorization 2025. [Google Scholar] [CrossRef]
- Oliyaei, N.; Ghorbani, M.; Moosavi-Nasab, M.; Sadeghimahoonak, A.R.; Maghsoudloo, Y. Effect of Temperature and Alkaline pH on the Physicochemical Properties of the Protein Isolates Extracted from the Whole Ungutted Lanternfish (Benthosema pterotum). J. Aquat. Food Prod. Technol. 2017, 26, 1134–1143. [Google Scholar] [CrossRef]
- Long, R.; Huang, Y.; Dabbour, M.; Mintah, B.K.; Pan, J.; Wu, M.; Zhang, S.; Qin, Z.; He, R.; Ma, H. Physical Processing-Assisted pH Shifting for Food Protein Modification: A Comprehensive Review. Foods 2025, 14, 2360. [Google Scholar] [CrossRef]
- Tang, C.H.; Ying, R.F.; Shi, L.E. Protein recovery from fish muscle by acid solubilization and isoelectric precipitation. Food Hydrocoll. 2021, 113, 106482. [Google Scholar] [CrossRef]
- Cortés-Ruiz, J.A.; Pacheco-Aguilar, R.; Ramírez-Suárez, J.C.; Lugo-Sánchez, M.E.; García-Orozco, K.D.; Sotelo-Mundo, R.R.; Peña-Ramos, A. Conformational changes in proteins recovered from jumbo squid muscle through pH shift washing treatments. Food Chem. 2016, 196, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Regenstein, J.M.; Zhou, P.; Yang, Y. Ultrasound-assisted modification of soy protein isolate for improved emulsifying properties. Food Hydrocoll. 2017, 63, 525–533. [Google Scholar] [CrossRef]
- Singh, A.; Benjakul, S.; Kijroongrojana, K. Ultrasound-assisted modification of squid ovary protein concentrate: Functional and structural properties. Ultrason. Sonochem. 2018, 40, 841–849. [Google Scholar] [CrossRef]
- Gao, K.; Rao, J.; Chen, B. Ultrasound improves solubility of commercial pea protein isolates. Food Hydrocoll. 2022, 131, 107823. [Google Scholar] [CrossRef]
- Sun, Y.; Yang, Y.; Jin, H.; Zhang, Y.; Ali, S.; Sheng, L. Ultrasound-assisted pH-shifting treatment: Effects on egg yolk protein interfacial adsorption and emulsification properties. Food Chem. 2025, 482, 144172. [Google Scholar] [CrossRef]
- Li, C.; Chen, L.; McClements, D.J.; Peng, X.; Xu, Z.; Meng, M.; Ji, H.; Qiu, C.; Long, J.; Jin, Z. Encapsulation of polyphenols in protein-based nanoparticles: Preparation, properties, and applications. Crit. Rev. Food Sci. Nutr. 2024, 64, 11341–11355. [Google Scholar] [CrossRef] [PubMed]
- Arredondo-Parada, I.; Torres-Arreola, W.; Suárez-Jiménez, G.M.; Ramírez-Suárez, J.C.; Juárez-Onofre, J.E.; Rodríguez-Félix, F.; Marquez-Rios, E. Effect of ultrasound on physicochemical and foaming properties of a protein concentrate from giant squid mantle. LWT 2020, 121, 108954. [Google Scholar] [CrossRef]
- Rajasekaran, B.; Singh, A.; Ponnusamy, A.; Patil, U.; Zhang, B.; Hong, H.; Benjakul, S. Ultrasound-treated fish myofibrillar protein and its stabilizing effect on shrimp oil-in-water emulsion. Ultrason. Sonochem. 2023, 98, 106513. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.P.; Ma, L.L.; Zheng, J.; Wang, J.T.; Wu, Q.; Song, S.; Zhou, D.Y. Effect of pH on the physicochemical and heat-induced gel properties of scallop Patinopecten yessoensis actomyosin. Fish. Sci. 2014, 80, 1073–1082. [Google Scholar] [CrossRef]
- Patil, U.; Gulzar, S.; Ma, L.; Zhang, B.; Benjakul, S. Pickering emulsion stabilized by fish myofibrillar proteins modified with tannic acid. Foods 2023, 12, 1556. [Google Scholar] [CrossRef]
- Kılınç, I. Pickering emulsion technology: An overview of stability and functionality in food processing. Food Nutr. Chem. 2025, 3, 354. [Google Scholar] [CrossRef]
- Gao, Y.; Lin, D.; Peng, H.; Zhang, R.; Zhang, B.; Yang, X. Low oil Pickering emulsion gels stabilised by bacterial cellulose nanofiber/soybean protein isolate. Int. J. Biol. Macromol. 2023, 247, 125623. [Google Scholar] [CrossRef]
- Cheon, J.; Haji, F.; Baek, J.; Wang, Q.; Tam, K.C. Pickering emulsions for functional food systems. J. Agric. Food Res. 2023, 11, 100510. [Google Scholar] [CrossRef]
- Nimaming, N.; Sadeghpour, A.; Murray, B.S.; Sarkar, A. Pickering emulsions stabilized by hybrid plant protein-flavonoid conjugate particles. Food Hydrocoll. 2024, 154, 110146. [Google Scholar] [CrossRef]
- Deng, L.; Liao, J.; Liu, W.; Liang, X.; Zhou, R.; Jiang, Y. Research Advances for Protein-Based Pickering Emulsions as Drug Delivery Systems. Pharmaceutics 2025, 17, 587. [Google Scholar] [CrossRef]
- Aghababaei, F.; Hadidi, M. Recent Advances in Potential Health Benefits of Quercetin. Pharmaceuticals 2023, 16, 1020. [Google Scholar] [CrossRef]
- McClements, D.J.; Öztürk, B. Nanotechnology to improve bioavailability of phytochemicals from waste streams. J. Agric. Food Chem. 2022, 70, 6884–6900. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Heussen, P.C.; Hazekamp, J.; Drost, E.; Velikov, K.P. Quercetin-loaded biopolymeric colloidal particles prepared by simultaneous precipitation. Food Chem. 2012, 133, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Q. Improved stability and bioavailability of quercetin by complexation with soy protein isolate. Food Chem. 2015, 171, 19–24. [Google Scholar] [CrossRef]
- Lin, J.; Yong, K.Y.A.; Zhou, Y.; Wang, Y.; Zhou, W. Improved bioaccessibility of quercetin by nanocomplexation with ultrasound-treated soy protein isolate. Food Chem. 2023, 406, 135004. [Google Scholar] [CrossRef]
- Ozogul, Y.; Duysak, O.; Ozogul, F.; Özkütük, A.S.; Türeli, C. Seasonal effects in the nutritional quality of the body structural tissue of cephalopods. Food Chem. 2008, 108, 847–852. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.C.; Turnay, J.; Fernández-Díaz, M.D.; Olmo, N.; Lizarbe, M.A.; Montero, P. Structural and physical properties of gelatin extracted from different marine species: A comparative study. Food Hydrocoll. 2002, 16, 25–34. [Google Scholar] [CrossRef]
- World Health Organization. Protein and Amino Acid Requirements in Human Nutrition; WHO Technical Report Series 935; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- Blanco-Pascual, N.; Fernández-Martín, F.; Montero, M.P. Effect of different protein extracts from Dosidicus gigas muscle co-products on edible films development. Food Hydrocoll. 2013, 33, 118–131. [Google Scholar] [CrossRef]
- Nisov, A.; Kakko, T.; Alakomi, H.L.; Lantto, R.; Honkapää, K. Comparison of enzymatic and pH shift methods to extract protein from whole Baltic herring (Clupea harengus membras) and roach (Rutilus rutilus). Food Chem. 2022, 373, 131524. [Google Scholar] [CrossRef]
- Mignino, L.A.; Paredi, M.E. Physicochemical and functional properties of myofibrillar proteins from molluscs. LWT 2006, 39, 35–42. [Google Scholar] [CrossRef]
- Fang, R.; Jing, H.; Chai, Z.; Zhao, G.; Stoll, S.; Ren, F.; Liu, F.; Leng, X. Design and characterization of protein-quercetin bioactive nanoparticles. J. Nanobiotechnol. 2011, 9, 19. [Google Scholar] [CrossRef]
- Wu, Z.; Yan, J.; Zhou, Z.; Xu, Q.; Zhong, Q.; Fang, X.; Huang, C.; He, X.; Li, L.; Li, Q. Preparation of soybean protein isolate-quercetin particles and its application in curcumin-camellia oil Pickering emulsion. Food Meas. 2024, 18, 2086–2100. [Google Scholar] [CrossRef]
- Liu, F.; Ma, C.; Gao, Y.; McClements, D.J. Food-grade covalent complexes and their application as nutraceutical delivery systems: A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 76–95. [Google Scholar] [CrossRef]
- Bai, L.; Lv, S.; Xiang, W.; Huan, S.; McClements, D.J.; Rojas, O.J. Oil-in-water Pickering emulsions via microfluidization with cellulose nanocrystals: 1. Formation and stability. Food Hydrocoll. 2019, 96, 699–708. [Google Scholar] [CrossRef]
- Zhang, T.; Xu, J.; Chen, J.; Wang, Z.; Wang, X.; Zhong, J. Protein nanoparticles for Pickering emulsions: A comprehensive review on their shapes, preparation methods, and modification methods. Trends Food Sci. Technol. 2021, 113, 26–41. [Google Scholar] [CrossRef]
- AOAC. Official Method of Analysis, 18th ed.; Association of Officiating Analytical Chemists: Arlington, VA, USA, 2015. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Alemán, A.; Pérez-Santín, E.; Bordenave-Juchereau, S.; Arnaudin, I.; Gómez-Guillén, M.C.; Montero, P. Squid gelatin hydrolysates with antihypertensive, anticancer and antioxidant activity. Food Res. Int. 2011, 44, 1044–1051. [Google Scholar] [CrossRef]
- Marín, D.; Alemán, A.; Montero, P.; Gómez-Guillén, M.C. Protein aggregation and thermal gelation of hake muscle with soy phosphatidylcholine liposomes. Food Hydrocoll. 2018, 82, 466–477. [Google Scholar] [CrossRef]
- Carrasco-Sandoval, J.; Aranda-Bustos, M.; Henríquez-Aedo, K.; López-Rubio, A.; Fabra, M.J. Bioaccessibility of different types of phenolic compounds co-encapsulated in alginate/chitosan-coated zein nanoparticles. LWT 2021, 149, 112024. [Google Scholar] [CrossRef]
- Alemán, A.; Montero, M.P.; Ramos, S.; Gómez-Guillén, M.C. Enrichment of surimi gels with water-in-oil emulsions formulated with virgin coconut oil and quercetin-loaded chitosan nanoparticles. Food Hydrocoll. 2025, 158, 110497. [Google Scholar] [CrossRef]
- Pascual-Silva, C.; Alemán, A.; Montero, M.P.; Gómez-Guillén, M.C. Extraction and characterization of Argentine red shrimp (Pleoticus muelleri) phospholipids as raw material for liposome production. Food Chem. 2022, 374, 131766. [Google Scholar] [CrossRef] [PubMed]
| Mince (M) | Protein Concentrate (PC) | |
|---|---|---|
| Moisture (%) | 82.83 ± 0.77 | 2.41 ± 0.43 |
| Protein (%) | 15.14 ± 0.23 | 89.63 ± 0.04 |
| Ash (%) | 1.20 ± 0.19 | 3.04 ± 0.05 |
| Fat (%) | 0.62 ± 0.13 | 4.86 ± 0.08 |
| mg/g Protein | Nutritional Score (%) | FAO/WHO Reference Pattern (mg/g) 1 | |
|---|---|---|---|
| Aspartic acid | 100.49 ± 0.34 | ||
| Threonine | 48.57 ± 0.35 | 211.17 | 23 |
| Serine | 50.22 ± 0.84 | ||
| Glutamic acid | 148.28 ± 0.87 | ||
| Glycine | 53.32 ± 0.08 | ||
| Alanine | 50.69 ± 0.16 | ||
| Cysteine | 5.97 ± 0.55 | ||
| Valine | 43.46 ± 0.62 | 111.44 | 39 |
| Methionine | 32.66 ± 2.33 | ||
| Isoleucine | 48.18 ± 0.43 | 160.60 | 30 |
| Leucine | 78.80 ± 0.18 | 133.56 | 59 |
| Tyrosine | 39.99 ± 0.38 | ||
| Phenylalanine | 43.32 ± 0.15 | ||
| Hydroxylysine | 3.87 ± 0.03 | ||
| Histidine | 23.44 ± 0.25 | 156.27 | 15 |
| Lysine | 76.70 ± 0.44 | 170.44 | 45 |
| Arginine | 83.25 ± 0.77 | ||
| Hydroxyproline | 12.14 ± 0.10 | ||
| Proline | 56.64 ± 0.32 | ||
| ∑ Hydrophobic | 407.13 ± 3.73 | ||
| ∑ TEAA 2 | 395.06 ± 2.50 | ||
| Met + Cys | 38.64 ± 2.88 | 175.59 | 22 |
| Phe + Tyr | 83.31 ± 0.53 | 219.24 | 38 |
| Yield (%) | Entrapment Efficiency (%) | |
|---|---|---|
| PQ-pH 2 | 94.5 ± 2.2 d | 81.2 ± 1.2 d |
| PQ-pH 4 | 72.2 ± 9.3 c | 57.2 ± 5.1 b |
| PQ-pH 7 | 24.2 ± 1.6 a | 65.7 ± 1.9 c |
| PQ-pH 8 | 36.6 ± 1.7 b | 39.9 ± 3.8 a |
| PQ-pH 10 | 92.5 ± 1.7 d | 81.9 ± 1.0 d |
| Apparent Viscosity η100 (mPa∙s) | κup (mPa⋅sn) | κdown (mPa⋅sn) | nup | ndown | |
|---|---|---|---|---|---|
| PQ-pH 2 | 8.42 ± 0.09 a | 8.87 ± 1.41 a | 8.88 ± 3.75 a | 0.938 ± 0.002 a | 0.944 ± 0.061 a |
| PQ-pH 4 | 2.26 ± 0.03 b | 4.52 ± 0.46 b | 4.51 ± 0.19 b | 0.864 ± 0.023 b | 0.863 ± 0.011 b |
| PQ-pH 7 | 1.88 ± 0.08 c | 0.93 ± 0.12 c | 2.73 ± 0.06 b | 1.180 ± 0.024 c | 0.878 ± 0.034 ab |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Guillén, M.C.; Alemán, A.; Boto, I.; López-Polo, J.; Montero, M.P. Structural Modulation of Musky Octopus Proteins by pH and Ultrasound: From Aggregates to Protein–Quercetin Emulsion Stabilisers. Molecules 2025, 30, 4570. https://doi.org/10.3390/molecules30234570
Gómez-Guillén MC, Alemán A, Boto I, López-Polo J, Montero MP. Structural Modulation of Musky Octopus Proteins by pH and Ultrasound: From Aggregates to Protein–Quercetin Emulsion Stabilisers. Molecules. 2025; 30(23):4570. https://doi.org/10.3390/molecules30234570
Chicago/Turabian StyleGómez-Guillén, María Carmen, Ailén Alemán, Ignacio Boto, Johana López-Polo, and María Pilar Montero. 2025. "Structural Modulation of Musky Octopus Proteins by pH and Ultrasound: From Aggregates to Protein–Quercetin Emulsion Stabilisers" Molecules 30, no. 23: 4570. https://doi.org/10.3390/molecules30234570
APA StyleGómez-Guillén, M. C., Alemán, A., Boto, I., López-Polo, J., & Montero, M. P. (2025). Structural Modulation of Musky Octopus Proteins by pH and Ultrasound: From Aggregates to Protein–Quercetin Emulsion Stabilisers. Molecules, 30(23), 4570. https://doi.org/10.3390/molecules30234570







