Construction of Donor–Acceptor Heterojunctions via Microphase Separation of Discotic Liquid Crystals with Ambipolar Transport
Abstract
1. Introduction
2. Results and Discussion
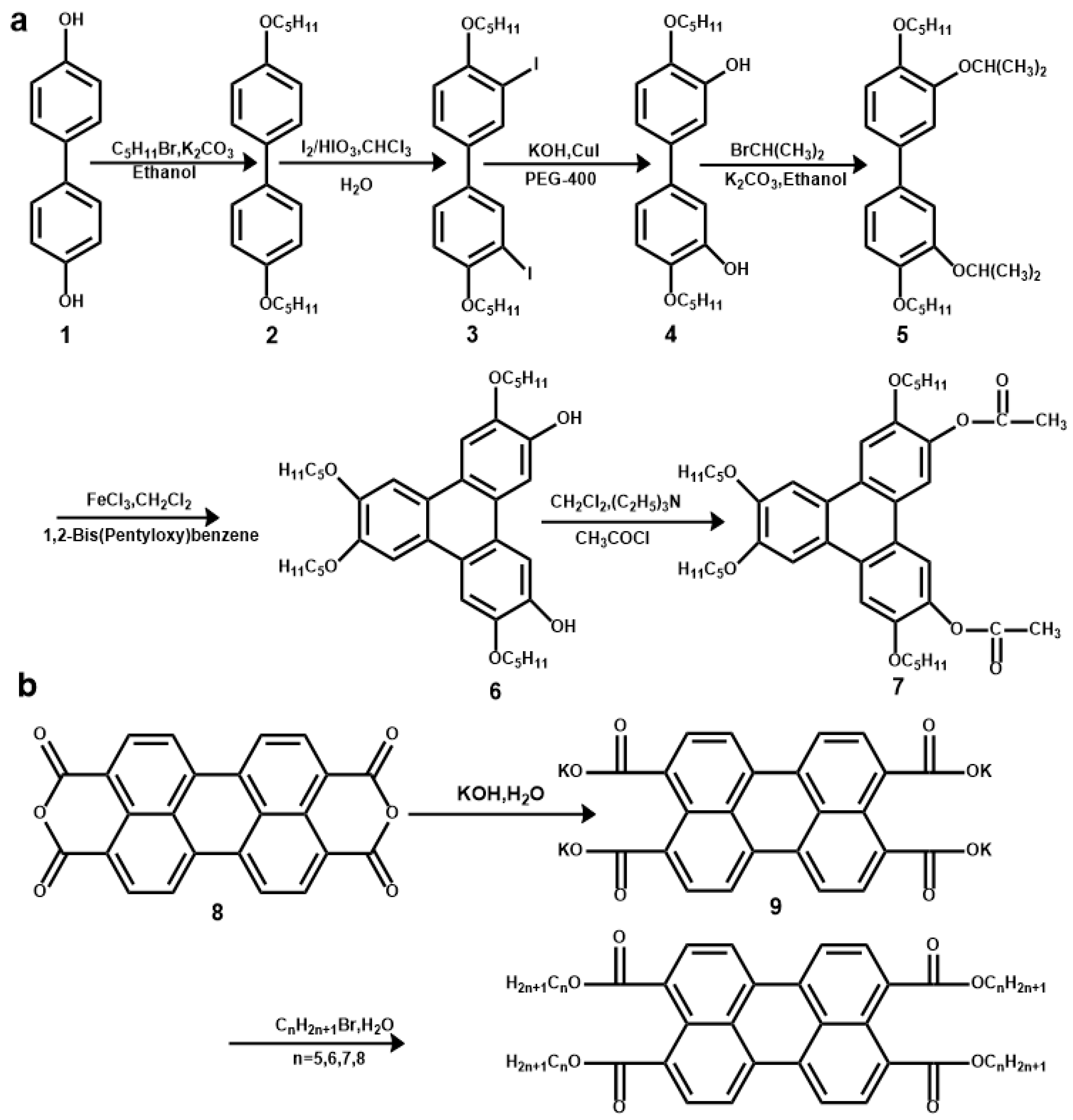
2.1. Synthesis of 3,6-di(Ethoxycarbonyl)-2,7,10,11-Tetrapentyloxytriphenylene and PTE
2.2. Thermal Properties and Phase Behaviors of T5E36 and PTECn
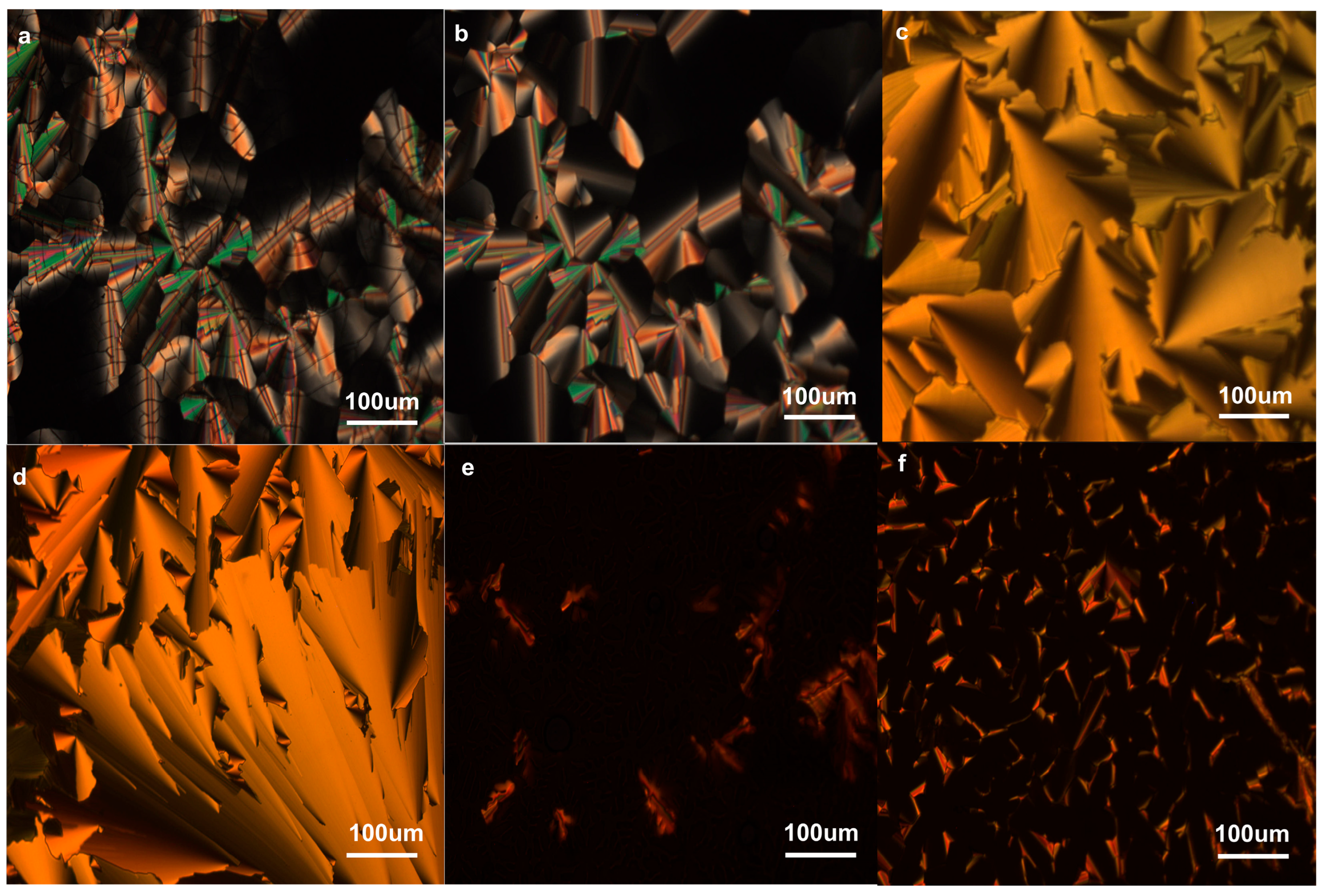
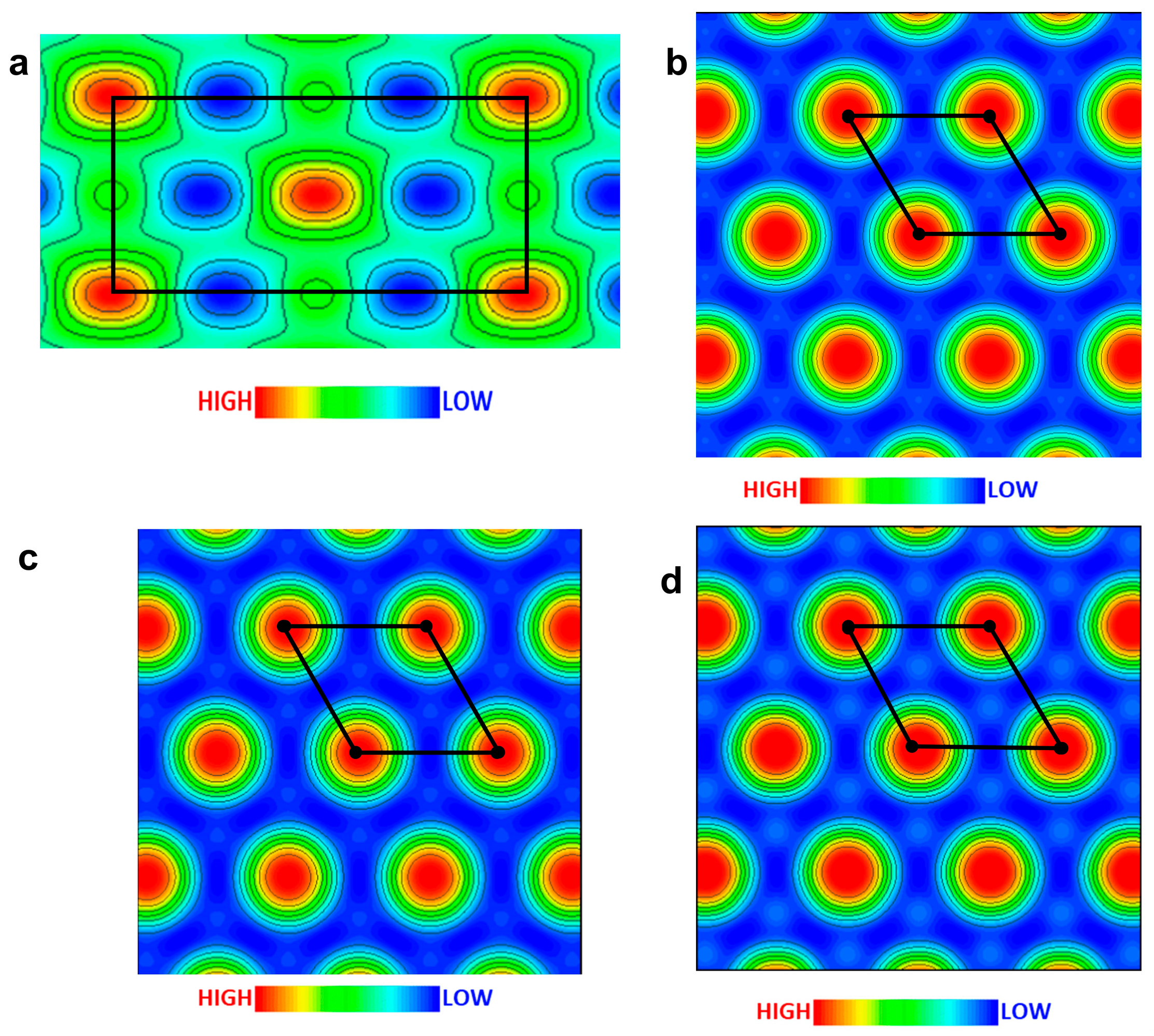
2.3. Mesophases of T5E36/PTECn Blend Systems
2.4. Steady-State Spectrum
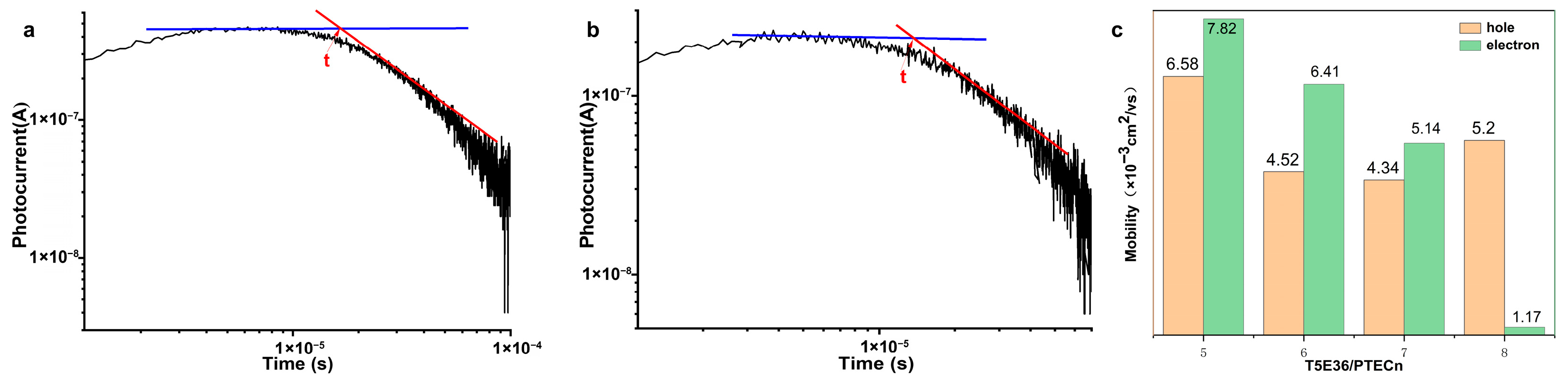
2.5. Charge Carrier Mobility
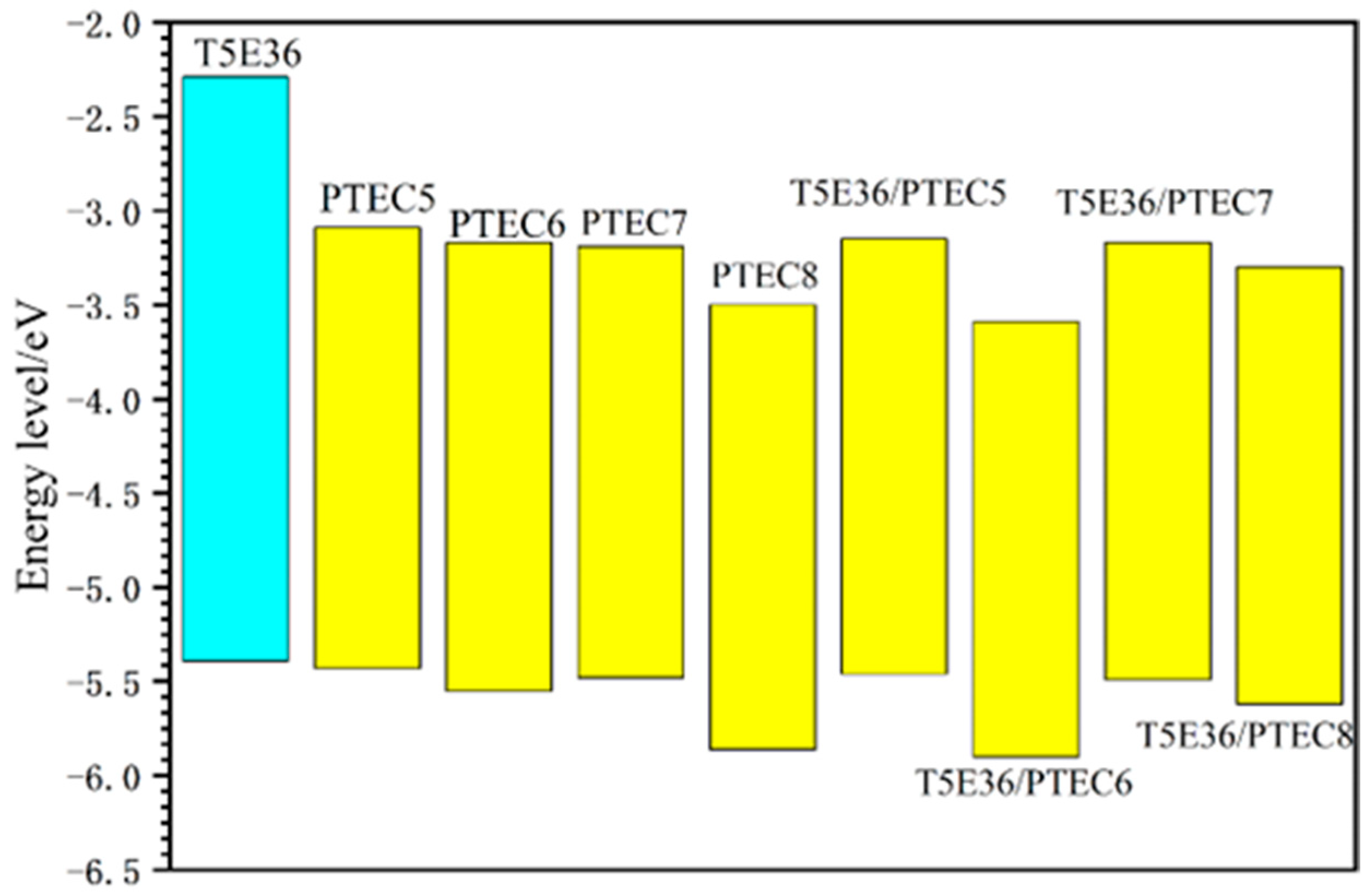
2.6. Energy Level Measurements
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lehn, J.-M. Perspectives in Supramolecular Chemistry From Molecular Recognition towards Molecular Information Processing and Self-organization. Angen. Chem. Int. Ed. Engl. 1990, 29, 1304–1319. [Google Scholar] [CrossRef]
- Fendler, J.H. Chemical Self-assembly for Electronic Applications. J. Am. Chem. Soc. 2001, 13, 3196–3210. [Google Scholar] [CrossRef]
- Whitesides, G.M.; Mathias, J.P.; Seto, C.T. Molecular Self-Assembly and Nanochemistry:A Chemical Strategy for the Synthesis of Nanostructures. Science 1991, 254, 1312–1319. [Google Scholar] [CrossRef]
- Zhang, S.G. Emerging biological materials through molecular self-assembly. Biotechnol. Adv. 2002, 20, 321–339. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.G. Fabrication of novel biomaterials through molecular self-assembly. Nat. Biotechnol. 2003, 21, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.; Miyake, K.; Sumaoka, J.; Komiyama, M.; Shigekawa, H. Effect of the Dipole-Dipole Interaction on the Self-Assembly of Cyclodextrin Inclusion Complexes. Jpn. J. Appl. Phys. 1999, 38, 3888–3891. [Google Scholar] [CrossRef]
- Kouwer, P.H.J.; Jager, W.F.; Mijs, W.J.; Picken, S.J. Specific interactions in discotic liquid crystals. J. Mater. Chem. 2003, 13, 458–469. [Google Scholar] [CrossRef]
- Rest, C.; Kandanelli, R.; Ferna’ndez, G. Strategies to create hierarchical self-assembled structures via cooperative non-covalent interactions. Chem. Soc. Rev. 2015, 44, 2543–2572. [Google Scholar] [CrossRef]
- Wang, J.; Liu, K.; Xing, R.; Yan, X. Peptide self-assembly: Thermodynamics and kinetics. Chem. Soc. Rev. 2016, 45, 5589–5604. [Google Scholar] [CrossRef]
- Lehn, J.-M.; Mascal, M.; DeCian, A. Molecular Recognition directed Self-assembly of Ordered Supramolecular Strands by Cocrystallization of Complementary Molecular Components. J. Chem. Soc. Chem. Commun. 1990, 1990, 479–481. [Google Scholar] [CrossRef]
- Kato, T.; Mizoshita, N.; Kishimoto, K. Functional liquid-crystalline assemblies: Self-organized soft materials. Angew. Chem. Int. Ed. Engl. 2005, 45, 38–68. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-K.; Noh, J.; Nayani, K.; Abbott, N.L. Soft matter from liquid crystals. Soft Matter 2019, 15, 6913–6929. [Google Scholar] [CrossRef] [PubMed]
- Bisoyi, H.K.; Li, Q. Liquid Crystals: Versatile Self-Organized Smart Soft Materials. Chem. Rev. 2022, 122, 4887–4926. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Yasuda, T.; Kamikawa, Y.; Yoshio, M. Self-assembly of functional columnar liquid crystals. Chem. Commun. 2009, 7, 729–739. [Google Scholar] [CrossRef]
- Foster, E.J.; Jones, R.B.; Lavigueur, C.; Williams, V.E. Structural Factors Controlling the Self-Assembly of Columnar Liquid Crystals. J. Am. Chem. Soc. 2006, 128, 8569–8574. [Google Scholar] [CrossRef]
- Kato, T. Hydrogen Bonding and the Self-Assembly of Supramolecular Liquid-Crystalline Materials. Macromol. Symp. 1995, 98, 311–326. [Google Scholar] [CrossRef]
- Lehmann, M.; Dechant, M.; Lambov, M.; Ghosh, T. Free Space in Liquid Crystals-Molecular Design, Generation, and Usage. Acc. Chem. Res. 2019, 52, 1653–1664. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Sadashiva, B.K.; Suresh, K.A. Liquid crystals of disc-like molecules. Pramana J. Phys. 1977, 9, 471–480. [Google Scholar] [CrossRef]
- Bushby, R.J.; Lozman, O.R. Discotic liquid crystals 25 years on. Curr. Opin. Colloid. Interface Sci. 2002, 7, 343–354. [Google Scholar] [CrossRef]
- Wohrle, T.; Wurzbach, I.; Kirres, J.; Kostidou, A.; Kapernaum, N.; Litterscheidt, J.; Haenle, J.C.; Staffeld, P.; Baro, A.; Giesse, F.; et al. Discotic Liquid Crystals. Chem. Rev. 2015, 116, 1139–1241. [Google Scholar] [CrossRef]
- Billard, J.; Dubois, J.; Nguyen Huu, T.; Zann, A. Une mesophase disquotique. Nou. J. Chim. 1978, 2, 535–539. [Google Scholar]
- Adam, D.; Schuhmacher, P.; Simmerer, J.; Haussling, L.; Siemensmeyer, K.; Etzbach, K.H.; Ringsdorf, H.; Haarer, D. Fast photoconduction in the highly ordered columnar phase of a discotic liquid crystal. Nature 1994, 371, 141–143. [Google Scholar] [CrossRef]
- Bala, I.; Singh, N.; Yadav, R.A.K.; De, J.; Gupta, S.P.; Singh, D.P.; Dubey, D.K.; Jou, J.-H.; Douali, R.; Pal, S.K. Room temperature perylene based columnar liquid crystals as solid-state fluorescent emitters in solution-processable organic light-emitting diodes. J. Mater. Chem. C 2020, 8, 12485–12494. [Google Scholar] [CrossRef]
- Keum, C.; Becker, D.; Archer, E.; Bock, H.; Kitzerow, H.; Gather, M.C.; Murawski, C. Organic Light-Emitting Diodes Based on a Columnar Liquid-Crystalline Perylene Emitter. Adv. Opt. Mater. 2020, 8, 2000414. [Google Scholar] [CrossRef]
- Wu, W.; Liu, Y.; Zhu, D. Pi-conjugated molecules with fused rings for organic field-effect transistors: Design, synthesis and applications. Chem. Soc. Rev. 2010, 39, 1489–1502. [Google Scholar] [CrossRef]
- Li, C.; Wonneberger, H. Perylene imides for organic photovoltaics: Yesterday, today, and tomorrow. Adv. Mater. 2012, 24, 613–636. [Google Scholar] [CrossRef]
- Zaumseil, J.; Sirringhaus, H. Electron and Ambipolar Transport in Organic Field-Effect Transistors. Chem. Rev. 2006, 107, 1296–1323. [Google Scholar] [CrossRef]
- Li, C.; Liu, M.; Pschirer, N.G.; Baumgarten, M.; Mullen, K. Polyphenylene-based materials for organic photovoltaics. Chem. Rev. 2010, 110, 6817–6855. [Google Scholar] [CrossRef]
- Gupta, R.K.; Sudhakar, A.A. Perylene-Based Liquid Crystals as Materials for Organic Electronics Applications. Langmuir 2019, 35, 2455–2479. [Google Scholar] [CrossRef]
- Cormier, R.A.; Gregg, B.A. Self-Organization in Thin Films of Liquid Crystalline Perylene Diimides. J. Phys. Chem. B 1997, 101, 11004–11006. [Google Scholar] [CrossRef]
- Cormier, R.A.; Gregg, B.A. Synthesis and Characterization of Liquid Crystalline Perylene Diimides. Chem. Mater. 1998, 10, 1309–1319. [Google Scholar] [CrossRef]
- Nagao, Y. Synthesis and properties of perylene pigments. Prog. Org. Coat. 1997, 31, 43–49. [Google Scholar] [CrossRef]
- Zhan, X.; Tan, Z.a.; Domercq, B.; An, Z.; Zhang, X.; Barlow, S.; Li, Y.; Zhu, D.; Kippelen, B.; Marder, S.R. A High-Mobility Electron-Transport Polymer with Broad Absorption and Its Use in Field-Effect Transistors and All-Polymer Solar Cells. J. Am. Chem. Soc. 2007, 129, 7246–7247. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Pathak, S.K.; Pradhan, B.; Rao, D.S.S.; Prasad, S.K.; Achalkumar, A.S. Self-assembly of luminescent N-annulated perylene tetraesters into fluid columnar phases. Soft Matter 2015, 11, 3629–3636. [Google Scholar] [CrossRef] [PubMed]
- Qiao, S.Y.; Liao, R.J.; Xie, M.S.; Song, X.L.; Zhang, A.; Fang, Y.; Zhang, C.X.; Yu, H.F. Synthesis and Optoelectronic Properties of Perylene Diimide-Based Liquid Crystals. Molecules 2025, 30, 799. [Google Scholar] [CrossRef] [PubMed]
- Wiebeler, C.; Vollbrecht, J.; Neuba, A.; Kitzerow, H.S.; Schumacher, S. Unraveling the electrochemical and spectroscopic properties of neutral and negatively charged perylene tetraethylesters. Sci. Rep. 2021, 11, 16097. [Google Scholar] [CrossRef]
- Tang, C.W. Two-layer organic photovoltaic cell. Appl. Phys. Lett. 1986, 48, 183–185. [Google Scholar] [CrossRef]
- Yu, G.; Gao, J.; Hummelen, J.C.; Wudi, F.; Heeger, A.J. Polymer Photovoltaic Cells: Enhanced Efficiencies via a Network of Internal Donor-Acceptor Heterojunctions. Science 1995, 270, 1789–1791. [Google Scholar] [CrossRef]
- Mikroyannidis, J.A.; Stylianakis, M.M.; Suresh, P.; Sharma, G.D. Efficient hybrid bulk heterojunction solar cells based on phenylenevinylene copolymer, perylene bisimide and TiO2. Sol. Energy Mater. Sol. Cells 2009, 93, 1792–1800. [Google Scholar] [CrossRef]
- Jiang, Y.Y.; Lu, L.H.; Yang, M.Y.; Zhan, C.; Xie, Z.Z.; Verpoort, F.; Xiao, S.Q. Taking the place of perylene diimide: Perylene tetracarboxylic tetraester as a building block for polymeric acceptors to achieve higher open circuit voltage in all-polymer bulk heterojunction solar cells. Polym. Chem. 2013, 4, 5612–5620. [Google Scholar] [CrossRef]
- Wang, F.Z.; Wu, H.; Guo, J.J.; Qiao, S.Y.; Wang, J.W.; Zhang, C.X.; Zhang, A.; Yu, H.F. Helical hexagonal columnar superlattice structures with high charge carrier mobility formed by hierarchical self-assembly of 3,6-substituted triphenylene esters. Mater. Chem. Front. 2025, 9, 1389–1400. [Google Scholar] [CrossRef]
- Bi, J.Z.; Wu, H.; Zhang, Z.H.; Zhang, A.; Yang, H.Z.; Feng, Y.W.; Fang, Y.; Zhang, L.N.; Wang, Z.R.; Qu, W.T.; et al. Highly ordered columnar superlattice nanostructures with improved charge carrier mobility by thermotropic self-assembly of triphenylene-based discotics. J. Mater. Chem. C 2019, 7, 12463–12469. [Google Scholar] [CrossRef]
- Hao, W.; Zhang, C.X.; Pu, J.L.; Wang, Y.F. A convenient synthesis method of 3,6-dihydroxy-2,7,10,11-tetrapentyloxytriphenylene from 4,4′-dihydroxybiphenyl with high yield. Liq. Cryst. 2014, 41, 1173–1178. [Google Scholar] [CrossRef]
- Laschat, S.; Baro, A.; Steinke, N.; Giesselmann, F.; Hagele, C.; Scalia, G.; Judele, R.; Kapatsina, E.; Sauer, S.; Schreivogel, A.; et al. Discotic liquid crystals: From tailor-made synthesis to plastic electronics. Angew. Chem. Int. Ed. 2007, 46, 4832–4887. [Google Scholar] [CrossRef]
- Anders, C.; Fischer, V.-M.; Tan, T.; Alaasar, M.; Waldecker, R.; Ke, Y.; Cao, Y.; Liu, F.; Tschierske, C. Modifying the liquid crystalline chessboard tiling—Soft reticular self-assembly of side-chain fluorinated polyphiles. J. Mater. Chem. C. 2025, 13, 37–53. [Google Scholar] [CrossRef]
- Stupnikov, A.A.; Bakirov, A.V.; Shcherbina, M.A.; Beginn, U.; Möller, M.; Chvalun, S.N. The effect of aliphatic chain length on the self-assembly of wedge-shaped cesium 3,4,5-tris(alkyloxy)benzenesulfonates. Mol. Syst. Des. Eng. 2023, 8, 181–188. [Google Scholar] [CrossRef]





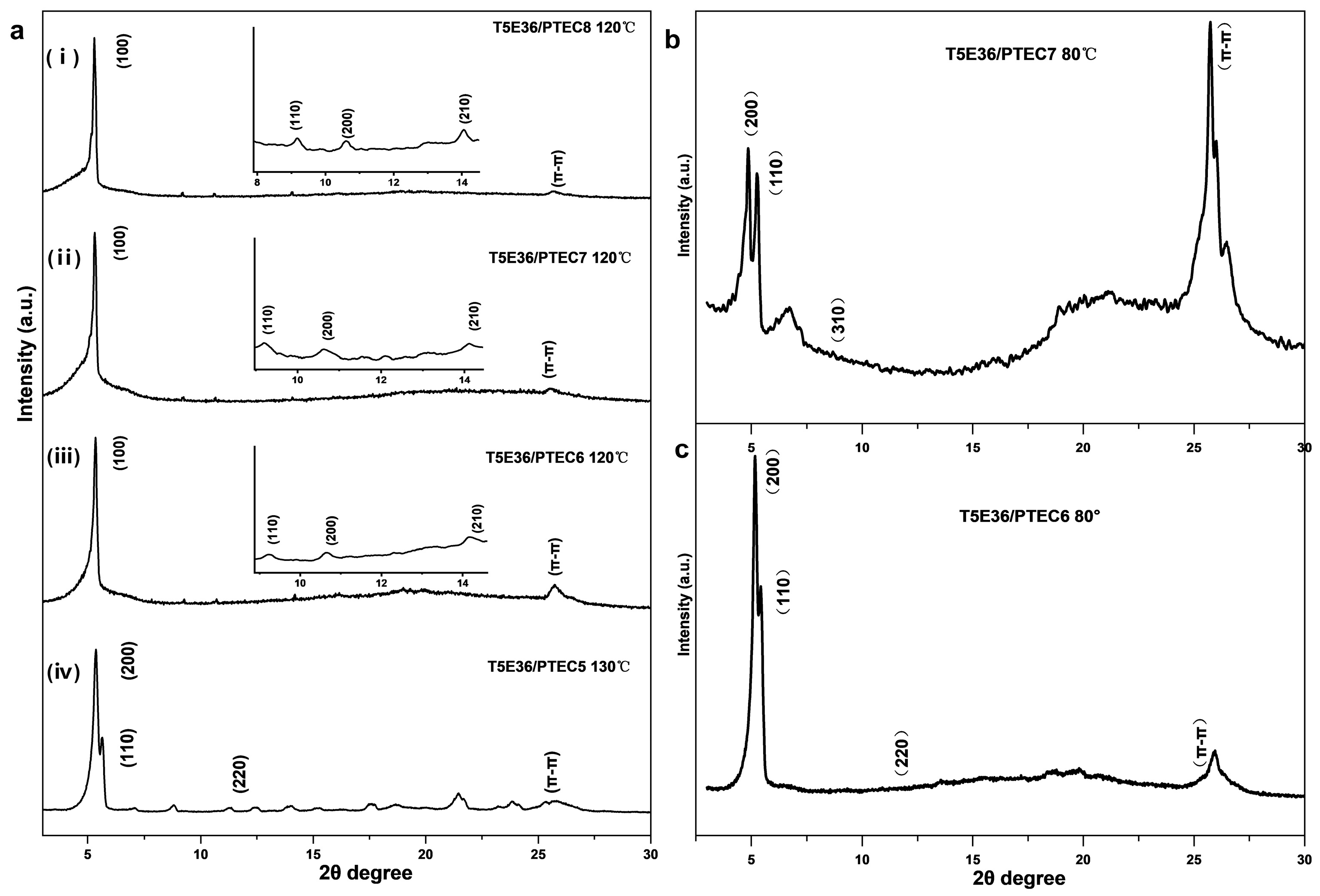

| Sample | d-Spacing (Å) | Miller Index (hkl) | Phase (Lattice Constants) |
|---|---|---|---|
| T5E36/PTEC5 (130 °C) | 16.32 | (200) | Colr (a = 32.64 Å b = 16.32 Å) |
| Colr/c2mm | 15.71 | (110) | |
| 7.81 | (220) | ||
| 3.55 | π-π | ||
| T5E36/PTEC6 (120 °C) | 16.51 | (100) | Colh (a = 19.06 Å) |
| Colh/p6mm | 9.5 | (110) | |
| 8.32 | (200) | ||
| 6.21 | (210) | ||
| T5E36/PTEC7 (120 °C) | 16.62 | (100) | Colh (a = 19.19 Å) |
| Colh/p6mm | 9.6 | (110) | |
| 8.28 | (200) | ||
| 6.28 | (210) | ||
| T5E36/PTEC8 (120 °C) | 16.67 | (100) | Colh (a = 19.25 Å) |
| Colh/p6mm | 9.62 | (110) | |
| 8.4 | (200) | ||
| 6.27 | (210) | ||
| T5E36/PTEC6 (80 °C) | 17.04 | (200) | Colr (a = 34.08 Å b = 18.56 Å) |
| Colr/c2mm | 16.11 | (110) | |
| 8.11 | (220) | ||
| 3.45 | π-π | ||
| T5E36/PTEC7 (80 °C) | 18.16 | (200) | Colr (a = 36.32 Å b = 18.83 Å) |
| Colr/c2mm | 16.7 | (110) | |
| 8.7 | (310) | ||
| 3.44 | π-π |
| Compound/Blend System | IP (eV) | Eg (eV) | HOMOUV (eV) | LUMOUV (eV) | HOMODFT (eV) | LUMODFT (eV) |
|---|---|---|---|---|---|---|
| T5E36 | 5.39 | 3.1 | −5.39 | −2.29 | −5.177 | −0.745 |
| PTEC5 | 5.55 | 2.38 | −5.55 | −3.17 | −5.475 | −2.670 |
| PTEC6 | 5.86 | 2.36 | −5.86 | −3.05 | −5.475 | −2.670 |
| PTEC7 | 5.9 | 2.31 | −5.9 | −3.59 | −5.473 | −2.669 |
| PTEC8 | 5.62 | 2.32 | −5.62 | −3.3 | −5.469 | −2.665 |
| T5E36/PTEC5 | 5.43 | 2.34 | −5.43 | −3.09 | ||
| T5E36/PTEC6 | 5.48 | 2.29 | −5.48 | −3.19 | ||
| T5E36/PTEC7 | 5.46 | 2.31 | −5.46 | −3.15 | ||
| T5E36/PTEC8 | 5.49 | 2.32 | −5.49 | −3.17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Xie, M.; Liu, Y.; Jia, G.; Liao, R.; Zhang, A.; Fang, Y.; Song, X.; Zhang, C.; Yu, H. Construction of Donor–Acceptor Heterojunctions via Microphase Separation of Discotic Liquid Crystals with Ambipolar Transport. Molecules 2025, 30, 3441. https://doi.org/10.3390/molecules30163441
Liu H, Xie M, Liu Y, Jia G, Liao R, Zhang A, Fang Y, Song X, Zhang C, Yu H. Construction of Donor–Acceptor Heterojunctions via Microphase Separation of Discotic Liquid Crystals with Ambipolar Transport. Molecules. 2025; 30(16):3441. https://doi.org/10.3390/molecules30163441
Chicago/Turabian StyleLiu, Heng, Mingsi Xie, Yaohong Liu, Gaojun Jia, Ruijuan Liao, Ao Zhang, Yi Fang, Xiaoli Song, Chunxiu Zhang, and Haifeng Yu. 2025. "Construction of Donor–Acceptor Heterojunctions via Microphase Separation of Discotic Liquid Crystals with Ambipolar Transport" Molecules 30, no. 16: 3441. https://doi.org/10.3390/molecules30163441
APA StyleLiu, H., Xie, M., Liu, Y., Jia, G., Liao, R., Zhang, A., Fang, Y., Song, X., Zhang, C., & Yu, H. (2025). Construction of Donor–Acceptor Heterojunctions via Microphase Separation of Discotic Liquid Crystals with Ambipolar Transport. Molecules, 30(16), 3441. https://doi.org/10.3390/molecules30163441









