Chemical Composition, Antioxidant, Antibacterial, and Antibiofilm Activities of Backhousia citriodora Essential Oil
Abstract
:1. Introduction
2. Results
2.1. Chemical Composition
2.2. Antioxidant Activity of BCEO
2.3. Antibacterial Activity of BCEO
2.3.1. Antibacterial Activity Analyzed by the Disk Diffusion Method
2.3.2. Antibacterial Activity Analyzed by the Broth Dilution Method to Determine Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
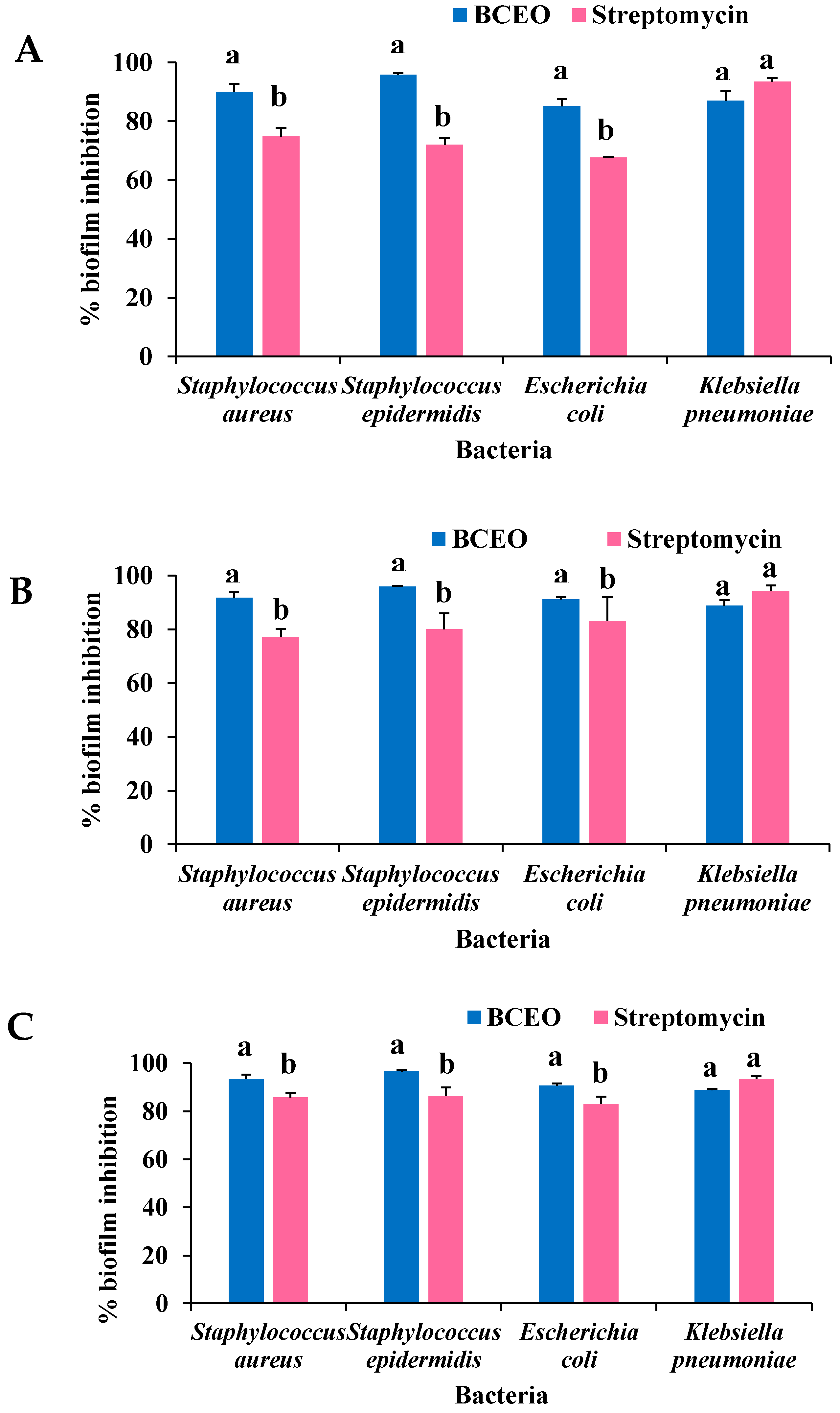
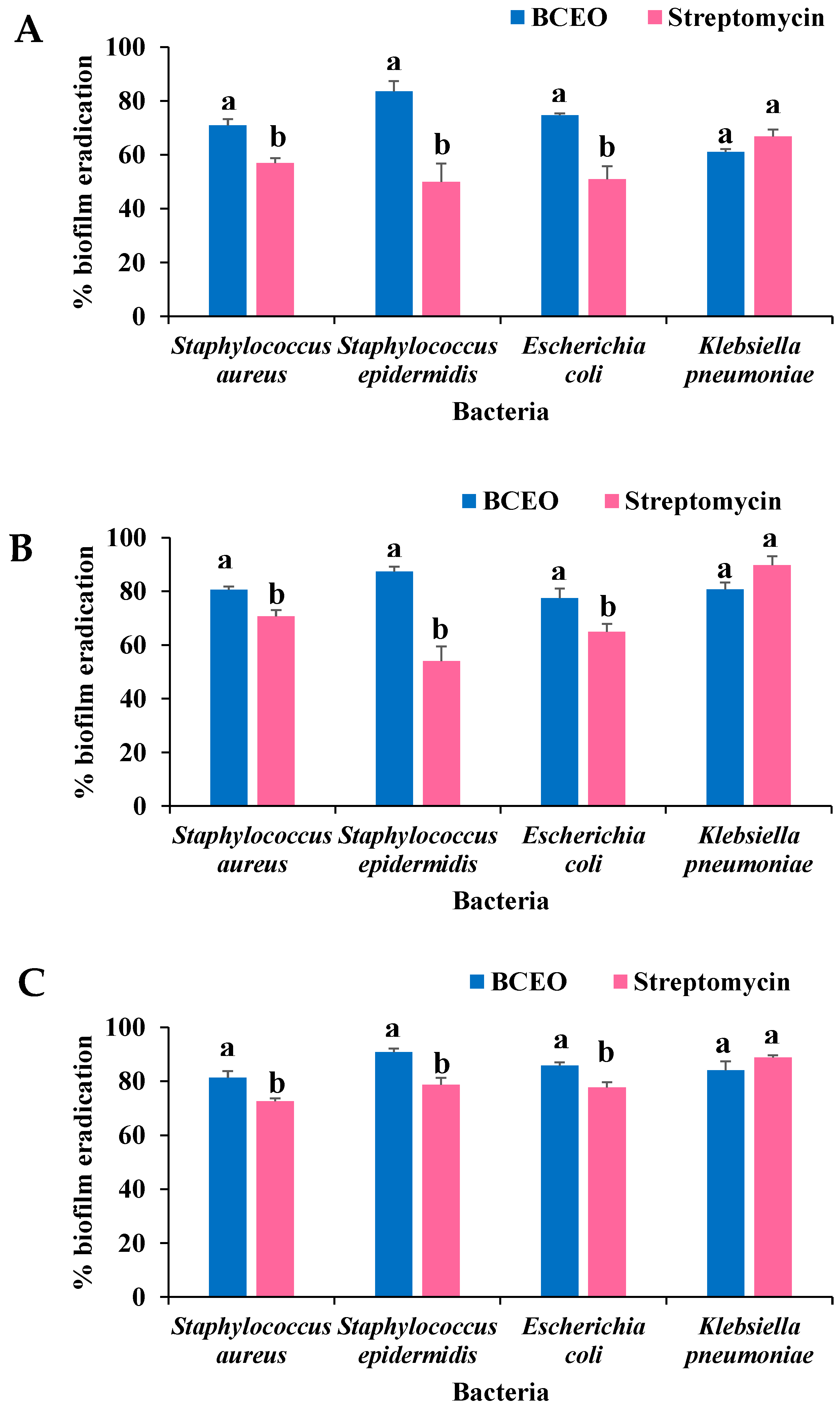
2.4. Antibiofilm Activity of BCEO
2.4.1. Biofilm Inhibitory Activity
2.4.2. Biofilm Eradication Activity
3. Discussion
4. Materials and Methods
4.1. Plant Material Collection and Identification
4.2. B. citriodora Essential Oil (BCEO) Extraction
4.3. Gas Chromatography–Flame Ionization Detection (GC–FID) and Gas Chromatography–Mass Spectrometry (GC–MS) Analyses of the BCEO
4.4. Measurement of Antioxidant Activity of BCEO
4.4.1. DPPH (2,2-diphenyl-1-picrylhydrazyl) Radical Scavenging Assay
4.4.2. Ferric Reducing Antioxidant Power Assay (FRAP)
4.5. Determination of Antibacterial Activity of BCEO
4.5.1. Microorganisms and Medium
4.5.2. Agar Disk Diffusion Method
4.5.3. Determination of the Minimum Inhibitory Concentration (MIC) and the Minimum Bactericidal Concentration (MBC)
4.6. Assessment of Antibiofilm Activity of BCEO
4.6.1. Preparation of Bacterial Cultures
4.6.2. Inhibition of Biofilm Formation, Prevention of Initial Bacteria Cell Attachment
4.6.3. Inhibition of Development of Pre-Formed Biofilms—Evaluation of Eradication of Biofilm Mass
4.6.4. Modified Crystal Violet Assay
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Kraker, M.E.A.; Stewardson, A.J.; Harbarth, S. Will 10 Million People Die a Year due to Antimicrobial Resistance by 2050? PLoS Med. 2016, 13, e1002184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Lack of New Antibiotics Threatens Global Efforts to Contain Drug-Resistant Infections. Available online: https://www.who.int/news/item/17-01-2020-lack-of-new-antibiotics-threatens-global-efforts-to-contain-drug-resistant-infections (accessed on 25 April 2022).
- World Health Organization. Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2018; Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 25 April 2022).
- van Duin, D.; Paterson, D.L. Multidrug-Resistant Bacteria in the Community. Infect. Dis. Clin. N. Am. 2016, 30, 377–390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davies, S.K.; Fearn, S.; Allsopp, L.P.; Harrison, F.; Ware, E.; Diggle, S.P.; Filloux, A.; McPhail, D.S.; Bundy, J.G. Visualizing Antimicrobials in Bacterial Biofilms: Three-Dimensional Biochemical Imaging Using TOF-SIMS. mSphere 2017, 2, e00211-17. [Google Scholar] [CrossRef] [Green Version]
- Balasubramanian, S.; Aubin-Tam, M.-E.; Meyer, A.S. 3D Printing for the Fabrication of Biofilm-Based Functional Living Materials. ACS Synth. Biol. 2019, 8, 1564–1567. [Google Scholar] [CrossRef] [Green Version]
- Borges, A.; Meireles, A.; Mergulhão, F.; Melo, L.; Simões, M. Biofilm control with enzymes. In Recent Trends in Biofilm Science and Technology; Elsevier: Amsterdam, The Netherlands, 2020; pp. 249–271. [Google Scholar]
- Khatoon, Z.; McTiernan, C.D.; Suuronen, E.J.; Mah, T.-F.; Alarcon, E.I. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018, 4, e01067. [Google Scholar] [CrossRef] [Green Version]
- Angiolella, L.; Sacchetti, G.; Efferth, T. Antimicrobial and Antioxidant Activities of Natural Compounds. Evid. Based Complement. Altern. Med. 2018, 2018, 1–3. [Google Scholar] [CrossRef]
- Botterweck, A.A.M.; Verhagen, H.; Goldbohm, R.; Kleinjans, J.; van den Brandt, P. Intake of butylated hydroxyanisole and butylated hydroxytoluene and stomach cancer risk: Results from analyses in the Netherlands Cohort Study. Food Chem. Toxicol. 2000, 38, 599–605. [Google Scholar] [CrossRef] [Green Version]
- Felter, S.P.; Zhang, X.; Thompson, C. Butylated hydroxyanisole: Carcinogenic food additive to be avoided or harmless antioxidant important to protect food supply? Regul. Toxicol. Pharmacol. 2021, 121, 104887. [Google Scholar] [CrossRef]
- Yang, X.; Sun, Z.; Wang, W.; Zhou, Q.; Shi, G.; Wei, F.; Jiang, G. Developmental toxicity of synthetic phenolic antioxidants to the early life stage of zebrafish. Sci. Total Environ. 2018, 643, 559–568. [Google Scholar] [CrossRef]
- Alabdaly, Y.Z.; Al-Hamdany, E.K.; Abed, E.R. Toxic effects of butylated hydroxytoluene in rats. Iraqi J. Veter. Sci. 2021, 35, 121–128. [Google Scholar] [CrossRef]
- US Food and Drug Administration. CFR—Code of Federal Regulations Title 21. Available online: https://www.accessdata.fda.gov/SCRIPTs/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=182.20 (accessed on 25 April 2022).
- Ríos, J.-L. Essential Oils. In Essential Oils in Food Preservation, Flavor and Safety; Elsevier: Amsterdam, The Netherlands, 2016; pp. 3–10. [Google Scholar]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharifi-Rad, J.; Sureda, A.; Tenore, G.C.; Daglia, M.; Sharifi-Rad, M.; Valussi, M.; Tundis, R.; Sharifi-Rad, M.; Loizzo, M.R.; Ademiluyi, A.O.; et al. Biological Activities of Essential Oils: From Plant Chemoecology to Traditional Healing Systems. Molecules 2017, 22, 70. [Google Scholar] [CrossRef] [PubMed]
- bin Jantan, I.; Moharam, B.A.K.; Santhanam, J.; Jamal, J.A. Correlation Between Chemical Composition and Antifungal Activity of the Essential Oils of Eight Cinnamomum. Species. Pharm. Biol. 2008, 46, 406–412. [Google Scholar] [CrossRef] [Green Version]
- Azizan, N.; Said, S.M.; Abidin, Z.Z.; Jantan, I. Composition and Antibacterial Activity of the Essential Oils of Orthosiphon stamineus Benth and Ficus deltoidea Jack against Pathogenic Oral Bacteria. Molecules 2017, 22, 2135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abu Bakar, A.; Ahmad, H.; Sulaiman, S.; Omar, B.; Ali, R.M. Evaluation of in vitro Bioactivity of Melaleuca cajuputi Powell Essential Oil against Aedes aegypti (L.) and Aedes albopictus (Skuse). Sains Malays. 2019, 48, 1919–1926. [Google Scholar] [CrossRef]
- Ogunwande, I.A.; Avoseh, O.N.; Igile, D.O.; Lawal, O.A.; Ascrizzi, R.; Guido, F. Chemical Constituents, Anti-nociceptive and Anti-inflammatory Activities of Essential Oil of Phyllanthus muellerianus. Nat. Prod. Commun. 2019, 14, 1934578X1984635. [Google Scholar] [CrossRef] [Green Version]
- Rahim, R.A.; Jayusman, P.; Muhammad, N.; Mohamed, N.; Lim, V.; Ahmad, N.; Mohamad, S.; Hamid, Z.A.; Ahmad, F.; Mokhtar, N.; et al. Potential Antioxidant and Anti-Inflammatory Effects of Spilanthes acmella and Its Health Beneficial Effects: A Review. Int. J. Environ. Res. Public Health 2021, 18, 3532. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.H.A.; Amarowicz, R.; Kandeil, A.; Ali, M.A.; Ibrahim, E.A. Antiviral activity of Lavandula angustifolia L. and Salvia officinalis L. essential oils against avian influenza H5N1 virus. J. Agric. Food Res. 2021, 4, 100135. [Google Scholar] [CrossRef]
- Huang, S.-Y.; Yao, N.; He, J.-K.; Pan, M.; Hou, Z.-F.; Fan, Y.-M.; Du, A.; Tao, J.-P. In vitro Anti-parasitic Activity of Pelargonium X. asperum Essential Oil Against Toxoplasma gondii. Front. Cell Dev. Biol. 2021, 9, 616340. [Google Scholar] [CrossRef]
- Salleh, W.M.N.H.W.; Khamis, S. Essential Oil Composition and Antioxidant Activity of Paramignya lobata. Chem. Nat. Compd. 2021, 57, 774–775. [Google Scholar] [CrossRef]
- Adorjan, B.; Buchbauer, G. Biological properties of essential oils: An updated review. Flavour Fragr. J. 2010, 25, 407–426. [Google Scholar] [CrossRef]
- Sabater-Jara, A.B.; Funes, M.P.; Pedreño, M.A.; Belchí-Navarro, S. Essential Oils of Thymbra capitata and Thymus hyemalis and Their Uses Based on Their Bioactivity. In Thymus; Rezaei, N., Ed.; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef] [Green Version]
- Franco, C.; Ferreira, O.; de Moraes, A.B.; Varela, E.; Nascimento, L.; Percário, S.; de Oliveira, M.; Andrade, E. Chemical Composition and Antioxidant Activity of Essential Oils from Eugenia patrisii Vahl, E. punicifolia (Kunth) DC., and Myrcia tomentosa (Aubl.) DC., Leaf of Family Myrtaceae. Molecules 2021, 26, 3292. [Google Scholar] [CrossRef]
- Fernandes, P.A.D.S.; Pereira, R.L.S.; dos Santos, A.T.L.; Coutinho, H.D.M.; Morais-Braga, M.F.B.; da Silva, V.B.; Costa, A.R.; Generino, M.E.M.; de Oliveira, M.G.; de Menezes, S.A.; et al. Phytochemical Analysis, Antibacterial Activity and Modulating Effect of Essential Oil from Syzygium cumini (L.) Skeels. Molecules 2022, 27, 3281. [Google Scholar] [CrossRef]
- Ferreira, O.O.; da Silva, S.H.M.; de Oliveira, M.S.; Andrade, E.H.D.A. Chemical Composition and Antifungal Activity of Myrcia multiflora and Eugenia florida Essential Oils. Molecules 2021, 26, 7259. [Google Scholar] [CrossRef]
- Nirmal, N.P.; Webber, D.; Mereddy, R.; Sultanbawa, Y. Biochemical and functional properties of indigenous Australian herbal infusions. Food Biosci. 2018, 26, 133–138. [Google Scholar] [CrossRef] [Green Version]
- Cock, I. Antimicrobial activity of Backhousia citriodora (lemon myrtle) methanolic extracts. Pharmacogn. Commun. 2013, 3, 58–63. [Google Scholar] [CrossRef] [Green Version]
- Shim, S.-Y.; Kim, J.-H.; Kho, K.-H.; Lee, M. Anti-inflammatory and anti-oxidative activities of lemon myrtle (Backhousia citriodora) leaf extract. Toxicol. Rep. 2020, 7, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Southwell, I. Backhousia citriodora F. Muell. (Lemon Myrtle), an Unrivalled Source of Citral. Foods 2021, 10, 1596. [Google Scholar] [CrossRef]
- Yabuta, Y.; Mukoyama, H.; Kaneda, Y.; Kimura, N.; Bito, T.; Ichiyanagi, T.; Ishihara, A.; Watanabe, F. A lemon myrtle extract inhibits glucosyltransferases activity of Streptococcus mutans. Biosci. Biotechnol. Biochem. 2018, 82, 1584–1590. [Google Scholar] [CrossRef]
- Almousawi, A.H.; Abed, H.A.; Ibrahim, Z.K.; Neamah, B.A.H. Lemon myrtle (Backhousia citriodora) leaf extract on biofilm formation of Pseudomonas aeruginosa. Int. J. Health Sci. 2022, 6, 721–727. [Google Scholar] [CrossRef]
- Santiago, J.D.A.; Cardoso, M.D.G.; da Cruz, F.A.; Palmieri, M.; de Souza, R.V.; Soares, L.; de Campos, J.; Andrade-Vieira, L. Cytogenotoxic effect of essential oil from Backhousia citriodora L. (Myrtaceae) on meristematic cells of Lactuca sativa L. S. Afr. J. Bot. 2017, 112, 515–520. [Google Scholar] [CrossRef]
- Southwell, I.A.; Russell, M.; Smith, R.L.; Archer, D.W. Backhousia citriodora F. Muell. (Myrtaceae), A Superior Source of Citral. J. Essent. Oil Res. 2000, 12, 735–741. [Google Scholar] [CrossRef]
- Doimo, L. Iso-Citrals and Iso-Geraniols in Lemon-Myrtle (Backhousia citriodora F. Muell.) Essential Oils. J. Essent. Oil Res. 2001, 13, 236–237. [Google Scholar] [CrossRef]
- Kean, O.B.; Yusoff, N.; Ali, N.A.M.; Subramaniam, V.; Yee, S.K. Chemical Composition and Antioxidant Properties of Volatile Oil. Open Conf. Proc. J. 2013, 4, 194. [Google Scholar] [CrossRef] [Green Version]
- Barra, A. Factors Affecting Chemical Variability of Essential Oils: A Review of Recent Developments. Nat. Prod. Commun. 2009, 4, 1147–1154. [Google Scholar] [CrossRef] [Green Version]
- Heikal, A.A.-E.M. Variation in the Essential Oil Content and its Composition in Eucalyptus cinerea Leaves and its Relation to Some Environmental Factors. J. Essent. Oil Bear. Plants 2017, 20, 995–1005. [Google Scholar] [CrossRef]
- Jardak, M.; Elloumi-Mseddi, J.; Aifa, S.; Mnif, S. Chemical composition, anti-biofilm activity and potential cytotoxic effect on cancer cells of Rosmarinus officinalis L. essential oil from Tunisia. Lipids Health Dis. 2017, 16, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Kedare, S.B.; Singh, R.P. Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol. 2011, 48, 412–422. [Google Scholar] [CrossRef] [Green Version]
- Moller, A.C.; Parra, C.; Said, B.; Werner, E.; Flores, S.; Villena, J.; Russo, A.; Caro, N.; Montenegro, I.; Madrid, A. Antioxidant and Anti-Proliferative Activity of Essential Oil and Main Components from Leaves of Aloysia polystachya Harvested in Central Chile. Molecules 2020, 26, 131. [Google Scholar] [CrossRef]
- Diniz do Nascimento, L.; De Moraes, A.A.B.; Da Costa, K.S.; Galúcio, J.M.P.; Taube, P.S.; Costa, C.M.L.; Cruz, J.N.; Andrade, E.H.D.A.; De Faria, L.J.G. Bioactive Natural Compounds and Antioxidant Activity of Essential Oils from Spice Plants: New Findings and Potential Applications. Biomolecules 2020, 10, 988. [Google Scholar] [CrossRef] [PubMed]
- Olszowy, M.; Dawidowicz, A. Essential oils as antioxidants: Their evaluation by DPPH, ABTS, FRAP, CUPRAC, and β-carotene bleaching methods. Mon. Chem. -Chem. Mon. 2016, 147, 2083–2091. [Google Scholar] [CrossRef]
- Sadeer, N.B.; Montesano, D.; Albrizio, S.; Zengin, G.; Mahomoodally, M.F. The Versatility of Antioxidant Assays in Food Science and Safety—Chemistry, Applications, Strengths, and Limitations. Antioxidants 2020, 9, 709. [Google Scholar] [CrossRef] [PubMed]
- Cerretani, L.; Bendini, A. Rapid assays to evaluate the antioxidant capacity of phenols in virgin olive oil. In Olives and Olive Oil in Health and Disease Prevention; Elsevier Inc.: Amsterdam, The Netherlands, 2010; pp. 625–635. ISBN 978-0-12-374420-3. [Google Scholar]
- Spiegel, M.; Kapusta, K.; Kołodziejczyk, W.; Saloni, J.; Żbikowska, B.; Hill, G.A.; Sroka, Z. Antioxidant Activity of Selected Phenolic Acids–Ferric Reducing Antioxidant Power Assay and QSAR Analysis of the Structural Features. Molecules 2020, 25, 3088. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, R.; Coppo, E.; Marchese, A.; Daglia, M.; Sobarzo-Sánchez, E.; Nabavi, S.F.; Nabavi, S.M. Phytochemicals for human disease: An update on plant-derived compounds antibacterial activity. Microbiol. Res. 2017, 196, 44–68. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, D.F.; Nora, F.M.D.; Lucas, B.N.; De Menezes, C.R.; Cichoski, A.J.; Giacomelli, S.R.; Wagner, R.; Barin, J.S. Oxygen introduction during extraction and the improvement of antioxidant activity of essential oils of basil, lemon and lemongrass. Cienc. Rural 2017, 47, e20170045. [Google Scholar] [CrossRef] [Green Version]
- Elshamy, A.I.; Abd-ElGawad, A.M.; El-Amier, Y.A.; El Gendy, A.E.G.; Al-Rowaily, S.L. Interspecific variation, antioxidant and allelopathic activity of the essential oil from three Launaea species growing naturally in heterogeneous habitats in Egypt. Flavour Fragr. J. 2019, 34, 316–328. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; Elshamy, A.I.; El-Amier, Y.A.; El Gendy, A.E.-N.G.; Al-Barati, S.A.; Dar, B.A.; Al-Rowaily, S.L.; Assaeed, A.M. Chemical composition variations, allelopathic, and antioxidant activities of Symphyotrichum squamatum (Spreng.) Nesom essential oils growing in heterogeneous habitats. Arab. J. Chem. 2019, 13, 4237–4245. [Google Scholar] [CrossRef]
- Mogoşanu, G.D.; Grumezescu, A.M.; Bejenaru, C.; Bejenaru, L.E. Natural products used for food preservation. In Food Preservation; Elsevier: Amsterdam, The Netherlands, 2017; pp. 365–411. [Google Scholar]
- Majolo, F.; Martins, A.; Rehfeldt, S.; Henriques, J.A.P.; Contini, V.; Goettert, M.I. Approaches for the treatment of neurodegenerative diseases related to natural products. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2021; Volume 69, pp. 1–63. [Google Scholar]
- Bharti, S.K. Essential Oil of Cymbopogon Citratus Against Diabetes: Validation by In vivo Experiments and Computational Studies. J. Bioanal. Biomed. 2013, 5, 194–203. [Google Scholar] [CrossRef] [Green Version]
- Anggraeni, N.I.; Hidayat, I.W.; Rachman, S.D. Ersanda Bioactivity of essential oil from lemongrass (Cymbopogon citratus Stapf) as antioxidant agent. AIP Conf. Proc. 2018, 1927, 30007. [Google Scholar] [CrossRef]
- Quintero, W.L.; Moreno, E.M.; Pinto, S.M.L.; Sanabria, S.M.; Stashenko, E.; García, L.T. Immunomodulatory, trypanocide, and antioxidant properties of essential oil fractions of Lippia alba (Verbenaceae). BMC Complement. Med. Ther. 2021, 21, 187. [Google Scholar] [CrossRef] [PubMed]
- Ehsani, A.; Alizadeh, O.; Hashemi, M.; Afshari, A.; Aminzare, M. Phytochemical, antioxidant and antibacterial properties of Melissa officinalis and Dracocephalum moldavica essential oils. Vet. Res. Forum Int. Q. J. 2017, 8, 223–229. [Google Scholar]
- Bayala, B.; Coulibaly, A.Y.; Djigma, F.W.; Nagalo, B.M.; Baron, S.; Figueredo, G.; Lobaccaro, J.-M.A.; Simpore, J. Chemical composition, antioxidant, anti-inflammatory and antiproliferative activities of the essential oil of Cymbopogon nardus, a plant used in traditional medicine. Biomol. Concepts 2020, 11, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Chi, P.T.L.; Van Hung, P.; Le Thanh, H.; Phi, N.T.L. Valorization of Citrus Leaves: Chemical Composition, Antioxidant and Antibacterial Activities of Essential Oils. Waste Biomass Valorization 2019, 11, 4849–4857. [Google Scholar] [CrossRef]
- Pucci, M.; Raimondo, S.; Zichittella, C.; Tinnirello, V.; Corleone, V.; Aiello, G.; Moschetti, M.; Conigliaro, A.; Fontana, S.; Alessandro, R. Biological Properties of a Citral-Enriched Fraction of Citrus limon Essential Oil. Foods 2020, 9, 1290. [Google Scholar] [CrossRef] [PubMed]
- Bouzenna, H.; Hfaiedh, N.; Giroux-Metges, M.-A.; Elfeki, A.; Talarmin, H. Biological properties of citral and its potential protective effects against cytotoxicity caused by aspirin in the IEC-6 cells. Biomed. Pharmacother. 2017, 87, 653–660. [Google Scholar] [CrossRef]
- Choudhary, P.; Singh, S.; Agarwal, V. Microbial Biofilms. In Bacterial Biofilms; Dincer, S., Sümengen Özdenefe, M., Arkut, A., Eds.; IntechOpen: London, UK, 2020. [Google Scholar]
- Wilkinson, J.M.; Hipwell, M.; Ryan, A.T.; Cavanagh, H.M.A. Bioactivity of Backhousia citriodora: Antibacterial and Antifungal Activity. J. Agric. Food Chem. 2003, 51, 76–81. [Google Scholar] [CrossRef]
- Chao, S.; Young, G.; Oberg, C.; Nakaoka, K. Inhibition of methicillin-resistant Staphylococcus aureus (MRSA) by essential oils. Flavour Fragr. J. 2008, 23, 444–449. [Google Scholar] [CrossRef]
- da Silva Júnior, A.Q.; da Silva, D.S.; Figueiredo, P.L.B.; Sarrazin, S.L.F.; Bouillet, L.E.M.; de Oliveira, R.B.; Maia, J.G.S.; Mourão, R.H.V. Seasonal and circadian evaluation of a citral-chemotype from Lippia alba essential oil displaying antibacterial activity. Biochem. Syst. Ecol. 2019, 85, 35–42. [Google Scholar] [CrossRef]
- Hayes, A.; Markovic, B. Toxicity of Australian essential oil Backhousia citriodora (Lemon myrtle). Part 1. Antimicrobial activity and in vitro cytotoxicity. Food Chem. Toxicol. 2002, 40, 535–543. [Google Scholar] [CrossRef]
- Thielmann, J.; Muranyi, P.; Kazman, P. Screening essential oils for their antimicrobial activities against the foodborne pathogenic bacteria Escherichia coli and Staphylococcus aureus. Heliyon 2019, 5, e01860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beikzadeh, S.; Akbarinejad, A.; Swift, S.; Perera, J.; Kilmartin, P.A.; Travas-Sejdic, J. Cellulose acetate electrospun nanofibers encapsulating Lemon Myrtle essential oil as active agent with potent and sustainable antimicrobial activity. React. Funct. Polym. 2020, 157, 104769. [Google Scholar] [CrossRef]
- Mahizan, N.A.; Yang, S.-K.; Moo, C.L.; Song, A.A.-L.; Chong, C.-M.; Chong, C.-W.; Abushelaibi, A.; Lim, S.-H.-E.; Lai, K.-S. Terpene Derivatives as a Potential Agent against Antimicrobial Resistance (AMR) Pathogens. Molecules 2019, 24, 2631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elyemni, M.; El Ouadrhiri, F.; Lahkimi, A.; Elkamli, T.; Bouia, A.; Eloutassi, N. Chemical Composition and Antimicrobial Activity of Essential Oil of Wild and Cultivated Rosmarinus officinalis from Two Moroccan Localities. J. Ecol. Eng. 2022, 23, 214–222. [Google Scholar] [CrossRef]
- Rathore, S.; Mukhia, S.; Kapoor, S.; Bhatt, V.; Kumar, R.; Kumar, R. Seasonal variability in essential oil composition and biological activity of Rosmarinus officinalis L. accessions in the western Himalaya. Sci. Rep. 2022, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, H.B.M.; Selis, N.D.N.; Sampaio, B.A.; Júnior, M.N.S.; de Carvalho, S.P.; de Almeida, J.B.; Almeida, P.P.; da Silva, I.B.S.; Oliveira, C.N.T.; Brito, T.L.S.; et al. Citral modulates virulence factors in methicillin-resistant Staphylococcus aureus. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yoplac, I.; Vargas, L.; Robert, P.; Hidalgo, A. Characterization and antimicrobial activity of microencapsulated citral with dextrin by spray drying. Heliyon 2021, 7, e06737. [Google Scholar] [CrossRef]
- Chueca, B.; Pagán, R.; García-Gonzalo, D. Oxygenated monoterpenes citral and carvacrol cause oxidative damage in Escherichia coli without the involvement of tricarboxylic acid cycle and Fenton reaction. Int. J. Food Microbiol. 2014, 189, 126–131. [Google Scholar] [CrossRef]
- Nirmal, N.; Mereddy, R.; Li, L.; Sultanbawa, Y. Formulation, characterisation and antibacterial activity of lemon myrtle and anise myrtle essential oil in water nanoemulsion. Food Chem. 2018, 254, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Prakash, A.; Vadivel, V. Citral and linalool nanoemulsions: Impact of synergism and ripening inhibitors on the stability and antibacterial activity against Listeria monocytogenes. J. Food Sci. Technol. 2019, 57, 1495–1504. [Google Scholar] [CrossRef]
- Yang, S.-K.; Yusoff, K.; Ajat, M.; Thomas, W.; Abushelaibi, A.; Akseer, R.; Lim, S.-H.-E.; Lai, K.-S. Disruption of KPC-producing Klebsiella pneumoniae membrane via induction of oxidative stress by cinnamon bark (Cinnamomum verum J. Presl) essential oil. PLoS ONE 2019, 14, e0214326. [Google Scholar] [CrossRef] [Green Version]
- Połeć, K.; Broniatowski, M.; Wydro, P.; Hąc-Wydro, K. The impact of β-myrcene—The main component of the hop essential oil—On the lipid films. J. Mol. Liq. 2020, 308, 113028. [Google Scholar] [CrossRef]
- Li, Z.-H.; Cai, M.; Liu, Y.-S.; Sun, P.L.; Luo, S.-L. Antibacterial Activity and Mechanisms of Essential Oil from Citrus medica L. var. sarcodactylis. Molecules 2019, 24, 1577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Du, C.; Li, Q.; Hu, A.; Peng, R.; Sun, F.; Zhang, W. Inhibition mechanism and antibacterial activity of natural antibacterial agent citral on bamboo mould and its anti-mildew effect on bamboo. R. Soc. Open Sci. 2021, 8, 202244. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Song, K.; Zhang, X.; Sun, Y.; Sui, Y.; Chen, Y.; Jia, Z.; Sun, H.; Sun, Z.; Xia, X. Antimicrobial Activity and Possible Mechanism of Action of Citral against Cronobacter sakazakii. PLoS ONE 2016, 11, e0159006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.F.; Ferreira, S.; Oliveira, R.; Domingues, F.C. Effect of Coriander Oil (Coriandrum sativum) on Planktonic and Biofilm Cells of Acinetobacter baumannii. Nat. Prod. Commun. 2013, 8, 1934578X1300800. [Google Scholar] [CrossRef] [Green Version]
- Melo, R.S.; Azevedo, M.A.; Pereira, A.M.G.; Rocha, R.R.; Cavalcante, R.M.B.; Matos, M.N.C.; Lopes, P.H.R.; Gomes, G.A.; Rodrigues, T.H.S.; dos Santos, H.S.; et al. Chemical Composition and Antimicrobial Effectiveness of Ocimum gratissimum L. Essential Oil Against Multidrug-Resistant Isolates of Staphylococcus aureus and Escherichia coli. Molecules 2019, 24, 3864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borotová, P.; Galovičová, L.; Vukovic, N.L.; Vukic, M.; Tvrdá, E.; Kačániová, M. Chemical and Biological Characterization of Melaleuca alternifolia Essential Oil. Plants 2022, 11, 558. [Google Scholar] [CrossRef]
- Kačániová, M.; Galovičová, L.; Valková, V.; Ďuranová, H.; Štefániková, J.; Čmiková, N.; Vukic, M.; Vukovic, N.L.; Kowalczewski, P. Chemical Composition, Antioxidant, In Vitro and In Situ Antimicrobial, Antibiofilm, and Anti-Insect Activity of Cedar atlantica Essential Oil. Plants 2022, 11, 358. [Google Scholar] [CrossRef]
- Morshdy, A.E.M.A.; El-Tahlawy, A.S.; Qari, S.H.; Qumsani, A.T.; Bay, D.H.; Sami, R.; Althubaiti, E.H.; Mansour, A.M.A.; Aljahani, A.H.; Hafez, A.E.-S.E.; et al. Anti-Biofilms’ Activity of Garlic and Thyme Essential Oils against Salmonella typhimurium. Molecules 2022, 27, 2182. [Google Scholar] [CrossRef] [PubMed]
- Cáceres, M.; Hidalgo, W.; Stashenko, E.; Torres, R.; Ortiz, C. Essential Oils of Aromatic Plants with Antibacterial, Anti-Biofilm and Anti-Quorum Sensing Activities against Pathogenic Bacteria. Antibiotics 2020, 9, 147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ben Abdallah, F.; Lagha, R.; Gaber, A. Biofilm Inhibition and Eradication Properties of Medicinal Plant Essential Oils against Methicillin-Resistant Staphylococcus aureus Clinical Isolates. Pharmaceuticals 2020, 13, 369. [Google Scholar] [CrossRef] [PubMed]
- Mizan, M.F.R.; Ashrafudoulla, M.; Hossain, M.I.; Cho, H.-R.; Ha, S.-D. Effect of essential oils on pathogenic and biofilm-forming Vibrio parahaemolyticus strains. Biofouling 2020, 36, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Kerekes, E.B.; Vidács, A.; Takó, M.; Petkovits, T.; Vágvölgyi, C.; Horváth, G.; Balázs, V.L.; Krisch, J. Anti-Biofilm Effect of Selected Essential Oils and Main Components on Mono- and Polymicrobic Bacterial Cultures. Microorganisms 2019, 7, 345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, S.; Liu, G.; Li, J.; Chen, J.; Li, L.; Li, Z.; Zhang, X.; Zhang, S.; Thorne, R.F.; Zhang, S. Antimicrobial Activity of Lemongrass Essential Oil (Cymbopogon flexuosus) and Its Active Component Citral Against Dual-Species Biofilms of Staphylococcus aureus and Candida Species. Front. Cell. Infect. Microbiol. 2020, 10, 603858. [Google Scholar] [CrossRef]
- Porfírio, E.M.; Melo, H.M.; Pereira, A.M.G.; Cavalcante, T.T.A.; Gomes, G.A.; de Carvalho, M.G.; Costa, R.A.; Júnior, F.E.A.C. In Vitro Antibacterial and Antibiofilm Activity of Lippia alba Essential Oil, Citral, and Carvone against Staphylococcus aureus. Sci. World J. 2017, 2017, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngome, M.T.; Alves, J.G.L.F.; Meira, A.C.F.D.O.; Machado, P.D.S.; Mondragón-Bernal, O.L.; Piccoli, R.H. Linalool, citral, eugenol and thymol: Control of planktonic and sessile cells of Shigella flexneri. AMB Express 2018, 8, 105. [Google Scholar] [CrossRef]
- Ortega-Ramirez, L.A.; Gutiérrez-Pacheco, M.M.; Vargas-Arispuro, I.; González-Aguilar, G.A.; Martínez-Téllez, M.A.; Ayala-Zavala, J.F. Inhibition of Glucosyltransferase Activity and Glucan Production as an Antibiofilm Mechanism of Lemongrass Essential Oil against Escherichia coli O157:H7. Antibiotics 2020, 9, 102. [Google Scholar] [CrossRef] [Green Version]
- Man, A.; Santacroce, L.; Iacob, R.; Mare, A.; Man, L. Antimicrobial Activity of Six Essential Oils Against a Group of Human Pathogens: A Comparative Study. Pathogens 2019, 8, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moghaddam, M.; Mehdizadeh, L.; Najafgholi, H.M.; Pirbalouti, A.G. Chemical composition, antibacterial and antifungal activities of seed essential oil of Ferulago angulata. Int. J. Food Prop. 2018, 21, 158–170. [Google Scholar] [CrossRef] [Green Version]
- Patterson, J.E.; McElmeel, L.; Wiederhold, N.P. In Vitro Activity of Essential Oils Against Gram-Positive and Gram-Negative Clinical Isolates, Including Carbapenem-Resistant Enterobacteriaceae. Open Forum Infect. Dis. 2019, 6, ofz502. [Google Scholar] [CrossRef] [PubMed]
- Elshafie, H.S.; Ghanney, N.; Mang, S.M.; Ferchichi, A.; Camele, I. An In Vitro Attempt for Controlling Severe Phytopathogens and Human Pathogens Using Essential Oils from Mediterranean Plants of Genus Schinus. J. Med. Food 2016, 19, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.R.; Rosário, D.K.A.; Silva, P.I.; Carneiro, J.C.S.; Pimentel Filho, N.J.; Bernardes, P.C. Cinnamon Essential Oil Reduces Adhesion of Food Pathogens to Polystyrene. Int. Food Res. J. 2019, 26, 1103–1110. [Google Scholar]
- Semiz, G.; Semiz, A.; Mercan-Doğan, N. Essential oil composition, total phenolic content, antioxidant and antibiofilm activities of four Origanum species from southeastern Turkey. Int. J. Food Prop. 2018, 21, 194–204. [Google Scholar] [CrossRef] [Green Version]
- Vijayalakshmi, M.; Ruckmani, K. Ferric reducing anti-oxidant power assay in plant extract. Bangladesh J. Pharmacol. 2016, 11, 570–572. [Google Scholar] [CrossRef] [Green Version]
- Adiguzel, A.; Ozer, H.; Kilic, H.; Cetin, B. Screening of antimicrobial activity of essential oil and methanol extract of Satureja hortensis on foodborne bacteria and fungi. Czech J. Food Sci. 2008, 25, 81–89. [Google Scholar] [CrossRef] [Green Version]
- Sandasi, M.; Leonard, C.; Viljoen, A. The effect of five common essential oil components on Listeria monocytogenes biofilms. Food Control 2008, 19, 1070–1075. [Google Scholar] [CrossRef]
- Djordjevic, D.; Wiedmann, M.; McLandsborough, L.A. Microtiter Plate Assay for Assessment of Listeria monocytogenes Biofilm Formation. Appl. Environ. Microbiol. 2002, 68, 2950–2958. [Google Scholar] [CrossRef] [Green Version]
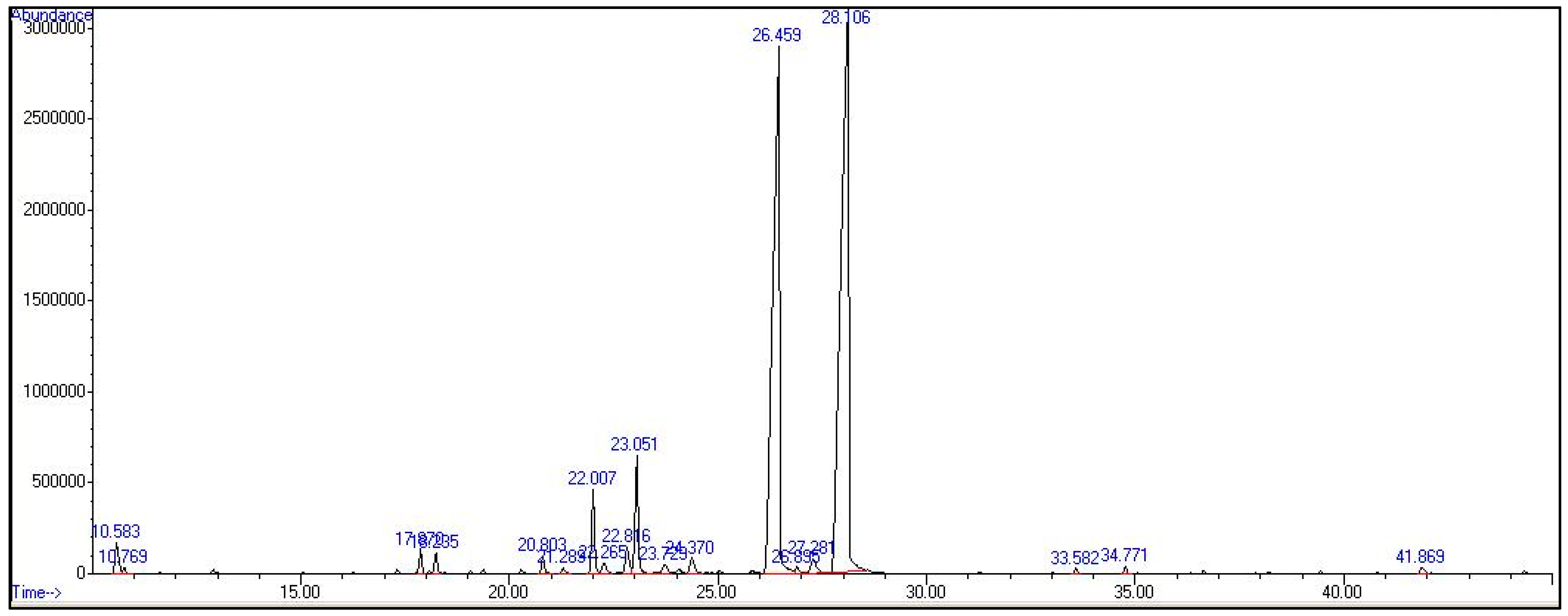
| N | Compound a | Molecular Formula | Compound b Group | RT c | KI d | Area Percentage (%) * |
|---|---|---|---|---|---|---|
| 1 | 6-methyl-5-hepten-2-one | C8H14O | Other | 10.583 | 994 | 1.02 |
| 2 | β-Myrcene | C10H16 | MH | 10.769 | 998 | 0.22 |
| 3 | (2-methylprop-1-enyl)-cyclohexa-1,5-diene | C10H14 | Other | 12.904 | 1030 | 0.12 |
| 4 | Rosefuran | C10H14O | OM | 17.870 | 1105 | 0.39 |
| 5 | Linalool | C10H18O | OM | 18.235 | 1110 | 0.53 |
| 6 | p-mentha-E-2,8(9)-dien-1-ol | C10H16O | OM | 20.279 | 1145 | 0.10 |
| 7 | Trifluoroacetyl-lavandulol | C12H17F3O2 | Other | 20.803 | 1154 | 0.33 |
| 8 | Citronella | C10H18O | OM | 21.289 | 1163 | 0.11 |
| 9 | Cyclopropene | C3H4 | Other | 22.007 | 1175 | 1.40 |
| 10 | α-Phellandren-8-ol | C10H16O | OM | 22.265 | 1179 | 0.32 |
| 11 | Cyclohexane, ethenyl- | C8H12 | Other | 23.051 | 1192 | 2.72 |
| 12 | Trans-p-menth-2-en-1,8-diol | C10H18O2 | OM | 23.729 | 1204 | 0.20 |
| 13 | β-Methylcrotonaldehyde | C5H8O | Other | 24.060 | 1211 | 0.18 |
| 14 | Cis-Carveol | C10H16O | OM | 24.370 | 1216 | 0.10 |
| 15 | Tetracyclo [3.3.0(2,6).0(3,9)]decan-2-ol | C10H14O | OM | 25.032 | 1229 | 0.42 |
| 16 | Neral | C10H16O | OM | 26.459 | 1256 | 37.65 |
| 17 | Geranial | C10H16O | OM | 28.106 | 1272 | 52.13 |
| 18 | Cinnamic acid | C9H8O2 | Other | 32.551 | 1379 | 0.12 |
| 19 | 1-Propanesulfonothioic acid | C6H14O2S2 | Other | 33.582 | 1383 | 0.11 |
| 20 | α-Gurjunene | C15H24 | SH | 34.771 | 1407 | 0.15 |
| 21 | Germacrene B | C15H24 | SH | 41.869 | 1556 | 0.18 |
| Total | 98.50 | |||||
| OM | 91.95 | |||||
| MH | 0.22 | |||||
| SH | 0.33 | |||||
| Others | 6.00 |
| Bacteria | Zone of Inhibition (mm) | |
|---|---|---|
| BCEO | Gentamicin | |
| Staphylococcus aureus | 31.13 ± 0.29 a | 22.30 ± 0.60 b |
| Staphylococcus epidermidis | 50.17 ± 0.29 a | 24.30 ± 0.60 b |
| Escherichia coli | 20.33 ± 0.58 a | 20.00 ± 0.00 a |
| Klebsiella pneumoniae | 12.67 ± 0.58 b | 18.00 ± 0.00 a |
| Bacteria | BCEO | Streptomycin | ||
|---|---|---|---|---|
| MIC | MBC | MIC | MBC | |
| Staphylococcus aureus | 6.25 | 50.00 | 15.63 | 125.00 |
| Staphylococcus epidermidis | 6.25 | 50.00 | 10.00 | 64.00 |
| Escherichia coli | 12.50 | 50.00 | 7.81 | 15.63 |
| Klebsiella pneumoniae | 12.50 | 50.00 | 3.91 | 31.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, A.C.; Tang, S.G.H.; Zin, N.M.; Maisarah, A.M.; Ariffin, I.A.; Ker, P.J.; Mahlia, T.M.I. Chemical Composition, Antioxidant, Antibacterial, and Antibiofilm Activities of Backhousia citriodora Essential Oil. Molecules 2022, 27, 4895. https://doi.org/10.3390/molecules27154895
Lim AC, Tang SGH, Zin NM, Maisarah AM, Ariffin IA, Ker PJ, Mahlia TMI. Chemical Composition, Antioxidant, Antibacterial, and Antibiofilm Activities of Backhousia citriodora Essential Oil. Molecules. 2022; 27(15):4895. https://doi.org/10.3390/molecules27154895
Chicago/Turabian StyleLim, Ann Chie, Shirley Gee Hoon Tang, Noraziah Mohamad Zin, Abdul Mutalib Maisarah, Indang Ariati Ariffin, Pin Jern Ker, and Teuku Meurah Indra Mahlia. 2022. "Chemical Composition, Antioxidant, Antibacterial, and Antibiofilm Activities of Backhousia citriodora Essential Oil" Molecules 27, no. 15: 4895. https://doi.org/10.3390/molecules27154895
APA StyleLim, A. C., Tang, S. G. H., Zin, N. M., Maisarah, A. M., Ariffin, I. A., Ker, P. J., & Mahlia, T. M. I. (2022). Chemical Composition, Antioxidant, Antibacterial, and Antibiofilm Activities of Backhousia citriodora Essential Oil. Molecules, 27(15), 4895. https://doi.org/10.3390/molecules27154895










