Biological and Cosmetical Importance of Fermented Raw Materials: An Overview
Abstract
1. Introduction
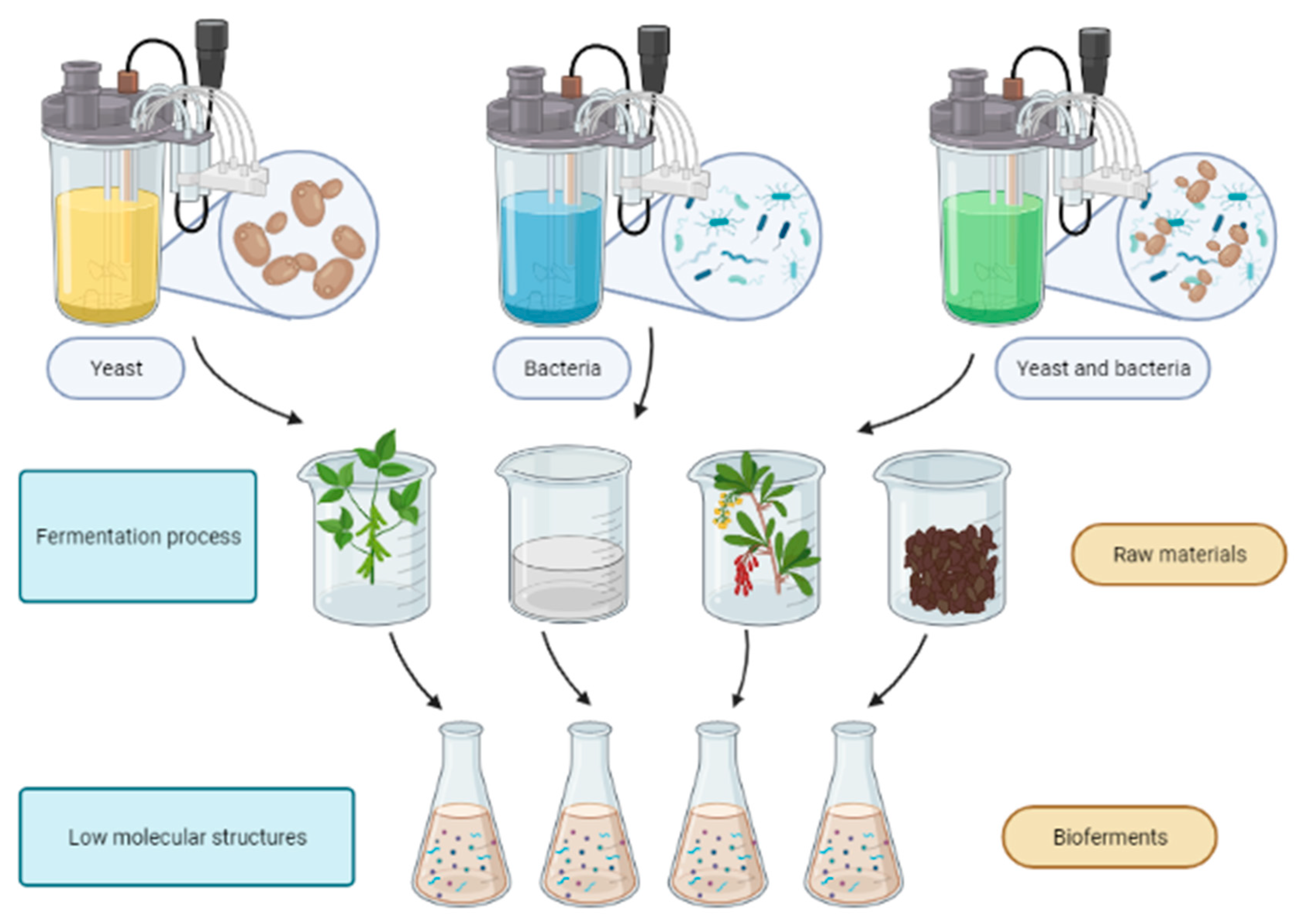
2. Overview of Bioferments
2.1. Microbial Products in Cosmetics
2.2. Soybean
2.3. Skimmed Milk
2.4. Ginseng
2.5. Aloe Vera
2.6. Berries
2.7. Agro-Food Waste
2.8. Herbal and Plant Mixtures
| Raw Material | Microorganisms | Properties | References |
|---|---|---|---|
| Blackberry 1 | Lactobacillus plantarum | Antioxidant, skin whitening | [11] |
| Fructus arctii1 | Grifola frondosa HB0071 | Anti-ageing, anti-wrinkle | [15] |
| Astragalus membranaceus var. mongholicus1 | Bacillus subtilis natto, Bacillus subtilis ATCC 7059 | Anti-ageing, anti-inflammatory, and skincare | [15] |
| Red ginseng 1 | Lactobacillus brevis or Saccharomyces cerevisiae | Anti-wrinkle, skincare, anti-inflammatory, and anti-allergenic | [15] |
| Prunus persica (L.) Batsch, Paeonia suffruticosa Andr., and Asparagus cochinchinensis (Loureiro) Merrill2 | Bifidobacterium bifidum | Antioxidant, skin whitening, and reduction of discoloration | [15] |
| Black ginseng 1 | Saccharomyces cerevisiae | Anti-wrinkle, antioxidant | [15] |
| Codonopsis lanceolata1 | Lactobacillus rhamnosus GG | Skin whitening, reduction of discoloration | [15] |
| clove, black galingale, betel, noni, green tea, and mangosteen 3 | Lactobacillus plantarum | Anti-inflammatory, promotes wound healing | [15] |
| Soybean 1 | Bifidobacterium animalis Saccharomyces cerevisiae Bacillus subtilis | Improve skin hydration and elasticity, antioxidant, skin whitening, and reduction of discoloration | [15,21,30] |
| Citrus unshiu peel 1 | Schizophyllum commune QG143 | Anti-ageing, anti-photo-aging | [15,59] |
| Wasabi root 1 | Lactobacillus | Antimicrobial, preservation, and antioxidant, anti-inflammatory | [21] |
| Lemon peel 1 | Lactobacillus lactis | Skin whitening, reduction of discoloration, and antioxidant | [21] |
| Tonka bean 1 | Lactobacillus | Anti-ageing, anti-photo-aging | [21] |
| Mannitol and maltodextrin: | Lactobacillus | Protects against allergens, free radicals, photo-aging, and pollution; increases skin regeneration; strengthens the protective barrier of sensitive skin; reduces redness and irritation; bacteriostatic effect against skin pathogens such as Staphylococcus aureus | [21] |
| Carrot root 1 | Bacillus ginsengisoli | Antioxidant, improving skin dullness, anti-ageing, and anti-wrinkle | [21] |
| Radish root 1 | Lactobacillus casei | Antioxidant, heals damaged cells, clears toxins, nourishes the skin, and anti-ageing | [21] |
| Sea kelp 1 | Lactobacillus | Natural film, excellent oil-free moisturizer | [21] |
| Skim milk | S. thermophilus YIT 2001, S. thermophilus YIT 2084 | Skin hydration, antioxidant, and maintains proper pH of the skin | [26] |
| Aloe vera 1 | L. plantarum YIT 0102 | The greatest (400%) skin hydration effect | [26] |
| Soybean milk | Bifidobacterium breve Yakult | Improves skin hydration and elasticity | [26] |
| Goji berry (Lycium barbarum L.) 1 | Lactobacillus rhamnosus, Lactobacillus reuteri Bacillus velezensis or Lactobacillus rhamnosus, Lactobacillus plantarum, Bacillus velezensis Bacillus licheniformis | Antioxidant, skin whitening, and anti-ageing | [53] |
| Walnut, Moutan Cortex Radicis and asparagus root 4 | B. bifidum | Antioxidant, skin whitening, and anti-photo-aging | [62] |
| Glycyrrhiza glabra, Broussonetia kazinoki, Angelica gigas, Atractylodes macrocephala, Poria cocos, Morus alba (root), Paeonia albiflora and Lithospermum officinale 1 | Phellinus linteus | Skin whitening, reduction of discoloration | [63] |
| Camellia sinensis (black, green, and white tea) 1 | Alcaligenes piechaudii CC-ESB2 | Antioxidant, skin whitening, and reduction of discoloration | [64] |
2.9. Cosmetic Kombucha
2.10. Bio-Oils
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Natural Cosmetics Market Size, Share, Trends Analysis Report By Product (Skin Care, Hair Care, Fragrance, Color Cosmetics), By Distribution Channel (Supermarket/Hypermarket, Online), and Segment Forecasts, 2019–2025. Available online: https://www.grandviewresearch.com/industry-analysis/natural-cosmetics-market (accessed on 23 May 2022).
- Rosholt, A.P. Regulatory Perspective of Natural and Organic Cosmetic Products. In Formulating, Packaging, and Marketing of Natural Cosmetic Products, 1st ed.; Nava, D., Lambros, K., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2011; pp. 49–65. [Google Scholar] [CrossRef]
- Juliano, C.; Magrini, G.A. Cosmetic Ingredients as Emerging Pollutants of Environmental and Health Concern. A Mini-Review. Cosmetics 2017, 4, 11. [Google Scholar] [CrossRef]
- Fonseca-Santos, B.; Corrêa, M.; Chorilli, M. Sustainability, natural and organic cosmetics: Consumer, products, efficacy, toxicological and regulatory considerations. Braz. J. Pharm. Sci. 2015, 51, 17–26. [Google Scholar] [CrossRef]
- COSMOS Natural and Organic Certification for Cosmetics. Available online: https://www.cosmos-standard.org/ (accessed on 23 May 2022).
- Ecocert|Act for a More Sustainable World. Available online: https://www.ecocert.com/en/home/ (accessed on 23 May 2022).
- U.S. Food and Drug Administration—Cosmetics Laws & Regulations. Available online: https://www.fda.gov/cosmetics/cosmetics-guidance-regulation/cosmetics-laws-regulations (accessed on 23 May 2022).
- Cosmetics European Commission. Available online: https://ec.europa.eu/growth/sectors/cosmetics_en (accessed on 23 May 2022).
- Choi, N.M.; Choi, J.I. Fermented Cosmetics. Korean Patent KR1430591, 18 August 2014. [Google Scholar]
- Joon, S.S.; Song, H.X.; So, H.P.; Keon, S.L.; Young, M.P.; Soo, N.P. Antioxidative and antiaging activities and component analysis of Lespedeza cuneata G. don extracts fermented with Lactobacillus pentosus. J. Microbiol. Biotechnol. 2017, 27, 1961–1970. [Google Scholar] [CrossRef]
- Yoseph, C.; Avanti, C.; Hadiwidjaja, M.; Briliani, K. The importance of fermented plant extract for anti aging in cosmetics product. World J. Pharm. Res. 2021, 10, 1330–1343. [Google Scholar]
- Mathur, H.; Beresford, T.P.; Cotter, P.D. Health benefits of lactic acid bacteria (LAB) fermentates. Nutrients 2020, 12, 1679. [Google Scholar] [CrossRef]
- Mi-sun, L.; Shun-Hua, L. Research on the perception of fermented cosmetics and the satisfaction level of ‘Korean Medicine Herb’ cosmetics. Asian J. Beauty Cosmetol. 2010, 8, 285–292. [Google Scholar]
- Tan, H. Hair Dye Prepared from Bio-Ferment and Having Antipruritic and Antistatic Effects. Chinese Patent WO2018032837, 22 February 2018. [Google Scholar]
- Sivamaruthi, B.S.; Chaiyasut, C.; Kesika, P. Cosmeceutical importance of fermented plant extracts: A short review. Int. J. Appl. Pharm. 2018, 10, 31–34. [Google Scholar] [CrossRef]
- Rajput, K.N.; Patel, K.C.; Trivedi, U.B. A novel cyclodextrin glucanotransferase from an alkaliphile Microbacterium terrae KNR 9: Purification and properties. 3Biotech 2016, 6, 168. [Google Scholar] [CrossRef]
- Singh, P.; Patil, Y.; Rale, V. Biosurfactant production: Emerging trends and promising strategies. J. Appl. Microbiol. 2018, 126, 2–13. [Google Scholar] [CrossRef]
- Bezerra, K.G.O.; Rufino, R.D.; Luna, J.M.; Sarubbo, L.A. Saponins and microbial biosurfactants: Potential raw materials for the formulation of cosmetics. Biotechnol. Prog. 2018, 34, 1482–1493. [Google Scholar] [CrossRef]
- Gupta, P.L.; Rajput, M.; Oza, T.; Ujwalkumar, T.; Gaurav, S. Eminence of Microbial Products in Cosmetic Industry. Nat. Prod. Bioprospect. 2019, 9, 267–278. [Google Scholar] [CrossRef]
- Chung-Yi, W.; Chang-Chai, N.; Hsin-Tang, L.; Yuan-Tay, S. Free radical-scavenging and tyrosinase-inhibiting activities of extracts from sorghum distillery residue. J. Biosci. Bioeng. 2011, 111, 554–556. [Google Scholar] [CrossRef]
- Singh, V.K. Bioferments in beauty infusing antioxidant and microbiome benefits into skin. Cosmet. Toilet. 2022, 137, 59–68. [Google Scholar]
- Kim, M.; Kim, W.B.; Koo, K.Y.; Kim, B.R.; Kim, D.; Lee, S.; Son, H.J.; Hwang, D.Y.; Kim, D.S.; Lee, C.Y.; et al. Optimal fermentation conditions of hyaluronidase inhibition activity on Asparagus cochinchinensis Merrill by Weissella cibaria. J. Microbiol. Biotechnol. 2017, 27, 701–708. [Google Scholar] [CrossRef]
- Wang, G.H.; Chen, C.Y.; Tsai, T.H.; Chen, C.K.; Cheng, C.Y.; Huang, Y.H.; Hsieh, M.C.; Chung, Y.C. Evaluation of tyrosinase inhibitory and anti oxidant activities of Angelica dahurica root extracts for four different probiotic bacteria fermentations. J. Biosci. Bioeng. 2017, 123, 679–684. [Google Scholar] [CrossRef]
- Kang, Y.M.; Hong, C.H.; Kang, S.H.; Seo, D.S.; Kim, S.O.; Lee, H.Y.; Sim, H.J.; An, H.J. Anti-Photoaging Effect of Plant Extract Fermented with Lactobacillus buchneri on CCD-986sk Fibroblasts and HaCaT Keratinocytes. J. Funct. Biomater. 2020, 11, 3. [Google Scholar] [CrossRef]
- Miyazaki, K.; Hanamizu, T.; Sone, T.; Chiba, K.; Kinoshita, T.; Yoshikawa, S. Topical application of Bifidobacterium -fermented soy milk extract containing genistein and daizein improves rheological and physiological properties of skin. Int. J. Cosmet. Sci. 2004, 55, 473–479. [Google Scholar]
- Izawa, N.; Sone, T. Cosmetic Ingredients Fermented by Lactic Acid Bacteria. Microb. Prod. 2014, 2, 233–242. [Google Scholar] [CrossRef]
- Miyazaki, K. Availability of soy isoflavones and fermented soy products in foods and cosmetics. Foods Food Ingred. J. Jpn. 2002, 204, 5–11. [Google Scholar]
- Kobayashi, K.; Horii, Y.; Watanabe, S.; Kubo, Y.; Koguchi, K.; Hoshi, Y.; Matsumoto, K.; Soda, K. Comparison of soybean cultivars for enhancement of the polyamine contents in the fermented soybean natto using Bacillus subtilis (natto). Biosci. Biotechnol. Biochem. 2017, 81, 587–594. [Google Scholar] [CrossRef]
- Leksono, B.Y.; Cahyanto, M.N.; Utami, T. Antioxidant activity of isoflavone aglycone from fermented black soymilk supplemented with sucrose and skim milk using indonesian indigenous lactic acid bacteria. Appl. Food Biotechnol. 2021, 8, 285–295. [Google Scholar] [CrossRef]
- Chae, G.Y.; Ha, B.J. The Comparative Evaluation of Fermented and Non-fermented Soybean Extract on Antioxidation and Whitening. Toxicol. Res. 2011, 27, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Zhu, N.; Wang, M.; Wei, G.J.; Lin, J.K.; Yang, C.S.; Ho, C.T. Identification of reaction products of (-)-epigallocatechin, (-)-epigallocatechin gallate and pyrogallol with 2,2- diphenyl-1 picrylhydrazyl radical. Food Chem. 2001, 73, 2345–2349. [Google Scholar] [CrossRef]
- Cho, S.H.; Park, B.Y.; Kim, J.H.; Kim, Y.K.; Lee, J.M.; Ahn, C.N. Effect of r-oryzanol on lipid oxidation of linoleic acids and ground pork. J. Anim. Sci. Technol. 2006, 48, 587–594. [Google Scholar] [CrossRef]
- Liaqat, H.M.; Hameed, M.; Iftikhar, M.U. Case series study on glutathione used as a skin whitening agent. Indo Am. J. Pharm. Sci. 2020, 7, 1863–1868. [Google Scholar]
- Toro-Funes, N.; Bosch-Fuste, J.; Latorre-Moratalla, M.L.; Veciana-Nogues, M.T.; Vidal-Carou, M.C. Biologically active amines in fermented and non-fermented commercial soybean products from the Spanish market. Food Chem. 2015, 173, 1119–1124. [Google Scholar] [CrossRef]
- Namgung, H.J.; Park, H.J.; Cho, I.H.; Choi, H.K.; Kwon, D.Y.; Shim, S.M.; Kim, Y.S. Metabolite profiling of doenjang, fermented soybean paste, during fermentation. J. Sci. Food Agric. 2010, 90, 1926–1935. [Google Scholar] [CrossRef]
- Izawa, N.; Sone, T. Development of skin care ingredients using lactic acid bacteria and Bifidobacterium. Fragr. J. 2012, 40, 72–77. [Google Scholar]
- Zhang, T.; Zhang, Z.; Yan, H.; Li, D.; Yang, Z.; Guo, M. Effects of stabilizers and exopolysaccharides on physiochemical properties of fermented skim milk by Streptococcus thermophilus ST1. Afr. J. Biotechnol. 2012, 11, 6123–6130. [Google Scholar] [CrossRef]
- Weindl, G.; Schaller, M.; Schäfer-Korting, M.; Korting, H.C. Hyaluronic acid in the treatment and prevention of skin diseases: Molecular biological, pharmaceutical and clinical aspects. Ski. Pharmacol. Physiol. 2004, 17, 207–213. [Google Scholar] [CrossRef]
- Hati, S.; Patel, N.; Mandal, S. Comparative Growth Behaviour and Biofunctionality of Lactic Acid Bacteria During Fermentation of Soy Milk and Bovine Milk. Probiotics Antimicrob. Proteins 2018, 10, 277–283. [Google Scholar] [CrossRef]
- Korcz, E.; Varga, L.; Kerenyi, Z. Relationship between total cell counts and exopolysaccharide production of Streptococcus thermophilus T9 in reconstituted skim milk. LWT-Food Sci. Technol. 2021, 148, 111775. [Google Scholar] [CrossRef]
- Pham, Q.L.; Jang, H.J.; Kim, K.B. Anti-wrinkle effect of fermented black ginseng on human fibroblasts. Int. J. Mol. Med. 2017, 39, 681–686. [Google Scholar] [CrossRef]
- Park, J.L.; An, J.; Lee, D.L.; Kim, H.Y.; Ha, J.; Cho, C.H.; Seo, D.W.; Kim, K.B. Effects of anti-wrinkle and skin-whitening fermented black ginseng on human subjects and underlying mechanism of action. J. Toxicol. Environ. Health 2020, 83, 470–484. [Google Scholar] [CrossRef]
- Jin, Y.; Jung, S.Y.; Kim, Y.J.; Lee, D.Y.; Min, J.W.; Wang, C.; Yang, D.C. Microbial ketonization of ginsenosides F1 and C-K by Lactobacillus brevis. Antonie Van Leeuwenhoek 2014, 106, 1215–1221. [Google Scholar] [CrossRef]
- Lee, H.S.; Kim, M.R.; Park, Y.; Park, H.J.; Chang, U.J.; Kim, S.Y.; Suh, H.J. Fermenting Red Ginseng Enhances Its Safety and Efficacy as a Novel Skin Care Anti-Aging Ingredient: In Vitro and Animal Study. J. Med. Food 2012, 15, 1015–1023. [Google Scholar] [CrossRef]
- Dini, I.; Laneri, S. The New Challenge of Green Cosmetics: Natural Food Ingredients for Cosmetic Formulations. Molecules 2021, 26, 3921. [Google Scholar] [CrossRef]
- Sone, T.; Hanamizu, T.; Chiba, K. Plant ingredients fermented by lactic acid bacteria. Fragrance 2005, 33, 65–70. [Google Scholar]
- Robinson, M.; Visscher, M.; Laruffa, A.; Wickett, R. Natural moisturizing factors (NMF) in the stratum corneum (SC). I. Effects of lipid extraction and soaking. J. Cosmet. Sci. 2010, 61, 13–22. [Google Scholar]
- Hai, Z.; Ren, Y.; Hu, J.; Wang, H.; Qin, Q.; Chen, T. Evaluation of the treatment effect of Aloe vera fermentation in burn injury healing using a rat model. Mediat. Inflamm. 2019. [Google Scholar] [CrossRef]
- Ro, H.S.; Jang, H.J.; Kim, G.R.; Park, S.J.; Lee, H.Y. Enhancement of the Anti-Skin Wrinkling Effects of Aloe arborescens Miller Extracts Associated with Lactic Acid Fermentation. Evid. Based Complement. Altern. Med. Ecam 2020. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Wei, K.; Lu, J.; Wei, J.; Hu, X.; Chen, T. A Clinic Trial Evaluating the Effects of Aloe Vera Fermentation Gel on Recurrent Aphthous Stomatitis. Can. J. Infect. Dis. Med. Microbiol. 2020, 2020, 8867548. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Jeong, D.H.; Kim, S.; Lee, S.W.; Sin, H.S.; Yu, K.Y.; Jeong, S.I.; Kim, S.Y. Fermentation of Blackberry with L. plantarum JBMI F5 Enhance the Protection Effect on UVB-Mediated Photoaging in Human Foreskin Fibroblast and Hairless Mice through Regulation of MAPK/NF-κB Signaling. Nutrients 2019, 11, 2429. [Google Scholar] [CrossRef] [PubMed]
- Qiong, L.; Yizhong, C.; Jun, Y.; Mei, S.; Harold, C. Hypoglycemic and hypolipidemic effects and antioxidant activity of fruit extracts from Lycium barbarum. Life Sci. 2004, 76, 137–149. [Google Scholar] [CrossRef]
- Liu, Y.; Cheng, H.; Liu, H.; Ma, R.; Ma, J.; Fang, H. Fermentation by Multiple Bacterial Strains Improves the Production of Bioactive Compounds and Antioxidant Activity of Goji Juice. Molecules 2019, 24, 3519. [Google Scholar] [CrossRef]
- Markkinen, N.; Laaksonen, O.; Nahku, R.; Kuldjarv, R.; Yang, B. Impact of lactic acid fermentation on acids, sugars, and phenolic compounds in black chokeberry and sea buckthorn juices. Food Chem. 2019, 286, 204–215. [Google Scholar] [CrossRef]
- Kaprasob, R.; Kerdchoechuen, O.; Laohakunjit, N.; Sarkar, D.; Shetty, K. Fermentation-based biotransformation of bioactive phenolics and volatile compounds from cashew apple juice by select lactic acid bacteria. Process. Biochem. 2017, 59, 141–149. [Google Scholar] [CrossRef]
- Luo, C.; Chen, Y.; Liu, X.; Wang, X.; Li, X.; Zhao, Y.; Wei, L. Engineered biosynthesis of cyclic lipopeptide locillomycins in surrogate host Bacillus velezensis FZB42 and derivative strains enhance antibacterial activity. Appl. Microbiol. Biotechnol. 2019, 103, 4467–4481. [Google Scholar] [CrossRef]
- Hur, S.J.; Lee, S.Y.; Kim, Y.; Choi, I.; Kim, G. Effect of fermentation on the antioxidant activity in plant-based foods. Food Chem. 2014, 160, 346–356. [Google Scholar] [CrossRef]
- Chreptowicz, K.; Sternicka, M.K.; Kowalska, P.D.; Mierzejewska, J. Screening of yeasts for the production of 2-phenylethanol (rose aroma) in organic waste-based media. Lett. Appl. Microbiol. 2018, 66, 153–160. [Google Scholar] [CrossRef]
- Jun, T.B.; Hyun, J.K.; Gyoung, B.K.; Hyeong, B.P.; Geun, S.L. Protective Effects of Fermented Citrus Unshiu Peel Extract against Ultraviolet-A-induced Photoageing in Human Dermal Fibrobolasts. Phytother. Research 2012, 26, 1851–1856. [Google Scholar] [CrossRef]
- Hai-li, G. Optimization of Extraction and Purification of Condition and Evaluation of Antioxidant Activities of the Total Flavonoids in Dendrobium Officinale Leaves; Sci-Tech University: Zhenjiang, China, 2019. [Google Scholar]
- Bajpai, P. Use of Mixed Cultures. In Single Cell Protein Production from Lignocellulosic Biomass. SpringerBriefs in Molecular Science; Springer: Singapore, 2017. [Google Scholar] [CrossRef]
- Yuanyuan, L.; Ting, Y.; Luoyi, S.; Jie, Z. Liping, L. Study on the properties of Dendrobiumofficinale fermentation broth as functional raw material of cosmetics. J. Cosmet. Dermatol. 2022, 21, 1216–1223. [Google Scholar] [CrossRef]
- Wang, G.H.; Chen, C.Y.; Lin, C.P.; Huang, C.L.; Lin, C.; Cheng, C.Y.; Chung, Y.C. Tyrosinase inhibitory and antioxidant activities of three Bifidobacterium bifidum-fermented herb extracts. Ind. Crops Prod. 2016, 89, 376–382. [Google Scholar] [CrossRef]
- Cha, J.Y.; Yang, H.J.; Moon, H.I.; Cho, Y.S. Inhibitory effect and mechanism on melanogenesis from fermented herbal composition for medical or food uses. Food Res. Int. 2012, 45, 225–231. [Google Scholar] [CrossRef]
- Kim, Y.C.; Choi, S.Y.; Park, E.Y. Anti-melanogenic effects of black, green, and white tea extracts on immortalized melanocytes. J. Vet. Sci. 2015, 16, 135–143. [Google Scholar] [CrossRef]
- Dutta, H.; Paul, S.K. Kombucha drink: Production, quality, and safety aspects. Prod. Manag. Beverages 2019, 1, 259–288. [Google Scholar] [CrossRef]
- Marsh, A.J.; O’Sullivan, O.; Hill, C.; Ross, R.P.; Cotter, P.D. Sequence-based analysis of the bacterial and fungal compositions of multiple Kombucha (tea fungus) samples. Food Microbiol. 2014, 38, 171–178. [Google Scholar] [CrossRef]
- Ziemlewska, A.; Nizioł-Łukaszewska, Z.; Bujak, T.; Zagórska-Dziok, M.; Wójciak, M.; Sowa, I. Effect of fermentation time on the content of bioactive compounds with cosmetic and dermatological properties in Kombucha Yerba Mate extracts. Sci. Rep. Nat. 2021, 11, 18792. [Google Scholar] [CrossRef]
- Aguilar-Toalá, J.; Garcia-Varela, R.; Garcia, H.; Mata-Haro, V.; González-Córdova, A.; Vallejo-Cordoba, B.; Hernández-Mendoza, A. Postbiotics: An evolving term within the functional foods field. Trends Food Sci. Technol. 2018, 75, 105–114. [Google Scholar] [CrossRef]
- Egert, M.; Simmering, R.; Riedel, C.U. The Association of the Skin Microbiota with Health, Immunity, and Disease. Clin. Pharmacol. Ther. 2017, 102, 62–69. [Google Scholar] [CrossRef]
- Ciardiello, T.; Pinto, D.; Marotta, L.; Giuliani, G.; Rinaldi, F. Effects of Fermented Oils on Alpha-Biodiversity and Relative Abundance of Cheek Resident Skin Microbiota. Cosmetics 2020, 7, 34. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Majchrzak, W.; Motyl, I.; Śmigielski, K. Biological and Cosmetical Importance of Fermented Raw Materials: An Overview. Molecules 2022, 27, 4845. https://doi.org/10.3390/molecules27154845
Majchrzak W, Motyl I, Śmigielski K. Biological and Cosmetical Importance of Fermented Raw Materials: An Overview. Molecules. 2022; 27(15):4845. https://doi.org/10.3390/molecules27154845
Chicago/Turabian StyleMajchrzak, Weronika, Ilona Motyl, and Krzysztof Śmigielski. 2022. "Biological and Cosmetical Importance of Fermented Raw Materials: An Overview" Molecules 27, no. 15: 4845. https://doi.org/10.3390/molecules27154845
APA StyleMajchrzak, W., Motyl, I., & Śmigielski, K. (2022). Biological and Cosmetical Importance of Fermented Raw Materials: An Overview. Molecules, 27(15), 4845. https://doi.org/10.3390/molecules27154845






