Abstract
Artemisia abrotanum L. (southern wormwood) is a plant species with an important position in the history of European and Asian medicine. It is a species famous as a medicinal plant in Central Asia, Asia Minor, and in South-East and Central Europe. The raw materials obtained from this species are Abrotani herba and Abrotani folium. In the traditional European medicine, they have been used successfully most of all in liver and biliary tract diseases, in parasitic diseases in children and as antipyretic medication. In the official European medicine, this plant species is recommended by the French Pharmacopoeia for use in homeopathy. In many European countries, it is used traditionally in allopathy. The latest studies on the biological activity of extracts from the aboveground parts of the plant and/or the leaves, and/or the essential oil have provided evidence of other possible applications related to their antibacterial, antifungal, antioxidant, anticancer, and antiallergic properties. The latest studies have also focused on the repellent activity of the essential oil of this species and the possibility to use it in the prevention of diseases in which insects are the vectors. The main substances obtained from the plant that are responsible for this activity are: the essential oil, coumarins, phenolic acids, and flavonoids. Some of the latest investigations emphasize the large differences in the composition of the essential oil, determined by the geographical (climatic) origin of the plant. A. abrotanum is recommended by the European Cosmetic Ingredients Database (CosIng) as a source of valuable cosmetic ingredients. Additionally, the leaves of this species possess a well-established position in the food industry. This plant species is also the object of biotechnological studies.
1. Introduction
The awarding of the 2015 Nobel Prize in Medicine for the discovery of artemisinin [1,2], a sesquiterpenoid lactone, in Artemisia annua (annual mugwort) and proving its effectiveness in the treatment of malaria sparked a huge interest in the world of science in the chemistry and biological activity of other Artemisia species [3,4,5,6,7]. One of the better-known medicinal species of this genus in Europe and Asia is Artemisia abrotanum L. (Asteraceae). This species occurs in the south-eastern part of Europe and in the countries of Central and North-Western Europe. This species also has its natural habitats in Asia Minor, Central Asia, and on the Arabian Peninsula [8]. The herb of this species—Abrotani herba— occupies an important position in the traditional medicine of European countries [8,9,10,11]. It is recommended most often for the treatment of diseases of the liver and the biliary tract. It is also used as an effective anthelmintic in children, and as an antipyretic. Recent professional studies have proven previously unknown biological activities of extracts from the herb and/or the leaves, and/or the distilled-off essential oil, namely antibacterial, antifungal, antioxidant, anticancer, and antiallergic properties [8,12]. Apart from the essential oil, the coumarins, phenolic acids and flavonoids present in the chemical composition of the plant are also responsible for the abovementioned biological effects. This species is also traditionally an object of interest of the food industry [13]. A recent development was its approval by the European CosIng (Cosmetic Ingredients) database for use in the production of cosmetics and its growing role as a cosmetic plant [14]. The aim of this article is to present the current state of knowledge regarding the position of A. abrotanum in the history of medicine, its importance in modern traditional medicine, the chemical composition, new directions of biological activity and the resulting possible new therapeutic applications and prospects for use in cosmetology. The aim was also presentation of biotechnological studies of this species and its position in the food industry.
2. Position in the History of European and Asian Medicine
A. abrotanum is one of those medicinal herbs that can be found already in the first herbals of our civilization. Descriptions of it were included in both Dioscorides and Pliny the Elder [15,16]. Noteworthy is the origin of the name “abrotanum”, which, according to Theophrastus of Eresos, may refer to a certain constitution of the body, namely to very thin and frail people (αβρος (abrós) in Greek means ‘slim, delicate’) [17]. This reference is significant because it corresponds exactly to the constitution of patients who were originally recommended to use this herb, namely those suffering from chronic respiratory ailments. Of the lung diseases, the most difficult to treat over the millennia had been tuberculosis, leading to persistent wasting of the body, but chronic bronchitis or asthma may have clinically manifested themselves in this way as well. In holistic Traditional Chinese Medicine (TCM), this condition is called “Lung-Qi weakening, with particular weakening of the Yin of the lungs”, and is the dominant indication for TCM treatment with this herb to this day [18]. Apart from bronchopulmonary ailments, A. abrotanum was also used in a number of other diseases, and its effects were widely known. In the Arab world, Avicenna, citing Galen among others, recommended its use in the absence of appetite, ulcers, and skin diseases, for hair growth, and as an amulet against evil spirits [19]. The Slavs considered A. abrotanum to be a cult “church herb”, used, for example, for making wreaths, hence its Polish name “boże drzewko” (“God’s little tree”) [20]. In the Middle Ages, applications from antiquity were adopted, and in Germanic areas in the 10th century, the spectrum of treatments with Artemisia abrotanum included broadly understood difficulty in breathing, pain from the liver, spleen, bladder and uterus, anthelmintic treatment, wound healing, to assist the stomach, intensify diuresis, and deter evil spirits [21]. A hundred years later, Hildegard of Bingen insisted on using this herb in compresses for leg pain [22]. In the Renaissance era, due to the warming and drying properties of A. abrotanum, it was recommended for use in ailments caused by cold and humidity, which mainly concerned the female genital tract and the functional circuits of the liver and spleen, according to the holistic nomenclature. This view was shared by the “Fathers of Botany”—Otto Brunfels [23], Hieronymus Bock [24], and Leonhart Fuchs [25], and half a century later also by their worthy successor—Jacobus Tabernaemontanus [26]. At the apex of the development of holistic thought in Western medicine, the following secondary properties were attributed to A. abrotanum: “dispersing, diluting, (surface) opening and cleansing effects” [27]. One of the most important Polish renaissance herbals—“Zielnik” by Simon Syrenius, also mentions the plant’s drying and warming effects as well as its effectiveness as a remedy against “venoms and poisons” (after Dioscorides and Pliny), repelling “venomous animals”, and in diseases of the lungs and the digestive system, and in women’s diseases [28]. A. abrotanum was also used as an antipyretic, against internal parasites and externally in the treatment of scalp and hair diseases, and as an anti-inflammatory medication in eye diseases and ulcers of various origins. The plant was used to prepare an oil with analgesic and diuretic properties, wine stimulating the appetite and effective against jaundice, and vodka for respiratory ailments [28]. In 19th-century folk medicine, the herb A. abrotanum was known as a medicine for skin diseases (added to baths), for throat and ear ailments, for sprains (in the form of compresses), and for women’s and childhood diseases [29].
3. General Information on the Species
A. abrotanum has many synonymous Latin names, including Abrotanum alpestre Jord., A. ambiguum Jord. & Fourr., A. brachylobium Jord. & Fourr., A. congestum Jord. & Fourr., A. incanescens Jord. & Fourr., A. mas Garsault, A. pauciflorum Jord. & Fourr., A. pedunculare Jord. & Fourr., A. platylobum Jord. & Fourr., A. pulverulentum Jord. & Fourr., A. rhodanicum Jord. & Fourr., A. suave Jord. & Fourr., A. virgatum Jord. & Fourr., A. viridulum Jord. & Fourr., A. xerophilum Jord. & Fourr., Artemisia abrotanifolium Salisb., A. altissima Ehrh., A. altissima Ehrh. ex DC. A. anethifolia Fisch., A. anethifolia Fisch. ex DC. [30,31,32]. The species is also known by numerous common names, for example: lad’s love, lemonwood, old man, slovenwood, southern wormwood, and southernwood (in English), and nayqatamisa (ajmara), abrotone, armoise aurone, armoise citronnelle, aurone mâle, aurone, citronelle aurone, citronnelle (in French). In German, there are only two names—Eberraute and Stabwurz [31,32,33,34,35,36]. In Polish, the species is customarily called “boże drzewko” (literally “God’s little tree”, a counterpart of the English name “Our Lord’s wood”) [33]. The pharmaceutical raw material is Artemisiae abrotani herba—the herb of the A. abrotanum plant—the flowering tips of its shoots. The dry herb has a grey-green colour, a slightly bitter taste and a citrusy, spicy aroma [33,37]. In traditional medicine, the leaves of the plant—Artemisiae abrotani folium, also constitute the raw material [11]. A. abrotanum is a semi-shrub reaching a height of 0.7–1.5 m [38]. The shoots of this species grow upright and have soft twigs. Young twigs are blue-green and older ones are brown. The stem is covered with secretory hairs [39]. The stems are highly branched with dense foliage. The grey-green leaves have numerous covering hairs on the upper side; the underside of the leaves is smooth. The leaves growing in the lower part of the stem are doubly pinnate and have ensiform sections, while in the upper parts they take on a singly pinnate, tripartite, and also ensiform shape. The whole plant has a strong distinctive lemon-like aroma [5,33,37,40,41].
The tiny yellow tubular flowers are gathered in spherical or ovoid-spherical hanging heads that form panicles. In Central Europe, flowering of the plant begins in August and lasts until September. Due to the climatic conditions, the plant in this region does not bear fruit. The fruits are small oblong achenes [5,33,37,40,41]. Cariological studies of the plant have shown that its specimens are most often diploid (2n = 2x = 18), less often tetraploid (2n = 4x = 36) [38]. A. abrotanum is a species native to Central Asia—in particular Armenia, Iran and Russia, and also to Asia Minor—Turkey and Europe, including Albania and Croatia [36]. The species also occurs in Central and North-Western Europe [42].
A. abrotanum likes warm, sunny, and humus-rich sites, with well-drained, not too moist soils [33,43]. In Central and North-Western Europe, the species reproduces only vegetatively by division or from hardwood and softwood cuttings. In other parts of the world, if the species grows in a warm climate, it reproduces by seed. The first harvest takes place in the second year after planting. During the flowering period, the tips of the shoots are collected, which are then dried in shaded and airy natural drying enclosures. Larger batches of the raw material are dried in drying rooms with the temperature increased to 35 °C. It is also advisable to trim annually the lower, lignified parts of the plant. This treatment improves the branching and ‘bouncing-back’ of the herb, and increases the yield [33,43]. After drying, the woody parts are discarded and the non-lignified herb is placed in airtight packages and stored in the dark [39,41,43].
4. Chemical Composition
Analyses of the compounds present in A. abrotanum have focused primarily on the essential oil present in the herb of the species [9,11,40]. The composition and concentration of A. abrotanum essential oil are variable and dependent on the cultivation site, among other things [9]. The dominant fraction of the oil are compounds with a monoterpenoid structure. The remaining fractions are compounds of sesquiterpenoid, diterpenoid, triterpenoid, or spiroterpenoid structures, and phenylpropanoid derivatives (Table 1).

Table 1.
Chemical composition of A. abrotanum essential oil.
In 2020, Lithuanian researchers determined the composition of the essential oil obtained from the herb of A. abrotanum plants growing in Lithuania, which were at various stages of plant development. The compound that was present in the highest amounts was piperitone. The remaining substances isolated in large amounts were: 1,4-cineole, lavandulyl butanoate, aromadendrene, and isogermacrene D. The essential oil content in the herb of the plant was the highest at the stage of flower-bud setting (38.48%), and the lowest at the stage of leaf growth and development (20.38%) [9].
Egyptian researchers conducted a study in 2020 evaluating an influence of trace elements on productivity of A. abrotanum. The main components isolated from essential oil of A. abrotanum aerial parts were: 2-hydroxy-1,8-cineole, β-eudesmol, and camphor. In the experiment the scientists treated A. abrotanum with foliar spray containing iron (Fe), magnesium (Mg) or manganese (Mn). The foliar application of Fe (3 g/L), Mg (8 g/L) or Mn (300 mg/L) significantly increased values of growth, yield, major constituents of essential oil and chemical composition of the plant in comparison with control [46].
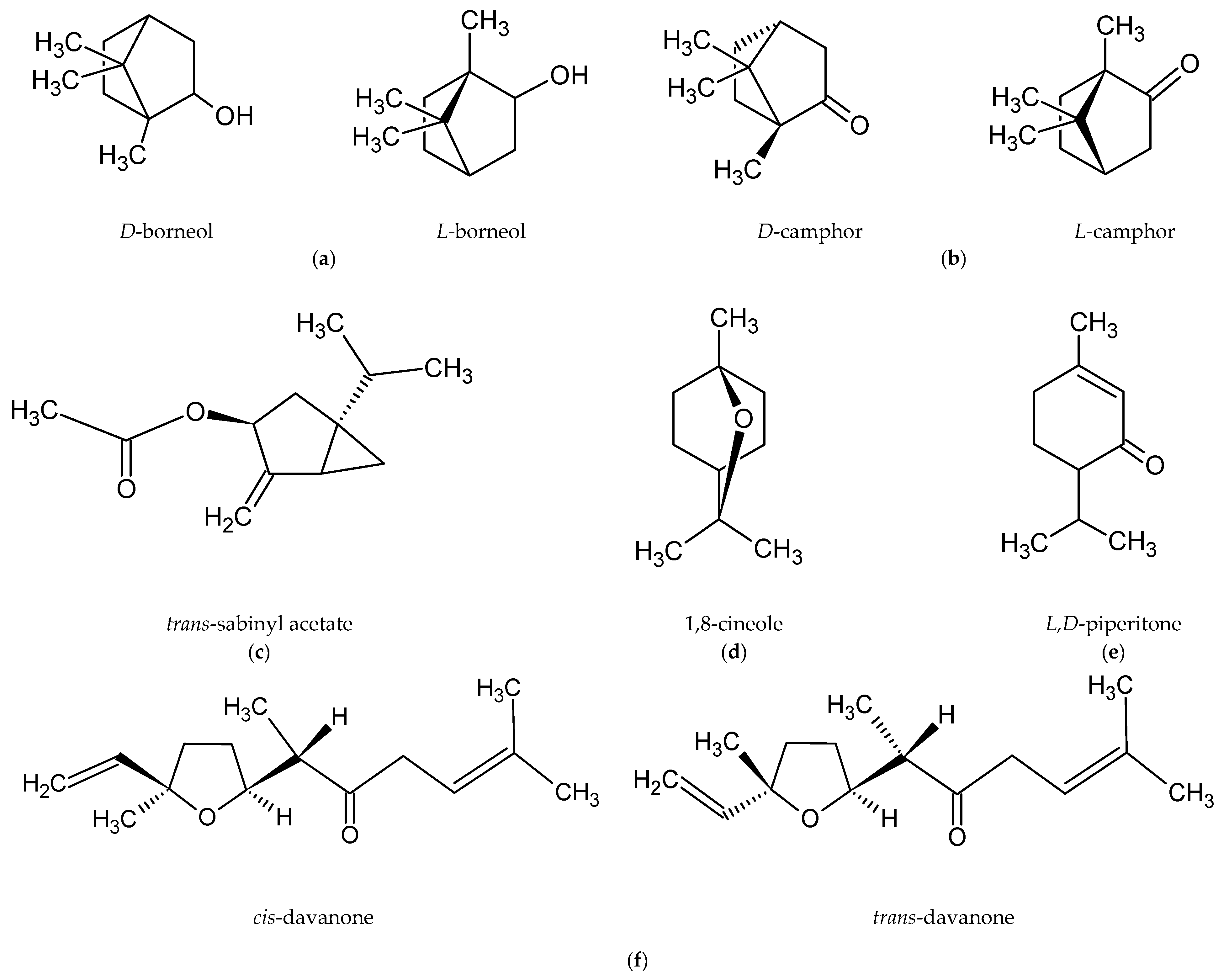
Romanian scientists have reported that the main component of the essential oil of A. abrotanum herb was davanone and its derivatives. The researchers also isolated, in small amounts, 1-terpineol, trans-piperitol, and estragole [47]. In contrast, the dominant compound in the essential oil of plants harvested in Cuba was trans-sabinyl acetate [44], and in the essential oil from plants harvested in Iraq borneol [48]. The main components in plants harvested in Crimea were 1,8-cineole and camphor [49]. The chemical structures of the most abundant compounds in A. abrotanum essential oil are shown in Figure 1.
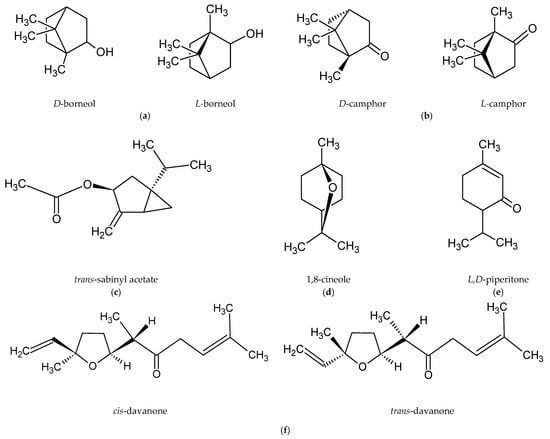
Figure 1.
Chemical structure of the dominant compounds in A. abrotanum essential oil: (a) borneol; (b) camphor; (c) trans-sabinyl acetate; (d) 1,8-cineole; (e) piperitone; (f) davanone.
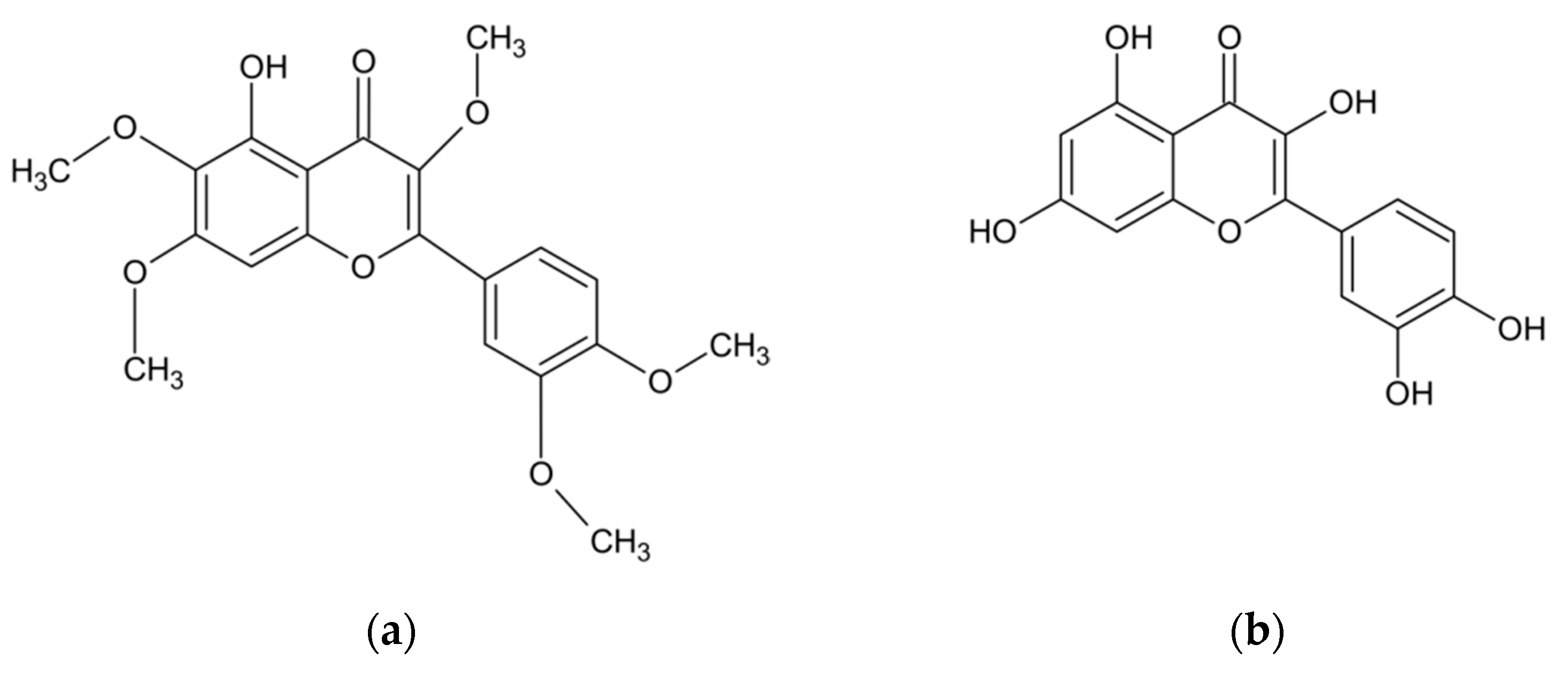
In addition to the essential oil, the herb of A. abrotanum also contains compounds with phenolic structures. The research conducted at JSS University in Mysore (India) has documented the presence of phenolic compounds in the amount of 9.35 mg/g dry weight in terms of gallic acid, and the presence of flavonoids in the amount of 11.0 mg/g dry weight in terms of rutoside [34]. Another study has proven the presence of phenolic acids. This group of compounds, present in the leaves of A. abrotanum plants growing in Saudi Arabia, was dominated by rosmarinic, chlorogenic, caffeic and isochlorogenic acids [40]. The structures of the compounds are shown in Figure 2. Other studies, in turn, identified the flavonoids present in the plant, including apigenin, hyperoside, quercetin, quercitrin, kaempferol, luteolin, myricetin, rutoside, and also lactone - artemisinin [33,37,40]. Among other compounds isolated from the herb of A. abrotanum were also coumarins, including isofraxidine, umbelliferone, scopoletin, herniarin, and esculetin [33,37,47,50,51]. Moreover, the herb has been found to contain the next sesquiterpenoid lactone—santonin [50], an alkaloid—abrotine [33,37], tannins, organic acids [44], sterols [52], and resin [33,37,53,54]. The chemical composition of the A. abrotanum plant is shown in Table 2.
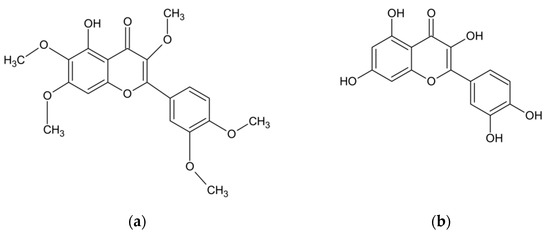
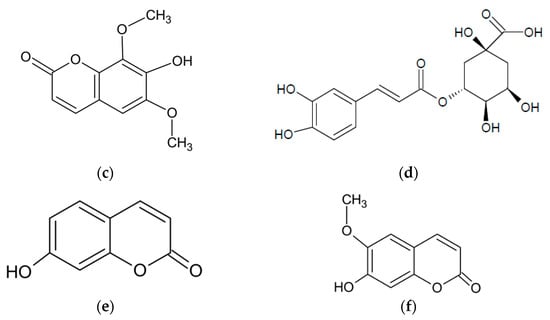
Figure 2.
Chemical structure of the most frequent compounds isolated from A. abrotanum: artemisetin (a); quercetin (b); isofraxidine (c); chlorogenic acid (d); umbelliferone (e), scopoletin (f).

Table 2.
Chemical composition of A. abrotanum.
5. Application in Traditional European and Non-European Medicine
In European traditional medicine, Abrotani herba is used in diseases of the liver, such as atony, contractile states of the bile ducts and stagnation of or insufficient bile secretion. Herbal infusions are used to improve digestion, increase appetite, and also as an astringent in diarrhea. It is also recommended to use infusions as an aid in cases of anorexia, flatulence, and hypoacidity [14,33,37,39]. Extracts from the plant have also been used as a remedy for frostbite, lymphadenitis, and in the treatment of epilepsy [37,39,43]. The species has been used in the treatment of malaria [54].The leaves have been used to improve liver function, stimulate menstruation, and relax smooth muscles. Other indications have included cancer prophylaxis and therapy, and men have used the plant as a hair growth agent [55]. The plant is also used as an anthelmintic agent in the treatment of oxyurosis and ascariasis in children [33]. In Bosnia and Herzegovina, the species has also been used in the treatment of jaundice [58], and in Turkey in the treatment of fever [59]. In traditional Indian medicine, Siddha, A. abrotanum is used as a repellent to repel insects and parasites [60].
6. Application in Modern Phytotherapy and Position in Official European Medicine
The herb A. abrotanum can be found primarily in homeopathic medicines. The French Pharmacopoeia states that their production is based on a 65% mother tincture, prepared from fresh herb of A. abrotanum. The tincture should contain at least 0.1% m/m of ortho-dihydroxycinnamic acid derivatives, expressed in terms of chlorogenic acid [61,62]. These preparations are indicated for the treatment of inflammation of the colon, rosacea, frostbite, and inflammation of the lymph nodes and mucous membranes [37,43]. Homeopathic medicines from A. abrotanum are also indicated for people suffering from depression and anxiety [63]. The European Medicines Agency (EMA) has also authorized the use of homeopathic preparations from A. abrotanum in farm animals used for food production [11].
Artemisiae abrotani herba and Artemisiae abrotani folium do not have the status of pharmacopoeial raw materials in European countries. The species and the raw materials obtained from it are, however, successfully used in official European allopathic medicine. A good example is the research conducted at Havelhoehe Research Institute and Charite’ University Medical Centre in Berlin, and the University of Witten/Herdecke in Herdecke (Germany). For a period of two years (2004–2005), it was recorded the prescribing by primary care doctors and specialist doctors of preparations containing plant extracts from plants of the family Asteraceae. Over that time, preparations with A. abrotanum extracts were recommended to 202 patients. The preparations included both homeopathic remedies and herbal mixtures as well as medicines containing only A. abrotanum extract. The indications for the use of these medications were usually diagnosed non-infectious inflammations of the intestines or of the large intestine [45,61].
In official European medicine similar as in traditional medicine the most often the whole plant extracts are used [11]. They contain the mixture of flavonoids, coumarins, phenolic acids and essential oils. These groups of metabolites with polyphenolic structures are important plant components with among others antioxidant and antitumor activities. For documented antibacterial and antifungal properties the components of essential oil are responsible. Synergic action might be created between these groups of polyphenols. In the phytotherapy as the galenic formulation the infusions and tinctures from A. abrotanum herb are recommended. Moreover, this raw material is endorsed for use in choleretic and gastric mixtures, as well as for strengthening and soothing baths. A. abrotanum is also the subject of patents in the field of pharmacy in Europe [43,64]. The mechanisms of action of A. abrotanum extracts and/or essential oil are summarized in Table 3.

Table 3.
Directions of biological activity of A. abrotanum confirmed by scientific research.
7. New Directions of Biological Activity of Extracts from the Herb and/or Leaves, and/or Essential Oil Confirmed by Scientific Research
7.1. Antibacterial and Antifungal Activity
The studies of the antibacterial and antifungal activities of ethanolic extracts from the herb of A. abrotanum were performed by Suresh et al. Using the disc diffusion method and the serial dilution method, the effect of the extracts on the bacteria: Bacillus subtilis, B. stearothermophilus, Micrococcus luteus, Klebsiella pneumoniae, Pseudomonas cepacia, Salmonella typhi, and the fungi: Candida albicans, Saccharomyces cerevisiae, Trichosporon beigelii, was tested. The control treatment was performed with 1 mg/mL penicillin for the bacteria and 500 μL/mL amphotericin B for the fungi. The extract proved to be effective against all the pathogens except B. subtillis. The maximum zone of growth inhibition among the bacteria was obtained for P. cepacia (28.6 mm) after using the A. abrotanum ethanolic extract at 30 mg/mL. The inhibition zone for the control was 31.4 mm. Among the fungi, significant activity was demonstrated with the use of 30 mg/mL extract for T. beigelii (17 mm) and S. cerevisiae (17 mm), compared with the control samples for which the zones of growth inhibition were 21 mm and 26 mm, respectively. It was also shown that the antimicrobial effect of the extract depended on its concentration [65].
Researchers from Romania have examined the activity of the essential oil obtained from the herb of A. abrotanum against a strain of Candida albicans. The disc diffusion method was also adopted as the test method. The measured zone of growth inhibition was 23.5 mm for the oil obtained by steam distillation and 21.5 mm for the oil obtained by extraction with dichloromethane. The growth inhibition zone for nystatin as the control was 20.5 mm. The results of the study indicate that A. abrotanum exhibits stronger antifungal activity than nystatin, which is a medication commonly used in the treatment of fungal infections [47].
Iranian researchers have also investigated the activity of the essential oil of A. abrotanum against pathogenic microorganisms. They tested the effect of the oil on the following bacteria: Staphylococcus aureus, Bacillus subtillis, Salmonella typhi, Escherichia coli, and the fungus Candida albicans. For this purpose, they used the method of diffusion in agar wells. The use of 15, 25 or 40 μL of the essential oil was shown to inhibit the growth of S. aureus, while the use of 10, 15, 25 or 40 μL of the essential oil inhibited the growth of E. coli and C. albicans. The authors of the study indicate that the antimicrobial effect of A. abrotanum can be attributed to the compounds contained in the plant, such as borneol, cymene, camphor, terpineol, 1,8-cineole, and aromadendrene [63].
Another study determined the activity of the essential oil isolated from the herb of A. abrotanum against the bacteria Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus and Proteus vulgaris, and against the fungi Candida albicans, Aspergillus flavus, and Fusarium oxysporum. The experiment was carried out using the disk diffusion method. With 25 μL of the essential oil, the zone of growth inhibition was 19.67 mm for E. coli, 10.33 mm for P. aeruginosa, 16.89 mm for S. aureus, and 18.89 mm for P. vulgaris. For the control with 10 μL/mL gentamicin, the zones of growth inhibition were 11 mm, 25 mm, 25 mm, and 23 mm, respectively. Among the tested fungi, the essential oil showed activity only against A. flavus (the growth inhibition zone was 11.89 mm after using 25 μL of a 75% solution of the essential oil diluted with Tween 40) [66].
In the another study, the action of the methanolic extract from the A. abrotanum plant and of the individual components of the extract were tested against the bacteria: Listeria monocytogenes, Staphylococcus aureus, Escherichia coli, Bacillus cereus, Pseudomonas aeruginosa and Micrococcus flavus, and against the fungi: Penicillium ochrochloron, P. funiculosum, Candida albicans, Aspergillus ochraceus, A. niger and A. flavus using the microdilution method. The extract showed good antibacterial properties and moderate antifungal properties against the pathogens tested. The action of quercitrin and isochlorogenic acid against the bacteria was shown to be as strong as that of streptomycin, which served as a positive control [55].
Ukrainian researchers have conducted a study evaluating antimicrobial activity of Artemisia L. herb extracts, including A. abrotanum extract. Therefore, the team has prepared ethanol extracts of A. abrotanum, A. vulgaris and A. absinthium with three concentration of the solvent: 40%, 70% and 90%. Micromethod of diffusion in agar has been employed in antimicrobial activity assessment. Moderate inhibition of the growth of bacteria: Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus gordonii, Enterococcus faecalis, Escherichia coli, Citrobacter freundii, Pseudomonas aeruginosa; methicilin suseptible: Staphylococcus aureus and Staphylococcus epidermis; methicillin resistant: Staphylococcus aureus and Staphylococcus haemolyticus and macrolides resistant: Propionibacterium acnes strains under influence of A. abrotanum herb ethanolic extract has been noted. The strongest bacteriostatic activity of the 70% ethanol extract has been displayed against Propionibacterium acnes MLS (8.71 mm. = inhibition zone) in comparison with the control (70% ethanol). The results also showed a decrement of the growth of Candida albicans and Candida tropicalis colonies as well as a significant inhibition of Aspergillus niger spore germination (13.32 mm.). Moreover, the study has also evaluated a synergistic action of A. abrotanum ethanol herb extract with erythromycin against Staphylococcus aureus with efflux mechanism of MLS-resistance. Only extract with 90% ethanol displayed a synergistic action with erythromycin. The results demonstrated inhibition zone 6.83 mm. with 1.95 µg/mL concentration of erythromycin and 7.48 mm. with 31.25 µg/mL of erythromycin. Of note, erythromycin alone didn’t exhibit any action against Staphylococcus aureus with efflux mechanism of MLS-resistance so did medium without erythromycin [12].
7.2. Antioxidant Effect
The antioxidant potential of an ethanolic extract from A. abrotanum herb was tested using the DPPH method. The results of the study showed moderate antioxidant activity of the plant extract. The IC50 value for the extract was 284.5 µg/mL, compared with the ascorbic acid control, for which the IC50 was 17.34 µg/mL [40].
Under another study the antioxidant activity of the essential oil isolated from the herb of A. abrotanum was tested. For this purpose, they determined the percentage inhibition of peroxidation of egg yolk lipids using the thiobarbituric acid method (TBARs). The reducing potential was investigated using the potassium hexacyanoferrate(III) method. In the TBARs test, 82.34% inhibition of lipid peroxidation was recorded after application of 1000 μL of the essential oil. The reducing potential of 100 μL of the oil was 135.97 μg of butylated hydroxyanisole equivalent [66].
The latest study conducted in 2020 has also confirmed the antioxidant activity of A. abrotanum. The effects of the methanolic extract from the leaves of A. abrotanum and of the individual components of the extract were determined using the β-carotene bleaching (BCB) method, the ability to reduce Fe(III) ions (FRAP test) and DPPH. The IC50 values recorded in the β-carotene bleaching test and the DPPH test were 35.4 μg/mL and 27.1 μg/mL, respectively, whereas for the butylated hydroxytoluene (BHT) control the IC50 was 2.7 μg/mL and 3.3 μg/mL, respectively. In the FRAP test, the IC50 was 39.1 μg/mL, whereas for the trolox control the IC50 was 3.2 μg/mL. The best results were obtained for rosmarinic acid (IC50 in the bleaching test was 3.1 μg/mL, 2.7 μg/mL in the DPPH test, and 3.5 μg/mL in the FRAP test) [55].
7.3. Antitumour Effect
The antitumour activity of the essential oil obtained from the herb of A. abrotanum was tested against the rhabdomyosarcoma (RD) cell line. The growth or inhibition of cancer cell proliferation was measured using the dye 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), which, when influenced by mitochondrial dehydrogenase present in living cells, changes colour from orange to dark blue. The viability of RD cells after the application of the essential oil at concentrations of 25, 50 and 100 μg/mL was 29.679%, 20.833%, and 20.256%, respectively. The negative control with DMSO and the positive control with methotrexate showed 32.179% and 18.205% survival of RD cells, respectively. The results of the study prove that the essential oil of A. abrotanum, especially in concentrations of 50 and 100 μg/mL, has an antitumour effect, probably due to the presence of compounds such as: borneol, cymene, camphor, terpineol, 1,8-cineole, and aromadendrene [63].
Under another study an ethanolic extract from the leaves of A. abrotanum and its individual components show antiproliferative activity against T-cell lymphoblastic leukemia cell lines (Jurkat line), breast adenocarcinoma (MCF-7 line), cervical adenocarcinoma (HeLa line), colorectal adenocarcinoma (HT-29 line), and embryonic human kidney cell lines (HEK-293 line). The MTT-mediated assay was used to determine the change in cell viability. The antiproliferative effect of the extract against all the lines, except for the HEK-293 line, was proven. Chlorogenic acid and isochlorogenic acid showed the greatest activity against these lines [55].
7.4. Allergy Symptom-Alleviating Effect
Swedish researchers have assessed the effect of a nasal spray containing the essential oil and flavonoids derived from A. abrotanum on the course of allergic rhinitis. The essential oil contained large amounts of 1,8-cineole, davanone and linalool, with centaureidine dimethylether, casticin and quercetin dominant among the flavonoids. The preparation was administered to 12 patients with known allergic rhinitis and/or bronchial obstruction, or with allergic rhinitis with coexisting allergic conjunctivitis or with exercise-induced asthma. The preparation was applied immediately after noticing the characteristic allergic symptoms of the nose. The assessment of the preparation’s effects was based on a questionnaire filled in by patients who subjectively assessed the symptoms. Five minutes after application of the spray, the patients reported alleviation of nasal symptoms similar to the use of an antihistamine medication or chromoglycan. The soothing effect lasted up to several hours. Seven patients with symptoms of allergic conjunctivitis also reported improvement in ocular symptoms, and three patients with obstructive bronchial disease reported improvement in bronchial symptoms. The results of the study indicate that a nasal spray with a mixture of essential oils and flavonoids present in A. abrotanum can be effective in the treatment of allergic rhinitis and in the treatment of accompanying symptoms [56].
7.5. Insect-Repelling Action
The toluene extract from the herb of A. abrotanum were tested whether they could be used as a repellent. The effect of the plant extract was tested against Ixodes ricinus (common tick, vector of e.g., Lyme disease and tick-borne encephalitis) and Aedes aegypti (Egyptian mosquito, vector of e.g., Dengue virus) [50].
The experiment with Ixodes ricinus consisted in placing in a cage with ticks tissue papers soaked with the toluene extract from the A. abrotanum herb or with individual components of this extract. The control sample was tissue soaked only with ethanol. Records were kept of the tendency of ticks to avoid surfaces with the applied extract [50].
The experiment with Aedes aegypti was carried out in the open field and in a laboratory. In the laboratory, female mosquitoes were kept in a mesh enclosure and fasted for 2–3 days; then the tester placed in it a hand with the previously applied extract of A. abrotanum suspended in ethanol or with individual components of this extract. The other hand of the tester, without the extract applied, served as the control. The number of mosquitoes that landed on the hands was counted over a specified time period. In the field experiment, the tester similarly exposed a hand with the extract or its ingredients applied to it [50].
After 4 and 8 h from the time of applying the ethanolic suspension of the toluene extract from the herb A. abrotanum, the recorded repellency rates were, respectively, 69.1% and 56.8% against Ixodes ricinus, and 100% and 86.7% against Aedes aegypti. For the control sample with DEET (N,N-dietylo-m-toluamid), the repellency rates after 4 and 8 h were, respectively, 83.4% and 94.4% against Ixodes ricinus, and 100% and 95.8% against Aedes aegypti. The components that showed the highest percentage of repellency against Ixodes ricinus were camphor, coumarin and thujyl alcohol, and against Aedes aegypti coumarin, chlorogenic acid and caffeic acid [50].
7.6. Action against Animal Parasites
The antiparasitic activity of an ethanolic extract from the leaves of A. abrotanum has been investigated in an animal model (mouse). Rodents infected with Hymenolepis nana (dwarf tapeworm) or with rodent pinworms (Syphacia obvelata and Aspiculuris tetraptera) were given the ethanolic extract from A. abrotanum leaves, dissolved in water, for five consecutive days. The administration of the extract was shown to significantly reduce the egg count of all three parasites in the faeces of the mice. On the seventh day of therapy, no eggs were detected in the faeces. The research results indicate that the traditional use of A. abrotanum extracts as an anti-parasitic agent has been justified [67].
7.7. Antimalarial Activity
The potential antimalarial properties of silver nanoparticles from two Artemisia species: A. abrotanum and A. arborescens were studied. In the experiment, the hemocompatibilty of the nanoparticles using a microplate reader to measure the absorbance of hemoglobin release in supernatant were evaluated. The test was carried out on parasitized (P. falciparum) red blood cells (pRBCs). The cells were incubated for 24 and 48 h at 37 and 41 °C and at concentrations of nanoparticles: 0.6 µg/mL; 1.25 µg/mL; 2.5 µg/mL; 5 µg/mL and 7.5 µg/mL. Hemolysis assay revealed a better hemo-biocompatibility of A. abrotanum silver nanoparticles (A. abrotanum—AgNPs) than A. arborescens silver nanoparticles (A. arborescens—AgNPs). Moreover, hemolytic activity increased in dose dependent manner. To evaluate parasite growth inhibition the team exposed pRBCs to the influence of different nanoparticles in concentrations ranging from 0.6 to 7.5 µg/mL for 24 and 48 h. Significant inhibition of the growth of the parasite in the presence of A. arborescens—AgNPs has been observed.
In the next in vitro test evaluating an influence of the nanoparticles on maturation and death of P. falciparum parasite the scientists used non hemolytic concentration (2.5 µg/mL). Different mechanism of action of the two Artemisia species was noted. A. abrotanum-AgNPs exhibited antiplasmodial activity leading to parasite death in comparison with the control after 24 and 48 h of treatment. Contrariwise, A. arborescens-AgNPs antiplasmodial effect was associated with the parasite maturation stage blockage from trophozoite to ring stage. Futhermore, Avitabile et. al. measured IC50 of different silver nanoparticles. The results demonstrated that A. abrotanum-AgNPs displayed better antiplasmodial effect considering 50%, 90% and 99% inhibition concentrations [68].
The properties described above are summarized in Table 3.
8. Application in Cosmetology and in the Food Industry
A. abrotanum, like other Artemisia species, is also used in the production of cosmetics.The European CosIng database advises that A. abrotanum herb, leaf and stem extracts can be used as a protective, moisturizing, and caring agent for use on the skin [14]. Such extracts can be found in face and hand creams, serums, body lotions, scrubs, milks, and pedicure masks. Products containing A. abrotanum are offered by Polish companies such as Organic life, Perfecta, German: Dr Hauschka, English: Bulldog Natural, Lush, American: Jack Black, Physicians Formula, Aveeno, and French: Decléor [14]. Interestingly, according to a pilot study from 2021 A. abrotanum leaf extract offers promising results as an ingredient of nail gel used in patients with nail plate surface abnormalities [69]. Moreover, because of antibacterial activity against macrolides resistant Propionibacterium acnes strain, A. abrotanum extract has a potential as an active compound in cosmetics for acne-prone skin [12].
The leaves of A. abrotanum, due to their pleasant aroma, are used to flavour meats, salads, and cottage cheese. As flavourings, they are sometimes added to confectionery as well as to alcoholic beverages such as vermouths and liqueurs [13,33,41]. The herb of A. abrotanum can be found as an ingredient in teas [41].
9. Safety of Use
At Havelhoehe Research Institute and Charite’ University Medical Centre in Berlin, and the University of Witten/Herdecke in Herdecke (Germany), adverse effects associated with the use of preparations based on plants from the Asteraceae family had been documented for two years. Of the 236 patients taking homeopathic remedies, herbal mixtures, or single-ingredient preparations from A. abrotanum extracts, only two were found to produce side effects. The reported ailments, after taking a preparation composed of extracts of A. abrotanum and Matricaria recutita, included stomach pain and allergy. However, no serious side effects were reported [61].
10. Biotechnological Research
A review of the scientific literature has shown that A. abrotanum is a species that so far has sporadically been the object of research in the field of plant biotechnology. A protocol for the micro-propagation of this species has been only developed. Young leaves of the plant were sterilized and placed in the MS (Murashige and Skoog) medium supplemented with 4.44 µM 6-benzyladenine (BA) and 0.54 or 0.81 µM naphthyl-1-acetic acid (NAA). Abundant callus growth and shoot formation were achieved. With the addition of 4.44 μM BA and 0.54 μM of NAA, 3.61 shoots per explant were obtained (31 shoots in total), and with the addition of 4.44 μM BA and 0.81 μM NAA, 4.05 shoots per explant were obtained (total of 38 shoots). The obtained shoots were rooted using the MS medium without the growth regulators or supplemented with 0.49 µM indole acetic acid (IAA) or 0.54 µM NAA. Afterwards, the plants were transplanted into soil-filled pots [70].
11. Conclusions
Artemisia abrotanum is a species with an important position in the history of European, Middle-Eastern and Asian medicine (including especially Traditional Chinese Medicine (TCM)). Currently, it also has an important position in traditional European and Asian medicine (especially in the Middle East). The French Pharmacopoeia recommends the plant for use in official homeopathic medicine. In many European and Middle Eastern countries, this species is traditionally used in allopathic medicine. Recent phytochemical studies of this species have proven the variability of the chemical composition of its essential oil. Pharmacological studies, in turn, have provided evidence of new, previously unknown directions of biological activity of extracts from the herb and/or leaves, and/or essential oil of the plant—antimicrobial, antioxidant, anticancer, and antiallergic effects. Some studies also indicate the possibility of using the plant in the prevention of diseases spread by insects, due to the repellent properties of the essential oil. This creates prospects for the medicinal use of the plant to an even greater extent. This species is also invariably used in the food industry as a spice and as a flavouring additive to alcoholic beverages and teas. A novelty is the possibility of using extracts from various parts of the plant in the production of cosmetics. The species is approved by the European CosIng database to be used for this purpose. In the light of contemporary research, A. abrotanum appears to be an attractive species with valuable medicinal, culinary, and cosmetic qualities.
Author Contributions
Data collection: H.E., A.S., J.Ś., P.K., A.R., E.K.; design of the study: H.E.; analysis and interpretation of the data: A.S., H.E., E.K., P.K., J.Ś., A.R., M.T.; drafting the manuscript: H.E., A.S., P.K., A.R.; critical revision of the manuscript: H.E., A.S., M.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by grand numbers: N42/DBS/000010 and N42/DBS/000136 supported by the Polish Ministry of Science and Higher Education.
Conflicts of Interest
The authors declare no conflict of interest.
- Efferth, T.; Zacchino, S.; Georgiev, M.I.; Liu, L.; Wagner, H.; Panossian, A. Nobel Prize for artemisinin brings phytotherapy into the spotlight. Phytomedicine 2015, 22, A1–A3. [Google Scholar] [CrossRef]
- Su, X.Z.; Miller, L.H. The discovery of artemisinin and the Nobel Prize in Physiology or Medicine. Sci. China Life Sci. 2015, 58, 1175–1179. [Google Scholar] [CrossRef]
- Pellicer, J.; Saslis-Lagoudakis, C.H.; Carrió, E.; Ernst, M.; Garnatje, T.; Grace, O.M.; Gras, A.; Mumbrú, M.; Vallès, J.; Vitales, D.; et al. A phylogenetic road map to antimalarial Artemisia species. J. Ethnopharmacol. 2018, 225, 1–9. [Google Scholar] [CrossRef]
- Garcia, L.C. A Review of Artemisia annua L.: Its genetics, biochemical characteristics, and anti-malarial efficacy. Int. J. Sci. Technol. 2015, 5, 38–46. [Google Scholar]
- Abad, M.J.; Bedoya, L.M.; Apaza, L.; Bermejo, P. The Artemisia L. genus: A review of bioactive essential oils. Molecules 2012, 17, 2542–2566. [Google Scholar] [CrossRef] [PubMed]
- Ekiert, H.; Świątkowska, J.; Klin, P.; Rzepiela, A.; Szopa, A. Artemisia annua—Importance in traditional medicine and current state of knowledge on the chemistry, biological activity and possible applications. Planta Med. 2021. [Google Scholar] [CrossRef]
- Numonov, S.; Sharopov, F.; Salimov, A.; Sukhrobov, P.; Atolikshoeva, S.; Safarzoda, R.; Habasi, M.; Aisa, H. Assessment of artemisinin contents in selected Artemisia species from Tajikistan (Central Asia). Medicines 2019, 6, 23. [Google Scholar] [CrossRef]
- Melzig, M.F. Artemisia abrotanum L, Eberraute. Z. Phyther. 2019, 40, 283–288. [Google Scholar] [CrossRef]
- Saunoriute, S.; Ragažinskiene, O.; Ivanauskas, L.; Marksa, M. Essential oil composition of Artemisia abrotanum L. during different vegetation stages in Lithuania. Chemija 2020, 31, 52–56. [Google Scholar] [CrossRef]
- Svanberg, I.; De Vahl, E. “It may also have prevented churchgoers from falling asleep”: Southernwood, Artemisia abrotanum L. (fam. Asteraceae), in the church bouquet, and its contemporary presence as a heritage plant in Sweden. J. Ethnobiol. Ethnomed. 2020, 16, 1–12. [Google Scholar] [CrossRef]
- European Medicines Agency Committee for Veterinary Medicinal Products. Artemisia Abrotanum Summary Report; European Medicines Agency: London, UK, 1999. [Google Scholar]
- Hrytsyk, R.A.; Kutsyk, R.V.; Yurchyshyn, O.I.; Struk, O.A.; Kireev, I.V.; Grytsyk, A.R. The investigation of antimicrobial and antifungal activity of some Artemisia L. species. Pharmacia 2021, 68, 93–100. [Google Scholar] [CrossRef]
- Trendafilova, A.; Moujir, L.M.; Sousa, P.M.C.; Seca, A.M.L. Research advances on health effects of edible Artemisia species and some sesquiterpene lactones constituents. Foods 2020, 10, 65. [Google Scholar] [CrossRef] [PubMed]
- European Commission Cosing CosIng—Cosmetic Database. Available online: https://ec.europa.eu/growth/tools-databases/cosing/ (accessed on 4 April 2020).
- Berendes, J. Des Pedanios Dioskurides aus Anazarbos Arzneimittellehre in fünf Büchern. Übersetzt und mit Erklärungen Versehen; Band III.; Ferdinand Enke: Stuttgart, Germany, 1902. [Google Scholar]
- Plinius, S.G. Naturalis Historia (Naturgeschichte); Metzler/Metzler-Verlag: Stuttgart, Germany, 1855; Volume XXI. [Google Scholar]
- Hegi, G. Illustrierte Flora von Mitteleuropa; Verlag, J.F.L., Ed.; Wiley: München, Germany, 1931; Volume VI. [Google Scholar]
- Krassnig, K. TCM mit westlichen Pflanzen. Dtsch. Z. Akupunkt. 2012, 55, 52. [Google Scholar] [CrossRef]
- Avicenna. Kanon der Medizin. Übersetzung und Bearbeitung Durch Gerhard von Cremona, Arnaldus de Villanova und Andrea Alpago (1450–1521); Abrotanum: Basel, Switzerland, 1556. [Google Scholar]
- Romańska, K. Etnofarmacja i etnomedycyna na łamach czasopisma “Lud” w latach 1946–1986. In Historia Leków Naturalnych; Kuźnicka, B., Ed.; Polska Akademia Nauk: Warsaw, Poland, 1999; Volume 5, p. 145. [Google Scholar]
- Madaus, G. Lehrbuch der Biologischen Heilmittel; Band, I., Ed.; Georg Olms Verlag: Hildesheim, NY, USA, 1976. [Google Scholar]
- Reier, H. Hildegard von Bingen Physica. Nach der Textausgabe; von J. P. Migne, Paris 1882 ins Deutsche übersetzt, Herbert Reier: Kiel, Germany, 1980. [Google Scholar]
- Brunfels, O. Ander Teyl des Teütschen Contrafayten Kreüterbůchs; Hans Schott: Strasburg, Germany, 1537. [Google Scholar]
- Bock, H. New Kreütter Bůch.; Wendel Rihel: Strasburg, Germany, 1539. [Google Scholar]
- Fuchs, L. New Kreütterbuch; Michael Isingrin: Basel, Switzerland, 1543. [Google Scholar]
- Tabernaemontanus. Neuw Kreuterbuch; Nicolaus Basseus: Franckfurt am Mayn, Germany, 1588. [Google Scholar]
- Lémery, N. Dictionnaire Universel Des Drogues Simples; Christoph Friedrich Richtern: Paris, France, 1699. [Google Scholar]
- Syrennivs, S. Zielnik Herbarzem z Języka Łacinskiego Zowią; Drukarnia Bazylego Skalskiego: Kraków, Poland, 1613. [Google Scholar]
- Arabas, I. Leki roślinne w “Dziełach Wszystkich” Oskara Kolberga. In Historia leków Naturalnych; Kuźnicka, B., Ed.; Polska Akademia Nauk: Warsaw, Poland, 1986; Volume 1, pp. 91–161. [Google Scholar]
- The Plant List. Available online: http://www.theplantlist.org/ (accessed on 12 March 2020).
- Plants of the Word online. Kew science. 2020. Available online: http://www.plantsoftheworldonline.org/taxon/urn:lsid:ipni.org:names:306365-2 (accessed on 13 March 2021).
- GBIF.org. GBIF Home Page. 2020. Available online: https://www.gbif.org (accessed on 12 March 2020).
- Rumińska, A.; Ożarowski, A. Leksykon Roślin Leczniczych; Państwowe Wydawnictwo Rolnicze i Leśne: Warszawa, Poland, 1990. [Google Scholar]
- GRIN. Available online: https://www.ars-grin.gov/ (accessed on 2 June 2020).
- Missouri Botanical Garden. Available online: https://www.tropicos.org/home (accessed on 15 June 2020).
- The Herb Society of America. Artemisia: An Essential Guide. Available online: https://www.herbsociety.org/hsa-learn/hsa-publications/hsa-essential-guides.html#item7738862 (accessed on 4 April 2020).
- Ożarowski, A.; Jaroniewski, W. Rośliny Lecznicze i ich Praktyczne Zastosowanie; Panacea: Warsaw, Poland, 1987. [Google Scholar]
- Watson, L.E.; Bates, P.L.; Evans, T.M.; Unwin, M.M.; Estes, J.R. Molecular phylogeny of Subtribe Artemisiinae (Asteraceae), including Artemisia and its allied and segregate genera. BMC Evol. Biol. 2002, 2, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Suresh, J.; Elango, K.; Dhanabal, S.P.; Paramakrishnan, N.; Suresh, B. A comparative pharmacognostical evaluation of two Artemisia species found in Nilgiris biosphere. Anc. Sci. Life 2007, 27, 7–13. [Google Scholar] [PubMed]
- Baiceanu, E.; Vlase, L.; Baiceanu, A.; Nanes, M.; Rusu, D.; Crisan, G. New polyphenols identified in Artemisiae abrotani herba extract. Molecules 2015, 20, 11063–11075. [Google Scholar] [CrossRef]
- Vaughan, J.; Geissler, C. The New Oxford Book of Food Plants, 2nd ed.; Oxford University Press: Oxford, UK, 2009; ISBN 0191609498. [Google Scholar]
- Podbielkowski, Z.; Sudnik-Wójcikowska, B. Słownik Roślin Użytkowych; Wydanie, V.I., Ed.; Państwowe Wydawnictwo Rolnicze i Leśne: Warsaw, Poland, 2003. [Google Scholar]
- Volak, J.; Stodola, J.; Severa, F. Rośliny Lecznicze; Państwowe Wydawnictwo Rolnicze i Leśne: Warsaw, Poland, 1987. [Google Scholar]
- Pino, J.A.; Marbot, R.; Martí, M.P. Leaf oil of Artemisia abrotanum L. grown in Cuba. J. Essent. Oil Res. 2011, 23, 119–120. [Google Scholar] [CrossRef]
- Muangphrom, P.; Misaki, M.; Suzuki, M.; Shimomura, M.; Suzuki, H.; Seki, H.; Muranaka, T. Identification and characterization of (+)-α-bisabolol and 7-epi-silphiperfol-5-ene synthases from Artemisia abrotanum. Phytochemistry 2019, 164, 144–153. [Google Scholar] [CrossRef]
- Khalid, K.A.; El-Gohary, A.E. Productivity of wormwood (Artemisia abrotanum) enhanced by trace elements. Bull. Natl. Res. Cent. 2020, 44, 1–10. [Google Scholar] [CrossRef]
- Obistioiu, D.; Cristina, R.T.; Schmerold, I.; Chizzola, R.; Stolze, K.; Nichita, I.; Chiurciu, V. Chemical characterization by GC-MS and in vitro activity against Candida albicans of volatile fractions prepared from Artemisia dracunculus, Artemisia abrotanum, Artemisia absinthium and Artemisia vulgaris. Chem. Cent. J. 2014, 8, 6. [Google Scholar] [CrossRef]
- Aruba, O.S.; Jasim, G.A.; Nasser, A.A. Detection of terpenes of Iraqi Artemisia abrotanum L. by GC/MS in hexane extract. Al Mustansiriyah J. Pharm. Sci. 2019, 19, 239–248. [Google Scholar]
- Khodakov, G.V.; Kotikov, I.V.; Pankovetskii, V.N. Component composition of essential oil from Artemisia abrotanum and A. dracunculus. Chem. Nat. Compd. 2009, 45, 755–758. [Google Scholar] [CrossRef]
- Tunón, H.; Thorsell, W.; Mikiver, A.; Malander, I. Arthropod repellency, especially tick (Ixodes ricinus), exerted by extract from Artemisia abrotanum and essential oil from flowers of Dianthus caryophyllum. Fitoterapia 2006, 77, 257–261. [Google Scholar] [CrossRef]
- Bergendorff, O.; Sterner, O. Spasmolytic flavonols from Artemisia abrotanum. Planta Med. 1995, 61, 370–371. [Google Scholar] [CrossRef] [PubMed]
- Serkerov, S.V.; Ibragimova, S.I. New steroidal compound from Artemisia abrotanum. Chem. Nat. Compd. 2014, 50, 478–479. [Google Scholar] [CrossRef]
- Bora, K.S.; Sharma, A. The genus Artemisia: A comprehensive review. Pharm. Biol. 2011, 49, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Suresh, J.; Ahuja, J.; Paramakrishnan, N.; Sebastian, M. Total phenolic and total flavonoids content of aerial parts of Artemisia abrotanum Linn. and A. pallens Wall. Anal. Chem. Lett. 2012, 2, 186–191. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; El-Ansary, D.O.; Al-Mana, F.A.; Mahmoud, E.A. Polyphenol content and biological activities of Ruta graveolens L. and Artemisia abrotanum L. in Northern Saudi Arabia. Processes 2020, 8, 531. [Google Scholar] [CrossRef]
- Remberg, P.; Björk, L.; Hedner, T.; Sterner, O. Characteristics, clinical effect profile and tolerability of a nasal spray preparation of Artemisia abrotanum L. for allergic rhinitis. Phytomedicine 2004, 11, 36–42. [Google Scholar] [CrossRef]
- Ur Rashid, M.; Alamzeb, M.; Ali, S.; Ullah, Z.; Shah, Z.A.; Naz, I.; Khan, M.R. The chemistry and pharmacology of alkaloids and allied nitrogen compounds from Artemisia species: A review. Phyther. Res. 2019, 33, 2661–2684. [Google Scholar] [CrossRef]
- Tewari, D.; Mocan, A.; Parvanov, E.D.; Sah, A.N.; Nabavi, S.M.; Huminiecki, L.; Ma, Z.F.; Lee, Y.Y.; Horbanczuk, J.O.; Atanasov, A.G. Ethnopharmacological approaches for therapy of jaundice: Part I. Front. Pharmacol. 2017, 8, 1–18. [Google Scholar] [CrossRef]
- Willcox, M. Artemisia species: From traditional medicines to modern antimalarials-and back again. J. Altern. Complement. Med. 2009, 15, 101–109. [Google Scholar] [CrossRef]
- Quattrocchi, U. CRC World Dictionary of Medicinal and Poisonous Plants: Common Names, Scientific Names, Eponyms, Synonyms, and Etymology; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Jeschke, E.; Ostermann, T.; Lüke, C.; Tabali, M.; Kröz, M.; Bockelbrink, A.; Witt, C.M.; Willich, S.N.; Matthes, H. Remedies containing Asteraceae extracts. Drug Saf. 2009, 32, 691–706. [Google Scholar] [CrossRef]
- Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM) Southernwood for Homoeopathic Preparations. Available online: https://ansm.sante.fr/var/ansm_site/storage/original/application/fba9a2c564da57f105b5f4327037aa07.pdf (accessed on 15 March 2020).
- Almahdawy, S.S.; Said, A.M.; Abbas, I.S.; Dawood, A.H. The evaluation of antimicrobial and cytotoxic activity of the essential oil extracted from the aerial parts of southernwood herb (Artemisia abrotanum L.) that recently grown in Iraq. Asian J. Pharm. Clin. Res. 2017, 10, 384–387. [Google Scholar] [CrossRef]
- Podbielkowski, Z. Słownik Roślin Użytkowych; Państwowe Wydawnictwo Rolnicze i Leśne: Warsaw, Poland, 1980. [Google Scholar]
- Suresh, J.; Vasavi Reddy, A.; Rajan, D.; Ihsanullah, M.; Nayeemmullah Khan, M. Antimicrobial activity of Artemisia abrotanum and Artemisia pallens. Int. J. Pharmacogn. Phytochem. Res. 2011, 3, 18–21. [Google Scholar]
- Al-Zubairi, A.S.; Al-Mamary, M.A.; Al-Ghasani, E. The Antibacterial, Antifungal, and Antioxidant Activities of Essential Oil from Different Aromatic Plants. Glob. Adv. Res. J. Med. Med. Sci. 2017, 6, 224–233. [Google Scholar]
- Amirmohammadi, M.; Khajoenia, S.; Bahmani, M.; Rafieian-Kopaei, M.; Eftekhari, Z.; Qorbani, M. In vivo evaluation of antiparasitic effects of Artemisia abrotanum and Salvia officinalis extracts on Syphacia obvelata, Aspiculoris tetrapetra and Hymenolepis nana parasites. Asian Pac. J. Trop. Dis. 2014, 4, 5–9. [Google Scholar] [CrossRef]
- Avitabile, E.; Senes, N.; D’Avino, C.; Tsamesidis, I.; Pinna, A.; Medici, S.; Pantaleo, A. The potential antimalarial efficacy of hemocompatible silver nanoparticles from Artemisia species against P. falciparum parasite. PLoS ONE 2020, 15, e0238532. [Google Scholar] [CrossRef] [PubMed]
- Varothai, S.; Bunyaratavej, S.; Leeyaphan, C.; Phaitoonwattanakij, S.; Winayanuwattikun, W. Pilot study of the efficacy and safety of nail gel containing Artemisia abrotanum extract and glycerin in the treatment of nail plate surface abnormality. Siriraj Med. J. 2021, 73. [Google Scholar] [CrossRef]
- Bolyard, M. In vitro regeneration of Artemisia abrotanum L. by means of somatic organogenesis. In-Vitro Cell. Dev. Biol. 2018, 54, 127–130. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).