Prebiotic Potential of Oligosaccharides Obtained by Acid Hydrolysis of α-(1→3)-Glucan from Laetiporus sulphureus: A Pilot Study
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Materials
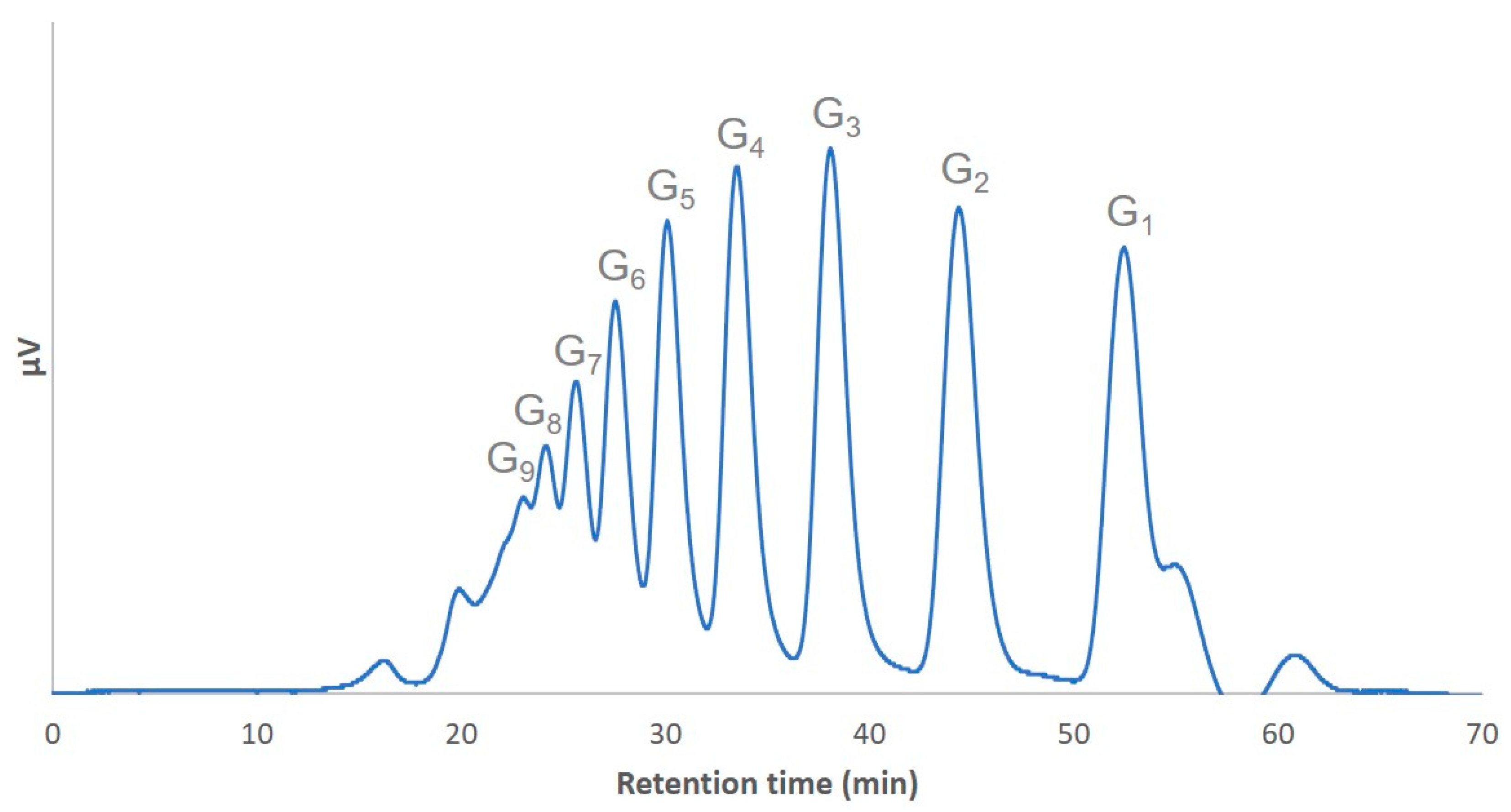
3.2. Preparation and Analysis of the α-(1→3)-Glucooligosaccharide Preparation
3.3. In Vitro Effects of α-(1→3)-GOS on Bacterial Growth
3.4. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Van den Abbeele, P.; Verstraete, W.; El Aidy, S.; Geirnaert, A.; Van de Wiele, T. Prebiotics, faecal transplants and microbial network units to stimulate biodiversity of the human gut microbiome. Microb. Biotechnol. 2013, 6, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Carding, S.; Verbeke, K.; Vipond, D.T.; Corfe, B.M.; Owen, L.J. Dysbiosis of the gut microbiota in disease. Microb. Ecol. Health Dis. 2015, 26, 26191. [Google Scholar] [CrossRef] [PubMed]
- Delzenne, N.M.; Neyrinck, A.M.; Cani, P.D. Gut microbiota and metabolic disorders: How prebiotic can work? Br. J. Nutr. 2013, 109, S81–S85. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.-Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J.; Michel, C. How to manipulate the microbiota: Prebiotics. In Microbiota of the Human Body; Schwiertz, A., Ed.; Springer International Publishing: Cham, Germany, 2016; pp. 143–155. [Google Scholar]
- Fernández, J.; Redondo-Blanco, S.; Gutierrez-del-Rio, I.; Miguelez, E.M.; Villar, C.J.; Lombo, F. Colon microbiota fermentation of dietary prebiotics towards short-chain fatty acids and their roles as anti-inflammatory and antitumour agents: A review. J. Funct. Foods 2016, 25, 511–522. [Google Scholar] [CrossRef]
- Moreno, F.J.; Corzo, N.; Montilla, A.; Villamiel, M.; Olano, A. Current state and latest advances in the concept, production and functionality of prebiotic oligosaccharides. Curr. Opin. Food Sci. 2017, 13, 50–55. [Google Scholar] [CrossRef]
- Villamiel, M.; Montilla, A.; Olano, A.; Corzo, N. Production and bioactivity of oligosaccharides derived from lactose. In Food Oligosaccharides: Production, Analysis and Bioactivity; Moreno, J., Sanz, M.L., Eds.; John Wiley & Sons Ltd.: Chichester, West Sussex, UK, 2014; pp. 137–167. [Google Scholar]
- Mano, M.C.R.; Neri-Numa, I.A.; da Silva, J.B.; Paulino, B.N.; Pessoa, M.G.; Pastore, G.M. Oligosaccharide biotechnology: An approach of prebiotic revolution on the industry. Appl. Microbiol. Biotechnol. 2018, 102, 17–37. [Google Scholar] [CrossRef]
- Tymczyszyn, E.E.; Santos, M.I.; Costa, M.D.C.; Illanes, A.; Gómez-Zavaglia, A. History, synthesis, properties, applications and regulatory issues of prebiotic oligosaccharides. In Carbohydrates Applications in Medicine; Gil, M.H., Ed.; Research Signpost: Kerala, India, 2014; pp. 127–154. [Google Scholar]
- Singh, S.P.; Jadaun, J.S.; Narnoliya, L.K.; Pandey, A. Prebiotic oligosaccharides: Special focus on fructooligosaccharides, its biosynthesis and bioactivity. Appl. Biochem. Biotechnol. 2017, 183, 613–635. [Google Scholar] [CrossRef]
- Barile, D.; Rastall, R.A. Human milk and related oligosaccharides as prebiotics. Curr. Opin. Biotechnol. 2013, 24, 214–219. [Google Scholar] [CrossRef]
- Seifert, A.; Freilich, S.; Kashi, Y.; Livney, Y.D. Protein-oligosaccharide conjugates as novel prebiotics. Polym. Adv. Technol. 2019, 30, 2577–2585. [Google Scholar] [CrossRef]
- Czerwonka, A.; Wiater, A.; Komaniecka, I.; Adamczyk, P.; Rzeski, W.; Pleszczyńska, M. Antitumour effect of glucooligosaccharides obtained via hydrolysis of α-(1→3)-glucan from Fomitopsis betulina. Mol. Biol. Rep. 2019, 46, 5977–5982. [Google Scholar] [CrossRef] [PubMed]
- Mussatto, S.I.; Mancilha, I.M. Non-digestible oligosaccharides: A review. Carbohydr. Polym. 2007, 68, 587–597. [Google Scholar] [CrossRef]
- Grün, C.H. Structure and biosynthesis of fungal α-glucans. Ph.D. Thesis, Utrecht University, Utrecht, The Netherlands, 30 January 2003. [Google Scholar]
- Synytsya, A.; Míčková, K.; Synytsya, A.; Jablonský, I.; Spěváček, J.; Erban, V.; Kováříková, E.; Čopíková, J. Glucans from fruit bodies of cultivated mushrooms Pleurotus ostreatus and Pleurotus eryngii: Structure and potential prebiotic activity. Carbohydr. Polym. 2009, 76, 548–556. [Google Scholar] [CrossRef]
- Wiater, A.; Paduch, R.; Choma, A.; Pleszczyńska, M.; Siwulski, M.; Dominik, J.; Janusz, G.; Tomczyk, M.; Szczodrak, J. Biological study on carboxymethylated (1→3)-α-d-glucans from fruiting bodies of Ganoderma lucidum. Int. J. Biol. Macromol. 2012, 51, 1014–1023. [Google Scholar] [CrossRef]
- Zhang, P.; Cheung, P.C. Evaluation of sulfated Lentinus edodes α-(1→3)-d-glucan as a potential antitumor agent. Biosci. Biotechnol. Biochem. 2002, 66, 1052–1056. [Google Scholar] [CrossRef]
- Huang, Q.; Zhang, L. Preparation, chain conformation and anti-tumor activities of water-soluble phosphated (1→3)-α-d-glucan from Poria cocos mycelia. Carbohydr. Polym. 2011, 83, 1363–1369. [Google Scholar] [CrossRef]
- Shimamura, A.; Uezono, Y.; Tsumori, H.; Mukasa, H. Assignment of 13C NMR signals for reduced nigerooligosaccharides prepared by partial acid hydrolysis of (1→3)-α-d-glucan. Carbohydr. Res. 1992, 233, 237–243. [Google Scholar] [CrossRef]
- Murosaki, S.; Muroyama, K.; Yamamoto, Y.; Kusaka, H.; Liu, T.; Yoshikai, Y. Immunopotentiating activity of nigerooligosaccharides for the T helper 1-like immune response in mice. Biosci. Biotechnol. Biochem. 1999, 63, 373–378. [Google Scholar] [CrossRef]
- Stacey, M.; Webber, J.M. Nigerose (3-O-α-d-glucopyranosyl-d-glucose). Partial hydrolysis of nigeran. In Methods in Carbohydrate Chemistry; Whistler, R.L., Wolfrom, M.L., Eds.; Academic Press Inc.: New York, NY, USA, 1962; pp. 339–341. [Google Scholar]
- Goffin, D.; Delzenne, N.; Blecker, C.; Hanon, E.; Deroanne, C.; Paquot, M. Will isomalto-oligosaccharides, a well-established functional food in Asia, break through the European and American market? The status of knowledge on these prebiotics. Crit. Rev. Food Sci. Nutr. 2011, 51, 394–409. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Prebiotics: Concept, definition, criteria, methodologies, and products. In Handbook of Prebiotics; Gibson, G.R., Roberfroid, M.B., Eds.; CRC Press: Boca Raton, FL, USA; London, UK; New York, NY, USA, 2008; pp. 57–86. [Google Scholar]
- Das, D.; Baruah, R.; Goyal, A. A food additive with prebiotic properties of an α-D-glucan from Lactobacillus plantarum DM5. Int. J. Biol. Macromol. 2014, 69, 20–26. [Google Scholar] [CrossRef]
- Champagne, C.P.; Raymond, Y.; Pouliot, Y.; Gauthier, S.F.; Lessard, M. Effect of bovine colostrum, cheese whey, and spray-dried porcine plasma on the in vitro growth of probiotic bacteria and Escherichia coli. Can. J. Microbiol. 2014, 60, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Polish Collection of Microorganisms. Available online: https://pcm.org.pl/search/E.%20faecalis%20PCM%20896 (accessed on 10 September 2020).
- Konishi, Y.; Shindo, K. Production of nigerose, nigerosyl glucose, and nigerosyl maltose by Acremonium sp. S4G13. Biosci. Biotechnol. Biochem. 1997, 61, 439–442. [Google Scholar] [CrossRef] [PubMed]
- Nakakuki, T. Present status and future prospects of functional oligosaccharide development in Japan. J. Appl. Glycosci. 2005, 52, 267–271. [Google Scholar] [CrossRef]
- Sanz, M.L.; Côté, G.L.; Gibson, G.R.; Rastall, R.A. Prebiotic properties of alternansucrase maltose-acceptor oligosaccharides. J. Agric. Food Chem. 2005, 53, 5911–5916. [Google Scholar] [CrossRef] [PubMed]
- Murosaki, S. Effects of intake of nigerooligosaccharides-supplemented syrup on the immune function and quality of life in healthy young adult subjects. Jpn. Pharmacol. Ther. 2002, 30, 81–90. [Google Scholar]
- Murosaki, S.; Muroyama, K.; Yamamoto, Y.; Liu, T.; Yoshikai, Y. Nigerooligosaccharides augment natural killer activity of hepatic mononuclear cells in mice. Int. Immunopharmacol. 2002, 2, 151–159. [Google Scholar] [CrossRef]
- Hirose, Y.; Murosaki, S.; Yamamoto, Y.; Ikematsu, H.; Nomoto, K. Nigerooligosaccharides augment mitogen-induced proliferation and suppresses activation-induced apoptosis of human peripheral blood mononuclear cells. Immunopharmacol. Immunotoxicol. 2004, 26, 387–399. [Google Scholar] [CrossRef]
- Hino, K.; Kurose, M.; Sakurai, T.; Inoue, S.I.; Oku, K.; Chaen, H.; Kohno, K.; Fukuda, S. Effect of dietary cyclic nigerosylnigerose on intestinal immune functions in mice. Biosci. Biotechnol. Biochem. 2006, 70, 2481–2487. [Google Scholar] [CrossRef]
- Wiater, A.; Paduch, R.; Pleszczyńska, M.; Próchniak, K.; Choma, A.; Kandefer-Szerszeń, M.; Szczodrak, J. α-(1→3)-D-glucans from fruiting bodies of selected macromycetes fungi and the biological activity of their carboxymethylated products. Biotechnol. Lett. 2011, 33, 787–795. [Google Scholar] [CrossRef]
- Hoeflinger, J.L.; Hoeflinger, D.E.; Miller, M.J. A dynamic regression analysis tool for quantitative assessment of bacterial growth written in Python. J. Microbiol. Methods 2017, 132, 83–85. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds α-(1→3)-d-glucan and α-(1→3)-oligosaccharides are available from the authors. |


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiater, A.; Waśko, A.; Adamczyk, P.; Gustaw, K.; Pleszczyńska, M.; Wlizło, K.; Skowronek, M.; Tomczyk, M.; Szczodrak, J. Prebiotic Potential of Oligosaccharides Obtained by Acid Hydrolysis of α-(1→3)-Glucan from Laetiporus sulphureus: A Pilot Study. Molecules 2020, 25, 5542. https://doi.org/10.3390/molecules25235542
Wiater A, Waśko A, Adamczyk P, Gustaw K, Pleszczyńska M, Wlizło K, Skowronek M, Tomczyk M, Szczodrak J. Prebiotic Potential of Oligosaccharides Obtained by Acid Hydrolysis of α-(1→3)-Glucan from Laetiporus sulphureus: A Pilot Study. Molecules. 2020; 25(23):5542. https://doi.org/10.3390/molecules25235542
Chicago/Turabian StyleWiater, Adrian, Adam Waśko, Paulina Adamczyk, Klaudia Gustaw, Małgorzata Pleszczyńska, Kamila Wlizło, Marcin Skowronek, Michał Tomczyk, and Janusz Szczodrak. 2020. "Prebiotic Potential of Oligosaccharides Obtained by Acid Hydrolysis of α-(1→3)-Glucan from Laetiporus sulphureus: A Pilot Study" Molecules 25, no. 23: 5542. https://doi.org/10.3390/molecules25235542
APA StyleWiater, A., Waśko, A., Adamczyk, P., Gustaw, K., Pleszczyńska, M., Wlizło, K., Skowronek, M., Tomczyk, M., & Szczodrak, J. (2020). Prebiotic Potential of Oligosaccharides Obtained by Acid Hydrolysis of α-(1→3)-Glucan from Laetiporus sulphureus: A Pilot Study. Molecules, 25(23), 5542. https://doi.org/10.3390/molecules25235542







