One-Step Extraction of Olive Phenols from Aqueous Solution Using β-Cyclodextrin in the Solid State, a Simple Eco-Friendly Method Providing Photochemical Stability to the Extracts
Abstract
1. Introduction
2. Results and discussion
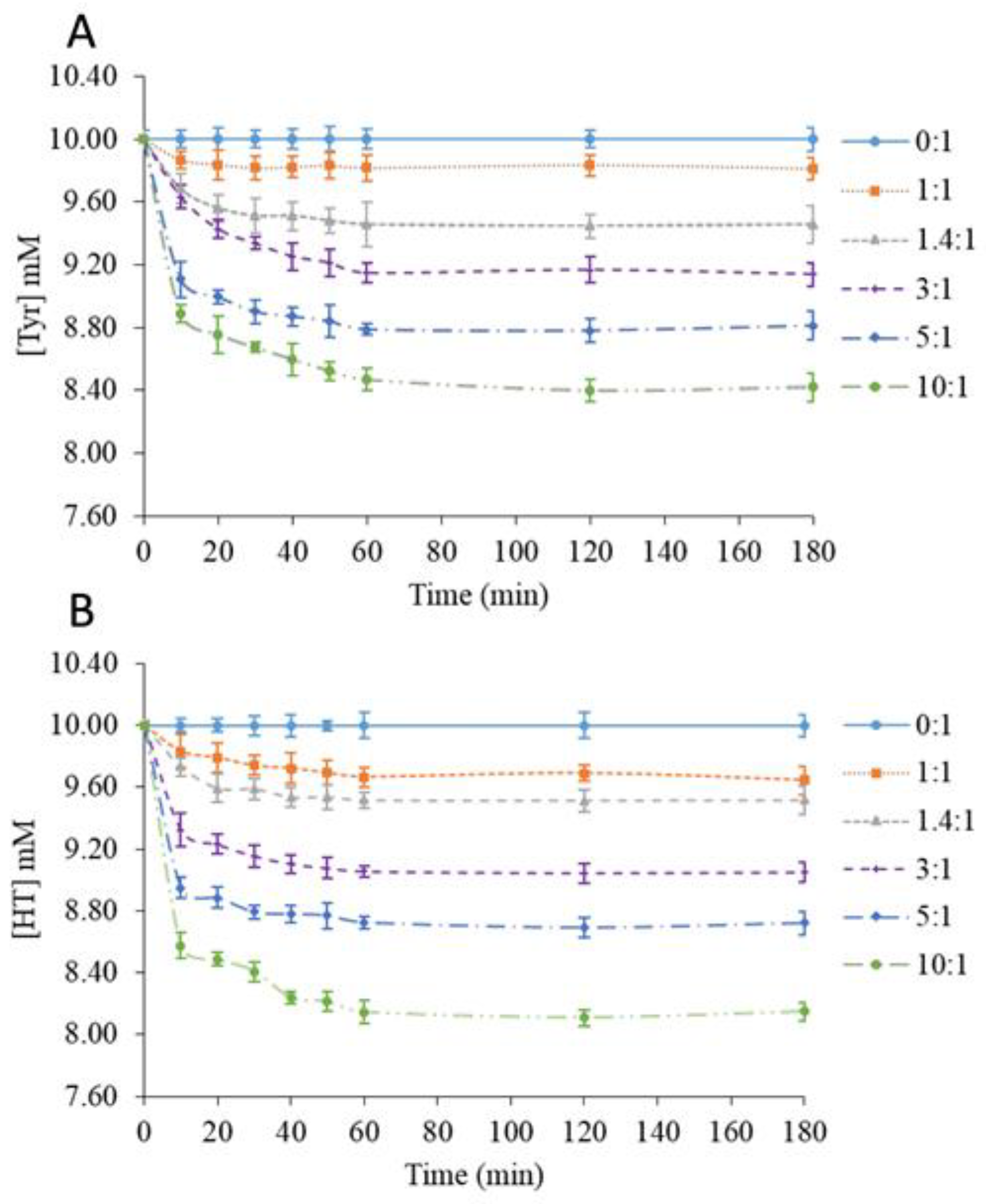
2.1. Influence of the β-CD/Phenol Molar Ratio
2.2. Influence of the Biophenol Concentration
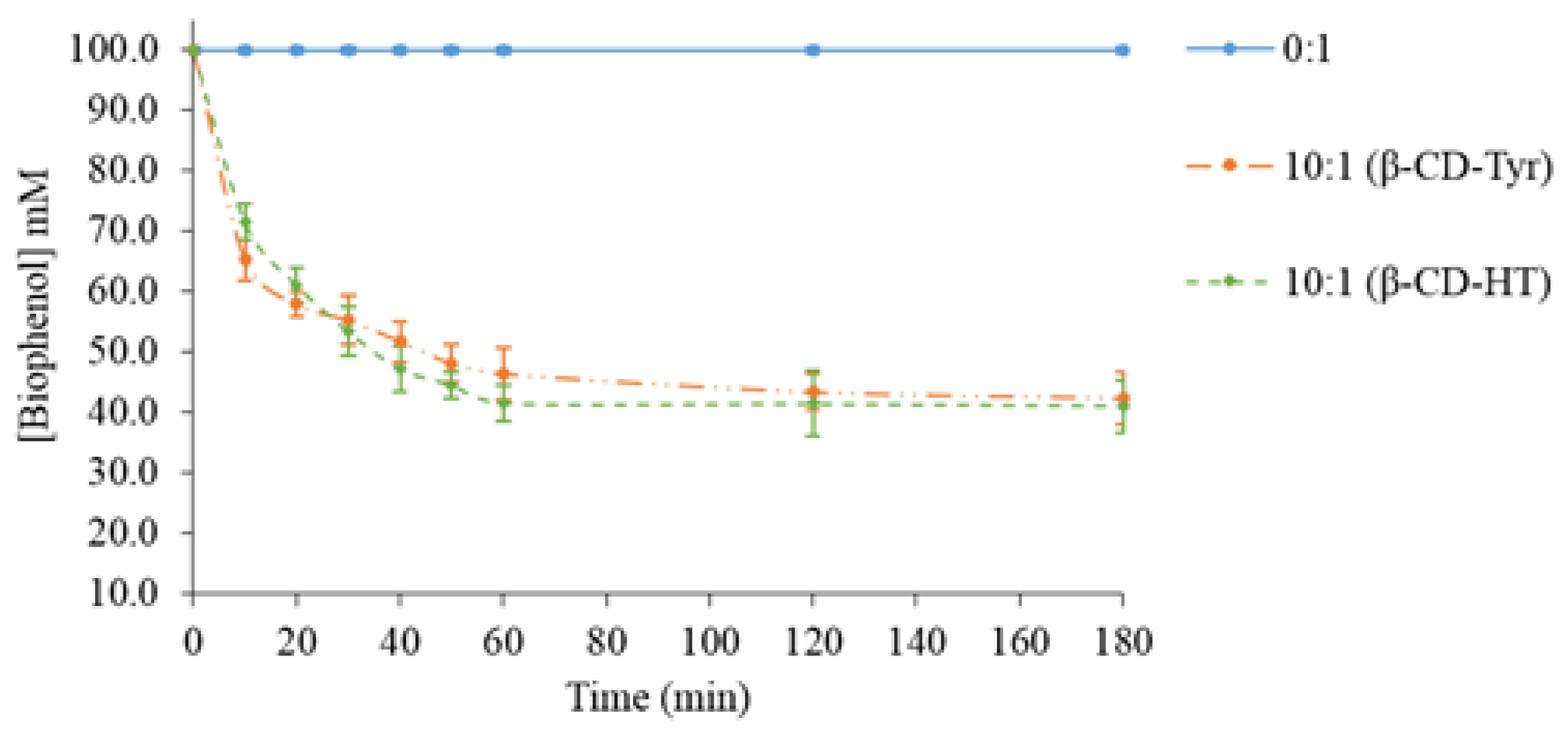
2.3. Photochemical Stability
2.4. Scanning Electron Microscopy (SEM)
3. Experimental
3.1. Materials
3.2. Spectroscopic Analysis
3.2.1. Standard Solution
3.2.2. Kinetic Analysis
3.3. Analysis of Complexes in the Solid State
3.3.1. Solid Recovery Procedure
3.3.2. Loading and Extraction Efficiencies
3.4. Photodegradation
3.5. Scanning Electron Microscopy
4. Conclusions
5. Patents
Author Contributions
Funding
International Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Tsagaraki, E.; Lazarides, H.N.; Petrotos, K.B. Olive Mill Wastewater Treatment; Springer: Boston, MA, USA, 2007; pp. 133–157. [Google Scholar]
- Rodis, P.S.; Karathanos, V.T.; Mantzavinou, A. Partitioning of olive oil antioxidants between oil and water phases. J. Agric. Food Chem. 2002, 50, 596–601. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality [Electronic Resource]: Incorporating 1st and 2nd Addenda, Recommendations, 3rd ed.; World Health Organization: Geneva, Switzerland, 2008; Volume 1, pp. 417–419. [Google Scholar]
- Iboukhoulef, H.; Amrane, A.; Kadi, H. Removal of phenolic compounds from olive mill wastewater by a Fenton-like system H2O2/Cu(II)-thermodynamic and kinetic modeling. Desalination Water Treat. 2016, 57, 1874–1879. [Google Scholar] [CrossRef]
- Chedeville, O.; Debacq, M.; Porte, C. Removal of phenolic compounds present in olive mill wastewaters by ozonation. Desalination 2009, 249, 865–869. [Google Scholar] [CrossRef]
- Comandini, P.; Lerma-García, M.J.; Massanova, P.; Simó-Alfonso, E.F.; Gallina Toschi, T. Phenolic profiles of olive mill wastewaters treated by membrane filtration systems. J. Chem. Technol. Biotechnol. 2015, 90, 1086–1093. [Google Scholar] [CrossRef]
- Aly, A.A.; Hasan, Y.N.Y.; Al-Farraj, A.S. Olive mill wastewater treatment using a simple zeolite-based low-cost method. J. Environ. Manag. 2014, 145, 341–348. [Google Scholar] [CrossRef]
- Abdelkreem, M. Adsorption of Phenol from Industrial Wastewater Using Olive Mill Waste. APCBEE Procedia 2013, 5, 349–357. [Google Scholar] [CrossRef]
- Aliakbarian, B.; Casazza, A.; Perego, P. Valorization of olive oil solid waste using high pressure—High temperature reactor. Food Chem. 2011, 128, 704–710. [Google Scholar] [CrossRef]
- Ena, A.; Pintucci, C.; Carlozzi, P. The recovery of polyphenols from olive mill waste using two adsorbing vegetable matrices. J. Biotechnol. 2012, 157, 573–577. [Google Scholar] [CrossRef]
- Fernández-Bolaños, J.G.; López, Ó.; López-García, M.Á.; Marset, A. Biological properties of hydroxytyrosol and its derivatives. In Olive Oil-Constituents, Quality, Health Properties and Bioconversions; IntechOpen: London, UK, 2012; pp. 20–24. [Google Scholar]
- Mortensen, A.; Aguilar, F.; Crebelli, R.; Di Domenico, A.; Dusemund, B.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Leblanc, J.; et al. Re-evaluation of β-cyclodextrin (E 459) as a food additive. EFSA J. 2016, 14, 4628. [Google Scholar]
- Zarzycki, P.K.; Fenert, B.; Głód, B.K. Cyclodextrins-based nanocomplexes for encapsulation of bioactive compounds in food, cosmetics, and pharmaceutical products: Principles of supramolecular complexes formation, their influence on the antioxidative properties of target chemicals, and recent advances in selected industrial applications. In Encapsulations, Nano-Technology in the Agri-Food Industry; Grumezescu, A.M., Ed.; Academic Press: London, UK, 2016; pp. 717–767. [Google Scholar]
- Aksamija, A.; Polidori, A.; Plasson, R.; Dangles, O.; Tomao, V. The inclusion complex of rosmarinic acid into beta-cyclodextrin: A thermodynamic and structural analysis by NMR and capillary electrophoresis. Food Chem. 2016, 208, 258–263. [Google Scholar] [CrossRef]
- Rescifina, A.; Chiacchio, U.; Iannazzo, D.; Piperno, A.; Romeo, G. β-cyclodextrin and caffeine complexes with catural polyphenols from olive and olive oils: NMR, thermodynamic, and molecular modeling studies. J. Agric. Food Chem. 2010, 58, 11876–11882. [Google Scholar] [CrossRef]
- López-García, M.Á.; López, Ó.; Maya, I.; Fernández-Bolaños, J.G. Complexation of hydroxytyrosol with β-cyclodextrins. An efficient photoprotection. Tetrahedron 2010, 66, 8006–8011. [Google Scholar] [CrossRef]
- Mourtzinos, I.; Salta, F.; Yannakopoulou, K.; Chiou, A.; Karathanos, V.T. Encapsulation of olive leaf extract in β-cyclodextrin. J. Agric. Food Chem. 2007, 55, 8088–8094. [Google Scholar] [CrossRef]
- Mourtzinos, I.; Papadakis, S.E.; Igoumenidis, P.; Karathanos, V.T. Encapsulation of Melissa Officinalis leaf’s active compounds in β-cyclodextrin and modified starch. Procedia Food Sci. 2011, 1, 1679–1685. [Google Scholar] [CrossRef]
- Wilkowska, A.; Ambroziak, W.; Czyżowska, A.; Adamiec, J. Effect of microencapsulation by spray-drying and freeze-drying technique on the antioxidant properties of blueberry (Vaccinium myrtillus) juice polyphenolic compounds. Pol. J. Food Nutr. Sci. 2016, 66, 11–16. [Google Scholar] [CrossRef]
- Hundre, S.Y.; Karthik, P.; Anandharamakrishnan, C. Effect of whey protein isolate and β-cyclodextrin wall systems on stability of microencapsulated vanillin by spray–freeze drying method. Food Chem. 2015, 174, 16–24. [Google Scholar] [CrossRef]
- García-Padial, M.; Martínez-Ohárriz, M.C.; Isasi, J.R.; Vélaz, I.; Zornoza, A. Complexation of tyrosol with cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 2013, 75, 241–246. [Google Scholar] [CrossRef]
- Ahmaruzzaman, M. Adsorption of phenolic compounds on low-cost adsorbents. Adv. Colloid Interface Sci. 2008, 143, 48–67. [Google Scholar] [CrossRef] [PubMed]
- Soto, M.L.; Moure, A.; Domínguez, H.; Parajó, J.C. Recovery, concentration and purification of phenolic compounds by adsorption. J. Food Eng. 2011, 105, 1–27. [Google Scholar] [CrossRef]
- Aoki, N.; Murai, H.; Hattori, K. Removal of phenolic compounds from aqueous solutions using ionic interaction between cyclodextrin derivatives and chitosan. Trans. Mater. Res. Soc. Jpn. 2010, 35, 809–812. [Google Scholar] [CrossRef][Green Version]
- Romo, A.; Peñas, F.J.; Isasi, J.R.; García-Zubiri, I.X.; González-Gaitano, G. Extraction of phenols from aqueous solutions by β-cyclodextrin polymers. Comparison of sorptive capacities with other sorbents. React. Funct. Polym. 2008, 68, 406–413. [Google Scholar] [CrossRef]
- Xiaohong, L.I.; Baowei, Z.; Kun, Z.H.U.; Xuekui, H.A.O. Removal of nitrophenols by adsorption using β-cyclodextrin modified zeolites. Chin. J. Chem. Eng. 2011, 19, 938–943. [Google Scholar]
- Wang, H.; Wang, Y.; Zhou, Y.; Han, P.; Lü, X. A facile removal of phenol in wastewater using crosslinked β-cyclodextrin particles with ultrasonic treatment: Removal of phenol in wastewater using β-cyclodextrin. Clean-Soil Air Water. 2014, 42, 51–55. [Google Scholar] [CrossRef]
- Jantarat, C.; Sirathanarun, P.; Ratanapongsai, S.; Watcharakan, P.; Sunyapong, S.; Wadu, A. Curcumin-Hydroxypropyl-beta-cyclodextrin inclusion complex preparation methods: Effect of common solvent evaporation, freeze drying, and pH shift on solubility and stability of curcumin. Trop. J. Pharm. Res. 2014, 13, 1215. [Google Scholar] [CrossRef]
- Kumar, N.S.; Suguna, M.; Subbaiah, M.V.; Reddy, A.S.; Kumar, N.P.; Krishnaiah, A. Adsorption of phenolic compounds from aqueous solutions onto chitosan-coated perlite beads as biosorbent. Ind. Eng. Chem. Res. 2010, 49, 9238–9247. [Google Scholar] [CrossRef]


| Solid Recovery (%) | Solid Recovery Efficiency (%) | Extraction Efficiency (%) | Loading Efficiency | ||
|---|---|---|---|---|---|
| mmol/100 g | mg/g | ||||
| 3:1 | 22.2 ± 3.9 | 69.2 ± 3.9 | 2.2 ± 0.2 | 2.9 ± 0.3 | 4.1 ± 0.4 |
| 5:1 | 51.7 ± 4.6 | 79.9 ± 4.6 | 6.1 ± 0.4 | 2.1 ± 0.1 | 2.9 ± 0.1 |
| 10:1 | 75.4 ± 5.1 | 89.5 ± 5.1 | 10.9 ± 1.2 | 1.3 ± 0.1 | 1.8 ± 0.1 |
| β-CD/HT Ratio | Solid Recovery (%) | Solid Recovery Efficiency (%) | Extraction Efficiency (%) | Loading Efficiency | |
|---|---|---|---|---|---|
| mmol/100 g | mg/g | ||||
| 3:1 | 31.4 ± 4.8 | 78.4 ± 4.8 | 3.4 ± 0.3 | 3.2 ± 0.2 | 4.9 ± 0.3 |
| 5:1 | 57.4 ± 5.4 | 85.6 ± 5.4 | 7.1 ± 0.6 | 2.2 ± 0.0 | 3.4 ± 0.1 |
| 10:1 | 79.1 ± 3.1 | 93.2 ± 3.1 | 13.6 ± 1.3 | 1.5 ± 0.1 | 2.3 ± 0.1 |
| β-CD/Phenol Ratio | Solid Recovery (%) | Solid Recovery Efficiency (%) | Extraction Efficiency (%) | Loading Efficiency | ||
|---|---|---|---|---|---|---|
| mmol/100 g | mg/g | |||||
| Tyr | 10:1 | 91.7 ± 6.2 | 93.1 ± 6.2 | 61.9 ± 4.9 | 5.9 ± 0.1 | 8.2 ± 0.1 |
| HT | 10:1 | 92.2 ± 3.1 | 93.6 ± 3.1 | 61.7 ± 3.1 | 5.9 ± 0.1 | 9.1 ± 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malapert, A.; Reboul, E.; Dangles, O.; Thiéry, A.; Sylla, N.; Tomao, V. One-Step Extraction of Olive Phenols from Aqueous Solution Using β-Cyclodextrin in the Solid State, a Simple Eco-Friendly Method Providing Photochemical Stability to the Extracts. Molecules 2021, 26, 4463. https://doi.org/10.3390/molecules26154463
Malapert A, Reboul E, Dangles O, Thiéry A, Sylla N, Tomao V. One-Step Extraction of Olive Phenols from Aqueous Solution Using β-Cyclodextrin in the Solid State, a Simple Eco-Friendly Method Providing Photochemical Stability to the Extracts. Molecules. 2021; 26(15):4463. https://doi.org/10.3390/molecules26154463
Chicago/Turabian StyleMalapert, Aurélia, Emmanuelle Reboul, Olivier Dangles, Alain Thiéry, N’nabinty Sylla, and Valérie Tomao. 2021. "One-Step Extraction of Olive Phenols from Aqueous Solution Using β-Cyclodextrin in the Solid State, a Simple Eco-Friendly Method Providing Photochemical Stability to the Extracts" Molecules 26, no. 15: 4463. https://doi.org/10.3390/molecules26154463
APA StyleMalapert, A., Reboul, E., Dangles, O., Thiéry, A., Sylla, N., & Tomao, V. (2021). One-Step Extraction of Olive Phenols from Aqueous Solution Using β-Cyclodextrin in the Solid State, a Simple Eco-Friendly Method Providing Photochemical Stability to the Extracts. Molecules, 26(15), 4463. https://doi.org/10.3390/molecules26154463







