Santamarine Shows Anti-Photoaging Properties via Inhibition of MAPK/AP-1 and Stimulation of TGF-β/Smad Signaling in UVA-Irradiated HDFs
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
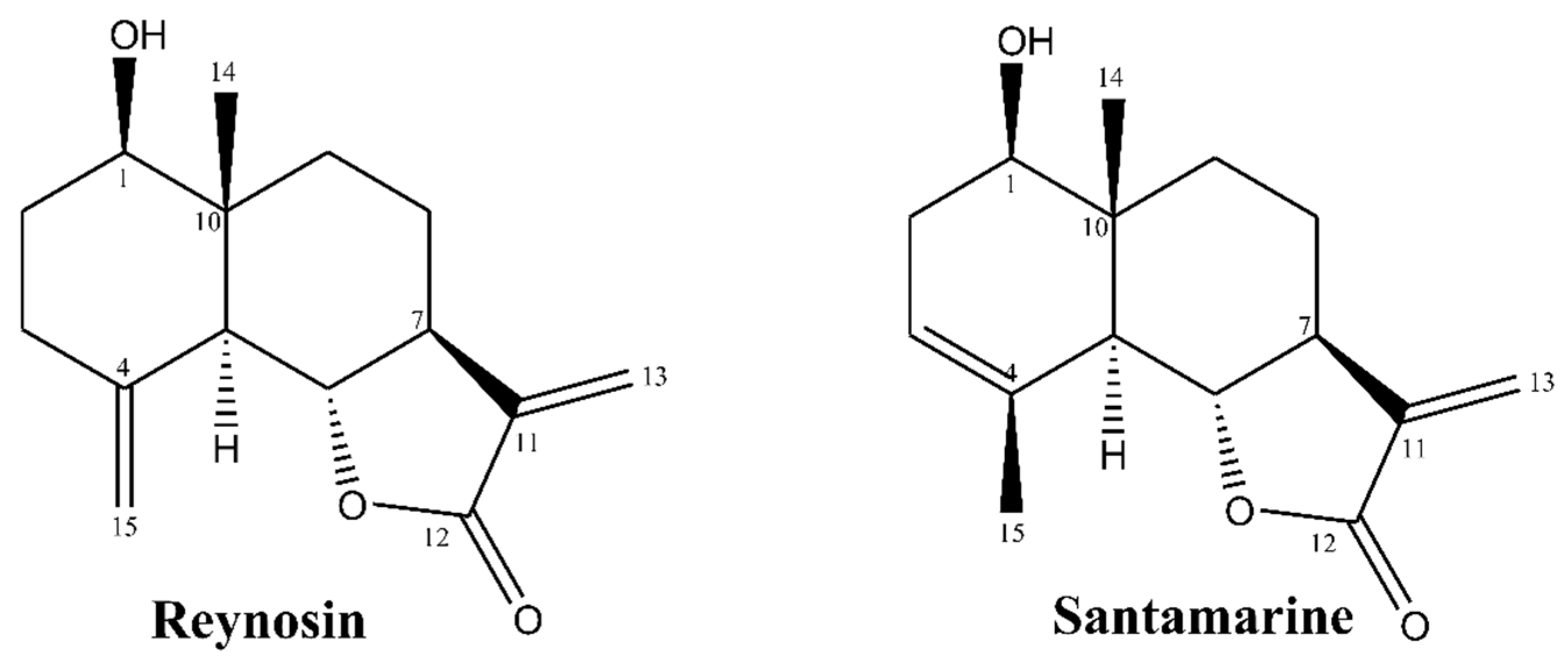
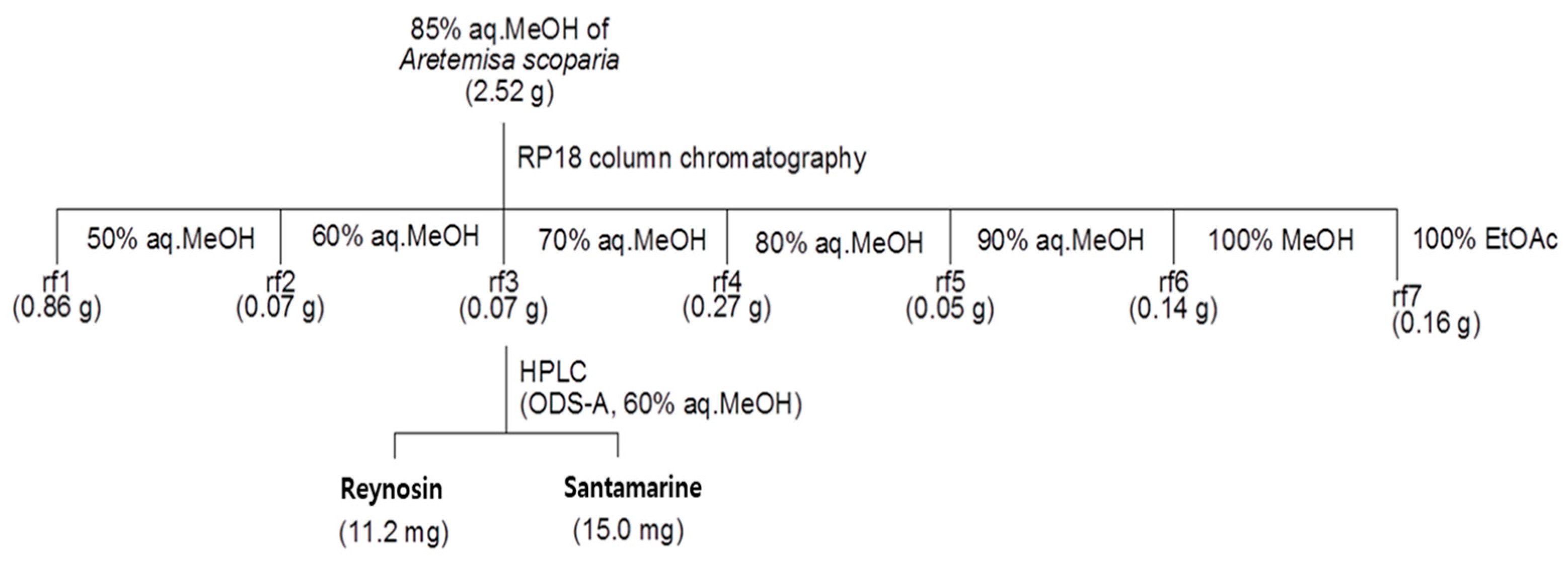
2.2. Isolation and Characterization of Reynosin and Santamarine
2.3. Cell Culture and Viability Assay
2.4. UVA Irradiation
2.5. DCFH-DA Cellular ROS Assay
2.6. Enzyme-Linked Immunosorbent Assay (ELISA)
2.7. Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR) Analysis
2.8. Western Blot
2.9. Immunofluorescence Staining
2.10. Statistical Analysis
3. Results
3.1. The Characterization of Reynosin and Santamarine
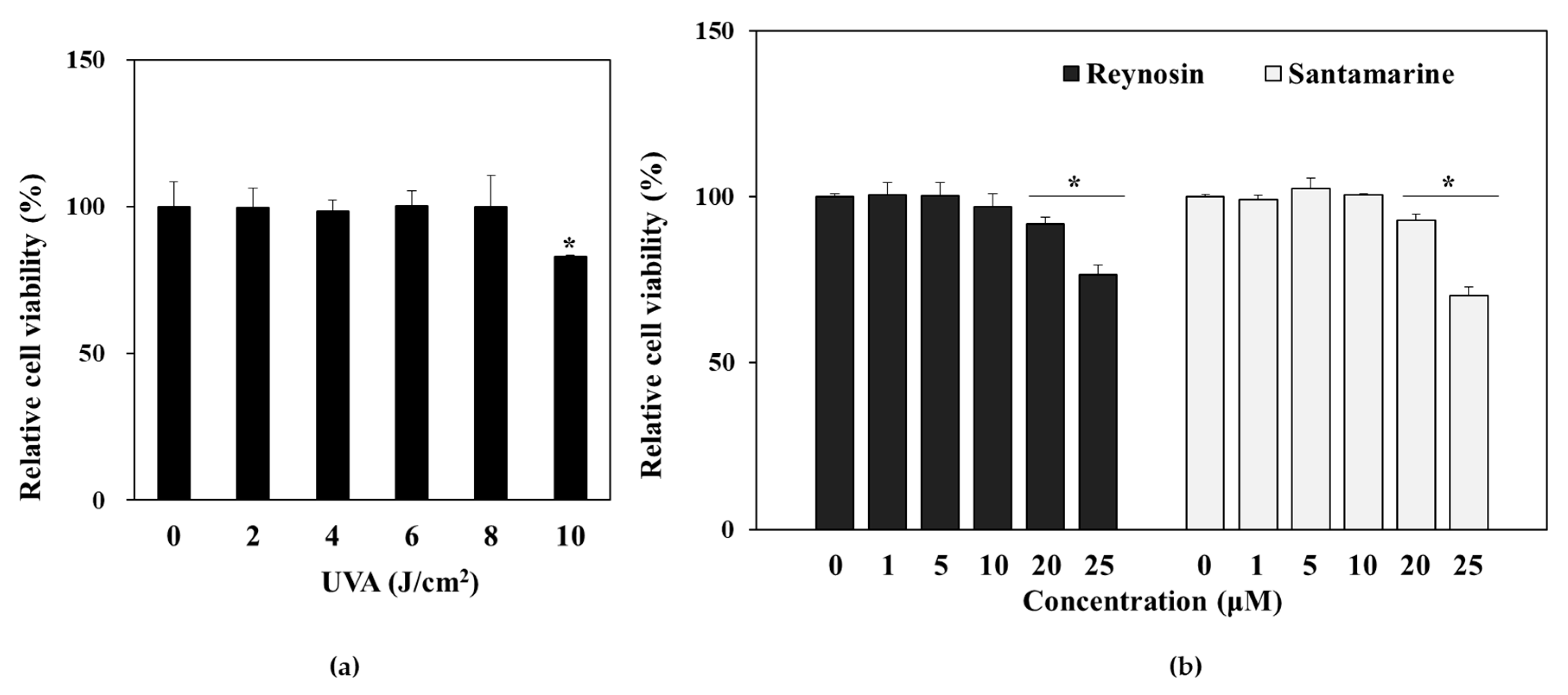
3.2. Effect of UVA Irradiation and Sample Treatment on the Viability of HDFs
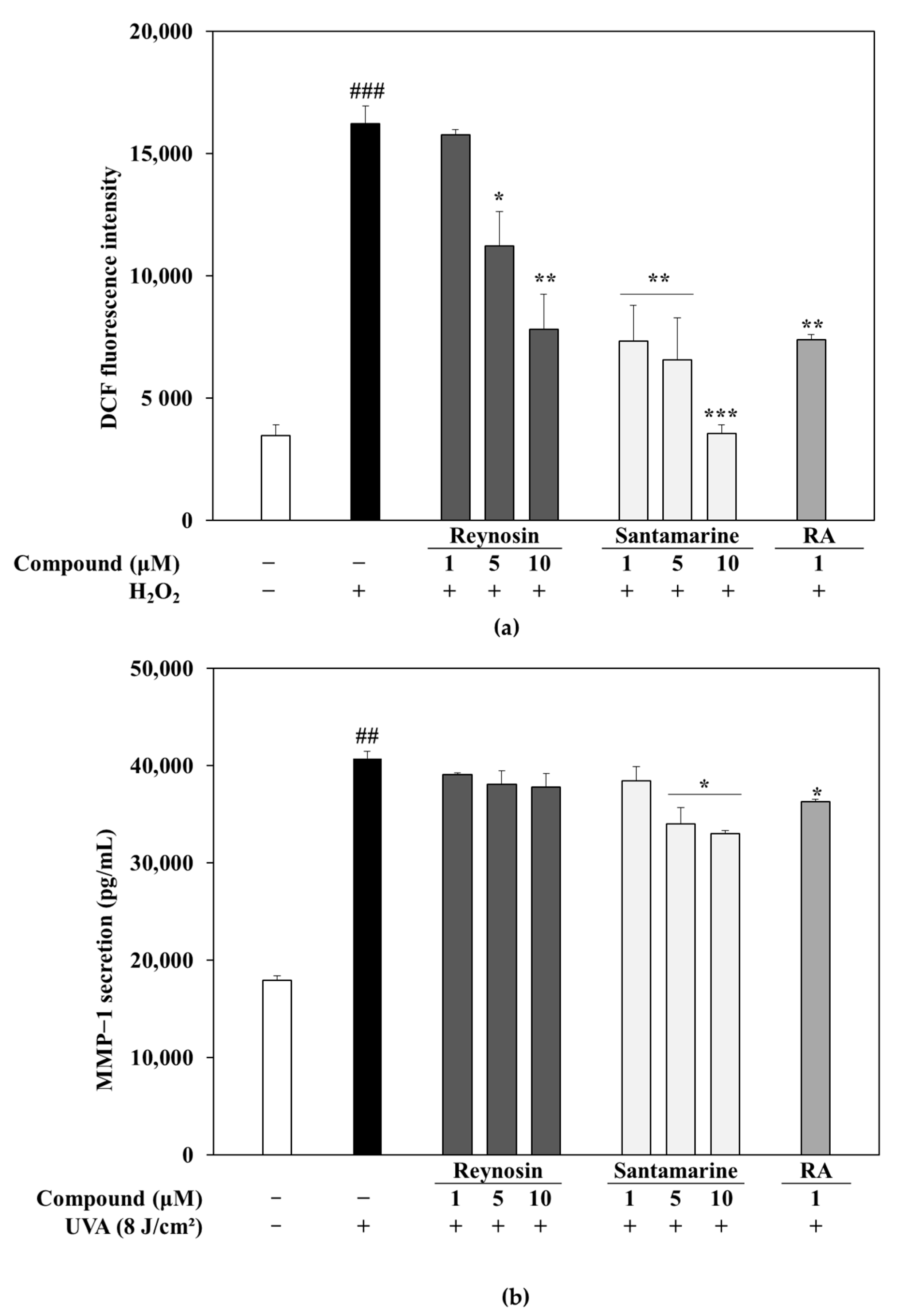
3.3. Measurement of Antioxidant Activity of Reynosin and Santamarine
3.4. Effect of Reynosin and Santamarine on UVA-Induced MMP-1 Secretion
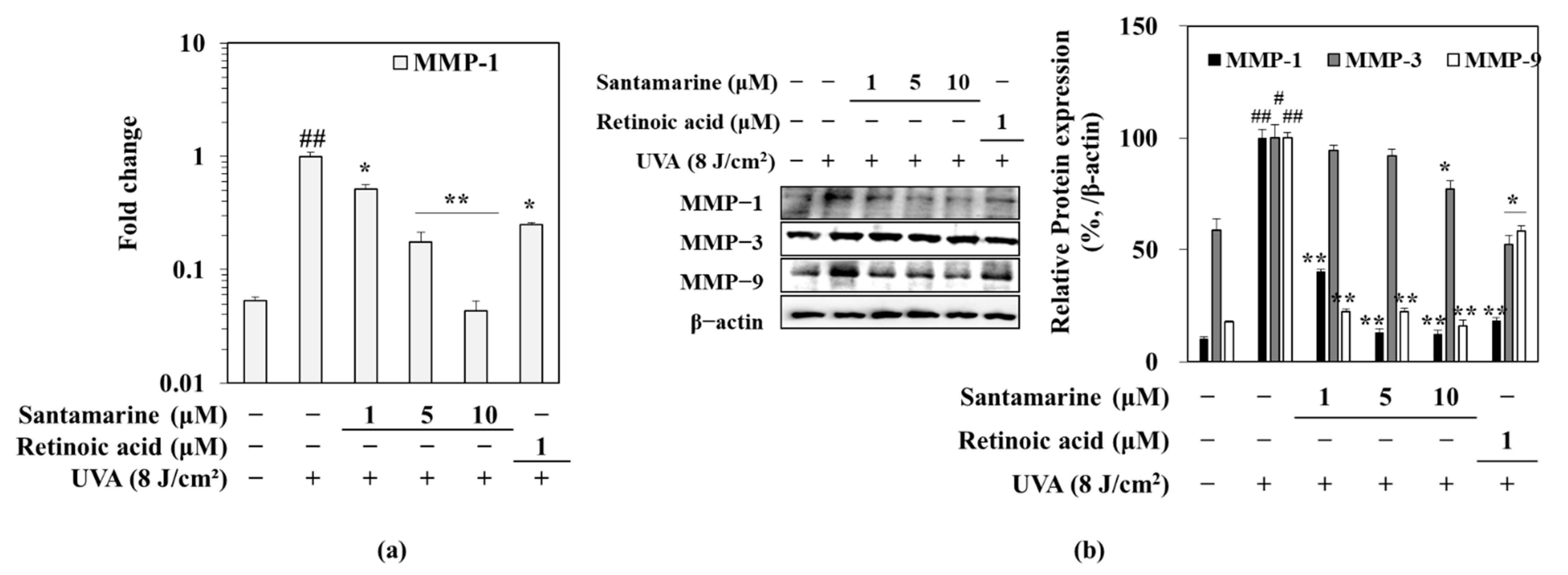
3.5. Santamarine Downregulated the UVA-Induced mRNA Expression of MMP-1, and Protein Expression of MMP-1, 3 and 9
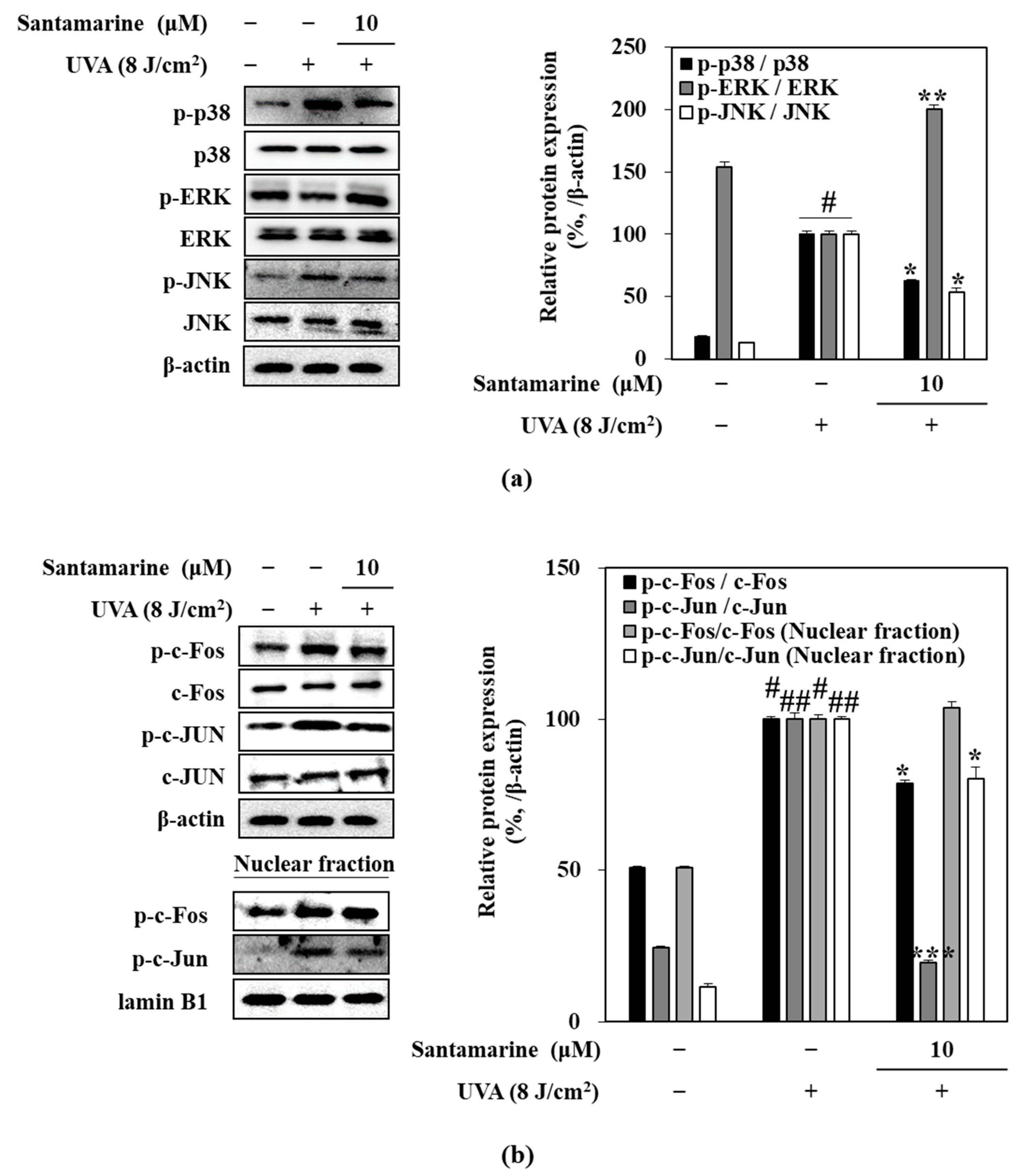
3.6. Santamarine Suppressed UVA-Induced Activation of MAPK/AP-1
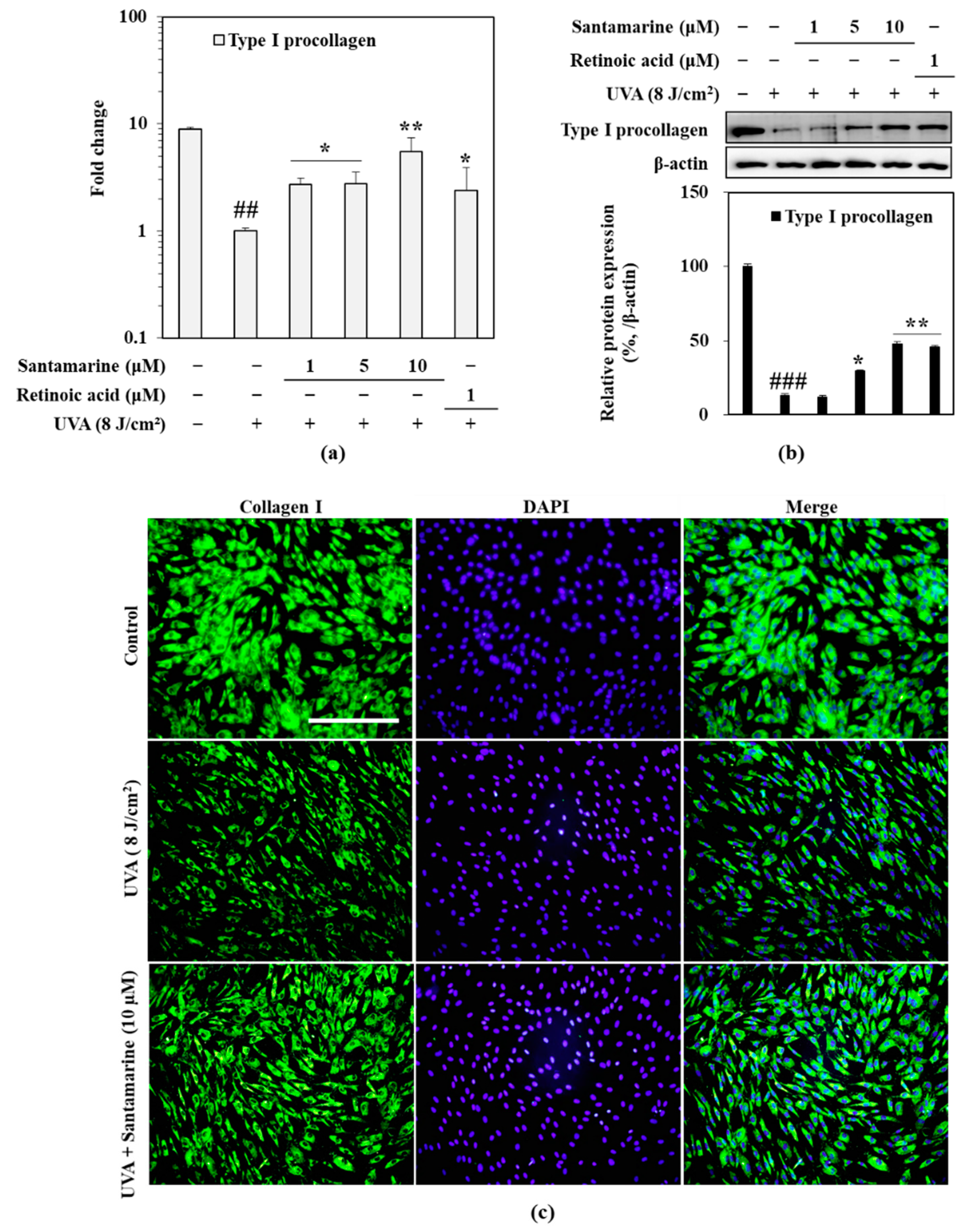
3.7. Santamarine Ameliorated UVA-Mediated Decrease in Type I Procollagen Expression
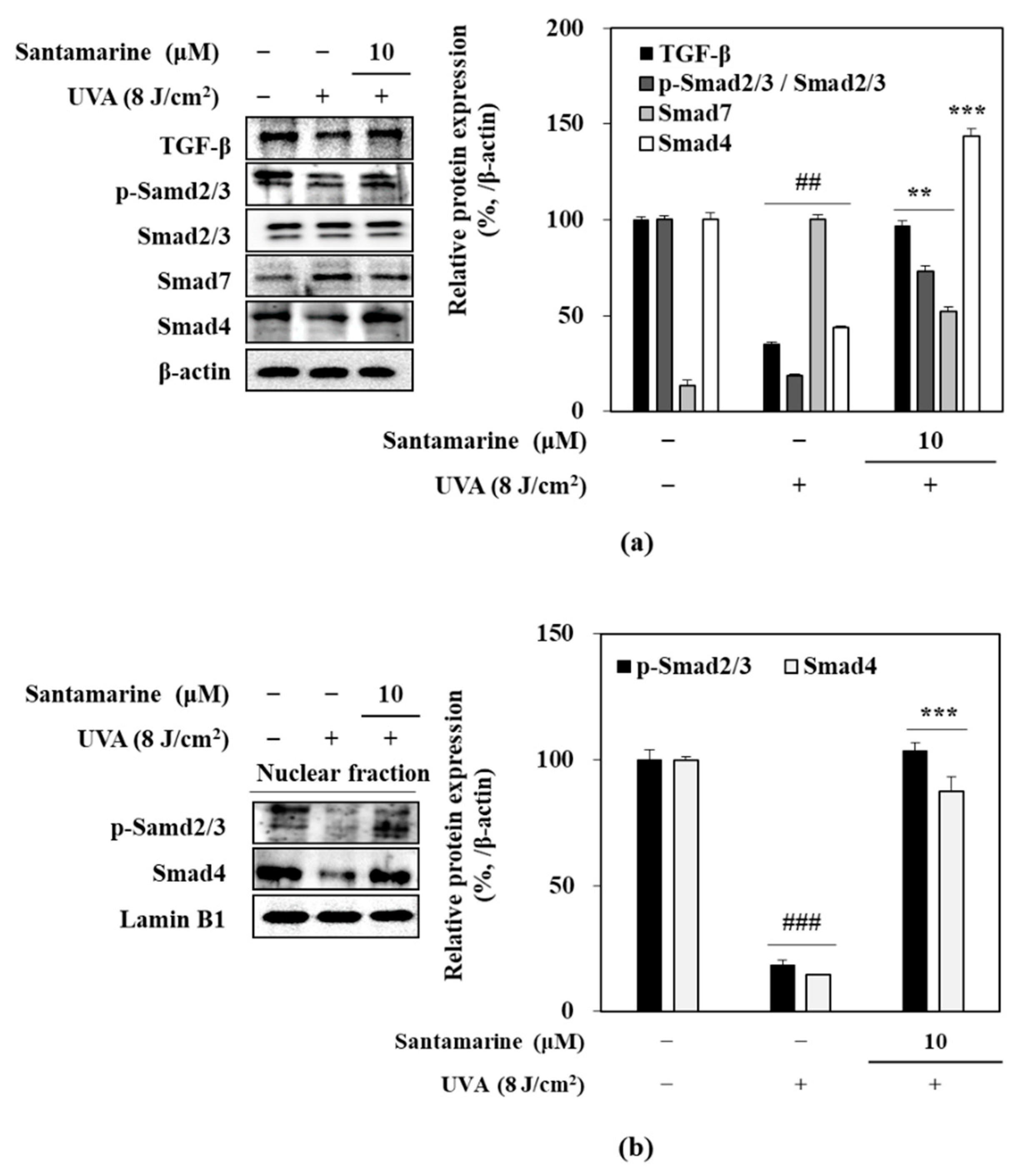
3.8. Effect of Santamarine on TGF-β/Smad Pathway
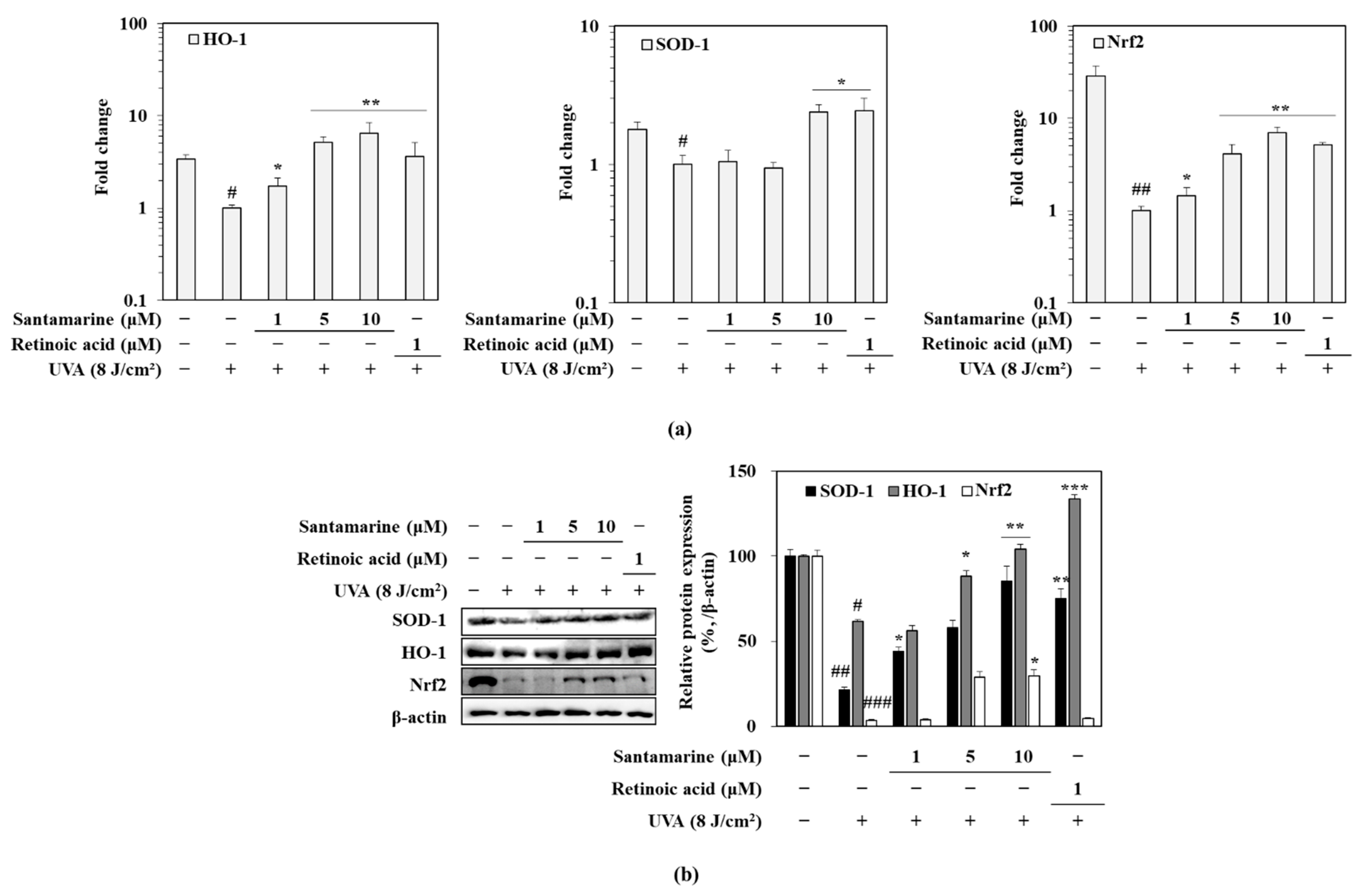
3.9. Santamarine Ameliorated UVA-Induced Deterioration of Nrf-Dependent Antioxidant Mechanism
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV Radiation and the Skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef] [PubMed]
- Meinhardt, M.; Krebs, R.; Anders, A.; Heinrich, U.; Tronnier, H. Wavelength-dependent penetration depths of ultraviolet radiation in human skin. J. Biomed. Opt. 2008, 13, 044030. [Google Scholar] [CrossRef]
- Bossi, O.; Gartsbein, M.; Leitges, M.; Kuroki, T.; Grossman, S.; Tennenbaum, T. UV irradiation increases ROS production via PKCδ signaling in primary murine fibroblasts. J. Cell. Biochem. 2008, 105, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Ou-Yang, H.; Stamatas, G.; Saliou, C.; Kollias, N. A Chemiluminescence Study of UVA-induced oxidative stress in human skin in vivo. J. Investig. Dermatol. 2004, 122, 1020–1029. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, K.S.; Kim, S.H.; Baek, S.-H.; Kim, H.Y.; Lee, C.; Kim, J.-R. Induction of Cellular Senescence by Secretory Phospholipase A2 in Human Dermal Fibroblasts through an ROS-Mediated p53 Pathway. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2009, 64A, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Bosch, R.; Philips, N.; Suárez-Pérez, J.; Juarranz, A.; Devmurari, A.; Chalensouk-Khaosaat, J.; González, S. Mechanisms of Photoaging and Cutaneous Photocarcinogenesis, and Photoprotective Strategies with Phytochemicals. Antioxidants 2015, 4, 248–268. [Google Scholar] [CrossRef]
- Lan, C.C.E.; Hung, Y.T.; Fang, A.H.; Ching-Shuang, W. Effects of irradiance on UVA-induced skin aging. J. Dermatol. Sci. 2019, 94, 220–228. [Google Scholar] [CrossRef]
- Jean, C.; Bogdanowicz, P.; Haure, M.-J.; Castex-Rizzi, N.; Fournié, J.-J.; Laurent, G. UVA-activated synthesis of metalloproteinases 1, 3 and 9 is prevented by a broad-spectrum sunscreen. Photodermatol. Photoimmunol. Photomed. 2011, 27, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Battie, C.; Jitsukawa, S.; Bernerd, F.; Del Bino, S.; Marionnet, C.; Verschoore, M. New insights in photoaging, UVA induced damage and skin types. Exp. Dermatol. 2014, 23, 7–12. [Google Scholar] [CrossRef]
- Fisher, G.J. The pathophysiology of photoaging of the skin. Cutis 2005, 75, 5–9. [Google Scholar]
- Tian, F.; Zhang, F.; Lai, X.; Wang, L.; Yang, L.; Wang, X.; Singh, G.; Li Zhong, J.L. Nrf2-mediated protection against UVA radiation in human skin keratinocytes. Biosci. Trends 2011, 5, 23–29. [Google Scholar] [CrossRef]
- Ryšavá, A.; Čížková, K.; Franková, J.; Roubalová, L.; Ulrichová, J.; Vostálová, J.; Vrba, J.; Zálešák, B.; Rajnochová Svobodová, A. Effect of UVA radiation on the Nrf2 signalling pathway in human skin cells. J. Photochem. Photobiol. B Biol. 2020, 209, 111948. [Google Scholar] [CrossRef]
- Chaiprasongsuk, A.; Onkoksoong, T.; Pluemsamran, T.; Limsaengurai, S.; Panich, U. Photoprotection by dietary phenolics against melanogenesis induced by UVA through Nrf2-dependent antioxidant responses. Redox Biol. 2016, 8, 79–90. [Google Scholar] [CrossRef]
- Stevanato, R.; Bertelle, M.; Fabris, S. Photoprotective characteristics of natural antioxidant polyphenols. Regul. Toxicol. Pharmacol. 2014, 69, 71–77. [Google Scholar] [CrossRef]
- Herranz-López, M.; Barrajón-Catalán, E. Antioxidants and Skin Protection. Antioxidants 2020, 9, 704. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Wang, L.; He, C.; Zhao, J.; Si, L.; Huang, H. Artemisia scoparia: Traditional uses, active constituents and pharmacological effects. J. Ethnopharmacol. 2021, 113960. [Google Scholar] [CrossRef] [PubMed]
- Stojanović, G.S.; Ickovski, J.D.; Đorđević, A.S.; Petrović, G.M.; Stepić, K.D.; Palić, I.R.; Stamenković, J.G. The First Report on Chemical Composition and Antimicrobial Activity of Artemisia scoparia Waldst. et Kit. Extracts. Nat. Prod. Commun. 2020, 15, 1934578X2091503. [Google Scholar] [CrossRef]
- Wang, X.; Huang, H.; Ma, X.; Wang, L.; Liu, C.; Hou, B.; Yang, S.; Zhang, L.; Du, G. Anti-inflammatory effects and mechanism of the total flavonoids from Artemisia scoparia Waldst. et kit. in vitro and in vivo. Biomed. Pharmacother. 2018, 104, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, Z.; Xie, Y.; Hu, H. Antitumor activity and mechanism of costunolide and dehydrocostus lactone: Two natural sesquiterpene lactones from the Asteraceae family. Biomed. Pharmacother. 2020, 125, 109955. [Google Scholar] [CrossRef]
- Babaei, G.; Aliarab, A.; Abroon, S.; Rasmi, Y.; Aziz, S.G.G. Application of sesquiterpene lactone: A new promising way for cancer therapy based on anticancer activity. Biomed. Pharmacother. 2018, 106, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Coronado-Aceves, E.W.; Velázquez, C.; Robles-Zepeda, R.E.; Jiménez-Estrada, M.; Hernández-Martínez, J.; Gálvez-Ruiz, J.C.; Garibay-Escobar, A. Reynosin and santamarine: Two sesquiterpene lactones from Ambrosia confertiflora with bactericidal activity against clinical strains of Mycobacterium tuberculosis. Pharm. Biol. 2016, 54, 2623–2628. [Google Scholar] [CrossRef]
- Muthusamy, V.; Piva, T.J. The UV response of the skin: A review of the MAPK, NFκB and TNFα signal transduction pathways. Arch. Dermatol. Res. 2010, 302, 5–17. [Google Scholar] [CrossRef]
- Polte, T.; Tyrrell, R.M. Involvement of lipid peroxidation and organic peroxides in UVA-induced matrix metalloproteinase-1 expression. Free Radic. Biol. Med. 2004, 36, 1566–1574. [Google Scholar] [CrossRef]
- Tu, Y.; Quan, T. Oxidative Stress and Human Skin Connective Tissue Aging. Cosmetics 2016, 3, 28. [Google Scholar] [CrossRef]
- Xu, Q.; Hou, W.; Zheng, Y.; Liu, C.; Gong, Z.; Lu, C.; Lai, W.; Maibach, H.I. Ultraviolet A-Induced Cathepsin K Expression Is Mediated via MAPK/AP-1 Pathway in Human Dermal Fibroblasts. PLoS ONE 2014, 9, e102732. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.G.; Lee, D.S.; Li, B.; Choi, Y.H.; Lee, S.H.; Kim, Y.C. Santamarin, a sesquiterpene lactone isolated from Saussurea lappa, represses LPS-induced inflammatory responses via expression of heme oxygenase-1 in murine macrophage cells. Int. Immunopharmacol. 2012, 13, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I.; Gilmour, P.S.; Jimenez, L.A.; MacNee, W. Oxidative stress and TNF-a induce histone Acetylation and NF-кB/AP-1 activation in Alveolar epithelial cells: Potential mechanism in gene transcription in lung inflammation. In Oxygen/Nitrogen Radicals: Cell Injury and Disease; Springer: New York, NY, USA, 2002; pp. 239–248. [Google Scholar]
- Ji, Z.; He, L.; Regev, A.; Struhl, K. Inflammatory regulatory network mediated by the joint action of NF-kB, STAT3, and AP-1 factors is involved in many human cancers. Proc. Natl. Acad. Sci. USA 2019, 116, 9453–9462. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.Y.; Choi, B.Y. Costunolide—A Bioactive Sesquiterpene Lactone with Diverse Therapeutic Potential. Int. J. Mol. Sci. 2019, 20, 2926. [Google Scholar] [CrossRef]
- Cheng, X.; Gao, W.; Dang, Y.; Liu, X.; Li, Y.; Peng, X.; Ye, X. Both ERK/MAPK and TGF-Beta/Smad signaling pathways play a role in the kidney fibrosis of diabetic mice accelerated by blood glucose fluctuation. J. Diabetes Res. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.S.; Chang, H.H.; Yeh, C.Y.; Chang, M.C.; Chan, C.P.; Kuo, H.Y.; Liu, H.C.; Liao, W.C.; Jeng, P.Y.; Yeung, S.Y.; et al. Transforming growth factor beta 1 increases collagen content and stimulates procollagen I and tissue inhibitor of metalloproteinase-1 production of dental pulp cells: Role of MEK/ERK and activin receptor-like kinase-5/Smad signaling. J. Formos. Med. Assoc. 2017, 116, 351–358. [Google Scholar] [CrossRef]
- Chen, S.J.; Yuan, W.; Mori, Y.; Levenson, A.; Trojanowska, M.; Varga, J. Stimulation of type I collagen transcription in human skin fibroblasts by TGF-β: Involvement of Smad 3. J. Investig. Dermatol. 1999, 112, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Liu, Z.; Chen, Y. Regulation of TGF- signaling by Smad7. Acta Biochim. Biophys. Sin. (Shanghai) 2009, 41, 263–272. [Google Scholar] [CrossRef] [PubMed]
| Position | δH | δC |
|---|---|---|
| 1 | 3.49 (1H, dd, J = 11.6, 4.5 Hz) | 78.8 |
| 2 | 1.80, 1.56 (1H, m) | 32.1 |
| 3 | 2.32 (1H, ddd, J = 13.5, 4.9, 1.8 Hz), 2.15 (1H, td, J = 13.5, 5.1) 2.15 (1H, td, J = 13.5, 5.1) | 34.8 |
| 4 | 145.1 | |
| 5 | 2.24 (1H, br d, J = 10.9 Hz) | 54.1 |
| 6 | 4.10 (1H, t, J = 10.9 Hz) | 81.5 |
| 7 | 2.61 (1H, tdd, J = 11.2, 3.1, 3.0 Hz) 5 | 50.7 |
| 8 | 2.08, 1.60 (1H, m) | 22.4 |
| 9 | 2.05 (1H, m), 1.38 (1H, td, J = 13.3, 3.8) | 36.9 |
| 10 | 44.2 | |
| 11 | 141.3 | |
| 12 | 171.7 | |
| 13 | 6.00 (1H, d, J = 3.1 Hz), 5.49 (1H, d, J = 3.0 Hz) | 117.3 |
| 14 | 0.80 (3H, s) | 12 |
| 15 | 4.95, 4.79 (1H, s) | 110.2 |
| Position | δH | δC |
|---|---|---|
| 1 | 3.49 (1H, dd, J = 11.6, 4.5 Hz) | 78.8 |
| 2 | 1.80, 1.56 (1H, m) | 32.1 |
| 3 | 2.32 (1H, ddd, J = 13.5, 4.9, 1.8 Hz), 2.15 (1H, td, J = 13.5, 5.1) 2.15 (1H, td, J = 13.5, 5.1) | 34.8 |
| 4 | 145.1 | |
| 5 | 2.24 (1H, br d, J = 10.9 Hz) | 54.1 |
| 6 | 4.10 (1H, t, J = 10.9 Hz) | 81.5 |
| 7 | 2.61 (1H, tdd, J = 11.2, 3.1, 3.0 Hz) 5 | 50.7 |
| 8 | 2.08, 1.60 (1H, m) | 22.4 |
| 9 | 2.05 (1H, m), 1.38 (1H, td, J = 13.3, 3.8) | 36.9 |
| 10 | 44.2 | |
| 11 | 141.3 | |
| 12 | 171.7 | |
| 13 | 6.00 (1H, d, J = 3.1 Hz), 5.49 (1H, d, J = 3.0 Hz) | 117.3 |
| 14 | 0.80 (3H, s) | 12 |
| 15 | 4.95, 4.79 (1H, s) | 110.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, J.H.; Kim, J.; Karadeniz, F.; Kim, H.R.; Park, S.Y.; Seo, Y.; Kong, C.-S. Santamarine Shows Anti-Photoaging Properties via Inhibition of MAPK/AP-1 and Stimulation of TGF-β/Smad Signaling in UVA-Irradiated HDFs. Molecules 2021, 26, 3585. https://doi.org/10.3390/molecules26123585
Oh JH, Kim J, Karadeniz F, Kim HR, Park SY, Seo Y, Kong C-S. Santamarine Shows Anti-Photoaging Properties via Inhibition of MAPK/AP-1 and Stimulation of TGF-β/Smad Signaling in UVA-Irradiated HDFs. Molecules. 2021; 26(12):3585. https://doi.org/10.3390/molecules26123585
Chicago/Turabian StyleOh, Jung Hwan, Junse Kim, Fatih Karadeniz, Hye Ran Kim, So Young Park, Youngwan Seo, and Chang-Suk Kong. 2021. "Santamarine Shows Anti-Photoaging Properties via Inhibition of MAPK/AP-1 and Stimulation of TGF-β/Smad Signaling in UVA-Irradiated HDFs" Molecules 26, no. 12: 3585. https://doi.org/10.3390/molecules26123585
APA StyleOh, J. H., Kim, J., Karadeniz, F., Kim, H. R., Park, S. Y., Seo, Y., & Kong, C.-S. (2021). Santamarine Shows Anti-Photoaging Properties via Inhibition of MAPK/AP-1 and Stimulation of TGF-β/Smad Signaling in UVA-Irradiated HDFs. Molecules, 26(12), 3585. https://doi.org/10.3390/molecules26123585






