Chemical Composition, Antibacterial, Anti-Inflammatory, and Enzyme Inhibitory Activities of Essential Oil from Rhynchanthus beesianus Rhizome
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Composition
2.2. Antibacterial Activity
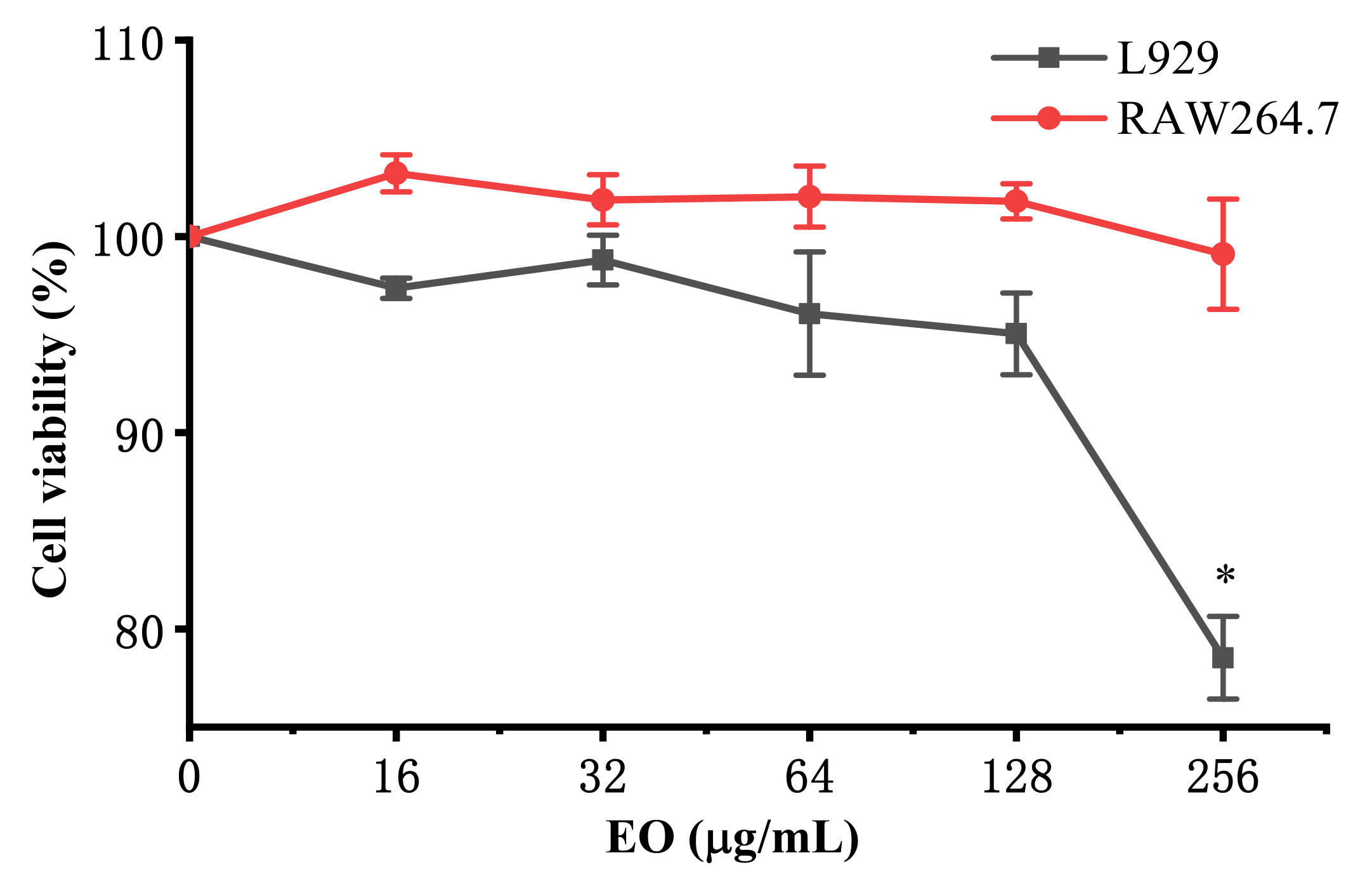
2.3. Anti-inflammatory Activity
2.4. Enzyme Inhibitory Activity
3. Materials and Methods
3.1. Plant Material
3.2. Isolation of Essential Oil
3.3. Essential Oil Analysis and Identification
3.4. Antibacterial Activity
3.5. Anti-Inflammatory Activity
3.6. Enzyme Inhibitory Activities
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Trong Le, N.; Viet Ho, D.; Quoc Doan, T.; Tuan Le, A.; Raal, A.; Usai, D.; Madeddu, S.; Marchetti, M.; Usai, M.; Rappelli, P.; et al. In Vitro Antimicrobial Activity of Essential Oil Extracted from Leaves of Leoheo domatiophorus Chaowasku, D.T. Ngo and H.T. Le in Vietnam. Plants 2020, 9, 453. [Google Scholar] [CrossRef]
- Buchbauer, G.; Bohusch, R. Biological activities of essential oils: An update. In Handbook of Essential Oils: Science, Technology, and Applications; Baser, K.H.C., Buchbauer, G., Eds.; CRC Press/Taylor & Francis Group: Boca Raton, FL, USA, 2010; pp. 281–321. [Google Scholar]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of essential oils. Ind. Crop. Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- The Plant List. Version 1.1. Available online: http://www.theplantlist.org/1.1/browse/A/Zingiberaceae/ (accessed on 25 December 2020).
- Jantan, I.B.; Yassin, M.S.M.; Chin, C.B.; Chen, L.L.; Sim, N.L. Antifungal activity of the essential oils of nine Zingiberaceae species. Pharm. Biol. 2003, 41, 392–397. [Google Scholar] [CrossRef]
- Tan, J.W.; Israf, D.A.; Tham, C.L. Major bioactive compounds in essential oils extracted from the rhizomes of Zingiber zerumbet (L) Smith: A mini-review on the anti-allergic and immunomodulatory properties. Front. Pharmacol. 2018, 9, 652. [Google Scholar] [CrossRef]
- Mahboubi, M. Zingiber officinale Rosc. essential oil, a review on its composition and bioactivity. Clin. Phytosci. 2019, 5, 6. [Google Scholar] [CrossRef]
- Balaji, S.; Chempakam, B. Anti-bacterial Effect of Essential Oils Extracted from Selected Spices of Zingiberaceae. Nat. Prod. J. 2018, 8, 70–76. [Google Scholar] [CrossRef]
- Phukerd, U.; Soonwera, M. Larvicidal and pupicidal activities of essential oils from Zingiberaceae plants against Aedes aegypti (Linn.) and Culex quinquefasciatus Say mosquitoes. Southeast Asian J. Trop Med. Public Health 2013, 44, 761–771. [Google Scholar]
- Tewtrakul, S.; Subhadhirasakul, S. Anti-allergic activity of some selected plants in the Zingiberaceae family. J. Ethnopharmacol. 2007, 109, 535–538. [Google Scholar] [CrossRef]
- The Plant List. Version 1.1. Available online: http://www.theplantlist.org/tpl1.1/search?q=Rhynchanthus (accessed on 14 October 2020).
- Gao, J.Y.; Yang, Z.H.; Ren, P.Y.; Li, Q.J. Reproductive ecology of Rhynchanthus beesianus WW Smith (Zingiberaceae) in South Yunnan, China: A ginger with bird pollination syndrome. J. Integr. Plant Biol. 2006, 48, 1294–1299. [Google Scholar] [CrossRef]
- Wu, D.L.; Larsen, K. Rhynchanthus J. D. Hooker. In Flora of China; Wu, Z.Y., Raven, P.H., Eds.; Science Press: Beijing, China; Missouri Botanical Garden Press: St. Louis, MO, USA, 2000; Volume 24, pp. 346–347. [Google Scholar]
- Chinese Materia Medica Editorial Committee. Zhong Hua Ben Cao [Chinese Materia Medica]; Shanghai Science and Technology Press: Shanghai, China, 1999; Volume 8, p. 648. [Google Scholar]
- CHMC-Chinese Herbal Medicine Company. The Chinese Traditional Medicine Resource Records; Science Press: Beijing, China, 1994; p. 1513. [Google Scholar]
- He, D.S. Yu Long Ben Cao [Yulong Materia Medica]; Yunnan Science and Technology Press: Kunming, China, 2016; p. 242. [Google Scholar]
- Kahkeshani, N.; Hadjiakhoondi, A.; Navidpour, L.; Akbarzadeh, T.; Safavi, M.; Karimpour-Razkenari, E.; Khanavi, M. Chemodiversity of Nepeta menthoides Boiss. & Bohse. essential oil from Iran and antimicrobial, acetylcholinesterase inhibitory and cytotoxic properties of 1,8-cineole chemotype. Nat. Prod. Res. 2018, 32, 2745–2748. [Google Scholar] [PubMed]
- Vuuren, S.V.; Viljoen, A.M. Antimicrobial activity of limonene enantiomers and 1,8-cineole alone and in combination. Flavour Fragr. J. 2007, 22, 540–544. [Google Scholar] [CrossRef]
- Yang, L.; Zhan, C.; Huang, X.; Hong, L.; Fang, L.; Wang, W.; Su, J. Durable Antibacterial Cotton Fabrics Based on Natural Borneol-Derived Anti-MRSA Agents. Adv. Healthc. Mater. 2020, 9, 2000186. [Google Scholar] [CrossRef]
- Donadu, M.G.; Trong Le, N.; Viet Ho, D.; Quoc Doan, T.; Tuan Le, A.; Raal, A.; Usai, M.; Marchetti, M.; Sanna, G.; Madeddu, S.; et al. Phytochemical Compositions and Biological Activities of Essential Oils from the Leaves, Rhizomes and Whole Plant of Hornstedtia bella Škorničk. Antibiotics 2020, 9, 334. [Google Scholar] [CrossRef]
- Alma, M.H.; Nitz, S.; Kollmannsberger, H.; Digrak, M.; Efe, F.T.; Yilmaz, N. Chemical composition and antimicrobial activity of the essential oils from the gum of Turkish pistachio (Pistacia vera L.). J. Agric. Food Chem. 2004, 52, 3911–3914. [Google Scholar] [CrossRef]
- Joshi, R.K. Chemical composition, in vitro antimicrobial and antioxidant activities of the essential oils of Ocimum Gratissimum, O. Sanctum and their major constituents. Indian J. Pharm. Sci. 2013, 75, 457–462. [Google Scholar] [CrossRef]
- Park, S.N.; Lim, Y.K.; Freire, M.O.; Cho, E.; Jin, D.; Kook, J.K. Antimicrobial effect of linalool and α-terpineol against periodontopathic and cariogenic bacteria. Anaerobe 2012, 18, 369–372. [Google Scholar] [CrossRef]
- Santoyo, S.; Cavero, S.; Jaime, L.; Ibanez, E.; Senorans, F.J.; Reglero, G. Chemical composition and antimicrobial activity of Rosmarinus officinalis L. essential oil obtained via supercritical fluid extraction. J. Food Prot. 2005, 68, 790–795. [Google Scholar] [CrossRef]
- Lorençoni, M.F.; Figueira, M.M.; e Silva, M.V.T.; Schmitt, E.F.P.; Endringer, D.C.; Scherer, R.; Barth, T.; Bertolucci, S.K.V.; Fronza, M. Chemical composition and anti-inflammatory activity of essential oil and ethanolic extract of Campomanesia phaea (O. Berg.) Landrum leaves. J. Ethnopharmacol. 2020, 252, 112562. [Google Scholar] [CrossRef]
- Owona, B.A.; Njayou, N.F.; Laufer, S.; Moundipa, P.F.; Schluesener, H.J. A fraction of stem bark extract of Entada africana suppresses lipopolysaccharide-induced inflammation in RAW 264.7 cells. J. Ethnopharmacol. 2013, 149, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Juergens, U.R. Anti-inflammatory properties of the monoterpene 1.8-cineole: Current evidence for co-medication in inflammatory airway diseases. Drug Res. 2014, 64, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Almeida, J.R.G.S.; Souza, G.R.; Silva, J.C.; Saraiva, S.R.G.D.; Junior, R.G.O.; Quintans, J.S.S.; Barreto, R.S.S.; Bonjardim, L.R.; Cavalcanti, S.C.H.; Junior, L.J.Q. Borneol, a bicyclic monoterpene alcohol, reduces nociceptive behavior and inflammatory response in mice. Sci. World J. 2013, 2013, 808460. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.K.; Cho, G.S.; Hwang, S.; Kim, B.W.; Lim, J.H.; Lee, J.C.; Kim, H.C.; Kim, W.K.; Kim, Y.S. Methyleugenol reduces cerebral ischemic injury by suppression of oxidative injury and inflammation. Free Radical. Res. 2010, 44, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Lee, H.J.; Jeon, Y.D.; Han, Y.H.; Kee, J.Y.; Kim, H.J.; Shin, H.J.; Kang, J.W.; Lee, B.S.; Kim, S.H.; et al. Alpha-pinene exhibits anti-inflammatory activity through the suppression of MAPKs and the NF-κB pathway in mouse peritoneal macrophages. Am. J. Chin. Med. 2015, 43, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.T.; Chen, C.J.; Lin, T.Y.; Tung, J.C.; Wang, S.Y. Anti-inflammation activity of fruit essential oil from Cinnamomum insularimontanum Hayata. Bioresour. Technol. 2008, 99, 8783–8787. [Google Scholar] [CrossRef] [PubMed]
- van de Laar, F.A.; Lucassen, P.L.; Akkermans, R.P.; van de Lisdonk, E.H.; Rutten, G.E.; van Weel, C. α-Glucosidase inhibitors for patients with type 2 diabetes: Results from a Cochrane systematic review and meta-analysis. Diabetes Care 2005, 28, 154–163. [Google Scholar] [CrossRef]
- Khan, R.M.M.; Chua, Z.J.Y.; Tan, J.C.; Yang, Y.; Liao, Z.; Zhao, Y. From Pre-Diabetes to Diabetes: Diagnosis, Treatments and Translational Research. Medicina 2019, 55, 546. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Taylor, R.H.; Goff, D.V.; Fielden, H.; Misiewicz, J.J.; Sarson, D.L.; Bloom, S.R.; Alberti, K.G.M.M. Scope and specificity of acarbose in slowing carbohydrate absorption in man. Diabetes 1981, 30, 951–954. [Google Scholar] [CrossRef]
- Oboh, G.; Ogunsuyi, O.B.; Adegbola, D.O.; Ademiluyi, A.O.; Oladun, F.L. Influence of gallic and tannic acid on therapeutic properties of acarbose in vitro and in vivo in Drosophila melanogaster. Biomed. J. 2019, 42, 317–327. [Google Scholar] [CrossRef]
- Basak, S.S.; Candan, F. Effect of Laurus nobilis L. essential oil and its main components on α-glucosidase and reactive oxygen species scavenging activity. Iran. J. Pharm. Res. 2013, 12, 367–379. [Google Scholar]
- Kaewnarin, K.; Rakariyatham, N. Inhibitory effects of phenolic compounds in Ocimum sanctum extract on the α-glucosidase activity and the formation of advanced glycation end-products. Chiang Mai J. Sci. 2017, 44, 203–214. [Google Scholar]
- Chang, T.S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef] [PubMed]
- Masum, M.N.; Yamauchi, K.; Mitsunaga, T. Tyrosinase inhibitors from natural and synthetic sources as skin-lightening agents. Rev. Agric. Sci. 2019, 7, 41–58. [Google Scholar] [CrossRef]
- Grutzendler, J.; Morris, J.C. Cholinesterase inhibitors for Alzheimer’s disease. Drugs 2001, 61, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Perry, N.S.L.; Houghton, P.J.; Theobald, A.; Jenner, P.; Perry, E.K. In-vitro inhibition of human erythrocyte acetylcholinesterase by Salvia lavandulaefolia essential oil and constituent terpenes. J. Pharm. Pharmacol. 2000, 52, 895–902. [Google Scholar] [CrossRef]
- Dohi, S.; Terasaki, M.; Makino, M. Acetylcholinesterase inhibitory activity and chemical composition of commercial essential oils. J. Agr. Food Chem. 2009, 57, 4313–4318. [Google Scholar] [CrossRef]
- de Oliveira, M.S.; da Cruz, J.N.; Silva, S.G.; da Costa, W.A.; de Sousa, S.H.B.; Bezerra, F.W.F.; Teixeira, E.; da Silva, N.J.N.; de Aguiar Andrade, E.H.; de Jesus Chaves Neto, A.M.; et al. Phytochemical profile, antioxidant activity, inhibition of acetylcholinesterase and interaction mechanism of the major components of the Piper divaricatum essential oil obtained by supercritical CO2. J. Supercrit. Fluids 2019, 145, 74–84. [Google Scholar] [CrossRef]
- Tian, M.Y.; Wu, X.H.; Lu, T.Y.; Zhao, X.G.; Wei, F.; Deng, G.D.; Zhou, Y. Phytochemical analysis, antioxidant, antibacterial, cytotoxic, and enzyme inhibitory activities of Hedychium flavum rhizome. Front. Pharmacol. 2020, 11, 572659. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Silva, T.C.; Justino, A.B.; Prado, D.G.; Koch, G.A.; Martins, M.M.; Santos, P.S.; Morais, S.A.L.; Goulart, L.R.; Cunha, L.C.S.; Sousa, R.M.F.; et al. Chemical composition, antioxidant activity and inhibitory capacity of α-amylase, α-glucosidase, lipase and non-enzymatic glycation, in vitro, of the leaves of Cassia bakeriana Craib. Ind. Crops Prod. 2019, 140, 111641. [Google Scholar] [CrossRef]
- Zardi-Bergaoui, A.; Jelizi, S.; Flamini, G.; Ascrizzi, R.; Jannet, B.H. Comparative study of the chemical composition and bioactivities of essential oils of fresh and dry seeds from Myoporum insulare R. Br. Ind. Crops Prod. 2018, 111, 232–237. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherston, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
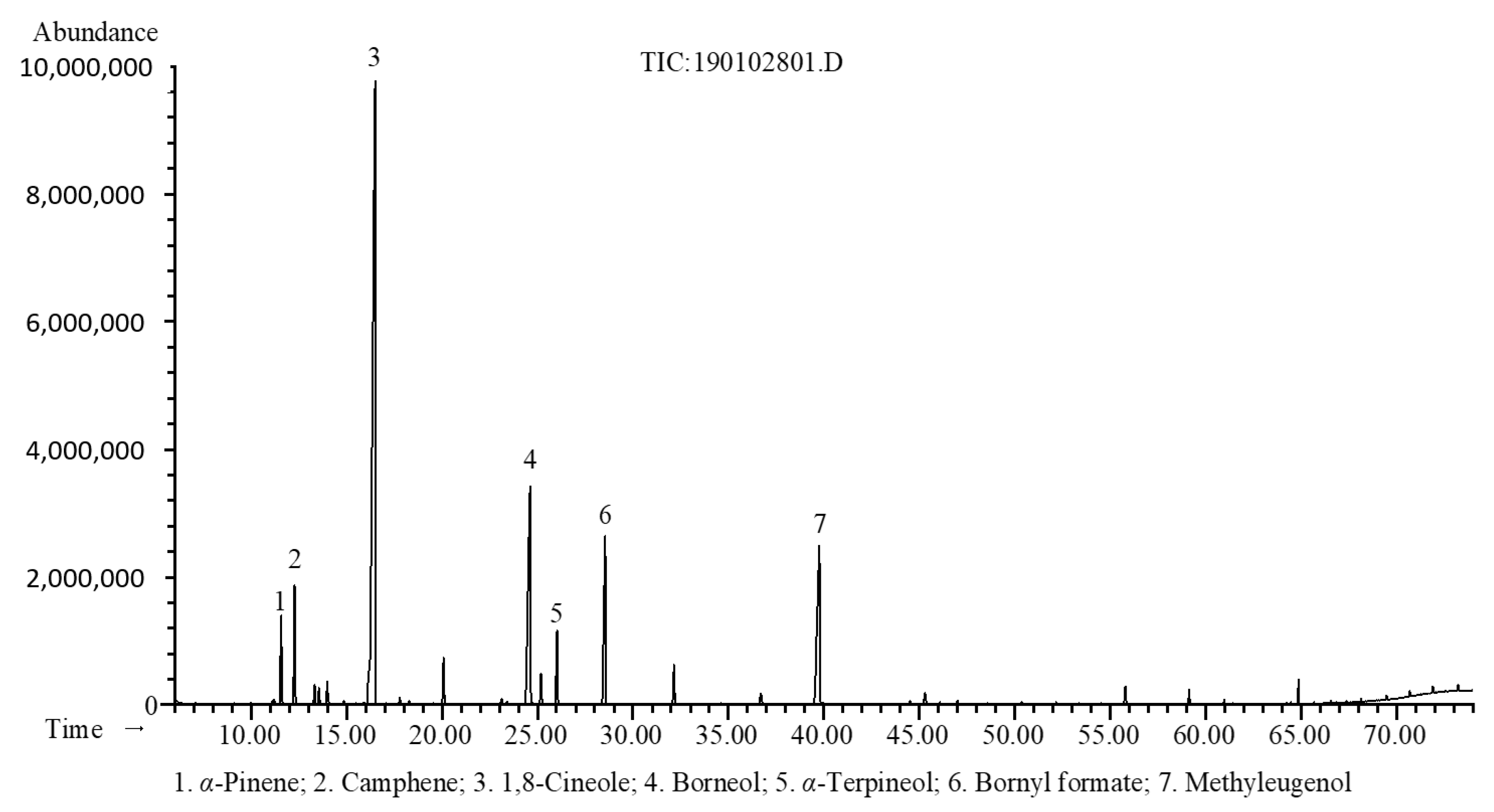

| Compounds a | RI b | RI c | RT (min) d | % Area | Identification e |
|---|---|---|---|---|---|
| Octane | 800 | 800 | 7.076 | 0.1 | MS, RI |
| Tricyclene | 926 | 925 | 11.109 | 0.1 | MS, RI |
| α-Thujene | 928 | 929 | 11.205 | 0.1 | MS, RI |
| α-Pinene | 936 | 937 | 11.575 | 2.5 | MS, RI |
| Camphene | 952 | 952 | 12.275 | 3.4 | MS, RI |
| Sabinene | 976 | 974 | 13.32 | 0.6 | MS, RI |
| β-Pinene | 981 | 979 | 13.559 | 0.5 | MS, RI |
| β-Myrcene | 991 | 991 | 13.991 | 0.7 | MS, RI |
| α-Phellandrene | 1008 | 1005 | 14.844 | 0.1 | MS, RI |
| α-Terpinene | 1020 | 1017 | 15.483 | tr e | MS, RI |
| p-Cymene | 1027 | 1022 | 15.926 | 0.1 | MS, RI |
| 1,8-Cineole | 1038 | 1032 | 16.495 | 47.6 | MS, RI |
| β-Ocimene | 1047 | 1037 | 17.055 | tr e | MS, RI |
| γ-Terpinene | 1061 | 1060 | 17.793 | 0.2 | MS, RI |
| trans-4-Thujanol | 1069 | 1070 | 18.279 | 0.1 | MS, RI |
| Terpinolene | 1092 | 1088 | 19.551 | 0.1 | MS, RI |
| Linalool | 1101 | 1099 | 20.085 | 1.5 | MS, RI |
| (+)-2-Bornanone | 1149 | 1144 | 23.128 | 0.2 | MS, RI |
| Citronellal | 1153 | 1152 | 23.4 | 0.1 | MS, RI |
| Borneol | 1173 | 1167 | 24.616 | 15.0 | MS, RI |
| 4-Terpineol | 1182 | 1177 | 25.187 | 0.9 | MS, RI |
| α-Terpineol | 1195 | 1190 | 26.033 | 2.7 | MS, RI |
| Bornyl formate | 1234 | 1226 | 28.544 | 7.6 | MS, RI |
| Bornyl acetate | 1290 | 1285 | 32.155 | 1.3 | MS, RI |
| Eugenol | 1361 | 1357 | 36.701 | 0.4 | MS, RI |
| Methyleugenol | 1409 | 1402 | 39.765 | 11.2 | MS, RI |
| α-Curcumene | 1487 | 1483 | 44.524 | 0.1 | MS, RI |
| Methylisoeugenol | 1499 | 1495 | 45.296 | 0.5 | MS, RI |
| β-Bisabolene | 1513 | 1509 | 46.073 | 0.1 | MS, RI |
| Sesquicineole | 1519 | 1516 | 46.436 | tr e | MS, RI |
| δ-Cadinene | 1529 | 1524 | 47.007 | 0.1 | MS, RI |
| 2-(3-Isopropenyl-4-methyl-4-vinylcyclohexyl)-2-propanol | 1555 | 1549 | 48.574 | 0.1 | MS, RI |
| (-)-Spathulenol | 1586 | 1577 | 50.354 | 0.1 | MS, RI |
| β-Eudesmol | 1660 | 1649 | 54.505 | 0.1 | MS, RI |
| Ambrial | 1815 | 1809 | 60.969 | 0.1 | MS, RI |
| Total identified | 98.3 |
| Bacterial Strains a | EO | Streptomycin | ||||
|---|---|---|---|---|---|---|
| DIZ b (mm) | MIC c (mg/mL) | MBC c (mg/mL) | DIZ b (mm) | MIC c (μg/mL) | MBC c (μg/mL) | |
| Gram positive | ||||||
| S. aureus | 9.78 ± 0.79 | 6.25 | 6.25 | 18.58 ± 0.41 | 0.78 | 1.56 |
| E. faecalis | 8.66 ± 0.80 | 6.25 | 6.25 | 7.34 ± 0.38 | 12.50 | 25.00 |
| B. subtilis | 10.54 ± 1.25 | 3.13 | 6.25 | 18.05 ± 1.56 | 0.39 | 0.78 |
| Gram negative | ||||||
| E. coli | 9.01 ± 0.59 | 6.25 | 12.50 | 18.71 ± 0.64 | 0.78 | 1.56 |
| P. aeruginosa | 7.29 ± 0.14 | 6.25 | 12.50 | 9.97 ± 0.63 | 3.13 | 12.50 |
| P. vulgaris | 10.56 ± 0.92 | 3.13 | 6.25 | 15.71 ± 0.77 | 0.39 | 1.56 |
| Samples | Enzyme Inhibitory Activity (IC50, mg/mL) 1 | |||
|---|---|---|---|---|
| α-Glucosidase | Tyrosinase | Acetylcholinesterase | Butyrylcholinesterase | |
| EO | 11.60 ± 0.25 a | 53.71 ± 4.89 a | 1.03 ± 0.18 a | 104.22 ± 11.61 a |
| Acarbose | 0.21 ± 0.01 b | |||
| Arbutin | 0.24 ± 0.06 b | |||
| Galanthamine * | 0.46 ± 0.04 b | 5.68 ± 0.41 b | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, X.; Chen, Q.; Lu, T.; Wei, F.; Yang, Y.; Xie, D.; Wang, H.; Tian, M. Chemical Composition, Antibacterial, Anti-Inflammatory, and Enzyme Inhibitory Activities of Essential Oil from Rhynchanthus beesianus Rhizome. Molecules 2021, 26, 167. https://doi.org/10.3390/molecules26010167
Zhao X, Chen Q, Lu T, Wei F, Yang Y, Xie D, Wang H, Tian M. Chemical Composition, Antibacterial, Anti-Inflammatory, and Enzyme Inhibitory Activities of Essential Oil from Rhynchanthus beesianus Rhizome. Molecules. 2021; 26(1):167. https://doi.org/10.3390/molecules26010167
Chicago/Turabian StyleZhao, Xiaoge, Qi Chen, Tingya Lu, Feng Wei, Yao Yang, Dan Xie, Huijuan Wang, and Minyi Tian. 2021. "Chemical Composition, Antibacterial, Anti-Inflammatory, and Enzyme Inhibitory Activities of Essential Oil from Rhynchanthus beesianus Rhizome" Molecules 26, no. 1: 167. https://doi.org/10.3390/molecules26010167
APA StyleZhao, X., Chen, Q., Lu, T., Wei, F., Yang, Y., Xie, D., Wang, H., & Tian, M. (2021). Chemical Composition, Antibacterial, Anti-Inflammatory, and Enzyme Inhibitory Activities of Essential Oil from Rhynchanthus beesianus Rhizome. Molecules, 26(1), 167. https://doi.org/10.3390/molecules26010167





