Lactoferrin and Its Derived Peptides: An Alternative for Combating Virulence Mechanisms Developed by Pathogens
Abstract
1. Introduction
2. Lactoferrin: General Features
2.1. Human and Bovine Lactoferrin
2.2. Lactoferrin-Derived Natural Peptides (Lactoferricins)
2.3. Lactoferrin-Derived Synthetic Peptides
3. Antibacterial Activity of Lactoferrin
3.1. Lactoferrin as a Bacteriostatic and Bactericidal Factor
3.2. Effect of Lactoferrin and Lactoferricins on the Bacterial Membrane Structure and Function
3.3. Effect of Lactoferrin and Lactoferricins on Microbial Biofilms
3.4. Effect of Lactoferrin and Lactoferricins on Bacterial Proteolytic and Oxidative Enzymes
3.5. Effect of Lactoferrin and Lactoferricins on Bacterial Toxins
3.6. Effect of Lf on Bacterial Adherence to Host Surfaces
4. Antiparasitic Activity of Lactoferrin
4.1. Main Treatments against Parasitic Diseases
4.2. Effect of Lactoferrin and Lactoferricins on Parasite Growth/Viability
4.3. Effect of Lactoferrin and Lactoferricins on The Parasite Structure
4.4. Effect of Lactoferrin and Lactoferricins on Parasite Virulence
5. Antifungal Activity of Lactoferrin
5.1. Fungal Diseases
5.2. Iron Requirement in Fungi and The Iron Chelation Effect of Lactoferrin
5.3. Direct or Indirect Interaction of Lactoferrin and Lactoferricins with the Fungal Cell Surface
5.4. Effect of Lactoferrin on Mitochondrial and Caspase-Dependent Regulated Cell Death
5.5. Fungal H+ ATPase (P3A-type) Is a Target of Lactoferrin, which Induces an Apoptosis-Like Process
5.6. Fungal Alteration of Responses to Stress due to Lactoferrin
5.7. Other Unspecified Antifungal Activities of Lactoferrin
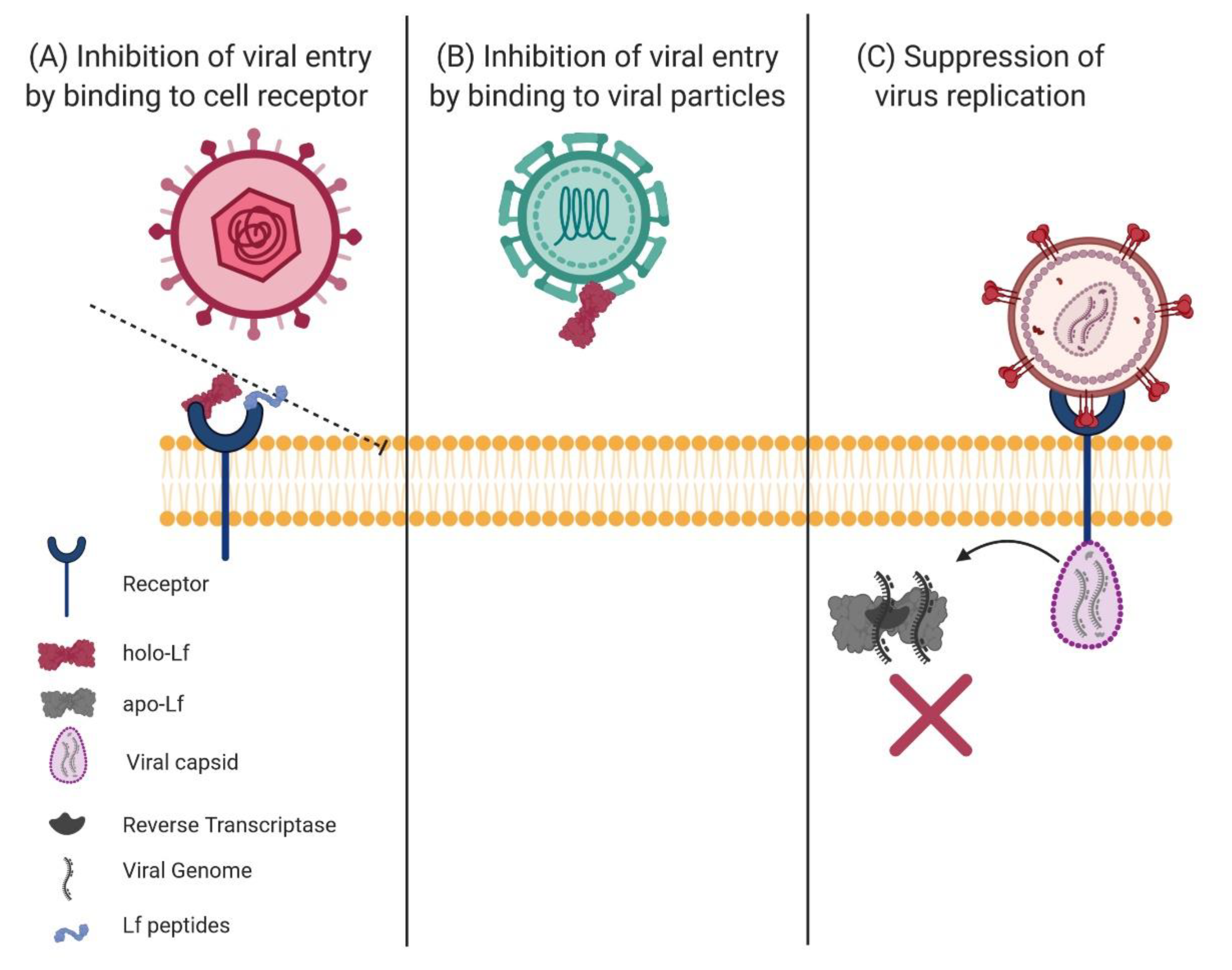
6. Antiviral Effect of Lactoferrin
6.1. Effect of Lactoferrin and Lactoferricins on the Viral Process of Infection
6.2. Binding of Lactoferrin to Target Cell Receptors
6.3. Binding of Lf to Viral Particles
7. Conclusion and Perspectives
Funding
Conflicts of Interest
References
- Frazer, D.M.; Anderson, G.J. The regulation of iron transport. BioFactors 2014, 40, 206–214. [Google Scholar] [CrossRef]
- Guo, S.; Frazer, D.M.; Anderson, G.J. Iron homeostasis: Transport, metabolism, and regulation. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 276–281. [Google Scholar] [CrossRef]
- Anderson, G.J.; Frazer, D.M. Current understanding of iron homeostasis. In Proceedings of the American. J. Clin. Nutr. 2017, 106, 1559S–1566S. [Google Scholar] [CrossRef]
- Ramm, G.A.; Ruddell, R.G. Hepatotoxicity of iron overload: Mechanisms of iron-induced hepatic fibrogenesis. Semin. Liver Dis. 2005, 25, 433–449. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, J.R.; Laakso, H.A.; Heinrichs, D.E. Iron acquisition strategies of bacterial pathogens. Microbiol. Spectr. 2016, 4, 43–85. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Castañeda, A.; Olivares-Trejo, J.J. Ehhmbp45 is a novel hemoglobin-binding protein identified in Entamoeba histolytica. FEBS Lett. 2008, 582, 2806–2810. [Google Scholar] [CrossRef] [PubMed]
- Benz, C.; Lo, W.; Fathallah, N.; Connor-Guscott, A.; Benns, H.J.; Urbaniak, M.D. Dynamic regulation of the Trypanosoma brucei transferrin receptor in response to iron starvation is mediated via the 3′UTR. PLoS ONE 2018, 13, e0206332. [Google Scholar] [CrossRef]
- Peterson, K.M.; Alderete, J.F. Iron uptake and increased intracellular enzyme activity follow host lactoferrin binding by Trichomonas vaginalis receptors. J. Exp. Med. 1984, 160, 398–410. [Google Scholar] [CrossRef]
- Reyes-López, M.; Serrano-Luna, J.D.J.; Negrete-Abascal, E.; León-Sicairos, N.; Guerrero-Barrera, A.L.; De la Garza, M. Entamoeba histolytica: Transferrin binding proteins. Exp. Parasitol. 2001, 99, 132–140. [Google Scholar] [CrossRef]
- López-Soto, F.; González-Robles, A.; Salazar-Villatoro, L.; León-Sicairos, N.; Piña-Vázquez, C.; Salazar, E.P.; de la Garza, M. Entamoeba histolytica uses ferritin as an iron source and internalises this protein by means of clathrin-coated vesicles. Int. J. Parasitol. 2009, 39, 417–426. [Google Scholar] [CrossRef]
- Ortíz-Estrada, G.; Calderón-Salinas, V.; Shibayama-Salas, M.; León-Sicairos, N.; de La Garza, M. Binding and endocytosis of bovine hololactoferrin by the parasite Entamoeba histolytica. Biomed. Res. Int. 2015, 2015, 375836. [Google Scholar] [CrossRef] [PubMed]
- Saha, M.; Sarkar, S.; Sarkar, B.; Sharma, B.K.; Bhattacharjee, S.; Tribedi, P. Microbial siderophores and their potential applications: A review. Environ. Sci. Pollut. Res. 2016, 23, 3984–3999. [Google Scholar] [CrossRef] [PubMed]
- Morgenthau, A.; Pogoutse, A.; Adamiak, P.; Moraes, T.F.; Schryvers, A.B. Bacterial receptors for host transferrin and lactoferrin: Molecular mechanisms and role in host-microbe interactions. Future Microbiol. 2013, 8, 1575–1785. [Google Scholar] [CrossRef] [PubMed]
- Mach, J.; Sutak, R. Iron in parasitic protists—From uptake to storage and where we can interfere. Metallomics 2020, 12, 1335–1347. [Google Scholar] [CrossRef]
- Castro, S.L.; Samaniego-Barron, L.; Serrano-Rubio, L.E.; Olvera, I.C.; Avalos-Gomez, C.; de la Garza, M. Lactoferrin: A powerful antimicrobial protein present in milk. Adv. Dairy Res. 2017, 05, 1–10. [Google Scholar] [CrossRef]
- De Mejia, E.G.; Dia, V.P. The role of nutraceutical proteins and peptides in apoptosis, angiogenesis, and metastasis of cancer cells. Cancer Metast. Rev. 2010, 29, 511–528. [Google Scholar] [CrossRef]
- Actor, J.; Hwang, S.-A.; Kruzel, M. Lactoferrin as a natural immune modulator. Curr. Pharm. Des. 2009, 15, 1956–1973. [Google Scholar] [CrossRef]
- Tsuda, H.; Fukamachi, K.; Xu, J.; Sekine, K.; Ohkubo, S.; Takasuka, N.; Iigo, M. Prevention of carcinogenesis and cancer metastasis by bovine lactoferrin. Proc. Japn. Acad. Ser. B Phys. Biol. Sci. 2006, 82, 208–215. [Google Scholar] [CrossRef]
- Rodrigues, L.; Teixeira, J.; Schmitt, F.; Paulsson, M.; Månsson, H.L. Lactoferrin and cancer disease prevention. Crit. Rev. Food Sci. Nutr. 2009, 49, 203–217. [Google Scholar] [CrossRef]
- Chen, Y.; Zheng, Z.; Zhu, X.; Shi, Y.; Tian, D.; Zhao, F.; Liu, N.; Hüppi, P.S.; Troy, F.A.; Wang, B. Lactoferrin promotes early neurodevelopment and cognition in postnatal piglets by upregulating the BDNF signaling pathway and polysialylation. Mol. Neurobiol. 2015, 52, 256–269. [Google Scholar] [CrossRef]
- Cornish, J. Lactoferrin promotes bone growth. BioMetals 2004, 17, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Lönnerdal, B. Nutritional roles of lactoferrin. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.P.; Paz, E.; Conneely, O.M. Multifunctional roles of lactoferrin: A critical overview. Cell. Mol. Life Sci. 2005, 62, 2540–2548. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Jiang, R.; Lönnerdal, B. Biochemical and molecular impacts of lactoferrin on small intestinal growth and development during early life. Biochem. Cell Biol. 2012, 90, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.A.; Lopez, V.; Lönnerdal, B. Mammalian lactoferrin receptors: Structure and function. Cell. Mol. Life Sci. 2005, 62, 2560–2575. [Google Scholar] [CrossRef]
- Karav, S.; German, J.B.; Rouquié, C.; Le Parc, A.; Barile, D. Studying lactoferrin N-glycosylation. Int. J. Mol. Sci. 2017, 18, 870. [Google Scholar] [CrossRef]
- Le Provost, F.; Nocart, M.; Guerin, G.; Martin, P. Characterization of the goat lactoferrin cDNA: Assignment of the relevant locus to bovine U12 synteny group. Biochem. Biophys. Res. Commun. 1994, 203, 1324–1332. [Google Scholar] [CrossRef]
- Wang, B.; Timilsena, Y.P.; Blanch, E.; Adhikari, B. Lactoferrin: Structure, function, denaturation and digestion. Crit. Rev. Food Sci. Nutr. 2019, 59, 580–596. [Google Scholar] [CrossRef]
- Levay, P.F.; Viljoen, M. Lactoferrin: A general review. Hematologica 1995, 80, 2552–2567. [Google Scholar]
- Cheng, J.B.; Wang, J.Q.; Bu, D.P.; Liu, G.L.; Zhang, C.G.; Wei, H.Y.; Zhou, L.Y.; Wang, J.Z. Factors affecting the lactoferrin concentration in bovine milk. J. Dairy Sci. 2008, 91, 970–976. [Google Scholar] [CrossRef]
- Tsuji, S.; Hirata, Y.; Mukai, F.; Ohtagaki, S. Comparison of lactoferrin content in colostrum between different cattle breeds. J. Dairy Sci. 1990, 73, 125–128. [Google Scholar] [CrossRef]
- Inoue, M.; Yamada, J.; Kitamura, N.; Shimazaki, K.I.; Andrén, A.; Yamashita, T. Immunohistochemical localization of lactoferrin in bovine exocrine glands. Tissue Cell 1993, 25, 791–797. [Google Scholar] [CrossRef]
- Weinberg, E.D. Human lactoferrin: A novel therapeutic with broad spectrum potential. J. Pharm. Pharmacol. 2001, 53, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Rosa, L.; Cutone, A.; Lepanto, M.S.; Paesano, R.; Valenti, P. Lactoferrin: A natural glycoprotein involved in iron and inflammatory homeostasis. Int. J. Mol. Sci. 2017, 18, 1985. [Google Scholar] [CrossRef] [PubMed]
- Drago-Serrano, M.E.; Campos-Rodríguez, R.; Carrero, J.C.; de la Garza, M. Lactoferrin: Balancing ups and downs of inflammation due to microbial infections. Int. J. Mol. Sci. 2017, 18, 501. [Google Scholar] [CrossRef]
- Van Snick, J.L.; Masson, P.L.; Heremans, J.F. The involvement of lactoferrin in the hyposideremia of acute inflammation. J. Exp. Med. 1974, 140, 1068–1084. [Google Scholar] [CrossRef]
- Bennett, R.M.; Kokocinski, T. Lactoferrin content of peripheral blood cells. Br. J. Haematol. 1978, 39, 509–521. [Google Scholar] [CrossRef]
- Baker, E.N.; Baker, H.M. A structural framework for understanding the multifunctional character of lactoferrin. Biochimie 2009, 91, 3–10. [Google Scholar] [CrossRef]
- Sipponen, T.; Savilahti, E.; Kolho, K.L.; Nuutinen, H.; Turunen, U.; Färkkilä, M. Crohn’s disease activity assessed by fecal calprotectin and lactoferrin: Correlation with Crohn’s disease activity index and endoscopic findings. Inflamm. Bowel Dis. 2008, 14, 40–46. [Google Scholar] [CrossRef]
- Groves, M.L. The Isolation of a red protein from milk. J. Am. Chem. Soc. 1960, 82, 3345–3350. [Google Scholar] [CrossRef]
- Montreuil, J.; Tonnelat, J.; Mullet, S. Preparation and properties of the lactosiderophilin (lactotransferrine) of human milk. Biochim. Biophys. Acta 1960, 45, 413–421. [Google Scholar] [CrossRef]
- Moguilevsky, N.; Retegui, L.A.; Masson, P.L. Comparison of human lactoferrins from milk and neutrophilic leucocytes. Relative molecular mass, isoelectric point, iron-binding properties and uptake by the liver. Biochem. J. 1985, 229, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Steijns, J.M.; Van Hooijdonk, A.C.M. Occurrence, structure, biochemical properties and technological characteristics of lactoferrin. Br. J. Nutr. 2000, 84, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Masson, P.L.; Heremans, J.F. Metal-combining properties of human lactoferrin (Red Milk Protein): 1. The involvement of bicarbonate in the reaction. Eur. J. Biochem. 1968, 6, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Testa, U. Proteins of Iron Metabolism; CRC Press: Boca Raton, FL, USA, 2002; ISBN 9780849386763. [Google Scholar]
- Baker, H.M.; Baker, E.N. Lactoferrin and iron: Structural and dynamic aspects of binding and release. BioMetals 2004, 17, 209–216. [Google Scholar] [CrossRef]
- Lönnerdal, B.; Iyer, S. Lactoferrin: Molecular structure and biological function. Annu. Rev. Nutr. 1995, 15, 93–110. [Google Scholar] [CrossRef]
- Jenssen, H.; Hancock, R.E.W. Antimicrobial properties of lactoferrin. Biochimie 2009, 91, 19–29. [Google Scholar] [CrossRef]
- Mirza, S.; Benjamin, W.H.; Coan, P.A.; Hwang, S.A.; Winslett, A.K.; Yother, J.; Hollingshead, S.K.; Fujihashi, K.; Briles, D.E. The effects of differences in pspA alleles and capsular types on the resistance of Streptococcus pneumoniae to killing by apolactoferrin. Microb. Pathog. 2016, 99, 209–219. [Google Scholar] [CrossRef]
- Arnold, R.R.; Russell, J.E.; Champion, W.J.; Brewer, M.; Gauthier, J.J. Bactericidal activity of human lactoferrin: Differentiation from the stasis of iron deprivation. Infect. Immun. 1982, 35, 792–799. [Google Scholar] [CrossRef]
- Salamah, A.A.; al-Obaidi, A.S. Effect of some physical and chemical factors on the bactericidal activity of human lactoferrin and transferrin against Yersinia pseudotuberculosis. New Microbiol. Off. J. Ital. Soc. Med. Odontoiatr. Clin. Microbiol. 1995, 18, 275–281. [Google Scholar]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Lönnerdal, B. Human milk: Bioactive proteins/peptides and functional properties. Nestle Nutr. Inst. Workshop Ser. 2016, 86, 97–107. [Google Scholar] [CrossRef]
- Bellamy, W.; Takase, M.; Wakabayashi, H.; Kawase, K.; Tomita, M. Antibacterial spectrum of lactoferricin B, a potent bactericidal peptide derived from the N-terminal region of bovine lactoferrin. J. Appl. Bacteriol. 1992, 73, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, W.; Takase, M.; Yamauchi, K.; Wakabayashi, H.; Kawase, K.; Tomita, M. Identification of the bactericidal domain of lactoferrin. Biochim. Biophys. Acta (BBA)/Protein Struct. Mol. 1992, 1121, 130–136. [Google Scholar] [CrossRef]
- Elass-Rochard, E.; Roseanu, A.; Legrand, D.; Trif, M.; Salmon, V.; Motas, C.; Montreuil, J.; Spik, G. Lactoferrin-lipopolysaccharide interaction: Involvement of the 28-34 loop region of human lactoferrin in the high-affinity binding to Escherichia coli 055B5 lipopolysaccharide. Biochem. J. 1995, 312, 839–845. [Google Scholar] [CrossRef]
- Van Der Kraan, M.I.A.; Groenink, J.; Nazmi, K.; Veerman, E.C.I.; Bolscher, J.G.M.; Nieuw Amerongen, A.V. Lactoferrampin: A novel antimicrobial peptide in the N1-domain of bovine lactoferrin. Peptides 2004, 25, 177–183. [Google Scholar] [CrossRef]
- Chen, P.W.; Ho, S.P.; Shyu, C.L.; Mao, F.C. Effects of bovine lactoferrin hydrolysate on the in vitro antimicrobial susceptibility of Escherichia coli strains isolated from baby pigs. Am. J. Vet. Res. 2004, 65, 131–137. [Google Scholar] [CrossRef]
- Oo, T.Z.; Cole, N.; Garthwaite, L.; Willcox, M.D.P.; Zhu, H. Evaluation of synergistic activity of bovine lactoferricin with antibiotics in corneal infection. J. Antimicrob. Chemother. 2010, 65, 1243–1251. [Google Scholar] [CrossRef]
- León-Sicairos, N.; Reyes-López, M.; Ordaz-Pichardo, C.; de la Garza, M. Microbicidal action of lactoferrin and lactoferricin and their synergistic effect with metronidazole in Entamoeba histolytica. Biochem. Cell Biol. 2006, 84, 327–336. [Google Scholar] [CrossRef]
- Gifford, J.L.; Hunter, H.N.; Vogel, H.J. Lactoferricin: A lactoferrin-derived peptide with antimicrobial, antiviral, antitumor and immunological properties. Cell. Mol. Life Sci. 2005, 62, 2588–2598. [Google Scholar] [CrossRef]
- Freiburghaus, C.; Janicke, B.; Lindmark-Månsson, H.; Oredsson, S.M.; Paulsson, M.A. Lactoferricin treatment decreases the rate of cell proliferation of a human colon cancer cell line. J. Dairy Sci. 2009, 92, 2477–2484. [Google Scholar] [CrossRef] [PubMed]
- Mader, J.S.; Salsman, J.; Conrad, D.M.; Hoskin, D.W. Bovine lactoferricin selectively induces apoptosis in human leukemia and carcinoma cell lines. Mol. Cancer Ther. 2005, 4, 612–624. [Google Scholar] [CrossRef] [PubMed]
- Vogel, H.J.; Schibli, D.J.; Jing, W.; Lohmeier-Vogel, E.M.; Epand, R.F.; Epand, R.M. Towards a structure-function analysis of bovine lactoferricin and related tryptophan- and arginine-containing peptides. Biochem. Cell Biol. 2002, 80, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.; Wong, J.H.; Ng, T.B. Recent studies on the antimicrobial peptides lactoferricin and lactoferrampin. Curr. Mol. Med. 2014, 14, 1139–1154. [Google Scholar] [CrossRef]
- Odell, E.W.; Sarra, R.; Foxworthy, M.; Chapple, D.S.; Evans, R.W. Antibacterial activity of peptides homologous to a loop region in human lactoferrin. FEBS Lett. 1996, 382, 175–178. [Google Scholar] [CrossRef]
- Bolscher, J.G.M.; Adão, R.; Nazmi, K.; van den Keybus, P.A.M.; van ’t Hof, W.; Nieuw Amerongen, A.V.; Bastos, M.; Veerman, E.C.I. Bactericidal activity of LFchimera is stronger and less sensitive to ionic strength than its constituent lactoferricin and lactoferrampin peptides. Biochimie 2009, 91, 123–132. [Google Scholar] [CrossRef]
- Xu, G.; Xiong, W.; Hu, Q.; Zuo, P.; Shao, B.; Lan, F.; Lu, X.; Xu, Y.; Xiong, S. Lactoferrin-derived peptides and lactoferricin chimera inhibit virulence factor production and biofilm formation in Pseudomonas aeruginosa. J. Appl. Microbiol. 2010, 109, 1311–1318. [Google Scholar] [CrossRef]
- Bolscher, J.; Nazmi, K.; Van Marle, J.; Van ’T Hof, W.; Veerman, E. Chimerization of lactoferricin and lactoferrampin peptides strongly potentiates the killing activity against Candida albicans. Biochem. Cell Biol. 2012, 90, 378–388. [Google Scholar] [CrossRef]
- Acosta-Smith, E.; Viveros-Jiménez, K.; Canizalez-Román, A.; Reyes-Lopez, M.; Bolscher, J.G.M.; Nazmi, K.; Flores-Villaseñor, H.; Alapizco-Castro, G.; de la Garza, M.; Martínez-Garcia, J.J.; et al. Bovine lactoferrin and lactoferrin-derived peptides inhibit the growth of Vibrio cholerae and other Vibrio species. Front. Microbiol. 2018, 8, 2633. [Google Scholar] [CrossRef]
- López-Soto, F.; León-Sicairos, N.; Nazmi, K.; Bolscher, J.G.; De La Garza, M. Microbicidal effect of the lactoferrin peptides Lactoferricin17-30, Lactoferrampin265-284, and Lactoferrin chimera on the parasite Entamoeba histolytica. BioMetals 2010, 23, 563–568. [Google Scholar] [CrossRef]
- Rodriguez, G.M.; Smith, I. Identification of an ABC transporter required for iron acquisition and virulence in Mycobacterium tuberculosis. J. Bacteriol. 2006, 188, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.E.; Rock, J.D.; Ridley, K.A.; Williams, P.H.; Ketley, J.M. Utilization of lactoferrin-bound and transferrin-bound iron by Campylobacter jejuni. J. Bacteriol. 2008, 190, 1900–1911. [Google Scholar] [CrossRef] [PubMed]
- Lisiecki, P. Transferrin and lactoferrin-human iron sources for enterococci. Polish J. Microbiol. 2017, 66, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Samaniego, B.L.; Luna, C.S.; Piña, V.C.; Suárez, G.F.; De La Garza, M. Two outer membrane proteins are bovine lactoferrin-binding proteins in Mannheimia haemolytica A1. Vet. Res. 2016, 47, 93. [Google Scholar] [CrossRef]
- Legrand, D.; Elass, E.; Carpentier, M.; Mazurier, J. Lactoferrin. Cell. Mol. Life Sci. 2005, 62, 2549–2559. [Google Scholar] [CrossRef]
- Appelmelk, B.J.; An, Y.Q.; Geerts, M.; Thijs, B.G.; de Boer, H.A.; MacLaren, D.M.; de Graaff, J.; Nuijens, J.H. Lactoferrin is a lipid A-binding protein. Infect. Immun. 1994, 62, 2628–2632. [Google Scholar] [CrossRef]
- Sallmann, F.R.; Baveye-Descamps, S.; Pattus, F.; Salmon, V.; Branza, N.; Spik, G.; Legrand, D. Porins OmpC and PhoE of Escherichia coli as specific cell-surface targets of human lactoferrin. J. Biol. Chem. 1999, 274, 16107–16114. [Google Scholar] [CrossRef]
- Vorland, L.H.; Ulvatne, H.; Rekdal, Ø.; Svendsen, J.S. Initial binding sites of antimicrobial peptides in Staphylococcus aureus and Escherichia coli. Scand. J. Infect. Dis. 1999, 31, 467–473. [Google Scholar] [CrossRef]
- Ellison, R.T.; LaForce, F.M.; Giehl, T.J.; Boose, D.S.; Dunn, B.E. Lactoferrin and transferrin damage of the Gram-negative outer membrane is modulated by Ca2+ and Mg2+. J. Gen. Microbiol. 1990, 136, 1437–1446. [Google Scholar] [CrossRef]
- Flores-Villaseñor, H.; Canizalez-Román, A.; Reyes-Lopez, M.; Nazmi, K.; de la Garza, M.; Zazueta-Beltrán, J.; León-Sicairos, N.; Bolscher, J.G.M. Bactericidal effect of bovine lactoferrin, LFcin, LFampin and LFchimera on antibiotic-resistant Staphylococcus aureus and Escherichia coli. BioMetals 2010, 23, 569–578. [Google Scholar] [CrossRef]
- Vorland, L.H. Lactoferrin: A multifunctional glycoprotein. APMIS 1999, 107, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Albar, A.; Almehdar, H.; Uversky, V.; Redwan, E. Structural Heterogeneity and Multifunctionality of Lactoferrin. Curr. Protein Pept. Sci. 2014, 15, 778–797. [Google Scholar] [CrossRef] [PubMed]
- Drago-Serrano, M.E.; de la Garza, M.; Luna, J.S.; Campos-Rodríguez, R. Lactoferrin-lipopolysaccharide (LPS) binding as key to antibacterial and antiendotoxic effects. Int. Immunopharmacol. 2012, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, X.; Hao, Y.; Teng, D.; Wang, J. Research and development on lactoferrin and its derivatives in China from 2011-2015. Biochem. Cell Biol. 2017, 95, 162–170. [Google Scholar] [CrossRef]
- Yamauchi, K.; Tomita, M.; Giehl, T.J.; Ellison, R.T. Antibacterial activity of lactoferrin and a pepsin-derived lactoferrin peptide fragment. Infect. Immun. 1993, 61, 719–728. [Google Scholar] [CrossRef]
- Van der Kraan, M.I.A.; van Marle, J.; Nazmi, K.; Groenink, J.; van’t Hof, W.; Veerman, E.C.I.; Bolscher, J.G.M.; Nieuw Amerongen, A.V. Ultrastructural effects of antimicrobial peptides from bovine lactoferrin on the membranes of Candida albicans and Escherichia coli. Peptides 2005, 26, 1537–1542. [Google Scholar] [CrossRef]
- León-Sicairos, N.; Angulo-Zamudio, U.A.; Vidal, J.E.; López-Torres, C.A.; Bolscher, J.G.M.; Nazmi, K.; Reyes-Cortes, R.; Reyes-López, M.; de la Garza, M.; Canizalez-Román, A. Bactericidal effect of bovine lactoferrin and synthetic peptide lactoferrin chimera in Streptococcus pneumoniae and the decrease in luxS gene expression by lactoferrin. BioMetals 2014, 27, 969–980. [Google Scholar] [CrossRef]
- Avalos-Gómez, C.; Reyes-López, M.; Ramírez-Rico, G.; Díaz-Aparicio, E.; Zenteno, E.; González-Ruiz, C.; De La Garza, M. Effect of apo-lactoferrin on leukotoxin and outer membrane vesicles of Mannheimia haemolytica A2. Vet. Res. 2020, 51, 36. [Google Scholar] [CrossRef]
- Hossain, F.; Moghal, M.M.R.; Islam, M.Z.; Moniruzzaman, M.; Yamazaki, M. Membrane potential is vital for rapid permeabilization of plasma membranes and lipid bilayers by the antimicrobial peptide lactoferricin B. J. Biol. Chem. 2019, 294, 10449–10462. [Google Scholar] [CrossRef]
- Leon-Sicairos, N.; Canizalez-Roman, A.; de la Garza, M.; Reyes-Lopez, M.; Zazueta-Beltran, J.; Nazmi, K.; Gomez-Gil, B.; Bolscher, J.G. Bactericidal effect of lactoferrin and lactoferrin chimera against halophilic Vibrio parahaemolyticus. Biochimie 2009, 91, 133–140. [Google Scholar] [CrossRef]
- Veerachamy, S.; Yarlagadda, T.; Manivasagam, G.; Yarlagadda, P.K. Bacterial adherence and biofilm formation on medical implants: A review. Proc. Inst. Mech. Eng. H 2014, 228, 1083–1099. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, J. Biofilms and antibiotic resistance: A genetic linkage. Trends Microbiol. 2002, 10, 264. [Google Scholar] [CrossRef]
- Nam, H.; Seo, H.S.; Bang, J.; Kim, H.; Beuchat, L.R.; Ryu, J.H. Efficacy of gaseous chlorine dioxide in inactivating Bacillus cereus spores attached to and in a biofilm on stainless steel. Int. J. Food Microbiol. 2014, 188, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, A.L.S.; Galdino, A.C.M.; de Mello, T.P.; de Ramos, L.S.; Branquinha, M.H.; Bolognese, A.M.; Neto, J.C.; Roudbary, M. What are the advantages of living in a community? A microbial biofilm perspective! Mem. Inst. Oswaldo Cruz 2018, 113, e180212. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Sarkar, S.; Das, B.; Bhattacharjee, S.; Tribedi, P. Biofilm, pathogenesis and prevention—A journey to break the wall: A review. Arch. Microbiol. 2016, 198, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.P.; Melo, L.D.R.; Vilas Boas, D.; Sillankorva, S.; Azeredo, J. Phage therapy as an alternative or complementary strategy to prevent and control biofilm-related infections. Curr. Opin. Microbiol. 2017, 39, 48–56. [Google Scholar] [CrossRef]
- Doolittle, M.M.; Cooney, J.J.; De Caldwell, D.E. Tracing the interaction of bacteriophage with bacterial biofilms using fluorescent and chromogenic probes. J. Ind. Microbiol. 1996, 16, 331–341. [Google Scholar] [CrossRef]
- Chen, M.J.; Zhang, Z.; Bott, T.R. Direct measurement of the adhesive strength of biofilms in pipes by micromanipulation. Biotechnol. Tech. 1998, 12, 875–880. [Google Scholar] [CrossRef]
- Stewart, P.S.; Costerton, J.W. Antibiotic resistance of bacteria in biofilms. Lancet 2001, 358, 135–138. [Google Scholar] [CrossRef]
- Ammons, M.C.B.; Ward, L.S.; Fisher, S.T.; Wolcott, R.D.; James, G.A. In Vitro susceptibility of established biofilms composed of a clinical wound isolate of Pseudomonas aeruginosa treated with lactoferrin and xylitol. Int. J. Antimicrob. Agents 2009, 33, 230–236. [Google Scholar] [CrossRef]
- Sheffield, C.L.; Crippen, T.L.; Poole, T.L. Destruction of single-species biofilms of Escherichia coli or Klebsiella pneumoniae subsp. pneumoniae by dextranase, lactoferrin, and lysozyme. Int. Microbiol. 2012, 15, 15. [Google Scholar] [CrossRef]
- Angulo-Zamudio, U.A.; Vidal, J.E.; Nazmi, K.; Bolscher, J.G.M.; Leon-Sicairos, C.; Antezana, B.S.; Canizalez-Roman, A.; León-Sicairos, N. Lactoferrin disaggregates pneumococcal biofilms and inhibits acquisition of resistance through its DNase activity. Front. Microbiol. 2019, 10, 2386. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, H.; Yamauchi, K.; Kobayashi, T.; Yaeshima, T.; Iwatsuki, K.; Yoshie, H. Inhibitory effects of lactoferrin on growth and biofilm formation of Porphyromonas gingivalis and Prevotella intermedia. Antimicrob. Agents Chemother. 2009, 53, 3308–3316. [Google Scholar] [CrossRef] [PubMed]
- Elkins, J.G.; Hassett, D.J.; Stewart, P.S.; Schweizer, H.P.; McDermott, T.R. Protective role of catalase in Pseudomonas aeruginosa biofilm resistance to hydrogen peroxide. Appl. Environ. Microbiol. 1999, 65, 4594–4600. [Google Scholar] [CrossRef]
- Sánchez-Gómez, S.; Ferrer-Espada, R.; Stewart, P.S.; Pitts, B.; Lohner, K.; Martínez De Tejada, G. Antimicrobial activity of synthetic cationic peptides and lipopeptides derived from human lactoferricin against Pseudomonas aeruginosa planktonic cultures and biofilms. BMC Microbiol. 2015, 15, 137. [Google Scholar] [CrossRef]
- De la Fuente-Núñez, C.; Reffuveille, F.; Haney, E.F.; Straus, S.K.; Hancock, R.E.W. Broad-spectrum anti-biofilm peptide that targets a cellular stress response. PLoS Pathog. 2014, 10. [Google Scholar] [CrossRef]
- Dashper, S.G.; Pan, Y.; Veith, P.D.; Chen, Y.Y.; Toh, E.C.Y.; Liu, S.W.; Cross, K.J.; Reynolds, E.C. Lactoferrin inhibits Porphyromonas gingivalis proteinases and has sustained biofilm inhibitory activity. Antimicrob. Agents Chemother. 2012, 56, 1548–1556. [Google Scholar] [CrossRef]
- Berlutti, F.; Ajello, M.; Bosso, P.; Morea, C.; Petrucca, A.; Antonini, G.; Valenti, P. Both lactoferrin and iron influence aggregation and biofilm formation in Streptococcus mutans. BioMetals 2004, 17, 271–278. [Google Scholar] [CrossRef]
- Quintieri, L.; Caputo, L.; Monaci, L.; Deserio, D.; Morea, M.; Baruzzi, F. Antimicrobial efficacy of pepsin-digested bovine lactoferrin on spoilage bacteria contaminating traditional Mozzarella cheese. Food Microbiol. 2012, 31, 64–71. [Google Scholar] [CrossRef]
- Rossi, C.; Serio, A.; Chaves-López, C.; Anniballi, F.; Auricchio, B.; Goffredo, E.; Cenci-Goga, B.T.; Lista, F.; Fillo, S.; Paparella, A. Biofilm formation, pigment production and motility in Pseudomonas spp. isolated from the dairy industry. Food Control 2018, 86, 241–248. [Google Scholar] [CrossRef]
- Quintieri, L.; Zühlke, D.; Fanelli, F.; Caputo, L.; Liuzzi, V.C.; Logrieco, A.F.; Hirschfeld, C.; Becher, D.; Riedel, K. Proteomic analysis of the food spoiler Pseudomonas fluorescens ITEM 17298 reveals the antibiofilm activity of the pepsin-digested bovine lactoferrin. Food Microbiol. 2019, 82, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Hendrixson, D.R.; Baker, E.N.; Murphy, T.F.; St Geme, J.W.; Plaut, A.G. Human milk lactoferrin inactivates two putative colonization factors expressed by Haemophilus influenzae. Proc. Natl. Acad. Sci. USA 1998, 95, 12641–12646. [Google Scholar] [CrossRef] [PubMed]
- Hendrixson, D.R.; Qiu, J.; Shewry, S.C.; Fink, D.L.; Petty, S.; Baker, E.N.; Plaut, A.G.; St. Geme, J.W. Human milk lactoferrin is a serine protease that cleaves Haemophilus surface proteins at arginine-rich sites. Mol. Microbiol. 2003, 47, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Lau, G.W.; Hassett, D.J.; Ran, H.; Kong, F. The role of pyocyanin in Pseudomonas aeruginosa infection. Trends Mol. Med. 2004, 10, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Kruzel, M.L.; Actor, J.K.; Radak, Z.; Bacsi, A.; Saavedra-Molina, A.; Boldogh, I. Lactoferrin decreases LPS-induced mitochondrial dysfunction in cultured cells and in animal endotoxemia model. Innate Immun. 2010, 16, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Melton-Celsa, A.R. Shiga toxin (Stx) classification, structure, and function. Microbiol. Spectr. 2014, 2, 2. [Google Scholar] [CrossRef]
- Fuller, C.A.; Pellino, C.A.; Flagler, M.J.; Strasser, J.E.; Weiss, A.A. Shiga toxin subtypes display dramatic differences in potency. Infect. Immun. 2011, 79, 1329–1337. [Google Scholar] [CrossRef]
- Kieckens, E.; Rybarczyk, J.; Barth, S.A.; Menge, C.; Cox, E.; Vanrompay, D. Effect of lactoferrin on release and bioactivity of Shiga toxins from different Escherichia coli O157:H7 strains. Vet. Microbiol. 2017, 202, 29–37. [Google Scholar] [CrossRef]
- Asadi, A.; Razavi, S.; Talebi, M.; Gholami, M. A review on anti-adhesion therapies of bacterial diseases. Infection 2019, 47, 13–23. [Google Scholar] [CrossRef]
- De Araújo, A.N.; Gimenes Giugliano, L. Lactoferrin and free secretory component of human milk inhibit the adhesion of enteropathogenic Escherichia coli to HeLa cells. BMC Microbiol. 2001, 1, 1–6. [Google Scholar] [CrossRef]
- Alugupalli, K.R.; Kalfas, S. Inhibitory effect of lactoferrin on the adhesion of Actinobacillus actinomycetemcomitans and Prevotella intermedia to fibroblasts and epithelial cells. APMIS 1995, 103, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Luna-Castro, S.; Aguilar-Romero, F.; Samaniego-Barrón, L.; Godínez-Vargas, D.; de La Garza, M. Effect of bovine apo-lactoferrin on the growth and virulence of Actinobacillus pleuropneumoniae. BioMetals 2014, 27, 891–903. [Google Scholar] [CrossRef] [PubMed]
- Di Biase, A.M.; Tinari, A.; Pietrantoni, A.; Antonini, G.; Valenti, F.; Conte, M.P.; Superti, F. Effect of bovine lactoferricin on enteropathogenic Yersinia adhesion and invasion in HEp-2 cells. J. Med. Microbiol. 2004, 53, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://biorender.com/.
- Gunn, A.; Pitt, S.J. Parasitology An Integrated Approach; Wiley-Blackwell: Hoboken, NJ, USA, 2012; ISBN 9780470684245. [Google Scholar]
- Moonah, S.N.; Jiang, N.M.; Petri, W.A.J. Host Immune response to intestinal amebiasis. PLoS Pathog. 2013, 9, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Baig, A.M. Pathogenesis of amoebic encephalitis: Are the amoebas being credited to an “inside job” done by the host immune response? Acta Trop. 2015, 148, 72–76. [Google Scholar] [CrossRef]
- Leitsch, D.; Williams, C.F.; Hrdý, I. Redox pathways as drug targets in microaerophilic parasites. Trends Parasitol. 2018, 34, 576–589. [Google Scholar] [CrossRef]
- Roe, F.J.C. Metronidazole: View of uses and toxicity. J. Antimicrob. Chemother. 1977, 3, 205–212. [Google Scholar] [CrossRef]
- Dingsdag, S.A.; Hunter, N. Metronidazole: An update on metabolism, structure-cytotoxicity and resistance mechanisms. J. Antimicrob. Chemother. 2018, 73, 265–279. [Google Scholar] [CrossRef]
- Hamill, R.J. Amphotericin B formulations: A comparative review of efficacy and toxicity. Drugs 2013, 73, 919–934. [Google Scholar] [CrossRef]
- Plantone, D.; Koudriavtseva, T. Current and future use of chloroquine and hydroxychloroquine in infectious, immune, neoplastic, and neurological diseases: A mini-review. Clin. Drug Investig. 2018, 38, 653–671. [Google Scholar] [CrossRef]
- Chatre, C.; Roubille, F.; Vernhet, H.; Jorgensen, C.; Pers, Y.M. Cardiac complications attributed to chloroquine and hydroxychloroquine: A systematic review of the literature. Drug Saf. 2018, 41, 919–931. [Google Scholar] [CrossRef] [PubMed]
- Price, R.N.; von Seidlein, L.; Valecha, N.; Nosten, F.; Baird, J.K.; White, N.J. Global extent of chloroquine-resistant Plasmodium vivax: A systematic review and meta-analysis. Lancet Infect. Dis. 2014, 14, 982–991. [Google Scholar] [CrossRef]
- Foley, M.; Tilley, L. Quinoline antimalarials: Mechanisms of action and resistance and prospects for new agents. Pharmacol. Ther. 1998, 79, 55–87. [Google Scholar] [CrossRef]
- Ravdin, J.I.; Guerrant, R.L. Role of adherence in cytopathogenic mechanisms of Entamoeba histolytica. Study with mammalian tissue culture cells and human erythrocytes. J. Clin. Investig. 1981, 68, 1305–1313. [Google Scholar] [CrossRef]
- Tsutsumi, V.; Ramírez-Rosales, A.; Lanz-Mendoza, H.; Shibayama, M.; Chávez, B.; Rangel-López, E.; Martínez-Palomo, A. Entamoeba histolytica: Erythrophagocytosis, collagenolysis, and liver abscess production as virulence markers. Trans. R. Soc. Trop. Med. Hyg. 1992, 86, 170–172. [Google Scholar] [CrossRef]
- Latour, N.G.; Reeves, R.E. An iron-requirement for growth of Entamoeba histolytica in culture, and the antiamebal activity of 7-iodo-8-hydroxy-quinoline-5-sulfonic acid. Exp. Parasitol. 1965, 17, 203–209. [Google Scholar] [CrossRef]
- Tachezy, J.; Kulda, J.; Bahníková, I.; Suchan, P.; Rázga, J.; Schrével, J. Tritrichomonas foetus: Iron acquisition from lactoferrin and transferrin. Exp. Parasitol. 1996, 83, 216–228. [Google Scholar] [CrossRef]
- León-Sicairos, N.; López-Soto, F.; Reyes-López, M.; Godínez-Vargas, D.; Ordaz-Pichardo, C.; de la Garza, M. Amoebicidal Activity of Milk, Apo-lactoferrin, sIgA and lysozyme. Clin. Med. Res. 2006, 4, 106–113. [Google Scholar] [CrossRef]
- Tomita, S.; Suzuki, C.; Wada, H.; Nomachi, M.; Imayasu, M.; Araki-Sasaki, K. Effects of lactoferrin on the viability and the encystment of Acanthamoeba trophozoites. Biochem. Cell Biol. 2017, 95, 48–52. [Google Scholar] [CrossRef]
- Frontera, L.S.; Moyano, S.; Quassollo, G.; Lanfredi-Rangel, A.; Rópolo, A.S.; Touz, M.C. Lactoferrin and lactoferricin endocytosis halt Giardia cell growth and prevent infective cyst production. Sci. Rep. 2018, 8, 18020. [Google Scholar] [CrossRef]
- Aguilar-Diaz, H.; Canizalez-Roman, A.; Nepomuceno-Mejia, T.; Gallardo-Vera, F.; Hornelas-Orozco, Y.; Nazmi, K.; Bolscher, J.G.M.; Carrero, J.C.; Leon-Sicairos, C.; Leon-Sicairos, N. Parasiticidal effect of synthetic bovine lactoferrin peptides on the enteric parasite Giardia intestinalis. Biochem. Cell Biol. 2016, 95, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Godínez, C.; González-Galindo, X.; Meza-Menchaca, T.; Bobes, R.J.; de la Garza, M.; León-Sicairos, N.; Laclette, J.P.; Carrero, J.C. Synthetic bovine lactoferrin peptide Lfampin kills Entamoeba histolytica trophozoites by necrosis and resolves amoebic intracecal infection in mice. Biosci. Rep. 2019, 39, BSR20180850. [Google Scholar] [CrossRef] [PubMed]
- Paredes, J.L.; Sparks, H.; White, A.C.; Martinez-Traverso, G.; Ochoa, T.; Castellanos-González, A. Killing of cryptosporidium sporozoites by lactoferrin. Am. J. Trop. Med. Hyg. 2017, 97, 774–776. [Google Scholar] [CrossRef] [PubMed]
- Bruni, N.; Capucchio, M.T.; Biasibetti, E.; Pessione, E.; Cirrincione, S.; Giraudo, L.; Corona, A.; Dosio, F. Antimicrobial activity of lactoferrin-related peptides and applications in human and veterinary medicine. Molecules 2016, 21, 752. [Google Scholar] [CrossRef]
- Turchany, J.M.; Mccaffery, J.M.; Aley, S.B.; Gillin, F.D. Ultrastructural effects of lactoferring binding on Giardia lamblia trophozoites. J. Eukaryot. Microbiol. 1997, 44, 68–72. [Google Scholar] [CrossRef]
- Dzitko, K.; Dziadek, B.; Dziadek, J.; Długońska, H. Toxoplasma gondii: Inhibition of the intracellular growth by human lactoferrin. Polish J. Microbiol. 2007, 56, 25–32. [Google Scholar]
- Tanaka, T.; Omata, Y.; Narisawa, M.; Saito, A.; Shimazaki, K.; Igarashi, I.; Hirumi, H.; Suzuki, N. Growth inhibitory effect of bovine lactoferrin on Toxoplasma gondii tachyzoites in murine macrophages: Role of radical oxygen and inorganic nitrogen oxide in Toxoplasma growth-inhibitory activity. Vet. Parasitol. 1997, 68, 27–33. [Google Scholar] [CrossRef]
- Tanaka, T.; Omata, Y.; Isamida, T.; Saito, A.; Shimazaki, K.; Yamauchi, K.; Suzuki, N. Growth inhibitory effect of bovine lactoferrin to Toxoplasma gondii tachyzoites in murine macrophages: Tyrosine phosphorylation in murine macrophages induced by bovine lactoferrin. J. Vet. Med. Sci. 1998, 60, 369–371. [Google Scholar] [CrossRef][Green Version]
- Lima, M.F.; Kierszenbaum, F. Lactoferrin effects on phagocytic cell function. I. Increased uptake and killing of an intracellular parasite by murine macrophages and human monocytes. J. Immunol. 1985, 134, 4176–4183. [Google Scholar]
- Gastelum-Martínez, A.; León-Sicairos, C.; Plata-Guzmán, L.; Soto-Castro, L.; León-Sicairos, N.; de la Garza, M. Iron-modulated virulence factors of Entamoeba histolytica. Future Microbiol. 2018, 13, 1329–1341. [Google Scholar] [CrossRef]
- Park, S.J.; Lee, S.M.; Lee, J.; Yong, T.S. Differential gene expression by iron-limitation in Entamoeba histolytica. Mol. Biochem. Parasitol. 2001, 114, 257–260. [Google Scholar] [CrossRef]
- Arantes, J.M.; Pedrosa, M.L.; Martins, H.R.; Veloso, V.M.; de Lana, M.; Bahia, M.T.; Tafuri, W.L.; Carneiro, C.M. Trypanosoma cruzi: Treatment with the iron chelator desferrioxamine reduces parasitemia and mortality in experimentally infected mice. Exp. Parasitol. 2007, 117, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Lehker, M.W.; Arroyo, R.; Alderete, J.F. The regulation by iron of the synthesis of adhesins and cytoadherence levels in the protozoan Trichomonas vaginalis. J. Exp. Med. 1991, 174, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.S.; Choi, H.K.; Min, D.Y.; Ha, S.E.; Ahn, M.H. Effect of iron on the virulence of Trichomonas vaginalis. J. Parasitol. 2001, 87, 457. [Google Scholar] [CrossRef]
- Das, S.; Stevens, T.; Castillo, C.; Villasenõr, A.; Arredondo, H.; Reddy, K. Lipid metabolism in mucous-dwelling amitochondriate protozoa. Int. J. Parasitol. 2002, 32, 655–675. [Google Scholar] [CrossRef]
- Goldston, A.M.; Powell, R.R.; Temesvari, L.A. Sink or swim: Lipid rafts in parasite pathogenesis. Trends Parasitol. 2012, 28, 417–426. [Google Scholar] [CrossRef]
- Castellanos-Castro, S.; Bolaños, J.; Orozco, E. Lipids in Entamoeba histolytica: Host-dependence and virulence factors. Front. Cell. Infect. Microbiol. 2020, 10, 1–16. [Google Scholar] [CrossRef]
- Welter, B.H.; Goldston, A.M.; Temesvari, L.A. Localization to lipid rafts correlates with increased function of the Gal/GalNAc lectin in the human protozoan parasite, Entamoeba histolytica. Int. J. Parasitol. 2011, 41, 1409–1419. [Google Scholar] [CrossRef]
- Humen, M.A.; Pérez, P.F.; Liévin-Le Moal, V. Lipid raft-dependent adhesion of Giardia intestinalis trophozoites to a cultured human enterocyte-like Caco-2/TC7 cell monolayer leads to cytoskeleton-dependent functional injuries. Cell. Microbiol. 2011, 13, 1683–1702. [Google Scholar] [CrossRef]
- Manque, P.M.; Eichinger, D.; Juliano, M.A.; Juliano, L.; Araya, J.E.; Yoshida, N. Characterization of the cell adhesion site of Trypanosoma cruzi metacyclic stage surface glycoprotein gp82. Infect. Immun. 2000, 68, 478–484. [Google Scholar] [CrossRef]
- Ruiz, R.C.; Favoreto, S.; Dorta, M.L.; Oshiro, M.E.M.; Ferreira, A.T.; Manque, P.M.; Yoshida, N. Infectivity of Trypanosoma cruzi strains is associated with differential expression of surface glycoproteins with differential Ca2+ signalling activity. Biochem. J. 1998, 330, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Sinnis, P.; Willnow, T.E.; Briones, M.R.S.; Herz, J.; Nussenzweig, V. Remnant lipoproteins inhibit malaria sporozoite invasion of hepatocytes. J. Exp. Med. 1996, 184, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Fritsch, G.; Sawatzki, G.; Treumer, J.; Jung, A.; Spira, D.T. Plasmodium falciparum: Inhibition in vitro with lactoferrin, desferriferrithiocin, and desferricrocin. Exp. Parasitol. 1987, 63, 1–9. [Google Scholar] [CrossRef]
- Dvořák, J.; Mashiyama, S.T.; Braschi, S.; Sajid, M.; Knudsen, G.M.; Hansell, E.; Lim, K.C.; Hsieh, I.; Bahgat, M.; Mackenzie, B.; et al. Differential use of protease families for invasion by schistosome cercariae. Biochimie 2008, 90, 345–358. [Google Scholar] [CrossRef] [PubMed]
- McKerrow, J.H.; Salter, J. Invasion of skin by Schistosoma cercariae. Trends Parasitol. 2002, 18, 193–195. [Google Scholar] [CrossRef]
- Satoskar, A.R.; Simon, G.L.; Hotez, P.J.; Tsuji, M. Medical Parasitology; Landes Bioscience: Austin, TX, USA, 2009; ISBN 9781498713672. [Google Scholar]
- Loukas, A.; Hotez, P.J.; Diemert, D.; Yazdanbakhsh, M.; McCarthy, J.S.; Correa-Oliveira, R.; Croese, J.; Bethony, J.M. Hookworm infection. Nat. Rev. Dis. Prim. 2016, 2, 16088. [Google Scholar] [CrossRef]
- Serrano-Luna, J.; Piña-Vázquez, C.; Reyes-López, M.; Ortiz-Estrada, G.; de la Garza, M. Proteases from Entamoeba spp. and pathogenic free-living amoebae as virulence factors. J. Trop. Med. 2013, 2013, 890603. [Google Scholar] [CrossRef]
- Marie, C.; Petri, W.A. Regulation of Virulence of Entamoeba histolytica. Annu. Rev. Microbiol. 2014, 68, 493–520. [Google Scholar] [CrossRef]
- Hernández-Nava, E.; Cuellar, P.; Nava, P.; Chávez-Munguía, B.; Schnoor, M.; Orozco, E.; Betanzos, A. Adherens junctions and desmosomes are damaged by Entamoeba histolytica: Participation of EhCPADH complex and EhCP112 protease. Cell. Microbiol. 2017, 19, e12761. [Google Scholar] [CrossRef]
- Sehgal, R.; Goyal, K.; Sehgal, A. Trichomoniasis and lactoferrin: Future prospects. Infect. Dis. Obstet. Gynecol. 2012, 2012, 536037. [Google Scholar] [CrossRef]
- Alderete, J.F.; Provenzano, D.; Lehker, M.W. Iron mediates Trichomonas vaginalis resistance to complement lysis. Microb. Pathog. 1995, 19, 93–103. [Google Scholar] [CrossRef] [PubMed]
- León-Sicairos, N.; Martínez-Pardo, L.; Sánchez-Hernández, B.; de la Garza, M.; Carrero, J.C. Oral lactoferrin treatment resolves amoebic intracecal infection in C3H/HeJ mice. Biochem. Cell Biol. 2012, 90, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Ordaz-Pichardo, C.; León-Sicairos, N.; Hernández-Ramírez, V.I.; Talamás-Rohana, P.; de la Garza, M. Effect of bovine lactoferrin in a therapeutic hamster model of hepatic amoebiasis. Biochem. Cell Biol. 2012, 90, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Omata, Y.; Satake, M.; Maeda, R.; Saito, A.; Shimazaki, K.; Yamauchi, K.; Uzuka, Y.; Tanabe, S.; Sarashina, T.; Mikami, T. Reduction of the infectivity of Toxoplasma gondii and Eimeria stiedai sporozoites by treatment with bovine lactoferricin. J. Vet. Med. Sci. 2001, 63, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Alimi, D.; Hajaji, S.; Rekik, M.; Abidi, A.; Gharbi, M.; Akkari, H. First report of the in vitro nematicidal effects of camel milk. Vet. Parasitol. 2016, 228, 153–159. [Google Scholar] [CrossRef]
- Anand, N.; Kanwar, R.K.; Sehgal, R.; Kanwar, J.R. Antiparasitic and immunomodulatory potential of oral nanocapsules encapsulated lactoferrin protein against Plasmodium berghei. Nanomedicine 2016, 11, 47–62. [Google Scholar] [CrossRef]
- Kassim, O.O.; Ako-Anai, K.A.; Torimiro, S.E.; Hollowell, G.P.; Okoye, V.C.; Martin, S.K. Inhibitory factors in breastmilk, maternal and infant sera against in vitro growth of Plasmodium falciparum malaria parasite. J. Trop. Pediatr. 2000, 46, 92–96. [Google Scholar] [CrossRef]
- Tanaka, T.; Omata, Y.; Saito, A.; Shimazaki, K.; Igarashi, I.; Suzuki, N. Growth inhibitory effects of bovine lactoferrin to Toxoplasma gondii parasites in murine somatic cells. J. Vet. Med. Sci. 1996, 58, 61–65. [Google Scholar] [CrossRef]
- Anand, N.; Sehgal, R.; Kanwar, R.K.; Dubey, M.L.; Vasishta, R.K.; Kanwar, J.R. Oral administration of encapsulated bovine lactoferrin protein nanocapsules against intracellular parasite Toxoplasma gondii. Int. J. Nanomedicine 2015, 10, 6355–6369. [Google Scholar]
- Lima, M.F.; Kierszenbaum, F. Lactoferrin effects on the interaction of blood forms of Trypanosoma cruzi with mononuclear phagocytes. Int. J. Parasitol. 1987, 17, 1205–1208. [Google Scholar] [CrossRef]
- Chander, J. Textbook of Medical Mycology; Jaypee Brothers Medical Pub: New Delhi, India, 2002; ISBN 81-88039-01-2. [Google Scholar]
- Latorre, B. Enfermedades de las Plantas Cultivadas. Cienc. Nvestig. Agrar. 2004, 31, 213. [Google Scholar] [CrossRef]
- Bowman, S.M.; Free, S.J. The structure and synthesis of the fungal cell wall. BioEssays 2006, 28, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Pontón, J. La pared celular de los hongos y el mecanismo de acción de la anidulafungina. Rev. Iberoam. Micol. 2008, 25, 78–82. [Google Scholar] [CrossRef]
- Shrivastava, S.; Chattopadhyay, A. Influence of cholesterol and ergosterol on membrane dynamics using different fluorescent reporter probes. Biochem. Biophys. Res. Commun. 2007, 356, 705–710. [Google Scholar] [CrossRef]
- Cepero, M.C.; Restrepo, S.; Franco, A.E. Biología de Hongos; Universidad de los Andes: Cundinamarca, Colombia, 2012; ISBN 9586957012. [Google Scholar]
- Walker, G.M.; White, N.A. Introduction to Fungal Physiology. In Fungi; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; pp. 1–35. [Google Scholar] [CrossRef]
- López-Martínez, R.I. Introducción. Importancia actual de la micología médica en México. Proc. Gac. Medica Mex. 2008, 144, 121–122. [Google Scholar]
- Cuenca-Estrella, M. Antifúngicos en el tratamiento de las infecciones sistémicas: Importancia del mecanismo de acción, espectro de actividad y resistencias. Rev. Esp. Quimioter. 2010, 23, 169–176. [Google Scholar]
- Fernandes, K.E.; Carter, D.A. The antifungal activity of lactoferrin and its derived peptides: Mechanisms of action and synergy with drugs against fungal pathogens. Front. Microbiol. 2017, 8, 2. [Google Scholar] [CrossRef]
- Kirkpatrick, C.H.; Green, I.; Rich, R.R.; Schade, A.L. Inhibition of growth of Candida albicans by iron-unsaturated lactoferrin: Relation to host-defense mechanisms in chronic mucocutaneous candidiasis. J. Infect. Dis. 1971, 124, 539–544. [Google Scholar] [CrossRef]
- Okutomi, T.; Abe, S.; Tansho, S.; Wakabayashi, H.; Kawase, K.; Yamaguchi, H. Augmented inhibition of growth of Candida albicans by neutrophils in the presence of lactoferrin. FEMS Immunol. Med. Microbiol. 1997, 18, 105–112. [Google Scholar] [CrossRef]
- Torres, A.; Martin Loeches, I. Invasive pulmonary aspergillosis in ventilator-associated pneumonia: The hidden enemy? Am. J. Respir. Crit. Care Med. 2020, 202, 1071–1073. [Google Scholar] [CrossRef]
- Zarember, K.A.; Sugui, J.A.; Chang, Y.C.; Kwon-Chung, K.J.; Gallin, J.I. Human polymorphonuclear leukocytes inhibit Aspergillus fumigatus conidial growth by lactoferrin-mediated iron depletion. J. Immunol. 2007, 178, 6367–6373. [Google Scholar] [CrossRef] [PubMed]
- Zarember, K.A.; Cruz, A.R.; Huang, C.Y.; Gallin, J.I. Antifungal activities of natural and synthetic iron chelators alone and in combination with azole and polyene antibiotics against Aspergillus fumigatus. Antimicrob. Agents Chemother. 2009, 53, 2654–2656. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.W.; Campbell, L.T.; Wilkins, M.R.; Pang, C.N.I.; Chen, S.; Carter, D.A. Synergy and antagonism between iron chelators and antifungal drugs in Cryptococcus. Int. J. Antimicrob. Agents 2016, 48, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Valenti, P.; Visca, P.; Antonini, G.; Orsi, N. Interaction between lactoferrin and ovotransferrin and Candida cells. FEMS Microbiol. Lett. 1986, 33, 271–275. [Google Scholar] [CrossRef]
- Nikawa, H.; Samaranayake, L.P.; Tenovuo, J.; Pang, K.M.; Hamada, T. The fungicidal effect of human lactoferrin on Candida albicans and Candida krusei. Arch. Oral Biol. 1993, 38, 1057–1063. [Google Scholar] [CrossRef]
- Nikawa, H.; Samaranayake, L.P.; Hamada, T. Modulation of the anti-Candida activity of apo-lactoferrin by dietary sucrose and tunicamycin in vitro. Arch. Oral Biol. 1995, 40, 581–584. [Google Scholar] [CrossRef]
- Xu, Y.Y.; Samaranayake, Y.H.; Samaranayake, L.P.; Nikawa, H. In Vitro susceptibility of Candida species to lactoferrin. Med. Mycol. 1999, 37, 35–41. [Google Scholar] [CrossRef]
- Viejo-Díaz, M.; Andrés, M.T.; Fierro, J.F. Modulation of in vitro fungicidal activity of human lactoferrin against Candida albicans by extracellular cation concentration and target cell metabolic activity. Antimicrob. Agents Chemother. 2004, 48, 1242–1248. [Google Scholar] [CrossRef]
- Bellamy, W.; Wakabayashi, H.; Takase, M.; Kawase, K.; Shimamura, S.; Tomita, M. Killing of Candida albicans by lactoferricin B, a potent antimicrobial peptide derived from the N-terminal region of bovine lactoferrin. Med. Microbiol. Immunol. 1993, 182, 97–105. [Google Scholar] [CrossRef]
- Bellamy, W.; Yamauchi, K.; Wakabayashi, H.; Takase, M.; Takakura, N.; Shimamura, S.; Tomita, M. Antifungal properties of lactoferricin B, a peptide derived from the N-terminal region of bovine lactoferrin. Lett. Appl. Microbiol. 1994, 18, 230–233. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Hiratani, T.; Uchida, K.; Yamaguchi, H. Antifungal spectrum and fungicidal mechanism of an N-terminal peptide of bovine lactoferrin. J. Infect. Chemother. 1996, 1, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Viejo-Díaz, M.; Andrés, M.T.; Fierro, J.F. Different anti-Candida activities of two human lactoferrin-derived peptides, Lfpep and kaliocin-1. Antimicrob. Agents Chemother. 2005, 49, 2583–2588. [Google Scholar] [CrossRef] [PubMed]
- Nikawa, H.; Samaranayake, L.P.; Tenovuo, J.; Hamada, T. The effect of antifungal agents on the in vitro susceptibility of Candida albicans to apo-lactoferrin. Arch. Oral Biol. 1994, 39, 921–923. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Abe, S.; Okutomi, T.; Tansho, S.; Kawase, K.; Yamaguchi, H. Cooperative anti-Candida effects of lactoferrin or its peptides in combination with azole antifungal agents. Microbiol. Immunol. 1996, 40, 821–825. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Abe, S.; Teraguchi, S.; Hayasawa, H.; Yamaguchi, H. Inhibition of Hyphal Growth of Azole-Resistant Strains of Candida albicans by Triazole Antifungal Agents in the Presence of Lactoferrin-Related Compounds. Antimicrob. Agents Chemother. 1998, 42, 1587–1591. [Google Scholar] [CrossRef]
- Kuipers, M.E.; De Vries, H.G.; Eikelboom, M.C.; Meijer, D.K.F.; Swart, P.J. Synergistic fungistatic effects of lactoferrin in combination with antifungal drugs against clinical Candida isolates. Antimicrob. Agents Chemother. 1999, 43, 2635–2641. [Google Scholar] [CrossRef]
- Acosta-Zaldívar, M.; Andrés, M.T.; Rego, A.; Pereira, C.S.; Fierro, J.F.; Côrte-Real, M. Human lactoferrin triggers a mitochondrial- and caspase-dependent regulated cell death in Saccharomyces cerevisiae. Apoptosis 2016, 21, 163–173. [Google Scholar] [CrossRef]
- Carmona-Gutierrez, D.; Eisenberg, T.; Büttner, S.; Meisinger, C.; Kroemer, G.; Madeo, F. Apoptosis in yeast: Triggers, pathways, subroutines. Cell Death Differ. 2010, 17, 763–773. [Google Scholar] [CrossRef]
- Kang, J.J.; Schaber, M.D.; Srinivasula, S.M.; Alnemri, E.S.; Litwack, G.; Hall, D.J.; Bjornsti, M.A. Cascades of mammalian caspase activation in the yeast Saccharomyces cerevisiae. J. Biol. Chem. 1999, 274, 3189–3198. [Google Scholar] [CrossRef]
- Andrés, M.T.; Acosta-Zaldívar, M.; Fierro, J.F. Antifungal mechanism of action of lactoferrin: Identification of H+-ATPase (P3A-type) as a new apoptotic-cell membrane receptor. Antimicrob. Agents Chemother. 2016, 60, 4206–4216. [Google Scholar] [CrossRef]
- Lai, Y.-W.; Pang, C.N.I.; Campbell, L.T.; Chen, S.C.A.; Wilkins, M.R.; Carter, D.A. Different pathways mediate amphotericin-lactoferrin drug synergy in Cryptococcus and Saccharomyces. Front. Microbiol. 2019, 10, 2195. [Google Scholar] [CrossRef] [PubMed]
- Samaranayake, Y.H.; Samaranayake, L.P.; Wu, P.C.; So, M. The antifungal effect of lactoferrin and lysozyme on Candida krusei and Candida albicans. APMIS 1997, 105, 875–883. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, H.; Uchida, K.; Yamauchi, K.; Teraguchi, S.; Hayasawa, H.; Yamaguchi, H. Lactoferrin given in food facilitates dermatophytosis cure in guinea pig models. J. Antimicrob. Chemother. 2000, 46, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Lodish, H.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Baltimore, D.; Darnell, J. Viruses: Structure, Function, and Uses, 4th ed.; W. H. Freeman: New York, NY, USA, 2000. [Google Scholar]
- Cann, A.J. Replication of Viruses. Encyclopedia of Virology; Elsvier: Amsterdam, The Netherlands, 2008; pp. 406–412. ISBN 9780123744104. [Google Scholar] [CrossRef]
- Sieczkarski, S.B.; Whittaker, G.R. Viral entry. Curr. Top. Microbiol. Immunol. 2004, 285, 1–23. [Google Scholar] [CrossRef]
- Berlutti, F.; Pantanella, F.; Natalizi, T.; Frioni, A.; Paesano, R.; Polimeni, A.; Valenti, P. Antiviral properties of lactoferrin\-a natural immunity molecule. Molecules 2011, 16, 6992–7012. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Hangoc, G.; Oliff, A.; Chen, L.T.; Shen, R.-N.; Broxmeyer, H.E. Protective influence of lactoferrin on mice infected with the polycythemia-inducing strain of Friend Virus Complex. Cancer Res. 1987, 47, 4184–4188. [Google Scholar]
- Van der Strate, B.W.A.; Beljaars, L.; Molema, G.; Harmsen, M.C.; Meijer, D.K.F. Antiviral activities of lactoferrin. Antiviral Res. 2001, 52, 225–239. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Oda, H.; Yamauchi, K.; Abe, F. Lactoferrin for prevention of common viral infections. J. Infect. Chemother. 2014, 20, 666–671. [Google Scholar] [CrossRef]
- Seganti, L.; Di Biase, A.M.; Marchetti, M.; Pietrantoni, A.; Tinari, A.; Superti, F. Antiviral activity of lactoferrin towards naked viruses. BioMetals 2004, 17, 295–299. [Google Scholar] [CrossRef]
- Valenti, P.; Marchetti, M.; Superti, F.; Amendolia, M.G.; Puddu, P.; Gessani, S.; Borghi, P.; Belardelli, F.; Antonini, G.; Seganti, L. Antiviral Activity of Lactoferrin; Springer: Boston, MA, USA, 1998; pp. 199–203. ISBN 978-1-4757-9070-2. [Google Scholar]
- Pietrantoni, A.; Fortuna, C.; Remoli, M.; Ciufolini, M.; Superti, F. Bovine lactoferrin inhibits Toscana Virus infection by binding to heparan sulphate. Viruses 2015, 7, 480–495. [Google Scholar] [CrossRef]
- Andersen, J.H.; Jenssen, H.; Sandvik, K.; Gutteberg, T.J. Anti-HSV activity of lactoferrin and lactoferricin is dependent on the presence of heparan sulphate at the cell surface. J. Med. Virol. 2004, 74, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, C.A.M.; Sousa, I.P.; Silva, J.L.; Oliveira, A.C.; Gonçalves, R.B.; Gomes, A.M.O. Inhibition of Mayaro virus infection by bovine lactoferrin. Virology 2014, 452–453, 297–302. [Google Scholar] [CrossRef]
- Nozaki, A.; Ikeda, M.; Naganuma, A.; Nakamura, T.; Inudoh, M.; Tanaka, K.; Kato, N. Identification of a lactoferrin-derived peptide possessing binding activity to hepatitis C virus E2 envelope protein. J. Biol. Chem. 2003, 278, 10162–10173. [Google Scholar] [CrossRef] [PubMed]
- Puddu, P.; Borghi, P.; Gessani, S.; Valenti, P.; Belardelli, F.; Seganti, L. Antiviral effect of bovine lactoferrin saturated with metal ions on early steps of human immunodeficiency virus type 1 infection. Int. J. Biochem. Cell Biol. 1998, 30, 1055–1063. [Google Scholar] [CrossRef]
- Redwan, E.M.; Uversky, V.N.; El-Fakharany, E.M.; Al-Mehdar, H. Potential lactoferrin activity against pathogenic viruses. C. R. Biol. 2014, 337, 581–595. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, K.; Motsuchi, W.; Tanaka, S.; Dosakol, S.I. Inhibition with lactoferrin of in vitro infection with human herpes virus. Japn. J. Med. Sci. Biol. 1994, 47, 73–85. [Google Scholar] [CrossRef]
- Marchetti, M.; Longhi, C.; Conte, M.P.; Pisani, S.; Valenti, P.; Seganti, L. Lactoferrin inhibits herpes simplex virus type 1 adsorption to Vero cells. Antiviral Res. 1996, 29, 221–231. [Google Scholar] [CrossRef]
- Superti, F.; Ammendolia, M.G.; Valenti, P.; Seganti, L. Antirotaviral activity of milk proteins: Lactoferrin prevents rotavirus infection in the enterocyte like cell line HT-29. Med. Microbiol. Immunol. 1997, 186, 83–91. [Google Scholar] [CrossRef]
- Viani, R.M.; Gutteberg, T.J.; Lathey, J.L.; Spector, S.A. Lactoferrin inhibits HIV-1 replication in vitro and exhibits synergy when combined with zidovudine. Aids 1999, 13, 1273–1274. [Google Scholar] [CrossRef]
- Andersen, J.H.; Osbakk, S.A.; Vorland, L.H.; Traavik, T.; Gutteberg, T.J. Lactoferrin and cyclic lactoferricin inhibit the entry of human cytomegalovirus into human fibroblasts. Antiviral Res. 2001, 51, 141–149. [Google Scholar] [CrossRef]
- Beaumont, S.L.; Maggs, D.J.; Clarke, H.E. Effects of bovine lactoferrin on in vitro replication of feline herpesvirus. Vet. Ophthalmol. 2003, 6, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Marr, A.K.; Jenssen, H.; Moniri, M.R.; Hancock, R.E.W.; Panté, N. Bovine lactoferrin and lactoferricin interfere with intracellular trafficking of Herpes simplex virus-1. Biochimie 2009, 91, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, C.A.M.; Casseb, S.M.M.; Gonçalves, R.B.; Silva, E.V.P.; Gomes, A.M.O.; Vasconcelos, P.F.C. Bovine lactoferrin activity against chikungunya and zika viruses. J. Gen. Virol. 2017, 98, 1749–1754. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.; Sun, W.-Z.; Ng, T.B. Lactoferrin as potential preventative and treatment for COVID-19. Int. J. Antimicrob. Agents 2020, 56, 106118. [Google Scholar] [CrossRef] [PubMed]
- Madadlou, A. Food proteins are a potential resource for mining cathepsin L inhibitory drugs to combat SARS-CoV-2. Eur. J. Pharmacol. 2020, 885, 173499. [Google Scholar] [CrossRef] [PubMed]
- Serrano, G.; Kochergina, I.; Albors, A.; Diaz, E.; Hueso, G.; Serrano, J.M. Liposomal lactoferrin as potential preventative and cure for COVID-19. Int. J. Res. Heal. Sci. 2020, 8, 8–15. [Google Scholar] [CrossRef]
- Yi, M.; Kaneko, S.; Yu, D.Y.; Murakami, S. Hepatitis C virus envelope proteins bind lactoferrin. J. Virol. 1997, 71, 5997–6002. [Google Scholar] [CrossRef]
- Liao, Y.; El-Fakkarany, E.; Lönnerdal, B.; Redwan, E.M. Inhibitory effects of native and recombinant full-length camel lactoferrin and its N and C lobes on hepatitis C virus infection of Huh7.5 cells. J. Med. Microbiol. 2012, 61, 375–383. [Google Scholar] [CrossRef]
- Redwan, E.R.M.; Tabll, A. Camel lactoferrin markedly inhibits hepatitis C virus genotype 4 infection of human peripheral blood leukocytes. J. Immunoass. Immunochem. 2007, 28, 267–277. [Google Scholar] [CrossRef]
- El-Fakharany, E.M.; Sánchez, L.; Al-Mehdar, H.A.; Redwan, E.M. Effectiveness of human, camel, bovine and sheep lactoferrin on the hepatitis C virus cellular infectivity: Comparison study. Virol. J. 2013, 10, 199. [Google Scholar] [CrossRef]
- Superti, F.; Giansanti, F.; Valenti, P.; Siciliano, R.; Rega, B.; Antonini, G. Involvement of bovine lactoferrin metal saturation, sialic acid and protein fragments in the inhibition of rotavirus infection. Biochim. Biophys. Acta 2001, 1528, 107–115. [Google Scholar] [CrossRef]
- Pietrantoni, A.; Dofrelli, E.; Tinari, A.; Ammendolia, M.G.; Puzelli, S.; Fabiani, C.; Donatelli, I.; Superti, F. Bovine lactoferrin inhibits Influenza A virus induced programmed cell death in vitro. Biometals 2010, 23, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Swart, P.J.; Harmsen, M.C.; Kuipers, M.E.; Van Dijk, A.A.; Van Der Strate, B.W.A.; Van Berkel, P.H.C.; Nuijens, J.H.; Smit, C.; Witvrouw, M.; De Clercq, E.; et al. Charge modification of plasma and milk proteins results in antiviral active compounds. J. Pept. Sci. 1999, 5, 563–576. [Google Scholar] [CrossRef]
- Ammendolia, M.G.; Agamennone, M.; Pietrantoni, A.; Lannutti, F.; Siciliano, R.A.; de Giulio, B.; Amici, C.; Superti, F. Bovine lactoferrin-derived peptides as novel broad-spectrum inhibitors of influenza virus. Pathog. Glob. Health 2012, 106, 12–19. [Google Scholar] [CrossRef]
- Harmsen, M.C.; Swart, P.J.; de Béthune, M.P.; Pauwels, R.; De Clercq, E.; The, T.H.; Meijer, D.K. Antiviral effects of plasma and milk proteins: Lactoferrin shows potent activity against both human immunodeficiency virus and human cytomegalovirus replication in vitro. J. Infect. Dis. 1995, 172, 380–388. [Google Scholar] [CrossRef] [PubMed]
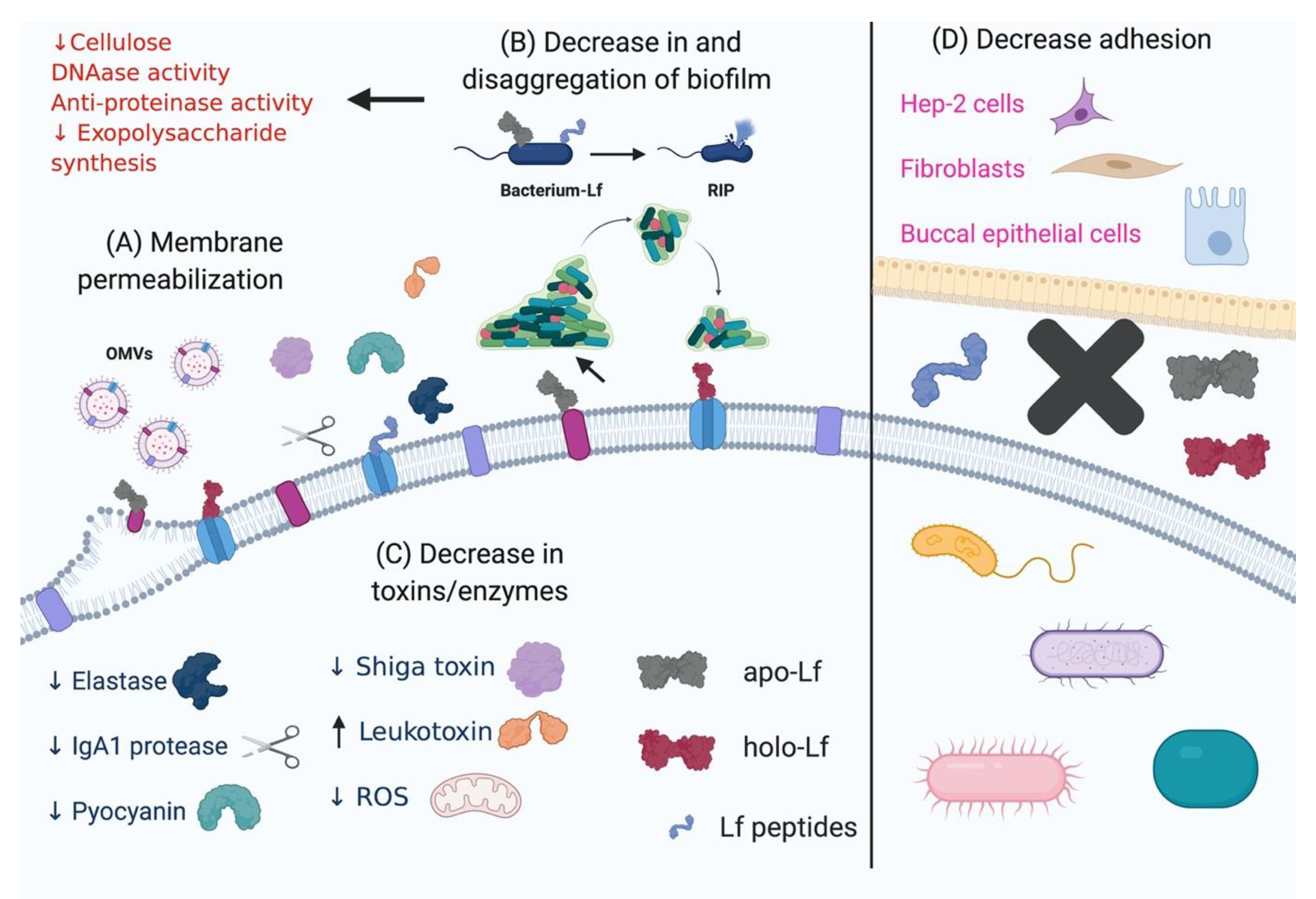
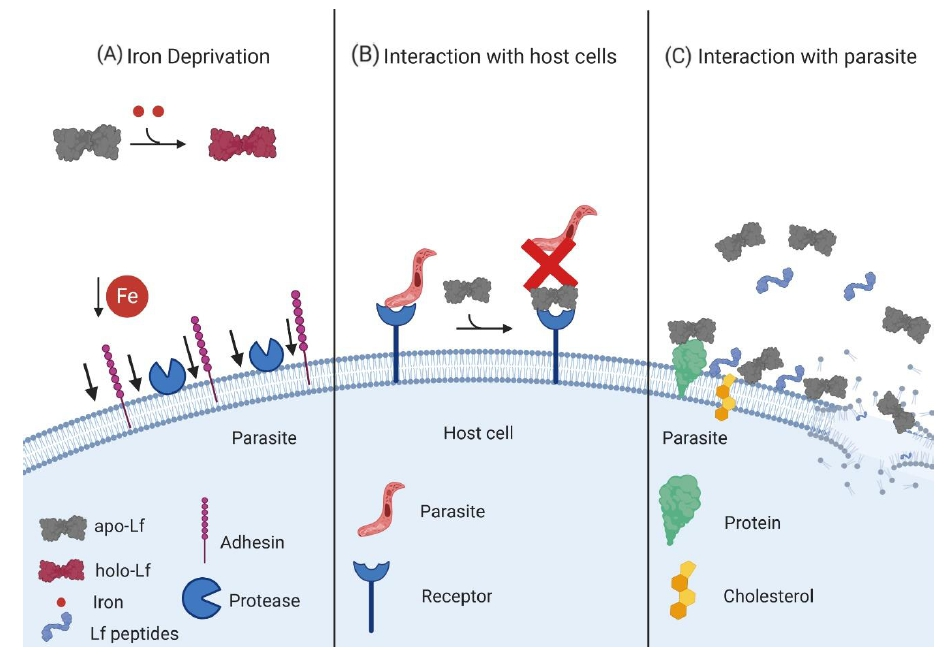
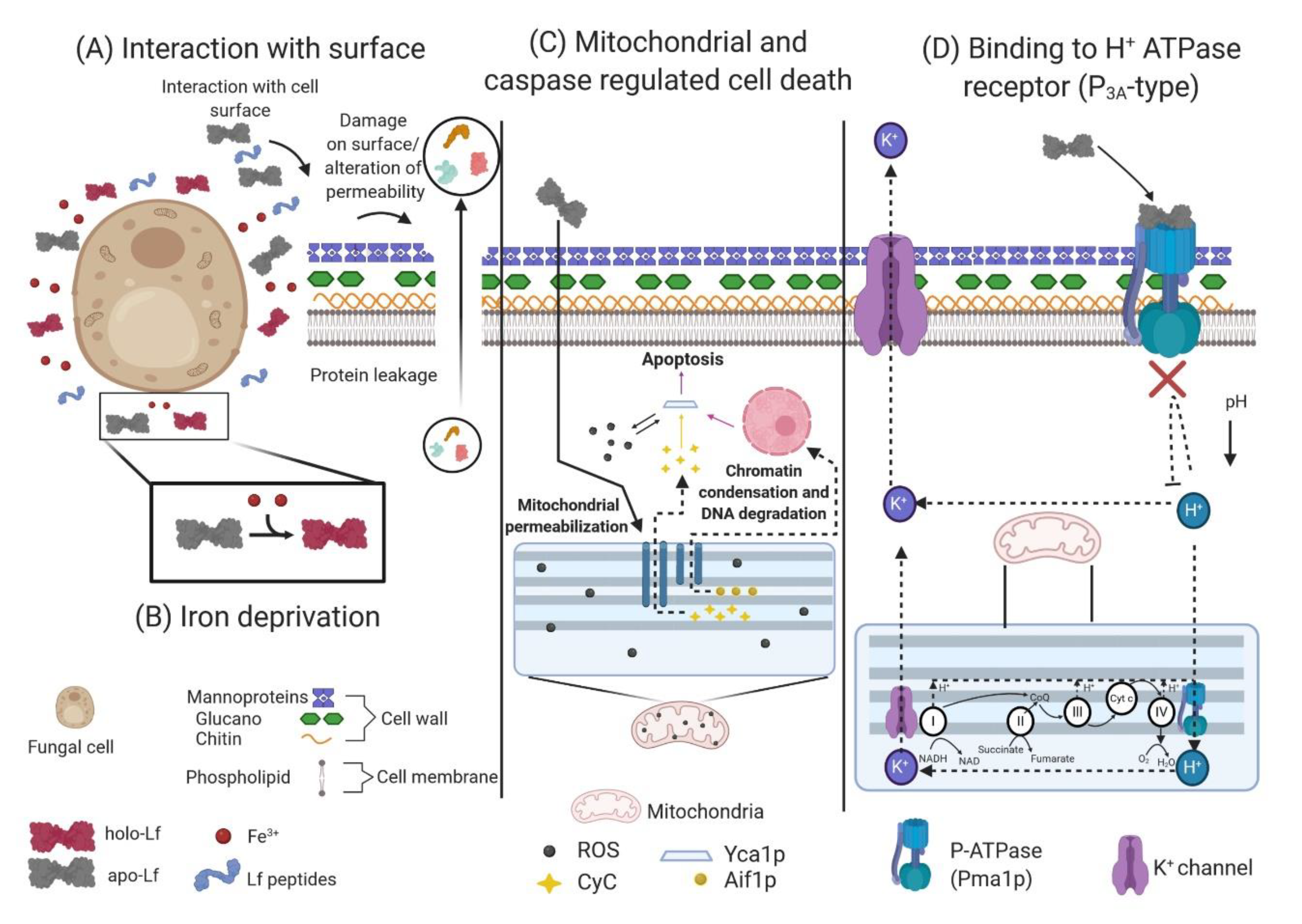
| Pathogens (Bacteria) | Source and Type of Lf/Iron-Saturated Condition | Effect on Viability/Growth/ Concentration | Other Effects In Vitro/Concentration | Ref. | |
|---|---|---|---|---|---|
| Actinobacillus pleuropneumoniae | apo-bLf (N) | - | Inhibited adhesion on porcine buccal epithelial cells: 0.8 µM | [123] | |
| A. actinomycetemcomitans | hLf, bLf (N) | - | Inhibited adhesion on fibroblasts: 0.5–2500 pg/mL | [122] | |
| Prevotella intermedia | |||||
| Escherichia coli | hLf (N) | - | Inhibited adhesion to HeLa cells: 20 and 30 mM | [121] | |
| bLf (N), bLfcin (N) | Bactericidal activity bLf: 13 µg/mL | Released LPS: bLf: 2 mg/mL, Lfcin: 100 µg/mL. Appearance of membrane “blisters” bLfcin: 100 µg/mL | [86] | ||
| Lfcin (N), Lframpin (S) | Bactericidal activity: Lframpin and Lfcin: 20 µM | Membrane damage and release of vesicle-like structures: Lframpin and Lfcin: 20 µM | [87] | ||
| LfcinB (N) | Bactericidal activity: 3 µM | Membrane permeabilization: 3 µM | [90] | ||
| apo-Lfb (N), Lfcin (N), Lfampin (S), Lfchimera (S) | Bactericidal activity: bLf, Lfcin, Lfampin: 20 and 40 µM, Lfchimera: 1 µM bLf and peptides + LPS (10–100-fold): counteracts the inhibitory effect of Lf. Synergistic effect with antibiotics: Lfampin, Lfcin 10 µM + ampicillin | Lfchimera induced membrane permeabilization: 1 µM | [81] | ||
| Staphylococcus aureus | |||||
| E. coli | Lf (N) | - | Antibiofilm activity: 40 µg/mL | [102] | |
| Klebsiella pneumoniae | |||||
| Mannheimia haemolytica | apo-bLf (N) holo-bLf (N) | Bactericidal activity apo-Lfb: 12 µM | Membrane permeabilization and damage, increased the release of OMVS Apo-Lfb: 2–10 µM: Increased secretion of Lkt 2–10 µM | [89] | |
| Pseudomonas aeruginosa | apo-bLf (N), Lfcin (N), Lfampin (S), Lfchimera (S) | Bactericidal activity: Lfb: 9.4 µM, Lfcin: 2.9 µM, Lfampin: 5.8 µM, Lfcin + Lfampin: 1.4 µM, Lfchimera: 0.9 µM | Inhibited pyocyanin, elastase and biofilm production: 1, 5, 25 µM | [68] | |
| Synthetic cationic peptides and lipopeptides from hLf | Bactericidal activity: 8–128 mg/mL depending on the peptide | Antibiofilm activity 10-fold major MIC | [106] | ||
| apo-bLf (N) | Bactericidal activity: 2% | Antibiofilm activity: 2% | [101] | ||
| Pseudomonas fluorescens | bLf hydrolysate (N) | - | Antibiofilm activity: 3 mg/mL | [112] | |
| Prevotella intermedia | hLf (N), apo-bLf (N), holo-bLf (N), LfcinB (N) | Bactericidal activity: hLf, apo-bLf, and holo-bLf: 0.13 to 8 mg/mL, Lfcin B at 0.006 to 0.4 mg/mL Synergistic effect with antibiotics: 0.1 or 10 g/mL of ABPC, CPFX, CAM, or MINO + 0.5 mg/mL native bLf or apo-bLf | Antibiofilm activity: P. gingivalis hLf, apo-, holo-bLf: 0.008 mg/mL LfcinB: 0.4 mg/mL P. intermedia: apo-, holo-bLf: >0.31 mg/mL, hLf: >0.13 mg/mL, LfcinB: 0.4 mg/mL | [104] | |
| Porphyromonas gingivalis | |||||
| apo-bLf (N), holo-bLf (N) | - | Anti-proteinase activity: 5 mg/mL Inhibited biofilm formation: 0.065 mg/mL | [108] | ||
| Streptococcus mutants | apo-bLf (N), holo-bLf (N) | Bactericidal activity apo- and holo-Lfb: 20 µg/mL | Decreased aggregation and biofilm development: 20 µg/mL | [109] | |
| Streptococcus pneumoniae | apo-bLf (N), Lfcin (N) Lfampin (S), Lfchimera (S) | - | Diminished adhesion on laryngeal, lung and nasopharyngeal human cells: 40 µM bLf and 10 µM peptides Eradicated pneumococcal preformed biofilms: 40 and 80 µM bLf eDNAase activity: 40 µM bLf | [103] | |
| apo-bLf (N), Lfcin (N) Lfampin (S), Lfchimera (S) | apo-Lfb: 40 µM Lfcin, Lfampin, Lfchimera: 10 µM | Ultrastructural damage: 40 µM for all peptides | [88] | ||
| STEC (Stx-producing Escherichia coli) | apo-bLf (N) | - | Decreased Stx2 secretion: 0.1, 1, 10 mg/mL Diminished verotoxicity: 0.1 or 1 mg/mL Protease activity: 1000, 100, 10, 1 mg/mL | [119] | |
| Vibrio cholerae O1 and non-O1 strains | bLf (N), Lfcin (N), Lfampin (S), Lfchimera (S) | Bactericidal activity: bLf: 40 mM, bLFcin: 20 μM, Lfampin: 20 μM, Lfchimera: 5 μM Synergistic effect with antibiotics: 1 μM Lfchimera + 2.5 µg/mL chloramphenicol 10 μM LF + 2.5 µg/mL chloramphenicol | Membrane permeabilization, vesicularization and membrane damage Lfchimera: 5 μM; bLf, Lfcin and Lfampin: 20 μM | [70] | |
| Vibrio parahaemolyticus | Lfchimera (S) | Lfchimera: 40 µM antibiotics 5μM Lfchimera + 5 µg/mL ampicillin, gentamicin, kanamycin | Membrane permeabilization, vesicularization and membrane damage Lfchimera: 40 μM | [91] | |
| Yersinia enterocolitica/pseudotuberculosis | bLfcin (S) | Bactericidal activity: 4 mg/mL | Enhanced adhesion on Hep-2 cells Bacterial internalization is inhibited | [124] |
| Pathogen (Parasite) | Source and Type of Lf/Iron-Saturated Condition | Effect on Viability/Growth Concentration | Other Effects in Vitro Concentration | Effects In Vivo Dose | Ref. |
|---|---|---|---|---|---|
| Acanthamoeba spp. | apo-bLf (N) | Decreased viability Growth inhibition 10 µM | Decreased encystment ratio. The cyst could not retransform to trophozoite. 10 µM | - | [142] |
| Cryptosporidium parvum | bLf (N) | Decreased viability 1–10 mg/mL | Decreased infectivity to HCT-8 cells 10 mg/mL | - | [146] |
| E. histolytica | apo-hLf(N), apo-bLf (N), Lfcin (N) | Decreased viability, 12.5–100 µM, 64.7–647 µM | Synergism with metronidazole 31.25 µM 323–453 µM | - | [60] |
| Lfampin (S) | Decreased viability 250–1000 µM | Killed trophozoites by lysis, 250 µM | Resolved amoebic intracecal infection in mice: 10 mg/kg daily for 4 days Lfcin17–30, LfcinB: absence of amoebic trophozoites in the lumen of 75% of the animals or a decrease in parasitic load | [145] | |
| apo-bLf (N) | - | - | Resolved intracecal infection in C3H/HeJ mice: 63.14% totally and 36.86% partially. Increased IgA, Induced Th2 response, 20 mg/kg daily for 7 days | [176] | |
| apo-bLf (N) | - | - | Resolved hepatic amoebiasis in hamster (decrease of lesions) Proteins, enzymes and hepatic cells returned to normal parameters: 2.5 mg/100 g mass daily for 8 days | [177] | |
| Human milk (N), apo-hLf (N), apo-bLf (N) | Decreased viability 5–20%, 1 mg/mL | Synergism with lysozyme and IgA: 1 mg/mL. Synergism with porcine milk: 1 mg/mL | - | [141] | |
| Lfcin17-30 (N), Lfampin265-284 (S), Lfchimera (S) | Decreased viability, 25–100 µM | - | - | [71] | |
| E. stiedai | Lfcin (N) | - | Decreased infectivity to rabbit hepatobiliary cells, 100–1000 µg/mL | Decreased number of oocysts in the feces of rabbits inoculated with treated sporozoites. Fewer abscesses and bile ducts were not swollen: 1000 µg/mL | [178] |
| Giardia intestinalis | bLf (N), Lfcin 17–30 (N), Lfampin 265–284 (S), Lfchimera (S) | Decreased viability Growth inhibition, 40 µM | Synergism with metronidazole, albendazole Peptides 40 µM. Morphological alterations: Electron-dense material in cytoplasm, reorganization of flagellum, displacement of adherent disk, membrane disruption, shrunken and distorted peripheral vacuoles. Apoptosis: 40 µM | - | [144] |
| bLf (N), Lfcin (N) | Growth inhibition, 12.5–50 µM, 1.3–3.9 µM | Morphological alterations: Vesiculation of ER, enlargement of nuclear envelope, delocalization and electron-dense PVs, changes in the cytoskeleton, invaginations and protrusions of plasma membrane, induced differentiation to cyst. Production of futile cysts: 12.5 µM, 2.6 µM | - | [143] | |
| Haemonchus contortus | Camel’s milk (N) | - | Inhibited egg hatching Inhibition of motility 5–100 mg/mL | - | [179] |
| Plasmodium berghei | apo-buLf (N) | - | - | Decreased infectivity to RBCs Reduced parasite load in mice Histopathology: Spleen: Decreased pigmentation. Liver: Decreased inflammation. Less accumulation of histiocytes and lymphocytes. Increased ROS and NO production. Expression of various miRNA genes required in Fe regulation. Upregulation of innate immune cytokines Increased Th1 response Increased survival of infected mice: 12 g bLf (Fe-bLf)/kg of diet | [180] |
| Plasmodium falciparum | apo-hLf (N) | Growth inhibition (RBC preincubation) 30 µM | Parasites could not develop from ring stages to trophozoites, 30 µM | - | [166] |
| hLf (N) (isolated fraction) | Growth inhibition 2 mg/mlL | - | - | [181] | |
| Toxoplasma gondii | apo-bLf (N) holo-bLf (N) | Lf suppressed the intracellular growth of parasites, 100–1000 µg/mL | - | - | [182] |
| Lfcin (N) | - | Decreased infectivity to MEC 100–1000 µg/mL | Increased survival of mice infected with treated sporozoites. Mice did not show clinical signs of infection: 1000 µg/mL | [178] | |
| holo-hLf (N) | Significantly inhibited the intracellular growth, 0.1–100 µg/mL | - | - | [149] | |
| native-bLf (N) apo-bLf (N) | - | Decreased infectivity to macrophages (mice J7741). Decreased number of tachyzoites per macrophage. Increased production of NO in macrophages, 20 µg/mL | Histopathology: Liver: No signs of pathology or infection Decreased parasite load Spleen: Decreased parasite load Increased ROS/NO production Increased cytokine production Elevated levels of different iron regulators Increased survival of infected mice: 12 g/kg of diet | [183] | |
| Trypanosoma cruzi | hLf (N) | - | Monocyte and macrophage stimulation Increased phagocytic capacity Stimulated intracellular killing capacity of HBM or MPM | - | [152] |
| apo-hLf (N) | - | Macrophages had greater capacity to internalize and kill trypomastigotes: 10 µg/mL | - | [184] |
| Pathogen (Fungal) | Source and Type of Lf/iron-Saturated Condition | Concentration | Effect on Viability | Ref. |
|---|---|---|---|---|
| Absidia corymbifera | bLfcin (N) | In Vitro MIC: 40->80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Aspergillus clavatus | bLfcin (N) | In Vitro MIC: >80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Aspergillus fumigatus | apo-hLf (N), apo-hLf(N) + amphotericin B | In Vitro IC50: ~10 (conidia)-80 μg/mL (hyphae) IC50: 10 nM | Iron deprivation | [198,199] |
| bLfcin (N) | In Vitro MIC: 45–80 μg/mL | Interaction with cell surface/alteration of cell membrane | [207,208] | |
| Aspergillus flavus | bLfcin (N) | In Vitro MIC: >80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Aspergillus niger | bLfcin (N) | In Vitro MIC: 30–80 μg/mL | Interaction with cell surface/alteration of cell membrane | [207,208] |
| Aspergillus versicolor | bLfcin (N) | In Vitro MIC: 10 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Candida albicans | apo-hLf (N), ovotransferrin (N) | In Vitro 5–200 μg/Ml 1 mg/mL 5 μM | Iron deprivation Interaction with cell surface/alteration of cell membrane H+ ATPase (P3A-type) | [195,201,202,203,205,218,220] |
| apo-bLf (N) | In Vitro 20 μg/mL | Interaction with cell surface/alteration of cell membrane | [204] | |
| bLfcin (N) | In Vitro 10–60 μg/mL bLfcin (100 µg/mL) + fluconazole or itraconazole (25 µg/mL) | Interaction with the cell surface | [206,207,208,212] | |
| bLF (N), Lfampin (S), bLfcin (N) | In Vitro 20 μM | Interaction with cell surface/alteration of cell membrane | [87] | |
| Lfpep (S), kaliocin-1 (S) | In Vitro MIC: 18.7 μM MIC: 150 μM | Interaction with cell surface/alteration of cell membrane | [209] | |
| apo-hLf (N)+ nystatin apo-hLf (N) + amphotericin B apo-hLf (N) + clotrimazole apo-hLf(N) + miconazole apo-hLf (N) + 5-fluorocytosine apo-hLf (N) + tunicamycin | In Vitro MIC: apo-hLf (20 μg/mL) + nystatin (2.0μg/mL) apo-hLf (20 μg/mL) + amphotericin B (0.4 μg/mL) apo-hLf (20 μg/mL) + clotrimazole (10 μg/mL) apo-hLf (20 μg/mL) + miconazole (4 μg/mL) apo-hLf (20 μg/mL) + 5-fluorocytosine (4 μg/mL) apo-hLf (20 μg/mL) + tunicamycin (40 μg/mL) | Interaction with cell surface/alteration of cell membrane | [210] | |
| apo-bLf (N) + amphotericin B apo-bLf (N) + fluconazole apo-bLf (N) + murine neutrophils | In Vitro apo-bLf (0.5–98 mg/mL) + amphotericin B (0.06–0.2 μg/mL) apo-bLf (0.5–98 mg/mL) + fluconazole (10 μg/mL) apo-bLf (110 μg/mL) + murine neutrophils (40 μg/mL) | Iron deprivation Interaction with cell surface/alteration of cell membrane | [196,213] | |
| bLfcin (N) + clotrimazole bLfcin (N) + ketoconazole bLfcin (N) + fluconazole bLfcin (N) + itraconazole | In Vitro bLf (100 μg/mL) + clotrimazole (12.5 ng/mL) bLf (100 μg/mL) + ketoconazole (3.1 ng/mL) bLf (100 μg/mL) + fluconazole (1000 ng/mL) bLf (100 μg/mL) + itraconazole (12.5 ng/mL) bLfcin (3.1 mg/mL) + clotrimazole (12.5 ng/mL) bLfcin (3.1 mg/mL) + ketoconazole (12.5 ng/mL) bLfcin (3.1 mg/mLl) + fluconazole (4000 ng/mL) bLfcin (3.1 mg/mL) +itraconazole (12.5 ng/mL) | Interaction with cell surface/alteration of cell membrane Synergistic activity with azoles | [211] | |
| Candida glabrata | apo-bLf (N) | In Vitro 20 μg/mL | Interaction with cell surface/alteration of cell membrane | [204] |
| bLfcin (N) | In Vitro MIC: 80->80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] | |
| Lfpep (S), kaliocin-1(S) | In Vitro MIC: 9.3 μM MIC: 150 μM | Interaction with cell surface/alteration of cell membrane | [209] | |
| apo-bLf (N)+ amphotericin B apo-bLf (N)+ fluconazole | In Vitro MIC: apo-bLf (<5–57 mg/mL) + amphotericin B (0.1-0.4 μg/mL) apo-bLf (<5–57 mg/mL) + fluconazole (24–156 μg/mL) | Interaction with cell surface/alteration of cell membrane | [213] | |
| Candida guilliermondii | apo-bLf (N) | In Vitro 20 μg/mL | Interaction with cell surface/alteration of cell membrane | [204] |
| bLfcin (N) | In Vitro MIC: 5–40 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] | |
| Lfpep (S), kaliocin-1 (S) | In Vitro MIC: 9.3 μM MIC: 150 μM | Interaction with cell surface/alteration of cell membrane | [209] | |
| Candida kefyr | bLfcin (N) | In Vitro MIC: 2.5–10 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Candida krusei | apo-hLf (N) | In Vitro 5–200 μg/mL | Interaction with cell surface/alteration of cell membrane | [202,203,204,220] |
| bLfcin (N) | In Vitro MIC: 10–20 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] | |
| Lfpep (S), kaliocin-1 (S) | In Vitro MIC: 4.7 μM MIC: 150 μM | Interaction with cell surface/alteration of cell membrane | [209] | |
| Candida parapsilosis | apo-bLf (N) | In Vitro 20 μg/mL | Interaction with cell surface/alteration of cell membrane | [204] |
| bLfcin (N) | In Vitro MIC: 20–80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] | |
| Lfpep (S), kaliocin-1 (S) | In Vitro MIC: 9.3 μM MIC: 150 μM | Interaction with cell surface/alteration of cell membrane | [209] | |
| Candida tropicalis | apo-bLf (N) | In Vitro 20 μg/mL | Interaction with cell surface/alteration of cell membrane | [204] |
| bLfcin (N) | In Vitro MIC: 0.31–1.25 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] | |
| Lfpep (S), kaliocin-1 (S) | In Vitro MIC: 9.3 μM MIC: 150 μM | Interaction with cell surface/alteration of cell membrane | [209] | |
| Cladosporium trichoides | bLfcin (N) | In Vitro MIC: 5 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Cryptococcus curvatus | bLfcin (N) | In Vitro MIC: 3-9 μg/mL | Interaction with cell surface/alteration of cell membrane | [207] |
| Cryptococcus gattii | apo-bLf (N) | In Vitro MIC: 64 μg/mL | Iron deprivation | [200] |
| Cryptococcus neoformans | apo-bLf (N) apo-bLf (N) + amphotericin B | In Vitro MIC: 32–64 μg/mL apo-bLf (8 μg/mL) + amphotericin B (0.25 μg/mL) | Iron deprivation Altered responses to stress | [200,219] |
| bLfcin (N) | In Vitro MIC: 0.63 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] | |
| Cryptococcus uniguttulatus | bLfcin (N) | In Vitro MIC: 3–6 μg/mL | Interaction with cell surface/alteration of cell membrane | [207] |
| Epidermophyton floccosum | bLfcin (N) | In Vitro MIC: 0.31–2.5 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Exophiala dermatidis | bLfcin (N) | In Vitro MIC: 2.5 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Fonsecaea pedroi | bLfcin (N) | In Vitro MIC: 5 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Fusarium moniliforme | bLfcin (N) | In Vitro MIC: 2.5–5 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Microsporum canis | bLfcin (N) | In Vitro MIC: 40 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Microsporum gypseum | bLfcin (N) | In Vitro MIC: 20–40 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Mucor circinelloides | bLfcin (N) | In Vitro MIC: >80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Mucor racemosus | bLfcin (N) | In Vitro MIC: >80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Nannizzia gypsea | bLfcin (N) | In Vitro MIC: 30->60 μg/mL | Interaction with cell surface/alteration of cell membrane | [207] |
| Nannizzia incurvata | bLfcin (N) | In Vitro MIC: 6–18 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Nannizzia otae | bLfcin (N) | In Vitro MIC: 12–60 μg/mL | Interaction with cell surface/alteration of cell membrane | [207] |
| Paracoccidioides brasiliensis | bLfcin (N) | In Vitro MIC: 0.63–1.25 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Penicillium expansum | bLfcin (N) | In Vitro MIC: >80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Penicillum notatum | bLfcin (N) | In Vitro MIC: >80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Penicillium pinophilum | bLfcin (N) | In Vitro MIC: 3–45 μg/mL | Interaction with cell surface/alteration of cell membrane | [207] |
| Penicillium vermiculatum | bLfcin (N) | In Vitro MIC: 6–45 μg/mL | Interaction with cell surface/alteration of cell membrane | [207] |
| Phialophora verrucosa | bLfcin (N) | In Vitro MIC: 5–10 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Rhizopus oryzae | bLfcin (N) | In Vitro MIC: 60 ≥ 80 μg/mL | Interaction with cell surface/alteration of cell membrane | [207,208] |
| Saccharomyces cerevisiae | apo-hLf (N) | In Vitro 1.56–6.25 µM | Mitochondrial and caspase-dependent regulated cell death | [216] |
| apo-bLf (N) | In Vitro MIC: 16 μg/mL | Iron deprivation | [200] | |
| bLfcin (N) | In Vitro MIC: 0.63 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] | |
| Sporothrix schenckii | bLfcin (N) | In Vitro MIC: 2.5–10 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Trichophyton cutaneum | bLfcin (N) | In Vitro MIC: 1.25–2.5 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Trichophyton mentagrophytes | bLfcin (N) | In Vitro MIC: 6 ≥ 80 µg/mL | Interaction with cell surface/alteration of cell membrane | [207,208] |
| Trichophyton rubrum | bLfcin (N) | In Vitro MIC: 12 ≥ 80 µg/mL | Interaction with cell surface/alteration of cell membrane | [207,208] |
| Trichphyton shoenleinii | bLfcin (N) | In Vitro MIC: >80 μg/ml | Interaction with cell surface/alteration of cell membrane | [208] |
| Trichophyton spp. | hLf (N) bLf (N) bLfcin (N) | In Vitro MIC: hLf (400, 800 and 13 mg/L) bLf (50, 100 and 13 mg/L) bLfcin (3.1, 6.3, 13 mg/L) In vivo Guinea pigs Doses: 2.5 g/kg/day for 28 days | Interaction with cell surface/alteration of cell membrane | [221] |
| Trichophyton tonsurans | bLfcin (N) | In Vitro MIC: 5–40 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Trichophyton violaceum | bLfcin (N) | In Vitro MIC: 40 ≥ 80 μg/mL | Interaction with cell surface/alteration of cell membrane | [208] |
| Trichosporon cutaneum | bLfcin (N) | In Vitro MIC: 6–18 μg/mL | Interaction with cell surface/alteration of cell membrane | [207] |
| Pathogen (DNA Virus) | Source and Type of Lf/iron-Saturated Condition | Effect on Cell Viral Infection/Concentration | Other Effects In Vitro/Concentration | Ref. |
| Feline herpesvirus (FHV-1) | bLf (N) | In Vitro: Inhibition of viral replication 0.5–10 mg/mL | [242] | |
| Human cytomegalovirus (HCMV) | apo-bLf (N) and holo-hLf (N) | In Vitro Prevented virus adsorption and penetration into the host cells 1 mg/mL | [237] | |
| apo-hLf (N), holo-hLf (N), apo-bLf(N), apo-gLf (N) and cyclic bLfcin (S) | In Vitro Prevented virus adsorption and penetration into the host cells IC50: bLf: 0.7 μM, hLf: 1.1 μM, gLf: 3.4 μM and cyclic bLfcin: 5 μM | [241] | ||
| Pathogen (RNA virus) | Source and type of Lf/iron-saturated condition | Effect on cell viral infection/concentration | Other effects In Vitro/concentration | Ref. |
| Chikungunya virus | apo-bLf (N) | In Vitro Prevention of viral infection IC50: 0.2 mg/mL Decreased viral replication 1.0 mg/mL | [244] | |
| Hepatitis C virus (HCV) | hLf (N), cLf (N), bLf (N) and oLf (N) | In Vitro Prevented viral adsorption and penetration into host cells hLf, bLf and oLf: 0.25 and 0.5 mg/mL cLf: 100, 150, 200, 250 and 500 μg/mL | [251] | |
| holo-cLf (N) | In Vitro Prevented viral adsorption and penetration into the host cells 1 mg/mL | [250] | ||
| cLf (N), cLf N lobe (N), cLf C lobe (N), rcLf (S) and rcLf N lobe (S) | In Vitro Blocks viral entry/viral infection cLf and rcLf: 0.5–1.0 mg/mL Inhibition of viral replication cLf and rcLf: 0.75–1.25 mg/mL cLf N and C lobes: 0.25–1.25 mg/mL rcLf C lobe: 1.0–1.25 mg/mL | [249] | ||
| Human immunodeficiency virus (HIV-1) | hLf (N) and bLf (N) | In Vitro Inhibition/prevention of viral infection IC50: hLf: 75 μg/mL bLf: 40 μg/mL | [214] | |
| holo-bLf (N) and apo-bLf (N) | In Vitro Prevented viral adsorption and penetration into host cells 0–20 μg/mL | [235] | ||
| apo-hLf (N), apo-bLf (N) with zidovudine | In Vitro Prevented viral adsorption and penetration into host cells IC50: apo-hLf: 9.6 μM and apo-bLf: 2.4 μM Zidovudine: 0.25–0.001 μM | [240] | ||
| Herpes simplex virus-1 (HSV-1) | holo-hLf (N) and apo-bLf (N) | In Vitro Prevented viral adsorption and penetration into host cells 1 mg/mL | [237] | |
| apo-hLf (N), apo-bLf (N), | In Vitro Prevented viral adsorption and penetration into host cells IC50: apo-hLf (1.41 μM) and apo-bLf (0.12 μM) | [238] | ||
| bLf (N) and b-Lfcin (N) | In Vitro Prevented viral adsorption and penetration into host cells IC50: bLf: 0.6 μM, bLfcin: 14.6 μM | [243] | ||
| Mayarovirus (MAYV) | apo-bLf (N) | In Vitro Prevented viral adsorption and penetration into host cells 1 mg/mL | [233] | |
| Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) | Liposomal bLf (S) (LactyferrinTM) | Oral supplementation LactyferrinTM: 32 mg/10 mL and vitamin C 12 mg/10 mL Dose for treatment for COVID-19 64-96 mg/6 h daily to cure COVID-19 (256–384 mg/d). Dose can be increased to 128 mg/6 h (512 mg) if needed. Preventive dose for COVID 19: 64 mg two to three times daily (128–192 mg/d). Lactyferrin syrup for pregnant women and infants (glycerosome encapsulation, alcohol free) - Pregnant women and infants under the age of two. - Mothers: 64 mg (20 mL) twice a day (128 mg/d). - Infants: 32 mg (10 mL) twice daily. - Zinc defense syrup: 10-30 mg/d (10–30 mL) LF nasal drops (Lactyferrin) to relieve acute sinusitis, alterations in smell and taste, and dry cough. In acute cases, we recommend applying two drops to each nostril every 4–6 h. | [247] | |
| Simian rotavirus SA11 | apo-bLf (N), holo-bLf (N), Zn-bLf (S), Mn-bLf (S) | In Vitro Inhibition of cytopathic effect EC50: apo-bLf: 50 μg/mL holo-bLf: 46 μg/mL Zn-bLf and Mn-bLf: 62 μg/mL | [253] | |
| apo-bLf (N) and holo-bLf (N) | In Vitro Prevented viral adsorption and penetration into host cells 25 μM | [239] | ||
| Zika virus | apo-bLf (N) | Prevention of viral infection IC50: 0.4 mg/mL Decreased viral replication 1.0 mg/mL | [244] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zarzosa-Moreno, D.; Avalos-Gómez, C.; Ramírez-Texcalco, L.S.; Torres-López, E.; Ramírez-Mondragón, R.; Hernández-Ramírez, J.O.; Serrano-Luna, J.; de la Garza, M. Lactoferrin and Its Derived Peptides: An Alternative for Combating Virulence Mechanisms Developed by Pathogens. Molecules 2020, 25, 5763. https://doi.org/10.3390/molecules25245763
Zarzosa-Moreno D, Avalos-Gómez C, Ramírez-Texcalco LS, Torres-López E, Ramírez-Mondragón R, Hernández-Ramírez JO, Serrano-Luna J, de la Garza M. Lactoferrin and Its Derived Peptides: An Alternative for Combating Virulence Mechanisms Developed by Pathogens. Molecules. 2020; 25(24):5763. https://doi.org/10.3390/molecules25245763
Chicago/Turabian StyleZarzosa-Moreno, Daniela, Christian Avalos-Gómez, Luisa Sofía Ramírez-Texcalco, Erick Torres-López, Ricardo Ramírez-Mondragón, Juan Omar Hernández-Ramírez, Jesús Serrano-Luna, and Mireya de la Garza. 2020. "Lactoferrin and Its Derived Peptides: An Alternative for Combating Virulence Mechanisms Developed by Pathogens" Molecules 25, no. 24: 5763. https://doi.org/10.3390/molecules25245763
APA StyleZarzosa-Moreno, D., Avalos-Gómez, C., Ramírez-Texcalco, L. S., Torres-López, E., Ramírez-Mondragón, R., Hernández-Ramírez, J. O., Serrano-Luna, J., & de la Garza, M. (2020). Lactoferrin and Its Derived Peptides: An Alternative for Combating Virulence Mechanisms Developed by Pathogens. Molecules, 25(24), 5763. https://doi.org/10.3390/molecules25245763







