Bioactive Flavonoid Glycosides and HPLC and UPLC Quantification of Commercial Astragali Complanati Semen
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural Elucidation of the Isolated Compounds
2.2. HPLC and UPLC Analysis of Astragali Complanati Semen
2.2.1. The Optimization of Fingerprint Chromatographic Conditions
2.2.2. The Optimization of Complanatuside Quantification Analysis Chromatographic Conditions
2.2.3. Optimization of Extraction Conditions
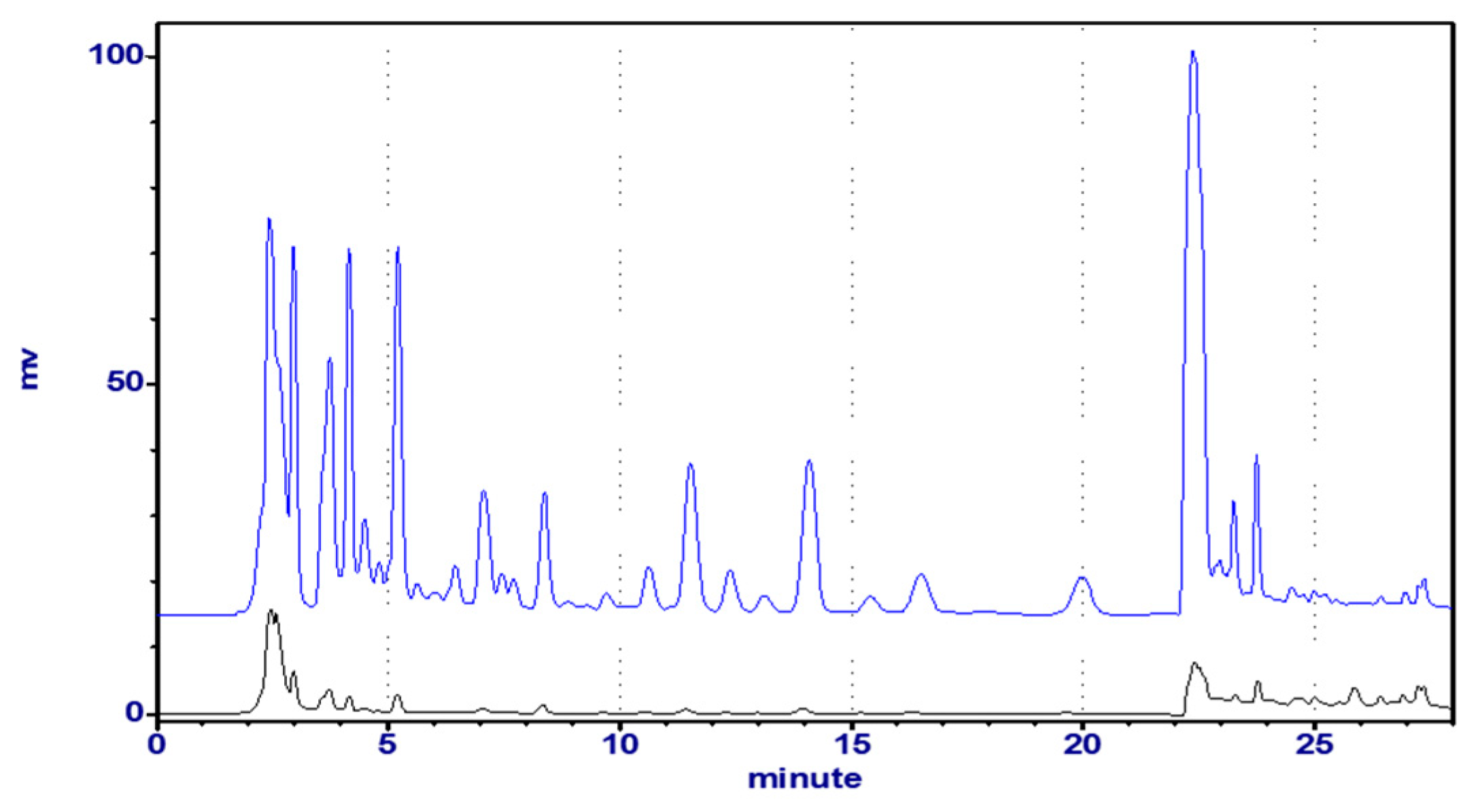
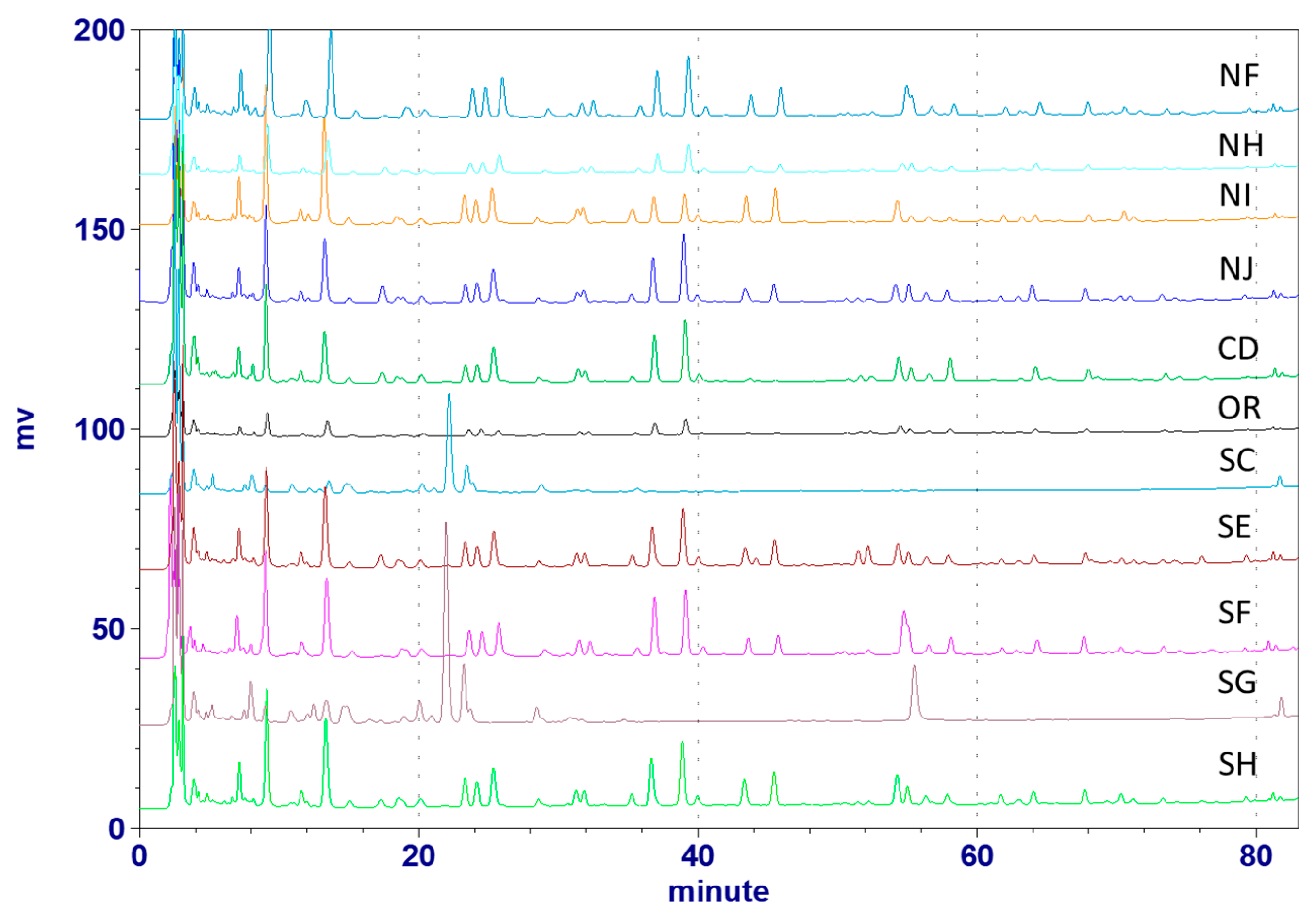
2.2.4. Chromatographic Analyses of Astragali Complanati Semen
2.2.5. Validation of Quantitative Analysis Method
2.2.6. Quantitative Determination of Astragali Complanati Semen
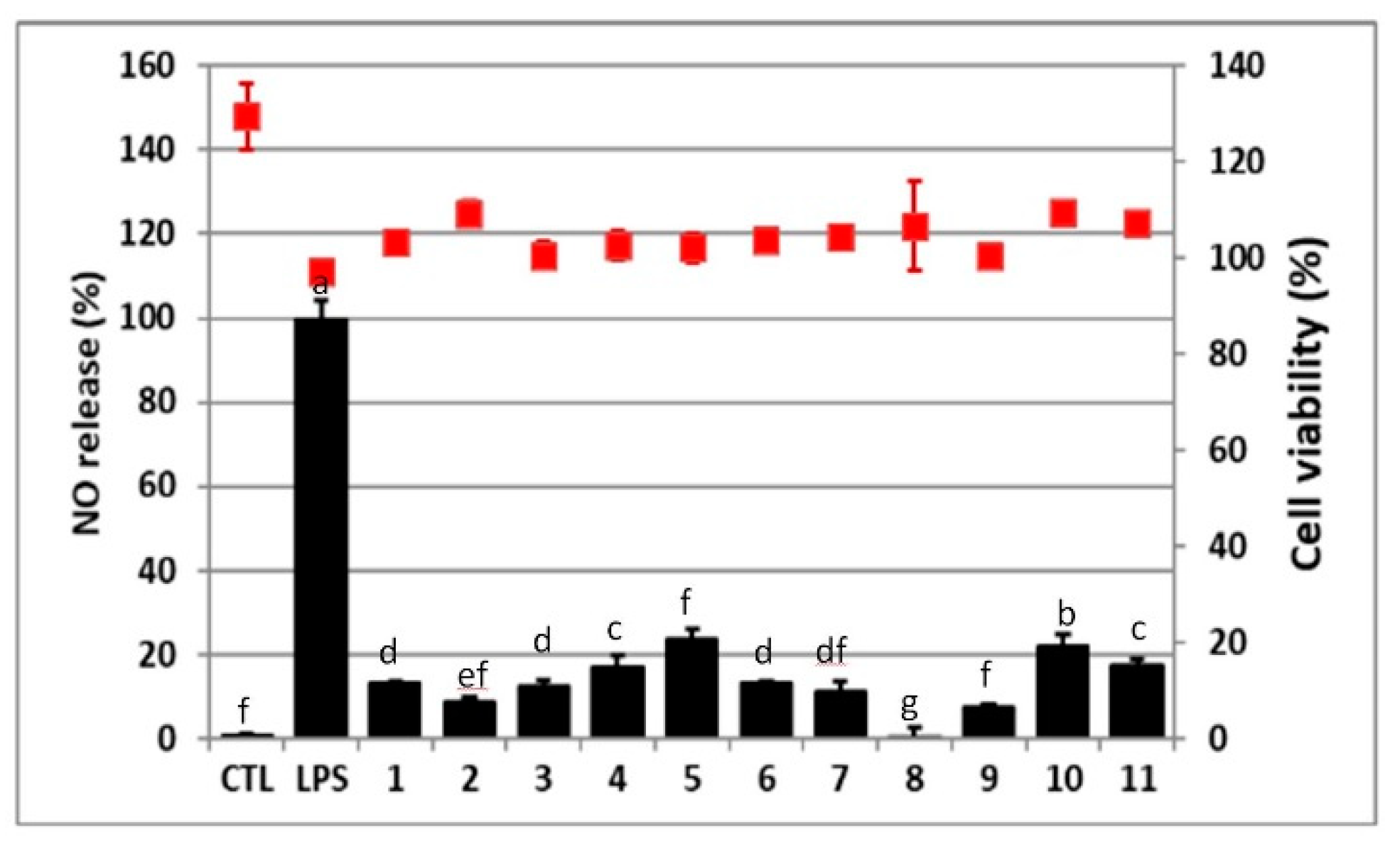
2.3. The Isolates and Fractions of the Antioxidation and Anti-NO Production Activities
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectroscopic Data
3.5. Sample Preparation for HPLC and UPLC Assays
3.6. HPLC Fingerprint Assay
3.7. Complanatuside Quantification Analysis
3.8. Validation Procedure
3.9. Scavenging Activity of DPPH Radical Assay
3.10. LPS-Induced NO Production of Anti-Inflammatory Assays
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SMMC-7721 | human hepatocarcinoma |
| 1D/2D NMR | 1Dimensional/2Dimensional Nuclear Magnetic Resonance |
| EtOH | Ethanol |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| CDCl3 | Chloroform |
| MS | Mass Spectrometry |
| ESIMS | Electrospray Ionization Mass Spectrometry |
| NMR | Nuclear Magnetic Resonance |
| LPS | Lipopolysaccharides |
| UPLC | Ultra Performance Liquid Chromatography |
| HPLC | High Performance Liquid Chromatography |
| ED50 | 50% Effective Dose |
| IC50 | 50% Inhibitory Concentration |
| DEPT | Distortionless Enhancement by Polarization Transfer |
| HMBC | Heteronuclear Multiple Bonds Correlation |
| COSY | Correlation Spectroscopy |
| Rt | Retention time |
| UV | Ultraviolet spectroscopy |
| IR | Infrared spectroscopy |
| HPLC-UV | High Performance Liquid Chromatography with Ultraviolet Detector |
| MeOH | Methanol |
| LOD | Level of Development |
| LOQ | Limit Of Quantitation |
| RSD | Relative Standard Deviation |
| SD | Standard Deviation |
| ANOVA | Analysis Of Variance |
| ELISA | Enzyme-Linked Immunosorbent Assay |
References
- Committee on Chinese Medicine and Pharmacy. Taiwan Herbal Pharmacopoeia, 3rd ed.; Ministry of Health and Welfare: Taipei, Taiwan, 2019; pp. 52–53.
- Pharmacopoeia Commission of People’s Republic of China. Pharmacopoeia of the People’s Republic of China; Medical Science & Technology Press: Beijing, China, 2010; Volume 1, p. 171.
- Ng, Y.-F.; Tang, P.C.-T.; Sham, T.-T.; Lam, W.-S.; Mok, D.K.; Chan, S.-W. Semen Astragali Complanati: An ethnopharmacological, phytochemical and pharmacological review. J. Ethnopharmacol. 2014, 155, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Yang, J.; Analyses, N. Analytical Chemistry Manual VII Fascicle; Chemical Industry Press: Beijing, China, 1999; pp. 901–902. [Google Scholar]
- Martin, C.; Nolen, H.; Podolnick, J.; Wang, R. Current and emerging therapies in premature ejaculation: Where we are coming from, where we are going. Int. J. Urol. 2017, 24, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Cheng, Y.; Wu, K.; Ma, Q.; Jiang, J.; Yan, Z. Paroxetine in the treatment of premature ejaculation: A systematic review and meta-analysis. BMC Urol. 2019, 19, 2. [Google Scholar] [CrossRef] [PubMed]
- Angulo, J.; Peiró, C.; Sanchez-Ferrer, C.F.; Gabancho, S.; Cuevas, P.; Gupta, S.; Sáenz de Tejada, I. Differential effects of serotonin reuptake inhibitors on erectile responses, NO-production, and neuronal NO synthase expression in rat corpus cavernosum tissue. Br. J. Pharm. 2001, 134, 1190–1194. [Google Scholar] [CrossRef]
- Cui, B.; Kinjo, J.; Nakamura, M.; Nohara, T. A novel acylated flavonoid glycoside from Astragalus complanatus. Tetrahedron Lett. 1991, 32, 6135–6138. [Google Scholar] [CrossRef]
- Cui, B.L.; Lu, Y.R.; Wei, L.X. Studies on chemical constituents of Astragalus complanatus R. BR. Yao Xue Xue Bao 1989, 24, 189–193. [Google Scholar]
- Cui, B.; Nakamura, M.; Kinjo, J.; Nohara, T. Structures of three new acylated flavonol glycosides from Astragalus complanatus R. BR. Chem. Pharm. Bull. 1992, 40, 1943–1945. [Google Scholar] [CrossRef][Green Version]
- Cui, B.; Nakamura, M.; Kinjo, J.; Nohara, T. Chemical constituents of Atragali semen. Chem. Pharm. Bull. 1993, 41, 178–182. [Google Scholar] [CrossRef]
- Sham, T.T.; Zhang, H.; Mok, D.K.W.; Chan, S.W.; Wu, J.H.; Tang, S.Y.; Chan, C.O. Chemical anaylsis of Astragali Complanati Semen and Its Hypocholesterolemic effect using serum metabolomics based on gas chromatography-mass spectrometry. Antioxidants 2017, 6, 57. [Google Scholar] [CrossRef]
- Cui, B.; Sakai, Y.; Takeshita, T.; Kinjo, J.; Nohara, T. Four new oleanene derivatives from the seeds of Astragalus complanatus. Chem. Pharm. Bull. 1992, 40, 136–138. [Google Scholar] [CrossRef]
- Liu, C.Y.; Gu, Z.L.; Zhou, W.X.; Guo, C.Y. Effect of Astragalus complanatus flavonoid on anti-liver fibrosis in rats. World J. Gastroenterol. 2005, 11, 5782–5786. [Google Scholar] [CrossRef] [PubMed]
- Xue, B.; Li, J.; Chai, Q.; Liu, Z.; Chen, L. Effect of total flavonoid fraction of Astragalus complanatus R. Brown on angiotensin II-induced portal-vein contraction in hypertensive rats. Phytomedicine 2008, 15, 759–762. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Dong, Y.; Li, X.; Peng, L. Total flavonoids from Astragalus complanatus attenuates lung injury following paraquat poisoning in rats through inhibiting excessive endoplasmic reticulum stress and c-Jun N-terminal kinase pathway. Chin. Crit. Care Med. 2014, 26, 383–387. [Google Scholar] [CrossRef]
- Hu, Y.W.; Liu, C.Y.; Du, C.M.; Zhang, J.; Wu, W.Q.; Gu, Z.L. Induction of apoptosis in human hepatocarcinoma SMMC-7721 cells in vitro by flavonoids from Astragalus complanatus. J. Ethnopharmacol. 2009, 123, 293–301. [Google Scholar] [CrossRef]
- Qi, L.; Liu, C.Y.; Wu, W.Q.; Gu, Z.L.; Guo, C.Y. Protective effect of flavonoids from Astragalus complanatus on radiation induced damages in mice. Fitoterapia 2011, 82, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.A.; Fan, X.H.; Zhang, Z.Q.; Li, T.; Zhu, C.P.; Zhang, X.R.; Song, W. Extraction, antioxidant capacity and identification of Semen Astragali Complanati (Astragalus complanatus R. Br.) phenolics. Food Chem. 2013, 141, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; He, W.; Li, C.; Chen, Q.; Han, L.; Liu, E.; Wang, T. Antioxidative flavonol glycosides from the flowers of Abelmouschus manihot. J. Nat. Med. 2013, 67, 78–85. [Google Scholar] [CrossRef]
- Lu, Y.; Sun, Y.; Foo, L.Y.; McNabb, W.C.; Molan, A.L. Phenolic glycosides of forage legume Onobrychis viciifolia. Phytochemistry 2000, 55, 67–75. [Google Scholar] [CrossRef]
- Xu, S.; Duan, W.; Fang, L.; Sun, Y.; Wang, X. Isolation and characterization of chemical constituents from the petals of Nelumbo nucifera. Asian J. Chem. 2012, 24, 4619–4622. [Google Scholar]
- Beninger, C.W.; Hosfield, G.L. Flavonol glycosides from Montcalm dark red kidney bean: Implications for the genetics of seed coat color in Phaseolus vulgaris L. J. Agric. Food Chem. 1999, 47, 4079–4082. [Google Scholar] [CrossRef]
- Saito, N.; Toki, K.; Honda, T.; Tatsuzawa, F. Floral pigments isolated from the sky-blue flowers of Oxypetalum caeruleum. Heterocycles 2012, 85, 1427–1436. [Google Scholar]
- Hari Kishore, P.; Vijaya Bhaskar Reddy, M.; Gunasekar, D.; Marthanda Murthy, M.; Caux, C.; Bodo, B. A new coumestan from Tephrosia calophylla. Chem. Pharm. Bull. 2003, 51, 194–196. [Google Scholar] [CrossRef]
- Kirecci, S.L.; Simsek, A.; Yuksel, A.; Gurdal, H.; Gurbuz, Z.G.; Usanmaz, S. Relevance of seminal plasma nitric oxide levels and the efficacy of SSRI treatment on lifelong premature ejaculation. Andrologia 2014, 46, 1169–1175. [Google Scholar] [CrossRef] [PubMed]
- Hirakura, K.; Morita, M.; Nakajima, K.; Sugama, K.; Takagi, K.; Niitsu, K.; Ikeya, Y.; Maruno, M.; Okada, M. Phenolic glucosides from the root of Pueraria lobata. Phytochemistry 1997, 46, 921–928. [Google Scholar] [CrossRef]
- Fiorentino, A.; D’Abrosca, B.; Pacifico, S.; Mastellone, C.; Piscopo, V.; Monaco, P. Spectroscopic identification and antioxidant activity of glucosylated carotenoid metabolites from Cydonia vulgaris fruits. J. Agric. Food. Chem. 2006, 54, 9592–9597. [Google Scholar] [CrossRef]
- Zhang, Q.A.; Fan, X.H.; Zhang, Z.Q.; Wang, Q.; Wang, J. Antioxidant for phenolic extracts from Semen Astragali Complanati. Nat. Prod. Res. Dev. 2012, 23, 955–958. [Google Scholar]
- Xiao, A.Z.; Wang, Z.; Gu, S.C.; Xin, Y.M. The anti-aging effect of Semen Astragali Complanati. Flight Surg. 2004, 32, 155–156. (In Chinese) [Google Scholar]
- Wei, C.P.; Zhou, D.H. Study on anti-aging effects of flavonoids of Astragali Complanati on subacute aging mice induced by D-galactose. Chin. Gen. Pract. 2010, 18, 160–162. [Google Scholar]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Kim, M.S.; Pang, G.C.; Lee, M.W. Flavonoids from the leaves of Rubus coreanum. Yakhak Hoeji 1997, 41, 1–6. [Google Scholar]
- Cassidy, A.; Franz, M.; Rimm, E.B. Dietary flavonoid intake and incidence of erectile dysfunction. Am. J. Clin. Nutr. 2016, 103, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Rangkadilok, N.; Sitthimonchai, S.; Worasuttayangkurn, L.; Mahidol, C.; Ruchirawat, M.; Satayavivad, J. Evaluation of free radical scavenging and antityrosinase activities of standardized longan fruit extract. Food Chem. Toxicol. 2007, 45, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Chiou, W.F.; Lin, J.J.; Chen, C.F. Andrographolide suppresses the expression of inducible nitric oxide synthase in macrophage and restores the vasoconstriction in rat aorta treated with lipopolysaccharide. Br. J. Pharmacol. 1998, 125, 327–334. [Google Scholar] [CrossRef] [PubMed]
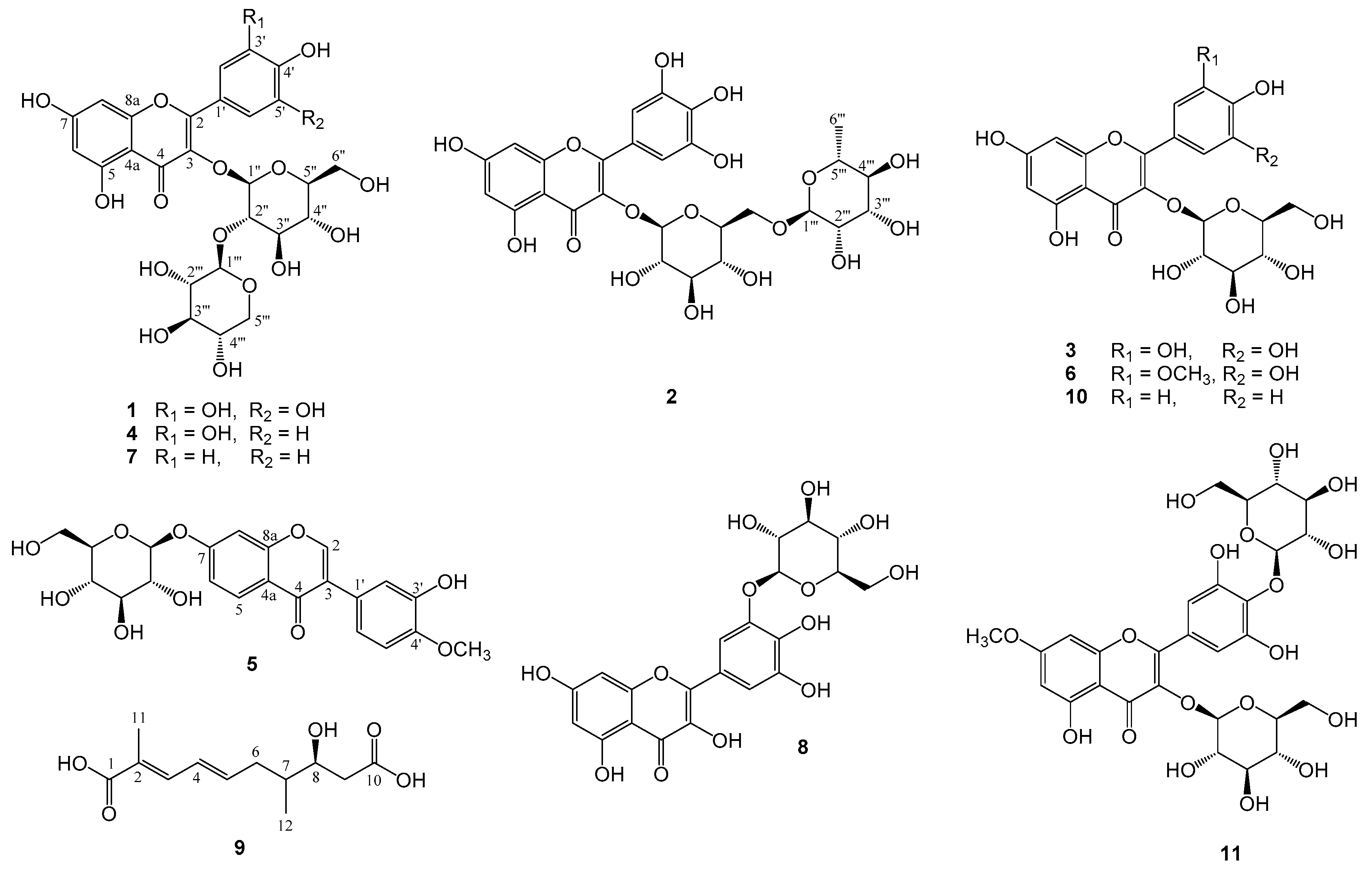
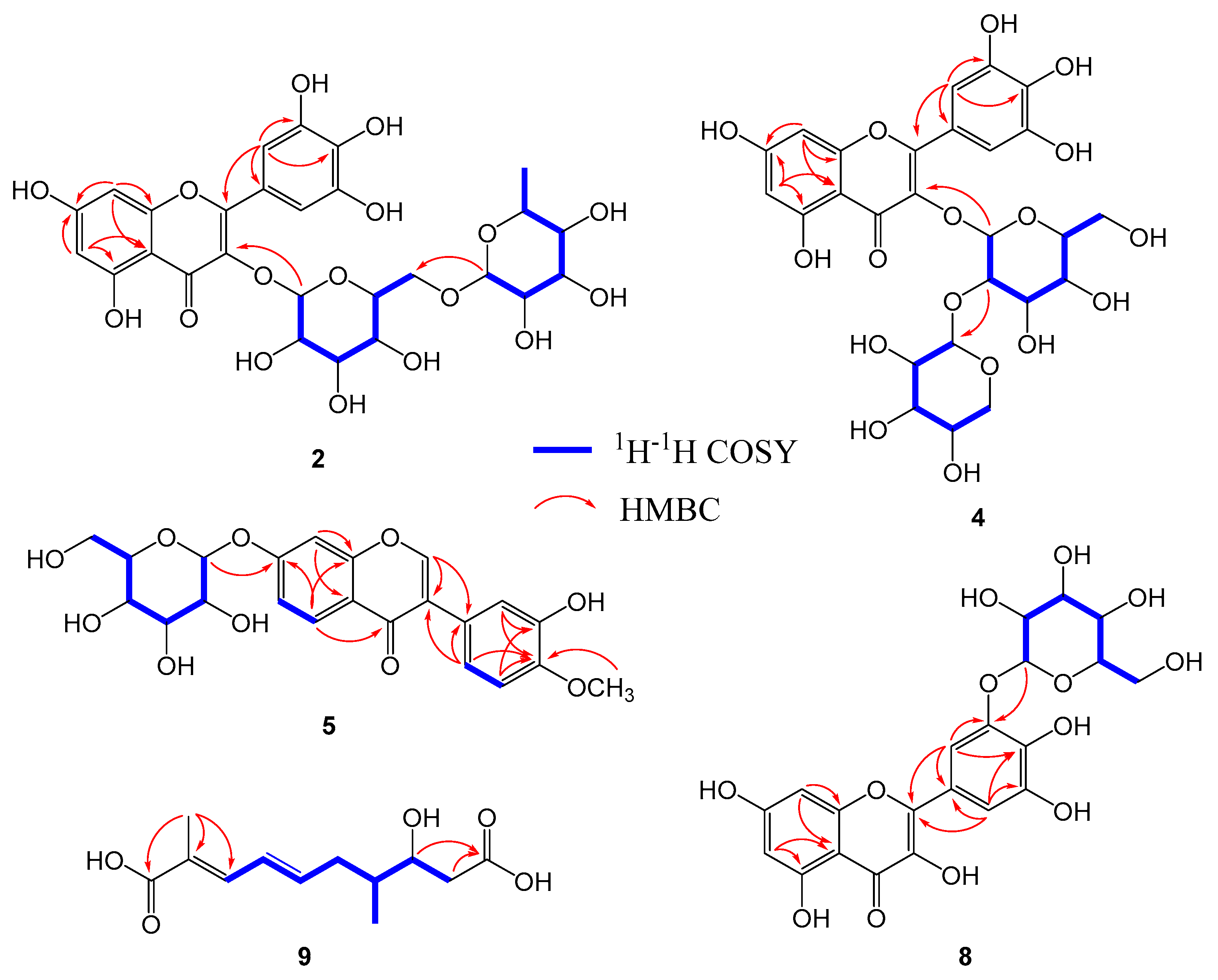
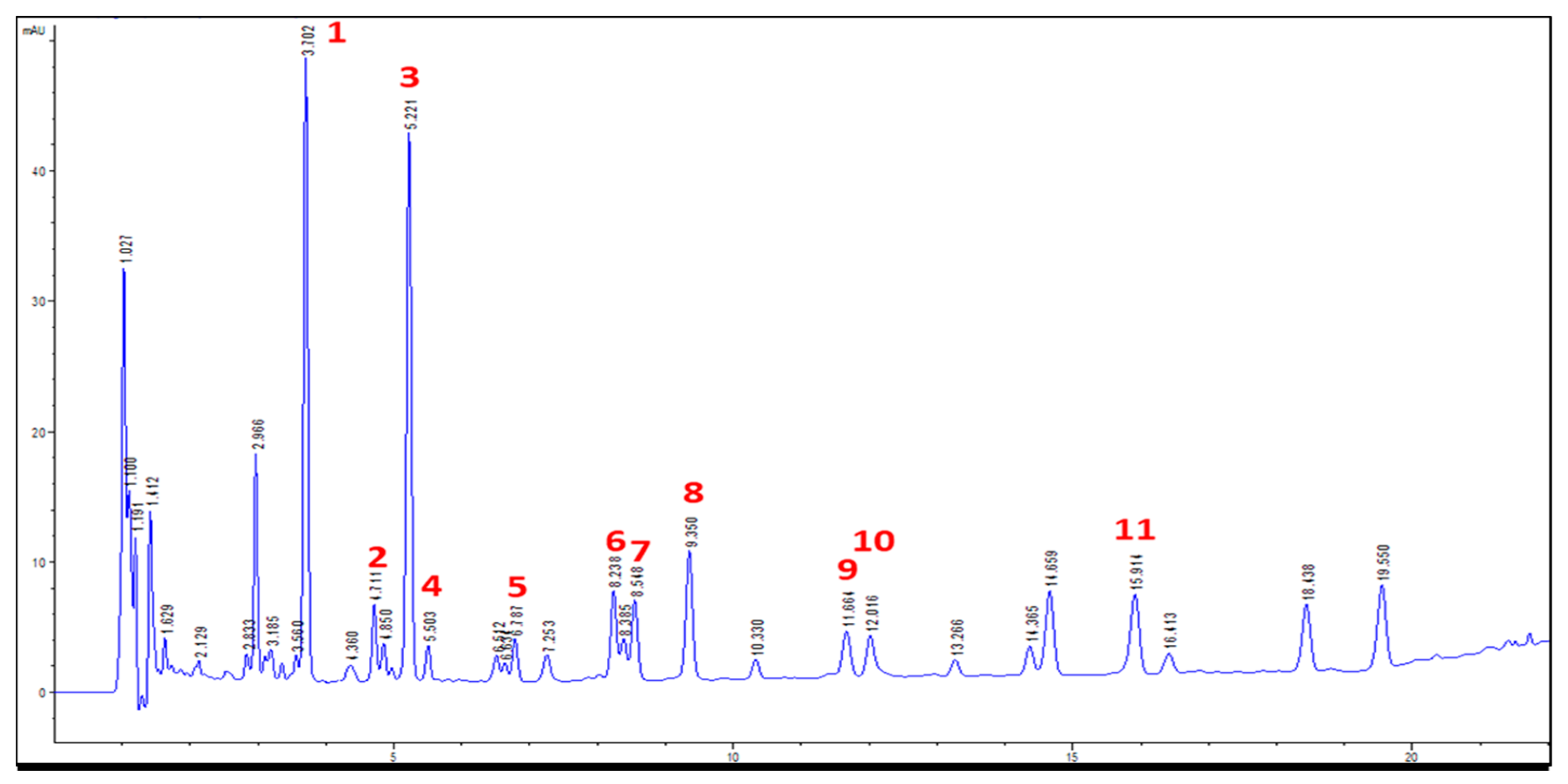
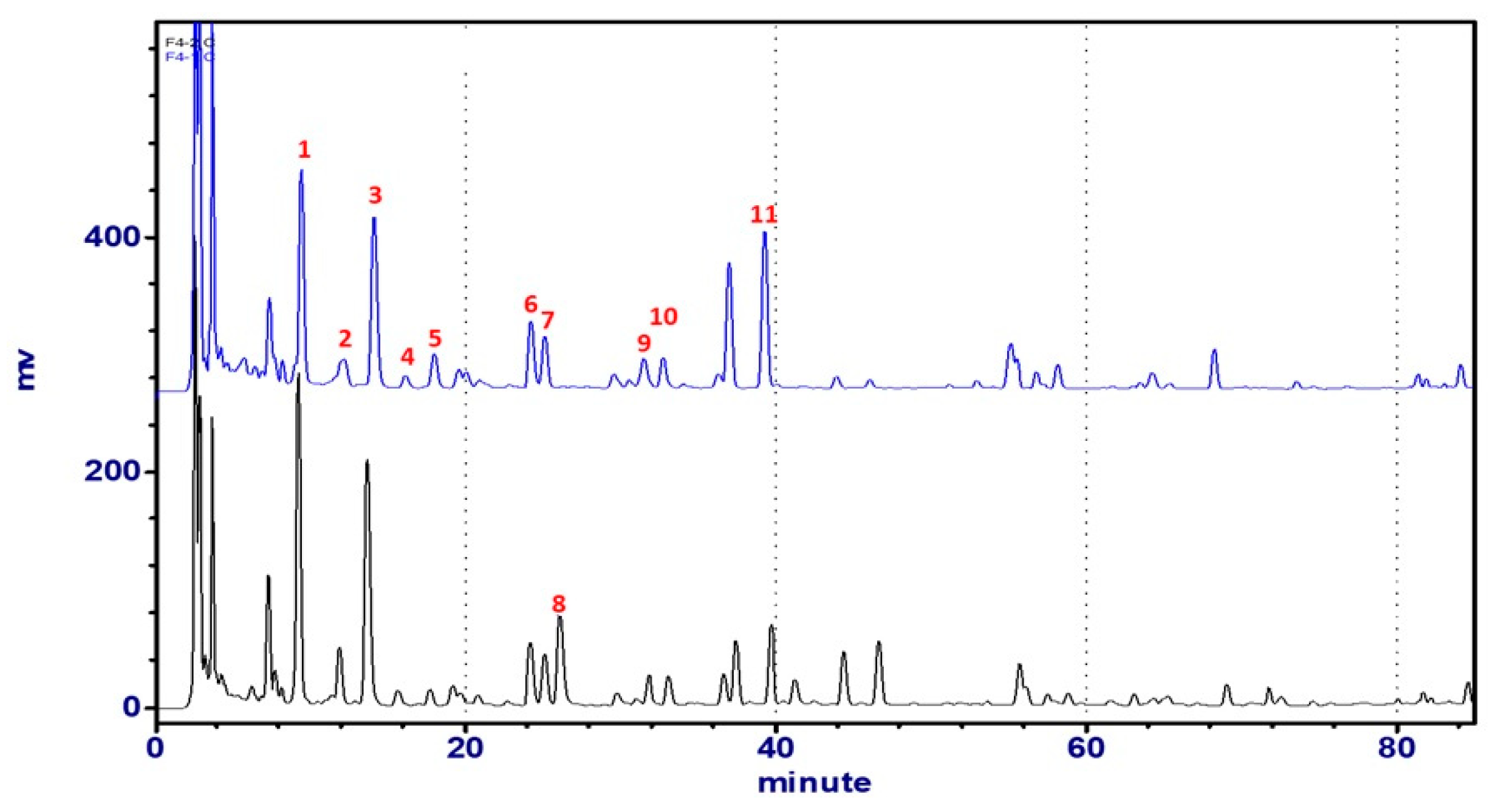
| No. | 1 a | 2 a | 3 b | 4 a | 5 b | 6 a | 7 a | 8 b | 10 b | 11 b |
|---|---|---|---|---|---|---|---|---|---|---|
| 2 | 158.4 | 159.4 | 156.5 | 158.4 | 153.8 | 158.3 | 158.3 | 146.6 | 156.7 | 156.6 |
| 3 | 135.3 | 135.8 | 133.7 | 135.1 | 124.6 | 135.6 | 135.0 | 136.3 | 133.5 | 134.2 |
| 4 | 179.5 | 179.4 | 177.6 | 179.6 | 175.1 | 179.3 | 179.6 | 176.1 | 177.8 | 177.9 |
| 4a | 105.8 | 105.6 | 104.1 | 105.8 | 118.7 | 105.7 | 105.7 | 103.3 | 104.3 | 105.3 |
| 5 | 163.1 | 163.0 | 161.4 | 163.1 | 127.3 | 162.9 | 163.1 | 164.2 | 161.5 | 161.1 |
| 6 | 99.7 | 100.0 | 98.8 | 99.8 | 115.9 | 99.8 | 99.9 | 98.5 | 99.1 | 98.2 |
| 7 | 165.8 | 166.4 | 164.3 | 165.9 | 161.7 | 165.9 | 166.1 | 164.2 | 164.4 | 165.5 |
| 8 | 94.5 | 94.9 | 93.6 | 94.6 | 103.7 | 94.7 | 94.7 | 93.90 | 94.0 | 92.6 |
| 8a | 158.4 | 158.5 | 156.5 | 158.3 | 157.3 | 158.6 | 158.5 | 156.4 | 156.7 | 156.2 |
| 1′ | 122.1 | 122.3 | 120.2 | 123.2 | 123.9 | 121.9 | 122.8 | 121.3 | 121.2 | 123.8 |
| 2′ | 109.9 | 110.3 | 108.7 | 116.1 | 116.6 | 106.7 | 135.0 | 107.7 | 115.5 | 116.0 |
| 3′ | 146.5 | 146.4 | 145.6 | 146.1 | 146.2 | 148.9 | 116.2 | 146.6 | 131.3 | 130.9 |
| 4′ | 137.9 | 138.0 | 136.8 | 149.7 | 147.9 | 138.7 | 161.5 | 137.5 | 160.2 | 159.5 |
| 5′ | 146.5 | 146.4 | 145.6 | 117.3 | 112.2 | 146.1 | 116.2 | 146.6 | 131.3 | 130.9 |
| 6′ | 109.9 | 110.3 | 108.7 | 123.4 | 120.1 | 111.1 | 135.0 | 110.9 | 115.5 | 116.0 |
| OCH3 | 55.9 | 57.0 | 56.4 | |||||||
| 1″ | 101.0 | 102.4 | 101.0 | 100.9 | 100.2 | 103.8 | 100.8 | 102.9 | 101.2 | 101.0 |
| 2″ | 82.2 | 75.7 | 74.1 | 82.3 | 73.3 | 75.9 | 82.4 | 73.5 | 74.5 | 74.4 |
| 3″ | 76.9 | 78.2 | 76.7 | 77.0 | 76.6 | 78.1 | 77.1 | 76.1 | 767 | 76.7 |
| 4″ | 70.9 | 71.4 | 70.0 | 71.0 | 69.9 | 71.4 | 71.1 | 69.9 | 70.2 | 70.1 |
| 5″ | 78.2 | 77.2 | 77.7 | 78.2 | 77.4 | 78.4 | 78.2 | 77.4 | 77.7 | 77.7 |
| 6″ | 62.3 | 68.6 | 61.2 | 62.4 | 60.9 | 62.5 | 62.4 | 60.9 | 61.1 | 61.0 |
| 1‴ | 105.2 | 104.8 | 105.4 | 105.5 | 100.1 | |||||
| 2‴ | 74.8 | 72.1 | 74.9 | 75.0 | 73.4 | |||||
| 3‴ | 78.3 | 72.1 | 78.4 | 78.4 | 76.6 | |||||
| 4‴ | 70.5 | 74.0 | 71.0 | 71.0 | 69.8 | |||||
| 5‴ | 66.5 | 69.7 | 66.6 | 66.7 | 77.2 | |||||
| 6‴ | 17.9 | 60.8 |
| Method Validation Item | Complanatuside (11) | |
|---|---|---|
| Concentration (μg/mL) | RSD % | |
| Injection precision (n = 5) | 15.0 | 1.35 |
| Intraday (n = 3) | 60.0 | 0.80 |
| 15.0 | 1.35 | |
| 1.9 | 1.00 | |
| Inter-day (n = 9) | 60.0 | 1.18 |
| 15.0 | 2.01 | |
| 1.9 | 2.99 | |
| Reproducibility (n = 5) | SF (0.5 g) | 4.25 |
| Stability (n = 5) | SF (0.5 g) | 1.04 |
| LOD (S/N = 3 μg/mL, n = 1) | 0.019 | - |
| LOQ (S/N = 10 μg/mL, n = 1) | 0.038 | - |
| No. | Original (mg) | Addition (mg) | Detection (mg) | Recovery (%) |
|---|---|---|---|---|
| 1 | 0.6001 | 0.389 | 1.0051 | 104.12 |
| 2 | 0.6006 | 0.389 | 0.9870 | 99.34 |
| 3 | 0.6013 | 0.389 | 0.9983 | 102.07 |
| 4 | 0.6002 | 0.389 | 0.9930 | 100.99 |
| 5 | 0.6001 | 0.389 | 1.0208 | 108.14 |
| Average | 0.6005 | 0.389 | 1.0009 | 102.93 |
| RSD (%) | 0.085 | - | 1.30 | 3.30 |
| Samples | Complanatuside (%) | Samples | Complanatuside (%) |
|---|---|---|---|
| NF | 0.106 ± 0.004 * | SC | 0 # |
| NH | 0.098 ± 0.001 * | SE | 0.115 ± 0.004 ** |
| NI | 0.095 ± 0.002 * | SF | 0.120 ± 0.002 ** |
| NJ | 0.134 ± 0.002 ** | SG | 0# |
| CD | 0.116 ± 0.006 * | SH | 0.109 ± 0.003 ** |
| OR | 0.062 ± 0.002 | THP/CHP | 0.06 |
| The Extract | Removal Effect (200 μg/mL, %) | ED50 (μg/mL) | Compound | Removal Effect (100 μg/mL, %) | ED50 (μM) |
|---|---|---|---|---|---|
| 80E | 53.34 ± 1.26 | 180.42 ± 5.22 | 1 | 98.93 ± 0.01 a | 14.34 ± 0.59 ab |
| 80EH | 10.67 ± 1.17 | (-) b* | 2 | 98.39 ± 0.00 a | 16.59 ± 0.85 b |
| 80ED0 | 13.12 ± 0.72 | (-) b* | 3 | 99.73 ± 0.13 a | 19.27 ± 0.46 b |
| 80ED25 | 99.23 ± 0.33 | 37.39 ± 0.33 | 4 | 94.54 ± 0.13 b | 39.36 ± 1.26 d |
| 80ED50 | 96.20 ± 3.00 | 50.89 ± 0.13 | 5 | 58.77 ± 0.13 c | 200.06 ± 0.63 g |
| 80ED75 | 57.31 ± 1.18 | 164.29 ± 3.58 | 6 | 97.23 ± 0.13 ab | 33.18 ± 1.64 c |
| 80ED95 | 64.61 ± 1.64 | 139.74 ± 3.39 | 7 | 1.97 ± 1.01 f | (-) c* |
| 80EDEA | 34.29 ± 0.62 | (-) b* | 8 | 99.55 ± 0.13 a | 14.77 ± 0.44 a |
| 9 | 5.37 ± 3.79 e | (-) c* | |||
| 10 | 31.57 ± 1.39 d | (-) c* | |||
| 11 | 0.15 ± 0.28 g | (-) c* | |||
| Vitamin C | - d* | 4.34 ± 0.28 | Vitamin C | - | 24.64 ± 1.58 d |
| Vitamin E | - | 12.67 ± 0.09 | Vitamin E | - | 29.42 ± 0.21 f |
Sample Availability: Samples of the compounds 1–11 are available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuo, J.C.-L.; Zhang, L.-J.; Huang, H.-T.; Liaw, C.-C.; Lin, Z.-H.; Liu, M.; Kuo, Y.-H. Bioactive Flavonoid Glycosides and HPLC and UPLC Quantification of Commercial Astragali Complanati Semen. Molecules 2020, 25, 4762. https://doi.org/10.3390/molecules25204762
Kuo JC-L, Zhang L-J, Huang H-T, Liaw C-C, Lin Z-H, Liu M, Kuo Y-H. Bioactive Flavonoid Glycosides and HPLC and UPLC Quantification of Commercial Astragali Complanati Semen. Molecules. 2020; 25(20):4762. https://doi.org/10.3390/molecules25204762
Chicago/Turabian StyleKuo, Jenny Chun-Ling, Li-Jie Zhang, Hung-Tse Huang, Chia-Ching Liaw, Zhi-Hu Lin, Min Liu, and Yao-Haur Kuo. 2020. "Bioactive Flavonoid Glycosides and HPLC and UPLC Quantification of Commercial Astragali Complanati Semen" Molecules 25, no. 20: 4762. https://doi.org/10.3390/molecules25204762
APA StyleKuo, J. C.-L., Zhang, L.-J., Huang, H.-T., Liaw, C.-C., Lin, Z.-H., Liu, M., & Kuo, Y.-H. (2020). Bioactive Flavonoid Glycosides and HPLC and UPLC Quantification of Commercial Astragali Complanati Semen. Molecules, 25(20), 4762. https://doi.org/10.3390/molecules25204762





