Innovative Approaches for Recovery of Phytoconstituents from Medicinal/Aromatic Plants and Biotechnological Production
Abstract
1. Introduction
2. Challenges in the Process “from Plants to Pure Molecules”
3. An Overview of the Biotechnological Aspects for Obtaining Phytochemicals from MAP
4. Recovery of Phytochemicals from Biotechnological Production
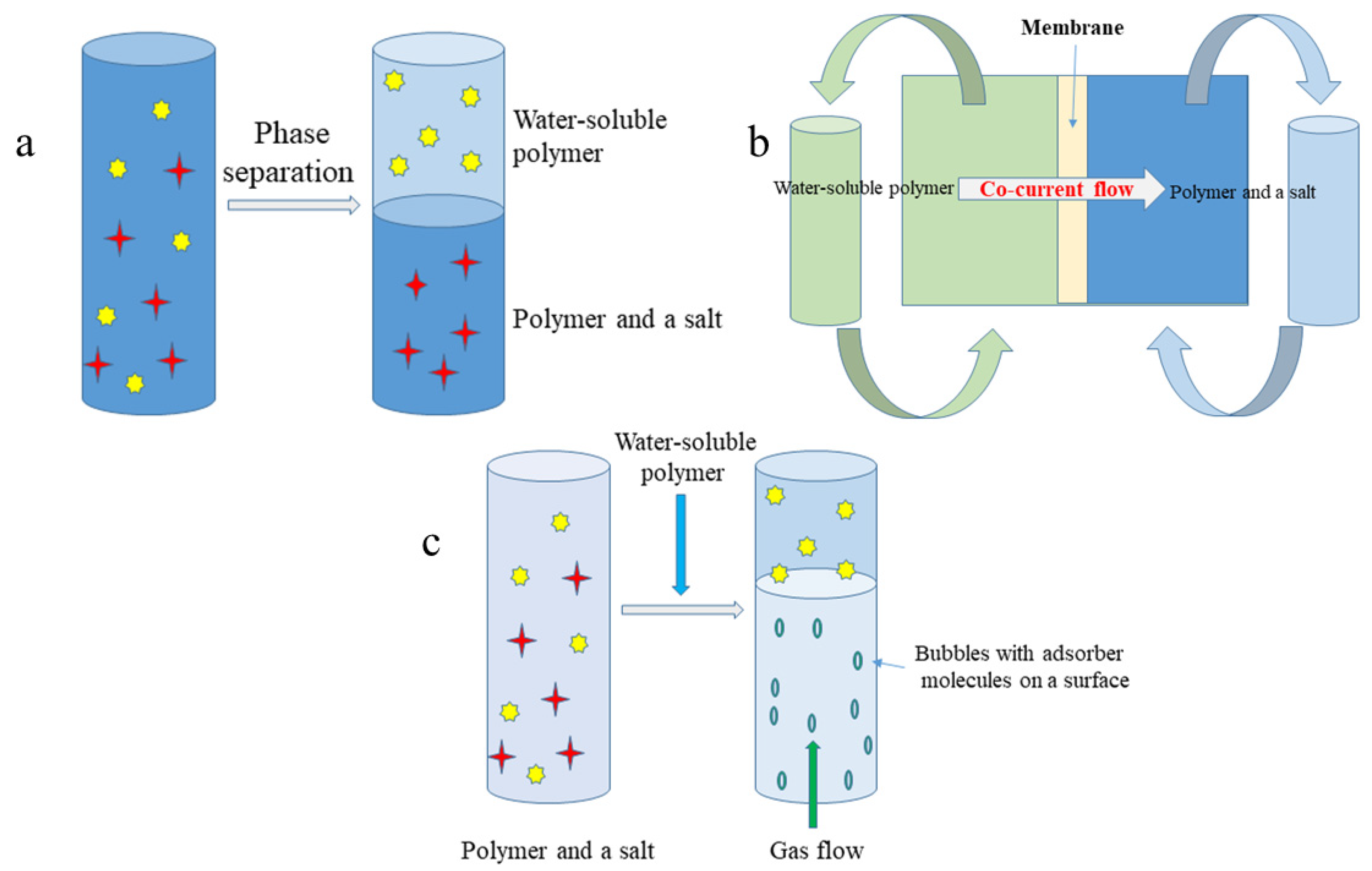
4.1. Non-Disruptive Processing
4.1.1. Liquid-Liquid Extraction
4.1.2. Natural Deep Eutectic Solvents
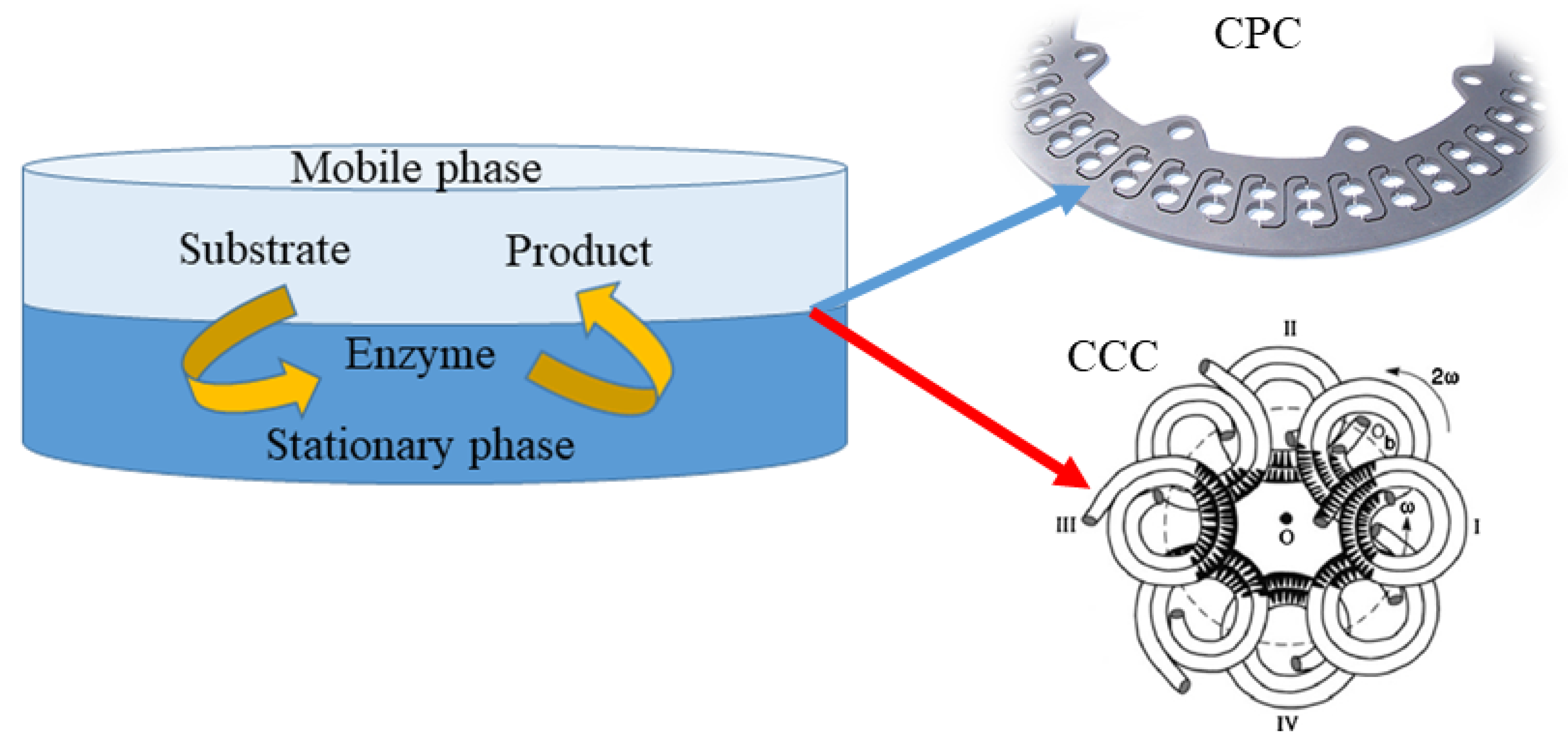
4.1.3. Counter Current Chromatography and Centrifugal Partition Chromatography
4.1.4. Ultrasonic Assisted Extraction
4.1.5. Supercritical Fluid Extraction
4.1.6. Supercritical Reactive Extraction
4.2. The Recovery of Metabolites from Disrupted Cells
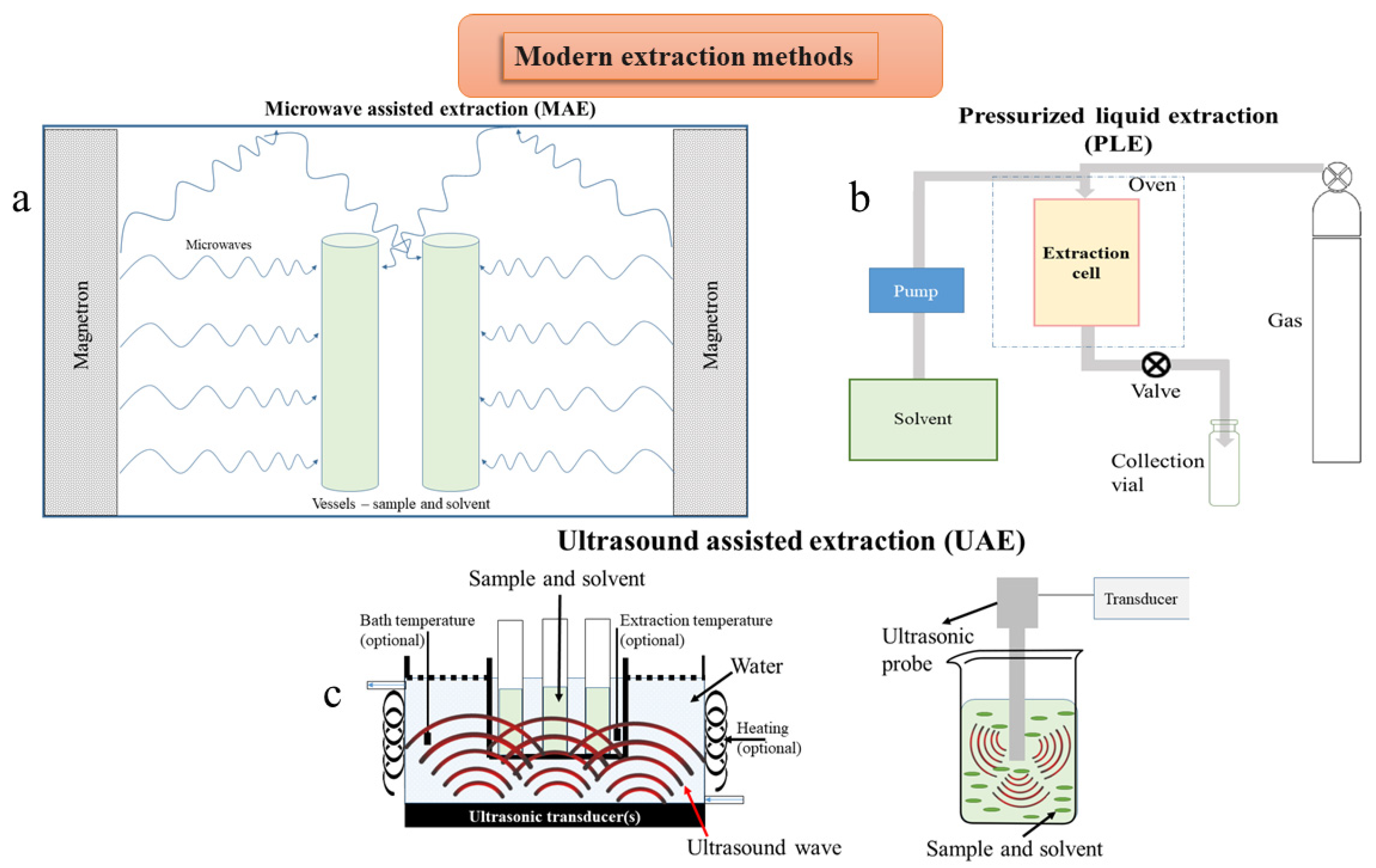
4.2.1. Microwave Assisted Extraction
4.2.2. Ultrasound Assisted Extraction
4.3. Simultaneous (Bio)Conversion and Separation with Application of Counter Current Chromatography
5. Advanced Extractions Techniques for Obtaining Biologically Active Compounds from MAP
5.1. Classical and Modern Extraction Techniques
5.1.1. Microwave Assisted Extraction
5.1.2. Pressurized Liquid Extraction
5.1.3. Ultrasound Assisted Extraction
5.2. Other Extraction Techniques
5.2.1. Ohmic Assisted Technologies
5.2.2. High Voltage Electric Discharge
5.2.3. Pulsed Electric Field
6. Experimental Design of Phytoconstituents’ Production and Recovery
7. Future Directions
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Murthy, H.N.; Georgiev, M.I.; Park, S.Y.; Dandin, V.S.; Paek, K.Y. The safety assessment of food ingredients derived from plant cell, tissue and organ cultures: A review. Food Chem. 2015, 176, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Croteau, R.; Kutchan, T.M.; Lewis, N.G. Natural products (secondary metabolites). In Biochemistry & Molecular Biology of Plants; Buchanan, B., Gruissem, W., Jones, R., Eds.; American Society of Plant Physiologists: Rockville, MD, USA, 2000; pp. 1250–1318. [Google Scholar]
- WHO Global Report on Traditional and Complementary Medicine; World Health Organization: Geneva, Switzerland, 2019; Available online: https://www.who.int/traditional-complementary-integrative-medicine/WhoGlobalReportOnTraditionalAndComplementaryMedicine2019.pdf?ua=1 (accessed on 9 January 2020).
- Puértolas, E.; Koubaa, M.; Barba, F.J. An overview of the impact of electrotechnologies for the recovery of oil and high-value compounds from vegetable oil industry: Energy and economic cost implications. Food Res. Int. 2016, 80, 19–26. [Google Scholar] [CrossRef]
- Bucar, F.; Wube, A.; Schmid, M. Natural product isolation, how to get from biological material to pure compounds. Nat. Prod. Rep. 2013, 30, 525–545. [Google Scholar] [CrossRef] [PubMed]
- Coors, A.; Brosch, M.; Kahl, E.; Khalil, R.; Michels, B.; Laub, A.; Franke, K.; Gerber, B.; Fendt, M. Rhodiola rosea root extract has antipsychotic-like effects in rodent models of sensorimotor gating. J. Ethnopharmacol. 2019, 235, 320–328. [Google Scholar] [CrossRef]
- Luo, C.; Xu, X.; Wei, X.; Feng, W.; Huang, H.; Liu, H.; Xu, R.; Lin, J.; Han, L.; Zhang, D. Natural medicines for the treatment of fatigue: Bioactive components, pharmacology, and mechanisms. Pharmacol. Res. 2019, 148, 104409. [Google Scholar] [CrossRef]
- Allen, D.; Bilz, M.; Leaman, D.J.; Miller, R.M.; Timoshyna, A.; Window, J. European Red List of Medicinal Plants; Publications Office of the European Union: Luxembourg, 2014; Available online: https://op.europa.eu/en/publication-detail/-/publication/d426ab4d-fc82-11e5-b713-01aa75ed71a1/language-en, (accessed on 20 September 2019).
- CORDIS. Final Report Summary, CROPS2INDUSTRY (Non-Food Crops-to-Industry Schemes in EU27). 2014. Available online: https://cordis.europa.eu/project/rcn/100484/reporting/en (accessed on 2 August 2019).
- Cragg, G.M.; Schepartz, S.A.; Suffness, M.; Grever, M.R. The taxol supply crisis. New NCI policies for handling the large-scale production of novel natural product anticancer and anti-HIV agents. J. Nat. Prod. 1993, 56, 1657–1668. [Google Scholar] [CrossRef]
- Knoess, W.; Wiesner, J. The globalization of traditional medicines: Perspectives related to the European Union regulatory environment. Engineering 2019, 5, 22–31. [Google Scholar] [CrossRef]
- Carvalho, A.C.B.; Lana, T.N.; Perfeito, J.P.S.; Silveira, D. The Brazilian market of herbal medicinal products and the impacts of the new legislation on traditional medicines. J. Ethnopharmacol. 2018, 212, 29–35. [Google Scholar] [CrossRef]
- Georgiev, M.I.; Weber, J.; Maciuk, A. Bioprocessing of plant cell cultures for mass production of targeted compounds. Appl. Microbiol. Biotechnol. 2009, 83, 809–823. [Google Scholar] [CrossRef]
- Lim, E.-K.; Bowles, D. Plant production systems for bioactive small molecules. Curr. Opin. Biotechnol. 2012, 23, 271–277. [Google Scholar] [CrossRef]
- Wilson, S.A.; Roberts, S.C. Recent advances towards development and commercialization of plant cell culture processes for the synthesis of biomolecules. Plant Biotechnol. J. 2012, 10, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, M.I.; Eibl, R.; Zhong, J.J. Hosting the plant cells in vitro: Recent trends in bioreactors. Appl. Microbiol. Biotechnol. 2013, 97, 3787–3800. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, M.; Weber, J. Bioreactors for plant cells: Hardware configuration and internal environment optimization as tools for wider commercialization. Biotechnol. Lett. 2014, 36, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- Eibl, R.; Meier, P.; Stutz, I.; Schildberger, D.; Huhn, T.; Eibl, D. Plant cell culture technology in the cosmetics and food industries: Current state and future trends. Appl. Microbiol. Biotechnol. 2018, 102, 8661–8675. [Google Scholar] [CrossRef]
- Available online: www.phytonbiotech.com (accessed on 4 June 2019).
- Zhong, J.J. Plant cell culture for production of paclitaxel and other taxanes. J. Biosci. Bioeng. 2002, 94, 591–599. [Google Scholar] [CrossRef]
- Murthy, H.N.; Georgiev, M.I.; Kim, Y.-S.; Jeong, C.-S.; Kim, S.-J.; Paek, K.-Y. Ginsenosides: Prospective for sustainable biotechnological production. Appl. Microbiol. Biotechnol. 2014, 98, 6243–6254. [Google Scholar] [CrossRef]
- Kim, H.K.; Choi, Y.H.; Verpoorte, R. NMR based metabolomics: Where do we stand, where do we go? Trends Biotechnol. 2011, 29, 267–275. [Google Scholar] [CrossRef]
- Walsh, G. Biopharmaceutical benchmarks. Nature Biotechnol. 2010, 28, 917–924. [Google Scholar] [CrossRef]
- Dos Santos, N.V.; de Carvalho Santos-Ebinuma, V.; Pessoa Jr, A.; Brandão Pereira, J.F. Liquid-liquid extraction of biopharmaceuticals from fermented broth: Trends and future prospects. J. Chem. Technol. Biotechnol. 2018, 93, 1845–1863. [Google Scholar] [CrossRef]
- Stanbury, P.F.; Whitaker, A.; Hall, S.J. The recovery and purification of fermentation products. In Principles of Fermentation Technologym; Stanbury, P.F., Whitaker, A., Hall, S.J., Eds.; Elsevier: Oxford, UK, 2017; pp. 619–686. [Google Scholar]
- Yang, Z. Natural Deep Eutectic Solvents and Their Applications in Biotechnology; Springer: Berlin, Germany, 2018; pp. 1–27. [Google Scholar]
- Carta, G.; Jungbauer, A. Protein Chromatography: Process Development and Scale-Up; Wiley-VCH: Weinheim, Germany, 2010. [Google Scholar]
- Aguilar, O. Aqueous two phase systems for the recovery and purification of bioproducts from plants and vegetable tissues. In Aqueous Two Phase Systems for Bioprocess Development for the Recovery of Biological Products; Rito-Palomares, M., Benavides, J., Eds.; Springer Nature: Cham, Germany, 2017; pp. 123–140. [Google Scholar]
- Rosa, P.A.J.; Azevedo, A.M.; Sommerfeld, S.; Bäcker, W.; Aires-Barros, M.R. Aqueous two phase extraction as a platform in the biomanufacturing industry: Economical and environmental sustainability. Biotechnol. Adv. 2011, 29, 559–567. [Google Scholar] [CrossRef]
- Doran, P.M. Bioprocess Engineering Principles; Academic press: Waltham, MA, USA, 2005. [Google Scholar]
- Riedl, W.; Raiser, T. Membrane-supported extraction of biomolecules with aqueous two phase systems. Desalination 2008, 224, 160–167. [Google Scholar] [CrossRef]
- Riedl, W.; Mollet, D.; Grundle, G. Using Membrane-Supported Liquid–Liquid Extraction for the Measurement of Extraction Kinetics. Chimia 2011, 65, 370–372. [Google Scholar] [CrossRef] [PubMed]
- Bi, P.Y.; Li, D.Q.; Dong, H.R. A novel technique for the separation and concentration of penicillin G from fermentation broth: Aqueous two phase flotation. Sep. Purif. Technol. 2009, 69, 205–209. [Google Scholar] [CrossRef]
- Tikhomiroff, C.; Allais, S.; Klvana, M.; Hisiger, S.; Jolicoeur, M. Continuous selective extraction of secondary metabolites from Catharanthus roseus hairy roots with silicon oil in a two-liquid phase bioreactor. Biotechnol. Prog. 2002, 18, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Ohtani, T.; Kondo, K. Comparison of solvent extraction and supported liquid membrane permeation using an ionic liquid for concentrating penicillin G. J. Membr. Sci. 2007, 289, 92–96. [Google Scholar] [CrossRef]
- Maugeri, Z.; Domínguez de María, P. Benzaldehyde lyase (BAL)-catalyzed enantioselective C-C bond formation in deep-eutectic solvents− buffer mixtures. J. Mol. Catal. B: Enzym. 2014, 107, 120–123. [Google Scholar] [CrossRef]
- Yang, Z.; Wen, Q. Deep eutectic solvents as a new reaction medium for biotransformations. In Ionic Liquid based Surfactant Science: Formulation, Characterization and Applications; Paul, B.K., Moulik, S.P., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 517–531. [Google Scholar]
- Khodaverdiana, S.; Dabirmanesh, B.; Heydarib, A.; Dashtban-Moghadama, E.; Khajeha, K.; Ghazi, F. Activity, stability and structure of laccase in betaine based natural deep eutectic solvents. Int. J. Biol. Macromol. 2018, 107, 2574–2579. [Google Scholar] [CrossRef]
- Francisco, M.; van den Bruinhorst, A.; Kroon, M.C. Low-transition temperature mixtures (LTTMs): A new generation of designer solvents. Angew. Chem. 2013, 52, 3074–3085. [Google Scholar] [CrossRef]
- Dai, Y.; van Spronsen, J.; Witkamp, G.J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef]
- Espino, M.; de los Ángeles Fernández, M.; Gomez, F.J.V.; Boiteux, J.; Silva, M.F. Green analytical chemistry metrics: Towards a sustainable phenolics extraction from medicinal plants. Microchem. J. 2018, 141, 438–443. [Google Scholar] [CrossRef]
- Liu, Y.; Li, J.; Fu, R.; Zhang, L.; Wang, D.; Wang, D. Enhanced extraction of natural pigments from Curcuma longa L. using natural deep eutectic solvents. Ind. Crop. Prod. 2019, 140, 111620. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Ravi, H.K.; Khadhraoui, B.; Hilali, S.; Perino, S.; Tixier, A.S.F. Review of Alternative Solvents for Green Extraction of Food and Natural Products: Panorama, Principles, Applications and Prospects. Molecules 2019, 24, 3007. [Google Scholar] [CrossRef] [PubMed]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 7, 70–71. [Google Scholar] [CrossRef] [PubMed]
- Zainal-Abidin, M.H.; Hayyan, M.; Hayyan, A.; Jayakumar, N.S. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Anal. Chim. Acta 2017, 979, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.F.; Wang, X.Q.; Peng, X.; Wang, W.; Zhao, C.J.; Zu, Y.G.; Fu, Y.J. Fast and green extraction and separation of main bioactive flavonoids from Radix Scutellariae. Ind. Crops Prod. 2015, 63, 175–181. [Google Scholar] [CrossRef]
- Jeong, K.M.; Ko, J.; Zhao, J.; Jin, Y.; Yoo, D.E.; Han, S.Y.; Lee, J. Multi-functioning deep eutectic solvents as extraction and storage media for bioactive natural products that are readily applicable to cosmetic products. J. Clean. Prod. 2017, 151, 87–95. [Google Scholar] [CrossRef]
- Espino, M.; Solari, M.; de los Ángeles Fernández, M.; Boiteux, J.; Gómez, M.R.; Silva, M.F. Nades-mediated folk plant extracts as novel antifungal agents against Candida albicans. J. Pharm. Biomed. Anal. 2019, 167, 15–20. [Google Scholar] [CrossRef]
- Espino, M.; de los Ángeles Fernández, M.; Gomez, F.J.V.; Silva, M.F. Natural designer solvents for greening analytical chemistry. Trends Analyt. Chem. 2016, 76, 126–136. [Google Scholar] [CrossRef]
- Gutiérrez, M.C.; Ferrer, M.L.; Mateo, C.R.; Monte, F.D. Freeze-drying of aqueous solutions of deep eutectic solvents: A suitable approach to deep eutectic suspensions of self-assembled structures. Langmuir 2009, 25, 5509–5515. [Google Scholar] [CrossRef]
- Panić, M.; Elenkov, M.M.; Roje, M.; Cvjetko Bubalo, M.; Radojčić Redovniković, I. Plant-mediated stereoselective biotransformations in natural deep eutectic solvents. Process Biochem. 2018, 66, 133–139. [Google Scholar] [CrossRef]
- Yang, T.X.; Zhao, L.Q.; Wang, J.; Song, G.L.; Liu, H.M.; Cheng, H.; Yang, Z. Improving whole cell biocatalysis by addition of deep eutectic solvents and natural deep eutectic solvents. ACS Sustain. Chem. Eng. 2017, 5, 5713–5722. [Google Scholar] [CrossRef]
- Ma, X.C.; Sun, C.; Huang, S.S.; Wang, J.K.; Zhang, B.J.; Li, F.Y.; Wang, G.; Deng, S.; Cui, J. Preparative isolation and purification of four prenylflavanones from microbial biotransformation of kurarinone by high-speed counter-current chromatography. Sep. Purif. Technol. 2010, 76, 140–145. [Google Scholar] [CrossRef]
- Xia, G.; Lin, C.; Liu, S. Tannase-mediated biotransformation assisted separation and purification of theaflavin and epigallocatechin by high speed counter current. Microsc. Res. Tech. 2016, 79, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Schwienheer, C.; Prinz, A.; Zeiner, T.; Merz, J. Separation of active laccases from Pleurotus sapidus culture supernatant using aqueous two phase systems in centrifugal partition chromatography. J. Chromatogr. B 2015, 1002, 1–7. [Google Scholar] [CrossRef]
- Rincón, E.; Balu, A.M.; Luque, R.; Serrano, L. Mechanochemical extraction of antioxidant phenolic compounds from Mediterranean and medicinal Laurus nobilis: A comparative study with other traditional and green novel techniques. Ind. Crop Prod. 2019, 141, 111805. [Google Scholar] [CrossRef]
- Dhanani, T.; Shah, S.; Gajbhiye, N.; Kumar, S. Effect of extraction methods on yield, phytochemical constituents and antioxidant activity of Withania somnifera. Arab. J. Chem. 2017, 10, S1193–S1199. [Google Scholar] [CrossRef]
- Srivastava, S.; Cahill, D.M.; Adholeya, A. Optimal method selection for biocompatible extraction of rosmarinic acid from mycorrhizal hairy roots of Ocimum basilicum. Biotechnol. Rep. 2019, 20, e00325. [Google Scholar] [CrossRef]
- Jiao, J.; Gai, Q.Y.; Fu, J.Y.; Ma, W.; Peng, X.; Tan, S.N.; Efferth, T. Efficient production of isoflavonoids by Astragalus membranaceus hairy root cultures and evaluation of antioxidant activities of extracts. J. Agric. Food Chem. 2014, 62, 12649–12658. [Google Scholar] [CrossRef]
- Rodrigues, M.F.F.; Sousa, I.M.O.; Vardanega, R.; Nogueira, G.C.; Meireles, M.A.A.; Foglio, M.A.; Marchese, J.A. Techno-economic evaluation of artemisinin extraction from Artemisia annua L. using supercritical carbon dioxide. Ind. Crop. Prod. 2019, 132, 336–343. [Google Scholar] [CrossRef]
- Smith, R.M. Supercritical fluids in separation science, The dreams, the reality and the future. J. Chromatogr. A 1999, 856, 83–115. [Google Scholar] [CrossRef]
- Szarka, S.; Gyurjan, I.; Laszlo, M.; Hethelyi, E.; Kuzovkina, I.N.; Lemberkovics, E.; Szoke, E. GC-MS Studies of thiophenes in the supercritical fluid CO2 and solvent extracts of Tagetes patula L. Chromatographia 2010, 71, 1039–1047. [Google Scholar] [CrossRef]
- Anuar, N.; Markom, M.; Khairedin, S.; Johari, N.A. Production and Extraction of Quercetin and (+)-Catechin from Phyllanthus niruri Callus Culture. Int. J. Biotechnol. Bioeng. 2012, 6, 968–971. [Google Scholar]
- Thakre, N.; Prajapati, A.K.; Mahapatra, S.P.; Kumar, A.; Khapre, A.; Pal, D. Modeling and optimization of reactive extraction of citric acid. J. Chem. Eng. Data 2016, 61, 2614–2623. [Google Scholar] [CrossRef]
- Djas, M.; Henczka, M. Reactive extraction of citric acid using supercritical carbon dioxide. J. Supercrit. Fluids 2016, 117, 59–63. [Google Scholar] [CrossRef]
- Afzal, W.; Liu, X.; Prausnitz, J.M. High solubilities of carbon dioxide in tetraalkyl phosphonium based ionic liquids and the effect of diluents on viscosity and solubility. J. Chem. Eng. Data 2014, 59, 954–960. [Google Scholar] [CrossRef]
- Henczka, M.; Djas, M. Reactive extraction of succinic acid using supercritical carbon dioxide. Sep. Sci. Technol. 2018, 53, 655–661. [Google Scholar] [CrossRef]
- Kursinszki, L.; Hank, H.; László, I.; Szőke, É. Simultaneous analysis of hyoscyamine, scopolamine, 6β-hydroxyhyoscyamine and apoatropine in Solanaceous hairy roots by reversed phase high-performance liquid chromatography. J Chromatogr. A 2005, 1091, 32–39. [Google Scholar] [CrossRef]
- Wahby, I.; Arráez-Román, D.; Segura-Carretero, A.; Ligero, F.; Caba, J.M.; Fernández-Gutiérrez, A. Analysis of choline and atropine in hairy root cultures of Cannabis sativa L. by capillary electrophoresis-electrospray mass spectrometry. Electrophoresis 2006, 27, 2208–2215. [Google Scholar] [CrossRef]
- Hahn, T.; Lang, S.; Ulber, R.; Muffler, K. Novel procedures for the extraction of fucoidan from brown algae. Process Biochem. 2012, 47, 1691–1698. [Google Scholar] [CrossRef]
- Kadam, S.U.; Tiwari, B.K.; O’Donnell, C.P. Application of novel extraction technologies for bioactives from marine algae. J. Agric. Food Chem. 2013, 61, 4667–4675. [Google Scholar] [CrossRef]
- Cassol, L.; Rodrigues, E.; Pelayo, C.; Noreña, Z. Extracting phenolic compounds from Hibiscus sabdariffa L. calyx using microwave assisted extraction. Ind. Crop. Prod. 2019, 133, 168–177. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Meullemiestre, A.; Turk, M.; Perino, S.; Fabiano-Tixier, A.S.; Abert-Vian, M. Review of green food processing techniques. Preservation, transformation, and extraction. Innov. Food Sci. Emerg. Technol. 2017, 41, 357–377. [Google Scholar] [CrossRef]
- Gao, M.; Song, B.Z.; Liu, C.Z. Dynamic microwave assisted extraction of flavonoids from Saussurea medusa Maxim cultured cells. Biochem. Eng. J. 2006, 32, 79–83. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, J.H. Development and optimization of a novel simultaneous microwave assisted extraction and adsorbent treatment process for separation and recovery of paclitaxel from plant cell cultures. Sep. Purif. Technol. 2011, 80, 240–245. [Google Scholar] [CrossRef]
- Romanik, G.; Gilgenast, E.; Przyjazny, A.; Kamiński, M. Techniques of preparing plant material for chromatographic separation and analysis. J. Biochem. Biophys. Methods 2007, 70, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.; Gai, Q.Y.; Zhang, L.; Wang, W.; Luo, M.; Zu, Y.G.; Fu, Y.J. High-speed homogenization coupled with microwave assisted extraction followed by liquid chromatography-tandem mass spectrometry for the direct determination of alkaloids and flavonoids in fresh Isatis tinctoria L. hairy root cultures. Anal. Bioanal. Chem. 2015, 407, 4841–4848. [Google Scholar] [CrossRef]
- Jiao, J.; Gai, Q.Y.; Luo, M.; Peng, X.; Zhao, C.J.; Fu, Y.J.; Ma, W. Direct determination of astragalosides and isoflavonoids from fresh Astragalus membranaceus hairy root cultures by high speed homogenization coupled with cavitation-accelerated extraction followed by liquid chromatography-tandem mass spectrometry. RSC Adv. 2015, 5, 34672–34681. [Google Scholar] [CrossRef]
- Li, S.M.; Fu, Y.J.; Zu, Y.G.; Zu, B.S.; Wang, Y.; Efferth, T. Determination of paclitaxel and its analogues in the needles of Taxus species by using negative pressure cavitation extraction followed by HPLC-MS-MS. J. Sep. Sci. 2009, 32, 3958–3966. [Google Scholar] [CrossRef]
- Liu, W.; Fu, Y.J.; Zu, Y.G.; Kong, Y.; Zhang, L.; Zu, B.S.; Efferth, T. Negative-pressure cavitation extraction for the determination of flavonoids in pigeon pea leaves by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2009, 1216, 3841–3850. [Google Scholar] [CrossRef]
- Krause, J.; Merz, J. Comparison of enzymatic hydrolysis in a centrifugal partition chromatograph and stirred tank reactor. J. Chromatogr. A 2017, 1504, 64–70. [Google Scholar] [CrossRef]
- Song, X.; Cui, L.; Li, J.; Yan, H.; Li, L.; Wen, L.; Geng, Y.; Wang, D. A novel bioreactor for highly efficient biotransformation of resveratrol from polydatin with high-speed counter-current chromatography. LWT Food Sci. Technol. 2019, 103, 192–198. [Google Scholar] [CrossRef]
- Wang, D.; Zhao, H.; Zhu, H.; Wen, L.; Yu, J.; Li, L.; Chen, L.; Geng, Y. A novel method for highly efficient biotransformation and separation of isoflavone aglycones from soybean with high-speed counter-current chromatography. Ind. Crops Prod. 2019, 129, 224–230. [Google Scholar] [CrossRef]
- Wang, D.; Khan, M.S.; Cui, L.; Song, X.; Zhu, H.; Ma, T.; Li, X.; Sun, R. A novel method for the highly efficient biotransformation of genistein from genistin using a high-speed counter-current chromatography bioreactor. RSC Adv. 2019, 9, 4892–4899. [Google Scholar] [CrossRef]
- Krause, J.; Oeldorf, T.; Schembecker, G.; Merz, J. Enzymatic hydrolysis in an aqueous organic two phase system using centrifugal partition chromatography. J. Chromatogr. A 2015, 1391, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Nioi, C.; Riboul, D.; Destrac, P.; Marty, A.; Marchal, L.; Condoret, J.S. The centrifugal partition reactor, a novel intensified continuous reactor for liquid=liquid enzymatic reactions. Biochem. Eng. J. 2015, 103, 227–233. [Google Scholar] [CrossRef]
- Berthod, A.; Carda-Broch, S. Use of ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate in countercurrent chromatography. Anal. Bioanal. Chem. 2004, 380, 168–177. [Google Scholar] [CrossRef]
- Ruiz-Angel, M.J.; Pino, V.; Carda-Broch, S.; Berthod, A. Solvent systems for countercurrent chromatography: An aqueous two phase liquid system based on a room temperature ionic liquid. J. Chromatogr. A 2007, 1151, 65–73. [Google Scholar] [CrossRef]
- Handa, S.S. An overview of extraction techniques for medicinal and aromatic plants. In Extraction Technologies for Medicinal and Aromatic Plants; Handa, S.S., Khanuja, S.P.S., Longo, G., Rakesh, D.D., Eds.; International Centre for Science and High Technology: Trieste, Italy, 2008; pp. 21–54. [Google Scholar]
- Ghafoor, K.; Al-Juhaimi, F.Y.; Choi, Y.H. Supercritical fluid extraction of phenolic compounds and antioxidants from grape (Vitis labrusca B.) seeds. Plant Foods Hum. Nutr. 2012, 67, 407–414. [Google Scholar] [CrossRef]
- Wang, L.; Wu, M.; Liu, H.M.; Ma, Y.X.; Wang, X.D.; Qin, G.Y. Subcritical Fluid Extraction of Chinese Quince Seed: Optimization and Product Characterization. Molecules 2017, 22, 528. [Google Scholar] [CrossRef]
- Toubane, A.; Rezzoug, S.A.; Besombes, C.; Daoud, K. Optimization of accelerated solvent extraction of Carthamus caeruleus L. Evaluation of antioxidant and anti-inflammatory activity of extracts. Ind. Crops Prod. 2017, 97, 620–631. [Google Scholar] [CrossRef]
- Fang, X.; Wang, J.; Hao, J.; Li, X.; Guo, N. Simultaneous extraction, identification and quantification of phenolic compounds in Eclipta prostrata using microwave assisted extraction combined with HPLC-DAD-ESI-MS/MS. Food Chem. 2015, 188, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Solana, R.; Vázquez-Araújo, L.; Salgado, J.M.; Domínguez, J.M.; Cortés-Diéguez, S. Optimization of the process of aromatic and medicinal plant maceration in grape marc distillates to obtain herbal liqueurs and spirits. J. Sci. Food Agric. 2016, 96, 4760–4771. [Google Scholar] [CrossRef] [PubMed]
- Luque de Castro, M.D.; Priego-Capote, F. Soxhlet extraction: Past and present panacea. J. Chromatogr. A 2010, 1217, 2383–2389. [Google Scholar] [CrossRef] [PubMed]
- Roby, M.H.H.; Sarhan, M.A.; Selim, K.A.H.; Khalel, K.I. Evaluation of antioxidant activity, total phenols and phenolic compounds in thyme (Thymus vulgaris L.), sage (Salvia officinalis L.), and marjoram (Origanum majorana L.) extracts. Ind. Crops Prod. 2013, 43, 827–831. [Google Scholar] [CrossRef]
- Ahmad, R.; Ahmad, N.; Shehzad, A. Solvent and temperature effects of accelerated solvent extraction (ASE) with Ultra-high-pressure liquid chromatography (UHPLC-PDA) technique for determination of Piperine and its ICP-MS analysis. Ind. Crop. Prod. 2019, 136, 37–49. [Google Scholar] [CrossRef]
- Mouahid, A.; Dufour, C.; Badens, E. Supercritical CO2 extraction from endemic Corsican plants; comparison of oil composition and extraction yield with hydrodistillation method. J. CO2 Util. 2017, 20, 263–273. [Google Scholar] [CrossRef]
- Du, F.Y.; Xiao, X.H.; Luo, X.J.; Li, G.K. Application of ionic liquids in the microwave assisted extraction of polyphenolic compounds from medicinal plants. Talanta 2009, 78, 1177–1184. [Google Scholar] [CrossRef]
- Nabet, N.; Gilbert-López, B.; Madani, K.; Herrero, M.; Ibáñez, E.; Mendiola, J.A. Optimization of microwave assisted extraction recovery of bioactive compounds from Origanum glandulosum and Thymus fontanesii. Ind. Crops Prod. 2019, 129, 395–404. [Google Scholar] [CrossRef]
- Dahmoune, F.; Nayak, B.; Moussi, K.; Remini, H.; Madani, K. Optimization of microwave- assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem. 2015, 166, 585–595. [Google Scholar] [CrossRef]
- Liu, X.; Jing, X.; Li, G. A process to acquire essential oil by distillation concatenated liquid-liquid extraction and flavonoids by solid-liquid extraction simultaneously from Helichrysum arenarium (L.) Moench inflorescences under ionic liquid microwave mediated. Sep. Purif. Technol. 2019, 209, 164–174. [Google Scholar]
- Liu, T.; Sui, X.; Lei, R.Z.; Yuangang, Y.; Zhang, Z.L.; Zhang, Y.; Zhang, Z. Application of ionic liquids based microwave assisted simultaneous extraction of carnosic acid, rosmarinic acid and essential oil from Rosmarinus officinalis. J. Chromatogr. A 2011, 1218, 8480–8489. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, L.; Zu, Y.; Zhao, C.; Zhang, L.; Zhang, Y.; Zhang, Z.; Wang, W. Development of an ionic liquid based microwave assisted method for simultaneous extraction and distillation for determination of proanthocyanidins and essential oil in Cortex cinnamomic. Food Chem. 2012, 135, 2514–2521. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.F.; Zhao, R.N.; Dong, L.J.; Zhao, X.Y.; Su, J.X.; Zhao, M.; Li, L.; Bian, Y.J.; Zhang, L.J. Dual-cooled solvent-free microwave extraction of Salvia officinalis L. essential oil and evaluation of its antimicrobial activity. Ind. Crops Prod. 2018, 120, 71–76. [Google Scholar] [CrossRef]
- Ivanović, M.; Alañón, M.E.; Arráez-Román, D.; Segura-Carretero, A. Enhanced and green extraction of bioactive compounds from Lippia citriodora by tailor-made natural deep eutectic solvents. Food Res. Int. 2018, 111, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Chizoba Ekezie, F.G.; Sun, D.W.; Cheng, J.H. Acceleration of microwave assisted extraction processes of food components by integrating technologies and applying emerging solvents: A review of latest developments. Trends Food Sci. Technol. 2017, 67, 160–172. [Google Scholar] [CrossRef]
- Yu, Y.; Chen, B.; Chen, Y.; Xie, M.; Duan, H.; Li, Y.; Duan, G. Nitrogen-protected microwave assisted extraction of ascorbic acid from fruit and vegetables. J. Sep. Sci. 2009, 32, 4227–4233. [Google Scholar] [CrossRef]
- Nieto, A.; Borrull, F.; Pocurull, E.; Marcé, R.M. Pressurized liquid extraction: A useful technique to extract pharmaceuticals and personal-care products from sewage sludge. Trends Anal. Chem. 2010, 29, 752–764. [Google Scholar] [CrossRef]
- Panja, P. Green extraction methods of food polyphenols from vegetable materials. Curr. Opin. Food Sci. 2018, 23, 173–182. [Google Scholar] [CrossRef]
- Fierascu, I.; Georgiev, M.I.; Ortan, A.; Fierascu, R.C.; Avramescu, S.M.; Ionescu, D.; Sutan, A.; Brinzan, A.; Ditu, L.M. Phyto-mediated metallic nano-architectures via Melissa officinalis L.: Synthesis, characterization and biological properties. Sci. Rep. 2017, 7, 12428. [Google Scholar] [CrossRef]
- Plaza, M.; Turner, C. Pressurized hot water extraction of bioactives. Trends Anal. Chem. 2015, 71, 39–54. [Google Scholar] [CrossRef]
- Bursać Kovačević, D.; Barba, F.J.; Granato, D.; Galanakis, C.M.; Herceg, Z.; Dragović- Uzelac, V.; Putnik, P. Pressurized Hot Water Extraction (PHWE) for the green recovery of bioactive compounds and steviol glycosides from Stevia rebaudiana Bertoni Leaves. Food Chem. 2018, 254, 150–157. [Google Scholar] [CrossRef]
- Gomes, S.V.F.; Portugal, L.A.; dos Anjos, J.P.; de Jesus, O.N.; de Oliveira, E.J.; David, J.P.; David, J.M. Accelerated solvent extraction of phenolic compounds exploiting a Box-Behnken design and quantification of five flavonoids by HPLC-DAD in Passiflora species. Microchem. J. 2017, 132, 28–35. [Google Scholar] [CrossRef]
- Herrero, M.; Plaza, M.; Cifuentes, A.; Ibáñez, E. Green processes for the extraction of bioactives from Rosemary: Chemical and functional characterization via ultra-performance liquid chromatography-tandem mass spectrometry and in-vitro assays. J. Chromatogr. A 2010, 1217, 2512–2520. [Google Scholar] [CrossRef] [PubMed]
- Nastić, N.; Švarc-Gajić, J.; Delerue-Matos, C.; Barroso, M.F.; Soares, C.; Moreira, M.M.; Morais, S.; Mašković, P.; Srček, V.G.; Slivac, I.; et al. Subcritical water extraction as an environmentally-friendly technique to recover bioactive compounds from traditional Serbian medicinal plants. Ind. Crops Prod. 2018, 111, 579–589. [Google Scholar] [CrossRef]
- Carr, A.G.; Mammucari, R.; Foster, N.R. A review of subcritical water as a solvent and its utilisation for the processing of hydrophobic organic compounds. Chem. Eng. J. 2011, 172, 1–17. [Google Scholar] [CrossRef]
- Arapitsas, P.; Turner, C. Pressurized solvent extraction and monolithic column-HPLC/DAD analysis of anthocyanins in red cabbage. Talanta 2008, 74, 1218–1223. [Google Scholar] [CrossRef]
- Lupacchini, M.; Mascitti, A.; Giachi, G.; Tonucci, L.; d’Alessandro, N.; Martinez, J.; Colacino, E. Sonochemistry in non-conventional, green solvents or solvent free reactions. Tetrahedron 2017, 73, 609–653. [Google Scholar] [CrossRef]
- Liu, X.; Ahlgren, S.; Korthout, H.A.A.J.; Salomé-Abarca, L.F.; Bayona, L.M.; Verpoorte, R.; Choi, Y.H. Broad range chemical profiling of natural deep eutectic solvent extracts using a high performance thin layer chromatography-based method. J. Chromatogr. A 2018, 1532, 198–207. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Parniakov, O.; Deng, Q.; Patras, A.; Koubaa, M.; Grimi, N.; Boussetta, N.; Tiwari, B.K.; Vorobiev, E.; Lebovka, N.; et al. Application of non-conventional extraction methods: Toward a sustainable and green production of valuable compounds from mushrooms. Food Eng. Rev. 2016, 8, 214–234. [Google Scholar] [CrossRef]
- Jia, X.J.; Zhang, C.; Qiu, J.F.; Wang, L.L.; Bao, J.L.; Wang, K.; Zhang, Y.L.; Chen, M.W.; Wan, J.B.; Su, H.X.; et al. Purification, structural characterization and anticancer activity of the novel polysaccharides from Rhynchosia minima root. Carbohydr. Polym. 2015, 132, 67–71. [Google Scholar] [CrossRef]
- Fernando, P.S.; Sanjeewa, K.K.A.; Samarakoon, K.W.; Lee, W.W.; Kim, H.S.; Kang, N.; Ranasinghe, P.; Lee, H.S.; Jeon, Y.J. A fucoidan fraction purified from Chnoospora minima; a potential inhibitor of LPS-induced inflammatory responses. Int. J. Biol. Macromol. 2017, 104, 1185–1193. [Google Scholar] [CrossRef]
- Fan, T.; Hu, J.G.; Fu, L.D.; Zhang, L.J. Optimization of enzymolysis-ultrasonic assisted extraction of polysaccharides from Momordica charantia L. by response surface methodology. Carbohydr. Polym. 2015, 115, 701–706. [Google Scholar] [CrossRef]
- Lin, J.A.; Kuo, C.H.; Chen, B.Y.; Li, Y.; Liu, Y.C.; Chen, J.H.; Shieh, C.J. A novel enzyme assisted ultrasonic approach for highly efficient extraction of resveratrol from Polygonum cuspidatum. Ultrason. Sonochem. 2016, 32, 258–264. [Google Scholar] [CrossRef]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Chen, S.; Xing, X.H.; Huang, J.J.; Xu, M.S. Enzyme assisted extraction of flavonoids from Ginkgo biloba leaves: Improvement effect of flavonol transglycosylation catalyzed by Penicillium decumbens cellulase. Enzyme Microb. Technol. 2011, 48, 100–105. [Google Scholar] [CrossRef]
- Puri, M.; Sharma, D.; Barrow, C.J. Enzyme assisted extraction of bioactives from plants. Trends Biotechnol. 2012, 30, 37–44. [Google Scholar] [CrossRef]
- Miron, T.L.; Herrero, M.; Ibáñez, E. Enrichment of antioxidant compounds from lemon balm (Melissa officinalis) by pressurized liquid extraction and enzyme assisted extraction. J. Chromatogr. A 2013, 1288, 1–9. [Google Scholar] [CrossRef]
- Knirsch, M.C.; dos Santos, C.A.; Martins de Oliveira, A.A.; Vicente, S.; Vessoni Penna, T.C. Ohmic heating, a review. Trends Food Sci. Technol. 2010, 21, 436–441. [Google Scholar] [CrossRef]
- Gavahian, M.; Farahnaky, A. Ohmic assisted hydrodistillation technology: A review. Trends Food Sci. Technol. 2018, 72, 153–161. [Google Scholar] [CrossRef]
- Gavahian, M.; Farhoosh, R.; Javidnia, K.; Shahidi, F.; Farahnaky, A. Effect of applied voltage and frequency on extraction parameters and extracted essential oils from Mentha piperita by ohmic assisted hydrodistillation. Innov. Food Sci. Emerg. Technol. 2015, 29, 161–169. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Nikmaram, N.; Esteghlal, S.; Khaneghah, A.M.; Niakousari, M.; Barba, F.J.; Roohinejad, S.; Koubaa, M. Efficiency of Ohmic assisted hydrodistillation for the extraction of essential oil from oregano (Origanum vulgare subsp. viride) spices. Innov. Food Sci. Emerg. Technol. 2017, 41, 172–178. [Google Scholar] [CrossRef]
- Locke, B.R.; Sato, M.; Sunka, P.; Hoffmann, M.R.; Chang, J.S. Electrohydraulic discharge and nonthermal plasma for water treatment. Ind. Eng. Chem. Res. 2006, 45, 882–905. [Google Scholar] [CrossRef]
- Martynenko, A.; Kudra, T. Electrically-induced transport phenomena in EHD drying, A review. Trends Food Sci. Technol. 2016, 54, 63–73. [Google Scholar] [CrossRef]
- Dalvi-Isfahan, M.; Hamdami, N.; Le-Bail, A.; Xanthakis, E. The principles of high voltage electric field and its application in food processing: A review. Food Res. Int. 2016, 89, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Roselló-Soto, E.; Barba, F.J.; Parniakov, O.; Galanakis, C.M.; Lebovka, N.; Grimi, N.; Vorobiev, E. High voltage electrical discharges, pulsed electric field, and ultrasound assisted extraction of protein and phenolic compounds from olive kernel. Food Bioproc. Tech. 2015, 8, 885–894. [Google Scholar] [CrossRef]
- Sarkis, J.R.; Boussetta, N.; Tessaro, I.C.; Marczak, L.D.F.; Vorobiev, E. Application of pulsed electric fields and high voltage electrical discharges for oil extraction from sesame seeds. J. Food Eng. 2015, 153, 20–27. [Google Scholar] [CrossRef]
- Fincan, M.; De Vito, F.; Dejmek, P. Pulsed electric field treatment for solid-liquid extraction of red beetroot pigment. J. Food Eng. 2004, 64, 381–388. [Google Scholar] [CrossRef]
- Guderjan, M.; Töpfl, S.; Angersbach, A.; Knorr, D. Impact of pulsed electric field treatment on the recovery and quality of plant oils. J. Food Eng. 2005, 67, 281–287. [Google Scholar] [CrossRef]
- Pina-Pérez, M.C.; Rivas, A.; Martínez, A.; Rodrigo, D. Effect of thermal treatment, microwave, and pulsed electric field processing on the antimicrobial potential of açaí (Euterpe oleracea), stevia (Stevia rebaudiana Bertoni), and ginseng (Panax quinque folius L.) extracts. Food Control 2018, 90, 98–104. [Google Scholar] [CrossRef]
- Dahmoune, F.; Remini, H.; Dairi, S.; Aoun, O.; Moussi, K.; Bouaoudia-Madi, N.; Adjeroud, N.; Kadri, N.; Lefsih, K.; Boughani, L.; et al. Ultrasound assisted extraction of phenolic compounds from P. lentiscus L. leaves: Comparative study of artificial neural network (ANN) versus degree of experiment for prediction ability of phenolic compounds recovery. Ind. Crop. Prod. 2015, 77, 251–261. [Google Scholar] [CrossRef]
- Wong, K.H.; Li, G.Q.; Li, K.M.; Razmovski-Naumovski, V.; Chan, K. Optimisation of Pueraria isoflavonoids by response surface methodology using ultrasonic assisted extraction. Food Chem. 2017, 231, 231–237. [Google Scholar] [CrossRef]
- Amdoun, R.; Benyoussef, E.H.; Benamghar, A.; Khelifi, L. Prediction of hyoscyamine content in Datura stramonium L. hairy roots using different modeling approaches: Response Surface Methodology (RSM), Artificial Neural Network (ANN) and Kriging. Biochem. Eng. J. 2019, 144, 8–17. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, Q.; Mao, G.; Zou, Y.; Feng, W.; Zheng, D.; Wang, W.; Zhou, L.; Zhang, T.; Yang, J.; et al. Optimization of enzyme assisted extraction and characterization of polysaccharides from Hericium erinaceus. Carbohydr. Polym. 2014, 101, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Milutinović, M.; Radovanović, N.; Ćorović, M.; Šiler-Marinković, S.; Rajilić-Stojanović, M.; Dimitrijević-Branković, S. Optimisation of microwave assisted extraction parameters for antioxidants from waste Achillea millefolium dust. Ind. Crops Prod. 2015, 77, 333–341. [Google Scholar] [CrossRef]
- Xu, W.J.; Zhai, J.W.; Cui, Q.; Liu, J.Z.; Luo, M.; Fu, Y.J.; Zu, Y.G. Ultra-turrax based ultrasound assisted extraction of five organic acids from honeysuckle Lonicera japonica Thunb. and optimization of extraction process. Sep. Purif. Technol. 2016, 166, 73–82. [Google Scholar] [CrossRef]
- Ameer, K.; Bae, S.W.; Jo, Y.; Lee, H.G.; Ameer, A.; Kwon, J.H. Optimization of microwave assisted extraction of total extract, stevioside and rebaudioside-A from Stevia rebaudiana (Bertoni) leaves, using response surface methodology (RSM) and artificial neural network (ANN) modelling. Food Chem. 2017, 229, 198–207. [Google Scholar] [CrossRef]
- Benmoussa, H.; Elfalleh, W.; He, S.; Romdhane, M.; Benhamou, A.; Chawech, R. Microwave hydrodiffusion and gravity for rapid extraction of essential oil from Tunisian cumin (Cuminum cyminum L.) seeds: Optimization by response surface methodology. Ind. Crops Prod. 2018, 124, 633–642. [Google Scholar] [CrossRef]
- Gallo-Molina, A.C.; Castro-Vargasa, H.I.; Garzón-Méndez, W.F.; Martínez Ramírez, J.A.; Rivera Monroy, Z.J.; King, J.W.; Parada-Alfonso, F. Extraction, isolation and purification of tetrahydrocannabinol from the Cannabis sativa L. plant using supercritical fluid extraction and solid phase extraction. J. Supercrit. Fluids 2019, 146, 208–216. [Google Scholar] [CrossRef]
- Pilkington, J.L.; Preston, C.; Gomes, R.L. Comparison of response surface methodology (RSM) and artificial neural networks (ANN) towards efficient extraction of artemisinin from Artemisia annua. Ind. Crops Prod. 2014, 58, 15–24. [Google Scholar] [CrossRef]
| Term | Definition |
|---|---|
| ANN | Artificial neural network, computational modelling technique, based on simulations of the biological brain, with which can be drawn conclusions through generalization |
| AFE | Accelerated fluid extraction, extraction method based on solvent with high pressure and temperature |
| CAE | Cavitation accelerated extraction, extraction method based on occurring cavitation due to the passage of ultrasound waves in the liquid medium |
| CCC | Counter-current chromatography, chromatographic techniques used for preparative isolation and purification of natural products |
| CPC | Centrifugal partition chromatography, chromatographic techniques used for preparative isolation and purification of natural products |
| EAAE | Enzyme assisted aqueous extraction, extraction method enzyme assisted in aqueous medium |
| EACP | Enzyme assisted cold pressing, extraction method enzyme assisted, used especially for obtaining oils |
| GAE | Gallic acid equivalents |
| HVED | High voltage electric discharge, non-conventional extracting method based on corona discharge, which through the control of voltage, can enhance heat and mass transfer; the process can be enhanced by pulsed high voltage electrical discharges in water or liquid medium |
| ILMHDE | Microwave assisted hydro distillation concatenated liquid-liquid extraction, extraction technique based on microwave concatenated with a liquid-liquid extraction installation, with two columns, which can separate essential oils (in the first separation column) and extract some components from the hydrosol (in the second separation column) |
| ILMSED | Ionic liquid based microwave assisted simultaneous extraction and distillation, microwave assisted simultaneous extraction and distillation, which use as solvents ionic liquids |
| LLE | Liquid-liquid extraction, extraction method based on the relative solubilities of compounds in two different immiscible liquids |
| Low energy-high efficiency extraction | Extracting processes with reduced energy consumption, decreased quantity of raw material, and increased yield of the final biologically active compounds |
| MAE | Microwave assisted extraction, extraction method based on microwave power with damaging of cells’ structure |
| MASDE | Microwave assisted simultaneous distillation extraction, extraction method with two simultaneous processes, microwave assisted extraction and the distillation of essential oils |
| NADES | Natural deep eutectic solvents, solvents based on more compounds produced from plant based primary metabolites liquid at ambient temperature having unusual solvent properties |
| OAHD | Ohmic assisted extraction, nonconventional extracting method that relies on ohmic heating by passing an electrical current through materials, instead of conductive heat transfer |
| PEF | Pulsed electric field, extraction techniques that use short pulses of electricity (from μs to ms) under high intensity electric fields (kV/cm), which leads to the formation of pores on the cell membranes with improving the extraction and diffusion processes, causing the permeabilization of the cell membrane |
| PHWE | Pressurized hot water extraction, techniques that perform the extraction under pressure, with water as a solvent |
| PLE | Pressurized liquid extraction, extraction method that performs the extraction under pressure |
| RSM | Response surface methodology, statistical methods used for process optimization and modelling through empirical models |
| SFE | Supercritical fluid extraction, extraction method that performs the extraction under pressure with CO2 as a solvent |
| SFME | Solvent-free microwave extraction, modified Clevenger-type equipment to obtain essential oils based on microwave |
| SLE | Solid-liquid extraction, a multi-step extraction technique, resulting in the release of the target product with or without disruption of the cells with a counter current operation |
| UAE | Ultrasonic assisted extraction, extraction method based on repeated compression/expansion cycles caused by ultrasonic waves without the disruption of cell wall/membranes |
| UAEE | Ultrasonic assisted enzymatic extraction, extraction method based simultaneous on ultrasound and enzymatic treatment |
| WEPO | Water extraction and particle formation on-line, extraction method that performs the extraction under pressure with a mixture of CO2 and ethanol or water and particles as a solvent with formation on-line |
| Extraction Technique | Advantages | Disadvantages | Ref. |
|---|---|---|---|
| Classical techniques | - Large amounts of biomass | - High amounts of solvents - Low yield of bioactive compounds - Consumes energy - Plant wastes still found in active compounds | [89] |
| Pressurized liquid extraction | |||
| Supercritical fluid extraction | - Increased selectivity - Avoid sample oxidization in the presence of air - Small amounts of solvents | - Expensive equipment - Extra cost for fluids | [90] |
| Subcritical fluid extraction | - Increased selectivity - Green solvent | - Expensive equipment | [91] |
| Accelerated solvent extraction | - Decreased time consumption and solvent use | - Need for high temperature and pressure - Expensive equipment | [92] |
| Microwave assisted | - Increased amounts of solvent - Increased extraction time - microwaves are non-contact heat source - microwaves can selectively heat the materials | - Expensive equipment | [93] |
| Ultrasound assisted | - Increased amounts of solvent - Can be used as pre-treatment for other techniques, increasing the yield of extraction - Breaking the walls of cells | - Good optimization of the parameters | [57] |
| Plant Material | Family Name | Extraction Method | Plant Organ | Conditions | Obtained Compounds | Ref. |
|---|---|---|---|---|---|---|
| Thyme, Thymus vulgaris L. Sage, Salvia officinalis L. Marjoram, Origanum majorana L. | Lamiaceae | Maceration | Leaves | Room temperature Time (hour), 72 Solvents: methanol, ethanol, diethyl ether, and hexane | Caffeic acid, p-coumaric acid, ferulic acid, cinnamic acid (hydroxycinnamic acids) carnosic acid (diterpene), rosmarinic acid (caffeic acid ester), apigenin (flavone) | [96] |
| Chamomile, Matricaria recutita L. Coriander, Coriandrum sativum L. Liquorice, Glycyrrhiza glabra L. Southern blue gum, Eucalyptus globulus Labill. | Asteraceae Apiaceae Fabaceae Myrtaceae | Alcoholic maceration | Flowers Seeds Roots Leaves | Solvent: grape marc distillates, alcohol content (v/v), 70%, 55%, 40%. Concentration of plants in the macerate (g/L) 40, 25, 10 | Terpenes | [94] |
| Rosemary, Rosmarinus officinalis L., Immortelle, Helichrysum italicum (Roth) G. Don fil. | Lamiaceae Asteraceae | Hydrodistillation | Leaves and flower | 500 kg of dried plant Time (hour), 3 Without any sample pre-treatment | Monoterpenoids | [98] |
| Plant Material | Family Name | Extraction Method | Plant Organ | Conditions | Obtained Compounds | Extraction Yields (Content) | Ref. |
|---|---|---|---|---|---|---|---|
| Guava, Psidium guajava Linn. China root, Smilax china L. | Myrtaceae Smilacaceae | ILs-MAE | Tubers | 1.0 g of accurately weighed sample, extracted with 20 mL of different ILs’ solution; extraction time (min) 10 at 70 °C; extraction time (min) 10 at 60 °C | Gallic acid, ellagic acid, (phenolic acids), quercetin (flavonol), trans-resveratrol (stilbenoid) | 79.5–93.8% | [99] |
| Rosemary, Rosmarinus officinalis L. | Lamiaceae | PLE SFE WEPO | Leaves | PLE: Temperature (°C), 50, 100, 150, 200; static extraction time (min), 20; warming, up time (min), 5,7, 9 SFE: Temperature (°C), 40; pressure (bar), 150; CO2 flow rate (g/min), 60 WEPO: Temperature (°C) = 200; flow rate of supercritical water (mL/min), 0.2 | Carnosic (diterpene) and rosmarinic acids (caffeic acid ester) | 17.8–37.9%/ 0.5–6.5%/4% | [115] |
| Rosemary, Rosmarinus officinalis L. | Lamiaceae | ILMSED | Fresh leaves | Microwave irradiation frequency (GHz), 2.45; power (W), 120–700 | Carnosic (diterpene), rosmarinic acids (caffeic acid ester), and essential oil | 0.49–33.29%/0.07–3.97%/18.5–23.1% | [103] |
| Ginkgo, Ginkgo biloba Linn. | Ginkgoaceae | EAE | Leaves | time (hours), 30; ethanol-water ratio of 3:7 (v/v); pH 6 | Flavonoids | 14–28.3% | [127] |
| Cinnamon, Cinnamomum verum J. Presl | Lauraceae | ILMSED | Inner bark | Microwave irradiation frequency (GHz), 2.45; power (W), 120–700 | Proanthocyanidins and essential oil | 1.24–4.58% | [104] |
| Lemon balm, Melissa officinalis L. | Lamiaceae | PLE | Leaves | time (min), 10; temperature (°C), 150 °C; extracting time (min), 20; warming-up time (min), 7 | Rosmarinic acid (caffeic acid ester), salvianolic acid, caffeic acid (hydroxycinnamic acid) | 12.8–60.5% | [129] |
| Lemon balm, Melissa officinalis L. | Lamiaceae | EAE | Leaves | Solid/liquid ratio, 1:20; temperature (°), 50; time (hours), 2 | Rosmarinic acid (caffeic acid ester), salvianolic acid, caffeic acid (hydroxycinnamic acid). | 56.2–65.2% | [129] |
| Myrtle, Myrtus communis L. | Myrtaceae | MAE | Leaves and flowers | Microwave power (W), 400–600; extraction time (s), 30–90; liquid-to-solid ratio (mL/g), 20–40; ethanol proportion (%), 20–100 | α-Pinene1,8-cineole, linalool and linalyl acetate (terpene, terpene alcohols) | 20– 60% | [101] |
| Baikal skullcap or Chinese skullcap, Scutellaria baicalensis Georgi | Lamiaceae | NADES -MAE | - | The mole ratio of choline chloride to lactic acid, 3:1, 2:1, 1:1, 1:2, 1:3, and 1:4; water content in choline chloride-lactic acid (%), 0, 20, 40, 60, 80. | Baicalin (flavone glycoside) Wogonoside (glycosides of wogonin); Baicalein (flavone) Wogonin (O-methylated flavone) | 79.5–84.1% | [46] |
| Bitter melon, Momordica charantia L. | Cucurbitaceae | UAEE | Fruit | Extraction time (min), 30–50; pH, 3–5; enzyme concentration (%), 1.5-2.5 | Polysaccharides | 29.75% (predicted value 29.8%) | [124] |
| Asian knotweed, Polygonum cuspidatum Siebold & Zucc. | Polygonaceae | UAEE | - | Power (W), 150; temperature (°C), 70; rotation (rpm), 150; frequency (kHz), 40 | Resveratrol (stilbenoid) | 3.76 mg/g– 11.88 mg/g | [125] |
| Lemon balm, Melissa officinalis L. | Lamiaceae | ASE | Leaves | Temperature (°C), 100; pressure (psi), 1500; static time (min), 5; static cycles, 1; flush volume (%), 60; purge time (s), 120. | Gallic acid, chlorogenic acid, ferulic acid (hydroxycinnamic acids), rosmarinic acid (caffeic acid ester), quercetin (flavonol), rutin (glycoside), apigenin (flavone) | – | [111] |
| Rosemary, Rosmarinus officinalis L., Juniper, Juniperus communis ssp. nana, Immortelle, Helichrysum italicum (Roth) G. Don fil. Mastic tree, Pistacia lentiscus L. | Lamiaceae Cupressaceae Asteraceae Anacardiaceae | SFE | Leaves and flower | Pressure (bar), 300; temperature (K), 313; flow rate of CO2 (kg/h), 0.4 | Verbenone, germacrene D, bornyl acetate, ferruginol, trans-caryophyllene, elemol, γ-cadinene, geraniol or β-eudesmol (terpenes) | 2–8%/23.1–50%/10–23% | [98] |
| Passion flower, Passiflora alata Curtis, P. capsularis, P. cincinnata, P. edulis f. flavicarpa, P. edulis f. edulis, P. galbana, P. gibertii, P. maliformis, P. malacophylla, P. morifolia, P. mucronata, P. quadrangularis, P. racemosa, P. setacea, P. suberosa P. vitifolia, P. tenuifila | Passifloraceae | ASE | Leaves | Temperature (°C), 40–80; ethanol concentration (% w/w), 40–100; number of extraction cycles, 1–5 | Isoorientin, orientin, vitexin, isovitexin, rutin (flavone, apigenin flavone glucoside, glycoside) | 11.07–47.73% | [114] |
| Lemon verbena, Lippia citriodora Kunth | Verbenaceae | NADES-MAE | Leaves | Choline chloride:lactic acid, 1:2; choline chloride:tartaric acid, 2:1; choline chloride:xylitol, 2:1; choline chloride:fructose:water, 2:1:1; choline chloride:sucrose: water, 4:1:2; MAE conditions: Power (W), 700; pressure (bar), 18; temperature (°C), 65; time (min), 20 | Gardoside, ixoside, verbascoside, verbascoside, luteolin-7-diglucuronide, apigenin-7-O-diglucuronide (iridoids, phenylpropanoids and flavonoids) | 7.25–15.63 mg/g 5.43–9.02 mg/g | [106] |
| Sage, Salvia officinalis L. | Lamiaceae | SFME | - | Soaking 71% water for 1 h; extraction time (min), 20–60; humidity of the matrix (%), 60-80 | Odoriferous oxygenated monoterpenes and terpene hydrocarbons | 0.85–1.29%/ 0.85–1.59% | [105] |
| Candy leaf, Stevia rebaudiana Bertoni | Asteraceae | PHWE | Leaves | Pressure (Pa), 10.34; static extraction time (min), 5, 10; temperature (°C), 100, 130, 160; cycle number: 1, 2, 3 | Total phenolic; condensed tannins; chlorophyll A and B total carotenoid content | 5.22–9.33 mg/g 1.37–2.29 mg/g 3.42–3.84 mg/g | [113] |
| Ginkgo, Ginkgo biloba Linn. Ginseng, Panax ginseng | Ginkgoaceae Araliaceae | NADES, UAE | Leaves Stems | malic acid:choline chloride, 1:1; malic acid:glucose, 1:1; choline chloride:glucose, 5:2; malic acid:proline, 1:1; glucose:fructose-sucrose, 1:1:1; glycerol:proline:sucrose, 9:4:1; time (min), 30; temperature (°C), 40 | Ginkgolides (terpenic lactones) Ginsenosides (panaxosides) | – | [120] |
| Blackberry, Morus nigra L. Wall germander, Teucrium chamaedrys L. Bigroot geranium, Geranium macrorrhizum L. Comfrey, Symphytum officinale L. | Moraceae Lamiaceae Geraniaceae Boraginaceae | SWE | Leaves Flowers Leaves Leaves | Sample to distilled water ratio, 1:40; extraction temperature (°C), 60–200; pressure (bar), 10 extraction time (min), 30 | Gallic acid, protocatechuic acid, catechin, chlorogenic acid, caffeic acid (phenolic acids), rutin (glycoside), quercetin (flavonol) | – | [116] |
| Oregano, Origanum glandulosum Desf., Algerian thyme, Thymus fontanesii Boiss. & Reut. | Lamiaceae | MAE | Leaves and flowers | Solvent composition, 0, 50, 100% ethanol in water; extraction time (min), 1, 5.5, 10; temperature (°C), 30, 90, 150; microwave extraction reactor, 850 W and 2455 MHz | Gallocatechin (flavan-3-ol) rosmarinic acid (caffeic acid ester) | 5.32–18.39% | [100] |
| Immortelle, Helichrysum arenarium L. Moench | Asteraceae | ILMHDE | Inflorescences | Microwave power (W), 120–700 | Essential oil; astragalin (3-O-glucoside of kaempferol); quercetin (flavonol); luteolin (flavone); kaempferol (flavonol); apigenin (flavone) | 5.12mg/g (essential oil), 3.05mg/g (total flavonoids) | [102] |
| Plant Material | Family Name | Extraction Method | Plant Organ | Conditions | Obtained Compounds | Extraction Yield | Ref. |
|---|---|---|---|---|---|---|---|
| Beetroot, Beta vulgaris L., 1753 | Pedaliaceae | PEF | Tubers | Nine electric pulses at a constant field strength of 1 kV/cm, with a pulse length of 10 µs; time (min), 60 | Betanin (aglycone) | 60–80% | [139] |
| Peppermint, Mentha piperita L. | Lamiaceae | OAHD | Fresh aerial parts | Frequency (kHz), 20, 50, 100; intensity (V), 220, 380; time (min), 60 | Increased yields of essential oil | 2.29–2.58% | [132] |
| Olive, Olea europaea L. | Oleaceae | HVED | Kernel | Needle plate geometry electrodes; voltage (kV), 40; time (µs), 10 | Protein, phenolic compounds | 555.8–607.5 mg GAE/L | [137] |
| Sesame, Sesamum indicum L. | Pedaliaceae | HVED | Seeds | Disc electrode in the bottom (3.5 cm of diameter) and one needle electrode in the top; voltage (kV), 40; time (µs), 10 | Enhanced oil content | 4.9–22.4% | [138] |
| Oregano, Origanum vulgare subsp. viride | Lamiaceae | OAHD | Dried plant | Intensity (V), 100, 150, 200; temperature (°), 50; time (min), 5 | Increased yields of essential oil | 9.5–10.6% | [133] |
| Plant | Family Name | Optimized Process | Process Parameters | Design Method | Independent Variables | Ref. |
|---|---|---|---|---|---|---|
| Lion’s mane mushroom, Hericium erinaceus (Bull.) Persoon | Hericiaceae | EAE | Polysaccharides’ yield | BBD | pH, temperature time | [145] |
| Mongolian milkvetch, Astragalus membranaceus Schischkin hairy root cultures | Fabaceae | Optimal biomass production | Isoflavonoids | BBD | Culture temperature, sucrose concentration, inoculum size, and harvest time | [59] |
| Yarrow, Achillea millefolium L., 1753 | Asteraceae | MAE | Polyphenolic compounds, flavonoid content, antioxidant activity | CCD | Extraction time, ethanol concentration, liquid/solid ratio, and microwave power | [146] |
| Woad, Isatis tinctoria L. hairy root cultures | Brassicaceae | High speed homogenization coupled with MAE | Alkaloids and flavonoids | BBD | Homogenization time, extraction temperature, microwave power, and extraction time | [77] |
| Mongolian milkvetch, Astragalus membranaceus Schischkin hairy root cultures | Fabaceae | High speed homogenization coupled with CAE | Isoflavonoids | BBD | Negative pressure, homogenization time, liquid/solid ratio, and extraction time | [78] |
| Golden-and-silver honeysuckle, Lonicera japonica Thunb. | Caprifoliaceae | Ultra-turrax based UAE | Organic acids | BBD | Ethanol concentration, time, and liquid/solid ratio | [147] |
| Candy leaf, Stevia rebaudiana (Bertoni) | Asteraceae | MAE | Stevioside rebaudioside-A | CCD | Microwave, power extraction temperature time | [148] |
| Cumin, Cuminum cyminum L. | Apiaceae | MHD | Essential oil | CCD | microwave irradiation time, microwave irradiation power, and moisture content | [149] |
| Hemp, Cannabis sativa L. | Cannabaceae | SFE | Tetrahydrocannabinol | CCD | Pressure, temperature, and co-solvent concentration | [150] |
| Jimsonweed, Datura stramonium hairy roots cultures | Solanaceae | Elicitation | Hyoscyamine | Polynomial models of 3 and 4 degrees | Exposure time and salicylic acid concentration | [144] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fierascu, R.C.; Fierascu, I.; Ortan, A.; Georgiev, M.I.; Sieniawska, E. Innovative Approaches for Recovery of Phytoconstituents from Medicinal/Aromatic Plants and Biotechnological Production. Molecules 2020, 25, 309. https://doi.org/10.3390/molecules25020309
Fierascu RC, Fierascu I, Ortan A, Georgiev MI, Sieniawska E. Innovative Approaches for Recovery of Phytoconstituents from Medicinal/Aromatic Plants and Biotechnological Production. Molecules. 2020; 25(2):309. https://doi.org/10.3390/molecules25020309
Chicago/Turabian StyleFierascu, Radu Claudiu, Irina Fierascu, Alina Ortan, Milen I. Georgiev, and Elwira Sieniawska. 2020. "Innovative Approaches for Recovery of Phytoconstituents from Medicinal/Aromatic Plants and Biotechnological Production" Molecules 25, no. 2: 309. https://doi.org/10.3390/molecules25020309
APA StyleFierascu, R. C., Fierascu, I., Ortan, A., Georgiev, M. I., & Sieniawska, E. (2020). Innovative Approaches for Recovery of Phytoconstituents from Medicinal/Aromatic Plants and Biotechnological Production. Molecules, 25(2), 309. https://doi.org/10.3390/molecules25020309











