Pomegranate Peel as Suitable Source of High-Added Value Bioactives: Tailored Functionalized Meat Products
Abstract
1. Introduction
2. Main Bioactive Constituents Present in Pomegranate Peel
2.1. Primary Metabolites
2.1.1. Polysaccharides
2.1.2. Proteins and Amino Acid
2.2. Secondary Metabolites
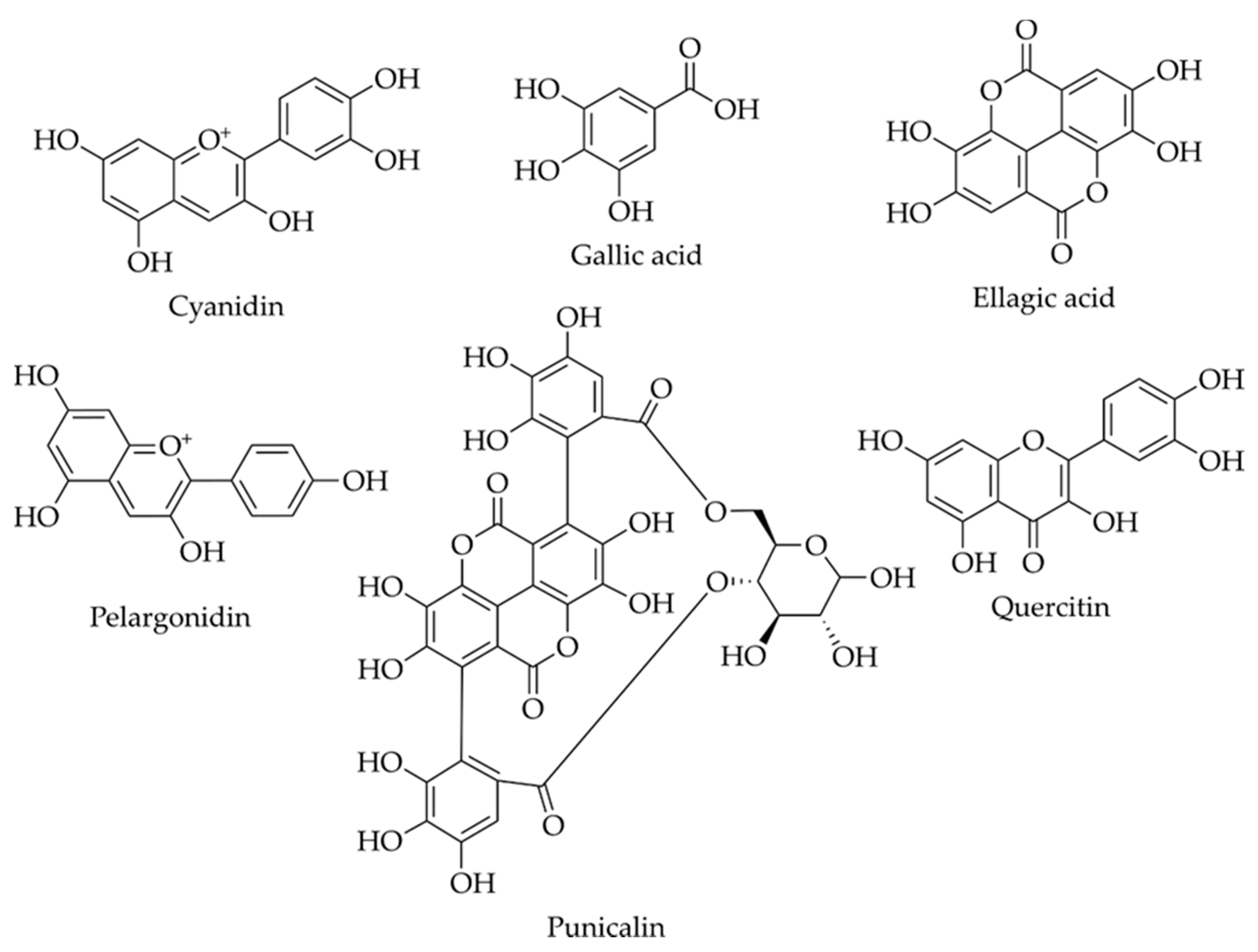
2.2.1. Phenolic Acids
2.2.2. Flavonoids
2.2.3. Tannins
3. Biological Activities of Pomegranate Peel
3.1. Antioxidant Activity
3.2. Antimicrobial Activity
3.3. Other Beneficial Health Properties
4. Role of Pomegranate By-Products in the Formulation of Meat Products
5. Conclusions and Future Trends
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Das, A.K.; Nanda, P.K.; Madane, P.; Biswas, S.; Das, A.; Zhang, W.; Lorenzo, J.M. A comprehensive review on antioxidant dietary fibre enriched meat-based functional foods. Trends Food Sci. Technol. 2020, 99, 323–336. [Google Scholar] [CrossRef]
- Gullón, B.; Gagaoua, M.; Barba, F.J.; Gullón, P.; Zhang, W.; Lorenzo, J.M. Seaweeds as promising resource of bioactive compounds: Overview of novel extraction strategies and design of tailored meat products. Trends Food Sci. Technol. 2020, 100, 1–18. [Google Scholar] [CrossRef]
- Cunha, L.C.M.; Monteiro, M.L.G.; Lorenzo, J.M.; Munekata, P.E.S.; Muchenje, V.; de Carvalho, F.A.L.; Conte-Junior, C.A. Natural antioxidants in processing and storage stability of sheep and goat meat products. Food Res. Int. 2018, 111, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, J.M.; Munekata, P.E.S.; Sant, A.S.; Baptista, R.; Barba, F.J.; Toldrá, F.; Mora, L.; Trindade, M.A. Main characteristics of peanut skin and its role for the preservation of meat products. Trends Food Sci. Technol. 2018, 77, 1–10. [Google Scholar] [CrossRef]
- Domínguez, R.; Pateiro, M.; Gagaoua, M.; Barba, F.J.; Zhang, W.; Lorenzo, J.M. A comprehensive review on lipid oxidation in meat and meat products. Antioxidants 2019, 8, 429. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, J.M.; Batlle, R.; Gómez, M. Extension of the shelf-life of foal meat with two antioxidant active packaging systems. LWT Food Sci. Technol. 2014, 59, 181–188. [Google Scholar] [CrossRef]
- Smaoui, S.; Hlima, H.B.; Mtibaa, A.C.; Fourati, M.; Sellem, I.; Elhadef, K.; Ennouri, K.; Mellouli, L. Pomegranate peel as phenolic compounds source: Advanced analytical strategies and practical use in meat products. Meat Sci. 2019, 158, 107914. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.S.; Pateiro, M.; Campagnol, P.C.B.; Domínguez, R. Healthy Spanish salchichón enriched with encapsulated n-3 long chain fatty acids in konjac glucomannan matrix. Food Res. Int. 2016, 89, 289–295. [Google Scholar] [CrossRef]
- de Carvalho, F.A.L.; Lorenzo, J.M.; Pateiro, M.; Bermúdez, R.; Purriños, L.; Trindade, M.A. Effect of guarana (Paullinia cupana) seed and pitanga (Eugenia uniflora L.) leaf extracts on lamb burgers with fat replacement by chia oil emulsion during shelf life storage at 2 °C. Food Res. Int. 2019, 125, 108554. [Google Scholar] [CrossRef]
- Heck, R.T.; Saldaña, E.; Lorenzo, J.M.; Correa, L.P.; Fagundes, M.B.; Cichoski, A.J.; de Menezes, C.R.; Wagner, R.; Campagnol, P.C.B. Hydrogelled emulsion from chia and linseed oils: A promising strategy to produce low-fat burgers with a healthier lipid profile. Meat Sci. 2019, 156, 174–182. [Google Scholar] [CrossRef]
- Franco, D.; Martins, A.J.; López-Pedrouso, M.; Cerqueira, M.A.; Purriños, L.; Pastrana, L.M.; Vicente, A.A.; Zapata, C.; Lorenzo, J.M. Evaluation of linseed oil oleogels to partially replace pork backfat in fermented sausages. J. Sci. Food Agric. 2020, 100, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Pateiro, M.; Barba, F.J.; Domínguez, R.; Sant’Ana, A.S.; Mousavi Khaneghah, A.; Gavahian, M.; Gómez, B.; Lorenzo, J.M. Essential oils as natural additives to prevent oxidation reactions in meat and meat products: A review. Food Res. Int. 2018, 113, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, J.M.; Pateiro, M.; Domínguez, R.; Barba, F.J.; Putnik, P.; Kovačević, D.B.; Shpigelman, A.; Granato, D.; Franco, D. Berries extracts as natural antioxidants in meat products: A review. Food Res. Int. 2018, 106, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Falowo, A.B.; Mukumbo, F.E.; Idamokoro, E.M.; Lorenzo, J.M.; Afolayan, A.J.; Muchenje, V. Multi-functional application of Moringa oleifera Lam. in nutrition and animal food products: A review. Food Res. Int. 2018, 106, 317–334. [Google Scholar] [CrossRef] [PubMed]
- Žugčić, T.; Abdelkebir, R.; Alcantara, C.; Collado, M.C.; García-Pérez, J.V.; Meléndez-Martínez, A.J.; Režek Jambrak, A.; Lorenzo, J.M.; Barba, F.J. From extraction of valuable compounds to health promoting benefits of olive leaves through bioaccessibility, bioavailability and impact on gut microbiota. Trends Food Sci. Technol. 2019, 83, 63–77. [Google Scholar] [CrossRef]
- Calderón-Oliver, M.; López-Hernández, L.H. Food Vegetable and Fruit Waste Used in Meat Products. Food Rev. Int. 2020, 00, 1–27. [Google Scholar] [CrossRef]
- Munekata, P.E.S.; Rocchetti, G.; Pateiro, M.; Lucini, L.; Domínguez, R.; Lorenzo, J.M. Addition of plant extracts to meat and meat products to extend shelf-life and health-promoting attributes: An overview. Curr. Opin. Food Sci. 2020, 31, 81–87. [Google Scholar] [CrossRef]
- Echegaray, N.; Gómez, B.; Barba, F.J.; Franco, D.; Estévez, M.; Carballo, J.; Marszałek, K.; Lorenzo, J.M. Chestnuts and by-products as source of natural antioxidants in meat and meat products: A review. Trends Food Sci. Technol. 2018, 82, 110–121. [Google Scholar] [CrossRef]
- Domínguez, R.; Gullón, P.; Pateiro, M.; Munekata, P.E.S.; Zhang, W.; Lorenzo, J.M. Tomato as potential source of natural additives for meat industry. A Review. Antioxidants 2020, 9, 73. [Google Scholar] [CrossRef]
- Putnik, P.; Bursać Kovačević, D.; Režek Jambrak, A.; Barba, F.J.; Cravotto, G.; Binello, A.; Lorenzo, J.M.; Shpigelman, A. Innovative “green” and novel strategies for the extraction of bioactive added value compounds from citruswastes - A review. Molecules 2017, 22, 680. [Google Scholar] [CrossRef]
- Franco, D.; Pateiro, M.; Rodríguez Amado, I.; López Pedrouso, M.; Zapata, C.; Vázquez, J.A.; Lorenzo, J.M. Antioxidant ability of potato (Solanum tuberosum) peel extracts to inhibit soybean oil oxidation. Eur. J. Lipid Sci. Technol. 2016, 118, 1891–1902. [Google Scholar] [CrossRef]
- Roselló-soto, E.; Barba, F.J.; Lorenzo, J.M.; Munekata, P.E.S.; Gómez, B.; Carlos, J. Phenolic pro fi le of oils obtained from “ horchata ” by-products assisted by supercritical-CO2 and its relationship with antioxidant and lipid oxidation parameters: Triple TOF-LC-MS-MS characterization. Food Chem. 2019, 274, 865–871. [Google Scholar] [CrossRef]
- Sharayei, P.; Azarpazhooh, E.; Zomorodi, S.; Ramaswamy, H.S. Ultrasound assisted extraction of bioactive compounds from pomegranate (Punica granatum L.) peel. LWT Food Sci. Technol. 2019, 101, 342–350. [Google Scholar] [CrossRef]
- Alexandre, E.M.C.; Silva, S.; Santos, S.A.O.; Silvestre, A.J.D.; Duarte, M.F.; Saraiva, J.A.; Pintado, M. Antimicrobial activity of pomegranate peel extracts performed by high pressure and enzymatic assisted extraction. Food Res. Int. 2019, 115, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Kaderides, K.; Papaoikonomou, L.; Serafim, M.; Goula, A.M. Microwave-assisted extraction of phenolics from pomegranate peels: Optimization, kinetics, and comparison with ultrasounds extraction. Chem. Eng. Process. Process Intensif. 2019, 137, 1–11. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Phenolic compounds as beneficial phytochemicals in pomegranate (Punica granatum L.) peel: A review. Food Chem. 2018, 261, 75–86. [Google Scholar] [CrossRef]
- Pirzadeh, M.; Caporaso, N.; Rauf, A.; Shariati, M.A.; Yessimbekov, Z.; Khan, M.U.; Imran, M.; Mubarak, M.S. Pomegranate as a source of bioactive constituents: A review on their characterization, properties and applications. Crit. Rev. Food Sci. Nutr. 2020, 0, 1–18. [Google Scholar] [CrossRef]
- Andrade, M.A.; Lima, V.; Sanches Silva, A.; Vilarinho, F.; Castilho, M.C.; Khwaldia, K.; Ramos, F. Pomegranate and grape by-products and their active compounds: Are they a valuable source for food applications? Trends Food Sci. Technol. 2019, 86, 68–84. [Google Scholar] [CrossRef]
- Hernández-Corroto, E.; Plaza, M.; Marina, M.L.; García, M.C. Sustainable extraction of proteins and bioactive substances from pomegranate peel (Punica granatum L.) using pressurized liquids and deep eutectic solvents. Innov. Food Sci. Emerg. Technol. 2020, 60, 102314. [Google Scholar] [CrossRef]
- Mushtaq, M.; Sultana, B.; Anwar, F.; Adnan, A.; Rizvi, S.S.H. Enzyme-assisted supercritical fluid extraction of phenolic antioxidants from pomegranate peel. J. Supercrit. Fluids 2015, 104, 122–131. [Google Scholar] [CrossRef]
- Bar-Ya’akov, I.; Tian, L.; Amir, R.; Holland, D. Primary metabolites, anthocyanins, and hydrolyzable tannins in the pomegranate fruit. Front. Plant Sci. 2019, 10, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Talekar, S.; Patti, A.F.; Vijayraghavan, R.; Arora, A. An integrated green biorefinery approach towards simultaneous recovery of pectin and polyphenols coupled with bioethanol production from waste pomegranate peels. Bioresour. Technol. 2018, 266, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Dranca, F.; Oroian, M. Extraction, purification and characterization of pectin from alternative sources with potential technological applications. Food Res. Int. 2018, 113, 327–350. [Google Scholar] [CrossRef]
- Balli, D.; Cecchi, L.; Khatib, M.; Bellumori, M.; Cairone, F.; Carradori, S.; Zengin, G.; Cesa, S.; Innocenti, M.; Mulinacci, N. Characterization of arils juice and peel decoction of fifteen varieties of punica Granatum L.: A focus on anthocyanins, ellagitannins and polysaccharides. Antioxidants 2020, 9, 238. [Google Scholar] [CrossRef]
- Gavlighi, H.A.; Tabarsa, M.; You, S.G.; Surayot, U.; Ghaderi-Ghahfarokhi, M. Extraction, characterization and immunomodulatory property of pectic polysaccharide from pomegranate peels: Enzymatic vs conventional approach. Int. J. Biol. Macromol. 2018, 116, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Abid, M.; Renard, C.M.G.C.; Watrelot, A.A.; Fendri, I.; Attia, H.; Ayadi, M.A. Yield and composition of pectin extracted from Tunisian pomegranate peel. Int. J. Biol. Macromol. 2016, 93, 186–194. [Google Scholar] [CrossRef]
- Shakhmatov, E.G.; Makarova, E.N.; Belyy, V.A. Structural studies of biologically active pectin-containing polysaccharides of pomegranate Punica granatum. Int. J. Biol. Macromol. 2019, 122, 29–36. [Google Scholar] [CrossRef]
- Fazio, A.; Iacopetta, D.; La Torre, C.; Ceramella, J.; Muià, N.; Catalano, A.; Carocci, A.; Sinicropi, M.S. Finding solutions for agricultural wastes: Antioxidant and antitumor properties of pomegranate Akko peel extracts and β-glucan recovery. Food Funct. 2018, 9, 6619–6632. [Google Scholar] [CrossRef]
- Han, B.; Baruah, K.; Cox, E.; Vanrompay, D.; Bossier, P. Structure-functional activity relationship of β-glucans from the perspective of immunomodulation: A Mini-Review. Front. Immunol. 2020, 11, 1–8. [Google Scholar] [CrossRef]
- Jalal, H.; Pal, M.A.; Ahmad, S.R.; Rather, M.; Andrabi, M.; Hamdani, S. Physico-chemical and functional properties of pomegranate peel and seed powder. J. Pharm. Innov. 2018, 7, 1127–1131. [Google Scholar]
- Rowayshed, G.; Salama, A.; Abul-Fadl, M.; Akila-Hamza, S.; Emad, A.M. Nutritional and chemical evaluation for pomegranate (Punica granatum L.) fruit peel and seeds powders by products. Middle East J. Appl. Sci. 2013, 3, 169–179. [Google Scholar]
- Hernández-Corroto, E.; Marina, M.L.; García, M.C. Extraction and identification by high resolution mass spectrometry of bioactive substances in different extracts obtained from pomegranate peel. J. Chromatogr. A 2019, 1594, 82–92. [Google Scholar] [CrossRef] [PubMed]
- El-Hadary, A.E.; Ramadan, M.F. Phenolic profiles, antihyperglycemic, antihyperlipidemic, and antioxidant properties of pomegranate (Punica granatum) peel extract. J. Food Biochem. 2019, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ye, T.; Yang, F.; Hu, M.; Liang, L.; He, H.; Li, Z.; Zeng, A.; Li, Y.; Yao, Y.; et al. Punica granatum (pomegranate) peel extract exerts potent antitumor and anti-metastasis activity in thyroid cancer. RSC Adv. 2016, 6, 84523–84535. [Google Scholar] [CrossRef]
- Rosas-Burgos, E.C.; Burgos-Hernández, A.; Noguera-Artiaga, L.; Kačániová, M.; Hernández-García, F.; Cárdenas-López, J.L.; Carbonell-Barrachina, Á.A. Antimicrobial activity of pomegranate peel extracts as affected by cultivar. J. Sci. Food Agric. 2017, 97, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Fanali, C.; Tripodo, G.; Dugo, P.; Muleo, R.; Dugo, L.; De Gara, L.; Mondello, L. Analysis of phenolic compounds in different parts of pomegranate (Punica granatum) fruit by HPLC-PDA-ESI/MS and evaluation of their antioxidant activity: Application to different Italian varieties. Anal. Bioanal. Chem. 2018, 410, 3507–3520. [Google Scholar] [CrossRef]
- Yan, L.; Zhou, X.; Shi, L.; Shalimu, D.; Ma, C.; Liu, Y. Phenolic profiles and antioxidant activities of six Chinese pomegranate (Punica granatum L.) cultivars. Int. J. Food Prop. 2017, 20, S94–S107. [Google Scholar] [CrossRef]
- Li, J.; He, X.; Li, M.; Zhao, W.; Liu, L.; Kong, X. Chemical fingerprint and quantitative analysis for quality control of polyphenols extracted from pomegranate peel by HPLC. Food Chem. 2015, 176, 7–11. [Google Scholar] [CrossRef]
- Ambigaipalan, P.; De Camargo, A.C.; Shahidi, F. Phenolic compounds of pomegranate byproducts (outer skin, mesocarp, divider membrane) and their antioxidant activities. J. Agric. Food Chem. 2016, 64, 6584–6604. [Google Scholar] [CrossRef]
- Mphahlele, R.R.; Fawole, O.A.; Makunga, N.P.; Linus Opara, U. Functional properties of pomegranate fruit parts: Influence of packaging systems and storage time. J. Food Meas. Charact. 2017, 11, 2233–2246. [Google Scholar] [CrossRef]
- Masci, A.; Coccia, A.; Lendaro, E.; Mosca, L.; Paolicelli, P.; Cesa, S. Evaluation of different extraction methods from pomegranate whole fruit or peels and the antioxidant and antiproliferative activity of the polyphenolic fraction. Food Chem. 2016, 202, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Gardeli, C.; Varela, K.; Krokida, E.; Mallouchos, A. Investigation of anthocyanins stability from pomegranate juice (Punica Granatum L. Cv Ermioni) under a simulated digestion process. Medicines 2019, 6, 90. [Google Scholar] [CrossRef] [PubMed]
- Ben-Simhon, Z.; Judeinstein, S.; Trainin, T.; Harel-Beja, R.; Bar-Yaakov, I.; Borochov-Neori, H.; Holland, D. A “white” anthocyanin-less pomegranate (Punica granatum L.) caused by an insertion in the coding region of the leucoanthocyanidin dioxygenase (LDOX; ANS) gene. PLoS ONE 2015, 10, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Romeo, F.V.; Ballistreri, G.; Fabroni, S.; Pangallo, S.; Li Destri Nicosia, M.G.; Schena, L.; Rapisarda, P. Chemical characterization of different sumac and pomegranate extracts effective against Botrytis cinerea rots. Molecules 2015, 20, 11941–11958. [Google Scholar] [CrossRef] [PubMed]
- Abid, M.; Yaich, H.; Cheikhrouhou, S.; Khemakhem, I.; Bouaziz, M.; Attia, H.; Ayadi, M.A. Antioxidant properties and phenolic profile characterization by LC–MS/MS of selected Tunisian pomegranate peels. J. Food Sci. Technol. 2017, 54, 2890–2901. [Google Scholar] [CrossRef]
- Wafa, B.A.; Makni, M.; Ammar, S.; Khannous, L.; Hassana, A.B.; Bouaziz, M.; Es-Safi, N.E.; Gdoura, R. Antimicrobial effect of the Tunisian Nana variety Punica granatum L. extracts against Salmonella enterica (serovars Kentucky and Enteritidis) isolated from chicken meat and phenolic composition of its peel extract. Int. J. Food Microbiol. 2017, 241, 123–131. [Google Scholar] [CrossRef]
- Moreira, H.; Slezak, A.; Szyjka, A.; Oszmianski, J.; Gasiorowski, K. Antioxidant and cancer chemopreventive activities of cistus and pomegranate polyphenols. Acta Pol. Pharm. 2017, 74, 688–698. [Google Scholar] [PubMed]
- Fernandes, L.; Pereira, J.A.C.; Lopéz-cortés, I.; Salazar, D.M.; Ramalhosa, E.C.D. Physicochemical changes and antioxidant activity of juice, skin, pellicle and seed of pomegranate (cv. Mollar de Elche ) at different stages of ripening. Food Technol. Biotechnol. 2015, 53, 397–406. [Google Scholar] [CrossRef]
- More, P.R.; Arya, S.S. A novel, green cloud point extraction and separation of phenols and flavonoids from pomegranate peel: An optimization study using RCCD. J. Environ. Chem. Eng. 2019, 7, 103306. [Google Scholar] [CrossRef]
- Rajha, H.N.; Mhanna, T.; El Kantar, S.; El Khoury, A.; Louka, N.; Maroun, R.G. Innovative process of polyphenol recovery from pomegranate peels by combining green deep eutectic solvents and a new infrared technology. LWT Food Sci. Technol. 2019, 111, 138–146. [Google Scholar] [CrossRef]
- Oualcadi, Y.; Sebban, M.F.; Berrekhis, F. Improvement of microwave-assisted Soxhlet extraction of bioactive compounds applied to pomegranate peels. J. Food Process. Preserv. 2020, 44, 1–12. [Google Scholar] [CrossRef]
- Kazemi, M.; Karim, R.; Mirhosseini, H.; Abdul Hamid, A. Optimization of pulsed ultrasound-assisted technique for extraction of phenolics from pomegranate peel of Malas variety: Punicalagin and hydroxybenzoic acids. Food Chem. 2016, 206, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.P.; Zhai, X.C.; Li, L.Q.; Wu, X.X.; Li, B. Response surface optimization of ultrasound-assisted polysaccharides extraction from pomegranate peel. Food Chem. 2015, 177, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, E.M.C.; Araújo, P.; Duarte, M.F.; de Freitas, V.; Pintado, M.; Saraiva, J.A. Experimental design, modeling, and optimization of high-pressure-assisted extraction of bioactive compounds from pomegranate peel. Food Bioprocess Technol. 2017, 10, 886–900. [Google Scholar] [CrossRef]
- Kandylis, P.; Kokkinomagoulos, E. Food applications and potential health benefits of pomegranate and its derivatives. Foods 2020, 9, 122. [Google Scholar] [CrossRef] [PubMed]
- Ismail, T.; Akhtar, S.; Sestili, P.; Riaz, M.; Ismail, A.; Labbe, R.G. Antioxidant, antimicrobial and urease inhibitory activities of phenolics-rich pomegranate peel hydro-alcoholic extracts. J. Food Biochem. 2016, 40, 550–558. [Google Scholar] [CrossRef]
- Alirezalu, K.; Pateiro, M.; Yaghoubi, M.; Alirezalu, A.; Peighambardoust, S.H.; Lorenzo, J.M. Phytochemical constituents, advanced extraction technologies and techno-functional properties of selected Mediterranean plants for use in meat products. A comprehensive review. Trends Food Sci. Technol. 2020, 100, 292–306. [Google Scholar] [CrossRef]
- Moreira, D.; Gullón, B.; Gullón, P.; Gomes, A.; Tavaria, F. Bioactive packaging using antioxidant extracts for the prevention of microbial food-spoilage. Food Funct. 2016, 7, 3273–3282. [Google Scholar] [CrossRef]
- Gullón, B.; Pintado, M.E.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Assessment of polyphenolic profile and antibacterial activity of pomegranate peel (Punica granatum) flour obtained from co-product of juice extraction. Food Control 2016, 59, 94–98. [Google Scholar] [CrossRef]
- Nur Hanani, Z.A.; Aelma Husna, A.B.; Nurul Syahida, S.; Nor Khaizura, M.A.B.; Jamilah, B. Effect of different fruit peels on the functional properties of gelatin/polyethylene bilayer films for active packaging. Food Packag. Shelf Life 2018, 18, 201–211. [Google Scholar] [CrossRef]
- Arun, K.B.; Jayamurthy, P.; Anusha, C.V.; Mahesh, S.K.; Nisha, P. Studies on activity guided fractionation of pomegranate peel extracts and its effect on antidiabetic and cardiovascular protection properties. J. Food Process. Preserv. 2017, 41, 1–12. [Google Scholar] [CrossRef]
- Kang, S.J.; Choi, B.R.; Lee, E.K.; Kim, S.H.; Yi, H.Y.; Park, H.R.; Song, C.H.; Lee, Y.J.; Ku, S.K. Inhibitory effect of dried pomegranate concentration powder on melanogenesis in B16F10 melanoma cells; involvement of p38 and PKA signaling pathways. Int. J. Mol. Sci. 2015, 16, 24219–24242. [Google Scholar] [CrossRef] [PubMed]
- Laosirisathian, N.; Saenjum, C.; Sirithunyalug, J.; Eitssayeam, S.; Sirithunyalug, B.; Chaiyana, W. The chemical composition, antioxidant and anti-tyrosinase activities, and irritation properties of sripanya Punica granatum peel extract. Cosmetics 2020, 7, 7. [Google Scholar] [CrossRef]
- Tortora, K.; Femia, A.P.; Romagnoli, A.; Sineo, I.; Khatib, M.; Mulinacci, N.; Giovannelli, L.; Caderni, G. Pomegranate by-products in colorectal cancer chemoprevention: Effects in Apc-mutated pirc rats and mechanistic studies In Vitro and Ex Vivo. Mol. Nutr. Food Res. 2018, 62, 1–10. [Google Scholar] [CrossRef]
- Deng, Y.; Li, Y.; Yang, F.; Zeng, A.; Yang, S.; Luo, Y.; Zhang, Y.; Xie, Y.; Ye, T.; Xia, Y.; et al. The extract from Punica granatum (pomegranate) peel induces apoptosis and impairs metastasis in prostate cancer cells. Biomed. Pharmacother. 2017, 93, 976–984. [Google Scholar] [CrossRef]
- Mastrogiovanni, F.; Mukhopadhya, A.; Lacetera, N.; Ryan, M.T.; Romani, A.; Bernini, R.; Sweeney, T. Anti-inflammatory effects of pomegranate peel extracts on in vitro human intestinal caco-2 cells and ex vivo porcine colonic tissue explants. Nutrients 2019, 11, 548. [Google Scholar] [CrossRef]
- Stojanović, I.; Šavikin, K.; Đedović, N.; Živković, J.; Saksida, T.; Momčilović, M.; Koprivica, I.; Vujičić, M.; Stanisavljević, S.; Miljković, Đ.; et al. Pomegranate peel extract ameliorates autoimmunity in animal models of multiple sclerosis and type 1 diabetes. J. Funct. Foods 2017, 35, 522–530. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Scientific opinion on the reevaluation of butylated hydroxyanisol – BHA (E 320) as a food additive. EFSA J. 2011, 9, 2392. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Scientific Opinion on the re-evaluation of butylated hydroxytoluene BHT (E 321) as a food additive. EFSA J. 2012, 10, 2588. [Google Scholar] [CrossRef]
- Turgut, S.S.; Soyer, A.; Işıkçı, F. Effect of pomegranate peel extract on lipid and protein oxidation in beef meatballs during refrigerated storage. Meat Sci. 2016, 116, 126–132. [Google Scholar] [CrossRef]
- Turgut, S.S.; Işıkçı, F.; Soyer, A. Antioxidant activity of pomegranate peel extract on lipid and protein oxidation in beef meatballs during frozen storage. Meat Sci. 2017, 129, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Morsy, M.K.; Mekawi, E.; Elsabagh, R. Impact of pomegranate peel nanoparticles on quality attributes of meatballs during refrigerated storage. LWT Food Sci. Technol. 2018, 89, 489–495. [Google Scholar] [CrossRef]
- Fourati, M.; Smaoui, S.; Ben Hlima, H.; Ennouri, K.; Chakchouk Mtibaa, A.; Sellem, I.; Elhadef, K.; Mellouli, L. Synchronised interrelationship between lipid/protein oxidation analysis and sensory attributes in refrigerated minced beef meat formulated with Punica granatum peel extract. Int. J. Food Sci. Technol. 2020, 55, 1080–1087. [Google Scholar] [CrossRef]
- Qin, Y.Y.; Zhang, Z.H.; Li, L.; Xiong, W.; Shi, J.Y.; Zhao, T.R.; Fan, J. Antioxidant effect of pomegranate rind powder extract, pomegranate juice, and pomegranate seed powder extract as antioxidants in raw ground pork meat. Food Sci. Biotechnol. 2013, 22, 1063–1069. [Google Scholar] [CrossRef]
- Firuzi, M.R.; Niakousari, M.; Eskandari, M.H.; Keramat, M.; Gahruie, H.H.; Mousavi Khaneghah, A. Incorporation of pomegranate juice concentrate and pomegranate rind powder extract to improve the oxidative stability of frankfurter during refrigerated storage. LWT Food Sci. Technol. 2019, 102, 237–245. [Google Scholar] [CrossRef]
- Ranucci, D.; Roila, R.; Andoni, E.; Braconi, P.; Branciari, R. Punica granatum and Citrus spp. extract mix affects spoilage microorganisms growth rate in vacuum-packaged cooked sausages made from pork meat, emmer wheat (Triticum dicoccum Schübler), almond (Prunus dulcis Mill.) and hazelnut (Corylus avellana L.). Foods 2019, 8, 664. [Google Scholar] [CrossRef]
- Shahamirian, M.; Eskandari, M.H.; Niakousari, M.; Esteghlal, S.; Hashemi Gahruie, H.; Mousavi Khaneghah, A. Incorporation of pomegranate rind powder extract and pomegranate juice into frozen burgers: Oxidative stability, sensorial and microbiological characteristics. J. Food Sci. Technol. 2019, 56, 1174–1183. [Google Scholar] [CrossRef]
- Andrés, A.I.; Petrón, M.J.; Adámez, J.D.; López, M.; Timón, M.L. Food by-products as potential antioxidant and antimicrobial additives in chill stored raw lamb patties. Meat Sci. 2017, 129, 62–70. [Google Scholar] [CrossRef]
- Bouarab-Chibane, L.; Ouled-Bouhedda, B.; Leonard, L.; Gemelas, L.; Bouajila, J.; Ferhout, H.; Cottaz, A.; Joly, C.; Degraeve, P.; Oulahal, N. Preservation of fresh ground beef patties using plant extracts combined with a modified atmosphere packaging. Eur. Food Res. Technol. 2017, 243, 1997–2009. [Google Scholar] [CrossRef]
- Zeeb, B.; Schöck, V.; Schmid, N.; Majer, L.; Herrmann, K.; Hinrichs, J.; Weiss, J. Mixing behaviour of WPI-pectin-complexes in meat dispersions: Impact of biopolymer ratios. Food Funct. 2017, 8, 333–340. [Google Scholar] [CrossRef]
- Sharma, P.; Yadav, S. Effect of incorporation of pomegranate peel and bagasse powder and their extracts on quality characteristics of chicken meat patties. Food Sci. Anim. Resour. 2020, 40, 388–400. [Google Scholar] [CrossRef] [PubMed]
- Santhi, D.; Kalaikannan, A.; Natarajan, A. Characteristics and composition of emulsion-based functional low-fat chicken meat balls fortified with dietary fiber sources. J. Food Process Eng. 2020, 43, 1–12. [Google Scholar] [CrossRef]
- Devatkal, S.K.; Thorat, P.; Manjunatha, M. Effect of vacuum packaging and pomegranate peel extract on quality aspects of ground goat meat and nuggets. J. Food Sci. Technol. 2014, 51, 2685–2691. [Google Scholar] [CrossRef] [PubMed]
- Hayrapetyan, H.; Hazeleger, W.C.; Beumer, R.R. Inhibition of Listeria monocytogenes by pomegranate (Punica granatum) peel extract in meat paté at different temperatures. Food Control 2012, 23, 66–72. [Google Scholar] [CrossRef]
| Technology | Extraction Conditions | Identification Method | Outcomes | Reference |
|---|---|---|---|---|
| SLE | Methanol: water (4:1, v/v) for 3 days at room temperature, LSR of 5:1 (mL/g) | HPLC-PDA | TPC: 188.9 mg GAE/g dw; TFC: 13.95 mg QE/g dw Identification of 23 phenolic compounds and 20 flavonoids | [43] |
| CPE | For total phenols: 8.22% Triton X-114, 4% NaCl at 36.80 °C and pH 4 For flavonoids: 8.27% Triton X-114, 4.06% NaCl at 34.30 °C and pH 5.07 | Not indicated | TPC: 205.2 mg of GAE/g; TFC: 60.05 mg of QE/g | [59] |
| SLE, IR and UAE | Solvents: water, 50% ethanol and 8 different DES. The temperature for the three technologies was fixed at 50 °C | HPLC-DAD | The combination of DES and IR leads to the highest yield of polyphenols (152 mg/g) with high antioxidant activity and good antimicrobial properties. Identification of caffeic acid, kaempferol, luteolin, protocatechuic acid, ellagic acid, chlorogenic acid, hydroxybenzoic acid, gallic acid and quercetin | [60] |
| MAE and UAE | For MAE: 50% aqueous ethanol; LSR: 60:1 mL/g; power, 600 W for 4 min For UAE: water, LSR: 32.2:1 mL/g, amplitude level: 39.8%, pulse duration/pulse interval ratio, 1.2/1 at 34.7 °C for 10 min | HPLC-UV–vis | TPC (MAE): 199.4 mg GAE/g; TPC (UAE): 119.82 mg GAE/g. Identification and quantification of punicalagin: 143.64 mg/g for MAE and 138.8 mg/g for UAE | [25] |
| MASE | TPC: 600 W, 24% ethanol for 35 min; TFC: 800 W, 30% ethanol for 25 min; TTC: 800 W, 30% ethanol for 15 min | UHPLC/ESI/MS | TPC: 373 mg GAE/g; TFC: 155 mg RE/g; TTC: 317 mg GAE/g. Identification of 13 phenolic compounds, mainly ellagitannins and granatin | [61] |
| PUAE | Intensity level: 105 W/cm2; duty cycle: 50% for 10 min | HPLC-UV-DAD | Punicalagin: 146.58 mg/g; ellagic acid: 20.66 mg/g; gallic acid: 0.053 mg/g | [62] |
| UAE | Ultrasonic amplitude 60% for 6.2 min | Not indicated | Yield: 13.1%; TPC: 42.2 mg GAE/g; DPPH: 88%; FRAP: 1824.6 μmol Fe2+/g; IC50: 0.51 mg/mL | [23] |
| UAE | Ultrasonic power of 148 W, 55 °C for 63 min using LSR: 24 mL/g | Not indicated | Polysaccharide yield: 13.658% | [63] |
| HIFU | Choline chloride:acetic acid:water (1:1:10 M), amplitude of 60% for 11 min | HPLC-ESI-Q-TOF/MS | Protein: 20 mg/g. Identification of 23 different peptides and of 20 different phenolic compounds, mainly punicalin, punicalagin, galloyl-HHDPhexoside, ellagic acid-hexoside, and ellagic acid | [29] |
| HPE | Depending on the variable evaluated, the conditions ranged between 356 and 600 MPa and 32–80% ethanol for 23–30 min | UHPLC-DAD/LC-DAD/ESI-MS | Yield: 31 mg/g; TPC: 52 mg GAE/g; TFC: 20 mg of QE/g; TTC: 2.02 mg of CAE/g; TAC: 86 mg cyd-3-glu/g; ABTS: 269 mg TE/g; DPPH: 314 mg TE/g; FRAP: 436 mg TE/g | [64] |
| HPE + EE | HPE conditions: 300 MPa for 15 min; EE: 4% pectinase and 4% cellulase for 15 min | HPLC-DAD-MSn | Total extraction yield: 41%; TPC: 207 mg GAE/g; DPPH: 334 mg TE/g Identification of quinic acid, a punicalin isomer, two bis-HHDP-glucoside isomers, 2-O-galloylpunicalagin, two punicalagin isomers, galloyl-HHDP-glucoside and digalloylpentoside | [24] |
| EASCFE | EE: cocktail enzyme (mixture of cellulase, pectinase and protease; 50:25:25) at 3.8%, 49 °C, pH 6.7 for 85 min; SCFE: ethanol as co-solvent (2 g/min), 55 °C, 300 bar for 100–120 min | HPLC-DAD-ESI–MS | TPC: 301.53 mg GAE/g Identification of p-coumaric acid, vanillic acid, gallic acid, caffeic acid, ferulic acid, syringic acid, sinapic acid | [30] |
| Meat Product | Material | Amount Used | Storage Conditions | Main Effects | References |
|---|---|---|---|---|---|
| Beef meatballs | PPAE | 0.5% and 1% | Refrigerated at 4 °C for 8 days | Decrease of lipid and protein oxidation. Prevents rancid odor formation. Improved shelf life | [80] |
| Frozen at −18 °C for 6 months | [81] | ||||
| LPP-NPs | 1% and 1.5% | Refrigerated at 4 °C for 15 days | Decreased peroxide, TBARS and total volatile base nitrogen contents. Improved microbiological quality. No negative impact on sensory properties | [82] | |
| Minced beef meat | PPEE | 0.1%, 0.5% and 1% | Refrigerated at 4 °C for 21 days | Reduction of oxidative deterioration. Inhibition of growth of spoilage microorganisms. Higher score for color, appearance, odor and overall acceptability | [83] |
| Raw ground pork meat | PRPE | 0.02% | Refrigerated at 4 °C for 12 days | Reduction in peroxide and TBARS values. Decreased lightness value. Better overall acceptability of the treated samples | [84] |
| Frankfurter | PRPE | 10 mg GAE/100 g | Refrigerated 4 °C for 60 days | Reduction in peroxide and TBARS values. Increase in L* value and reduction in a* and b* values | [85] |
| Cooked sausages | Naturmix WM® | 5‰ and 10 ‰ | Refrigerated at 4 °C for 60 days in vacuum-packaged | Delay of the growth of total viable count, psychrotrophic microbial counts and Lactobacillus spp. Better acceptability of the product. Enhanced shelf life | [86] |
| Beef burgers | PRPE | 0.01% | Frozen at −18 °C for 90 days | Retarded lipid oxidation. Reduced growth of aerobic bacteria. Improved the overall acceptability of the product | [87] |
| Beef patties | PP | 10 g/kg | Refrigerated at 4 °C for 12 days in a high O2 atmosphere | Reduction in TBARS levels. Decrease redness value. Modified the texture and taste | [89] |
| Lamb patties | POM | 1000 mg/kg | Refrigerated at 2 °C for 7 days | Reduced the growth of mesophile bacteria and psychrotrophic bacteria | [88] |
| Chicken patties | PP/PPAE | 2% and 6% | Refrigerated at 4 °C for 16 days | Increased the content of phenolics, fiber and ashes. Improved the water holding capacity, emulsion stability and cooking yield. Retarded lipid oxidation and microbial deterioration. | [91] |
| Low-fat chicken meatballs | PPP | 0.5%, 1%, 1.5% and 2% | Not indicated | Improved the fiber level. Decrease of sensory scores with the increase % of PPP | [92] |
| Ground goat meat and cooked nuggets | PPE | 1% | AP and VP for 25 days at 4 °C | Reduction in TBARS content. Extended shelf life | [93] |
| Meat paté | PPE | 7.5% (v/v) | Refrigerated at 4 °C for 46 days | Inhibition of Listeria monocytogenes | [94] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gullón, P.; Astray, G.; Gullón, B.; Tomasevic, I.; Lorenzo, J.M. Pomegranate Peel as Suitable Source of High-Added Value Bioactives: Tailored Functionalized Meat Products. Molecules 2020, 25, 2859. https://doi.org/10.3390/molecules25122859
Gullón P, Astray G, Gullón B, Tomasevic I, Lorenzo JM. Pomegranate Peel as Suitable Source of High-Added Value Bioactives: Tailored Functionalized Meat Products. Molecules. 2020; 25(12):2859. https://doi.org/10.3390/molecules25122859
Chicago/Turabian StyleGullón, Patricia, Gonzalo Astray, Beatriz Gullón, Igor Tomasevic, and José M. Lorenzo. 2020. "Pomegranate Peel as Suitable Source of High-Added Value Bioactives: Tailored Functionalized Meat Products" Molecules 25, no. 12: 2859. https://doi.org/10.3390/molecules25122859
APA StyleGullón, P., Astray, G., Gullón, B., Tomasevic, I., & Lorenzo, J. M. (2020). Pomegranate Peel as Suitable Source of High-Added Value Bioactives: Tailored Functionalized Meat Products. Molecules, 25(12), 2859. https://doi.org/10.3390/molecules25122859








