Phenolics Profile and Antioxidant Activity of Special Beers
Abstract
1. Introduction
2. Results
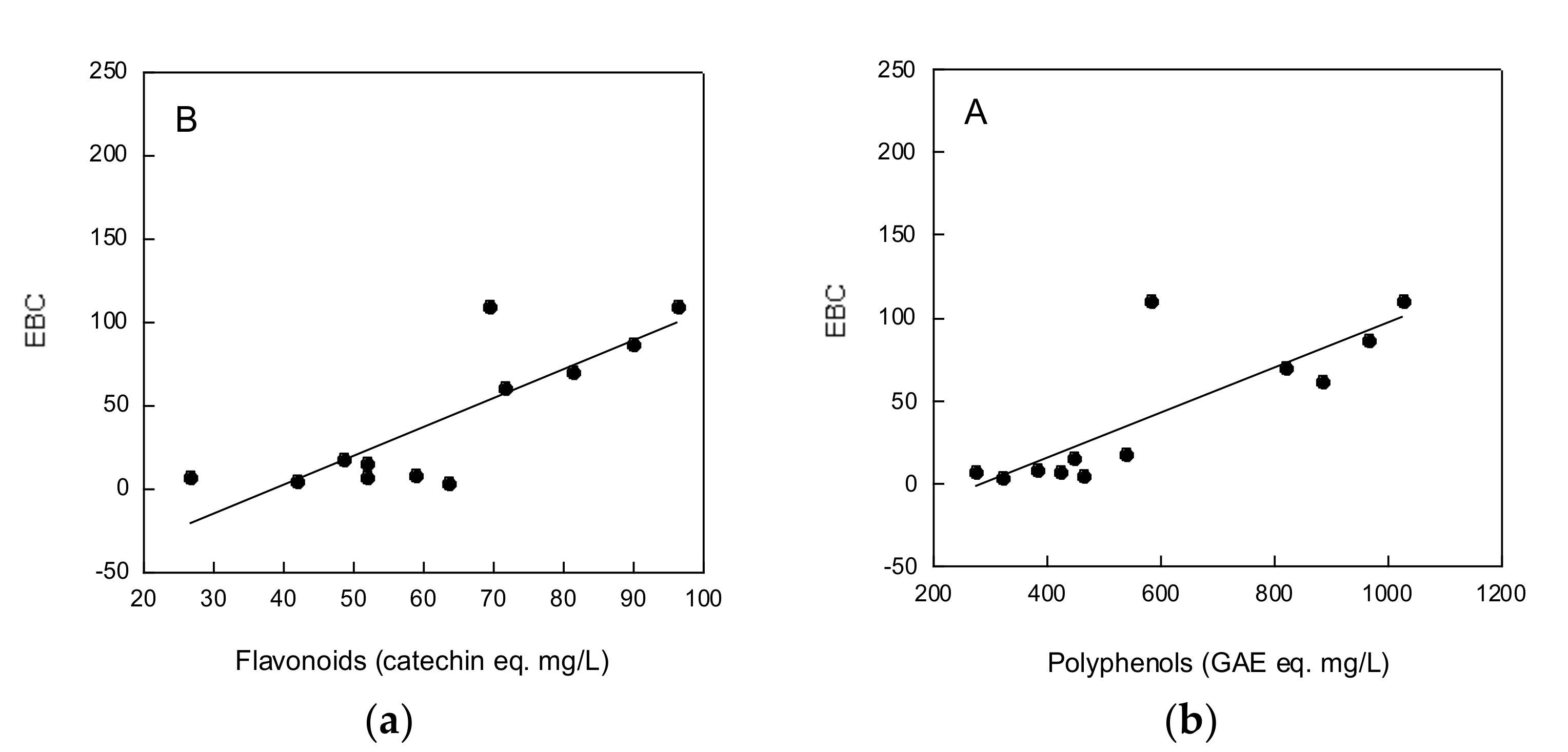
2.1. Beers’ Characterization
2.2. Total Polyphenols and Flavonoids Contents of Beers
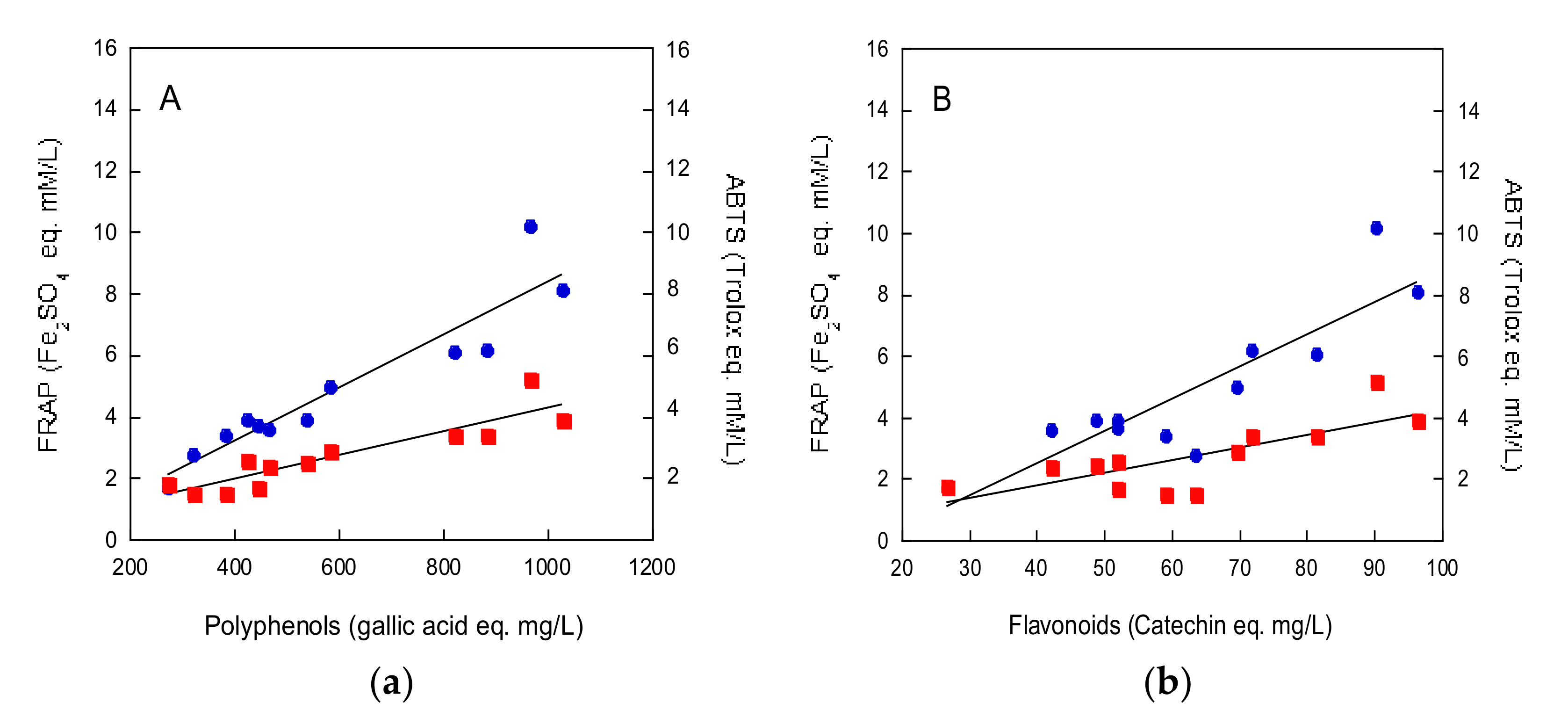
2.3. Beers Antioxidant Activity
2.4. Phenolics Profile Analyses
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Beers
4.3. Beers’ Analyses
4.4. Beer Treatment for Phenolics Profile Determination by High Performance Liquid Chromatography (HPLC)
4.5. HPLC Instrumentation
4.6. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Aruoma, O. Free radicals, oxidative stress and antioxidants in human health and diseases. J. Am. Oil Chem. Soc. 1998, 75, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130, 2073S–2085S. [Google Scholar] [CrossRef] [PubMed]
- Pulido, R.; Hernandez-Garcia, M.; Saura-Calixto, F. Contribution of beverages to the intake of lipophilic and hydrophilic antioxidants in the Spanish diet. Eur. J. Clin. Nutr. 2003, 57, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Rienks, J.; Barbaresko, J.; Nothlings, U. Association of polyphenol biomarkers with cardiovascular disease and mortality risk: A systematic review and meta-analysis of observational studies. Nutrients 2017, 9, e415. [Google Scholar] [CrossRef]
- Grosso, G.; Micek, A.; Godos, J.; Pajak, A.; Sciacca, S.; Galvano, F.; Giovannucci, E.L. Dietary flavonoid and lignan intake and mortality in prospective cohort studies: Systematic review and dose-response meta-analysis. Am. J. Epidemiol. 2017, 185, 1304–1316. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Callemien, D.; Jerkovic, V.; Rozenberg, R.; Collin, S. Hop as an interesting source of resveratrol for brewers: Optimization of the extraction and quantitative study by liquid chromatography/atmosferic pressure chemical ionization tandem mass spectrometry. J. Agric. Food Chem. 2005, 53, 424–429. [Google Scholar] [CrossRef]
- De Keukeleire, D.; de Cooman, L.; Rong, H.; Heyerick, A.; Kalita, J.; Milligan, S.R. Functional properties of hop polyphenols. Basic Life Sci. 1999, 66, 739–760. [Google Scholar]
- Gronbaek, M.; Deis, A.; Sorensen, T.I.; Becker, U.; Schnohr, P.; Jensen, G. Mortality associated with moderate intakes of wine, beer and spirits. Br. Med. J. 1995, 310, 1165–1169. [Google Scholar] [CrossRef]
- Gorinstein, S.; Caspi, A.; Libman, E.; Leontowicz, H.; Leontowicz, M.; Tahsma, Z.; Katrich, E.; Jastrzebski, Z.; Trakhtenberg, S. Bioactivity of beer and its influence on human metabolism. Int. J. Food Sci. Nutr. 2007, 58, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, N.M.; Palmer, B.F. Nutritional and health benefits of beer. Am. J. Med. Sci. 2000, 320, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, S.; Di Castelnuovo, A.; Donati, M.B.; Iacoviello, L.; de Gaetano, G. Wine, beer or spirit drinking in relation to fatal and non-fatal cardiovascular events: A meta-analysis. Eur. J. Epidemiol. 2011, 26, 833–850. [Google Scholar] [CrossRef] [PubMed]
- Nardini, M.; Garaguso, I. Characterization of bioactive compounds and antioxidant activity of fruit beers. Food Chem. 2020, 305, 125437. [Google Scholar] [CrossRef] [PubMed]
- Lugasi, A. Polyphenol content and antioxidant properties of beer. Acta Alimentaria 2003, 32, 181–192. [Google Scholar] [CrossRef]
- Granato, D.; Branco, G.F.; FariaJde, A.; Cruz, A.G. Characterization of Brazilian lager and brown ale beers based on color, phenolic compounds, and antioxidant activity using chemometrics. J. Sci. Food Agric. 2011, 91, 563–571. [Google Scholar] [CrossRef]
- Piazzon, A.; Forte, M.; Nardini, M. Characterization of phenolics content and antioxidant activity of different beer types. J. Agric. Food Chem. 2010, 58, 10677–10683. [Google Scholar] [CrossRef]
- Vinson, J.A.; Mandarano, M.; Hirst, M.; Trevithick, J.R.; Bose, P. Phenol antioxidant quantity and quality in foods: Beers and the effect of two types of beer on an animal model of atherosclerosis. J. Agric. Food Chem. 2003, 51, 5528–5533. [Google Scholar] [CrossRef]
- Gorjanovic, S.; Novakovic, M.; Potkonjak, N.; Leskosek-Cukalovic, I.; Suznjevic, D. Application of a novel antioxidative assay in beer analysis and brewing process monitoring. J. Agric. Food Chem. 2010, 58, 744–751. [Google Scholar] [CrossRef]
- Zhao, H.; Li, H.; Sun, G.; Yang, B.; Zhao, M. Assessment of endogenous antioxidative compounds and antioxidant activities of lager beers. J. Sci. Food Agric. 2013, 93, 910–917. [Google Scholar] [CrossRef]
- Nardini, M.; Ghiselli, A. Determination of free and bound phenolic acids in beer. Food Chem. 2004, 84, 137–143. [Google Scholar] [CrossRef]
- Montanari, L.; Perretti, G.; Natella, F.; Guidi, A.; Fantozzi, P. Organic and phenolic acids in beer. Lebensm. Wiss. Technol. 1999, 32, 535–539. [Google Scholar] [CrossRef]
- Floridi, S.; Montanari, L.; Marconi, O.; Fantozzi, P. Determination of free phenolics in wort and beer by coulometric array detection. J. Agric. Food Chem. 2003, 51, 1548–1554. [Google Scholar] [CrossRef]
- Jandera, P.; Skerikova, V.; Rehova, L.; Hajek, T.; Baldrianova, L.; Skopova, G.; Kellner, V.; Horna, A. RP-HPLC analysis of phenolic compounds and flavonoids in beverages and plant extracts using a CoulArray detector. J. Sep. Sci. 2005, 28, 1005–1022. [Google Scholar] [CrossRef] [PubMed]
- Vanbeneden, N.; Delvaux, F.; Delvaux, R. Determination of hydroxycinnamic acids and volatile phenols in wort and beer by isocratic high-performance liquid chromatography using electrochemical detection. J. Chromatogr. A 2006, 1136, 237–242. [Google Scholar] [CrossRef]
- McMurrough, I.; Roche, G.P.; Cleary, K.G. Phenolic acids in beers and worts. J. Inst. Brew. 1984, 90, 181–187. [Google Scholar] [CrossRef]
- Woffenden, H.M.; Ames, J.M.; Chandra, S. Relationships between antioxidant activity, color, and flavor compounds of crystal malt extract. J. Agric. Food Chem. 2001, 49, 5524–5530. [Google Scholar] [CrossRef]
- Guido, L.F.; Curto, A.F.; Boivin, P.; Benismail, N.; Goncalves, C.R.; Barros, A.A. Correlation of malt quality parameters and beer flavor stability: Multivariate analysis. J. Agric. Food Chem. 2007, 55, 728–733. [Google Scholar] [CrossRef]
- McMurrough, I.; Madigan, D.; Kelly, R.J. The role of flavonoid polyphenols in beer stability. J. Am. Soc. Brew. Chem. 1996, 54, 141–148. [Google Scholar]
- Drost, B.W.; Van der Berg, R.; Freijee, F.J.M.; Van der Velde, E.G.; Hollemans, M. Flavor stability. J. Am. Soc. Brew. Chem. 1990, 48, 124–131. [Google Scholar] [CrossRef]
- Bertuzzi, T.; Mulazzi, A.; Rastelli, S.; Donadini, G.; Rossi, F.; Spigno, G. Targeted healthy compounds in small and large-scale brewed beers. Food Chem. 2020, 310, 125935. [Google Scholar] [CrossRef]
- Perez-Jimenez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010, 64, S112–S120. [Google Scholar] [CrossRef] [PubMed]
- Hayes, D.; Angove, M.J.; Tucci, J.; Dennmis, C. Walnuts (Juglans regia) chemical composition and research in human health. Crit. Rev. Food Sci. Nutr. 2016, 56, 1231–1241. [Google Scholar] [CrossRef] [PubMed]
- Alasalvar, C.; Shahidi, F. Tree nuts: Composition, phytochemicals, and health effects: An overview. In Tree Nuts: Composition, Phytochemicals, and Health Effects; Alasalvar, C., Shahidi, F., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 1–10. [Google Scholar]
- Harnly, J.M.; Doherty, R.F.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Bhagwat, S.; Gebhardt, S. Flavonoid content of US fruits, vegetables and nuts. J. Agric. Food Chem. 2006, 54, 9966–9977. [Google Scholar] [CrossRef] [PubMed]
- De Vasconcelos, M.C.B.M.; Bennett, R.N.; Rosa, E.A.S.; Ferreira-Cardoso, J.V. Industrial processing effects on chesnutnfruits (Castanea Sativa Mill.). 2. Crude protein, free amino acids and phenolic phytochemicals. Int. J. Food Sci. Technol. 2009, 44, 2613–2619. [Google Scholar]
- De Vasconcelos, M.C.B.M.; Bennett, R.N.; Rosa, E.A.S.; Ferreira-Cardoso, J.V. Composition of European chesnut (Castanea Sativa Mill.) and association with health effects: Fresh and processed products. J. Sci. Food Agric. 2010, 90, 1578–1589. [Google Scholar] [CrossRef]
- De Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J.C. Quantitative analysis of flavan-3-ols in Spanish foodstuffs and beverages. J. Agric. Food Chem. 2000, 48, 5331–5337. [Google Scholar] [CrossRef]
- Moreira, A.S.P.; Nunes, F.M.; Simoes, C.; Maciel, E.; Domingues, P.; Domingues, M.R.M.; Coimbra, M.A. Data on coffee composition and mass spectrometry analysis of mixtures of coffee related carbohydrates, phenolic compounds and peptides. Data Brief 2017, 13, 145–161. [Google Scholar] [CrossRef]
- Nardini, M.; Cirillo, E.; Natella, F.; Scaccini, C. Absorption of phenolic acids in humans after coffee consumption. J. Agric. Food Chem. 2002, 50, 5735–5741. [Google Scholar] [CrossRef]
- Rodriguez-Carrasco, Y.; Gaspari, A.; Graziani, G.; Santini, S.; Ritieni, A. Fast analysis of polyphenols and alkaloids in cocoa-based products by ultra-high performance liquid chromatography and Orbitrap high resolution mass spectrometry (UHPLC-Q-Orbitrap-MS/MS). Food Res. Int. 2018, 111, 229–236. [Google Scholar] [CrossRef]
- Zhao, C.N.; Tang, G.Y.; Cao, S.Y.; Xu, X.Y.; Gan, R.; Liu, Q.; Mao, Q.Q.; Shang, A.; Li, H.B. Phenolic profiles and antioxidant activities of 30 tea infusion from green, black, oolong, white, yellow and dark teas. Antioxidants 2019, 8, 215. [Google Scholar] [CrossRef]
- Pastorino, G.; Cornara, L.; Soares, S.; Rodrigues, F.; Oliveira, M.B.P.P. Liquorice (Glycyrrhiza glabra): A phytochemical and pharmacological review. Phytother. Res. 2018, 32, 2323–2339. [Google Scholar] [CrossRef]
- Rizzato, G.; Scalabrin, E.; Radaelli, M.; Capodaglio, R.; Piccolo, O. A new exploration of licorice metabolome. Food Chem. 2017, 221, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Asl, M.N.; Hosseinzadeh, H. Review of pharmacological effects of Glycyrrhiza sp. and its bioactive compounds. Phytother. Res. 2008, 22, 709–724. [Google Scholar] [CrossRef] [PubMed]
- Fiore, C.; Eisenhut, M.; Ragazzi, E.; Zanchin, G.; Armanini, D. A history of the therapeutic use of liquorice in Europe. J. Ethnopharmacol. 2005, 99, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Ciulu, M.; Spano, N.; Pilo, M.I.; Sanna, G. Recent advances in the analysis of phenolic compounds in unifloral honeys. Molecules 2016, 21, 45. [Google Scholar] [CrossRef]
- Cianciosi, D.; Forbes-Hernandez, T.J.; Afrin, S.; Gasparrini, M.; Reboredo-Rodriguez, P.; Manna, P.P.; Zhang, J.; Bravo Lamas, l.; Martinez Florez, S.; Toyos, P.A.; et al. Phenolic compounds in honey and their associated health benefits: A review. Molecules 2018, 23, 2322. [Google Scholar] [CrossRef]
- Cook, N.C.; Samman, S. Review: Flavonoids-chemistry, metabolism, cardioprotective effects and dietary sources. J. Nutr. Biochem. 1996, 7, 66–76. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouysegu, L. Plant polyphenols: Chemical properties, biological activities and synthesis. Angew. Chem. Int. Ed. Engl. 2011, 50, 586–621. [Google Scholar] [CrossRef] [PubMed]
- Chamkha, M.; Cathala, B.; Cheynier, V.; Douillard, R. Phenolic compositionof champagnes from Chardonnay and Pinot Noir vintages. J. Agric. Food Chem. 2003, 51, 3179–3184. [Google Scholar] [CrossRef]
- Stockham, K.; Sheard, A.; Paimin, R.; Buddhadasa, S.; Duong, S.; Orbell, J.D.; Murdoch, T. Comparative studies on the antioxidant properties and polyphenolic content of wine from different growing regions and vintages, a pilot study to investigate chemical markers for climate changes. Food Chem. 2013, 140, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Garaguso, I.; Nardini, M. Polyphenols content, phenolics profile and antioxidant activity of organic red wines produced without sulfur dioxide/sulfites addition in comparison to conventional red wines. Food Chem. 2015, 179, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Nardini, M.; Garaguso, I. Effect of sulfites on antioxidant activity, total polyphenols and flavonoids measurements in white wine. Foods 2018, 7, 35. [Google Scholar] [CrossRef] [PubMed]
- Bourne, L.; Paganga, G.; Baxter, D.; Hughes, P.; Rice-Evans, C. Absorption of ferulic acid from low-alcohol beer. Free Radic. Res. 2000, 32, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Nardini, M.; Natella, F.; Scaccini, C.; Ghiselli, A. Phenolic acids from beer are absorbed and extensively metabolized in humans. J. Nutr. Biochem. 2006, 17, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Piazzon, A.; Vrhovsek, U.; Masuero, D.; Mattivi, F.; Mandoj, F.; Nardini, M. Antioxidant activity of phenolic acids and their metabolites: Synthesis and antioxidant properties of the sulfate derivatives of ferulic and caffeic acids and of the acyl glucuronide of ferulic acid. J. Agric. Food Chem. 2012, 60, 12312–12323. [Google Scholar] [CrossRef]
- Williamson, G.; Manach, C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am. J. Clin. Nutr. 2005, 81, 243S–255S. [Google Scholar] [CrossRef]
- Tome-Carneiro, J.; Larrosa, M.; Gonzales-Sarrias, A.; Tomas-Barberan, F.A.; Garcia-Conesa, M.T.; Espin, J.C. Resveratrol and clinical trials: The crossroad from in vitro studies to human evidence. Curr. Pharm. Design 2013, 19, 6064–6093. [Google Scholar] [CrossRef]
- Smoliga, J.M.; Baur, J.A.; Hausenblas, H.A. Resveratrol and health—A comprehensive review of human clinical trials. Mol. Nutr. Food Res. 2011, 55, 1129–1141. [Google Scholar] [CrossRef]
- Arranz, S.; Chiva-Blanch, G.; Valderas-Martinez, P.; Medina-Remon, A.; Lamuela-Raventos, R.M.; Estruch, R. Wine, beer, alcohol and polyphenols on cardiovascular disease and cancer. Nutrients 2012, 4, 759–781. [Google Scholar] [CrossRef] [PubMed]
- Van der Gaag, M.S.; Ubbink, J.B.; Sillanaukee, P.; Nikkari, S.; Hendriks, H.F.J. Effect of consumption of red wine, spirits and beer on serum homocysteine. Lancet 2000, 335, 1522. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 2002, 50, 3010–3014. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of 1 plasma (FRAP) as a measure of “Antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Pozo-Bayon, M.A.; Hernandez, M.T.; Martin-Alvarez, P.J.; Polo, M.C. Study of low molecular weight phenolic compounds during the aging of sparkling wines manufactured with red and white grape varieties. J. Agric. Food Chem. 2003, 51, 2089–2095. [Google Scholar] [CrossRef]
- Nardini, M.; Forte, M.; Vrhovsek, U.; Mattivi, F.; Viola, R.; Scaccini, C. White wine phenolics are absorbed and extensively metabolized in humans. J. Agric. Food Chem. 2009, 57, 2711–2718. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Beer Code | Food Added | Amount Added (g/L of Beer) | Ingredients |
|---|---|---|---|
| Special Beers: | |||
| WALN | Walnut | 35 | Water, barley malt, oats, walnut, hops, yeast |
| CHES | Chestnut | 40 | Water, barley malt, dried chestnut, hops, yeast |
| GTEA | Green tea | 9 | Water, barley malt, wheat malt, hops, yeast, green tea |
| COFF | Coffee | 35 | Water, barley malt, oats, coffee (80% Arabica, 20% Robusta), hops, yeast |
| COCO | Cocoa beans | 10 | Water, barley malt, oats, carob, cocoa beans, hops, yeast |
| HONE | Honey | 62 | Water, barley malt, wildflower honey, hops, yeast |
| LIQU Conventional Beers: | Licorice | 2 | Water, barley malt, wheat malt, hops, licorice, sugar, yeast |
| ALE 1 | - | - | Water, barley malt, corn, barley, hops, yeast |
| ALE 2 | - | - | Water, barley malt, sugar, hops, yeast |
| ALE 3 | - | - | Water, barley malt, caramelized barley malt, hops, yeast |
| LAGE 1 | - | - | Water, barley malt, maize, hops, yeast |
| LAGE 2 | - | - | Water, barley malt, barley, glucose syrup, hops, yeast |
| Beer Code | Style | Country of Production | Alcohol Strength (% vol) | pH a | IBU | Color EBC |
|---|---|---|---|---|---|---|
| Special Beers: | ||||||
| WALN | Ale | Italy | 4.7 | 4.47 | 30 | 87 |
| CHES | Lager | Italy | 8.0 | 4.64 | 7 | 61 |
| GTEA | Ale | Italy | 4.5 | 4.54 | 12 | 5 |
| COFF | Ale | Italy | 4.5 | 4.04 | 15 | 110 |
| COCO | Ale | Italy | 7.0 | 4.41 | 10 | 110 |
| HONE | Ale | Italy | 6.8 | 4.34 | 8 | 18 |
| LIQU | Ale | Italy | 9.0 | 4.60 | 22 | 70 |
| Conventional Beers: | ||||||
| ALE 1 | Ale | Belgium | 6.6 | 4.39 | 28 | 15 |
| ALE 2 | Ale | Italy | 5.2 | 4.61 | 25 | 8 |
| ALE 3 | Ale | Italy | 5.2 | 4.29 | 35 | 20 |
| LAGE 1 | Lager | Italy | 4.6 | 4.43 | 15 | 7 |
| LAGE 2 | Lager | Italy | 4.8 | 4.87 | 20 | 4 |
| Beer Code | Total Polyphenols Gallic acid Eq. mg/L | Total Flavonoids Catechin Eq. mg/L | FRAP Fe2SO4 Eq. mM | ABTS Trolox Eq. mM |
|---|---|---|---|---|
| Special Beers: | ||||
| WALN | 964.7 ± 9.6 a | 90.1 ± 1.8 a | 10.2 ± 0.02 a | 5.2 ± 0.05 a |
| CHES | 883.4 ± 10.9 b | 71.7 ± 0.9 b | 6.2 ± 0.08 b | 3.4 ± 0.03 b |
| GTEA | 464.4 ± 3.9 f | 42.0 ± 0.3 e | 3.6 ± 0.05 d | 2.4 ± 0.03 e |
| COFF | 582.7 ± 6.4 d | 69.5 ± 1.0 b | 5.0 ± 0.14 e | 2.9 ± 0.03 f |
| COCO | 1026.4 ± 3.0 a | 96.4 ± 2.0 c | 8.1 ± 0.10 c | 3.9 ± 0.04 c |
| HONE | 538.3 ± 8.3 e | 48.7 ± 1.0 f | 3.9 ± 0.01 f | 2.5 ± 0.03 d |
| LIQU | 819.7 ± 6.9 c | 81.4 ± 1.3 d | 6.1 ± 0.04 b | 3.4 ± 0.01 b |
| Conventional Beers: | ||||
| Ale 1 | 446.1 ± 12.6 f,i | 51.9 ± 1.1 g | 3.7 ± 0.17 d,f,h | 1.7 ± 0.03 g,h |
| Ale 2 | 382.7 ± 6.6 l | 59.0 ± 0.9 h | 3.4 ± 0.04 h | 1.5 ± 0.02 i |
| Ale 3 | 424.4 ± 8.7 g,f | 51.9 ± 1.3 f,g | 3.9 ± 0.01 f | 2.6 ± 0.02 d |
| LAGE 1 | 273.8 ± 4.1 h | 26.6 ± 0.1 l | 1.7 ± 0.02 g | 1.8 ± 0.03 g |
| LAGE 2 | 320.6 ± 8.6 m | 63.5 ± 0.8 i | 2.8 ± 0.04 i | 1.5 ± 0.06 h,i |
| Beer Code | ALE 1 | ALE 2 | ALE 3 | LAGE 1 | LAGE 2 |
|---|---|---|---|---|---|
| Phenolic Acids: | |||||
| Chlorogenic | nd | nd | nd | nd | nd |
| Vanillic | |||||
| Free | nd | nd | 2.09 ± 0.08 | nd | nd |
| Total | 2.80 ± 0.05 | 3.58 ± 0.07 | 4.65 ± 0.06 | 4.46 ± 0.12 | 2.3 ± 0.07 |
| Caffeic | |||||
| Free | nd | nd | 1.24 ± 0.10 | nd | nd |
| Total | 3.00 ± 0.20 | 3.38 ± 0.01 | 5.99 ± 0.11 | 1.70 ± 0.08 | 1.61 ± 0.04 |
| Syringic | |||||
| Free | nd | nd | 0.25 ± 0.01 | nd | nd |
| Total | 0.71 ± 0.09 | 0.67 ± 0.04 | 0.51 ± 0.03 | nd | 0.32 ± 0.01 |
| p-Coumaric | |||||
| Free | 0.53 ± 0.03 | 1.12 ± 0.05 | 0.68 ± 0.02 | 1.06 ± 0.05 | 0.35 ± 0.04 |
| Total | 2.00 ± 0.10 | 2.77 ± 0.09 | 2.13 ± 0.04 | 1.56 ± 0.08 | 0.77 ± 0.01 |
| Ferulic | |||||
| Free | 0.90 ± 0.03 | 11.03 ± 0.54 | 2.91 ± 0.11 | 2.12 ± 0.06 | 1.81 ± 0.03 |
| Total | 10.27 ± 1.00 | 19.90 ± 0.21 | 21.66 ± 0.55 | 11.0 ± 0.07 | 13.71 ± 0.49 |
| Sinapic | |||||
| Free | 0.41 ± 0.01 | 0.44 ± 0.21 | 0.98 ± 0.10 | 0.36 ± 0.04 | 1.07 ± 0.04 |
| Total | 4.80 ± 0.05 | 2.19 ± 0.07 | 3.95 ± 0.11 | 3.53 ± 0.03 | 3.07 ± 0.06 |
| Total Phenolic Acids a | 23.58 ± 1.56 | 32.49 ± 0.49 | 38.89 ± 0.90 | 22.25 ± 0.38 | 21.78 ± 0.68 |
| Flavonoids: | |||||
| Catechin | nd | nd | nd | nd | nd |
| Epicatechin | nd | nd | nd | nd | nd |
| Rutin | nd | nd | nd | nd | nd |
| Myricetin | nd | nd | nd | nd | nd |
| Quercetin | nd | nd | nd | nd | nd |
| Stilbenes: | |||||
| Resveratrol | nd | nd | nd | nd | nd |
| Beer Code | WALN | CHES | GTEA | COFF | COCO | HONE | LIQU |
|---|---|---|---|---|---|---|---|
| Phenolic Acids: | |||||||
| Chlorogenic | tr | nd | nd | 1.56 ± 0.10 | nd | nd | nd |
| Vanillic | |||||||
| Free | 0.92 ± 0.12 | 1.57 ± 0.03 | 0.87 ± 0.04 | 0.78 ± 0.03 | 1.14 ± 0.09 | 0.80 ± 0.05 | 1.03 ± 0.10 |
| Total | 2.16 ± 0.26 | 5.09 ± 0.06 | 2.82 ± 0.15 | 2.03 ± 0.14 | 3.39 ± 0.17 | 3.09 ± 0.22 | 2.32 ± 0.11 |
| Caffeic | |||||||
| Free | 0.52 ± 0.01 | 0.24 ± 0.01 | tr | 0.57 ± 0.02 | 0.50 ± 0.02 | tr | 0.56 ± 0.07 |
| Total | 3.16 ± 0.15 | 3.47 ± 0.03 | 1.48 ± 0.18 | 9.20 ± 0.21 | 3.69 ± 0.01 | 2.37 ± 0.17 | 3.71 ± 0.04 |
| Syringic | |||||||
| Free | tr | 0.40 ± 0.03 | 0.62 ± 0.02 | tr | 0.54 ± 0.02 | 0.27 ± 0.01 | 0.40 ± 0.03 |
| Total | tr | 1.24 ± 0.05 | 0.96 ± 0.04 | tr | 1.42 ± 0.05 | 1.24 ± 0.10 | 0.67 ± 0.03 |
| p-Coumaric | |||||||
| Free | 0.68 ± 0.01 | 1.02 ± 0.06 | 0.11 ± 0.01 | 0.36 ± 0.02 | 1.38 ± 0.08 | 0.21 ± 0.01 | 1.06 ± 0.06 |
| Total | 4.32 ± 0.24 | 3.36 ± 0.07 | 2.24 ± 0.16 | 1.93 ± 0.08 | 3.26 ± 0.13 | 1.75 ± 0.03 | 2.95 ± 0.14 |
| Ferulic | |||||||
| Free | 1.05 ± 0.02 | 1.81 ± 0.20 | 0.15 ± 0.01 | 0.63 ± 0.02 | 1.16 ± 0.03 | 0.43 ± 0.01 | 1.32 ± 0.08 |
| Total | 8.22 ± 0.17 | 27.55 ± 0.43 | 14.30 ± 0.40 | 20.50 ± 0.64 | 22.10 ± 0.73 | 19.20 ± 0.33 | 20.63 ± 0.87 |
| Sinapic | |||||||
| Free | 0.45 ± 0.01 | 0.97 ± 0.12 | 0.49 ± 0.01 | 0.24 ± 0.02 | 0.44 ± 0.01 | 0.55 ± 0.01 | 1.03 ± 0.04 |
| Total | 2.68 ± 0.06 | 4.74 ± 0.04 | 4.48 ± 0.08 | 2.52 ± 0.02 | 4.89 ± 0.05 | 6.73 ± 0.03 | 6.66 ± 0.07 |
| Totalphenolic Acids a | 20.54 ± 0.88 | 45.45 ± 0.68 | 26.28 ± 1.01 | 36.18 ± 1.09 | 38.75 ± 1.14 | 34.38 ± 0.88 | 36.94 ± 1.26 |
| Flavonoids: | |||||||
| Catechin | tr | 4.65 ± 0.13 | 2.98 ± 0.09 | tr | 4.58 ± 0.02 | tr | tr |
| Epicatechin | 1.80 ± 0.11 | 3.68 ± 0.12 | 3.09 ± 0.05 | 1.30 ± 0.07 | 1.83 ± 0.11 | 0.94 ± 0.05 | tr |
| Rutin | nd | nd | 0.68 ± 0.02 | nd | nd | 1.29 ± 0.02 | 0.92 ± 0.10 |
| Myricetin | 4.44 ± 0.27 | tr | 1.69 ± 0.05 | 0.39 ± 0.03 | 0.65 ± 0.02 | 2.67 ± 0.18 | 8.82 ± 0.07 |
| Quercetin | 6.55 ± 0.31 | tr | 1.17 ± 0.09 | 0.54 ± 0.02 | 1.52 ± 0.06 | 4.67 ± 0.23 | 2.63 ± 0.15 |
| Stilbenes: | |||||||
| Resveratrol | 0.26 ± 0.20 | 0.35 ± 0.02 | 0.32 ± 0.02 | 0.23 ± 0.01 | 0.31 ± 0.01 | 0.24 ± 0.01 | 0.20 ± 0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nardini, M.; Foddai, M.S. Phenolics Profile and Antioxidant Activity of Special Beers. Molecules 2020, 25, 2466. https://doi.org/10.3390/molecules25112466
Nardini M, Foddai MS. Phenolics Profile and Antioxidant Activity of Special Beers. Molecules. 2020; 25(11):2466. https://doi.org/10.3390/molecules25112466
Chicago/Turabian StyleNardini, Mirella, and Maria Stella Foddai. 2020. "Phenolics Profile and Antioxidant Activity of Special Beers" Molecules 25, no. 11: 2466. https://doi.org/10.3390/molecules25112466
APA StyleNardini, M., & Foddai, M. S. (2020). Phenolics Profile and Antioxidant Activity of Special Beers. Molecules, 25(11), 2466. https://doi.org/10.3390/molecules25112466







