Synthesis of AMPSA Polymeric Derivatives Monitored by Electrical Conductivity and Evaluation of Thermosensitive Properties of Resulting Microspheres
Abstract
1. Introduction
2. Results
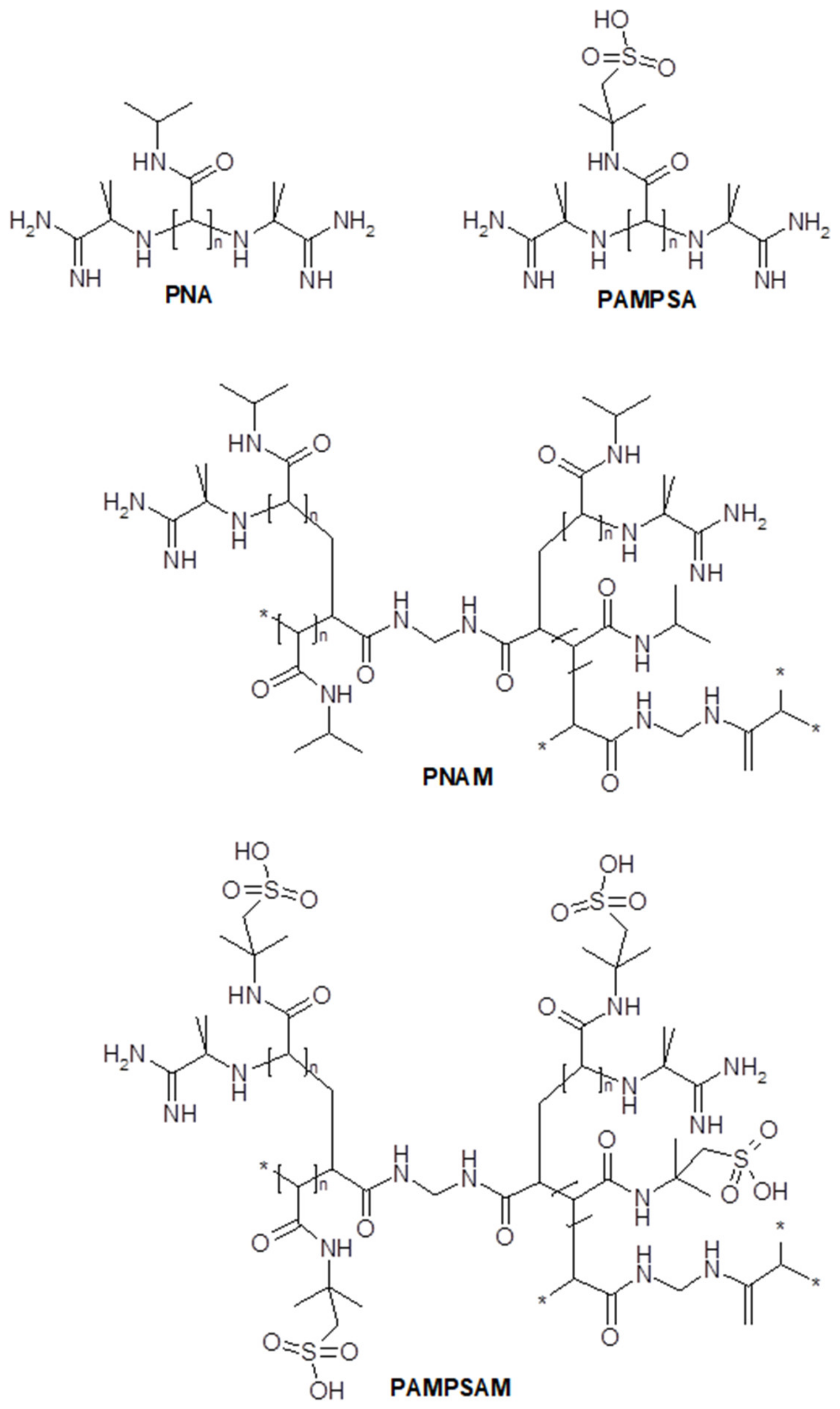
2.1. Synthesis
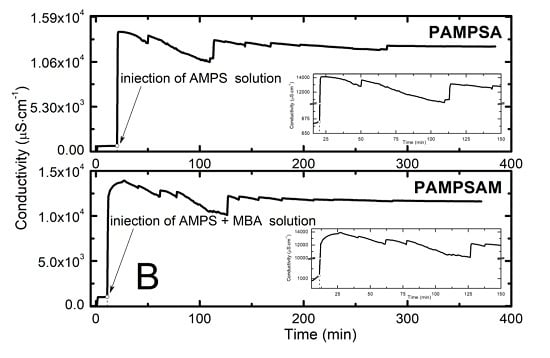
2.2. Conductivity Measurement Analysis
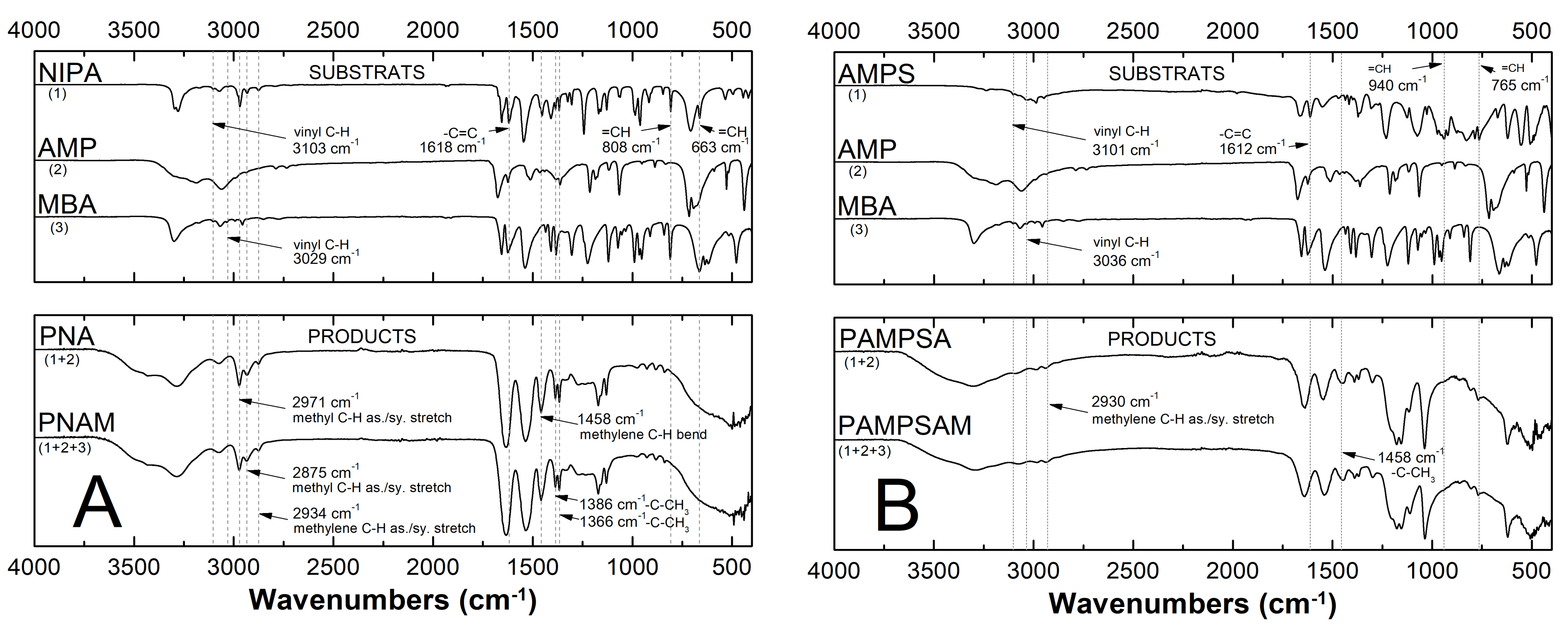
2.3. Attenuated Total Reflection Fourier Transform Infrared Spectroscopy Analysis (ATR-FTIR)
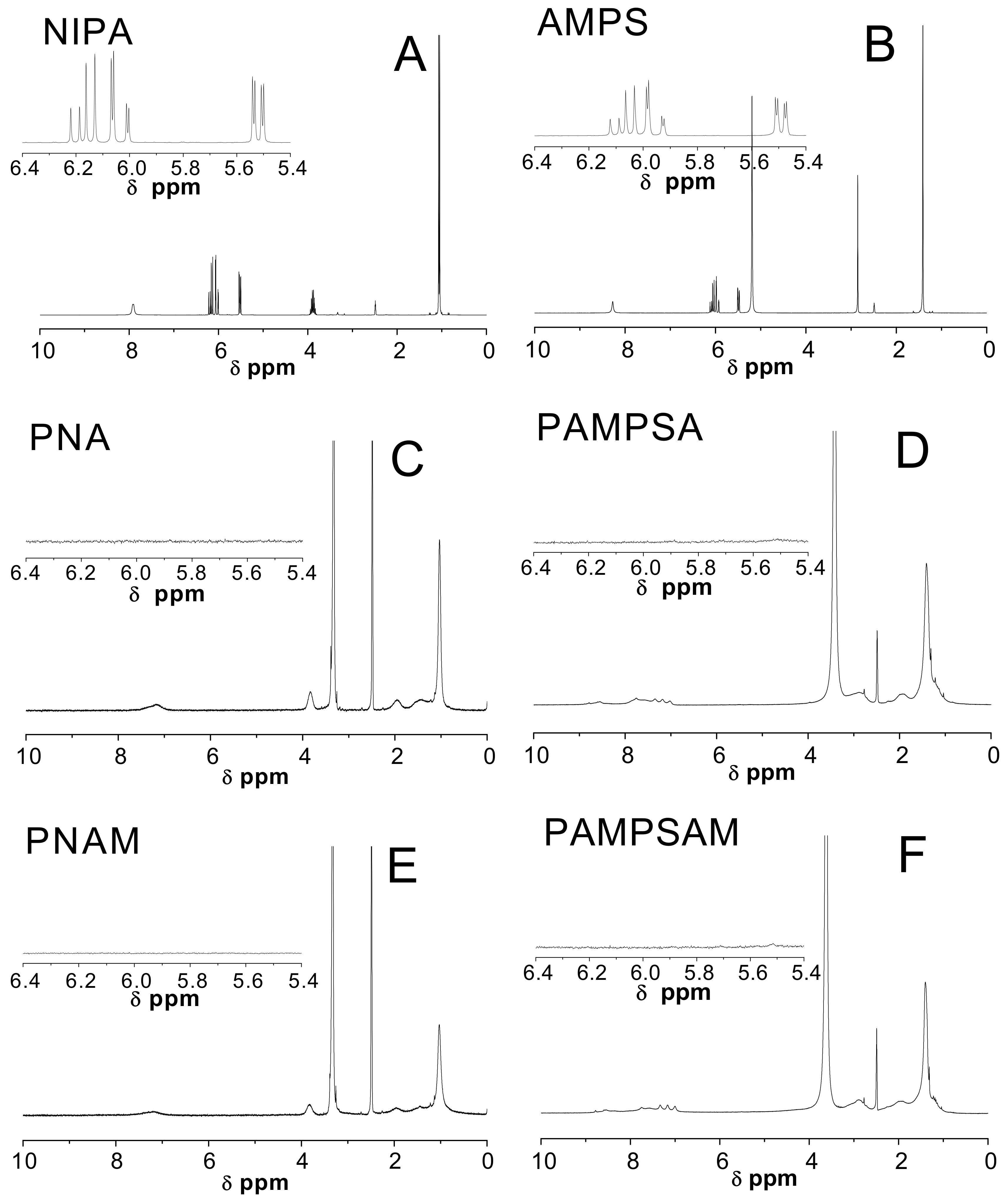
2.4. Nuclear Magnetic Resonance Spectroscopy Analysis (1H-NMR)
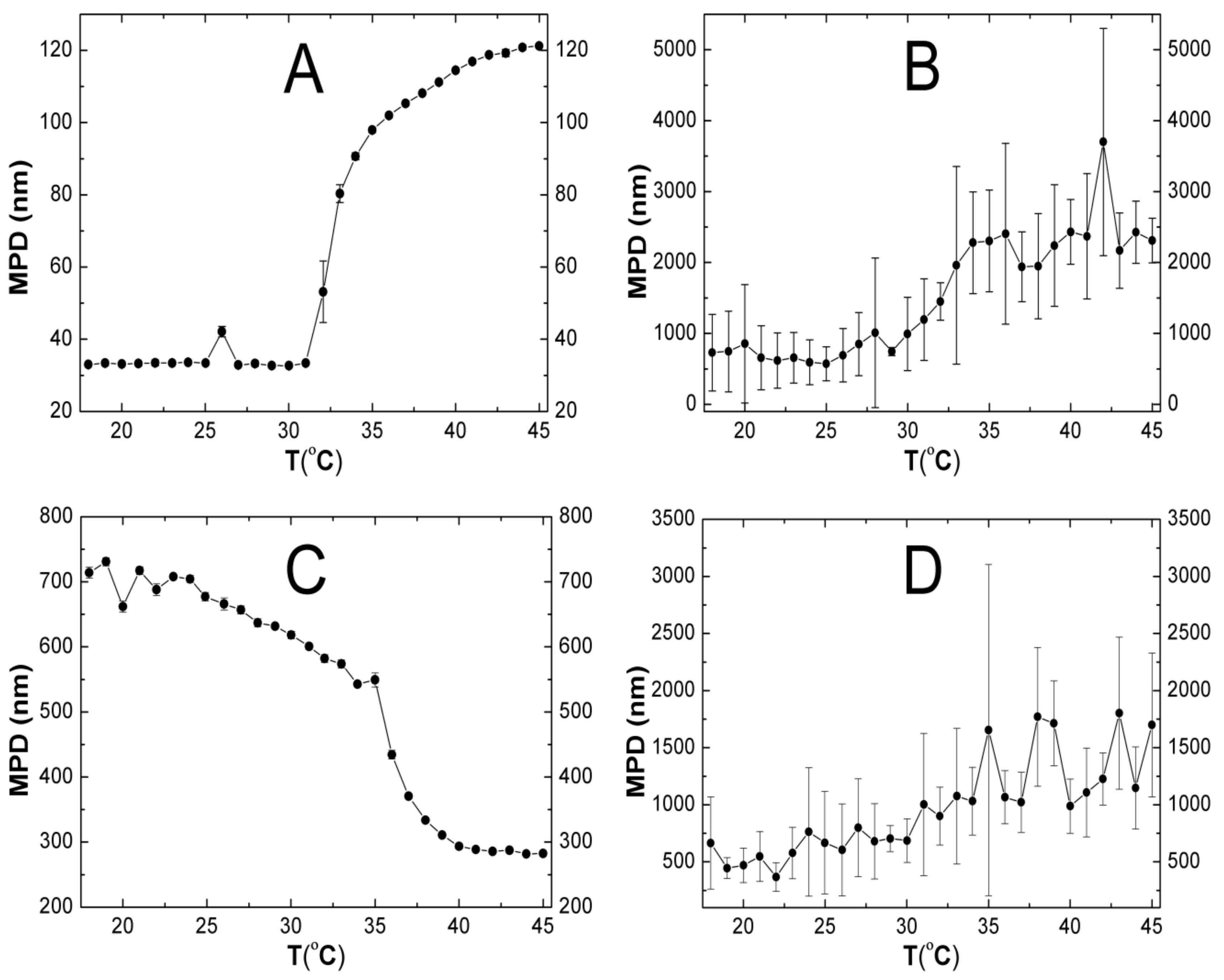
2.5. Hydrodynamic Diameter (HD)
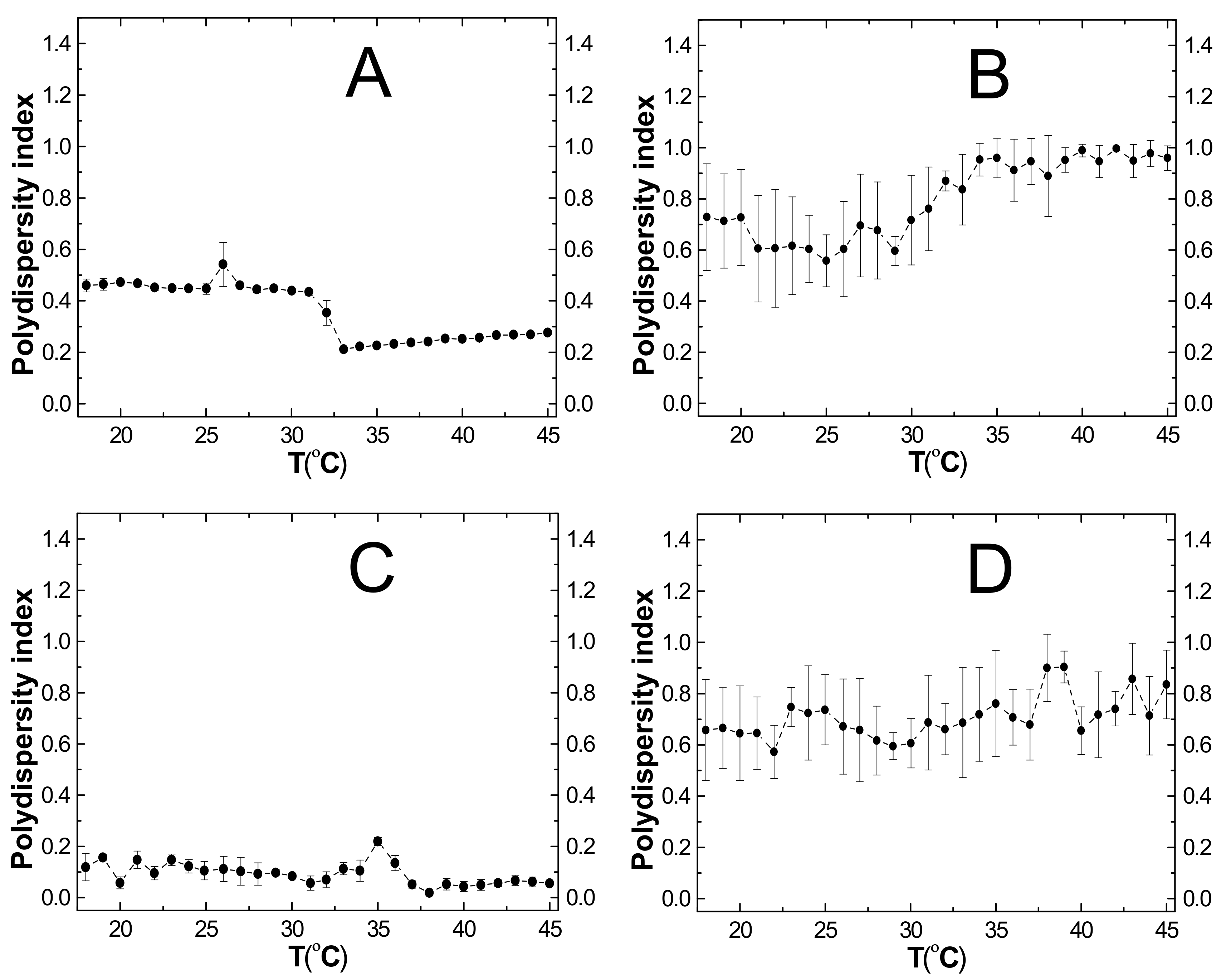
2.6. Polydispersity Index (PDI)
2.7. Zeta Potential (ZP)
2.8. Scanning Electron Microscopy with Energy Dispersive Spectroscopy System (SEM-EDS) Analysis
3. Discussion
3.1. Synthesis
3.2. Conductivity
3.3. ATR-FTIR
3.4. H-NMR
3.5. HD
3.6. PDI
3.7. ZP
3.8. SEM-EDS
4. Materials and Methods
4.1. Materials
4.2. Synthesis
4.3. Conductivity Measurements
4.4. Attenuated Total Reflection Fourier Transformed Infrared Spectroscopy measurements (ATR-FTIR)
4.5. Nuclear Magnetic Resonance Spectroscopy measurements (1H-NMR)
4.6. Hydrodynamic diameter (HD)and polydispersity index(PDI) measurements
4.7. Zeta Potential (ZP) Measurements
4.8. Scanning Electron Microscopy With Energy Dispersive Spectroscopy System (SEM-EDS) Measurements
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nakajima, K.; Honda, S.; Nakamura, Y.; Redondo, F.L.H.; Kohsaka, S.; Yamato, M.; Kikuchi, A.; Okano, T. Intact microglia are cultured and non-invasively harvested without pathological activation using a novel cultured cell recovery method. Biomaterials 2001, 22, 1213–1223. [Google Scholar] [CrossRef]
- Shimizu, T.; Yamato, M.; Kikuchi, A.; Okano, T. Cell sheet engineering for myocardial tissue reconstruction. Biomaterials 2003, 24, 2309–2316. [Google Scholar] [CrossRef]
- Pelton, R.H.; Chibante, P. Preparation of aqueous latices with N-isopropylacrylamide. Colloid. Surf. A 1986, 20, 247–256. [Google Scholar] [CrossRef]
- Pelton, R.H.; Pelton, H.M.; Morphesis, A.; Rowells, R.L. Particle sizes and electrophoretic mobilities of poly(N-isopropylacrylamide) latex. Langmuir 1989, 5, 816–818. [Google Scholar] [CrossRef]
- Ougizawa, T.; Inoue, T.; Kammer, H.W. UCST and LCST behavior in polymer blends. Macromolecules 1985, 18, 2089–2092. [Google Scholar] [CrossRef]
- Heyda, J.; Soll, S.; Yuan, J.; Dzubiella, J. Thermodynamic description of the LCST of charged thermoresponsive copolymers. Macromolecules 2014, 47, 2096–2102. [Google Scholar] [CrossRef]
- Saikia, A.K.; Aggarwal, S.; Mandal, U.K. Preparation and controlled drug release characteristics of thermoresponsive PEG/Poly (NIPAM-co-AMPS) hydrogels. Int. J. Polym. Mat. 2012, 62, 39–44. [Google Scholar] [CrossRef]
- Takegami, Y.; Yokoyama, Y.; Norisugi, O.; Nagatsuma, M.; Takata, K.; Rehman, M.U.; Matsunaga, K.; Yokoi’, H.; Fujiki, S.; Makino, T.; et al. Synthesis and characterization of high-quality skin-cooling sheets containing thermosensitive poly(N-isopropylacrylamide). J. Biom. Mat. Res. B: Appl. Biomater. 2011, 98B, 110–113. [Google Scholar] [CrossRef]
- Indulekha, S.; Arunkumar, P.; Bahadur, D.; Srivastava, R. Thermoresponsive polymeric gel as an on demand transdermal drug delivery system for pain management. Mater. Sci. Eng. C 2016, 62, 113–122. [Google Scholar] [CrossRef]
- Liu, L.; Wang, W.; Ju, X.J.; Xie, R.; Chu, L.Y. Smart thermo-triggered squirting capsules for nanoparticle delivery. Soft Matt. 2010, 6, 3759–3763. [Google Scholar] [CrossRef]
- Gong, C.; Qi, T.; Wei, X.; Qu, Y.; Wu, Q.; Luo, F.; Qian, Z. Thermosensitive polymeric hydrogels as drug delivery systems. Curr. Med. Chem. 2012, 20, 79–94. [Google Scholar] [CrossRef]
- Hamidi, M.; Azadi, A.; Rafiei, P. Hydrogel nanoparticles in drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1638–1649. [Google Scholar] [CrossRef]
- Gupta, P.; Vermani, K.; Garg, S. Hydrogels: From controlled release to pH-responsive drug delivery. Drug Discov. Today 2002, 7, 569–579. [Google Scholar] [CrossRef]
- Qiu, Y.; Park, K. Environment-sensitive hydrogels for drug delivery. Advc. Drug Deliv. Rev. 2001, 53, 321–339. [Google Scholar] [CrossRef]
- Shin, H.S.; Kim, S.Y.; Lee, Y.M. Indomethacin release behaviors from pH and thermoresponsive poly-(vinyl alcohol) and poly-(acrylic acid) IPN hydrogels for site-specific drug delivery. J. Appl. Polym. Sci. 1997, 65, 685–693. [Google Scholar] [CrossRef]
- Wei, H.; Cheng, C.; Chang, C.; Chen, W.Q.; Cheng, S.X.; Zhang, X.Z.; Zhuo, R.-X. Synthesis and applications of shell cross-linked thermoresponsive hybrid micelles based on poly-(N-isopropylacrylamide-co-3-(trimethoxysilyl)-propyl-methacrylate)-b-poly-(methyl methacrylate). Langmuir 2008, 24, 4564–4570. [Google Scholar] [CrossRef]
- Loh, X.J.; Wu, Y.L.; Seow, W.T.J.; Norimzan, M.N.I.; Zhang, Z.X.; Xu, F.J.; Kang, E.T.; Neoh, K.G.; Li, J. Micellization and phase transition behavior of thermosensitive poly-(N-isopropylacrylamide)-poly(ε-caprolactone)-poly-(N-isopropylacrylamide) triblock copolymers. Polymer 2008, 49, 5084–5094. [Google Scholar] [CrossRef]
- Gil, E.S.; Hudson, S.M. Stimuli-responsive polymers and their bioconjugates. Prog. Polym. Sci. 2004, 29, 1173–1222. [Google Scholar] [CrossRef]
- Taghizadeh, B.; Taranejoo, S.; Monemian, S.A.; Moghaddam, Z.S.; Daliri, K.; Derakhshankhah, H.; Derakhshani, Z. Classification of stimuli–responsive polymers as anticancer drug delivery systems. Drug Deliv. 2015, 22, 145–155. [Google Scholar] [CrossRef]
- Oerlemans, C.; Bult, W.; Bos, M.; Storm, G.; Frank, J.; Nijsen, W.; Hennink, W.E. Polymeric micelles in anticancer terapy: Targeting, imaging and triggered release. Pharm. Res. 2010, 27, 2569–2589. [Google Scholar] [CrossRef]
- Xin, Y.; Yin, M.; Zhao, L.; Meng, F.; Luo, L. Recent progress on nanoparticle-based drug delivery systems for cancer therapy. Cancer Biol. Med. 2017, 14, 228–241. [Google Scholar] [CrossRef]
- Kohori, F.; Sakai, K.; Aoyagi, T.; Yokoyama, M.; Yamato, M.; Sakurai, T. Control of adriamycin cytotoxic activity using thermally responsive polymeric micelles composed of poly(N-isopropylacrylamide- co-N,N-dimethylacrylamide)-b-poly(d,l-lactide). Colloids Surf. B Biointerfaces 1999, 16, 195–205. [Google Scholar] [CrossRef]
- Liu, S.Q.; Tong, Y.W.; Yang, Y.Y. Thermally sensitive micelles self-assembled from poly(N-isopropylacrylamide-co-N,N-dimethylacrylamide)-b-poly(d,l-lactide-co-glycolide) for controlled delivery of paclitaxel. Mol. BioSyst. 2005, 1, 158–165. [Google Scholar] [CrossRef]
- Liu, B.; Yang, M.; Li, R.; Ding, Y.; Qian, X.; Yu, L.; Jiang, X. The antitumor effect of novel docetaxel-loaded thermosensitive micelles. Eur. J. Pharm. Biopharm. 2008, 69, 527–534. [Google Scholar] [CrossRef]
- Qu, T.; Wang, A.; Yuan, J.; Shi, J.; Gao, Q. Preparation and characterization of thermo-responsive amphiphilic triblock copolymer and its self-assembled micelle for controlled drug release. Colloids Surf. B: Biointerfaces 2009, 72, 94–100. [Google Scholar] [CrossRef]
- Nakayama, M.; Okano, T.; Miyazaki, T.; Kohori, F.; Sakai, K.; Yokoyama, M. Molecular design of biodegradable polymeric micelles for temperature-responsive drug release. J. Control. Release 2006, 115, 46–56. [Google Scholar] [CrossRef]
- Wei, H.; Cheng, S.X.; Zhang, X.Z.; Zhuo, R.X. Thermo-sensitive polymeric micelles based on poly(N-isopropylacrylamide) as drug carriers. Prog. Polym. Sci. 2009, 34, 893–910. [Google Scholar] [CrossRef]
- Molina, M.; Giulbudagian, M.; Calderón, M. Positively charged thermoresponsive nanogels for anticancer drug delivery. Macromol. Chem. Phys. 2014, 215, 2414–2419. [Google Scholar] [CrossRef]
- Watkins, K.A.; Chen, R. pH-responsive, lysine-based hydrogels for the oral delivery of a wide size range of molecules. Int. J. Pharm. 2015, 478, 496–503. [Google Scholar] [CrossRef]
- Reyes-Ortega, F. 3-pH-responsive polymers: Properties, synthesis and applications. In Smart Polymers and Their Applications, 1st ed.; Aguilar De Armas, M.R., Román, J.S., Eds.; Woodhead Publishing in Materials: Cambridge, UK, 2014; pp. 45–92. ISBN 9780857096951. [Google Scholar]
- Roy, S.G.; De, P. pH responsive polymers with amino acids in the side chains and their potential applications. J. Appl. Polym. Sci. 2014, 131, 41084–41096. [Google Scholar] [CrossRef]
- Schilli, Ch.M.; Zhang, M.; Rizzardo, E.; Thang, S.H.; Chong, Y.; Edwards, K.; Karlsson, G.; Müller, A.H.E. A new double- responsive block copolymer synthesized via RAFT. polymerization: Poly(N-isopropylacrylamide)-block-poly(acrylic acid). Macromolecules 2004, 37, 7861–7866. [Google Scholar] [CrossRef]
- Kang, S.I.; Bae, Y.H. pH-Induced solubility transition of sulfonamide-based polymers. J. Controlled Release 2002, 80, 145–155. [Google Scholar] [CrossRef]
- Zhu, L.; Smith, P.P.; Boyes, S.G. pH-responsive polymers for imaging acidic biological environments in tumors. J. Polym. Sci. Part. B: Polym. Phys. 2013, 51, 1062–1067. [Google Scholar] [CrossRef]
- Musiał, W.; Michálek, J. The influence of low process temperature on the hydrodynamic radius of polyNIPAM-co-PEG thermosensitive nanoparticles presumed as drug carriers for bioactive proteins. Acta Pol. Pharm. 2015, 72, 161–169. [Google Scholar]
- Gola, A.; Knysak, T.; Musiał, W. The Influence of Anionic Initiator on the Selected Properties of Poly-N-Isopropyl Acrylamide Evaluated for Controlled Drug Delivery. Molecules 2017, 22, 23. [Google Scholar] [CrossRef]
- Kabiri, K.; Zohuriaan-Mehr, M.J.; Mirzadeh, H.; Kheirabadi, M. Solvent- ion- and pH-specific swelling of poly(2-acrylamido-2- methylpropane sulfonic acid) superabsorbing gels. J. Polym. Res. 2010, 17, 203–212. [Google Scholar] [CrossRef]
- Gospodinova, N.; Tomšík, E.; Omelchenko, O. How strong are strong poly(sulfonic acids)? An example of the poly(2-acrylamido-2-methyl-1-propanesulfonic acid). Europ. Polym. J. 2016, 74, 130–135. [Google Scholar] [CrossRef]
- Yoshida, H.; Furukawa, Y.; Tasumi, M. Structural studies and vibrational analyses of stable and less stable conformers of 1,3,5-hexatriene based on ab initio MO calculations. J. Mol. Struct. 1989, 194, 279–299. [Google Scholar] [CrossRef]
- Scherer, J.R.; Potts, W.J. Normal Coordinates for the Out-of-Plane Deformations of Vinyl Bromide. J. Chem. Phys. 1959, 30, 1527–1529. [Google Scholar] [CrossRef]
- Coates, J. Interpretation of infrared spectra, a practical approach. In Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; John Wiley & Sons Ltd.: Chichester, UK, 2000; pp. 10815–10837. ISBN 0471976709. [Google Scholar]
- Mishra, P.K.; Ekielski, A. The self-assembly of lignin and its application in nanoparticle synthesis: A Short Review. Nanomaterials 2019, 9, 243. [Google Scholar] [CrossRef]
- Mishra, P.K.; Wimmer, R. Aerosol assisted self-assembly as a route to synthesize solid and hollow spherical lignin colloids and its utilization in layer by layer deposition. UltrasonicsSonochemistry 2017, 35, 45–50. [Google Scholar] [CrossRef]
- Matthews, B.A.; Rhodes, C.T. Use of the Derjaguin, Landau, Verwey and Overbeek theory to interpret pharmaceutical suspension stability. J. Pharm. Sci. 1970, 59, 521–525. [Google Scholar] [CrossRef]
- Pelton, R. Temperature-sensitive aqueous microgels. Adv. Colloid Interface Sci. 2000, 85, 1–33. [Google Scholar] [CrossRef]
- McPhee, W.; Tam, K.C.; Pelton, R. Poly(N-isopropylacrylamide) latices prepared with sodium dodecyl sulfate. J. Colloid Interface Sci. 1993, 156, 24–30. [Google Scholar] [CrossRef]
- Kratz, K.; Eimer, W. Swelling properties of colloidal poly(N-Isopropylacrylamide) microgels in solution. Ber. Bunsenges. Phys. Chem. 1998, 102, 848–854. [Google Scholar] [CrossRef]
- Oh, K.S.; Bae, Y.C. Swelling behavior of submicron gel particles. J. Appl. Polym. Sci. 1998, 69, 109–114. [Google Scholar] [CrossRef]
- Ito, K. Competition between thermal decomposition and hydrolysis of 2,2′-azobis (2-amidinopropane) in aqueous solution. J. Polym. Sci. Polym. Chem. 1973, 11, 1673–1681. [Google Scholar] [CrossRef]
- Musiał, W.; Kokol, V.; Fecko, T.; Voncina, B. Morphological patterns of poly(N-isopropylacrylamide) derivatives synthesized with EGDMA, DEGDMA, and TEGDMA crosslinkers for application as thermosensitive drug carriers. Chem. Papers 2010, 64, 791–798. [Google Scholar] [CrossRef]
- Available online: https://www.iso.org/standard/65410.html (accessed on 23 March 2019).
Sample Availability: Samples of the compounds PNA, PAMPSA, PNAM, PAMPSAM are available from the authors. |



| Type of Polymer Microparticle System | Monomer [mol] | Cationic Initiator [mol] | Cross-Linker [mol] | |
|---|---|---|---|---|
| NIPA | AMPS | AMP | MBA | |
| PNA | 4.433 × 10−2 | - | 2.238 × 10−3 | - |
| PAMPSA | - | 2.453 × 10−2 | 1.232 × 10−3 | - |
| PNAM | 4.429 × 10−2 | - | 2.224 × 10−3 | 3.256 × 10−3 |
| PAMPSAM | - | 2.416 × 10−2 | 1.239 × 10−3 | 3.282 × 10−3 |
| Type of Polymer Microparticles System | Zeta Potential [mV] | pH | |||
|---|---|---|---|---|---|
| Measurement Temperatures [°C] | |||||
| 18 | 25 | 32 | 45 | 25 | |
| PNA | −1.36 ± 0.87 | −0.34 ± 0.83 | 1.65 ± 0.75 | 47.32 ± 1.88 | 7.39 |
| PAMPSA | −0.01 ± 0.37 | 0.15 ± 0.22 | −0.23 ± 0.05 | 0.06 ± 0.34 | 4.42 |
| PNAM | 8.90 ± 0.11 | 10.00 ± 0.33 | 11.66 ± 0.82 | 30.16 ± 1.54 | 7.50 |
| PAMPSAM | −0.09 ± 0.11 | −0.13 ± 0.01 | −0.13 ± 0.32 | −0.04 ± 0.11 | 3.99 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gola, A.; Sacharczuk, M.; Musiał, W. Synthesis of AMPSA Polymeric Derivatives Monitored by Electrical Conductivity and Evaluation of Thermosensitive Properties of Resulting Microspheres. Molecules 2019, 24, 1164. https://doi.org/10.3390/molecules24061164
Gola A, Sacharczuk M, Musiał W. Synthesis of AMPSA Polymeric Derivatives Monitored by Electrical Conductivity and Evaluation of Thermosensitive Properties of Resulting Microspheres. Molecules. 2019; 24(6):1164. https://doi.org/10.3390/molecules24061164
Chicago/Turabian StyleGola, Agnieszka, Magdalena Sacharczuk, and Witold Musiał. 2019. "Synthesis of AMPSA Polymeric Derivatives Monitored by Electrical Conductivity and Evaluation of Thermosensitive Properties of Resulting Microspheres" Molecules 24, no. 6: 1164. https://doi.org/10.3390/molecules24061164
APA StyleGola, A., Sacharczuk, M., & Musiał, W. (2019). Synthesis of AMPSA Polymeric Derivatives Monitored by Electrical Conductivity and Evaluation of Thermosensitive Properties of Resulting Microspheres. Molecules, 24(6), 1164. https://doi.org/10.3390/molecules24061164