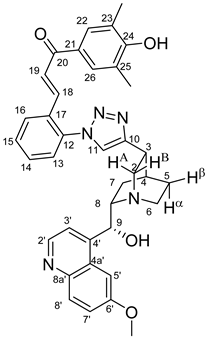
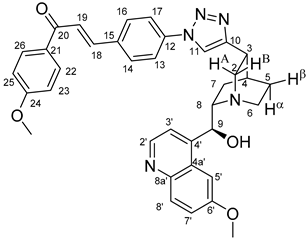
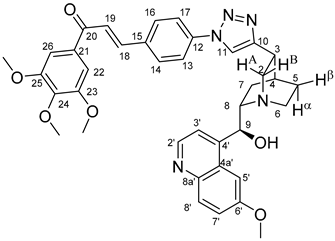
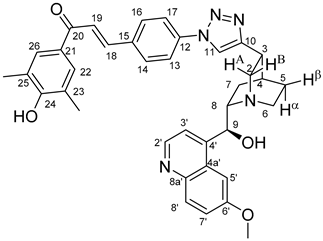
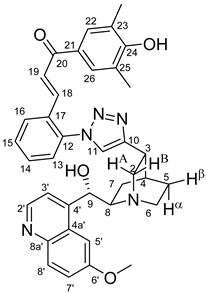
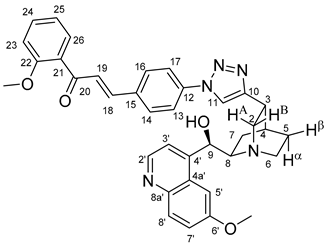
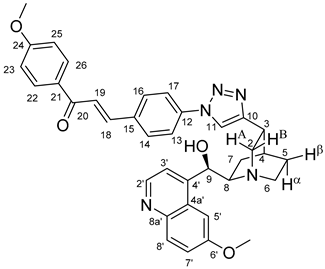
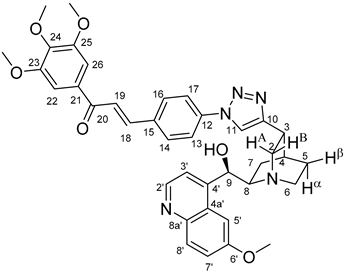
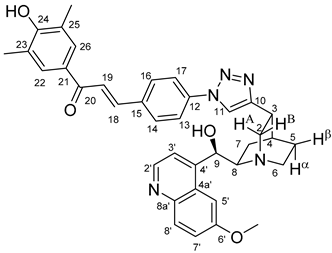
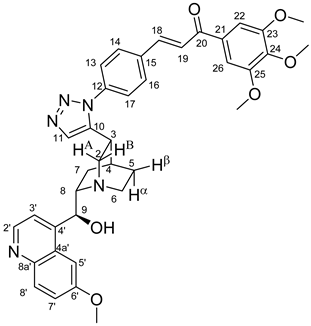
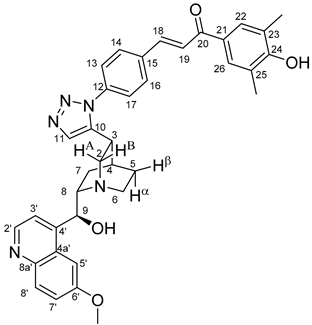
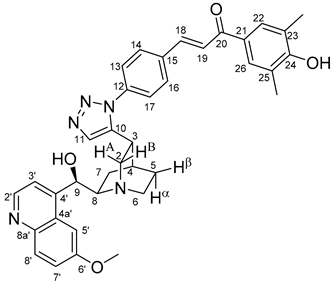
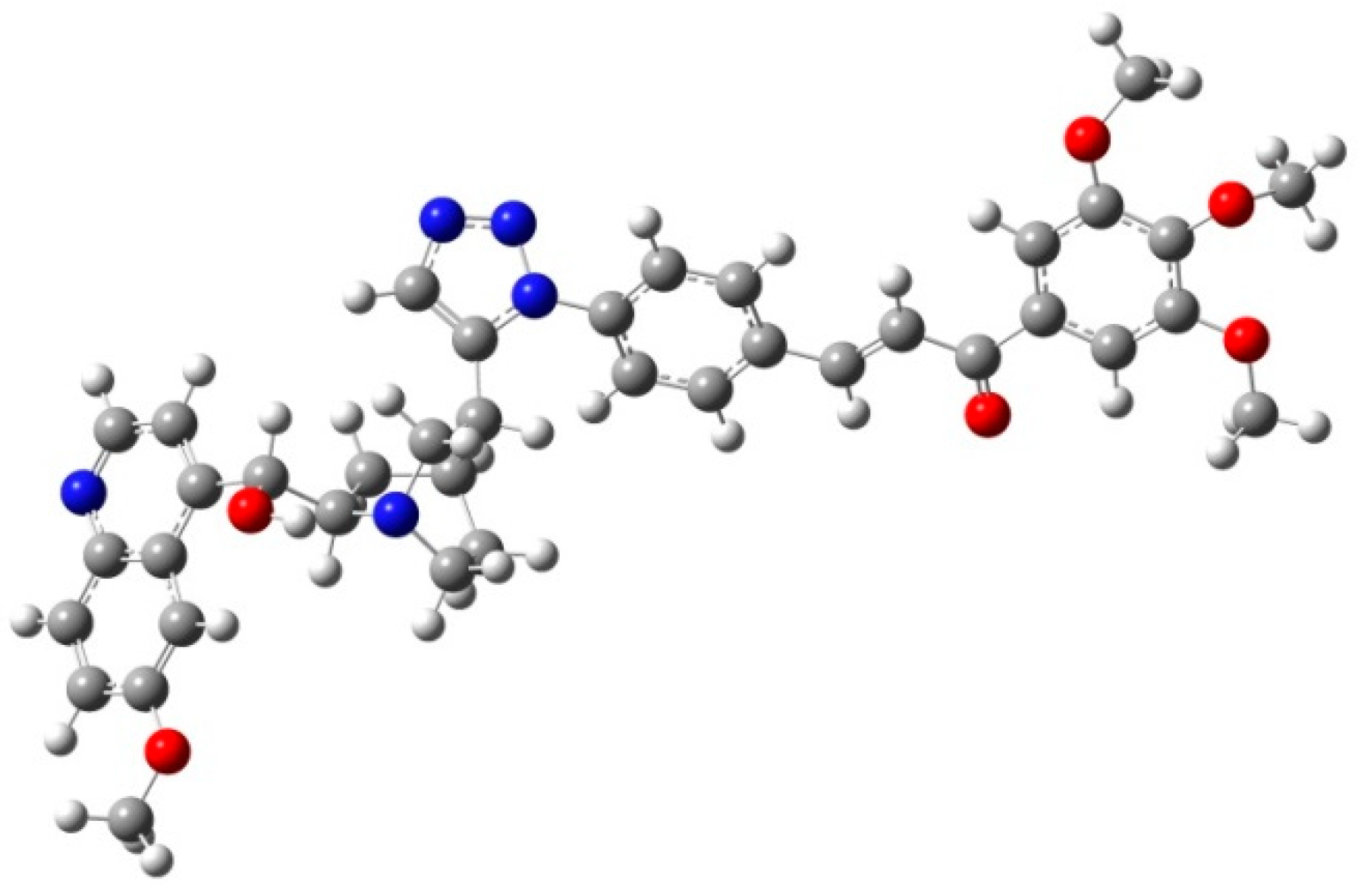
All fine chemicals were obtained from commercially available sources (Merck, Fluorochem, Molar Chemicals, VWR) and used without further purification. Dioxane was distilled from sodium benzophenone. Merck Kieselgel (230–400 mesh, 60 Å) was used for flash column chromatography. Melting points (uncorrected) were determined with a Büchi M-560. The 1H- and 13C NMR spectra of all compounds were recorded in CDCl3 or DMSO-d6 solution in 5 mm tubes at RT, on a Bruker DRX-500 spectrometer at 500 (1H) and 125 (13C) MHz, with the deuterium signal of the solvent as the lock and TMS as the internal standard. The HSQC, HMBC, COSY and NOESY spectra, which support the exact assignments the of 1H- and 13C NMR signals, were obtained by using the standard Bruker pulse programs. For each compound characterized in this session the numbering of atoms used for assignment of 1H- and 13C NMR signals do not correspond to IUPAC rules reflected from the given systematic names. The exact mass measurements were performed using a Q-TOF Premier mass spectrometer (Waters Corporation, 34 Maple St, Milford, MA, USA) in positive electrospray mode.
3.4. General Synthesis of Cinchona-Chalcone Hybrids with 1,5-Disubstituted Triazole Linkers
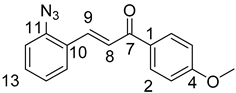
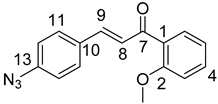
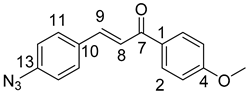
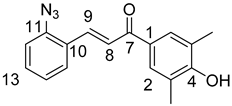
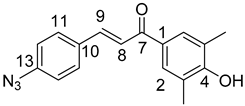
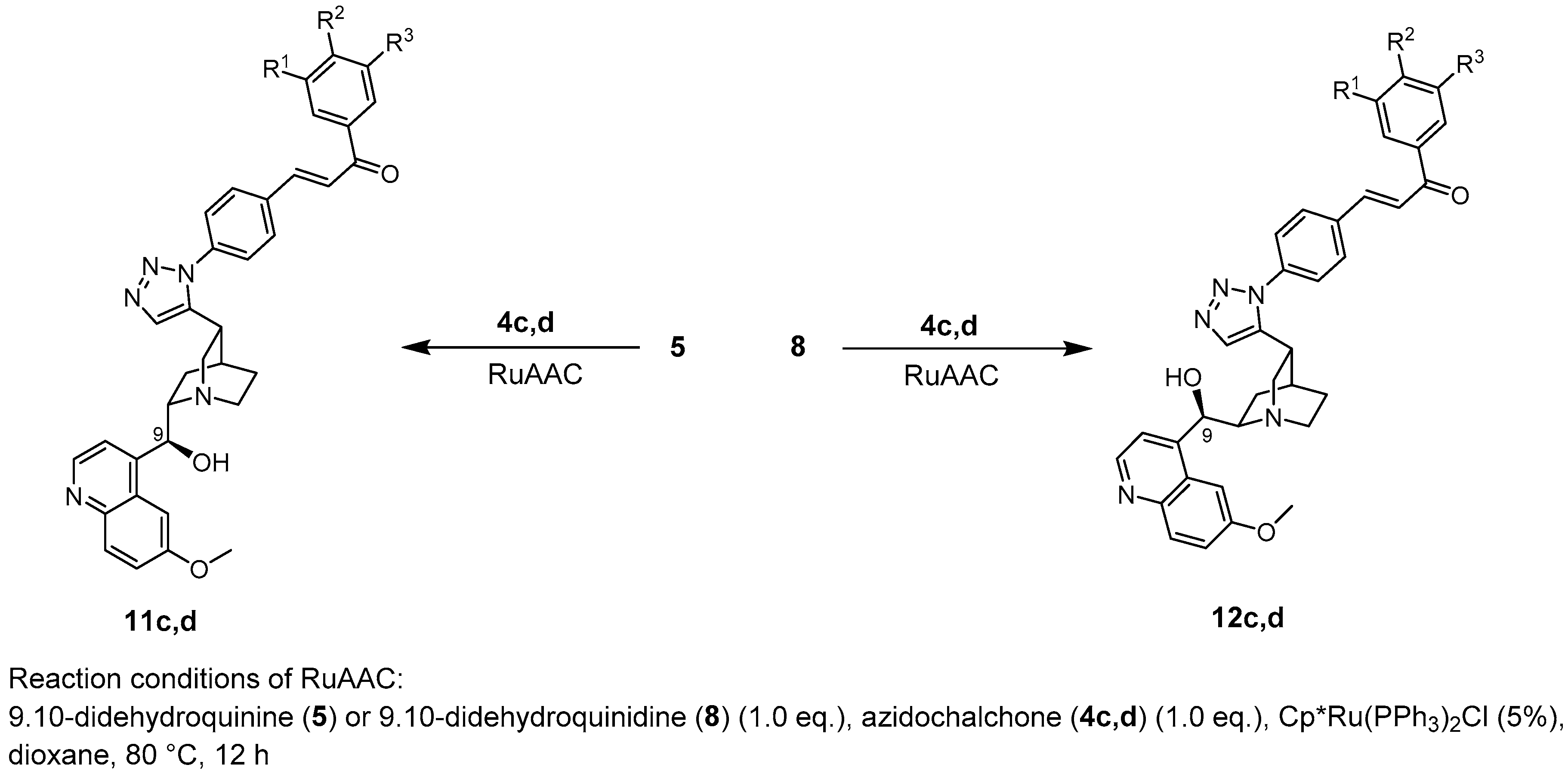
10,11-Didehydroquinine (5) or 10,11-didehydroquinidine (8) (322 mg, 1.0 mmol, 1.0 eq.), azidochalchone (2c,d, 4c,d) (1.0 mmol, 1.0 eq.) and pentamethylcyclopentadienylbis(triphenylphosphine)ruthenium(II) chloride (40 mg, 0,05 mmol, 5%) were dissolved in dioxane (4 mL). The resulting mixture was stirred at 80 °C for 12 h diluted with water (15 mL) and extracted with DCM (5 × 25 mL). The combined organic phase was washed with brine, dried on Na2SO4, and evaporated to dryness. The residue was purified by flash chromatography on silica gel, using DCM:MeOH mixtures (the ratio of DCM and MeOH was varied between 60:1 and 10:1). The analytical sample was sequentially crystallized by EtOH-water and Et2O.
Spectral characterization (
1H- and
13C-NMR data and HRMS) of the novel compounds can be found in the
Supplementary Materials.
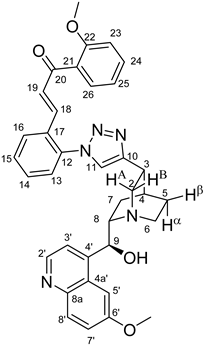
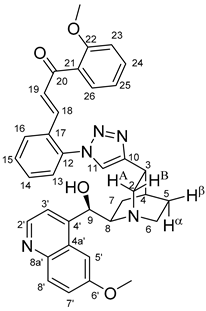
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(2-methoxyphenyl)prop-2-en-1-one (
6a)
Pale yellow solid; Yield: 578 mg (91%); MP.: ~121 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.68 (d,
J = 4.5 Hz, 1H, H2′), 8.26 (s, 1H, H11), 7.98 (dd,
J = 7.5 Hz, 1.5 Hz, 1H, H16), 7.90 (d,
J = 9.2 Hz, 1H, H8′), 7.66 (d,
J = 2.3 Hz, 1H, H5′), 7.55–7.59 (m, 3H, H3′ and H14 and H15), 7.41 (td,
J = 8.4 Hz, 1.7 Hz, 1H, H24), 7.38 (dd,
J = 7.5 Hz, 1.3 Hz, 1H, H13), 7.36 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 7.29 (dd,
J = 7.5 Hz, 1.7 Hz, 1H, H26), 7.17 (d,
J = 15.9 Hz, 1H, H19), 6.98 (d,
J = 8.4 Hz, 1H, H23), 1H, H25), 6.92 (td,
J = 7.5 Hz, 0.8 Hz, 6.88 (d,
J = 15.9 Hz, 1H, H18), 6.10 (br s, 1H, C9O
H), 5.96 (br m, 1H, H9), 3.97 (s, 3H, C6′OCH
3), 3.70 (s, 3H, C22OCH
3), 3.62 (br m, H8), 3.57 (br m, 1H, H2A), 3.39 (br m, 1H, H2B), 3.38 (br m, 1H, H3), 3.82 (br m, 1H, H6α), 3.06 (br m, 1H, H6β), 2.16 (br m, 1H, H4), 2.01 (br m, 1H, H5α), 1.86 (br m, 1H, H5β), 1.94 (br m, 1H, H7α), 1.39 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 192.7 (C20), 158.2 (C6′), 158.1 (C22), 147.0 (C4′), 148.9 (C10), 147.9 (C2′), 144.5 (C8a′), 137.2 (C18), 136.7 (C12), 133.5 (C21), 133.4 (C24), 131.8 (C8′), 131.4 (C14), 130.8 (C17), 130.7 (C15), 130.3 (C19), 128.9 (C16), 127.7 (C26), 127.1 (C13), 126.7 (C4a′), 125.1 (C11), 121.9 (C7′), 120.9 (C25), 119.6 (C3′), 112.8 (C23), 102.9 (C5′), 68.0 (C9), 60.3 (C8), 57.0 (C6′O
CH
3), 56.5 (C22O
CH
3), 54.5 (C2), 43.2 (C6), 31.9 (C3) 27.9 (C4), 25.3 (C5), 25.2 (C7); HRMS exact mass calcd. for C
36H
36N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27545.
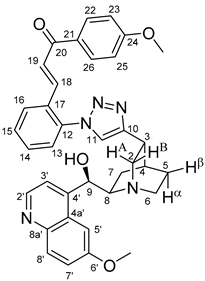
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-trimethoxyphenyl)prop-2-en-1-one (
6b)
Pale yellow solid; Yield: 433 mg (72%); MP.: ~151 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.62 (d,
J = 4.5 Hz, 1H, H2′), 8.29 (s, 1H, H11), 8.20 (dd,
J = 7.6 Hz, 1.5 Hz, 1H, H16), 7.99 (d,
J = 8.9 Hz, 2H, H22 and H26), 7.88 (d,
J = 9.1 Hz, 1H, H8′), 7.68 (d,
J = 15.6 Hz, 1H, H19), 7.61 (td,
J = 7.4 Hz, 1.3 Hz, 1H, H15), 7.59 (td,
J = 7.5 Hz, 1.6 Hz, 1H, H14), 7.56 (d,
J = 2.6 Hz, 1H, H5′), 7.49 (d,
J = 4.5 Hz, 1H, H3′), 7.45 (dd,
J = 7.5 Hz, 1.5 Hz, 1H, H13), 7.33 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 7.21 (d,
J = 15.6 Hz, 1H, H18), 7.01 (d,
J = 8.9 Hz, 2H, H23 and H25), 5.59 (br m, 1H, C9O
H), 5.41 (br s, 1H, C9), 3.89 (s, 3H, C6′OCH
3), 3.83 (s, 3H, C24OCH
3), 3.42 (br m, 1H, H8), 3.36 (br m, 1H, H2A), 3.24 (br m, 2H, H6α and H6β), 3.14 (br m, 1H, H3), 2.70 (br m, 1H, H2B), 2.14 (br m, 1H, H4), 1.86 (br m, 1H, H5α), 1.72 (br m, 1H, H7α), 1.68 (br m, 1H, H5β), 1.63 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 187.8 (C20), 163.9 (C24), 157.6 (C6′), 150.7 (C10), 148.6 (C4′), 147.9 (C2′), 144.5 (C8a′), 137.3 (C18), 137.0 (C12), 131.7 (C8′), 131.4 (C14 and C22 and C26), 131.1 (C17), 130.7 (C21), 130.5 (C15), 128.7 (C16), 127.4 (C4a′), 127.0 (C13), 125.8 (C19), 124.9 (C11), 121.4 (C7′), 119.7 (C3′), 114.6 (C23 and C25), 103.3 (C5′), 70.7 (C9), 60.8 (C8), 56.2 (C6′O
CH
3), 56.1 (C24O
CH
3), 55.7 (C2), 42.7 (C6), 33.0 (C3), 28.1 (C4), 27.1 (C7 and C5); HRMS exact mass calcd. for C
36H
36N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27608.
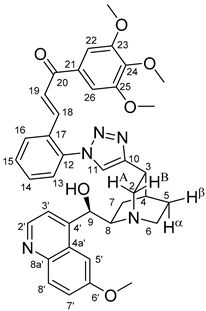
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one (
6c)
Yellow solid; Yield: 503 mg (74%); MP.: ~157 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.62 (d,
J = 4.5 Hz, 1H, H2′), 8.30 (s, 1H, H11), 8.22 (dd,
J = 7.6 Hz, 1.4 Hz, 1H, H16), 7.88 (d,
J = 9.2 Hz, 1H, H8′), 7.71 (d,
J = 15.6 Hz, 1H, H19), 7.62 (td,
J = 7.3 Hz, 1.1 Hz, 1H, H15, overlapped by H5′ and H14), 7.59 (td,
J = 7.6 Hz, 1.3 Hz, 1H, H14, overlapped by H5′ and H15), 7.59 (1H, H5′, overlapped by H14 and H15), 7.50 (d,
J = 4.3 Hz, 1H, H3′), 7.44 (d,
J = 7.4 Hz, 1H, H13), 7.33 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 7.28 (s, 2H, H22 and H26), 7.23 (d,
J = 15.6 Hz, 1H, H18), 5.78 (br m, 1H, C9O
H, overlapped by H9), 5.67 (br s, 1H, H9, overlapped by C9OH), 3.92 (s, 3H, C6′OCH
3), 3.84 (s, 6H, C23OCH
3 and C25OCH
3), 3.76 (s, 3H, C24OCH
3), 3.57 (br m, 1H, H6α), 3.50 (br m, 1H, H8), 3.30-3.41 (br m, 2H, H2A and H2B), 3.28 (br m, 1H, H3), 2.86 (br m, 1H, H6β), 2.16 (br m, 1H, H4), 1.91 (br m, 1H, H5α), 1.86 (br m, 1H, H7α), 1.77 (br m, 1H, H5β), 1.57 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 188.3 (C20), 157.7 (C6′), 153.5 (C23 and C25), 153.1 (C10), 150.2 (C4′), 147.9 (C2′), 144.5 (C8a′), 143.3 (C24), 137.8 (C18), 137.0 (C12), 133.1 (C21), 131.7 (C8′), 131.5 (C14), 130.9 (C17), 130.5 (C15), 128.8 (C16), 127.1 (C4a′), 127.0 (C13), 125.7 (C19), 124.9 (C11), 121.5 (C7′), 119.6 (C3′), 107.3 (C22 and C26), 103.2 (C5′), 70.3 (C9), 60.7 (C24OCH
3), 60.6 (C8), 56.9 (C23OCH
3 and C25OCH
3), 56.4 (C6′O
CH
3), 55.1 (C2), 42.9 (C6), 32.5 (C3), 28.3 (C4), 26.5 (C5), 22.4 (C7); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 662.29731, found
m/
z: 662.29623.
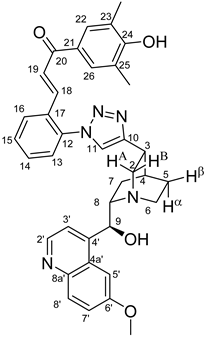
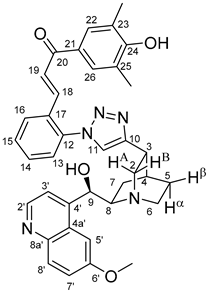
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
6d)
Orange solid; Yield: 184 mg (30%); MP.: ~179 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 9.01 (br s, 1H, C24O
H), 8.63 (d,
J = 4.5 Hz, 1H, H2′), 8.27 (s, 1H, H11), 8.18 (dd,
J = 7.7 Hz, 1.5 Hz, 1H, H16), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.65 (s, 2H, H22 and H26, overlapped by H19), 7.65 (d,
J = 15.6 Hz, 1H, H19, overlapped by H22 and H26), 7.60 (td,
J = 7.5 Hz, 1.2 Hz, 1H, H15), 7.56–7.58 (m, 2H, H5′ and H14), 7.50 (d,
J = 4.5 Hz, 1H, H3′), 7.42 (dd,
J = 7.6 Hz, 1.2 Hz, 1H, H13), 7.34 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 7.18 (d,
J = 15.6 Hz, 1H, H18), 5.69 (br m, 1H, C9O
H), 5.52 (br s, 1H, C9), 3.90 (s, 3H, C6′OCH
3), 3.47 (br m, 1H, H8), 3.46 (br m, 1H, H6α) 3.27–3.35 (br m, 2H, H2A and H2B), 3.22 (br m, 1H, H3), 2.79 (br m, 1H, H6β), 2.21 (s, 6H, C
H3), 2.16 (br m, 1H, H4), 1.87 (br m, 1H, H5α), 1.80 (br m, 1H, H7α), 1.72 (br m, 1H, H5β), 1.61 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 187.8 (C20), 158.9 (C24), 157.7 (C6′), 150.3 (C10), 148.3 (C4′), 147.9 (C2′), 144.5 (C8a′), 136.8 (C12), 136.5 (C18), 131.7 (C8′), 131.2 (C14 and C17), 130.5 (C15), 130.0 (C22 and C26), 129.3 (C21), 128.6 (C16), 127.2 (C4a′), 127.0 (C13), 126.1 (C19), 124.9 (C11), 124.8 (C23 and C25), 121.4 (C7′), 119.6 (C3′), 103.3 (C5′), 70.1 (C9) 60.6 (10), 56.4 (C6′O
CH
3), 55.3 (C2), 42.8 (C6), 32.7 (C3), 28.3 (C4), 26.8 (C5), 22.8 (C7), 16.8 (
CH
3); HRMS exact mass calcd. for C
37H
38N
5O
4 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29167.
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
S)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
6*d)
Yellow solid; Yield: 308 mg (50%); MP.: ~171 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 9.03 (br s, 1H, C24O
H), 8.66 (d,
J = 4.5 Hz, 1H, H2′), 8.34 (s, 1H, H11), 8.15 (dd,
J = 7.7 Hz, 1.5 Hz, 1H, H16), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.70 (d,
J = 2.6 Hz, 1H, H5′), 7.63 (s, 2H, H22 and H26, overlapped by H19), 7.63 (d,
J = 15.6 Hz, 1H, H19, overlapped by H22 and H26), 7.58 (td,
J = 7.6 Hz, 1.1 Hz, 1H, H15, overlapped by H3′), 7.52 (td,
J = 7.7 Hz, 1.3 Hz, 1H, H14), 7.50 (d,
J = 4.5 Hz, 1H, H3′, overlapped by H15), 7.34–7.36 (2H, H7′ and H13), 7.07 (d,
J = 15.6 Hz, 1H, H18), 6.34 (br m, 1H, C9O
H), 6.26 (br s, 1H, C9), 4.06 (br m, 1H, H6α), 4.02 (s, 3H, C6′OCH
3), 3.61–3.76 (br m, 4H, H8 and H2A and H2B and H3), 3.28 (br m, 1H, H6β), 2.30 (br m, 1H, H4), 2.22 (s, 6H, C
H3), 2.09–2.12 (br m, 2H, H7α and H5α), 2.00 (t,
J = 11.1 Hz, 1H, H5β), 1.37 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 187.7 (C20), 158.9 (C24), 158.5 (C6′), 148.1 (C10), 147.8 (C2′), 145.8 (C4′), 144.4 (C8a′), 136.6 (C12), 136.2 (C18), 131.8 (C8′), 131.2 (C17), 131.1 (C14), 130.6 (C15), 130.0 (C22 and C26), 129.2 (C21), 128.6 (C16), 127.0 (C13), 126.3 (C19), 126.2 (C4a′), 125.5 (C11), 124.8 (C23 and C25), 122.2 (C7′), 119.6 (C3′), 102.7 (C5′), 66.5 (C9) 60.1 (C8), 57.4 (C6′O
CH
3), 53.7 (C2), 43.5 (C6), 31.3 (C3), 27.7 (C4), 24.3 (C5), 18.7 (C7), 16.9 (
CH
3); HRMS exact mass calcd. for C
37H
38N
5O
4 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29091.
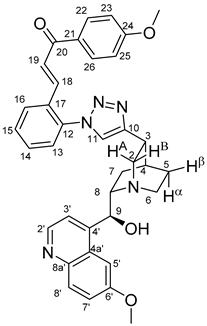
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(2-methoxyphenyl)prop-2-en-1-one (
7a)
Pale yellow solid; Yield: 337mg (56%); MP.: ~170 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.64 (d,
J = 4.4 Hz, 1H, H2′), 8.62 (s, 1H, H11), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.83–7.85 (m, 4H, H13 and H14 and H16 and H17), 7.59 (d,
J = 2.6 Hz, 1H, H5′), 7.46–7.53 (m, 4H, H3′ and H18 and H24 and H26), 7.39 (d,
J = 16.0 Hz, 1H, H19), 7.36 (dd,
J = 9.2 Hz, 2.6 Hz, 1H, H7′), 7.15 (d,
J = 8.3 Hz, 1H, H23), 7.03 (td,
J = 7.5 Hz, 0.8 Hz, 1H, H25), 5.83-5.71 (br s, 2H, C9O
H and H9) 3.94 (s, 3H, C6′OCH
3), 3.83 (s, 3H, C22OCH
3), 3.45–3.48 (br m, 4H, H8 and H2A and H2B and H6α), 3.28 (br m, 1H, H3), 2.89 (br m, 1H, H6β), 2.22 (br m, 1H, H4), 1.92 (br m, 1H, H5α), 1.85 (br m, 1H, H7α), 1.78 (br m, 1H, H5β), 1.51 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 192.5 (C20), 158.4 (C22), 157.9 (C6′), 150.1 (C10), 147.9 (C2′), 145.8 (C4′), 144.5 (C8a′), 141.4 (C18), 138.1 (C12), 135.2 (C15), 133.5 (C24), 131.7 (C8′), 130.3 (C14 and C16), 129.9 (C26), 129.4 (C21), 128.5 (C19), 127.0 (C4a′), 121.7 (C7′), 121.1 (C25), 120.8 (C11), 120.6 (C13 and C17), 119.6 (C3′), 113.1 (C23), 103.1 (C5′), 68.2 (C9), 56.6 (C6′O
CH
3), 56.5 (C22OCH
3), 60.4 (C8), 54.9 (C2), 43.0 (C6), 32.5 (C3), 27.5 (C4), 27.0 (C7 and C5); HRMS exact mass calcd. for C
36H
37N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27570.
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4methoxyphenyl)prop-2-en-1-one (
7b)
Pale yellow solid; Yield: 475 mg (79%); MP.: ~154 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.64 (d,
J = 4.5 Hz, 1H, H2′), 8.62 (s, 1H, H11), 8.11 (d,
J = 8.9 Hz, 2H, H22 and H26), 7.99 (d,
J = 8.7 Hz, 2H, H16 and H14), 7.88–7.90 (m, 4H, H8′ and H13 and H17 and H19), 7.68 (d,
J = 15.5 Hz, 1H, H18), 7.57 (d,
J = 2.6 Hz, 1H, H5′), 7.49 (d,
J = 4.5 Hz, 1H, H3′), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 7.05 (d,
J = 8.9 Hz, 2H, H23 and H25), 5.67 (br m, 1H, C9O
H), 5.50 (br s, 1H, C9), 3.91 (s, 3H, C6′OCH
3), 3.84 (s, 3H, C24OCH
3), 3.45 (br m, 1H, H6α), 3.39 (br m, 1H, H8), 3.27 (br m, 2H, H2A and H2B), 3.15 (br m, 1H, H3), 2.75 (br m, 1H, H6β), 2.19 (br m, 1H, H4), 1.86 (br m, 1H, H5α), 1.76 (br m, 1H, H7α), 1.70 (br m, 1H, H5β), 1.58 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 188.0 (C20), 163.9 (C24), 157.7 (C6′), 151.3 (C10), 148.4 (C4′), 147.9 (C2′), 144.5 (C8a′), 142.0 (C18), 138.2 (C12), 135.4 (C15), 131.7 (C8′), 131.4 (C23 and C25), 131.0 (C21), 130.5 (C14 and C16), 127.2 (C4a′), 123.8 (C19), 121.5 (C7′), 120.7 (C11), 120.5 (C13 and C17), 119.6 (C3′), 114.6 (C22 and C26), 103.3 (C5′), 70.2 (C9), 60.6 (C8), 56.4 (C6′O
CH
3), 56.1 (C24OCH
3), 55.3 (C2), 42.7 (C6), 32.9 (C3), 27.7 (C4), 27.0 (C7 and C5); HRMS exact mass calcd. for C
36H
36N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27553.
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one (
7c)
Yellow solid; Yield: 404 mg (61%); MP.: ~150 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.64 (d,
J = 4.5 Hz, 1H, H2′), 8.63 (s, 1H, H11), 8.02 (d,
J = 8.6 Hz, 2H, H16 and H14), 7.87–7.90 (m, 4H, H8′ and H13 and H17 and H19), 7.72 (d,
J = 15.5 Hz, 1H, H18), 7.57 (d,
J = 2.5 Hz, 1H, H5′), 7.50 (d,
J = 4.5 Hz, 1H, H3′), 7.39 (s, 2H, H22 and H26), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 5.77–5.44 (br s, 2H, C9O
H, H9), 3.92 (s, 3H, C6′OCH
3), 3.87 (s, 6H, C23OCH
3 and C25OCH
3), 3.76 (s, 3H, C24OCH
3), 3.40 (br m, 1H, H8), 3.32-3.25 (br m, 2H, H2A and H2B), 3.24 (br m, 1H, H3), 3.07 (br m, 1H, H6α, overlapped by HDO from solvent), 2.88 (br m, 1H, H6β), 2.20 (br m, 1H, H4), 1.88 (br m, 1H, H7α), 1.79 (br m, 1H, H5α), 1.73 (br m, 1H, H5β), 1.59 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 188.5 (C20), 157.7 (C6′), 153.5 (C23 and C25), 150.7 (C10), 149.8 (C4′), 147.9 (C2′), 144.5 (C8a′), 143.2 (C24), 142.5 (C18), 138.2 (C12), 135.4 (C15), 133.4 (C21), 131.7 (C8′), 130.7 (C14 and C16), 127.1 (C4a′), 123.7 (C19), 121.5 (C7′), 120.7 (C11), 120.5 (C13 and C17), 119.6 (C3′), 107.4 (C22 and C26), 103.2 (C5′), 70.3 (C9), 60.7 (C24OCH
3), 60.6 (C8), 57.0 (C23OCH
3 and C25OCH
3), 56.4 (C6′O
CH
3), 55.3 (C2), 42.6 (C6), 32.8 (C3), 27.6 (C4), 26.8 (C5), 26.5 (C7); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 662.29731, found
m/
z: 662.29683.
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
7d)
Yellow solid; Yield: 129 mg (21%); MP.: ~152 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.65 (d,
J = 4.4 Hz, 1H, H2, overlapped by H11), 8.65 (s, 1H, H11, overlapped by H2′), 7.94 (d,
J = 8.7 Hz, 2H, H16 and H14), 7.88 (d,
J = 9.2 Hz, 1H, H8′), 7.84 (d,
J = 15.6 Hz, 1H, H19), 7.81 (d,
J = 8.7 Hz, 2H, H13 and H17), 7.74 (s, 2H, H22 and H26), 7.69 (d,
J = 2.6 Hz, 1H, H5′), 7.61 (d,
J = 15.6 Hz, 1H, H18), 7.55 (d,
J = 4.4 Hz, 1H, H3′), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 6.02 (br s, 1H, H9), 3.98 (s, 3H, C6′OCH
3), 3.85 (br m, 1H, H6α), 3.58 (br m, 1H, H8), 3.57 (br m, 1H, H2B), 3.48 (br m, 1H, H2A), 3.41 (br m, 1H, H3), 3.06 (br m, 1H, H6β), 2.28 (br m, 1H, H4), 2.04 (s, 6H, C
H3), 2.02 (br m, 1H, H5α), 1.98 (br m, 1H, H7α), 1.88 (br m, 1H, H5β), 1.42 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 187.9 (C20), 158.8 (C24), 158.2 (C6′), 149.7 (C10), 147.9 (C2′), 147.1 (C4′), 144.5 (C8a′), 141.2 (C18), 137.9 (C12), 135.6 (C15), 131.8 (C8′), 130.4 (C14 and C16), 130.0 (C22 and C26), 129.6 (C21), 126.7 (C4a′), 124.9 (C23 and C25), 124.2 (C19), 122.0 (C7′), 121.1 (C11), 120.6 (C13 and C17), 119.5 (C3′), 103.0 (C5′), 67.9 (C9), 60.3 (C8), 57.1 (C6′O
CH
3), 54.4 (C2), 43.2 (C6), 32.0 (C3), 27.6 (C4), 25.4 (C5), 20.4 (C7), 16.9 (
CH
3); HRMS exact mass calcd. for C
37H
38N
5O
4 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29159.
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(2-methoxyphenyl)prop-2-en-1-one (
9a)
Yellow solid; Yield: 373 mg (62%); MP.: ~138 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.72 (s, 1H, H11), 8.64 (d,
J = 4.4 Hz, 1H, H2′), 7.97 (d,
J = 8.7 Hz, 1H, H26), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.96-7.92 (m, 3H, H14 and H15 and H16), 7.49 (d,
J = 7.8 Hz, 1H, H13), 7.60 (d,
J = 2.8 Hz, 1H, H5′), 7.54 (d,
J = 16.0 Hz, 1H, H18), 7.50 (1H, H24, overlapped by H3′), 7.45 (1H, H3′, overlapped by H24), 7.44 (d,
J = 16.0 Hz, 1H, H19), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 7.17 (d,
J = 8.3 Hz, 1H, H23), 7.04 (t,
J = 7.5 Hz, 1H, H25), 5.75–5.45 (br s, 2H, H9 and C9O
H), 3.92 (s, 3H, C6′OCH
3), 3.86 (s, 3H, C22OCH
3), 3.72 (br m, 1H, H2A), 3.18 (br m, H8), 3.12 (1H, H2B, overlapped by HDO from solvent), 3.11 (1H, H3, overlapped by HDO from solvent), 2.83 (br m, 2H, H6α and H6β), 2.16 (br m, 1H, H4), 1.95 (br m, 1H, H7β), 1.68 (br m, 2H, H5α and H5β), 1.45 (br m, 1H, H7α);
13C-NMR (DMSO-
d6 at 45 °C): 192.5 (C20), 158.4 (C22), 157.6 (C6′), 150.9 (C10), 149.3 (C4′), 147.9 (C2′), 144.5 (C8a′), 141.4 (C18), 138.3 (C12), 135.3 (C17), 133.5 (C24), 131.6 (C8′), 130.4 (C14 and C15), 130.0 (C13), 129.4 (C21), 128.6 (C19), 127.4 (C4a′), 121.5 (C7′), 121.1 (C25), 120.7 (C11 and C16 and C26), 119.6 (C3′), 113.1 (C23), 103.3 (C5′), 69.7 (C9), 60.8 (C8), 56.5 (C22OCH
3), 56.2 (C6′O
CH
3), 49.4 (C6), 48.1 (C2), 32.9 (C3) 28.0 (C4), 25.6 (C5), 23.7 (C7); HRMS exact mass calcd. for C
36H
36N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27540.
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-methoxyphenyl)prop-2-en-1-one (
9b)
Yellow solid; Yield: 259 mg (43%); MP.: ~129 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.60 (d,
J = 4.5 Hz, 1H, H2′), 8.40 (s, 1H, H11), 8.26 (m, 1H, H16), 8.04 (d,
J = 8.9 Hz, 2H, H22 and H26), 7.88 (d,
J = 9.2 Hz, 1H, H8′), 7.77 (d,
J = 15.6 Hz, 1H, H19), 7.64−7.69 (m, 2H, H14 and H15), 7.60 (d,
J = 7.1 Hz, 1H, H13), 7.59 (d,
J = 2.7 Hz, 1H, H5′), 7.47 (d,
J = 4.5 Hz, 1H, H3′), 7.36 (d,
J = 15.6 Hz, 1H, H18), 7.34 (dd,
J = 9.2 Hz, 2.6 Hz, 1H, H7′), 7.03 (d,
J = 8.8 Hz, 2H, H23 and H25), 5.57 (br m, 1H, H9 and C9O
H), 3.88 (s, 3H, C6′OCH
3), 3.84 (s, 3H, C24OCH
3), 3.69 (br m, 1H, H2A), 3.16 (br m, H8), 3.15 (1H, H3, overlapped by HDO from solvent), 3.12 (1H, H2B, overlapped by HDO from solvent), 2.81 (br m, 2H, H6α and H6β), 2.10 (br m, 1H, H4), 2.00 (br m, 1H, H7β), 1.66 (br m, 2H, H5α and H5β), 1.43 (br m, 1H, H7α);
13C-NMR (DMSO-
d6 at 45 °C): 187.8 (C20), 164.0 (C24), 157.6 (C6′), 150.3 (C10), 148.8 (C4′), 147.9 (C2′), 144.5 (C8a′), 137.3 (C18), 137.0 (C12), 131.6 (C8′), 131.5 (C14), 131.4 (C22 and C26), 130.7 (C21), 130.6 (C15), 128.9 (C16), 127.4 (C4a′), 127.1 (C13), 125.9 (C17), 125.8 (C19), 125.1 (C11), 121.5 (C7′), 119.7 (C3′), 114.6 (C23 and C25), 103.2 (C5′), 70.4 (C9), 61.0 (C8), 56.2 (C6′O
CH
3), 56.1 (C24OCH
3), 49.5 (C6), 48.4 (C2), 33.1 (C3) 28.6 (C4), 26.0 (C5), 23.7 (C7); HRMS exact mass calcd. for C
36H
37N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27521.
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one (
9c)
Yellow solid; Yield: 300 mg (50%); MP.: ~158 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.66 (br s, 1H, H2′), 8.45 (s, 1H, H11), 8.35 (m, 1H, H16), 7.93 (d,
J = 9.1 Hz, 1H, H8′), 7.86 (d,
J = 15.5 Hz, 1H, H19), 7.74 (1H, H15, overlapped by H14), 7.72 (1H, H14, overlapped by H15), 7.68 (m, 1H, H13), 7.62 (d,
J = 2.5 Hz, 1H, H5′), 7.51 (d,
J = 4.4 Hz, 1H, H3′), 7.43 (d,
J = 15.6 Hz, 1H, H18), 7.38 (dd,
J = 9.1 Hz, 2.5 Hz, 1H, H7′, overlapped by H22 and H26), 7.38 (s, 2H, H22 and H26, overlapped by H7′), 5.61 (br s, 1H, C9O
H), 5.56 (br m, 1H, H9), 3.92 (s, 3H, C6′OCH
3), 3.89 (s, 6H, C23OCH
3 and C25OCH
3), 3.79 (s, 3H, C24OCH
3), 3.78 (br m, 1H, H8), 3.73 (br m, 1H, H2A), 3.15 (1H, H3, overlapped by HDO from solvent), 3.13 (1H, H2B, overlapped by HDO from solvent), 2.84 (br m, 1H, H6α), 2.80 (br m, 1H, H6β), 2.12 (br m, 1H, H4), 2.00 (br m, 1H, H7β), 1.47 (br m, 1H, H7α), 1.69 (br m, 2H, H5α and H5β);
13C-NMR (DMSO-
d6 at 45 °C): 188.3 (C20), 157.4 (C6′), 153.5 (C23 and C25), 149.2 (C10), 147.9 (C2′), 146.8 (C4′), 144.5 (C8a′), 143.3 (C24), 137.9 (C18), 137.1 (C12), 133.1 (C21), 131.7 (C8′ and C14), 131.0 (C17), 130.7 (C15), 128.9 (C16), 127.2 (C13), 127.5 (C4a′), 125.7 (C19), 125.0 (C11), 121.4 (C7′), 119.6 (C3′), 107.3 (C22 and C26), 103.2 (C5′), 68.7 (C9), 60.8 (C8), 56.9 (C23OCH
3 and C24OCH
3 and C25OCH
3), 56.1 (C6′O
CH
3), 49.6 (C6), 48.5 (C2), 33.4 (C3) 28.8 (C4), 26.2 (C5), 23.7 (C7); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 662.29731, found
m/
z: 662.29652.
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
R)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one (
9*c)
Yellow solid; Yield: 102 mg (17%); MP.: ~151 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.66 (br s, 1H, H2′), 8.48 (s, 1H, H11), 8.36 (m, 1H, H16), 7.95 (d,
J = 9.2 Hz, 1H, H8′), 7.88 (d,
J = 15.6 Hz, 1H, H19), 7.75 (1H, H15, overlapped by H14), 7.74 (1H, H14, overlapped by H15), 7.66 (m, 1H, H13), 7.61 (d,
J = 2.5 Hz, 1H, H5′), 7.55 (d,
J = 4.3 Hz, 1H, H3′), 7.42 (d,
J = 15.6 Hz, 1H, H18, overlapped by H7′), 7.41 (dd,
J = 9.1 Hz, 2.5 Hz, 1H, H7′, overlapped by H18), 7.39 (s, 2H, H22 and H26), 6.00 (br s, 1H, C9O
H, overlapped by H9), 5.96 (br m, 1H, H9, overlapped by C9O
H), 4.13 (br m, 1H, H2A), 3.98 (s, 3H, C6′OCH
3), 3.89 (s, 6H, C23OCH
3 and C25OCH
3), 3.79 (s, 3H, C24OCH
3), 3.47 (1H, H2B, overlapped by HDO from solvent), 3.40 (2H, H8 and H3, overlapped by HDO from solvent), 3.18 (br m, 1H, H6α), 3.10 (br m, 1H, H6β), 2.25 (br m, 1H, H4), 2.19 (br m, 1H, H7β), 1.95 (br m, 1H, H7α), 1.86 (br m, 1H, H5α), 1.79 (br m, 1H, H5β);
13C-NMR (DMSO-
d6 at 45 °C): 188.3 (C20), 157.9 (C6′), 153.5 (C23 and C25), 149.2 (C10), 147.8 (C2′), 147.2 (C4′), 144.4 (C8a′), 143.2 (C24), 137.7 (C18), 137.0 (C12), 133.0 (C21), 131.7 (C8′ and C14), 131.0 (C17), 130.7 (C15), 129.0 (C16), 127.2 (C13), 126.8 (C4a′), 125.8 (C19), 125.2 (C11), 121.8 (C7′), 119.6 (C3′), 107.3 (C22 and C26), 102.8 (C5′), 68.7 (C9), 65.3 (C8), 56.9 (C23OCH
3 and C25OCH
3), 56.6 (C6′O
CH
3), 56.5 (C24OCH
3), 49.1 (C6), 48.1 (C2), 32.1 (C3) 28.1 (C4), 27.9 (C7), 24.3 (C5); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 662.29731, found
m/
z: 662.29604.
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
9d)
Orange solid; Yield: 226 mg (37%); MP.: ~174 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.59 (d,
J = 4.4 Hz, 1H, H2′), 8.37 (s, 1H, H11), 8.24 (dd,
J = 7.6 Hz, 2.5 Hz, 1H, H16), 7.88 (d,
J = 9.2 Hz, 1H, H8′), 7.73 (d,
J = 15.6 Hz, 1H, H19), 7.69 (s, 2H, H22 and H26), 7.66 (td,
J = 7.3 Hz, 2.2 Hz, 1H, H14, overlapped by H15), 7.64 (td,
J = 7.4 Hz, 2.1 Hz, 1H, H15, overlapped by H14), 7.58 (m, 2H, H5′ and H13), 7.45 (d,
J = 4.4 Hz, 1H, H3′), 7.33 (dd,
J = 9.1 Hz, 2.8 Hz, 1H, H7′), 7.32 (d,
J = 15.6 Hz, 1H, H18), 5.54 (br s, 2H, H9 and C9O
H), 3.88 (s, 3H, C6′OCH
3), 3.80 (br m, 2H, H6α and H6β), 3.71 (br m, 1H, H2A), 3.16 (br m, 1H, H8, overlapped by HDO from solvent), 3.13 (br m, 1H, H3, overlapped HDO from solvent), 3.09 (br m, 1H, H2B), 2.21 (s, 6H, C
H3), 2.09 (br m, 1H, H4), 1.98 (br m, 1H, H7β), 1.60–1.70 (br m, 2H, H5α and H5β), 1.44 (br m, 1H, H7α);
13C-NMR (DMSO-
d6 at 45 °C): 187.8 (C20), 158.9 (C24), 157.5 (C6′), 150.4 (C10), 149.1 (C4′), 147.8 (C2′), 144.5 (C8a′), 137.0 (C12), 136.6 (C18), 131.6 (C8′), 131.3 (two coalesced lines, C14 and C17), 130.5 (C15), 130.0 (C22 and C26), 129.3 (C21), 128.8 (C16), 127.5 (C4a′), 127.1 (C13), 126.2 (C19), 125.0 (C11), 124.8 (C23 and C25), 121.4 (C7′), 119.5 (C3′), 103.2 (C5′), 70.8 (C9), 61.2 (C8), 56.1 (C6′O
CH
3), 49.5 (C6), 48.5 (C2), 33.3 (C3) 28.6 (C4), 26.3 (C5), 23.9 (C7), 16.9 (
CH
3); HRMS exact mass calcd. for C
37H
38N
5O
4 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29137.
(
E)-3-{2-{4-{(1
S,3
R,4
S,6
R)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
9*d)
Yellow solid; Yield: 324 mg (53%); MP.: ~172 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 9.05 (br s, 1H, C24O
H), 8.57 (d,
J = 4.0 Hz, 1H, H2′), 8.42 (s, 1H, H11), 8.27 (dd,
J = 6.9 Hz, 2.4 Hz, 1H, H16), 7.91 (d,
J = 9.1 Hz, 1H, H8′), 7.78 (d,
J = 15.6 Hz, 1H, H19), 7.72 (s, 2H, H22 and H26), 7.66–7.68 (m, 2H, H14 and H15), 7.64 (d,
J = 2.6 Hz, 1H, H5′), 7.60 (dd,
J = 7.6 Hz, 2.3 Hz, 1H, H13), 7.51 (d,
J = 4.0 Hz, 1H, H3′), 7.36 (dd,
J = 9.1 Hz, 2.6 Hz, 1H, H7′), 7.32 (d,
J = 15.6 Hz, 1H, H18), 6.38 (br s, 1H, H9), 6.22 (br s, 1H, C9O
H), 4.39 (br m, 1H, H2A), 4.00 (s, 3H, C6′OCH
3), 3.69 (br m, 1H, H2B), 3.54 (br m, 1H, H3, overlapped by H8), 3.53 (br m, 1H, H8, overlapped by H3), 3.38 (br m, 1H, H6α), 3.26 (br m, 1H, H6β), 2.32 (br m, 1H, H7β, overlapped by H4), 2.31 (br m, 1H, H4, overlapped by H7β), 2.22 (s, 6H, C
H3), 1.95 (br m, 1H, H5α), 1.86 (br m, 1H, H5β), 1.19 (br m, 1H, H7α);
13C-NMR (DMSO-
d6 at 45 °C): 187.7 (C20), 159.0 (C24), 158.4 (C6′), 148.3 (C10), 147.8 (C2′), 145.9 (C4′), 144.4 (C8a′), 136.9 (C12), 136.3 (C18), 131.7 (C8′), 131.4 (C14), 130.7 (C15 and C17), 130.1 (C22 and C26), 129.3 (C21), 128.8 (C16), 127.2 (C13), 126.3 (C4a′ and C19), 125.3 (C11), 124.9 (C23 and C25), 122.2 (C7′), 119.5 (C3′), 102.6 (C5′), 66.9 (C9), 60.0 (C8), 59.9 (C6′O
CH
3), 48.9 (C6), 47.9 (C2), 31.5 (C3) 27.5 (C4), 23.3 (C5), 18.4 (C7), 16.9 (
CH
3); HRMS exact mass calcd. for C
37H
38N
5O
4 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29097.
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(2-methoxyphenyl)prop-2-en-1-one (
10a)
Yellow solid; Yield: 114 mg (19%); MP.: ~173 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.77 (s, 1H, H11), 8.65 (d,
J = 4.5 Hz, 1H, H2′), 7.93-7.96 (m, 4H, H13 and H14 and H16 and H17), 7.91 (d,
J = 9.2 Hz, 1H, H8′), 7.62 (d,
J = 2.6 Hz, 1H, H5′), 7.54 (d,
J = 16.0 Hz, 1H, H18), 7.48–7.53 (m, 3H, H3′ and H24 and H26), 7.45 (d,
J = 16.0 Hz, 1H, H19), 7.37 (dd,
J = 9.2 Hz, 2.6 Hz, 1H, H7′), 7.17 (d,
J = 8.3 Hz, 1H, H23), 7.04 (td,
J = 7.2 Hz, 0.9 Hz, 1H, H25), 6.42–6.08 (br s, 2H, H9 and C9O
H), 3.98 (s, 3H, C6′OCH
3), 3.86 (s, 3H, C22OCH
3), 3.68-3.73 (br s, 2H, H6α and H6β), 3.52 (br s, 1H, H8), 3.43 (br s, 1H, H3), 3.20–3.34 (br s, 2H, H2A and H2B), 2.35 (br s, 1H, H4), 2.25 (br s, 1H, H7β), 1.84–1.90 (br s, 2H, H5α and H5β), 1.28 (br s, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 192.5 (C20), 158.4 (C22), 158.1 (C6′), 149.2 (C10), 147.8 (C2′), 146.0 (C4′), 144.4 (C8a′), 141.4 (C18), 138.2 (C12), 135.4 (C15), 133.5 (C24), 131.8 (C8′), 130.5 (C14 and C16), 130.0 (C26), 129.4 (C21), 128.7 (C19), 126.7 (C4a′), 122.0 (C7′), 121.1 (C25), 121.0 (C11), 120.8 (C13 and C17), 119.4 (C3′), 113.1 (C23), 102.8 (C5′), 66.2 (C9), 60.1 (C8), 57.0 (C6′OCH
3), 56.5 (C22OCH
3), 49.0 (C2), 47.9 (C6), 31.5 (C3), 27.1 (C4), 26.0 (C7), 23.3 (C5); HRMS exact mass calcd. for C
36H
37N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27546.
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-methoxyphenyl)prop-2-en-1-one (
10b)
Yellow solid; Yield: 373 mg (62%); MP.: ~154 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.73 (s, 1H, H11), 8.64 (d,
J = 4.5 Hz, 1H, H2′), 8.13 (d,
J = 8.9 Hz, 2H, H22 and H26), 8.06 (d,
J = 8.6 Hz, 2H, H14 and H16), 7.99 (d,
J = 8.6 Hz, 2H, H13 and H17), 7.93 (d,
J = 15.6 Hz, 1H, H19), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.73 (d,
J = 15.6 Hz, 1H, H18), 7.60 (d,
J = 2.7 Hz, 1H, H5′), 7.46 (d,
J = 4.5 Hz, 1H, H3′), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 7.06 (d,
J = 8.9 Hz, 2H, H23 and H25), 5.54 (br s, 1H, C9O
H), 5.46 (br s, 1H, H9), 3.91 (s, 3H, C6′OCH
3), 3.85 (s, 3H, C24OCH
3), 3.65 (br s, 1H, H2A), 3.14 (1H, H8, overlapped by HDO from solvent), 3.06 (1H, H3, overlapped by HDO from solvent), 3.05 (1H, H2B, overlapped by HDO from solvent), 2.75 (br s, 2H, H6α and H6β), 2.13 (s, 1H, H4), 1.91 (s, 1H, H7β), 1.64 (m, 2H, H5α and H5β), 1.51 (s, 1H, H7α);
13C-NMR (DMSO-
d6 at 45 °C): 188.0 (C20), 163.9 (C24), 157.5 (C6′), 151.2 (C10), 149.3 (C4′), 147.9 (C2′), 144.6 (C8a′), 142.0 (C18), 138.3 (C12), 135.4 (C15), 131.4 (C22 and C26), 131.6 (C8′), 131.1 (C21), 130.7 (C14 and C16), 129.4 (C4a′), 123.9 (C19), 121.4 (C7′), 121.0 (C11), 120.8 (C14 and C16), 119.6 (C3′), 114.6 (C23 and C25), 103.4 (C5′), 70.8 (C9), 61.2 (C8), 56.1 (C6′O
CH
3 and C24OCH
3), 49.6 (C6), 48.3 (C2), 33.2 (C3), 28.2 (C4), 26.4 (C5), 24.2 (C7); HRMS exact mass calcd. for C
36H
37N
5O
4 [MH]
+, requires
m/
z: 602.27618, found
m/
z: 602.27550.
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one (
10c)
Yellow solid; Yield: 377 mg (57%); MP.: ~148 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.75 (s, 1H, H11), 8.65 (d,
J = 4.5 Hz, 1H, H2′), 8.10 (d,
J = 8.6 Hz, 2H, H14 and H16), 8.00 (d,
J = 8.6 Hz, 2H, H13 and H17), 7.93 (d,
J = 15.6 Hz, 1H, H19), 7.90 (d,
J = 9.2 Hz, 1H, H8′), 7.77 (d,
J = 15.6 Hz, 1H, H18), 7.61 (d,
J = 2.7 Hz, 1H, H5′), 7.47 (d,
J = 4.5 Hz, 1H, H3′), 7.41 (s, 2H, H22 and H26), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 5.66 (br s, 2H, C9O
H and H9), 3.93 (s, 3H, C6′OCH
3), 3.89 (s, 6H, C23OCH
3 and C25OCH
3), 3.77 (s, 3H, C24OCH
3), 3.22 (br m, 1H, H8), 3.70 (br m, 1H, H2A), 3.17 (1H, H3, overlapped by HDO from solvent), 3.26 (1H, H2B, overlapped by HDO from solvent), 2.87 (br m, 2H, H6α and H6β), 2.18 (br m, 1H, H4), 1.99 (br m, 1H, H7β), 1.45 (br m, 1H, H7α), 1.70 (br m, 2H, H5α and H5β);
13C-NMR (DMSO-
d6 at 45 °C): 188.5 (C20), 157.7 (C6′), 153.5 (C23 and C25), 150.7 (C10), 147.9 (C2′), 145.9 (C4′), 144.5 (C8a′), 143.2 (C24), 142.7 (C18), 138.4 (C12), 135.4 (C15), 133.4 (C21), 131.6 (C8′), 130.9 (C14 and C16), 127.3 (C4a′), 123.7 (C19), 121.7 (C7′), 120.8 (C11), 120.6 (C13 and C17), 119.6 (C3′), 107.4 (C22 and C26), 103.2 (C5′), 60.9 (C8), 60.7 (C24OCH
3), 57.0 (C23OCH
3 and C25OCH
3), 56.3 (C6′O
CH
3), 49.5 (C6), 48.3 (C2), 32.1 (C3) 28.0 (C4), 25.8 (C5), 23.4 (C7); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 662.29731, found
m/
z: 662.29668.
(
E)-3-{4-{4-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
10d)
Yellow solid; Yield: 148 mg (24%); MP.: ~188 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 9.06 (br s, 3H, C24O
H), 8.78 (s, 1H, H11), 8.64 (d,
J = 4.5 Hz, 1H, H2′), 8.05 (d,
J = 8.7 Hz, 2H, H14 and H16), 7.98 (d,
J = 8.7 Hz, 2H, H13 and H17), 7.90 (d,
J = 15.6 Hz, 1H, H19, overlapped by H8′), 7.90 (d,
J = 9.2 Hz, 1H, H8′, overlapped by H19), 7.77 (s, 2H, H22 and H26), 7.68 (d,
J = 15.6 Hz, 1H, H18, overlapped by H5′), 7.66 (1H, H5′, overlapped by H18), 7.49 (d,
J = 4.5 Hz, 1H, H3′), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 6.11 (br s, 1H, H9), 6.05 (br s, 1H, C9O
H), 4.14 (br m, 1H, H2A), 3.97 (s, 3H, C6′OCH
3), 3.48 (br m, 1H, H2B), 3.40 (br m, 1H, H8), 3.35 (br m, 1H, H3), 3.18 (br m, 1H, H6α), 3.08 (br m, 1H, H6β), 2.29 (br m, 1H, H4), 2.24 (s, 6H, C
H3), 2.19 (br m, 1H, H7β), 1.78–1.85 (br m, 2H, H5α and H5β), 1.34 (br m, 1H, H7α);
13C-NMR (DMSO-
d6 at 45 °C): 188.0 (C20), 158.8 (C6′), 158.1 (C24), 149.7 (C10), 147.8 (C2′), 147.2 (C4′), 144.5 (C8a′), 141.2 (C18), 138.1 (C12), 135.8 (C15), 131.7 (C8′), 130.6 (C14 and C16), 130.0 (C22 and C26), 129.6 (C21), 126.8 (C4a′), 124.9 (C23 and C25), 124.2 (C19), 121.9 (C7′), 121.0 (C11), 120.6 (C13 and C17), 119.5 (C3′), 103.0 (C5′), 68.2 (C9), 60.5 (C8), 56.8 (C6′O
CH
3), 49.1 (C6), 48.0 (C2), 32.1 (C3) 27.7 (C4), 24.4 (C5), 20.6 (C7), 16.9 (
CH
3); HRMS exact mass calcd. for C
37H
38N
5O
4 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29129.
(
E)-3-{4-{5-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one (
11c)
Brownish yellow solid; Yield: 264 mg (40%); MP.: ~152 °C (from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.64 (d,
J = 4.5 Hz, 1H, H2′), 8.06 (d,
J = 8.4 Hz, 2H, H16 and H14), 7.95 (d,
J = 15.6 Hz, 1H, H19), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.82 (s, 1H, H11), 7.76 (d,
J = 15.6 Hz, 1H, H18), 7.59 (d,
J = 8.4 Hz, 2H, H13 and H17), 7.49 (2H, H3′ and H5′), 7.41 (s, 2H, H22 and H26), 7.36 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 5.54 (br s, 1H, C9O
H), 5.27 (br s, 1H, H9), 3.88 (s, 3H, C6′OCH
3), 3.87 (s, 6H, C23OCH
3 and C25OCH
3), 3.77 (s, 3H, C24OCH
3), 3.25 (br s, 1H, H8), 3.13 (overlapped by HDO signal from solvent, 1H, H3), 1.85 (br s, 1H, H4), 1.68 (br s, 1H, H5α), 1.58 (t,
J = 10.9 Hz, 1H, H7α), 1.38 (br s, 1H, H5β), 1.24 (m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C): 188.4 (C20), 157.6 (C6′), 153.5 (C23 and C25), 147.9 (C2′), 144.5 (C8a′), 143.1 (C24), 142.5 (C18), 142.4 (C10), 138.1 (C12), 136.6 (C15), 133.3 (C21), 131.9 (C11), 131.7 (C8′), 130.4 (C14 and C16), 127.4 (C4a′), 126.4 (C13 and C17), 124.5 (C19), 121.3 (C7′), 119.7 (C3′), 107.3 (C22 and C26), 103.3 (C5′), 71.0 (C9), 60.7 (C8), 60.6 (C24OCH
3), 57.0 (C23OCH
3 and C25OCH
3), 56.1 (C6′O
CH
3), 31.1 (C3), 26.6 (C4), 24.2 (C5), 22.0 (C7); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 662.29731, found
m/
z: 662.29685.
(
E)-3-{4-{5-{(1
S,3
R,4
S,6
S)-6-[(
S)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
11d)
Brown solid; Yield: 193 mg (31%); MP.: ~176 °C (from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 9.02 (br s, 1H, C24OH), 8.64 (d,
J = 4.4 Hz, 1H, H2), 8.19 (d,
J = 8.5 Hz, 2H, H14 and H16), 8.09 (s, 1H, H11), 7.92 (d,
J = 15.5 Hz, 1H, H19), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.78 (s, 2H, H22 and H26), 7.68 (d,
J = 15.5 Hz, 1H, H18), 7.49 (d,
J = 4.5 Hz, 1H, H3′), 7.56 (d,
J = 8.2 Hz, 2H, H13 and H17), 7.52 (d,
J = 2.8 Hz, 1H, H5′), 7.35 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 5.61 (br s, 1H, C9OH), 5.33 (br s, 1H, H9), 3.89 (s, 3H, C6′OCH
3), 2.90-3.20 (6H, H2A and H2B and H3 and H6α and H6β and H8, overlapped by HDO signal from solvent), 2.23 (s, 6H, CH
3), 1.83 (br s, 1H, H4), 1.68 (br s, 1H, H5α), 1.56–1.59 (2H, H5β and H7α), 1.37 (br m, 1H, H7β);
13C-NMR (DMSO-
d6 at 45 °C, chemical shifts were determined from
1H-
13C HSQC- and
1H-
13C HMBC spectra): 187.9 (C20), 158.7 (C24), 157.5 (C6′), 147.9 (C2′), 144.5 (C8a′), 141.1 (C18), 139.8 (C10), 138.1 (C12), 136.8 (C15), 131.8 (C11), 131.6 (C8′), 130.2 (C14 and C16), 130.1 (C22 and C26), 129.5 (C21), 127.2 (C4a′), 126.4 (C13 and C17), 124.9 (C19 and C23 and C25), 121.4 (C7′), 119.7 (C3′), 103.3 (C5′), 70.1 (C9), 60.5 (C8), 56.2 (C6′OCH
3), 54.4 (C2), 42.1 (C6), 31.0 (C3), 26.6 (C4), 25.3 (C5), 23.2 (C7), 16.9 (CH
3); HRMS exact mass calcd. for C
37H
38N
5O
4 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29165.
(
E)-3-{4-{5-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one (
12c)
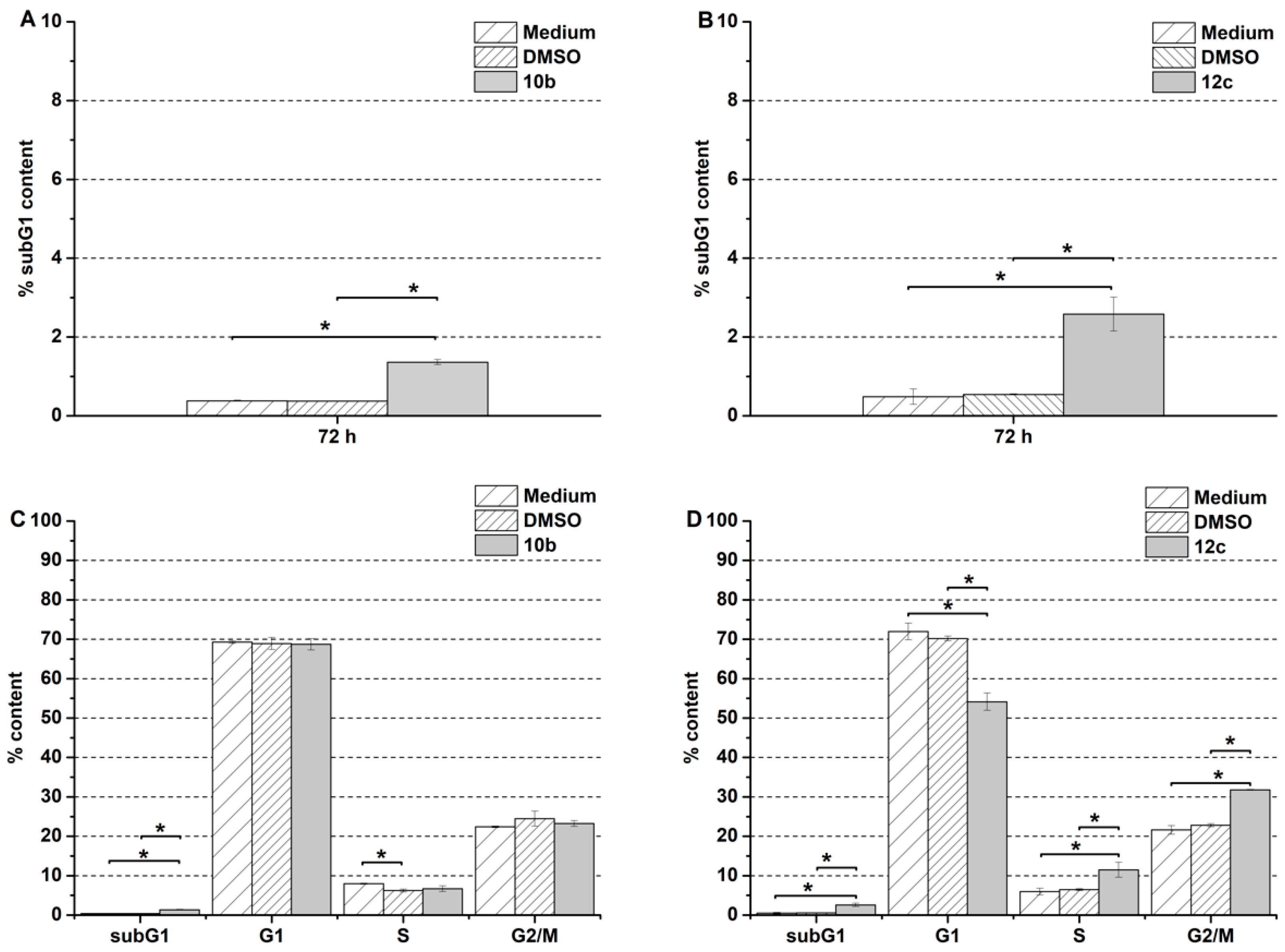
Yellow solid; Yield: 400 mg (60%); MP.: ~134 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 8.64 (d,
J = 4.5 Hz, 1H, H2′), 8.07 (d,
J = 8.4 Hz, 2H, H14 and H16, overlapped by H11), 8.06 (s, 1H, H11, overlapped by H14 and H16), 7.95 (d,
J = 15.6 Hz, 1H, H19), 7.89 (d,
J = 9.1 Hz, 1H, H8′), 7.78 (d,
J = 15.6 Hz, 1H, H18), 7.62 (d,
J = 8.4 Hz, 2H, H13 and H17), 7.49 (d,
J = 4.5 Hz, 1H, H3′), 7.41 (s, 2H, H22 and H26), 7.38 (d,
J = 2.7 Hz, 1H, H5′), 7.32 (dd,
J = 9.2 Hz, 2.7 Hz, 1H, H7′), 5.61 (br s, 2H, C9O
H), 5.34 (br s, 1H, H9), 3.87 (s, 6H, C23OCH
3 and C25OCH
3), 3.83 (s, 3H, C6′OCH
3), 3.76 (s, 3H, C24OCH
3), 3.31 (br m, 1H, H2A), 3.04 (t,
J = 8.8 Hz, 1H, H3), 2.97 (qua,
J = 7.2 Hz, 1H, H8), 2.81 (m, 1H, H2B), 2.64 (br m, 1H, H6α), 2.55 (m, 1H, H6β), 1.92 (dd,
J = 12.6 Hz, 10.3 Hz, 1H, H7β), 1.85 (br s, 1H, H4), 1.44 (br m, 1H, H5β), 1.32 (br m, 2H, H7α and H5α);
13C-NMR (DMSO-
d6 at 45 °C): 188.3 (C20), 157.3 (C6′), 153.4 (C23 and C25), 141.3 (C10), 147.9 (C2′), 149.6 (C4′), 144.2 (C8a′), 143.1 (C24), 142.6 (C18), 138.1 (C12), 136.4 (C15), 133.3 (C21), 131.6 (C8′), 130.5 (C14 and C16), 127.3 (C4a′), 124.1 (C19), 121.5 (C7′), 132.3 (C11), 126.3 (C13 and C17), 119.3 (C3′), 106.8 (C22 and C26), 102.8 (C5′), 71.1 (C9), 60.8 (C8), 60.7 (C24OCH
3), 56.7 (C23OCH
3 and C25OCH
3), 55.9 (C6′O
CH
3), 49.2 (C6), 49.0 (C2), 31.5 (C3) 27.5 (C4), 26.3 (C5), 22.7 (C7); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 662.29731, found
m/
z: 662.29687.
(
E)-3-{4-{5-{(1
S,3
R,4
S,6
R)-6-[(
R)-Hydroxy(6-methoxyquinolin-4-yl)methyl]quinuclidin-3-yl}-1
H-1,2,3-triazol-1-yl}phenyl}-1-(4-hydroxy-3,5-dimethylphenyl)prop-2-en-1-one (
12d)
Yellow solid; Yield: 317 mg (51%); MP.: ~177 °C (dec., from diethyl ether);
1H-NMR (DMSO-
d6 at 45 °C): 9.03 (br s, 1H, C24OH), 8.65 (d,
J = 4.4 Hz, 1H, H2′), 8.09 (s, 1H, H11), 8.04 (d,
J = 8.5 Hz, 2H, H14 and H16), 7.92 (d,
J = 15.6 Hz, 1H, H19), 7.89 (d,
J = 9.2 Hz, 1H, H8′), 7.78 (s, 2H, H22 and H26), 7.70 (d,
J = 15.6 Hz, 1H, H18), 7.60 (d,
J = 8.5 Hz, 2H, H13 and H17), 7.51 (d,
J = 4.5 Hz, 1H, H3′), 7.42 (br s, 1H, H5′), 7.34 (dd,
J = 9.2 Hz, 2.3 Hz, 1H, H7′), 5.83 (br s, 1H, C9OH), 5.58 (s, 1H, H9), 3.87 (s, 3H, C6′OCH
3), 3.53 (br s, 1H, H2A), 3.15 (1H, H3, overlapped by HDO signal from solvent), 3.11 (1H, H8, overlapped by HDO from solvent), 3.00 (m, 1H, H2B), 2.80 (m, 1H, H6α), 2.70 (m, 1H, H6β), 2.23 (s, 6H, CH
3), 2.02 (dd,
J = 12.6 Hz, 9.2 Hz, 1H, H7β), 1.90 (s, 1H, H4), 1.53 (q,
J = 9.2 Hz, 1H, H5α), 1.40 (t,
J = 10.3 Hz, 1H, H5β), 1.29 (m, 1H, H7α);
13C-NMR (DMSO-
d6 at 45 °C): 187.9 (C20), 158.9 (C24), 157.6 (C6′), 147.9 (C2′), 144.4 (C8a′), 141.1 (C18), 140.8 (C10), 137.7 (C12), 136.9 (C15), 132.3 (C11), 131.6 (C8′), 130.2 (C14 and C16), 130.1 (C22 and C26), 129.5 (C21), 127.1 (C4a′), 126.4 (C13 and C17), 124.9 (C23 and C25), 124.8 (C19), 121.5 (C7′), 119.7 (C3′), 103.0 (C5′), 70.2 (C9), 60.5 (C8), 56.2 (C6′OCH
3), 49.0 (C6), 48.8 (C2), 31.1 (C3), 27.3 (C4), 25.5 (C5), 21.6 (C7), 16.9 (CH
3); HRMS exact mass calcd. for C
38H
40N
5O
6 [MH]
+, requires
m/
z: 616.29183, found
m/
z: 616.29137.