2.1. Rate and Yield of Hydrogen Generation
Our previous report on organic acid-promoted hydrolysis of ammonia borane included rates and yields of hydrogen generation when using either tartaric acid or oxalic acid to promote hydrolysis. While both of these acids were capable of promoting hydrolysis with favorable results, for this paper we have chosen to use maleic acid to promote hydrolysis as it is more water-soluble than oxalic acid and its first pKa is lower than that of tartaric acid (1.94 compared to 2.89). Water solubility is an important factor for system design as a more soluble acid will require less water to become fully dissolved which allows for a more compact reaction vessel. Previous authors have shown the kinetics of acid promoted hydrolysis to be first order with pH, meaning for a given molarity of solution, an acid with a lower pKa will lead to more rapid hydrolysis [
4]. Additionally, maleic acid is cheaper than either tartaric acid or oxalic acid, making it attractive for commercial applications.
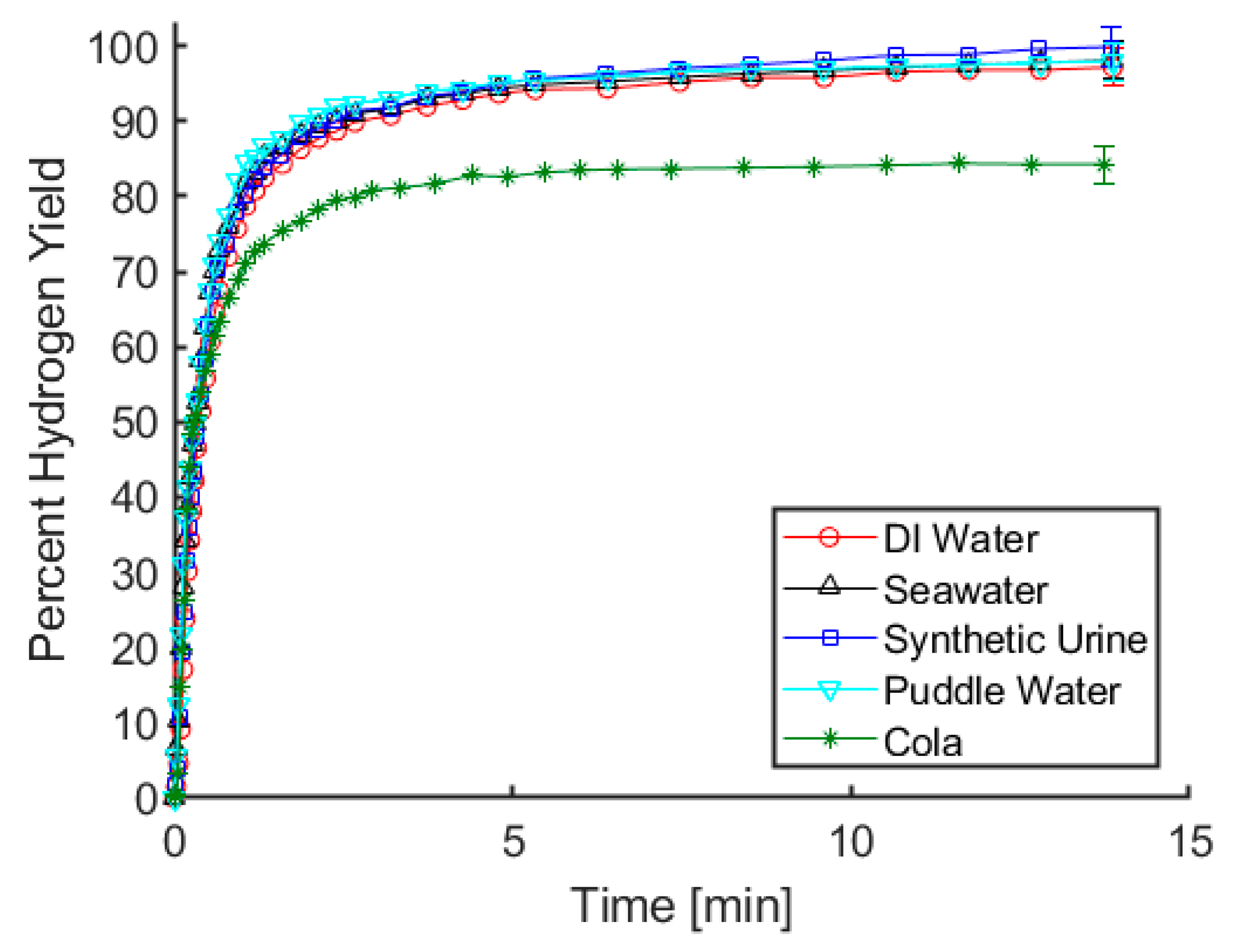
An important trait for our proposed hydrogen reactor is that for any given water source, the rate and yield of hydrogen release remain relatively constant. As can be seen in
Figure 1, there is virtually no difference in rate or yield of gas evolution when using maleic acid and deionized water, seawater, synthetic urine, or puddle water. Tests using a carbonated cola beverage resulted in an initial rate of hydrogen release that was comparable to the results using other fluids, but only 84% of the theoretical hydrogen yield was observed. It is notable that gas evolution occurred as soon as ammonia borane and cola were mixed, even prior to acid addition. Considering that the cola has a pH below 3, it is likely that the acidity of the soft drink resulted in the generation of some amount of hydrogen upon initial contact with ammonia borane. Using our test procedures, there is a brief moment between mixing the cola with the ammonia borane and connecting the vessel containing the solution to the gas measurement apparatus, and it is likely that the lower hydrogen yield can be attributed to gas generation during that time. From an application point of view, using the cola resulted in a great deal of foaming and left a sticky residue on the glassware used for these tests. While foaming solutions and sticky glassware are only mild nuisances in a laboratory setting, in a fielded system these factors could potentially lead to clogged tubing or moving parts becoming stuck. For these reasons, carbonated soft drinks such as cola are not an ideal water source for hydrolysis. For the remaining water sources, the results in
Figure 1 indicate that at least with respect to rate and yield of gas evolution, acid-promoted hydrolysis is relatively indifferent to the water source being used.
2.2. Hydrogen Purity
To ensure the PEMFC compatibility of hydrogen generated via organic acid-promoted hydrolysis of ammonia borane using low-quality water sources, Fourier transform infrared spectroscopy (FTIR) was used to detect contaminants within the gas products. Three contaminants were of particular interest: ammonia, because it is a byproduct of metal-catalyzed ammonia borane hydrolysis and is highly damaging to PEMFCs; carbon monoxide because it is also highly damaging to PEMFCs, and carbon dioxide because it readily dissolves in water and can be found in many water sources. Both ammonia and carbon monoxide have been found to be damaging to fuel cells at levels as low as single-digit parts per million, with specific examples of the extent of fuel cell degradation varying based on fuel cell design and operational parameters [
6,
7,
8,
9,
10]. While the influence of carbon dioxide contamination is not as severe at low concentrations, several reports have found gas mixtures that contain between 10–50% (100,000–500,000 ppm) of carbon dioxide to be damaging to fuel cells [
6,
7,
11]. Gas analysis tests were conducted using the same water sources that were used for comparison of the hydrogen generation rate and yield. The resulting spectrums, as well as two spectrums from the ammonia calibration curve that have been added for comparison, can be seen in
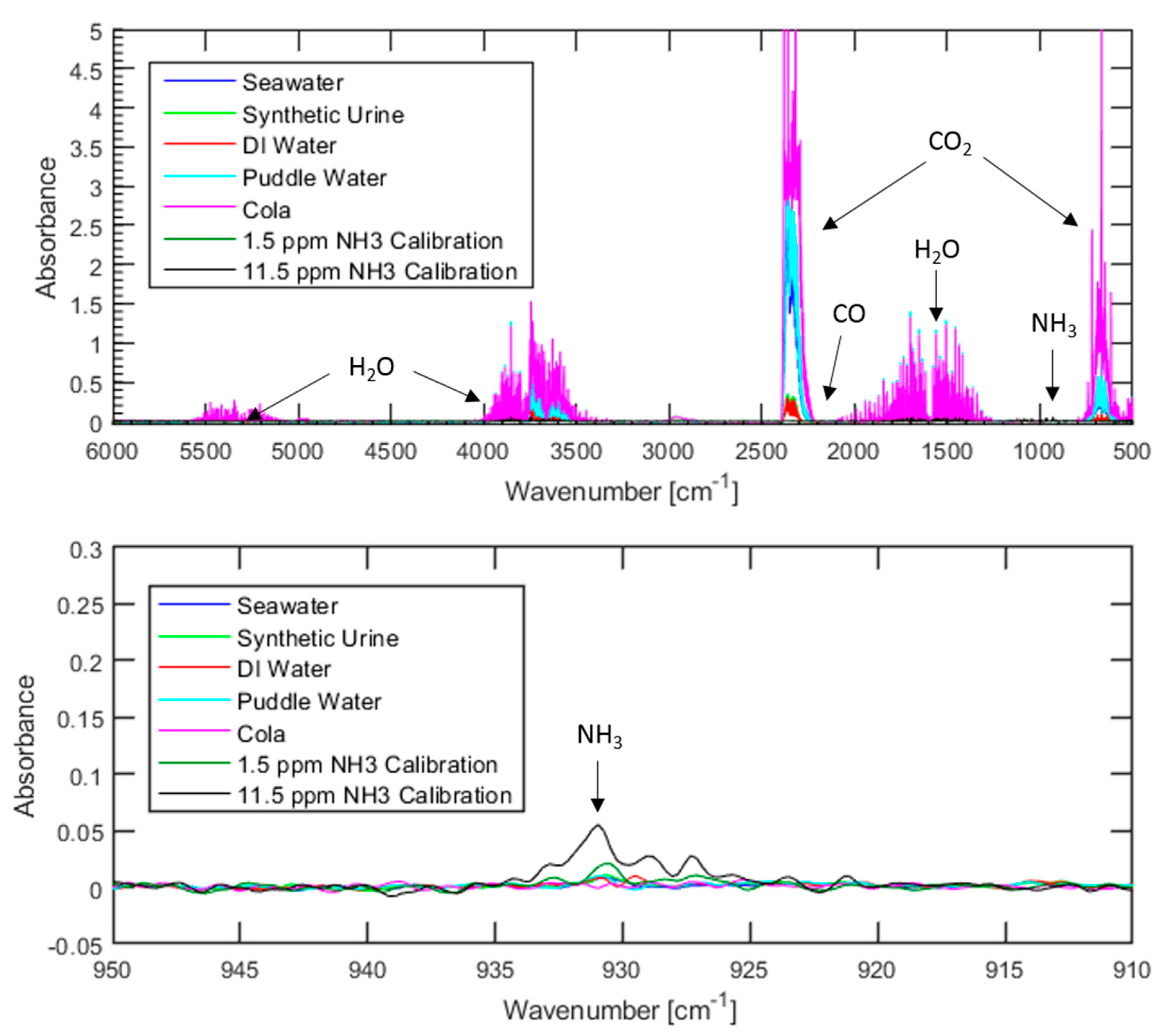
Figure 2 below
Regardless of the water source used, ammonia levels were below the 7.4 ppm lower detection limit associated with these conditions. Similarly, none of the tests resulted in carbon monoxide levels above the lower detection limit of 19.3 ppm. However, carbon dioxide was detected in each spectrum, with the concentration being highly dependent on the water source that is used. Not surprisingly, the cola resulted in the highest carbon dioxide levels with over 2500 ppm, while deionized water led to levels less than 300 ppm. Considering that previous studies concerning the influence of carbon dioxide on fuel cell performance typically investigate carbon dioxide concentrations of 100,000 ppm or greater, it is highly unlikely that 2500 ppm or less of carbon dioxide would lead to fuel cell degradation. A summary of the results of FTIR testing can be seen in
Table 1 below.
Because ammonia can damage fuel cells at concentrations below the 7.4 ppm lower detection limit of the FTIR tests discussed above, further testing was conducted using Dräger ammonia detection tubes capable of measuring ammonia levels as low as 0.25 ppm. Hydrolysis was conducted under similar conditions to those used during FTIR testing, with solutions being made using deionized water. The test was repeated several times, with the Dräger tubes confirming the absence of ammonia in excess of 0.25 ppm.
The presence of carbon dioxide in the tests with puddle water and seawater is expected as both are exposed to open air for long periods of time prior to use, allowing for carbon dioxide from the atmosphere to dissolve into the water. The solubility of carbon dioxide in water decreases as pH decreases, meaning that when acid is injected into the system the equilibrium between aqueous and gaseous carbon dioxide will shift to favor the gas. It is highly likely that carbon dioxide dissolved in the puddle water and seawater prior to use is responsible for the presence of carbon dioxide in the spectrum in
Figure 2. This hypothesis is further supported by the very low levels of carbon dioxide detected when using deionized water or synthetic urine, both of which have minimal exposure to open-air prior to use.
In order to directly compare the influence of water sources on hydrogen purity when using organic acids versus metal catalysts, similar tests to those discussed above were conducted by replacing the maleic acid with 10 wt.% platinum on a carbon support to catalyze hydrolysis. The platinum to ammonia borane loading was approximately 0.018:1 by mass, which is the same loading ratio as used by previous authors [
12]. Tests were conducted using seawater and deionized water, with the gas products being analyzed by FTIR. The resulting spectrums can be seen in
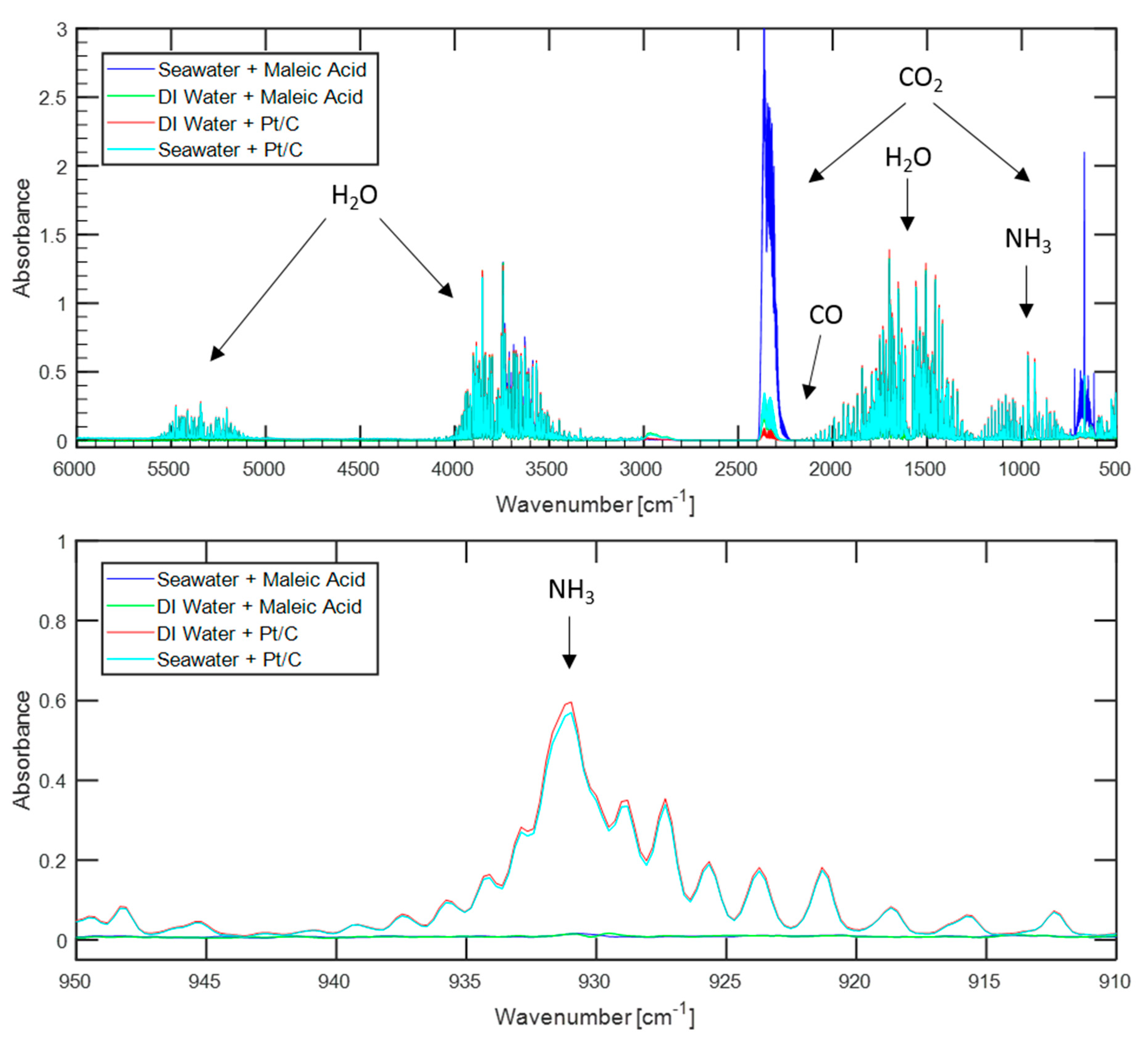
Figure 3, along with those from maleic acid promoted hydrolysis using the same water sources for ready comparison.
Using the platinum-catalyst resulted in approximately 1100 ppm of ammonia when using either seawater or deionized water, far more than the non-detectable amount (less than 7.4 ppm) evolved when using maleic acid. Such high levels of ammonia would necessitate a further ammonia sequestration system before hydrogen evolved via platinum-catalyzed hydrolysis could be used to power a PEMFC.
Lower levels of carbon dioxide were detected in the product gas stream when using the platinum catalyst than when using maleic acid. Using deionized water, the gas sample from platinum catalysis contained less than 26 ppm of carbon dioxide, while using maleic acid and the same water source resulted in nearly 300 ppm. Similarly, using platinum and seawater resulted in 350 ppm of carbon dioxide compared to 1700 ppm when using maleic acid. These results strengthen the argument that carbon dioxide enters the system dissolved in the water sources and is primarily released when the pH of the water source is lowered during acid addition.
The water source used for hydrolysis does not seem to have an influence on the levels of carbon monoxide or ammonia that are released during hydrolysis, but does have an influence on the levels of carbon dioxide that are released. The choice of reaction promoter does have a strong influence on the levels of ammonia and carbon dioxide detected in the hydrolysis gas products, with platinum catalysts resulting in high levels of ammonia and low levels of carbon dioxide. Carbon monoxide was never measured in excess of the 19.3 ppm lower detection limit for any of the combinations of water sources and reaction promoter that were investigated.
2.3. Hydrogen Reactor Design
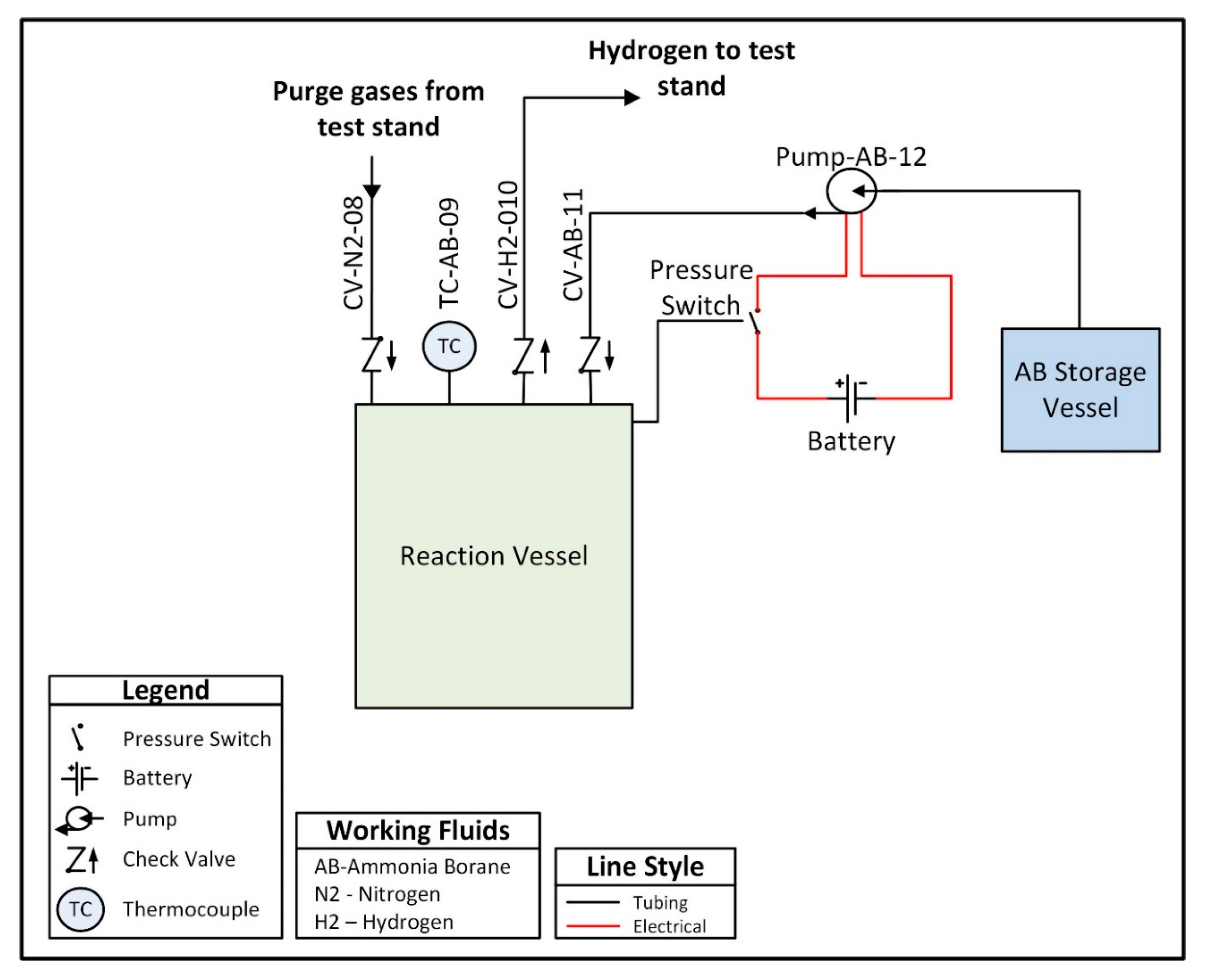
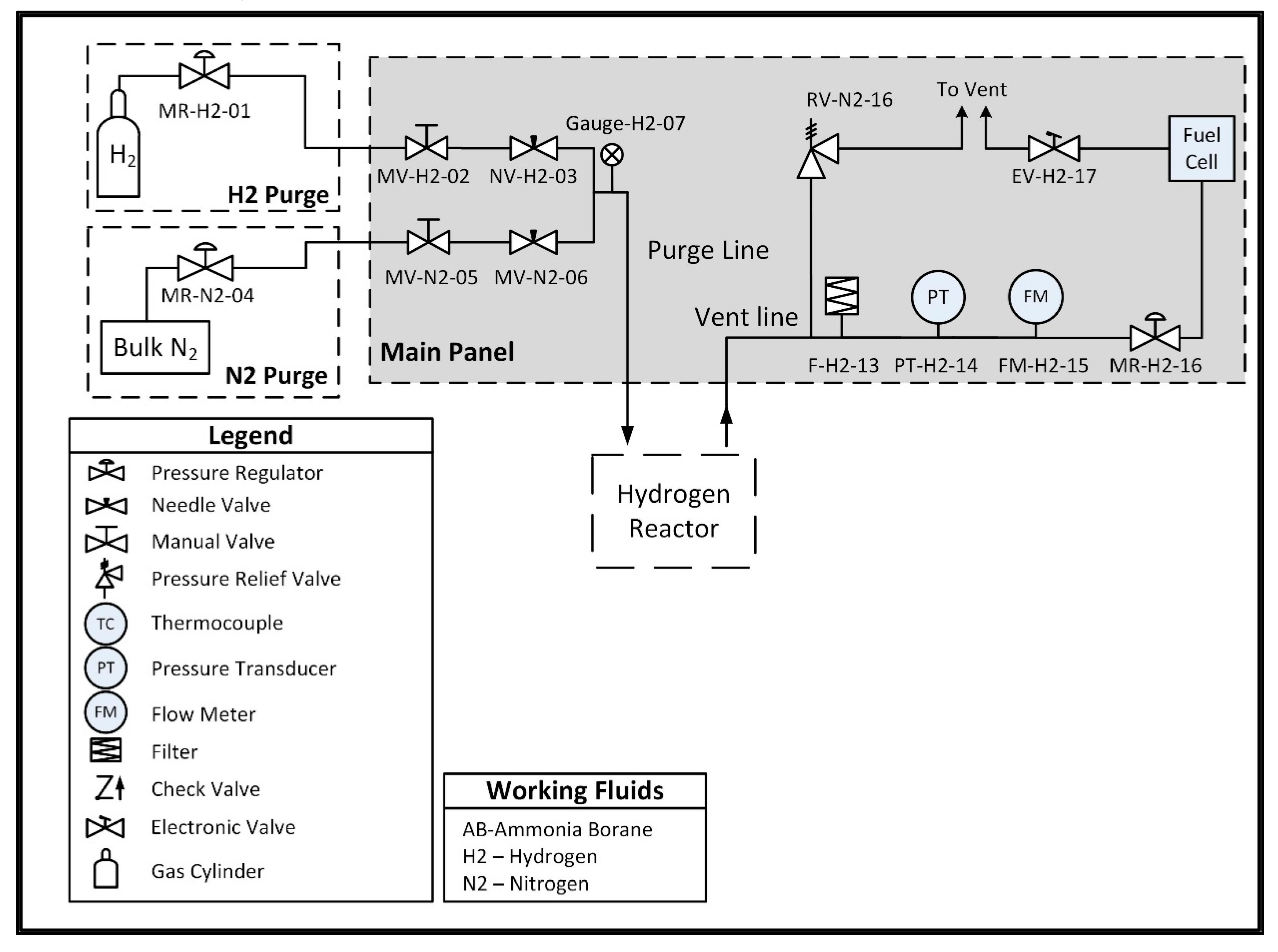
A custom hydrogen reactor was designed to accommodate acid promoted hydrolysis, and is comprised of two chambers connected by a peristaltic pump. One chamber, referred to as the ammonia borane chamber, is a lightweight vessel that houses an ammonia borane solution and is not subjected to internal pressure. The second vessel, referred to as the reaction chamber, houses an acid solution and is designed to withstand pressures up to 200 kPa. The peristaltic pump is used to move the solution from the ammonia borane chamber into the reaction chamber in order to initiate hydrolysis and generate hydrogen. A check valve between the reaction chamber and the peristaltic pump prevents fluid from the reaction chamber from back flowing into the line between the two chambers. The pump is intermittently powered using a 9 V battery and is controlled using a normally closed pressure switch which opens to break the circuit when the internal pressure exceeds 150 kPa, thus stopping the flow of ammonia borane solution and preventing a buildup of hydrogen pressure. The low-pressure switch is a passive control unit, meaning the only electrical input required is the power supply (in this case the 9 V battery) for the peristaltic pump. In future prototypes, the pump could be powered by the fuel cell once the system has reached steady-state operation, with an external power source only being required during the startup transient. The entirety of the hydrogen reactor and the control system is approximately 500 g. A plumbing and instrumentation diagram (PandID) of the reactor is shown in
Figure 4 below.
The design of this system creates a passively controlled, on-demand hydrogen generation scheme that can provide a wide range of gas evolution rates as required by the fuel cell to meet the load demand. This allows the system to operate at varying loads without wide fluctuations in hydrogen pressure. This is an important factor with regards to the safety of the system, especially when considering risk mitigation for the potential use case of military operations where the vessel could be compromised while filled with hydrogen gas. Operating at low pressures also allows for thinner vessel walls and lighter hardware which increases the energy density of the system.
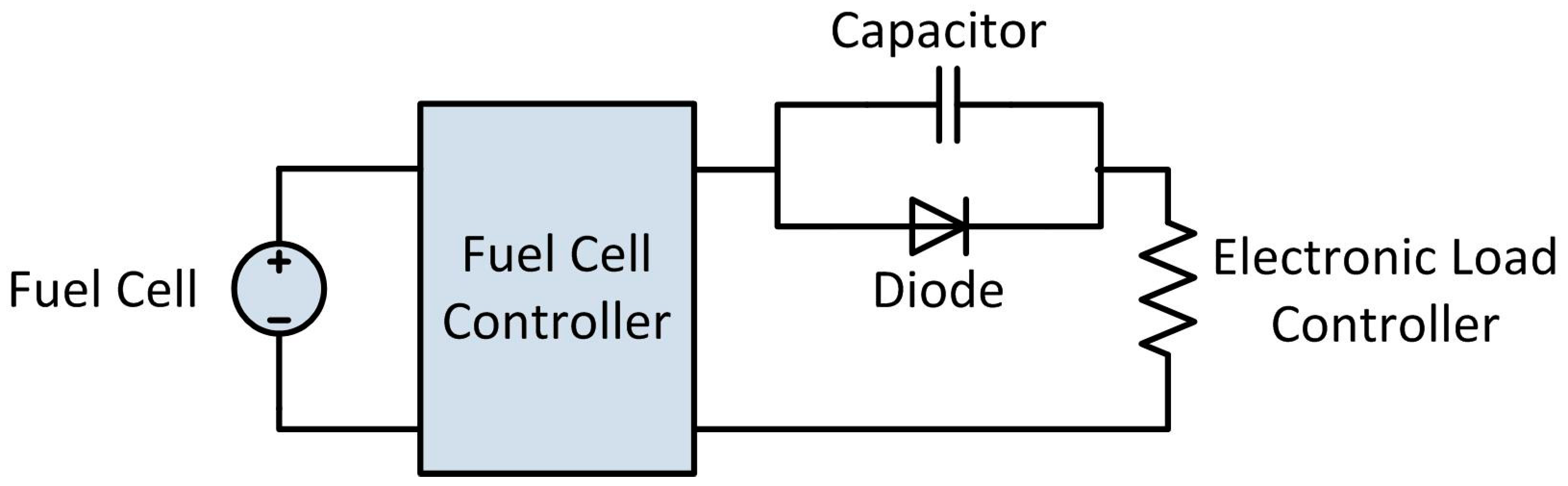
2.4. Power Production Testing
Using the hydrogen reactor detailed above and the test stand detailed in
Section 4.4, acid-promoted hydrolysis of ammonia borane was used to generate hydrogen and power a PEMFC in order to validate our proposed portable hydrogen generation system. Tests were conducted using 7.5 g of ammonia borane and 28.2 g of maleic acid, providing a 1:1 molar ratio of the two. The ammonia borane was dissolved into 30 g of water to form a 20 wt.% solution, while the maleic acid was dissolved into 145 g of water. The pressure switch was set to 152 kPa and the pressure regulator upstream of the fuel cell was set to 145 kPa, per the fuel cell manufacture’s specifications. The electronic load controller was set to draw constant current loads of 2 A for ten minutes, followed by ten minutes each of 3 A, and 4 A. Prior to the test, the fuel cell was operated using a facility hydrogen supply to allow it to reach steady-state temperature and operation prior to switching over to hydrogen released from ammonia borane.
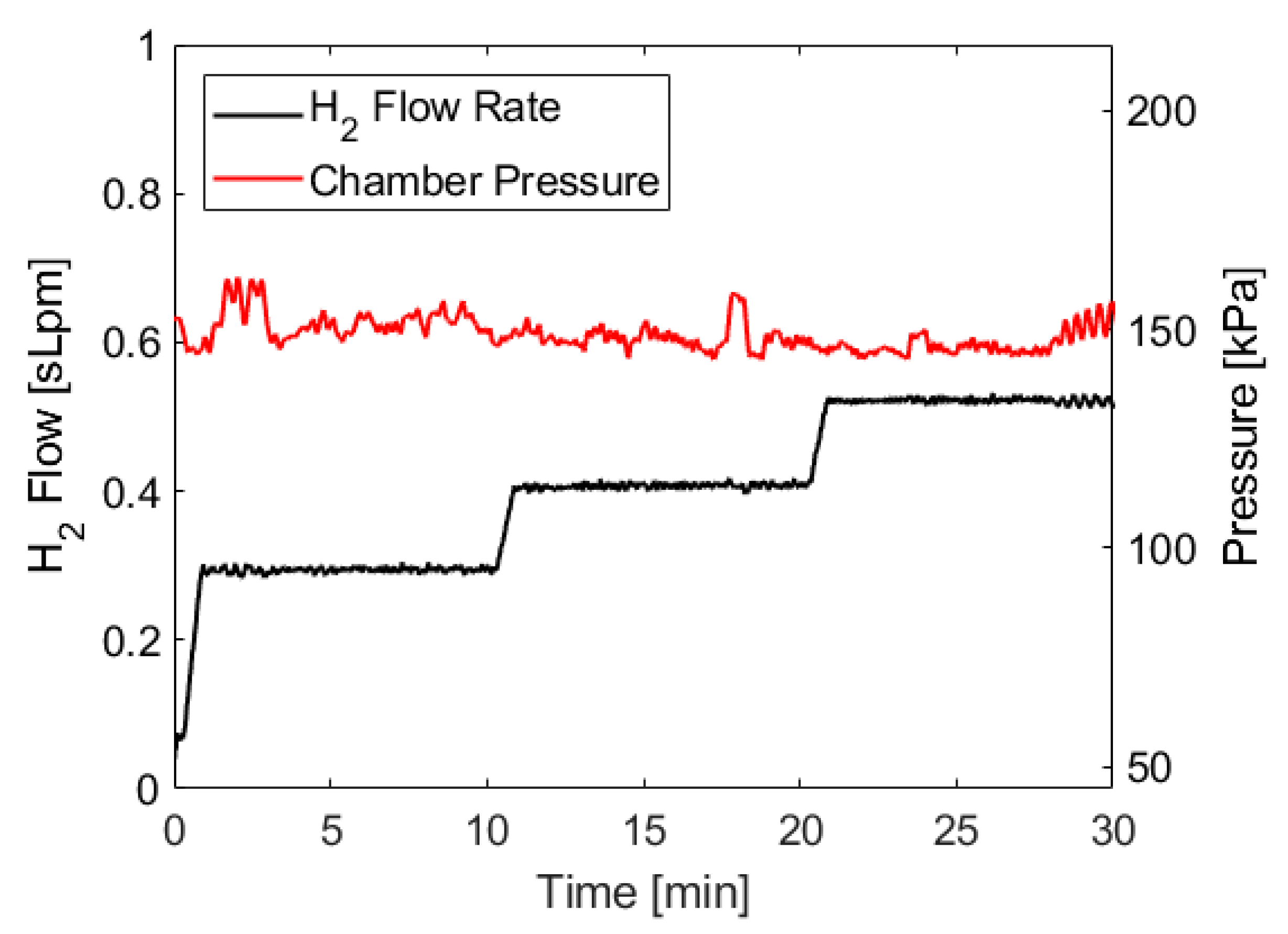
The pressure trace and hydrogen flow rate for the test described above are shown in
Figure 5. Because of the water management strategies employed by the fuel cell (described in
Section 4.4, the raw pressure trace and flow rate data have brief but significant dips and spikes, respectively, so the data shown below has been smoothed across 30 s moving intervals in order to show the average chamber pressure and flow rate and make it easier to identify pressure trends. The 30 s interval averages three cycles of water management events. These smoothed traces are more representative of what would be observed with a fuel cell stack that uses different water management strategies.
Figure 5 shows a relatively consistent pressure trace throughout the test, with notable deviations occurring at the beginning and at the end of the test. Initially, there is a brief pressure spike as the system ramps up to steady-state conditions. This behavior has been observed in all tests conducted with this apparatus, as there is a slight delay between ammonia borane solution being added to the reaction vessel and enough hydrogen being generated to build up sufficient pressure to engage the pressure switch. The duration of the startup transient is dependent on the volume of the reaction chamber and could be shortened by limiting the headspace in the reactor, which would also result in more frequent cycling of the pressure switch. At the end of the test, the ammonia borane solution pumping cycles become longer as the reaction kinetics slow due to decreasing acid concentration, similar to the behavior observed near the endpoint of acid-base titrations. This causes the pump to operate longer during each cycle in order to add enough ammonia borane to compensate for slower reaction kinetics, which leads to more exaggerated pressure fluctuations. Again, this behavior is consistent across all tests we have conducted. However, the duration of the fluctuating period at the end of the test does not appear to scale with the duration of the test, meaning approximately the same duration of increased fluctuations were observed during a 10 min test as a 30 min test.
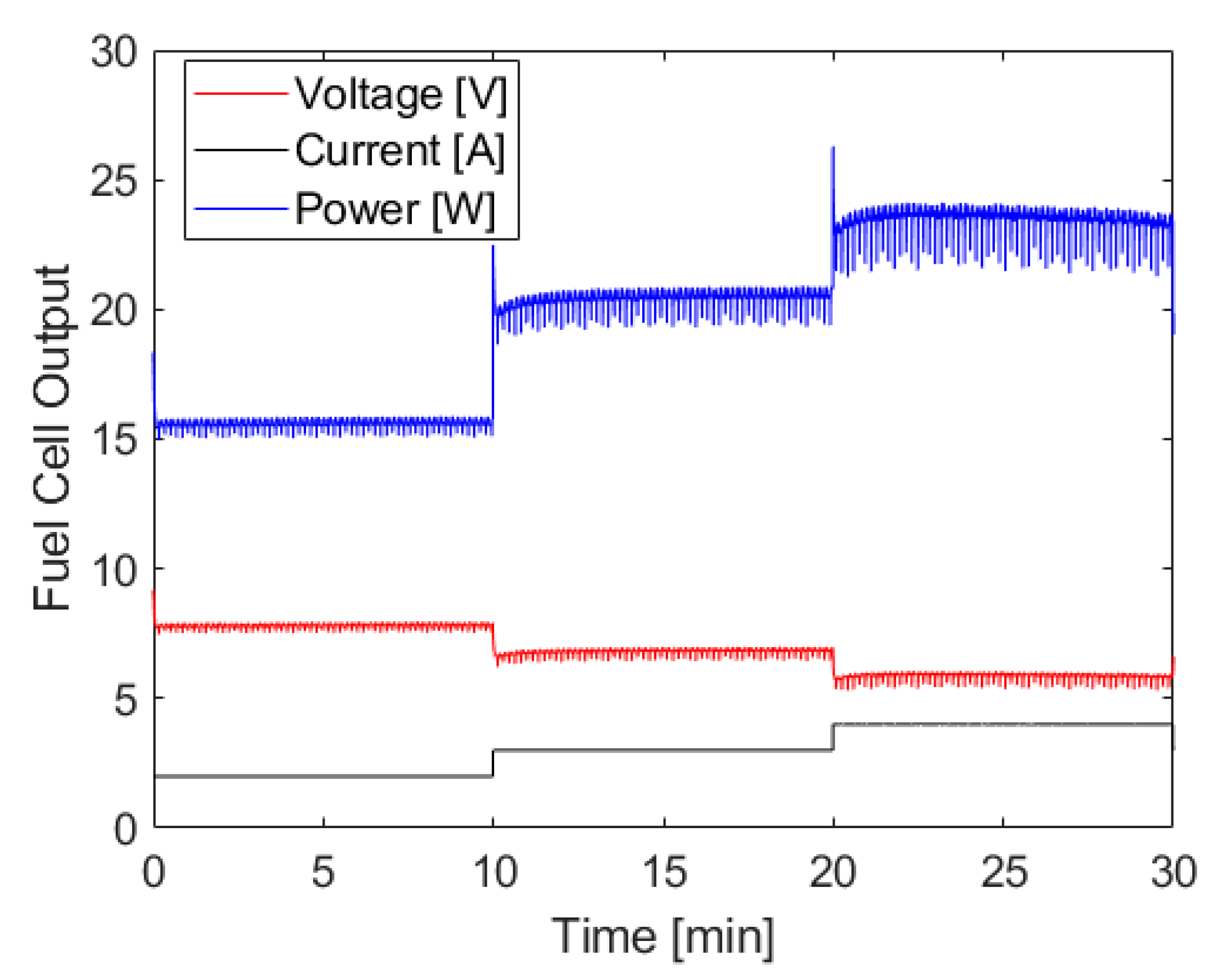
The fuel cell output generated using the hydrogen generation shown above can be seen in
Figure 6, and shows the hydrogen generation system’s ability to power the fuel cell at various loads without an associated increase in chamber pressure. There are brief voltage spikes whenever the load controller shifts from one current setting to another, which is due to the capacitor discharging down to the operating voltage of the fuel cell at the new current level. There are also small fluctuations in voltage and power that are due to the water management strategy of this fuel cell. Outside of these exceptions, the power output at each level is quite constant with only a slight decrease at the tail end of the test which aligns closely with the fluctuations in pressure described above. When averaging the power output across 30 s moving intervals to smooth power drops due to water management events, the standard deviation of power at each step of constant current demand is less than ± 0.1 W.
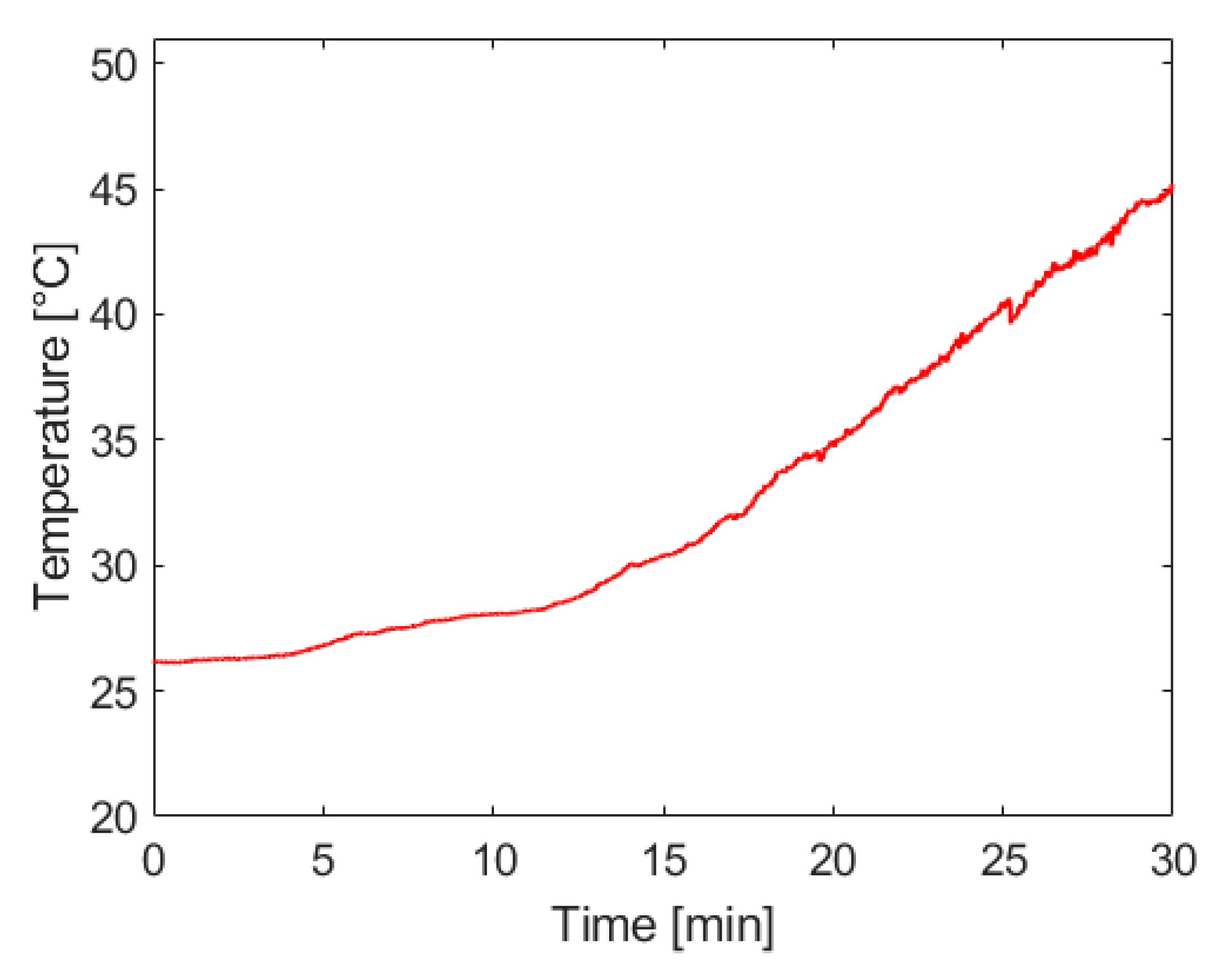
Figure 7 below shows that the rate of temperature rise in the reaction vessel increases with the rate of hydrogen generation, as would be expected from this exothermic reaction. The most significant concerns associated with system temperature rise are the potential of injuring the system operator and an increase of water vapor entrained in the hydrogen being supplied to the fuel cell. Future work on this system will include the introduction of passive heat management techniques that can maintain a safe operating temperature and extend the operational capacity of the system.