Influence of Oxygen-Containing Sulfur Flavor Molecules on the Stability of β-Carotene under UVA Irradiation
Abstract
1. Introduction
2. Results
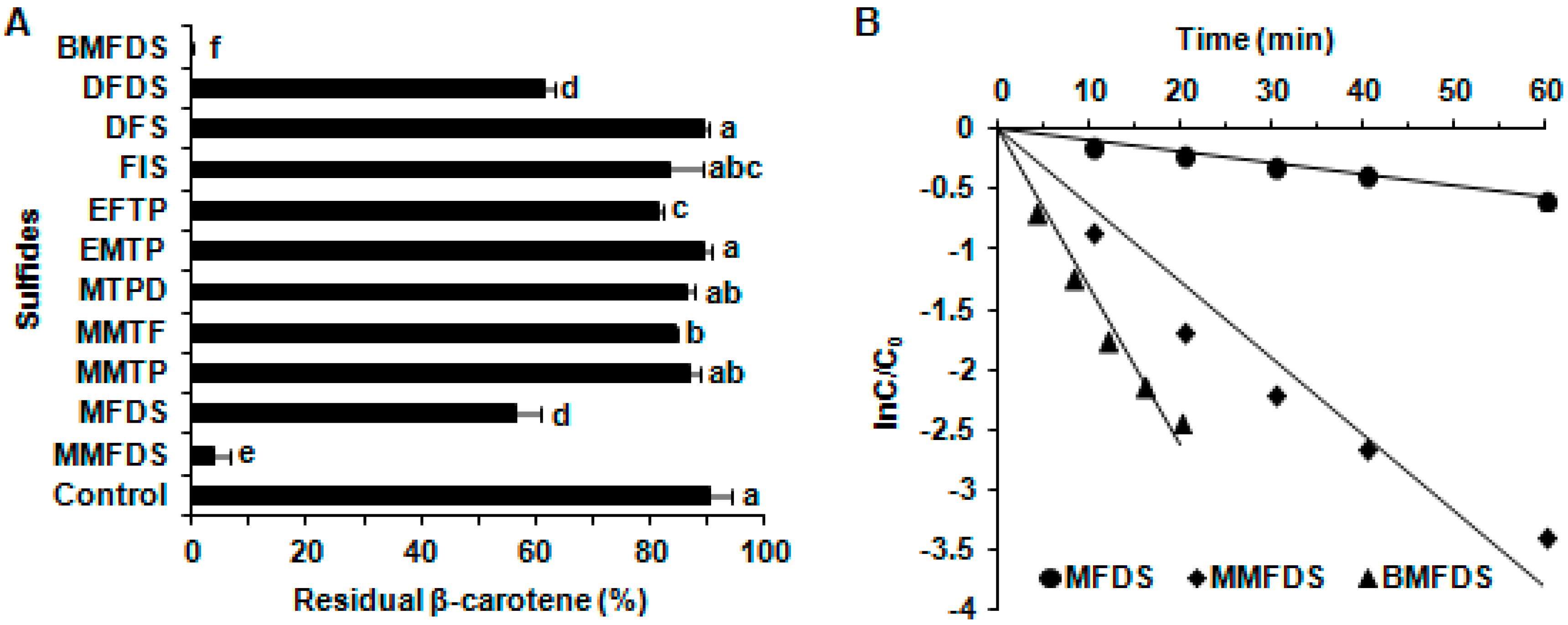
2.1. The Effects of Oxygen-Containing Sulfur Flavor Molecules on β-Carotene Degradation under UVA Irradiation
2.2. The Effects of Furan-Containing Sulfides on β-Carotene Degradation under UVA Irradiation
2.3. Kinetics of β-Carotene Degradation Treated with BMFDS under UVA Irradiation
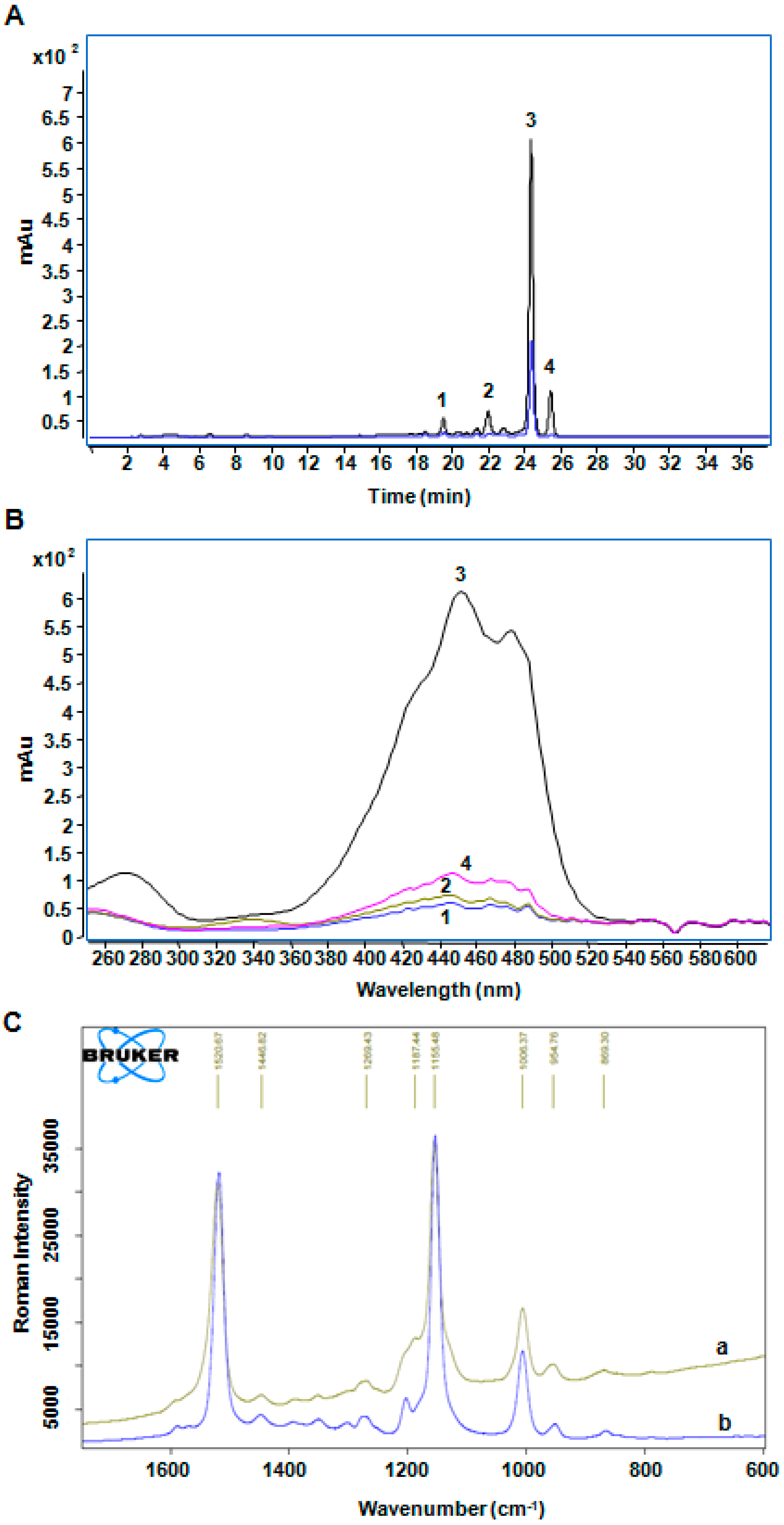
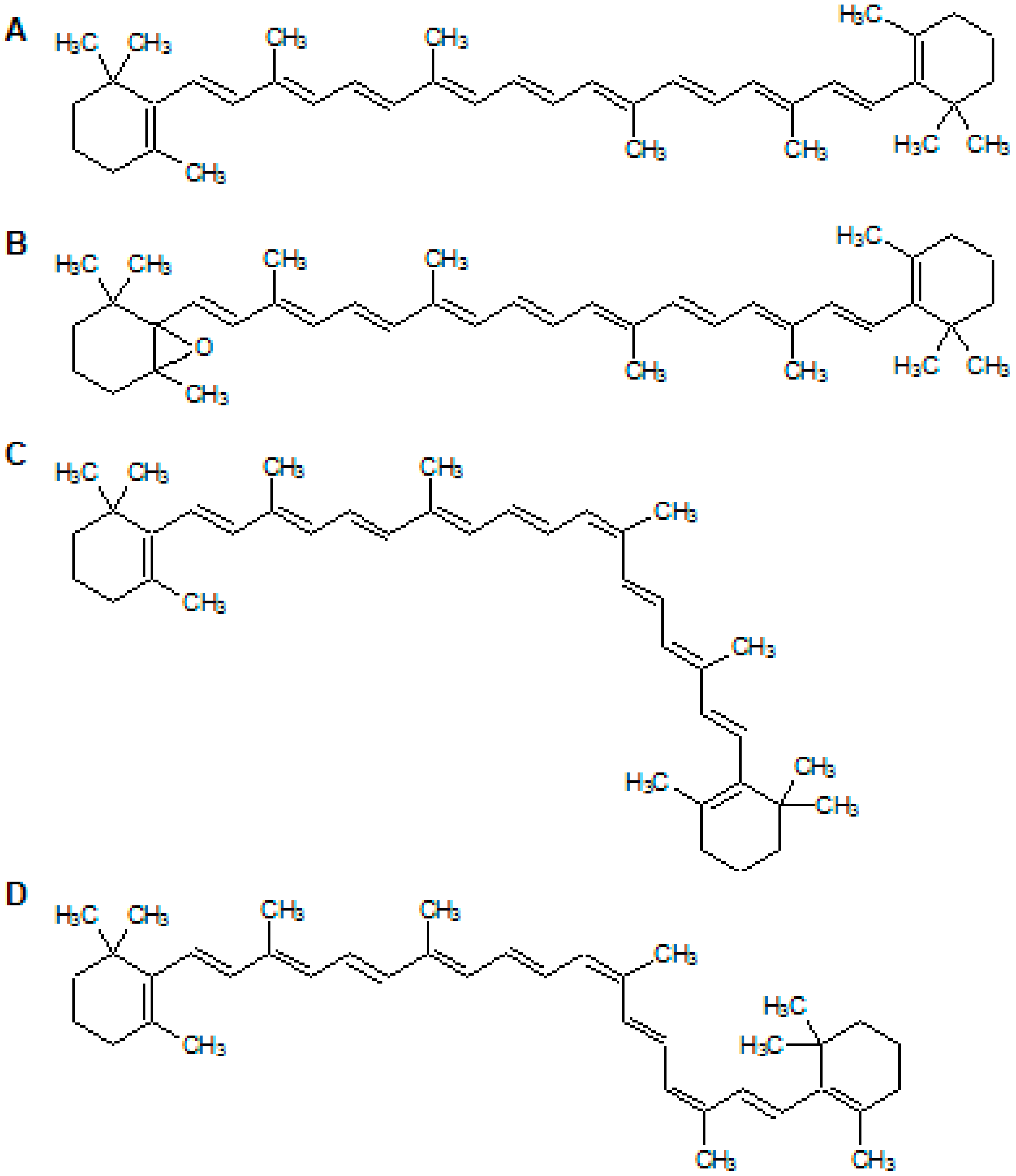
2.4. The Analysis of Photooxidation Products of β-Carotene Treated with BMFDS under UVA Irradiation
3. Discussion
4. Materials and Methods
4.1. Materials and Chemicals
4.2. Preparation of the Model Systems
4.3. Kinetic Analysis of β-Carotene Degradation
4.4. Degradation Kinetics Modeling
4.5. Analysis of β-Carotene Treated with UVA Irradiation and BMFDS
4.6. Determination of Degradation Products by Raman Spectroscopy
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Durante, M.; Lenucci, M.S.; D’Amico, L.; Piro, G.; Mita, G. Effect of drying and co-matrix addition on the yield and quality of supercritical CO2 extracted pumpkin (Cucurbita moschata Duch.) oil. Food Chem. 2014, 148, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, L.; Colle, I.; Buggenhout, S.V.; Palmero, P.; Loey, A.V.; Hendrickx, M. Carotenoid bioaccessibility in fruit-and vegetable-based food products as affected by product (micro) structural characteristics and the presence of lipids: A review. Trends Food Sci. Technol. 2014, 38, 125–135. [Google Scholar] [CrossRef]
- Hou, Z.Q.; Liu, Y.W.; Lei, F.; Gao, Y.X. Investigation into the in vitro release properties of β-carotene in emulsions stabilized by different emulsifiers. LWT Food Sci. Technol. 2014, 59, 867–873. [Google Scholar] [CrossRef]
- Gul, K.; Tak, A.; Singh, A.K.; Singh, P.; Yousuf, B.; Wani, A.A. Chemistry, encapsulation, and health benefits of β-carotene—A review. Cogent. Food Agric. 2015, 1, 1018696. [Google Scholar] [CrossRef]
- Tanaka, T.; Shnimizu, M.; Moriwaki, H. Cancer chemoprevention by carotenoids. Molecules 2012, 17, 3202–3242. [Google Scholar] [CrossRef]
- Huang, Q.R.; Yu, H.L.; Ru, Q.M. Bioavailability and delivery of nutraceuticals using nanotechnology. J. Food Sci. 2010, 75, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Salvia-Trujillo, L.; Qian, C.; Martín-Belloso, O.; McClements, D.J. Influence of particle size on lipid digestion and beta-carotene bioaccessibility in emulsions and nanoemulsions. Food Chem. 2013, 141, 1472–1480. [Google Scholar] [CrossRef]
- Salvia-Trujillo, L.; Qian, C.; Martin-Belloso, O.; McClements, D.J. Modulating β-carotene bioaccessibility by controlling oil composition and concentration in edible nanoemulsions. Food Chem. 2013, 139, 878–884. [Google Scholar] [CrossRef]
- Boon, C.S.; Mcclements, D.J.; Weiss, J.; Decker, E.A. Factors influencing the chemical stability of carotenoids in foods. Crit. Rev. Food Sci. 2010, 50, 515–532. [Google Scholar] [CrossRef]
- Achir, N.; Randrianatoandro, V.A.; Bohuon, P.; Laffargue, A.; Avallone, S. Kinetic study of β-carotene and lutein degradation in oils during heat treatment. Eur. J. Lipid Sci. Technol. 2010, 112, 349–361. [Google Scholar] [CrossRef]
- Guan, Y.; Wu, J.; Zhong, Q. Eugenol improves physical and chemical stabilities of nanoemulsions loaded with β-carotene. Food Chem. 2016, 194, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Ayu, D.F.; Andarwulan, N.; Hariyadi, P.; Purnomo, E.H. Effectof tocopherols, tocotrienols, β-carotene and chlorophyll on the photo-oxidative stability of red palm oil. Food Sci. Biotechnol. 2016, 25, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Q.; Zhong, Q.X. Thermal and UV stability of β-carotene dissolved in peppermint oil microemulsified by sunflower lecithin and Tween 20 blend. Food Chem. 2015, 174, 630–636. [Google Scholar] [CrossRef]
- Heymann, T.; Heinz, P.; Glomb, M.A. Lycopene inhibits the isomerization of β-carotene during quenching of singlet oxygen and free radicals. J. Agric. Food Chem. 2015, 63, 3279–3287. [Google Scholar] [CrossRef]
- Rajendran, V.; Chen, B.H. Isomerization of β-carotene by titanium tetrachloride catalyst. J. Chem. Sci. 2007, 119, 253–258. [Google Scholar] [CrossRef]
- Liu, Y.P.; Chen, H.T.; Yin, D.C.; Sun, B.G. Synthesis and odor evaluation of five new sulfur-containing ester flavor compounds from 4-ethyloctanoic acid. Molecules 2010, 15, 5104–5111. [Google Scholar] [CrossRef] [PubMed]
- Apitz-Castro, R.; Badimon, J.J.; Badimon, L. Effect of ajoene, the major antiplatelet compound from garlic, on platelet thrombus formation. Thromb. Res. 1992, 68, 145–155. [Google Scholar] [CrossRef]
- Naganawa, R.; Iwata, N.; Ishikawa, K.; Fukuda, H.; Fujino, T.; Suzuki, A. Inhibition of microbial growth by ajoene, a sulfur-containing compound derived from garlic. Appl. Environ. Microb. 1996, 62, 4238–4242. [Google Scholar]
- Taylor, P.; Noriega, R.; Farah, C.; Abad, M.J.; Arsenak, M.; Apitz, R. Ajoene inhibits both primary tumor growth and metastasis of B16/BL6 melanoma cells in C57BL/6 mice. Cancer Lett. 2006, 239, 298–304. [Google Scholar] [CrossRef]
- Dirsch, V.M.; Vollmar, A.M. Ajoene, a natural product with nonsteroidal anti-inflammatory drug (NSAID)-like properties. Biochem. Pharmacol. 2001, 61, 587–593. [Google Scholar] [CrossRef]
- Zhang, G.L.; Wu, H.T.; Zhu, B.W.; Shimoishi, Y.; Nakamura, Y.; Murata, Y. Effect of dimethyl sulfides on the induction of apoptosis in human leukemia Jurkat cells and HL-60 cells. Biosci. Biotechnol. Biochem. 2008, 72, 2966–2972. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.L.; Wu, H.T.; Zhu, B.W.; Shimoishi, Y.; Nakamura, Y.; Murata, Y. Induction of apoptosis by beta-carotene and dimethyl tetrasulfide assisted by UVA irradiation in HL-60 cells. Biosci. Biotechnol. Biochem. 2009, 73, 1014–1020. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sun, Y.; Ma, G.; Ye, X.; Kakuda, Y.; Meng, R. Stability of all-trans-beta-carotene under ultrasound treatment in a model system: Effects of different factors, kinetics and newly formed compounds. Ultrason. Sonochem. 2010, 17, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Arita, S.; Ando, S.; Hosoda, H.; Sakaue, K.; Nagata, T.; Murata, Y.; Shimoishi, Y.; Tada, M. Acceleration effect of sulfides on photodegradation of carotenoids by UVA irradiation. Biosci. Biotechnol. Biochem. 2005, 69, 1786–1789. [Google Scholar] [CrossRef] [PubMed]
- Lowe, G.M.; Vlismas, K.; Graham, D.L.; Carail, M.; Caris-Veyrat, C.; Young, A.J. The degradation of (all-E)-β-carotene by cigarette smoke. Free Radic. Res. 2009, 43, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Dan, Q.; Chen, Z.R.; Li, H.R. Effect of heating on solidβ-carotene. Food Chem. 2009, 112, 344–349. [Google Scholar]
- Onsekizoglu, P.; Gökmen, V.; Acar, J. Degradation of β-carotene with the effects of light and sulfur dioxide may be responsible for the formation of white spot in dried apricots. Eur. Food. Res. Technol. 2005, 221, 357–360. [Google Scholar] [CrossRef]
- Spada, J.C.; Noreña, C.P.Z.; Marczak, L.D.F.; Tessaro, I.C. Study on the stability of β-carotene microencapsulated with pinhão (Araucaria angustifolia seeds) starch. Carbohydr. Polym. 2012, 89, 1166–1173. [Google Scholar] [CrossRef]
- Chen, B.H.; Chen, T.M.; Chien, J.T. Kinetic model for studying the isomerization of alpha- and beta-carotene during heating and illumination. J. Agric. Food Chem. 1994, 42, 2391–2397. [Google Scholar] [CrossRef]
- Wei, Z.H.; Gao, Y.X. Physicochemical properties of β-carotene bilayer emulsions coated by milk proteins and chitosan–EGCG conjugates. Food Hydrocoll. 2016, 52, 590–599. [Google Scholar] [CrossRef]
- Zhang, G.L.; Zhu, B.W.; Nakamura, Y.; Shimoishi, Y.; Murata, Y. Structure-dependent photodegradation of carotenoids accelerated by dimethyl tetrasulfide under UVA irradiation. Biosci. Biotechnol. Biochem. 2008, 72, 2176–2183. [Google Scholar] [CrossRef][Green Version]
- Zhang, G.L.; Liang, Y.; Zhu, J.Y.; Jia, Q.; Gan, W.Q.; Sun, L.M.; Hou, H.M. Oxidative stress-mediated antiproliferative effects of furan-containing sulfur flavors in human leukemia Jurkat cells. Food Chem. 2015, 180, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lavelli, V.; Zanoni, B.; Zaniboni, A. Effect of water activity on carotenoid degradation in dehydrated carrots. Food Chem. 2007, 104, 1705–1711. [Google Scholar] [CrossRef]
- Knockaert, G.; Pulissery, S.K.; Lemmens, L.; Van, B.S.; Hendrickx, M.; Van, L.A. Carrot beta-carotene degradation and isomerization kinetics during thermal processing in the presence of oil. J. Agric. Food Chem. 2012, 60, 10312–10319. [Google Scholar] [CrossRef]
- Aparicio-Ruiz, R.; Mínguez-Mosquera, M.I.; Gandul-Rojas, B. Thermal degradation kinetics of lutein, β-carotene and β-cryptoxanthin in virgin olive oils. J. Food Compos. Anal. 2011, 24, 811–820. [Google Scholar] [CrossRef]
- Ahmed, J.; Shivhare, U.S.; Sandhu, K.S. Thermal degradation kinetics of carotenoids and visual color of papaya puree. J. Food Sci. 2002, 67, 2692–2695. [Google Scholar] [CrossRef]
- Saxena, A.; Maity, T.; Raju, P.S.; Bawa, A.S. Degradation kinetics of colour and total carotenoids in jackfruit (Artocarpus heterophyllus) bulb slices during hot air drying. Food Bioprocess Technol. 2012, 5, 672–679. [Google Scholar] [CrossRef]
- Ferreira, J.E.M.; Rodriguez-Amaya, D.B. Degradation of lycopene and β-carotene in model systems and in lyophilized guava during ambient storage: Kinetics, structure, and matrix effects. J. Food Sci. 2008, 73, 589–594. [Google Scholar] [CrossRef]
- Li, D.J.; Xiao, Y.D.; Zhang, Z.Y.; Liu, C. Light-induced oxidation and isomerization of all-trans-β-cryptoxanthin in a model system. J. Photochem. Photobiol. B 2015, 142, 51–58. [Google Scholar] [CrossRef]
- Chen, B.H.; Chen, Y.Y. Stability of chlorophylls and carotenoids in sweet potato leaves during microwave cooking. J. Agric. Food Chem. 1993, 41, 1315–1320. [Google Scholar] [CrossRef]
- Chen, B.H.; Huang, J.H. Degradation and isomerization of chlorophyll a and β-carotene as affected by various heating and illumination treatments. Food Chem. 1998, 62, 299–307. [Google Scholar] [CrossRef]
- Glaser, T.; Lienau, A.; Zeeb, D.; Krucker, M.; Dachtler, M.; Albert, K. Qualitative and quantitative determination of carotenoid stereoisomers in a variety of spinach samples by use of MSPD before HPLC-UV, HPLC-APCI-MS, and HPLC-NMR online coupling. Chromatographia 2003, 57, S19–S25. [Google Scholar] [CrossRef]
- Handelman, G.J.; Kuijk, F.J.G.M.V.; Chatterjee, A.; Krinsky, N.I. Characterization of products formed during the autoxidation of β-carotene. Free Radic. Biol. Med. 1991, 10, 427–437. [Google Scholar] [CrossRef]
- Zeb, A. Oxidation and formation of oxidation products of β-carotene at boiling temperature. Chem. Phys. Lipids 2012, 165, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, A.M.; Carmona, M.; Ordoudi, S.A.; Tsimidou, M.Z.; Alonso, G.L. Kinetics of individual crocetin ester degradation in aqueous extracts of saffron (Crocus sativus L.) upon thermal treatment in the dark. J. Agric. Food Chem. 2008, 56, 1627–1637. [Google Scholar] [CrossRef]
- Santos, J.; Herrero, M.; Mendiola, J.A.; Oliva-Teles, M.T.; Ibáñez, E.; Delerue-Matos, C.; Oliveira, M.B.P.P. Assessment of nutritional and metabolic profiles of pea shoots: The new ready-to-eat baby-leaf vegetable. Food Res. Int. 2014, 58, 105–111. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| FEMA Number | Name | Abbreviation | Structure |
|---|---|---|---|
| 3573 | Methyl (2-Methyl-3-furyl) disulfide | MMFDS |  |
| 3362 | Methyl furfuryl disulfide | MFDS | |
| 2720 | Methyl 3-methylthiopropionate | MMTP | |
| 3949 | 2-Methyl-3-(methylthio) furan | MMTF | |
| 2747 | 3-(Methylthio) propionaldehyde | MTPD | |
| 3343 | Ethyl 3-methylthiopropionate | EMTP | |
| 3674 | Ethyl 3-(furfurylthio) propionate | EFTP | |
| 3161 | Furfuryl isopropyl sulfide | FIS | |
| 3238 | Difurfuryl sulfide | DFS | |
| 3146 | Difurfuryl disulfide | DFDS | |
| 3259 | Bis (2-methyl-3-furyl) disulfide | BMFDS |
| Sulfides | K (min−1) | R2 | t1/2 (min) |
|---|---|---|---|
| MFDS | 0.0095 | 0.9959 | 72.96 |
| MMFDS | 0.0633 | 0.9356 | 10.95 |
| BMFDS | 0.131 | 0.9720 | 5.29 |
| R Zero-Order (c) | R First-Order (ln c) | R Second-Order (1/c) | k (min−1) | R2 | |
|---|---|---|---|---|---|
| Control | 0.9468 | 0.9512 | 0.9554 | 0.0012 | 0.9785 |
| BMFDS | 0.6376 | 0.9766 | 0.9141 | 0.1879 | 0.9537 |
| Peak | Retention Time (min) | Molecular Ion, Formula | Isomer | λ (nm) | Q-Ratio | ||
|---|---|---|---|---|---|---|---|
| Found | Reported | Found | Reported | ||||
| 1 | 19.467 | 553, C18H57O | All-trans-5,6-expoxy-β-carotene | 446 | 444 | ||
| 2 | 21.953 | 537, C18H57 | 13-cis-β-carotene | 340 446 | 340 445 | 0.41 | 0.41 |
| 3 | 24.319 | 537, C18H57 | All-trans-β-carotene | 451 | 455 | ||
| 4 | 25.399 | 537, C18H57 | 9,13-di-cis-β-carotene | 350 446 | 335 439 | 0.16 | 0.16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, G.-L.; Wu, H.-Y.; Liang, Y.; Song, J.; Gan, W.-Q.; Hou, H.-M. Influence of Oxygen-Containing Sulfur Flavor Molecules on the Stability of β-Carotene under UVA Irradiation. Molecules 2019, 24, 318. https://doi.org/10.3390/molecules24020318
Zhang G-L, Wu H-Y, Liang Y, Song J, Gan W-Q, Hou H-M. Influence of Oxygen-Containing Sulfur Flavor Molecules on the Stability of β-Carotene under UVA Irradiation. Molecules. 2019; 24(2):318. https://doi.org/10.3390/molecules24020318
Chicago/Turabian StyleZhang, Gong-Liang, Hong-Yan Wu, Ying Liang, Jie Song, Wei-Qi Gan, and Hong-Man Hou. 2019. "Influence of Oxygen-Containing Sulfur Flavor Molecules on the Stability of β-Carotene under UVA Irradiation" Molecules 24, no. 2: 318. https://doi.org/10.3390/molecules24020318
APA StyleZhang, G.-L., Wu, H.-Y., Liang, Y., Song, J., Gan, W.-Q., & Hou, H.-M. (2019). Influence of Oxygen-Containing Sulfur Flavor Molecules on the Stability of β-Carotene under UVA Irradiation. Molecules, 24(2), 318. https://doi.org/10.3390/molecules24020318






