Preparation and Antioxidant Activity of Ethyl-Linked Anthocyanin-Flavanol Pigments from Model Wine Solutions
Abstract
:1. Introduction
2. Results and Discussion
2.1. HSCCC Separation of Anthocyanins and (–)-Epicatechin
2.2. Dynamic Monitoring of Condensation Reactions between Anthocyanins and (–)-Epicatechin
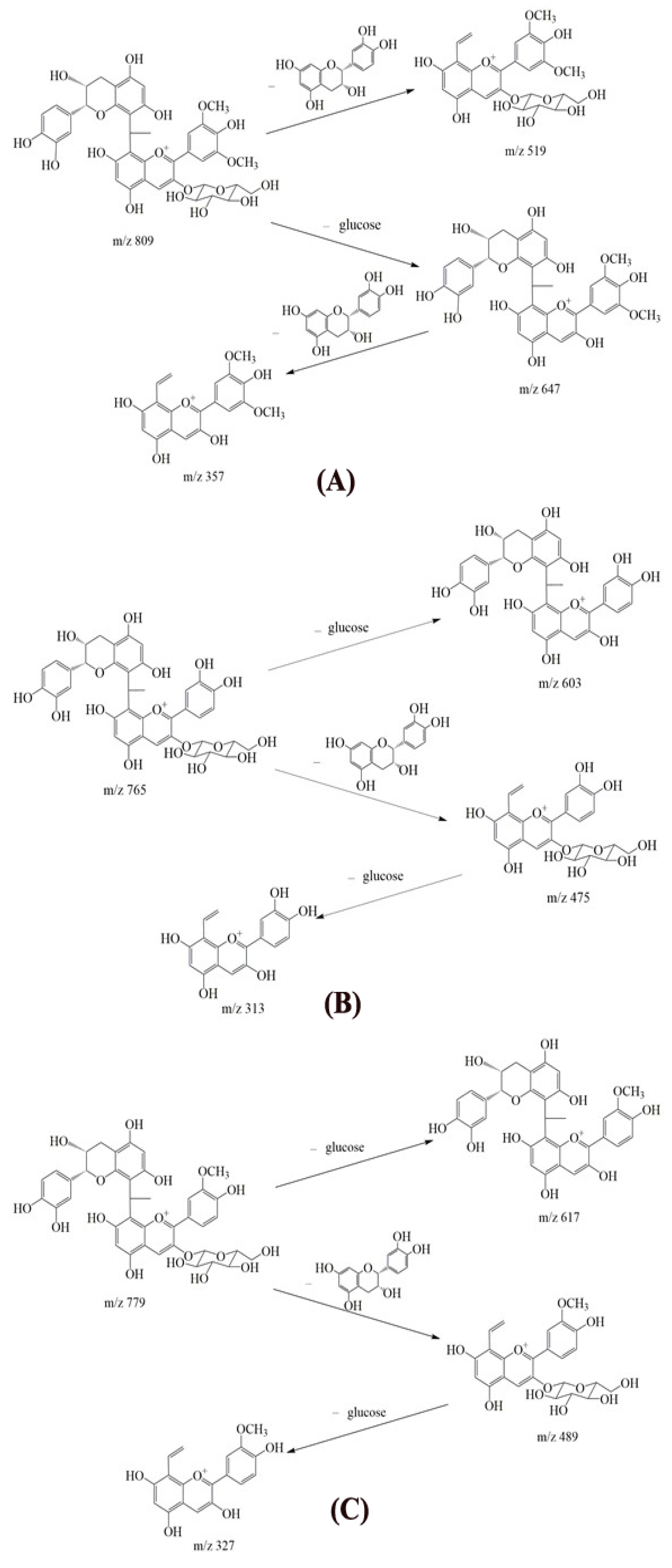
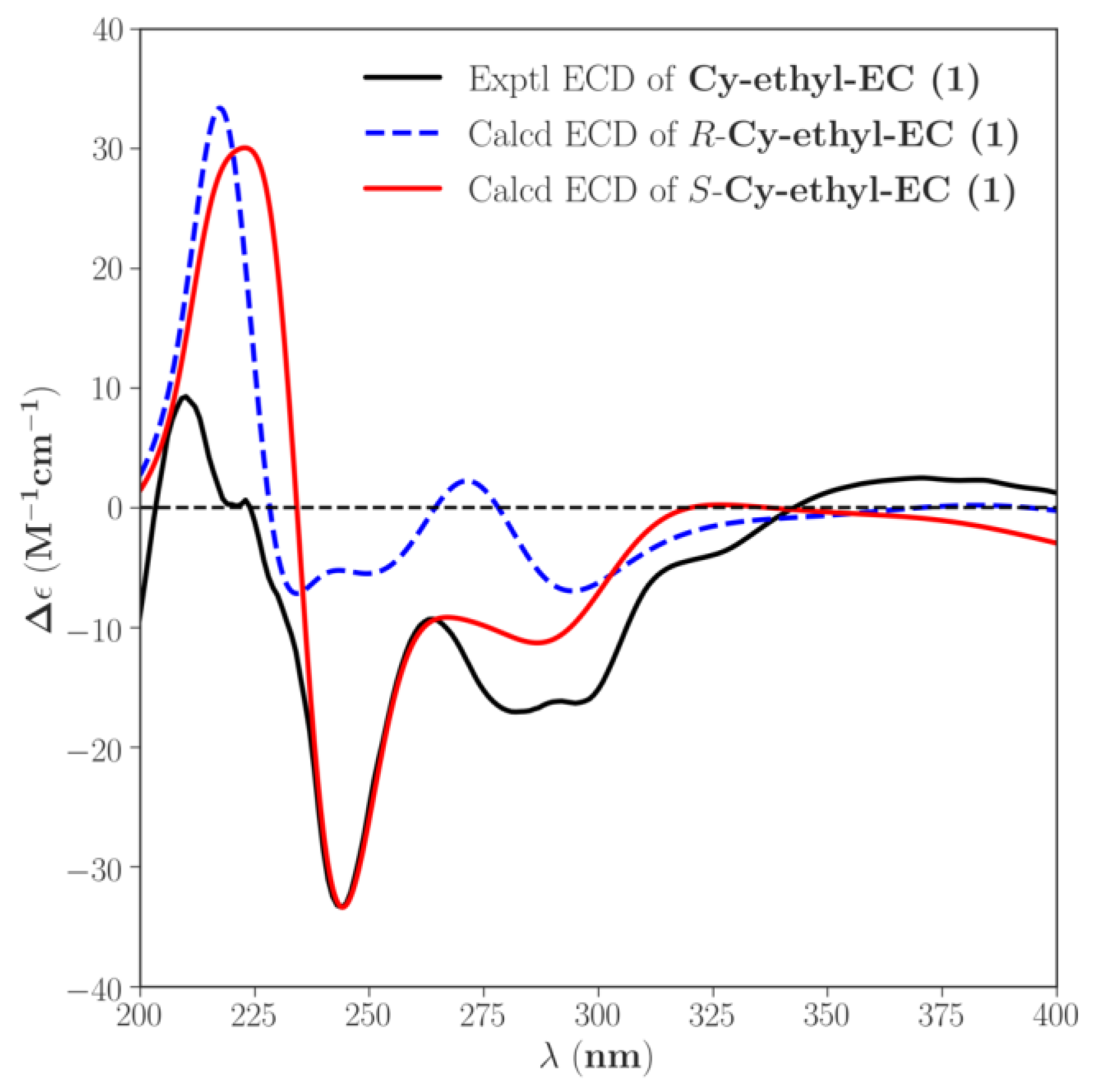
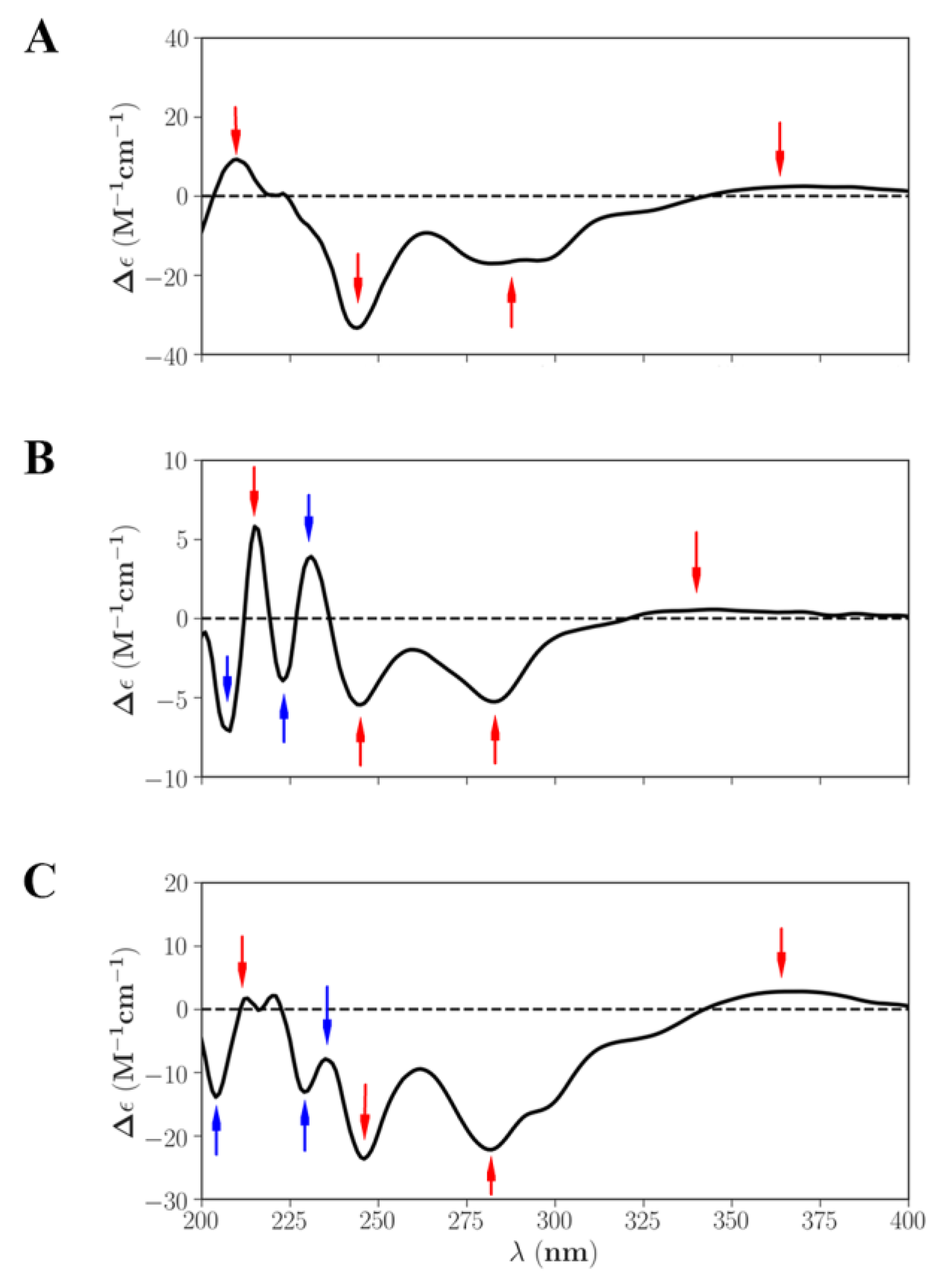
2.3. Structural Identification
2.4. Isolation of Individual Ethyl-Linked Anthocyanin-Flavanol Pigments by Preparative HPLC
2.5. Antioxidant Activity
3. Materials and Methods
3.1. Chemicals and Materials
3.2. Preparation of Anthocyanins and Flavanols by HSCCC
3.3. Model Wine Solution
3.4. Optimization of the Condensation Reaction between Anthocyanins and Flavanols Mediated by Acetaldehyde
3.5. HPLC-DAD Analysis
3.6. Isolation and Purification of Ethyl-Linked Anthocyanin-Flavanol Pigments
3.7. MS Analysis
3.8. ECD Analysis
3.8.1. Circular Dichroism (CD) Spectra
3.8.2. Conformational Analysis
3.8.3. ECD Calculation
3.9. Antioxidant Activity
3.9.1. DPPH Assay
3.9.2. ABTS Assay
3.9.3. FRAP Assay
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Timberlake, C.F.; Bridle, P. Interactions between anthocyanins, phenolic compounds, and acetaldehyde and their significance in red wines. Am. J. Enol. Viticult. 1976, 27, 97–105. [Google Scholar]
- Haslam, E. In vino veritas: Oligomeric procyanidins and the ageing of red wines. Phytochemistry 1980, 19, 2577–2582. [Google Scholar]
- Casassa, L.F.; Keller, M.; Harbertson, J.F. Regulated deficit irrigation alters anthocyanins, tannins and sensory properties of cabernet sauvignon grapes and wines. Molecules 2015, 20, 7820–7844. [Google Scholar] [CrossRef] [PubMed]
- Preys, S.; Mazerolles, G.; Courcoux, P.; Samson, A.; Fischer, U.; Hanafi, M.; Bertrand, D.; Cheynier, V. Relationship between polyphenolic composition and some sensory properties in red wines using multiway analyses. Anal. Chim. Acta 2006, 563, 126–136. [Google Scholar] [CrossRef]
- Ricardo-da-Silva, J.M.; Darmon, N.; Fernandez, Y.; Mitjavila, S. Oxygen free radical scavenger capacity in aqueous models of different procyanidins from grape seeds. J. Agric. Food Chem. 1991, 39, 1549–1552. [Google Scholar] [CrossRef]
- Zhao, G.; Gao, H.; Jie, Q.; Lu, W.; Wei, X. The molecular mechanism of protective effects of grape seed proanthocyanidin extract on reperfusion arrhythmias in rats in vivo. Biol. Pharm. Bull. 2010, 33, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Kruger, M.J.; Davies, N.; Myburgh, K.H.; Lecour, S. Proanthocyanidins, anthocyanins and cardiovascular diseases. Food Res. Int. 2014, 59, 41–52. [Google Scholar] [CrossRef]
- Renaud, S.; de Lorgeril, M. Wine, alcohol, platelets, and the french paradox for coronary heart disease. Lancet 1992, 339, 1523–1526. [Google Scholar] [CrossRef]
- Aviram, M.; Fuhrman, B. Polyphenolic flavonoids inhibit macrophage-mediated oxidation of LDL and attenuate atherogenesis. Atherosclerosis 1998, 137, S45–S50. [Google Scholar] [CrossRef]
- Spranger, I.; Sun, B.; Mateus, A.M.; Freitas, V.D.; Ricardo-da-Silva, J.M. Chemical characterization and antioxidant activities of oligomeric and polymeric procyanidin fractions from grape seeds. Food Chem. 2008, 108, 519–532. [Google Scholar] [CrossRef] [PubMed]
- Cueva, C.; Gil-Sanchez, I.; Ayuda-Duran, B.; Gonzalez-Manzano, S.; Gonzalez-Paramas, A.M.; Santos-Buelga, C.; Bartolome, B.; Moreno-Arribas, M.V. An integrated view of the effects of wine polyphenols and their relevant metabolites on gut and host health. Molecules 2017, 22, 99. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Leandro, M.C.; de Freitas, V.; Spranger, M.I. Fractionation of red wine polyphenols by solid phase extraction and liquid chromatography. J. Chromatogr. A 2006, 1128, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Boulton, R. The copigmentation of anthocyanins and its role in the color of red wine: A critical review. Am. J. Enol. Viticult. 2001, 52, 67–87. [Google Scholar]
- Schwarz, M.; Picazo-Bacete, J.J.; Winterhalter, P.; Hermosín-Gutiérrez, I. Effect of copigments and grape cultivar on the color of red wines fermented after the addition of copigments. J. Agric. Food Chem. 2005, 53, 8372–8381. [Google Scholar] [CrossRef] [PubMed]
- Nagel, C.W.; Wulf, L.W. Changes in the anthocyanins, flavonoids and hydroxycinnamic acid esters during fermentation and aging of merlot and cabernet sauvignon. Am. J. Enol. Viticult. 1979, 30, 111–116. [Google Scholar]
- Somers, T.C.; Michael, E.E. Grape pigment phenomena: Interpretation of major colour losses during vinification. J. Sci. Food Agric. 1979, 30, 623–633. [Google Scholar] [CrossRef]
- Es-Safi, N.E.; Cheynier, V.; Moutounet, M. Interactions between cyanidin 3-O-glucoside and furfural derivatives and their impact on food color changes. J. Agric. Food Chem. 2002, 50, 5586–5595. [Google Scholar] [CrossRef] [PubMed]
- Eglinton, J.; Griesser, M.; Henschke, P.; Kwiatkowski, M.; Parker, M.; Herderich, M. Yeast-mediated formation of pigmented polymers in red wine. In Red Wine Color; American Chemical Society: Washington, DC, USA, 2004; Volume 886, pp. 7–21. [Google Scholar]
- Es-Safi, N.E.; Fulcrand, H.; Cheynier, V.; Moutounet, M. Studies on the acetaldehyde-induced condensation of (−)-epicatechin and malvidin 3-O-glucoside in a model solution system. J. Agric. Food Chem. 1999, 47, 2096–2102. [Google Scholar] [CrossRef] [PubMed]
- Dueñas, M.; Fulcrand, H.; Cheynier, V. Formation of anthocyanin–flavanol adducts in model solutions. Anal. Chim. Acta 2006, 563, 15–25. [Google Scholar] [CrossRef]
- Salas, E.; Fulcrand, H.; Emmanuelle Meudec, A.; Cheynier, V. Reactions of anthocyanins and tannins in model solutions. J. Agric. Food Chem. 2003, 51, 7951. [Google Scholar] [CrossRef] [PubMed]
- Pissarra, J.; Mateus, N.; Rivas-Gonzalo, J.; Santos Buelga, C.; De Freitas, V. Reaction between malvidin 3-glucoside and (+)-catechin in model solutions containing different aldehydes. J. Food Sci. 2003, 68, 476–481. [Google Scholar] [CrossRef]
- Drinkine, J.; Lopes, P.; Kennedy, J.A.; Teissedre, P.L.; Saucier, C. Ethylidene-bridged flavan-3-ols in red wine and correlation with wine age. J. Agric. Food Chem. 2007, 55, 6292–6299. [Google Scholar] [CrossRef] [PubMed]
- Cheynier, V. Grape polyphenols and their reactions in wine. In Polyphenols; Martens, S., Treutter, T., Forkmann, G., Eds.; Freising-Weihenstephan: Bavaria, Germany, 2002; pp. 1–14. [Google Scholar]
- Escott, C.; Del Fresno, J.M.; Loira, I.; Morata, A.; Tesfaye, W.; González, M.D.C.; Suárez-Lepe, J.A. Formation of polymeric pigments in red wines through sequential fermentation of flavanol-enriched musts with non-Saccharomyces yeasts. Food Chem. 2018, 239, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Bindon, K.A.; McCarthy, M.G.; Smith, P.A. Development of wine colour and non-bleachable pigments during the fermentation and ageing of (Vitis vinifera L. cv.) cabernet sauvignon wines differing in anthocyanin and tannin concentration. LWT Food Sci. Technol. 2014, 59, 923–932. [Google Scholar] [CrossRef]
- Kunsági-Máté, S.; Szabó, K.; Nikfardjam, M.P.; Kollár, L. Determination of the thermodynamic parameters of the complex formation between malvidin-3-O-glucoside and polyphenols. Copigmentation effect in red wines. J. Biochem. biophys. Meth. 2006, 69, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Bindon, K.; Kassara, S.; Hayasaka, Y.; Schulkin, A.; Smith, P. Properties of wine polymeric pigments formed from anthocyanin and tannins differing in size distribution and subunit composition. J. Agric. Food Chem. 2014, 62, 11582–11593. [Google Scholar] [CrossRef] [PubMed]
- Hayasaka, Y.; Birse, M.; Eglinton, J.; Herderich, M. The effect of saccharomyces cerevisiae and saccharomyces bayanus yeast on colour properties and pigment profiles of a cabernet sauvignon red wine. Aust. J. Grape Wine Res. 2007, 13, 176–185. [Google Scholar] [CrossRef]
- Somers, T.C. The polymeric nature of wine pigments. Phytochemistry 1971, 10, 2175–2186. [Google Scholar] [CrossRef]
- Sun, B.; Spranger, M.I. Changes in phenolic composition of tinta miúda red wines after 2 years of ageing in bottle: Effect of winemaking technologies. Eur. Food Res. Technol. 2005, 221, 305–312. [Google Scholar] [CrossRef]
- Damianaki, A.; Bakogeorgou, E.; Kampa, M.; Notas, G.; Hatzoglou, A.; Panagiotou, S.; Gemetzi, C.; Kouroumalis, E.; Martin, P.M.; Castanas, E. Potent inhibitory action of red wine polyphenols on human breast cancer cells. J. Cell Biochem. 2000, 78, 429–441. [Google Scholar] [CrossRef]
- Pinder, R.M.; Sandler, M. Alcohol, wine and mental health: Focus on dementia and stroke. J. Psychopharmacol. 2004, 18, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Tresserra-Rimbau, A.; Estruch, R.; Martinez-Gonzalez, M.A.; Medina-Remon, A.; Castaner, O.; Corella, D.; Salas-Salvado, J.; Lamuela-Raventos, R.M. Effects of polyphenol, measured by a biomarker of total polyphenols in urine, on cardiovascular risk factors after a long-term follow-up in the predimed study. Oxid. Med. Cell. Longev. 2016, 2016, 2572606. [Google Scholar] [CrossRef] [PubMed]
- Greyling, A.; Bruno, R.M.; Draijer, R.; Mulder, T.; Thijssen, D.H.J.; Taddei, S.; Virdis, A.; Ghiadoni, L. Effects of wine and grape polyphenols on blood pressure, endothelial function and sympathetic nervous system activity in treated hypertensive subjects. J. Funct. Foods 2016, 27, 448–460. [Google Scholar] [CrossRef]
- Giovinazzo, G.; Grieco, F. Functional properties of grape and wine polyphenols. Plant Food. Hum. Nutr. 2015, 70, 454–462. [Google Scholar] [CrossRef] [PubMed]
- German, J.B.; Walzem, R.L. The health benefits of wine. Annu. Rev. Nutr. 2000, 20, 561–593. [Google Scholar] [CrossRef] [PubMed]
- Radovanovic, B.; Radovanovic, A. Free radical scavenging activity and anthocyanin profile of cabernet sauvignon wines from the balkan region. Molecules 2010, 15, 4213–4226. [Google Scholar] [CrossRef] [PubMed]
- Cavallini, G.; Straniero, S.; Donati, A.; Bergamini, E. Resveratrol requires red wine polyphenols for optimum antioxidant activity. J. Nutr. Health Aging 2016, 20, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Vinayagam, R.; Jayachandran, M.; Xu, B. Antidiabetic effects of simple phenolic acids: A comprehensive review. Phytother. Res. 2016, 30, 184–199. [Google Scholar] [CrossRef] [PubMed]
- Boam, T. Anti-androgenic effects of flavonols in prostate cancer. ecancermedicalscience 2015, 9, 585. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sun, B. Grape and wine polymeric polyphenols: Their importance in enology. Crit. Rev. Food Sci. 2017, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Brouillard, R. Flavonoids and flower colour. In The flavonoids; Harborne, J.B., Ed.; Chapman & Hall: London, UK, 1988; pp. 525–538. [Google Scholar]
- Burtch, C.E.; Mansfield, A.K.; Manns, D.C. Reaction kinetics of monomeric anthocyanin conversion to polymeric pigments and their significance to color in interspecific hybrid wines. J. Agric. Food Chem. 2017, 65, 6379–6386. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Liang, N.N.; Mu, L.; Pan, Q.H.; Wang, J.; Reeves, M.J.; Duan, C.Q. Anthocyanins and their variation in red wines I. Monomeric anthocyanins and their color expression. Molecules 2012, 17, 1571–1601. [Google Scholar] [CrossRef] [PubMed]
- Dipalmo, T.; Crupi, P.; Pati, S.; Clodoveo, M.; Luccia, A. Studying the evolution of anthocyanin-derived pigments in a typical red wine of southern Italy to assess its resistance to aging. LWT Food Sci. Technol. 2016, 71, 1–9. [Google Scholar] [CrossRef]
- Sun, B.; Fernandes, T.A.; Spranger, M.I. A new class of anthocyanin-procyanidin condensation products detected in red wine by electrospray ionization multi-stage mass spectrometry analysis. Rapid Commun. Mass Spectrom. 2010, 24, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Boido, E.; Alcalde-Eon, C.; Carrau, F.; Dellacassa, E.; Rivas-Gonzalo, J.C. Aging effect on the pigment composition and color of Vitis vinifera L. cv. Tannat wines. Contribution of the main pigment families to wine color. J. Agric. Food Chem. 2006, 54, 6692–6704. [Google Scholar] [CrossRef] [PubMed]
- Pati, S.; Losito, I.; Gambacorta, G.; La Notte, E.; Palmisano, F.; Zambonin, P.G. Simultaneous separation and identification of oligomeric procyanidins and anthocyanin-derived pigments in raw red wine by HPLC-UV-ESI-MSn. J. Mass Spectrom. 2006, 41, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Vega, D.; Gomez-Alonso, S.; Hermosin-Gutierrez, I. Identification, content and distribution of anthocyanins and low molecular weight anthocyanin-derived pigments in Spanish commercial red wines. Food Chem. 2014, 158, 449–458. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Liang, N.N.; Mu, L.; Pan, Q.H.; Wang, J.; Reeves, M.J.; Duan, C.Q. Anthocyanins and their variation in red wines. II. Anthocyanin derived pigments and their color evolution. Molecules 2012, 17, 1483–1519. [Google Scholar] [CrossRef] [PubMed]
- Nugroho, A.E.; Morita, H. Circular dichroism calculation for natural products. J. Nat. Med. 2014, 68, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Neves, A.C.; Fernandes, T.A.; Fernandes, A.L.; Mateus, N.; De Freitas, V.; Leandro, C.; Spranger, M.I. Evolution of phenolic composition of red wine during vinification and storage and its contribution to wine sensory properties and antioxidant activity. J. Agric. Food Chem. 2011, 59, 6550–6557. [Google Scholar] [CrossRef] [PubMed]
- Lingua, M.S.; Fabani, M.P.; Wunderlin, D.A.; Baroni, M.V. In vivo antioxidant activity of grape, pomace and wine from three red varieties grown in argentina: Its relationship to phenolic profile. J. Funct. Foods 2016, 20, 332–345. [Google Scholar] [CrossRef]
- Dumitriu, D.; Peinado, R.A.; Peinado, J.; de Lerma, N.L. Grape pomace extract improves the in vitro and in vivo antioxidant properties of wines from sun light dried Pedro Ximénez grapes. J. Funct. Foods 2015, 17, 380–387. [Google Scholar] [CrossRef]
- Li, D.; Zhang, Y.; Liu, Y.; Sun, R.; Xia, M. Purified anthocyanin supplementation reduces dyslipidemia, enhances antioxidant capacity, and prevents insulin resistance in diabetic patients. J. Nutr. 2015, 145, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, L.; Cui, Y.; Zhang, S.; Sun, B. Separation and purification of polyphenols from red wine extracts using high speed counter current chromatography. J. Chromatogr. B 2017, 1054, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Kan, L.; Nie, S.-P.; Hu, J.; Liu, Z.; Xie, M. Antioxidant activities and anthocyanins composition of seed coats from twenty-six kidney bean cultivars. J. Funct. Foods 2016, 26, 622–631. [Google Scholar] [CrossRef]
- Kahkonen, M.P.; Heinonen, M. Antioxidant activity of anthocyanins and their aglycons. J. Agric. Food Chem. 2003, 51, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, J.; Oliveira, J.; Cruz, L.; Teixeira, N.; Bras, N.F.; De Freitas, V.; Mateus, N. Antioxidant features of red wine pyranoanthocyanins: Experimental and theoretical approaches. J. Agric. Food Chem. 2014, 62, 7002–7009. [Google Scholar] [CrossRef] [PubMed]
- Muselík, J.; García-Alonso, M.P.; Martín-López, M.; Zemlicka, M.; Rivas-Gonzalo, J. Measurement of antioxidant activity of wine catechins, procyanidins, anthocyanins and pyranoanthocyanins. Int. J. Mol. Sci. 2007, 8, 797–809. [Google Scholar] [CrossRef]
- Shon, M.Y.; Lee, J.; Choi, J.H.; Choi, S.Y.; Nam, S.H.; Seo, K.I.; Lee, S.W.; Sung, N.J.; Park, S.K. Antioxidant and free radical scavenging activity of methanol extract of chungkukjang. J. Food Compos. Anal. 2007, 20, 113–118. [Google Scholar] [CrossRef]
- Azevedo, J.; Fernandes, I.; Faria, A.; Oliveira, J.; Fernandes, A.; de Freitas, V.; Mateus, N. Antioxidant properties of anthocyanidins, anthocyanidin-3-glucosides and respective portisins. Food Chem. 2010, 119, 518–523. [Google Scholar] [CrossRef]
- Ali, H.M.; Almagribi, W.; Al-Rashidi, M.N. Antiradical and reductant activities of anthocyanidins and anthocyanins, structure–activity relationship and synthesis. Food Chem. 2016, 194, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Li, L.X.; Zhang, S.T.; Cui, Y.; Li, Y.Y.; Luo, L.X.; Zhou, P.Y.; Sun, B.S. Preparative separation of cacao bean procyanidins by high-speed counter-current chromatography. J. Chromatogr. B 2016, 1036, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Santos, C.P.; Leandro, M.C.; De Freitas, V.; Spranger, M.I. High-performance liquid chromatography/electrospray ionization mass spectrometric characterization of new products formed by the reaction between flavanols and malvidin 3-glucoside in the presence of acetaldehyde. Rapid Commun. Mass Spectrom. 2007, 21, 2227–2236. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Barradas, T.; Leandro, C.; Santos, C.; Spranger, I. Formation of new stable pigments from condensation reaction between malvidin 3-glucoside and (−)-epicatechin mediated by acetaldehyde: Effect of tartaric acid concentration. Food Chem. 2008, 110, 344–351. [Google Scholar] [CrossRef] [PubMed]
- O’Boyle, N.M.; Vandermeersch, T.; Flynn, C.J.; Maguire, A.R.; Hutchison, G.R. Confab—Systematic generation of diverse low-energy conformers. J. Cheminform. 2011, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Bruhn, T.; Schaumloffel, A.; Hemberger, Y.; Bringmann, G. Specdis: Quantifying the comparison of calculated and experimental electronic circular dichroism spectra. Chirality 2013, 25, 243–249. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
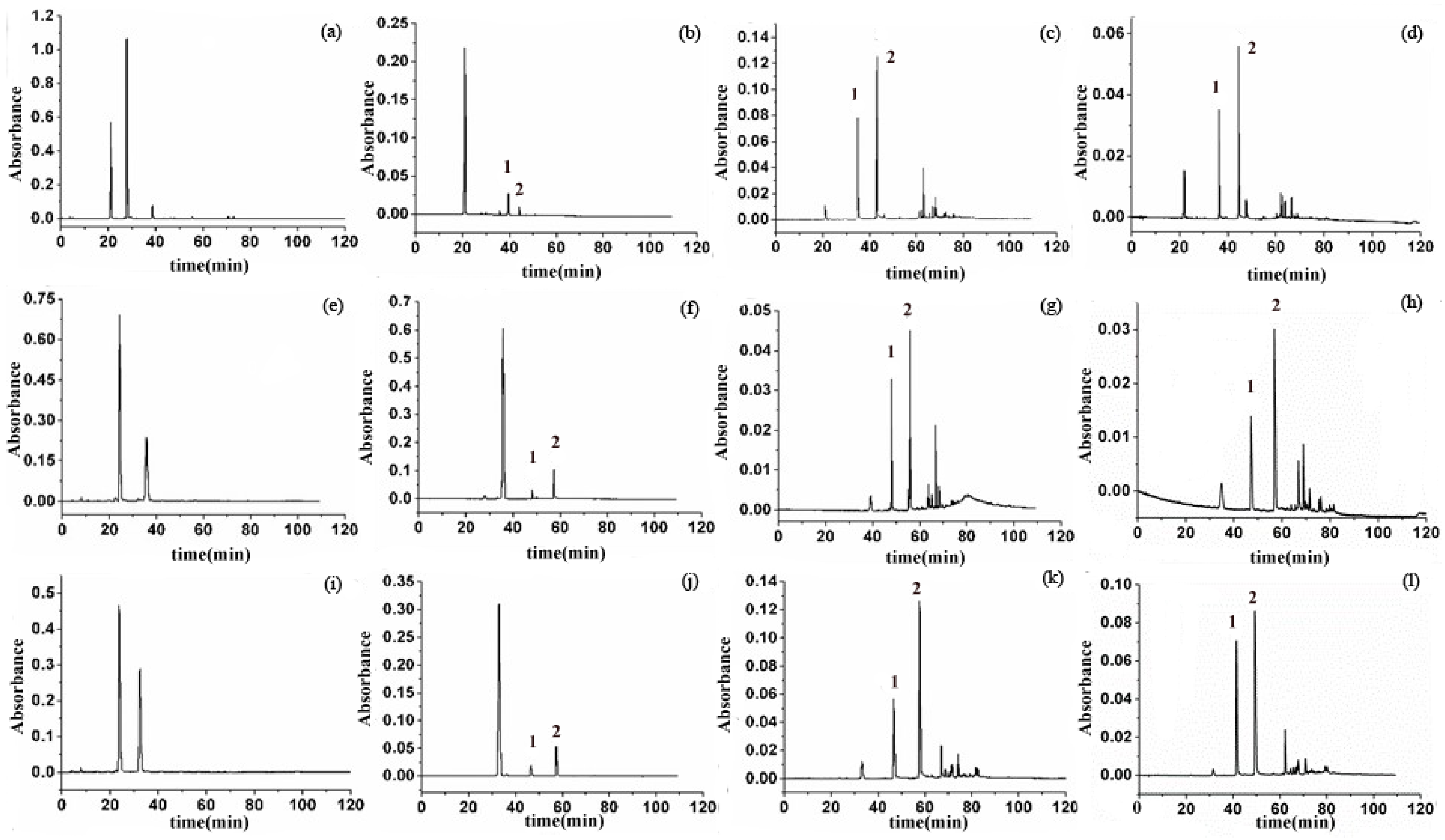
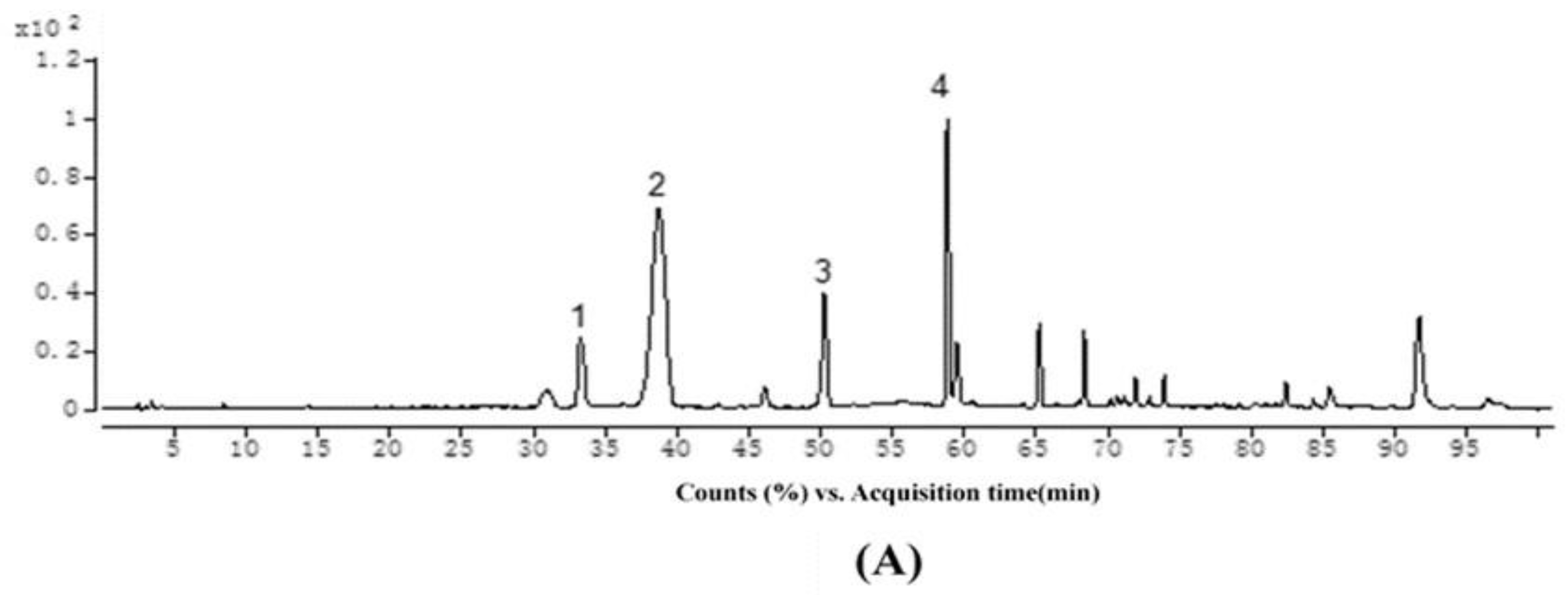
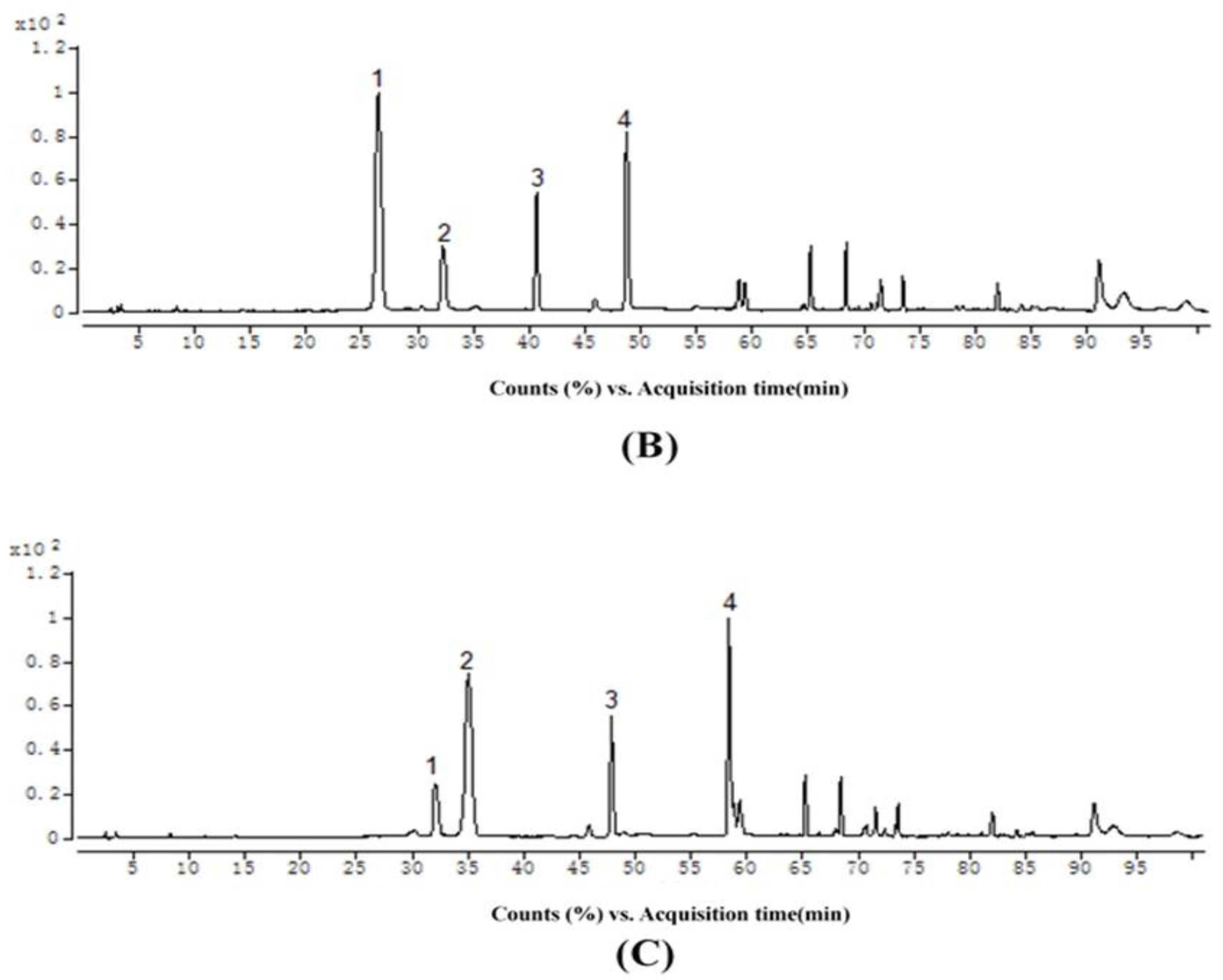

| Reactants (anthocyanin + EC) | Reaction Condition (Temperature, Molar Ratio (anthocyanin/EC/acetaldehyde)) | |||
|---|---|---|---|---|
| pH = 1.7 | Rate Constant K (h−1) | pH = 3.2 | Rate Constant K (h−1) | |
| Mv + EC | 25 °C, 1:1:10 | 0.0497 | 25 °C, 1:1:10 | 0.0028 |
| 25 °C, 1:3:10 | 0.0565 | 25 °C, 1:3:10 | 0.0065 | |
| 25 °C, 1:6:10 | 0.0919 | 25 °C, 1:6:10 | 0.0090 | |
| 30 °C, 1:6:10 | 0.1763 | 30 °C, 1:6:10 | 0.0191 | |
| 35 °C, 1:6:10 | 0.3207 | 35 °C, 1:6:10 | 0.0413 | |
| 40 °C, 1:6:10 | 0.7693 | 40 °C, 1:6:10 | 0.0910 | |
| Cy + EC | 25 °C, 1:6:10 | 0.1005 | 25 °C, 1:6:10 | 0.0066 |
| Pn + EC | 25 °C, 1:6:10 | 0.1630 | 25 °C, 1:6:10 | 0.0101 |
| Sample | No. | Compound | [M]+ | MS2 Product Ions (m/z) |
|---|---|---|---|---|
| Mv + EC | 1 | Mv | 493.1344 | 331.0812 |
| 2 | Mv-ethyl-EC (1) | 809.2293 | 647.1760, 519.1502, 357.0974 | |
| 3 | Mv-ethyl-EC (2) | 809.2289 | 647.1763, 519.1497, 357.0976 | |
| 4 | Mv-ethyl-EC-ethyl-EC | 1125.3240 | ||
| Cy + EC | 1 | Cy | 449.1082 | 287.0551 |
| 2 | Cy-ethyl-EC (1) | 765.2029 | 603.1496, 475.1239, 313.0713 | |
| 3 | Cy-ethyl-EC (2) | 765.2025 | 603.1500, 475.1235, 313.0715 | |
| 4 | Cy-ethyl-EC-ethyl-EC | 1081.2977 | ||
| Pn + EC | 1 | Pn | 463.1239 | 301.0707 |
| 2 | Pn-ethyl-EC (1) | 779.2184 | 617.1655, 489.1395, 327.0872 | |
| 3 | Pn-ethyl-EC (2) | 779.2182 | 617.1659, 489.1391, 327.0872 | |
| 4 | Pn-ethyl-EC-ethyl-EC | 1095.3126 |
| Compounds | DPPH (μmol/L) | ABTS (μmol/L) | FRAP (μmol/L) | |||
|---|---|---|---|---|---|---|
| EC50 | Linear Range | EC50 | Linear Range | FRAP Value | Linear Range | |
| Pn | 189 ± 3 c | 24.8–198.5 | 107 ± 2 c | 19.8–148.8 | 10.3 ± 0.0059 g | 24.3–194.8 |
| Mv | 190 ± 10 c | 24.3–194.8 | 106 ± 2 c | 19.5–146.1 | 8.9 ± 0.0070 h | 24.8–248.1 |
| Cy | 170 ± 5 d | 48.9–244.6 | 103 ± 1 d | 19.6–146.7 | 10.8 ± 0.0056 f | 12.2–146.7 |
| EC | 153 ± 4 e | 50.2–301.2 | 99 ± 1 e | 50.2–200.8 | 11.5 ± 0.0069 e | 25.1–200.8 |
| Pn-ethyl-EC (S) | 130 ± 4 f | 24–192 | 95 ± 2 f | 24–192 | 12.2 ± 0.0045 d | 24–192 |
| Pn-ethyl-EC (R) | 121 ± 2 f | 24.5–196.1 | 90 ± 2 g | 24.5–196.1 | 12.4 ± 0.0079 d | 25.6–205 |
| Mv-ethyl-EC (S) | 132 ± 1 f | 25.6–205 | 86 ± 1 h | 25.6–153.8 | 13.3 ± 0.0069 b | 24.8–198 |
| Mv-ethyl-EC (R) | 135 ± 2 f | 24.8–198 | 87 ± 2 g,h | 29.7–118.8 | 12.9 ± 0.0074 c | 12.2–145.8 |
| Cy-ethyl-EC (S) | 83 ± 2 g | 12.2–97.2 | 79 ± 3 i | 24.3–116.7 | 12.4 ± 0.0061 d | 12.2–146 |
| Cy-ethyl-EC (R) | 80 ± 3 g | 12.2–97.3 | 77 ± 3 i | 24.3–116.8 | 16.5 ± 0.0048 a | 12.5–150 |
| VC | 1030 ± 27 a | 125.5–2007.6 | 469 ± 4 a | 200.8–702.6 | ||
| Trolox | 921 ± 17 b | 200.4–1002 | 447 ± 2 b | 200.4–701.4 | 2.2 ± 0.0049 i | 200.4–1002 |
| FeSO4 | 400–2000.1 | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Zhang, M.; Zhang, S.; Cui, Y.; Sun, B. Preparation and Antioxidant Activity of Ethyl-Linked Anthocyanin-Flavanol Pigments from Model Wine Solutions. Molecules 2018, 23, 1066. https://doi.org/10.3390/molecules23051066
Li L, Zhang M, Zhang S, Cui Y, Sun B. Preparation and Antioxidant Activity of Ethyl-Linked Anthocyanin-Flavanol Pigments from Model Wine Solutions. Molecules. 2018; 23(5):1066. https://doi.org/10.3390/molecules23051066
Chicago/Turabian StyleLi, Lingxi, Minna Zhang, Shuting Zhang, Yan Cui, and Baoshan Sun. 2018. "Preparation and Antioxidant Activity of Ethyl-Linked Anthocyanin-Flavanol Pigments from Model Wine Solutions" Molecules 23, no. 5: 1066. https://doi.org/10.3390/molecules23051066
APA StyleLi, L., Zhang, M., Zhang, S., Cui, Y., & Sun, B. (2018). Preparation and Antioxidant Activity of Ethyl-Linked Anthocyanin-Flavanol Pigments from Model Wine Solutions. Molecules, 23(5), 1066. https://doi.org/10.3390/molecules23051066




