Avocado Seed: A Comparative Study of Antioxidant Content and Capacity in Protecting Oil Models from Oxidation
Abstract
1. Introduction
2. Results and Discussion
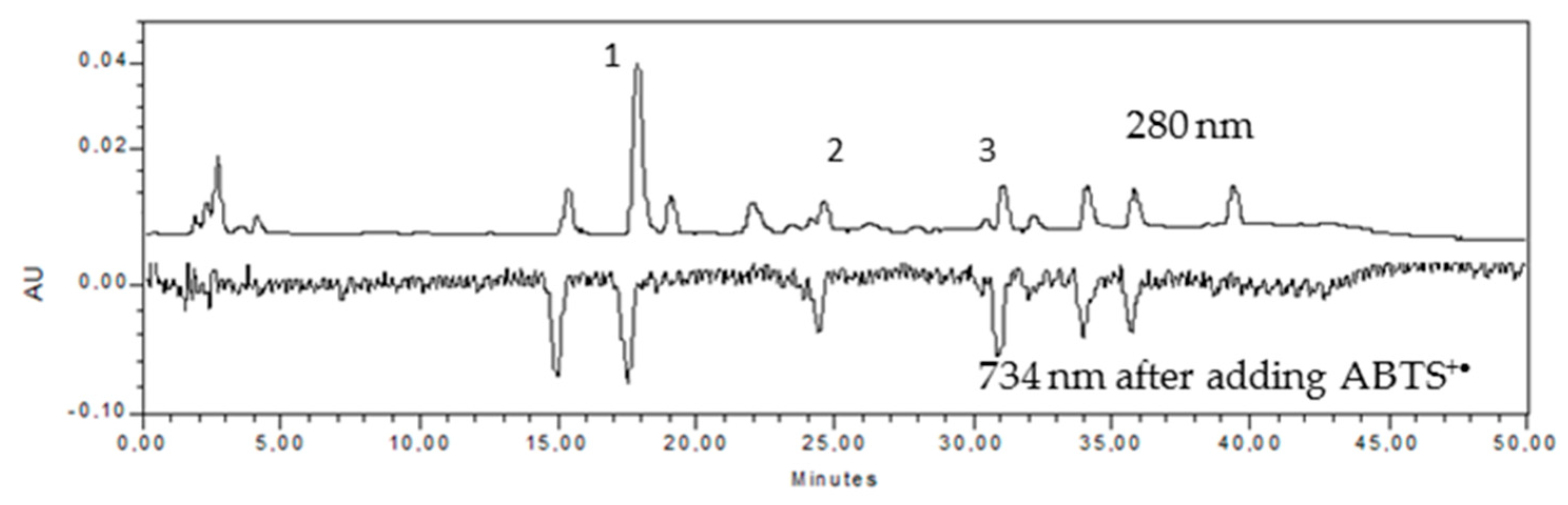
2.1.Total Polyphenol Content (TPC) and Radical Scavenging Activity
2.2. Protective Effect of ASE in Sunflower Oil Fatty Acid Mixture
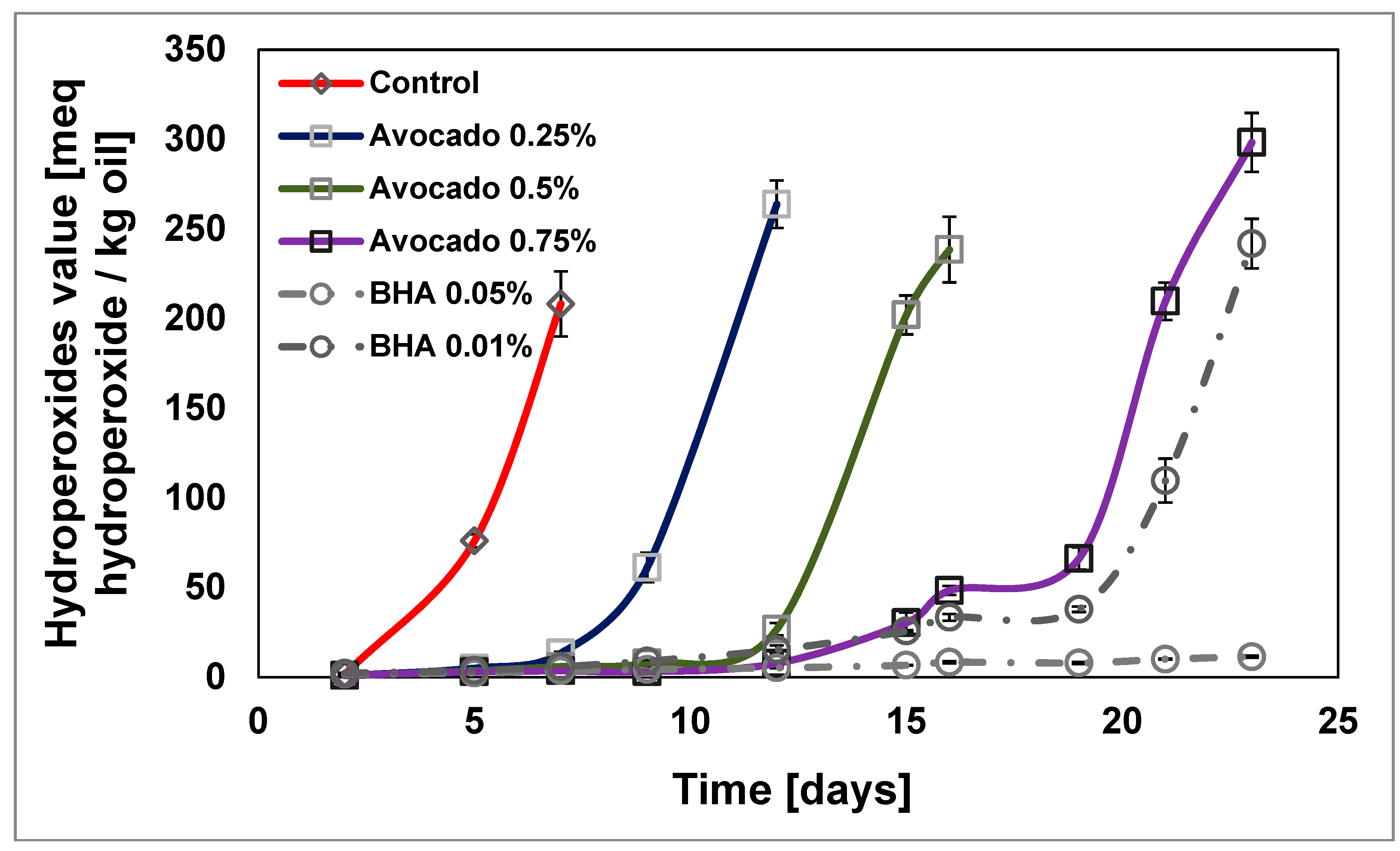
2.2.1. Primary Oxidation of Sunflower Oil by Peroxide Value
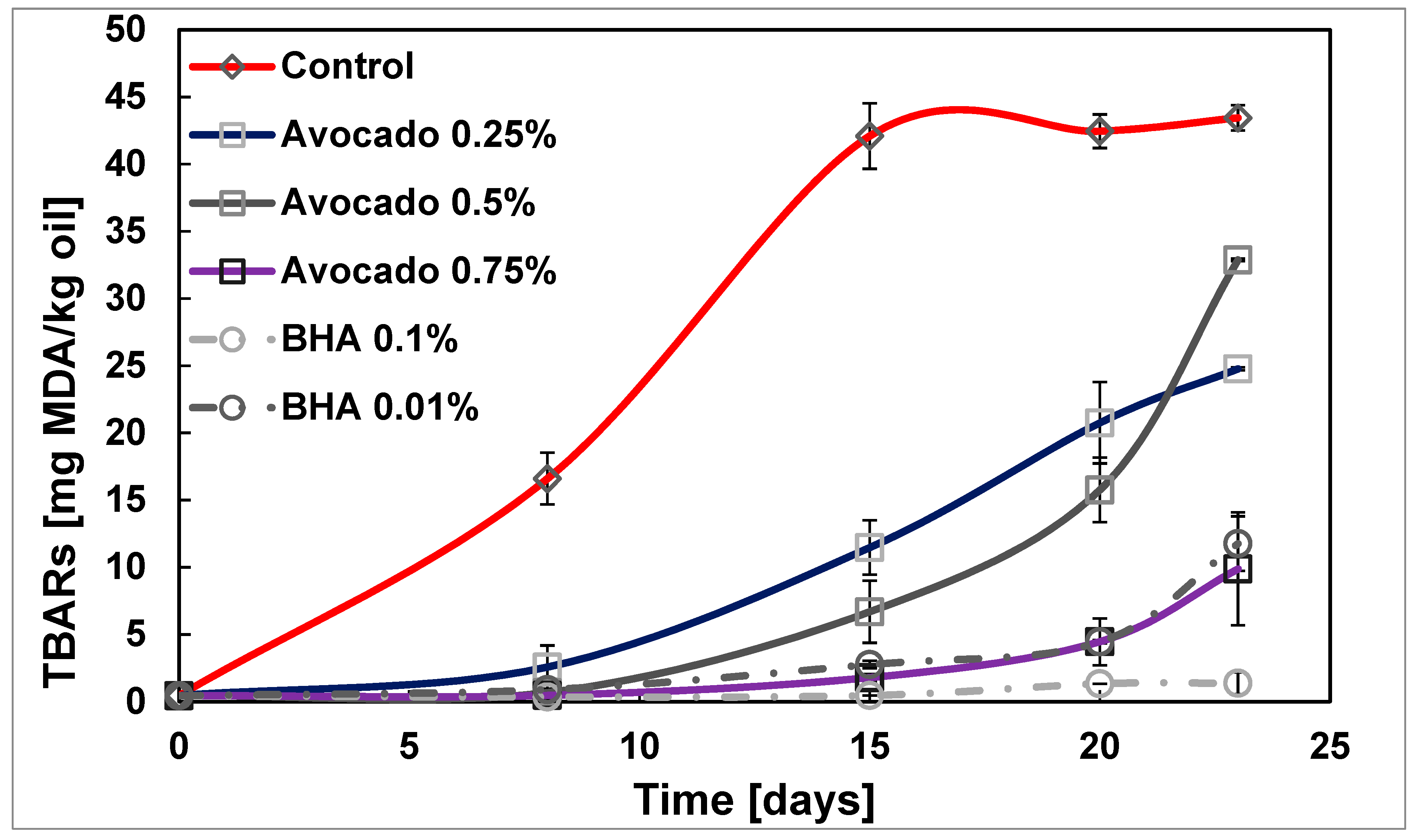
2.2.2. Secondary Oxidation of Sunflower Oil by TBARs
3. Materials and Methods
3.1. Sample and Extracts Preparation
3.2. Material and Reagents
3.3. Chemical Analysis
3.3.1. Total Polyphenol Content (TPC)
3.3.2. Radical Scavenging Activity
3.3.3. Determination of Methoxy Radical Scavenging Activity by EPR
3.3.4. High Performance Liquid Chromatography (HPLC)
3.3.5. Determination of Primary Oxidation with Peroxide Value (PV) and Secondary Oxidation with TBARs Method
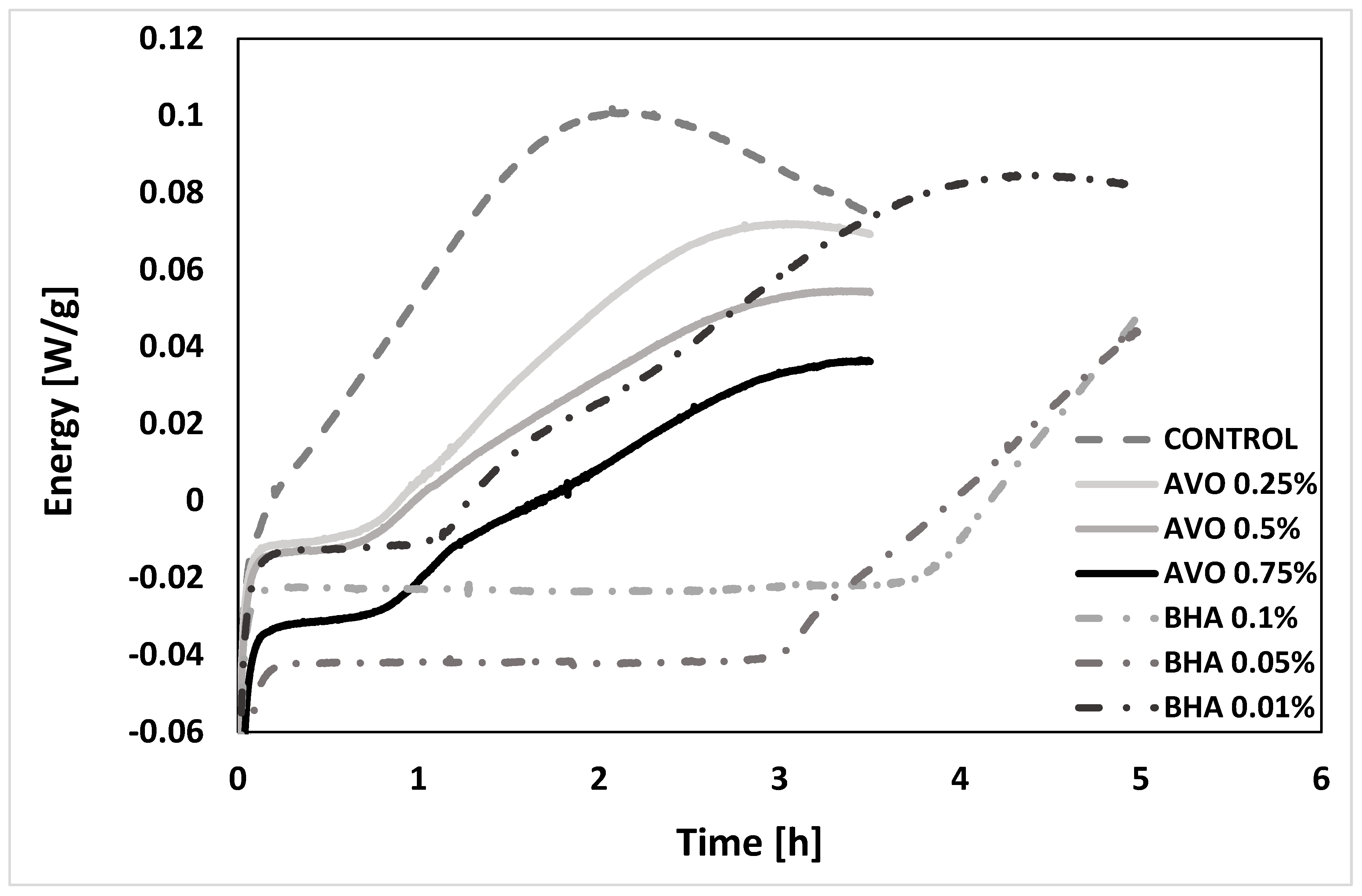
3.3.6. Oxidation Induction Time analysis (OIT-DSC)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- King, A.J.; Griffin, J.K.; Roslan, F. In vivo and in vitro addition of dried olive extract in poultry. J. Agric. Food Chem. 2014, 62, 7915–7919. [Google Scholar] [CrossRef] [PubMed]
- Perumalla, A.V.S.; Hettiarachchy, N.S. Green tea and grape seed extracts—Potential applications in food safety and quality. Food Res. Int. 2011, 44, 827–839. [Google Scholar] [CrossRef]
- Jordán, M.J.; Lax, V.; Rota, M.C.; Lorán, S.; Sotomayor, J.A. Relevance of carnosic acid, carnosol, and rosmarinic acid concentrations in the in vitro antioxidant and antimicrobial activities of Rosmarinus officinalis (L.) methanolic extracts. J. Agric. Food Chem. 2012, 60, 9603–9608. [Google Scholar] [CrossRef] [PubMed]
- Wettasinghe, M.; Shahidi, F.; Amarowicz, R.; Abou-Zaid, M.M. Phenolic acids in defatted seeds of borage (Borago officinalis L.). Food Chem. 2001, 75, 49–56. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Demirata, B.; Özyürek, M.; Çelik, S.E.; Bektaşoǧlu, B.; Berker, K.I.; Özyurt, D. Comparative evaluation of various total antioxidant capacity assays applied to phenolic compounds with the CUPRAC assay. Molecules 2007, 12, 1496–1547. [Google Scholar] [CrossRef] [PubMed]
- Yashin, A.; Yashin, Y.; Wang, J.Y.; Nemzer, B. Antioxidant and antiradical activity of Coffee. Antioxidants 2013, 2, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.; Chae, K.S.; Choi, H.R.; Lee, S.J.; Gim, S.W.; Kwon, G.T.; Lee, H.T.; Song, Y.C.; Kim, K.J.; Kong, H.S.; et al. Bioactive and pharmacokinetic characteristics of pre-matured black raspberry, Rubus occidentalis. Ital. J. Food Sci. 2018, 30, 428–439. [Google Scholar]
- Cano-Lamadrid, M.; Hernández, F.; Nowicka, P.; Carbonell-Barrachina, A.A.; Wojdyło, A. Formulation and storage effects on pomegranate smoothie phenolic composition, antioxidant capacity and color. LWT 2018, 96, 322–328. [Google Scholar] [CrossRef]
- Lubinska-Szczygieł, M.; Różańska, A.; Namieśnik, J.; Dymerski, T.; Shafreen, R.B.; Weisz, M.; Ezra, A.; Gorinstein, S. Quality of limes juices based on the aroma and antioxidant properties. Food Control 2018, 89, 270–279. [Google Scholar] [CrossRef]
- Giuffrè, A.M.; Zappia, C.; Capocasale, M. Effects of high temperatures and duration of heating on olive oil properties for food use and biodiesel production. J. Am. Oil Chem. Soc. 2017, 94, 819–830. [Google Scholar] [CrossRef]
- Giuffrè, A.M.; Capocasale, M.; Zappia, C.; Poiana, M. Influence of high temperature and duration of heating on the sunflower seed oil properties for food use and bio-diesel production. J. Oleo Sci. 2017, 66, 1193–1205. [Google Scholar] [CrossRef] [PubMed]
- Ayala-zavala, J.F.; Vega-vega, V.; Rosas-domínguez, C.; Palafox-carlos, H.; Villa-rodriguez, J.A.; Siddiqui, M.W.; Dávila-Aviña, J.E.; González-Aguilar, G.A. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 2011, 44, 1866–1874. [Google Scholar] [CrossRef]
- Wijngaard, H.; Hossain, M.B.; Rai, D.K.; Brunton, N. Techniques to extract bioactive compounds from food by-products of plant origin. Food Res. Int. 2012, 46, 505–513. [Google Scholar] [CrossRef]
- Lagha-Benamrouche, S.; Madani, K. Phenolic contents and antioxidant activity of orange varieties (Citrus sinensis L. and Citrus aurantium L.) cultivated in Algeria: Peels and leaves. Ind. Crops Prod. 2013, 50, 723–730. [Google Scholar] [CrossRef]
- Aguedo, M.; Kohnen, S.; Rabetafika, N.; Vanden Bossche, S.; Sterckx, J.; Blecker, C.; Beauve, C.; Paquot, M. Composition of by-products from cooked fruit processing and potential use in food products. J. Food Compos. Anal. 2012, 27, 61–69. [Google Scholar] [CrossRef]
- Wijngaard, H.H.; Brunton, N. The optimisation of solid-liquid extraction of antioxidants from apple pomace by response surface methodology. J. Food Eng. 2010, 96, 134–140. [Google Scholar] [CrossRef]
- Guerrero, M.S.; Torres, J.S.; Nuñez, M.J. Extraction of polyphenols from white distilled grape pomace: Optimization and modelling. Bioresour. Technol. 2008, 99, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Giuffrè, A.M. HPLC-DAD detection of changes in phenol content of red berry skins during grape ripening. Eur. Food Res. Technol. 2013, 237, 555–564. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, M.; Su, G.; Cai, M.; Zhou, C.; Huang, J.; Lin, L. The antioxidant activities and the xanthine oxidase inhibition effects of walnut (Juglans regia L.) fruit, stem and leaf. Int. J. Food Sci. Technol. 2015, 50, 233–239. [Google Scholar] [CrossRef]
- Villamil, L.; Astier, M.; Merlín, Y.; Ayala-Barajas, R.; Ramírez-García, E.; Martínez-Cruz, J.; Devoto, M.; Gavito, M.E. Management practices and diversity of flower visitors and herbaceous plants in conventional and organic avocado orchards in Michoacán, Mexico. Agroecol. Sustain. Food Syst. 2018, 42, 530–551. [Google Scholar] [CrossRef]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Estévez, M. Avocado by-products as inhibitors of color deterioration and lipid and protein oxidation in raw porcine patties subjected to chilled storage. Meat Sci. 2011, 89, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, J.G.; Borrás-Linares, I.; Lozano-Sánchez, J.; Segura-Carretero, A. Comprehensive characterization of phenolic and other polar compounds in the seed and seed coat of avocado by HPLC-DAD-ESI-QTOF-MS. Food Res. Int. 2018, 105, 752–763. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Smith, R.W.; Henao, J.J.A.; Stark, K.D.; Spagnuolo, P.A. Analytical method to detect and quantify Avocatin B in Hass Avocado Seed and pulp matter. J. Nat. Prod. 2018, 81, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, A.; Rodríguez-Sánchez, D.G.; Villarreal-Lara, R.; Navarro-Silva, J.M.; Senés-Guerrero, C.; Hernández-Brenes, C. Stability of the antimicrobial activity of acetogenins from avocado seed, under common food processing conditions, against Clostridium sporogenes vegetative cell growth and endospore germination. Int. J. Food Sci. Technol. 2017, 52, 2311–2323. [Google Scholar] [CrossRef]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Estévez, M. Avocado, sunflower and olive oils as replacers of pork back-fat in burger patties: Effect on lipid composition, oxidative stability and quality traits. Meat Sci. 2012, 90, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Gómez, F.S.; Sánchez, S.P.; Iradi, M.G.G.; Azman, N.A.M.; Almajano, M.P. Avocado seeds: Extraction optimization and possible use as antioxidant in food. Antioxidants 2014, 3, 439–454. [Google Scholar] [CrossRef] [PubMed]
- Pahua-Ramos, M.E.; Ortiz-Moreno, A.; Chamorro-Cevallos, G.; Hernández-Navarro, M.D.; Garduño-Siciliano, L.; Necoechea-Mondragón, H.; Hernández-Ortega, M. Hypolipidemic effect of Avocado (Persea americana Mill) seed in a hypercholesterolemic mouse model. Plant Foods Hum. Nutr. 2012, 67, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Kosińska, A.; Karamać, M.; Estrella, I.; Hernández, T.; Bartolomé, B.; Dykes, G.A. Phenolic compound profiles and antioxidant capacity of Persea americana Mill. peels and seeds of two varieties. J. Agric. Food Chem. 2012, 60, 4613–4619. [Google Scholar] [CrossRef] [PubMed]
- López-Yerena, A.; Guerra-Ramírez, D.; Jácome-Rincón, J.; Espinosa-Solares, T.; Reyes-Trejo, B.; Famiani, F.; Cruz-Castillo, J.G. Initial evaluation of fruit of accessions of Persea schiedeana Nees for nutritional value, quality and oil extraction. Food Chem. 2018, 245, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Andrade, M.J.; Kylli, P.; Estévez, M. Avocado (Persea americana Mill.) phenolics, in vitro antioxidant and antimicrobial activities, and inhibition of lipid and protein oxidation in porcine patties. J. Agric. Food Chem 2011, 59, 5625–5635. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Bostic, T.R.; Gu, L. Antioxidant capacities, procyanidins and pigments in avocados of different strains and cultivars. Food Chem. 2010, 122, 1193–1198. [Google Scholar] [CrossRef]
- Soong, Y.Y.; Barlow, P.J. Antioxidant activity and phenolic content of selected fruit seeds. Food Chem. 2004, 88, 411–417. [Google Scholar] [CrossRef]
- Tremocoldi, M.A.; Rosalen, P.L.; Franchin, M.; Massarioli, A.P.; Denny, C.; Daiuto, É.R.; Paschoal, J.A.R.; Melo, P.S.; de Alencar, S.M. Exploration of avocado by-products as natural sources of bioactive compounds. PLoS ONE 2018, 13, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Jónsdóttir, R.; Ólafsdóttir, G. Total phenolic compounds, radical scavenging and metal chelation of extracts from Icelandic seaweeds. Food Chem. 2009, 116, 240–248. [Google Scholar] [CrossRef]
- Tobolková, B.; Polovka, M.; Belajová, E.; Koreňovská, M.; Suhaj, M. Possibilities of organic and conventional wines differentiation on the basis of multivariate analysis of their characteristics (EPR, UV-Vis, HPLC and AAS study). Eur. Food Res. Technol. 2014, 239, 441–451. [Google Scholar] [CrossRef]
- Mocan, A.; Crișan, G.; Vlase, L.; Crișan, O.; Vodnar, D.C.; Raita, O.; Gheldiu, A.M.; Toiu, A.; Oprean, R.; Tilea, I. Comparative studies on polyphenolic composition, antioxidant and antimicrobial activities of Schisandra chinensis leaves and fruits. Molecules 2014, 19, 15162–15179. [Google Scholar] [CrossRef] [PubMed]
- Azman, N.A.M.; Peiró, S.; Fajarí, L.; Julià, L.; Almajano, M.P. Radical scavenging of white tea and its flavonoid constituents by electron paramagnetic resonance (EPR) spectroscopy. J. Agric. Food Chem. 2014, 62, 5743–5748. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Gong, J.; Liu, J.; Wu, X.; Zhang, Y. Antioxidant capacity of extract from edible flowers of Prunus mume in China and its active components. LWT-Food Sci. Technol. 2009, 42, 477–482. [Google Scholar] [CrossRef]
- Silva, B.M.; Andrade, P.B.; Valentão, P.; Ferreres, F.; Seabra, R.M.; Ferreira, M.A. Quince (Cydonia oblonga Miller) fruit (pulp, peel, and seed) and jam: Antioxidant activity. J. Agric. Food Chem. 2004, 52, 4705–4712. [Google Scholar] [CrossRef] [PubMed]
- Sendra, J.M.; Sentandreu, E.; Navarro, J.L. Kinetic model for the antiradical activity of the isolated p-catechol group in flavanone type structures using the free stable radical 2,2-diphenyl-1-picrylhydrazyl as the antiradical probe. J. Agric. Food Chem. 2007, 55, 5512–5522. [Google Scholar] [CrossRef] [PubMed]
- Kosar, M.; Dorman, D.; Baser, K.; Hiltunen, R. An Improved HPLC post-column methodology for the identification of free radical scavenging phytochemicals in complex mixtures. Chromatographia 2004, 60, 635–638. [Google Scholar] [CrossRef]
- Shi, S.; Zhou, H.; Zhang, Y.; Jiang, X.; Chen, X. Coupling HPLC to on-line, post-column (bio) chemical assays for high-resolution screening of bioactive compounds from complex mixtures. TrAC Trends Anal. Chem. 2009, 28, 865–877. [Google Scholar] [CrossRef]
- Koleva, I.I.; Niederla, H.A.G.; van Beek, T.A. Application of ABTS radical cation for selective on-line detection of radical scavengers in HPLC eluates. Anal. Chem. 2001, 73, 3373–3381. [Google Scholar] [CrossRef] [PubMed]
- Marinova, E.M.; Toneva, A.; Yanishlieva, N. Comparison of the antioxidative properties of caffeic and chlorogenic acids. Food Chem. 2009, 114, 1498–1502. [Google Scholar] [CrossRef]
- Dibert, K.; Cros, E.; Andrieu, J. Solvent extraction of oil and chlorogenic acid from green coffee part I: Equilibrium data. J. Food Eng. 1989, 10, 1–11. [Google Scholar] [CrossRef]
- Bertrand, C.; Noirot, M.; Doulbeau, S.; de Kochko, A.; Hamon, S.; Campa, C. Chlorogenic acid content swap during fruit maturation in Coffea pseudozanguebariae: Qualitative comparison with leaves. Plant Sci. 2003, 165, 1355–1361. [Google Scholar] [CrossRef]
- Fujioka, K.; Shibamoto, T. Chlorogenic acid and caffeine contents in various commercial brewed coffees. Food Chem. 2008, 106, 217–221. [Google Scholar] [CrossRef]
- Alarcon, E.; Campos, A.M.; Edwards, A.M.; Lissi, E.; Lopez-alarcon, C. Antioxidant capacity of herbal infusions and tea extracts: A comparison of ORAC-fluorescein and ORAC-pyrogallol red methodologies. Food Chem. 2008, 107, 1114–1119. [Google Scholar] [CrossRef]
- Pilar Almajano, M.; Carbó, R.; LóPez Jiménez, J.A.; Gordon, M.H. Antioxidant and antimicrobial activities of tea infusions. Food Chem. 2008, 108, 55–63. [Google Scholar] [CrossRef]
- Vaidya, B.; Eun, J. Effect of Temperature on Oxidation Kinetics of Walnut and Grape Seed Oil. Food Sci. Biotechnol. 2013, 22, 273–279. [Google Scholar] [CrossRef]
- Abdelazim, A.A.; Mahmoud, A.; Ramadan-Hassanien, M.F. Oxidative stability of vegetable oils as affected by sesame extracts during accelerated oxidative storage. J. Food Sci. Technol. 2011, 50, 868–878. [Google Scholar] [CrossRef] [PubMed]
- Wardhani, D.H.; Fuciños, P.; Vázquez, J.A.; Pandiella, S.S. Inhibition kinetics of lipid oxidation of model foods by using antioxidant extract of fermented soybeans. Food Chem. 2013, 139, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Ciftçi, O.N.; Kowalski, B.; Göğüş, F.; Fadiloğlu, S. Effect of the addition of a cocoa butter-like fat enzymatically produced from olive pomace oil on the oxidative stability of cocoa butter. J. Food Sci. 2009, 74, E184–E190. [Google Scholar] [CrossRef] [PubMed]
- Araújo, K.L.G.V.; Magnani, M.; Nascimento, J.A.; Souza, A.L.; Epaminondas, P.S.; Queiroz, N.; Queiroz, N.S.; Aquino, J.; Souza, A.G.; Costa, M.F.C.; Souza, A.L. By-products from fruit processing: One alternative antioxidant for use in soybean oil. J. Therm. Anal. Calorim. 2017, 130, 1229–1235. [Google Scholar] [CrossRef]
- Kodali, D.R. Oxidative stability measurement of high-stability oils by pressure differential scanning calorimeter (PDSC). J. Agric. Food Chem. 2005, 53, 7649–7653. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, J.A.; Arau, K.L.G.V.; Epaminondas, P.S.; Souza, A.S.; Magnani, M.; Souza, A.L.; Soledade, L.E.B.; Queiroz, N.; Souza, A.G. Ethanolic extracts of Moringa oleifera Lam. Evaluation of its potential as an antioxidant additive for fish oil. J. Therm. Anal. Calorim. 2013, 114, 833–838. [Google Scholar] [CrossRef]
- Quinchia, L.A.; Delgado, M.A.; Valencia, C.; Franco, J.M.; Gallegos, C. Natural and synthetic antioxidant additives for improving the performance of new biolubricant formulations. J. Agric. Food Chem. 2011, 59, 12917–12924. [Google Scholar] [CrossRef] [PubMed]
- Skowyra, M.; Gallego, M.G.; Segovia, F.; Almajano, M.P. Antioxidant Properties of Artemisia annua Extracts in Model Food Emulsions. Antioxidants 2014, 3, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Segovia, F.; Lupo, B.; Peiró, S.; Gordon, M.; Almajano, M. Extraction of Antioxidants from Borage (Borago officinalis L.) Leaves—Optimization by response surface method and application in oil-in-water emulsions. Antioxidants 2014, 3, 339–357. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Skowyra, M.; Falguera, V.; Azman, N.; Segovia, F.; Almajano, M. The Effect of Perilla frutescens extract on the oxidative stability of model food emulsions. Antioxidants 2014, 3, 38–54. [Google Scholar] [CrossRef] [PubMed]
- Azman, N.; Segovia, F.; Martínez-Farré, X.; Gil, E.; Almajano, M. Screening of antioxidant activity of Gentian Lutea root and its application in oil-in-water emulsions. Antioxidants 2014, 3, 455–471. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Maurya, S.; DeLampasona, M.P.; Catalan, C.A.N. A comparison of chemical, antioxidant and antimicrobial studies of cinnamon leaf and bark volatile oils, oleoresins and their constituents. Food Chem. Toxicol. 2007, 45, 1650–1661. [Google Scholar] [CrossRef] [PubMed]
- American Oil Chemists’ Society (AOCS). Peroxide Value (acetic acid-chloroform method). In Official Methods and Recommended Practices of the AOCS, 5th ed.; Official Method Cd 8-53; AOCS Press: Champaign, IL, USA, 1997. [Google Scholar]
- Gallego, M.G.; Gordon, M.H.; Segovia, F.J.; Skowyra, M.; Almajano, M.P. Antioxidant properties of three aromatic herbs (Rosemary, Thyme and Lavender) in oil-in-water emulsions. J. Am. Oil Chem. Soc. 2013, 1559–1568. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Seed Sample | Extraction Conditions | TPC [mg GAE/g DW] | ORAC [µmol TE/g DW] | ABTS [µmol TE/g DW] | DPPH [µmol TE/g DW] | FRAP [µmol TE/g DW] | Ref. |
|---|---|---|---|---|---|---|---|
| Persea schiedeana | Acetone/water/acetic acid (70:29.7:0.3, v/v/v), r.t. | 6.15 ± 0.08 | - | 58.45 ± 1.39 | 37.64 ± 4.81 | 42.81 ± 0.80 | [30] |
| Persea americana var. Hass | Acetone/water/acetic acid (70:29.7:0.3, v/v/v), r.t. | 16.5 | 137.2 | - | 52.7 | - | [32] |
| Persea americana var. Hass | Ethanol/water (1:1, v/v), 200 °C, 11 MPa | - | 310 ± 30 | 300 ± 20 | 15 ± 2 γ | - | [23] |
| Persea americana | Ethanol/Water (1:1, v/v), 70 °C | 88.2 ± 2.2 | - | 725 ± 39.4 | - | 1484 ± 15.7 | [33] |
| Persea americana | Ethanol/water (56:44, v/v), 63 °C | 45.01 | 616.48 | - | - | - | [27] |
| Persea americana var. Hass 1 and Fuerte 2 | Ethanol/Water (80:20, v/v), r.t. | 57.3 ± 2.7 1, 59.2 ± 6.9 2 | - | 645.8 ± 17.9 1, 580.8 ± 31.0 2 | 410.7 ± 35.8 1, 464.9 ± 32.7 2 | 656.9 ± 26.0 1,δ, 931.7 ± 65.6 2,δ | [34] |
| Persea americana var. Hass | Methanol/Water (80:20, v/v), 60 °C | 9.51 ± 0.161 | 210 | 94 | - | - | [29] |
| Persea americana | Soxhlet Methanol/water (75:25, v/v) | 292.00 ± 9.81 | - | 173.3 | - | - | [28] |
| Persea americana var. Hass 1 and Fuerte 2 | Methanol/water (70:30, v/v), r.t. | 35.11 1, 41.64 2 | - | 78.93 ± 26.73 1,β, 121.61 ± 31.87 2,β | 66.24 ± 24.84 1,β, 94.27 ± 30.47 2,β | CUPRAC: 141.67 ± 41.24 1,β, 184.42 ± 66.05 2,β | [31] |
| Persea americana var. Hass 1 and Fuerte 2 | Acetone/water (70:30, v/v), r.t. | 60.82 1, 69.12 2 | - | 158.29 ± 26.27 1,β, 194.80 ± 44.69 2,β | 130.26 ± 36.80 1,β, 167.50 ± 42.08 2,β | CUPRAC: 275.36 ± 59.09 1,β, 353.43 ± 75.83 2,β | [31] |
| Method | Methanol, 4 °C, 24 h | Ethanol/water (50:50, v/v) 4 °C, 24 h |
|---|---|---|
| TPC [mg GAE/g DW] | 25.35 ± 0.77 | 30.98 ± 0.68 |
| ORAC [µmol TE/g DW] | 1240 ± 70 (0.59 ± 0.03 1) | 1310 ± 40 |
| ABTS [µmol TE/g DW] | 123.74 ± 2.46 (0.15 ± 0.00 1) | 263.58 ± 17.85 |
| FRAP [µmol TE/g DW] | 316.60 ± 6.87 (0.19 ± 0.00 1) | 438.89 ± 7.32 |
| EPR [µmol FAE/g DW] | 0.53 ± 0.07 | - |
| Name | Retention Time (RT) | HPLC Peak Area | Concentration mg/L | HPLC-ABTS Peak Area | % | Antioxidant Capacity [mg GAE/L] | % Antioxidant Activity in the Total Extract |
|---|---|---|---|---|---|---|---|
| Procyanidin 1 * | 15.36 | 199,682 | - | 1,956,638 | 21.76 | 53.12 | 16.3 |
| Chlorogenic acid | 17.88 | 1,011,205 | 51.59 | 1,901,135 | 21.14 | 51.86 | 16.0 |
| (+)-Catechin | 24.59 | 157,538 | 20.10 | 977,869 | 10.88 | 30.84 | 9.5 |
| (−)-Epicatechin | 31.07 | 233,557 | 27.89 | 1,574,801 | 17.51 | 44.43 | 13.7 |
| Procyanidin 2 * | 32.21 | 80,373 | - | 265,837 | 2.96 | 14.63 | 4.5 |
| Procyanidin 3 * | 34.12 | 216,547 | - | 1,169,588 | 13.01 | 35.21 | 10.8 |
| Procyanidin 4 * | 35.82 | 192,750 | - | 960,683 | 10.68 | 30.45 | 9.4 |
| Catechin 1 ** | 39.40 | 205,953 | - | 184,943 | 2.06 | 12.79 | 3.9 |
| Sample | IT [days] 1 | OIT [min] 2 | PV10 [meq hydroperoxide/kg oil] 3 | PV Slope [meq Hydroperoxide/kg oil·days] | TBARs15 [mg MDA/kg oil] 4 | TBARs Slope [mg MDA/kg oil·days] |
|---|---|---|---|---|---|---|
| Control | 5.00 | 28.51 | - | 66.08 | 42.09 ± 1.92 a | 3.64 |
| Avocado 0.25% | 8.31 | 41.55 | 110.03 ± 13.28 a | 67.52 | 11.47 ± 1.60 b | 1.27 |
| Avocado 0.5% | 11.65 | 43.22 | 26.74 ± 3.50 b | 54.21 | 6.69 ± 1.04 b | 0.86 |
| Avocado 0.75% | 18.85 | 52.54 | 7.73 ± 0.98 c | 58.02 | 1.79 ± 0.38 c | 0.19 |
| BHA 0.01% | 19.17 | 4.54 | 15.37 ± 2.28 d | 51.00 | 2.76 ± 0.08 d | 0.26 |
| BHA 0.05% | - | 127.79 | 5.23 ± 0.19 e | - | 0.99 ± 0.00 e | 0.06 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segovia, F.J.; Hidalgo, G.I.; Villasante, J.; Ramis, X.; Almajano, M.P. Avocado Seed: A Comparative Study of Antioxidant Content and Capacity in Protecting Oil Models from Oxidation. Molecules 2018, 23, 2421. https://doi.org/10.3390/molecules23102421
Segovia FJ, Hidalgo GI, Villasante J, Ramis X, Almajano MP. Avocado Seed: A Comparative Study of Antioxidant Content and Capacity in Protecting Oil Models from Oxidation. Molecules. 2018; 23(10):2421. https://doi.org/10.3390/molecules23102421
Chicago/Turabian StyleSegovia, Francisco J., Gádor Indra Hidalgo, Juliana Villasante, Xavier Ramis, and María Pilar Almajano. 2018. "Avocado Seed: A Comparative Study of Antioxidant Content and Capacity in Protecting Oil Models from Oxidation" Molecules 23, no. 10: 2421. https://doi.org/10.3390/molecules23102421
APA StyleSegovia, F. J., Hidalgo, G. I., Villasante, J., Ramis, X., & Almajano, M. P. (2018). Avocado Seed: A Comparative Study of Antioxidant Content and Capacity in Protecting Oil Models from Oxidation. Molecules, 23(10), 2421. https://doi.org/10.3390/molecules23102421






