Increasing the Strength and Production of Artemisinin and Its Derivatives
Abstract
1. Introduction and Background
2. Increased Production of Artemisinin and Its Analogues
3. Advancements in Understanding the Mode of Action of Artemisinin and Related Drugs
4. Marine Sponges as a Source of Endoperoxides
5. Artemisinin Inspired Novel Antimalarial Compounds
6. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
Abbreviations
| ACTs | Artemisinin-based combination therapies |
| COSTREL | combinatorial super transformation of transplastomic recipient lines |
| ALDH1 | aldehyde dehydrogenase |
| DXR | 1-deoxy-d-xylulose-5-phosphate reductoisomerase |
| ELQ | Endochin-like quinolones |
References
- World Health Organisation WHO. World Malaria Report 2017; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- World Health Organization. World Health Statistics 2017: Monitoring Health for The SDGs; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- World Health Organization. World Malaria Report 2016; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Barber, B.E.; Rajahram, G.S.; Grigg, M.J.; William, T.; Anstey, N.M. World Malaria Report: Time to acknowledge Plasmodium knowlesi malaria. Malar. J. 2017, 16, 135. [Google Scholar] [CrossRef] [PubMed]
- Alonso, P.L.; Brown, G.; Arevalo-Herrera, M.; Binka, F.; Chitnis, C.; Collins, F.; Doumbo, O.K.; Greenwood, B.; Hall, B.F.; Levine, M.M.; et al. A research Agenda to underpin Malaria Eradication. PLoS Med. 2011, 8, e1000406. [Google Scholar] [CrossRef] [PubMed]
- Dondorp, A.M.; Nosten, F.; Yi, P.; Das, D.; Phyo, A.P.; Tarning, J.; Lwin, K.M.; Ariey, F.; Hanpithakpong, W.; Lee, S.J.; et al. Artemisinin resistance in Plasmodium falciparum malaria. N. Engl. J. Med. 2009, 361, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Phyo, A.P.; Nkhoma, S.; Stepniewska, K.; Ashley, E.A.; Nair, S.; McGready, R.; Moo, C.L.; Al-Saai, S.; Dondorp, A.M.; Lwin, K.M.; et al. Emergence of artemisinin-resistant malaria on the western border of Thailand: A longitudinal study. Lancet 2012, 379, 1960–1966. [Google Scholar] [CrossRef]
- Olliaro, P.; Wells, T.N.C. The global portfolio of new antimalarial medicines under development. Clin. Pharmacol. Ther. 2009, 85, 584–595. [Google Scholar] [CrossRef] [PubMed]
- Burrows, J.N.; van Huijsduijnen, R.H.; Möhrle, J.J.; Oeuvray, C.; Wells, T.N. Designing the next generation of medicines for malaria control and eradication. Malar. J. 2013, 12, 187. [Google Scholar] [CrossRef] [PubMed]
- Rackham, M.D.; Brannigan, J.A.; Rangachari, K.; Meister, S.; Wilkinson, A.J.; Holder, A.A.; Leatherbarrow, R.J.; Tate, E.W. Design and synthesis of high affinity inhibitors of Plasmodium falciparum and Plasmodium vivax N-myristoyltransferases directed by ligand efficiency dependent lipophilicity (LELP). J. Med. Chem. 2014, 57, 2773–2788. [Google Scholar] [CrossRef] [PubMed]
- Ashley, E.A.; Dhorda, M.; Fairhurst, R.M.; Amaratunga, C.; Lim, P.; Suon, S.; Sreng, S.; Anderson, J.M.; Mao, S.; Sam, B.; et al. Spread of Artemisinin Resistance in Plasmodium falciparum Malaria. N. Engl. J. Med. 2014, 371, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Vale, N.; Aguiar, L.; Gomes, P. Antimicrobial peptides: A new class of antimalarial drugs? Front. Pharmacol. 2014, 5, 275. [Google Scholar] [CrossRef] [PubMed]
- Roca-Feltrer, A.; Carneiro, I.; Armstrong Schellenberg, J.R.M. Estimates of the burden of malaria morbidity in Africa in children under the age of 5 years. Trop. Med. Int. Health 2008, 13, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Moutinho, P.R.; Gil, L.H.; Cruz, R.B.; Ribolla, P.E. Population dynamics, structure and behavior of Anopheles darlingi in a rural settlement in the Amazon rainforest of Acre, Brazil. Malar. J. 2011, 10, 174. [Google Scholar] [CrossRef] [PubMed]
- Kourtis, A.P.; Read, J.S.; Jamieson, D.J. Pregnancy and Infection. N. Engl. J. Med. 2014, 370, 2211–2218. [Google Scholar] [CrossRef] [PubMed]
- Hay, S.I.; Sinka, M.E.; Okara, R.M.; Kabaria, C.W.; Mbithi, P.M.; Tago, C.C.; Benz, D.; Gething, P.W.; Howes, R.E.; Patil, A.P.; et al. Developing global maps of the dominant Anopheles vectors of human malaria. PLoS Med. 2010, 7, e1000209. [Google Scholar] [CrossRef] [PubMed]
- Gurarie, D.; Zimmerman, P.A.; King, C.H. Dynamic regulation of single- and mixed-species malaria infection: Insights to specific and non-specific mechanisms of control. J. Theor. Biol. 2006, 240, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Baird, J.K. Neglect of Plasmodium vivax malaria. Trends Parasitol. 2007, 23, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Grimberg, B.T.; Mehlotra, R.K. Expanding the antimalarial drug arsenal-now, but how? Pharmaceuticals 2011, 4, 681–712. [Google Scholar] [CrossRef] [PubMed]
- Schantz-Dunn, J.; Nour, N.M. Malaria and pregnancy: A global health perspective. Rev. Obstet. Gynecol. 2009, 2, 186–192. [Google Scholar] [PubMed]
- Weathers, P.J.; Arsenault, P.R.; Covello, P.S.; McMickle, A.; Teoh, K.H.; Reed, D.W. Artemisinin production in Artemisia annua: Studies in planta and results of a novel delivery method for treating malaria and other neglected diseases. Phytochem. Rev. 2011, 10, 173–183. [Google Scholar] [CrossRef] [PubMed]
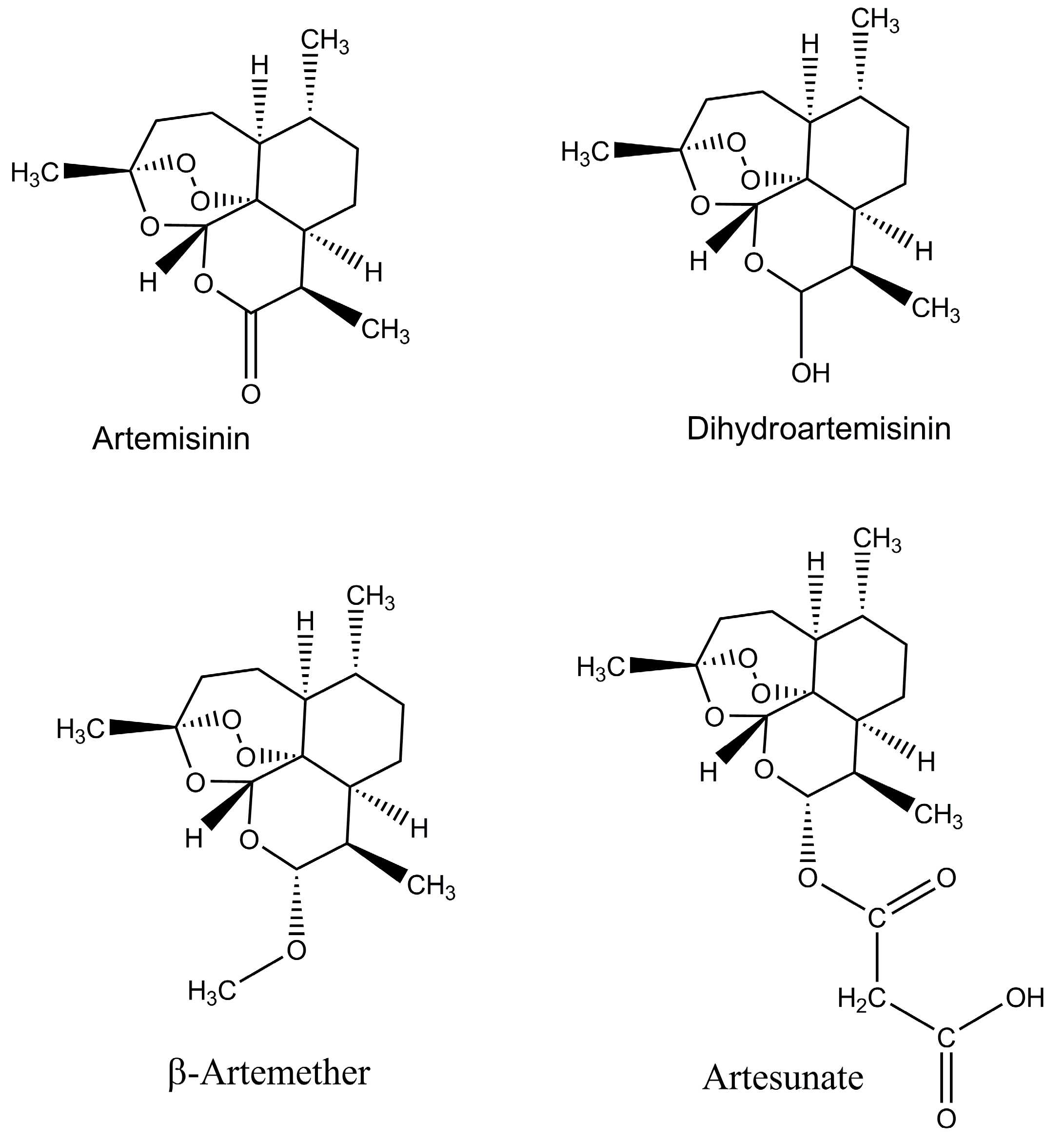
- Klayman, D. Qinghaosu (artemisinin): An antimalarial drug from China. Science 1985, 228, 1049–1055. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.Y.; Tan, R.X. Artemisinin, a miracle of traditional Chinese medicine. Nat. Prod. Rep. 2015, 32, 1617–1621. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y. Artemisinin—A Gift from Traditional Chinese Medicine to the World (Nobel Lecture). Angew. Chem. Int. Ed. 2016, 55, 10210–10226. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, D.; Goswami, A.; Saikia, P.P.; Barua, N.C.; Rao, P.G. Artemisinin and its derivatives: A novel class of anti-malarial and anti-cancer agents. Chem. Soc. Rev. 2010, 39, 435–454. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Li, H.; Yang, Y.; Hou, L. Anti-inflammatory and immunoregulatory functions of artemisinin and its derivatives. Mediat. Inflamm. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.K.; Xu, C.; Kalesh, K.A.; He, Y.; Lin, Q.; Wong, W.S.F.; Shen, H.M.; Wang, J. Artemisinin as an anticancer drug: Recent advances in target profiling and mechanisms of action. Med. Res. Rev. 2017, 37, 1492–1517. [Google Scholar] [CrossRef] [PubMed]
- Crespo-Ortiz, M.P.; Wei, M.Q. Antitumor activity of artemisinin and its derivatives: From a well-known antimalarial agent to a potential anticancer drug. J. Biomed. Biotechnol. 2012, 2012, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Vil, V.A.; Yaremenko, I.A.; Ilovaisky, A.I.; Terent’ev, A.O. Peroxides with Anthelmintic, Antiprotozoal, Fungicidal and Antiviral Bioactivity: Properties, Synthesis and Reactions. Molecules 2017, 22, 1881. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. World Malaria Report 2015; World Health Organization: Geneva, Switzerland, 2015; Volume 243. [Google Scholar]
- Beeson, J.G.; Boeuf, P.; Fowkes, F.J. Maximizing antimalarial efficacy and the importance of dosing strategies. BMC Med. 2015, 13, 110. [Google Scholar] [CrossRef] [PubMed]
- Mok, S.; Ashley, E.A.; Ferreira, P.E.; Zhu, L.; Lin, Z.; Yeo, T.; Chotivanich, K.; Imwong, M.; Pukrittayakamee, S.; Dhorda, M.; et al. Population transcriptomics of human malaria parasites reveals the mechanism of artemisinin resistance. Science 2015, 347, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Fairhurst, R.M.; Nayyar, G.M.L.; Breman, J.G.; Hallett, R.; Vennerstrom, J.L.; Duong, S.; Ringwald, P.; Wellems, T.E.; Plowe, C.V.; Dondorp, A.M. Artemisinin-Resistant Malaria: Research Challenges, Opportunities, and Public Health Implications. Am. J. Trop. Med. Hyg. 2012, 87, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Ariey, F.; Witkowski, B.; Amaratunga, C.; Beghain, J.; Langlois, A.-C.; Khim, N.; Kim, S.; Duru, V.; Bouchier, C.; Ma, L.; et al. A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature 2014, 505, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Slutsker, L.; Newman, R.D. Malaria scale-up progress: Is the glass half-empty or half-full? Lancet 2009, 373, 11–13. [Google Scholar] [CrossRef]
- Tun, K.M.; Imwong, M.; Lwin, K.M.; Win, A.A.; Hlaing, T.M.; Hlaing, T.; Lin, K.; Kyaw, M.P.; Plewes, K.; Faiz, M.A.; et al. Spread of artemisinin-resistant Plasmodium falciparum in Myanmar: A cross-sectional survey of the K13 molecular marker. Lancet Infect. Dis. 2015, 15, 415–421. [Google Scholar] [CrossRef]
- Karema, C.; Imwong, M.; Fanello, C.I.; Stepniewska, K.; Uwimana, A.; Nakeesathit, S.; Dondorp, A.; Day, N.P.; White, N.J. Molecular correlates of high-level antifolate resistance in Rwandan children with Plasmodium falciparum malaria. Antimicrob. Agents Chemother. 2010, 54, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, M.; Van Den Broek, I.; Van Herp, M.; Urrutia, P.P.P.; Van Overmeir, C.; Kyomuhendo, J.; Ndosimao, C.N.; Ashley, E.; Guthmann, J.-P. Varying efficacy of artesunate+amodiaquine and artesunate+sulphadoxine- pyrimethamine for the treatment of uncomplicated falciparum malaria in the Democratic Republic of Congo: A report of two in vivo studies. Malar. J. 2009, 8, 192. [Google Scholar] [CrossRef] [PubMed]
- Mutabingwa, T.K.; Anthony, D.; Heller, A.; Hallett, R.; Ahmed, J.; Drakeley, C.; Greenwood, B.M.; Whitty, C.J.M. Amodiaquine alone, amodiaquine+sulfadoxine-pyrimethamine, amodiaquine+artesunate, and artemether-lumefantrine for outpatient treatment of malaria in Tanzanian children: A four-arm randomised effectiveness trial. Lancet 2005, 365, 1474–1480. [Google Scholar] [CrossRef]
- Imwong, M.; Suwannasin, K.; Kunasol, C.; Sutawong, K.; Mayxay, M.; Rekol, H.; Smithuis, F.M.; Hlaing, T.M.; Tun, K.M.; van der Pluijm, R.W.; et al. The spread of artemisinin-resistant Plasmodium falciparum in the Greater Mekong subregion: A molecular epidemiology observational study. Lancet Infect. Dis. 2017, 17, 491–497. [Google Scholar] [CrossRef]
- Kolaczinski, J.; Macdonald, M.; Meek, S. Vector control to eliminate artemisinin resistant malaria in the greater mekong subregion. Lancet Infect. Dis. 2014, 14, 9–11. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Emergency Response to Artemisinin Resistance in the Greater Mekong Subregion: Regional Framework for Action 2013–2015; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Cui, L.; Yan, G.; Sattabongkot, J.; Cao, Y.; Chen, B.; Chen, X.; Fan, Q.; Fang, Q.; Jongwutiwes, S.; Parker, D.; et al. Malaria in the Greater Mekong Subregion: Heterogeneity and complexity. Acta Trop. 2012, 121, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Zwang, J.; Olliaro, P.; Barennes, H.; Bonnet, M.; Brasseur, P.; Bukirwa, H.; Cohuet, S.; D’Alessandro, U.; Djimdé, A.; Karema, C.; et al. Efficacy of artesunate-amodiaquine for treating uncomplicated falciparum malaria in sub-Saharan Africa: A multi-centre analysis. Malar. J. 2009, 8, 203. [Google Scholar] [CrossRef] [PubMed]
- Smithuis, F.; Kyaw, M.K.; Phe, O.; Win, T.; Aung, P.P.; Oo, A.P.P.; Naing, A.L.; Nyo, M.Y.; Myint, N.Z.H.; Imwong, M.; et al. Effectiveness of five artemisinin combination regimens with or without primaquine in uncomplicated falciparum malaria: An open-label randomised trial. Lancet Infect. Dis. 2010, 10, 673–681. [Google Scholar] [CrossRef]
- Price, R.N.; Douglas, N.M.; Anstey, N.M.; von Seidlein, L. Plasmodium vivax treatments: What are we looking for? Curr. Opin. Infect. Dis. 2011, 24, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Wells, T.N. Natural products as starting points for future anti-malarial therapies: Going back to our roots? Malar. J. 2011, 10, S3. [Google Scholar] [CrossRef] [PubMed]
- Nair, P.; Misra, A.; Singh, A.; Shukla, A.K.; Gupta, M.M.; Gupta, A.K.; Gupta, V.; Khanuja, S.P.S.; Shasany, A.K. Differentially expressed genes during contrasting growth stages of Artemisia annua for artemisinin content. PLoS ONE 2013, 8, e60375. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.; Shen, Q.; Yan, T.; Fu, X. Transgenic approach to increase artemisinin content in Artemisia annua L. Plant Cell Rep. 2014, 33, 605–615. [Google Scholar] [CrossRef] [PubMed]
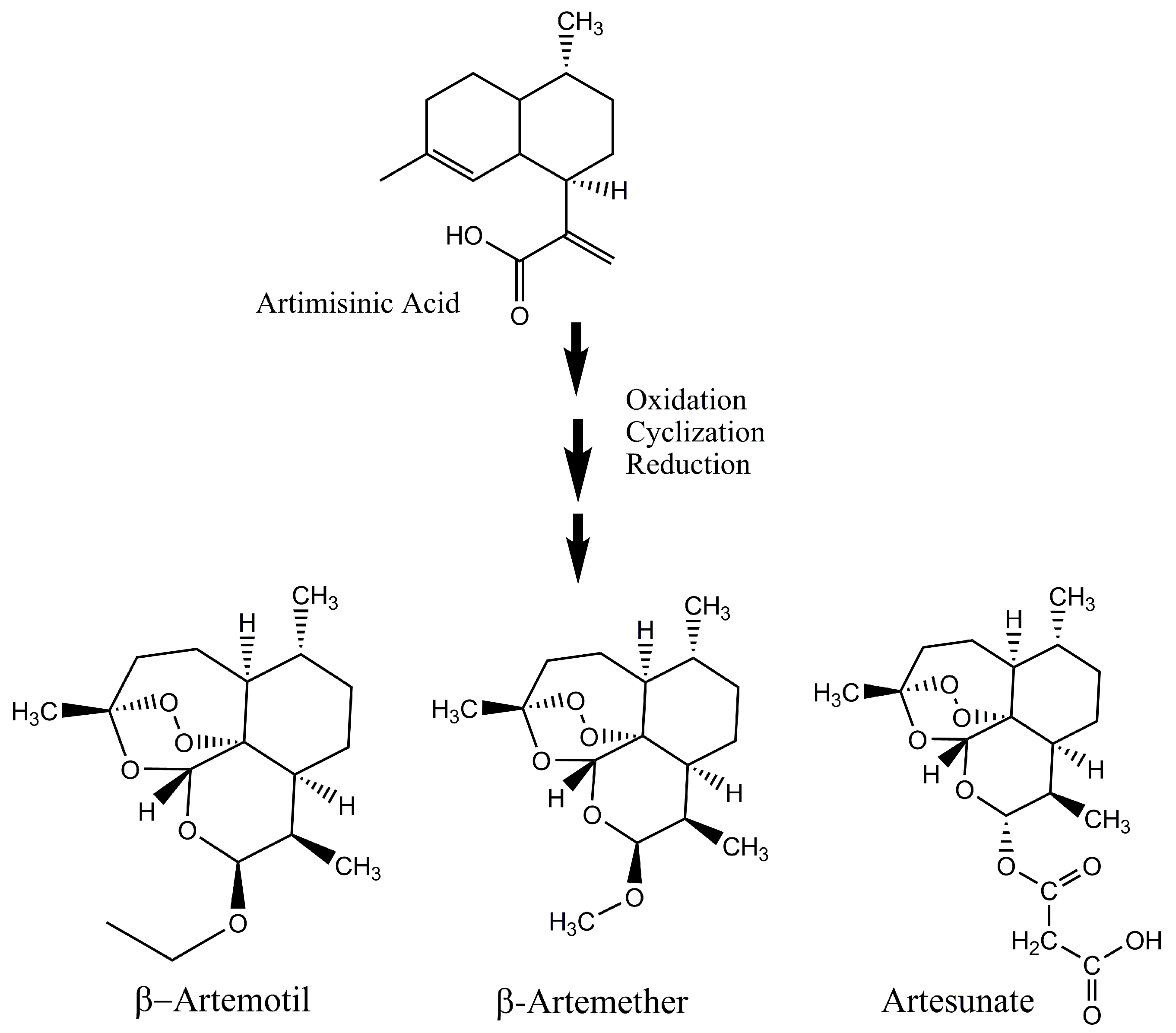
- Westfall, P.J.; Pitera, D.J.; Lenihan, J.R.; Eng, D.; Woolard, F.X.; Regentin, R.; Horning, T.; Tsuruta, H.; Melis, D.J.; Owens, A.; et al. Production of amorphadiene in yeast, and its conversion to dihydroartemisinic acid, precursor to the antimalarial agent artemisinin. Proc. Natl. Acad. Sci. USA 2012, 109, E111–E112. [Google Scholar] [CrossRef] [PubMed]
- Paddon, C.J.; Westfall, P.J.; Pitera, D.J.; Benjamin, K.; Fisher, K.; McPhee, D.; Leavell, M.D.; Tai, A.; Main, A.; Eng, D.; et al. High-level semi-synthetic production of the potent antimalarial artemisinin. Nature 2013, 496, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Paddon, C.J.; Keasling, J.D. Semi-synthetic artemisinin: A model for the use of synthetic biology in pharmaceutical development. Nat. Rev. Microbiol. 2014, 12, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Plan, M.R.; Carpenter, A.; Nielsen, L.K.; Vickers, C.E. Coupling gene regulatory patterns to bioprocess conditions to optimize synthetic metabolic modules for improved sesquiterpene production in yeast. Biotechnol. Biofuels 2017, 10, 43. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, K.R.; Silva, I.R.; Cherubim, J.P.; McPhee, D.; Paddon, C.J. Developing commercial production of semi-synthetic artemisinin, and of β-Farnesene, an Isoprenoid Produced by Fermentation of Brazilian Sugar. J. Braz. Chem. Soc. 2016, 27, 1339–1345. [Google Scholar] [CrossRef]
- Zhang, Y.; Nowak, G.; Reed, D.W.; Covello, P.S. The production of artemisinin precursors in tobacco. Plant Biotechnol. J. 2011, 9, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Farhi, M.; Marhevka, E.; Ben-Ari, J.; Algamas-Dimantov, A.; Liang, Z.; Zeevi, V.; Edelbaum, O.; Spitzer-Rimon, B.; Abeliovich, H.; Schwartz, B.; et al. Generation of the potent anti-malarial drug artemisinin in tobacco. Nat. Biotechnol. 2011, 29, 1072–1074. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, P.; Zhou, F.; Erban, A.; Karcher, D.; Kopka, J.; Bock, R. A new synthetic biology approach allows transfer of an entire metabolic pathway from a medicinal plant to a biomass crop. eLife 2016, 5, e13664. [Google Scholar] [CrossRef] [PubMed]
- Mbengue, A.; Bhattacharjee, S.; Pandharkar, T.; Liu, H.; Estiu, G.; Stahelin, R.V.; Rizk, S.S.; Njimoh, D.L.; Ryan, Y.; Chotivanich, K.; et al. A molecular mechanism of artemisinin resistance in Plasmodium falciparum malaria. Nature 2015, 520, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Miley, G.P.; Pou, S.; Winter, R.; Nilsen, A.; Li, Y.; Kelly, J.X.; Stickles, A.M.; Mather, M.W.; Forquer, I.P.; Pershing, A.M.; et al. ELQ-300 prodrugs for enhanced delivery and single dose cure of malaria. Antimicrob. Agents Chemother. 2015, 59, 5555–5560. [Google Scholar] [CrossRef] [PubMed]
- Nilsen, A.; LaCrue, A.N.; White, K.L.; Forquer, I.P.; Cross, R.M.; Marfurt, J.; Mather, M.W.; Delves, M.J.; Shackleford, D.M.; Saenz, F.E.; et al. Quinolone-3-Diarylethers: A New Class of Antimalarial Drug. Sci. Transl. Med. 2013, 5, 177ra37. [Google Scholar] [CrossRef] [PubMed]
- Winter, R.W.; Kelly, J.X.; Smilkstein, M.J.; Dodean, R.; Hinrichs, D.; Riscoe, M.K. Antimalarial Quinolones: Synthesis, potency, and mechanistic studies. Exp. Parasitol. 2008, 118, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, K.; Subramaniyan, M.; Rawat, K.; Kalamuddin, M.; Qureshi, M.I.; Malhotra, P.; Mohmmed, A.; Cornish, K.; Daniell, H.; Kumar, S. Compartmentalized Metabolic Engineering for Artemisinin Biosynthesis and Effective Malaria Treatment by Oral Delivery of Plant Cells. Mol. Plant 2016, 9, 1464–1477. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.D.; Kumar, S.; Daniell, H. Expression of β-glucosidase increases trichome density and artemisinin content in transgenic Artemisia annua plants. Plant Biotechnol. J. 2016, 14, 1034–1045. [Google Scholar] [CrossRef] [PubMed]
- Czechowski, T.; Larson, T.R.; Catania, T.M.; Harvey, D.; Brown, G.D.; Graham, I.A. Artemisia annua mutant impaired in artemisinin synthesis demonstrates importance of nonenzymatic conversion in terpenoid metabolism. Proc. Natl. Acad. Sci. USA 2016, 113, 15150–15155. [Google Scholar] [CrossRef] [PubMed]
- Arora, M.; Saxena, P.; Choudhary, D.K.; Abdin, M.Z.; Varma, A. Dual symbiosis between Piriformospora indica and Azotobacter chroococcum enhances the artemisinin content in Artemisia annua L. World J. Microbiol. Biotechnol. 2016, 32, 19. [Google Scholar] [CrossRef] [PubMed]
- Arora, M.; Saxena, P.; Abdin, M.Z.; Varma, A. Interaction between Piriformospora indica and Azotobacter chroococcum governs better plant physiological and biochemical parameters in Artemisia annua L. plants grown under in vitro conditions. Symbiosis 2017, 1–10. [Google Scholar] [CrossRef]
- Singh, D.; McPhee, D.; Paddon, C.J.; Cherry, J.; Maurya, G.; Mahale, G.; Patel, Y.; Kumar, N.; Singh, S.; Sharma, B.; et al. Amalgamation of Synthetic Biology and Chemistry for High-Throughput Nonconventional Synthesis of the Antimalarial Drug Artemisinin. Org. Process Res. Dev. 2017, 21, 551–558. [Google Scholar] [CrossRef]
- Hosseini, R.; Yazdani, N.; Garoosi, G. The presence of amorpha-4,11-diene synthase, a key enzyme in artemisinin production in ten Artemisia species. Daru 2011, 19, 332–337. [Google Scholar] [PubMed]
- Gilmore, K.; Kopetzki, D.; Lee, J.W.; Horváth, Z.; McQuade, D.T.; Seidel-Morgenstern, A.; Seeberger, P.H. Continuous synthesis of artemisinin-derived medicines. Chem. Commun. 2014, 50, 12652–12655. [Google Scholar] [CrossRef] [PubMed]
- Ikram, N.K.B.; Beyraghdar Kashkooli, A.; Peramuna, A.V.; Van Der Krol, A.R.; Bouwmeester, H.; Simonsen, H.T. Stable production of the antimalarial drug artemisinin in the moss Physcomitrella patens. Front. Bioeng. Biotechnol. 2017, 5, 47. [Google Scholar] [CrossRef] [PubMed]
- Ikram, N.K.B.K.; Simonsen, H.T. A Review of Biotechnological Artemisinin Production in Plants. Front. Plant Sci. 2017, 8, 1966. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.-W.; Han, L.; Zhang, Y.-H.; Chen, D.-F.; Simonsen, H.T. Sclareol production in the moss Physcomitrella patens and observations on growth and terpenoid biosynthesis. Plant Biotechnol. Rep. 2015, 9, 149–159. [Google Scholar] [CrossRef]
- Zhan, X.; Zhang, Y.-H.; Chen, D.-F.; Simonsen, H.T. Metabolic engineering of the moss Physcomitrella patens to produce the sesquiterpenoids patchoulol and α/β-santalene. Front. Plant Sci. 2014, 5, 636. [Google Scholar] [CrossRef] [PubMed]
- Gangl, D.; Zedler, J.A.Z.; Rajakumar, P.D.; Martinez, E.M.R.; Riseley, A.; Włodarczyk, A.; Purton, S.; Sakuragi, Y.; Howe, C.J.; Jensen, P.E.; et al. Biotechnological exploitation of microalgae. J. Exp. Bot. 2015, 66, 6975–6990. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Pohnert, G.; Wei, D. Extracellular metabolites from industrial microalgae and their biotechnological potential. Mar. Drugs 2016, 14, 191. [Google Scholar] [CrossRef] [PubMed]
- Sigamani, S.; Ramamurthy, D.; Natarajan, H. A review on potential biotechnological applications of microalgae. J. Appl. Pharm. Sci. 2016, 6, 179–184. [Google Scholar] [CrossRef]
- Hagemann, M.; Hess, W.R. Systems and synthetic biology for the biotechnological application of cyanobacteria. Curr. Opin. Biotechnol. 2018, 49, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wang, J.; Meldrum, D.R. Application of synthetic biology in cyanobacteria and algae. Front. Microbiol. 2012, 3, 344. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Pathak, J.; Sinha, R.P. Cyanobacterial factories for the production of green energy and value-added products: An integrated approach for economic viability. Renew. Sustain. Energy Rev. 2017, 69, 578–595. [Google Scholar]
- Choi, S.Y.; Lee, H.J.; Choi, J.; Kim, J.; Sim, S.J.; Um, Y.; Kim, Y.; Lee, T.S.; Keasling, J.D.; Woo, H.M. Photosynthetic conversion of CO2 to farnesyl diphosphate-derived phytochemicals (amorpha-4,11-diene and squalene) by engineered cyanobacteria. Biotechnol. Biofuels 2016, 9, 202. [Google Scholar] [CrossRef] [PubMed]
- Shandilya, A.; Chacko, S.; Jayaram, B.; Ghosh, I. A plausible mechanism for the antimalarial activity of artemisinin: A computational approach. Sci. Rep. 2013, 3, 2513. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, P.M.; Barton, V.E.; Ward, S.A. The molecular mechanism of action of artemisinin—The debate continues. Molecules 2010, 15, 1705–1721. [Google Scholar] [CrossRef] [PubMed]
- Robert, A.; Meunier, B. Is alkylation the main mechanism of action of the antimalarial drug artemisinin? Chem. Soc. Rev. 1998, 27, 273. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, C.-J.; Chia, W.N.; Loh, C.C.Y.; Li, Z.; Lee, Y.M.; He, Y.; Yuan, L.-X.; Lim, T.K.; Liu, M.; et al. Haem-activated promiscuous targeting of artemisinin in Plasmodium falciparum. Nat. Commun. 2015, 6, 10111. [Google Scholar] [CrossRef] [PubMed]
- Ismail, H.M.; Barton, V.E.; Panchana, M.; Charoensutthivarakul, S.; Biagini, G.A.; Ward, S.A.; O’Neill, P.M. Corrigendum to: A Click Chemistry-Based Proteomic Approach Reveals that 1,2,4-Trioxolane and Artemisinin Antimalarials Share a Common Protein Alkylation Profile. Angew. Chem. Int. Ed. 2016, 55, 10548. [Google Scholar] [CrossRef] [PubMed]
- Ismail, H.M.; Barton, V.; Phanchana, M.; Charoensutthivarakul, S.; Wong, M.H.L.; Hemingway, J.; Biagini, G.A.; O’Neill, P.M.; Ward, S.A. Artemisinin activity-based probes identify multiple molecular targets within the asexual stage of the malaria parasites Plasmodium falciparum 3D7. Proc. Natl. Acad. Sci. USA 2016, 113, 2080–2085. [Google Scholar] [CrossRef] [PubMed]
- Asano, M.; Iwahashi, H. Determination of the structures of radicals formed in the reaction of antimalarial drug artemisinin with ferrous ions. Eur. J. Med. Chem. 2017, 127, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.Q.; Wang, D.D.; Ding, L.; Cui, H.Y.; Jin, H.; Yang, X.Y.; Yang, J.S.; Qin, B. Mechanism of artemisinin phytotoxicity action: Induction of reactive oxygen species and cell death in lettuce seedlings. Plant Physiol. Biochem. 2015, 88, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Nagasundaram, N.; George Priya Doss, C.; Chiranjib, C.; Karthick, V.; Thirumal Kumar, D.; Balaji, V.; Siva, R.; Lu, A.; Ge, Z.; Zhu, H. Mechanism of artemisinin resistance for malaria PfATP6 L263 mutations and discovering potential antimalarials: An integrated computational approach. Sci. Rep. 2016, 6, 30106. [Google Scholar]
- Badshah, S.L.; Naeem, A. Bioactive thiazine and benzothiazine derivatives: Green synthesis methods and their medicinal importance. Molecules 2016, 21, 1054. [Google Scholar] [CrossRef] [PubMed]
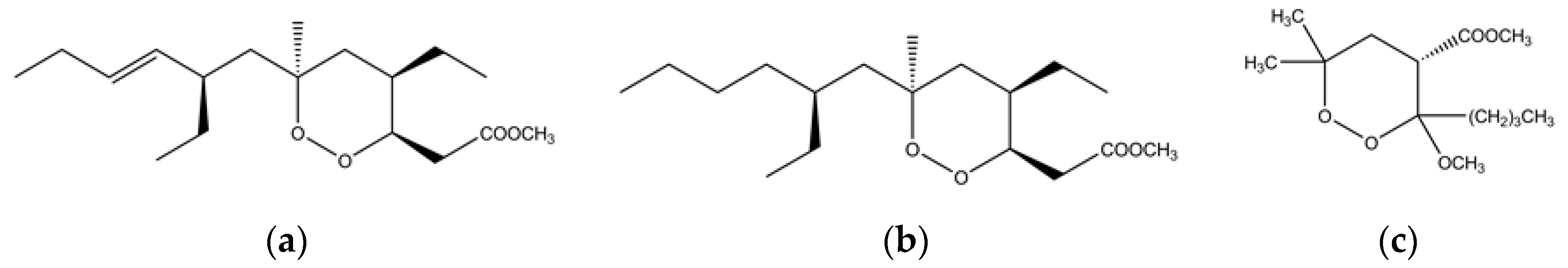
- Davis, R.A.; Duffy, S.; Fletcher, S.; Avery, V.M.; Quinn, R.J. Thiaplakortones A–D: Antimalarial thiazine alkaloids from the Australian marine sponge plakortis lita. J. Org. Chem. 2013, 78, 9608–9613. [Google Scholar] [CrossRef] [PubMed]
- Pouwer, R.H.; Deydier, S.M.; Van Le, P.; Schwartz, B.D.; Franken, N.C.; Davis, R.A.; Coster, M.J.; Charman, S.A.; Edstein, M.D.; Skinner-Adams, T.S.; et al. Total synthesis of thiaplakortone A: Derivatives as metabolically stable leads for the treatment of malaria. ACS Med. Chem. Lett. 2014, 5, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Persico, M.; Fattorusso, R.; Taglialatela-Scafati, O.; Chianese, G.; De Paola, I.; Zaccaro, L.; Rondinelli, F.; Lombardo, M.; Quintavalla, A.; Trombini, C.; et al. The interaction of heme with plakortin and a synthetic endoperoxide analogue: New insights into the heme-activated antimalarial mechanism. Sci. Rep. 2017, 7, 45485. [Google Scholar] [CrossRef] [PubMed]
- Skorokhod, O.A.; Davalos-Schafler, D.; Gallo, V.; Valente, E.; Ulliers, D.; Notarpietro, A.; Mandili, G.; Novelli, F.; Persico, M.; Taglialatela-Scafati, O.; et al. Oxidative stress-mediated antimalarial activity of plakortin, a natural endoperoxide from the tropical sponge Plakortis simplex. Free Radic. Biol. Med. 2015, 89, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Fattorusso, C.; Persico, M.; Calcinai, B.; Cerrano, C.; Parapini, S.; Taramelli, D.; Novellino, E.; Romano, A.; Scala, F.; Fattorusso, E.; et al. Manadoperoxides A–D from the indonesian sponge Plakortis cfr. simplex. Further insights on the structure-activity relationships of simple 1,2-dioxane antimalarials. J. Nat. Prod. 2010, 73, 1138–1145. [Google Scholar] [CrossRef] [PubMed]
- Fattorusso, E.; Parapini, S.; Campagnuolo, C.; Basilico, N.; Taglialatela-Scafati, O.; Taramelli, D. Activity against Plasmodium falciparum of cycloperoxide compounds obtained from the sponge Plakortis simplex. J. Antimicrob. Chemother. 2002, 50, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Campagnuolo, C.; Fattorusso, E.; Romano, A.; Taglialatela-Scafati, O.; Basilico, N.; Parapini, S.; Taramelli, D. Antimalarial polyketide cycloperoxides from the marine sponge Plakortis simplex. Eur. J. Org. Chem. 2005, 2005, 5077–5083. [Google Scholar] [CrossRef]
- Del Sol Jiménez, M.; Garzón, S.P.; Rodríguez, A.D. Plakortides M and N, bioactive polyketide endoperoxides from the Caribbean marine sponge Plakortis halichondrioides. J. Nat. Prod. 2003, 66, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.F.; Gao, H.F.; Kelly, M.; Hamann, M.T. Plakortides I–L, four new cyclic peroxides from an undescribed Jamaican sponge Plakortis sp. (Homosclerophorida, Plakinidae). Tetrahedron 2001, 57, 9379–9383. [Google Scholar] [CrossRef]
- Yu, H.B.; Liu, X.F.; Xu, Y.; Gan, J.H.; Jiao, W.H.; Shen, Y.; Lin, H.W. Woodylides A–C, new cytotoxic linear polyketides from the South China Sea sponge Plakortis simplex. Mar. Drugs 2012, 10, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Inbaneson, S.J.; Ravikumar, S. In vitro antiplasmodial activity of Clathria vulpina sponge associated bacteria against Plasmodium falciparum. Asian Pac. J. Trop. Dis. 2012, 2, 319–323. [Google Scholar] [CrossRef]
- Kumar, S.P.; Ravikumar, S. In vitro antiplasmodial activity of marine sponge Clathria vulpina extract against chloroquine sensitive Plasmodium falciparum. Asian Pac. J. Trop. Dis. 2014, 4, S162–S166. [Google Scholar] [CrossRef]
- Jamison, M.T.; Dalisay, D.S.; Molinski, T.F. Peroxide Natural Products from Plakortis zyggompha and the Sponge Association Plakortis halichondrioides-Xestospongia deweerdtae: Antifungal Activity against Cryptococcus gattii. J. Nat. Prod. 2016, 79, 555–563. [Google Scholar] [CrossRef] [PubMed]
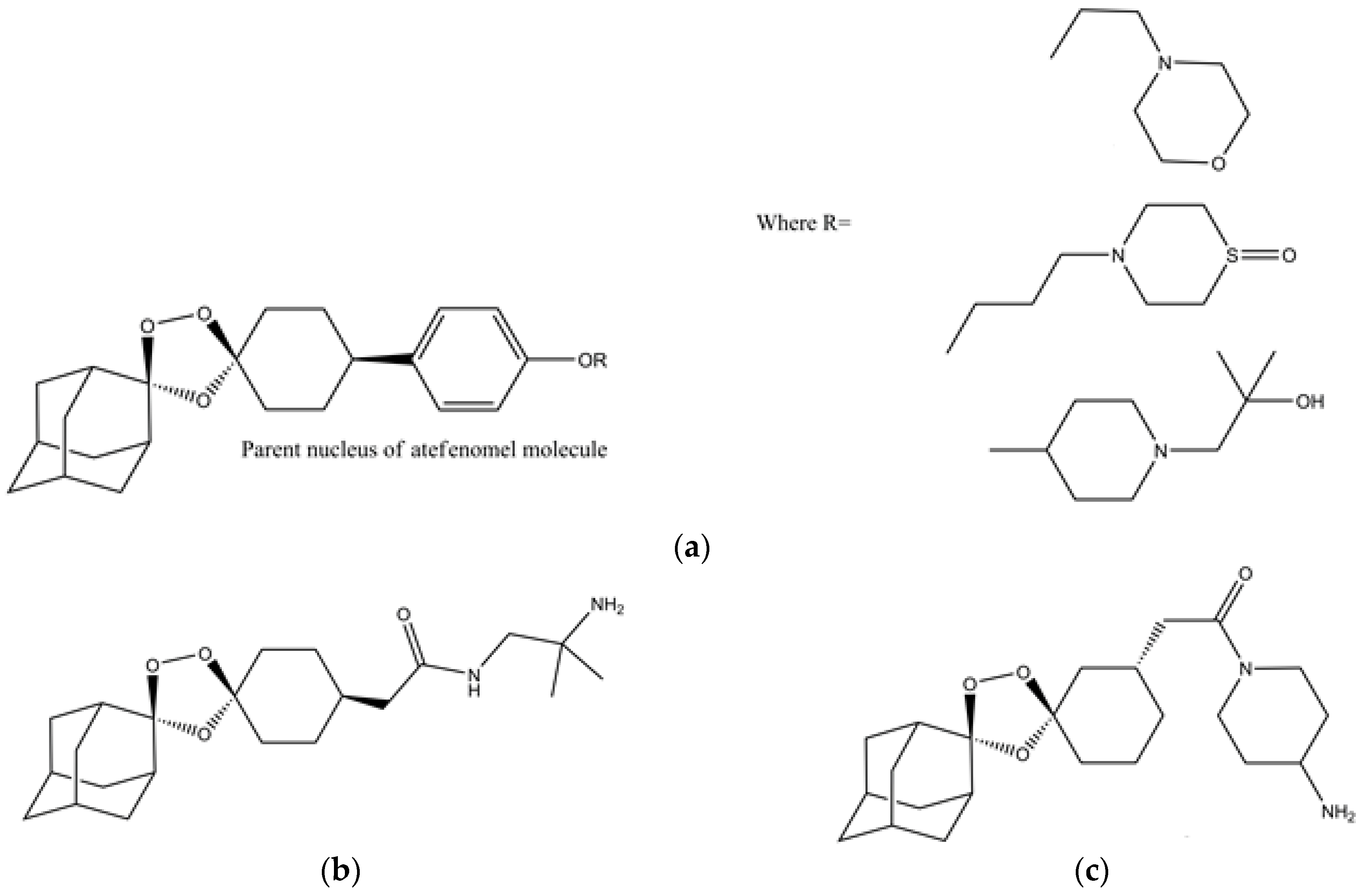
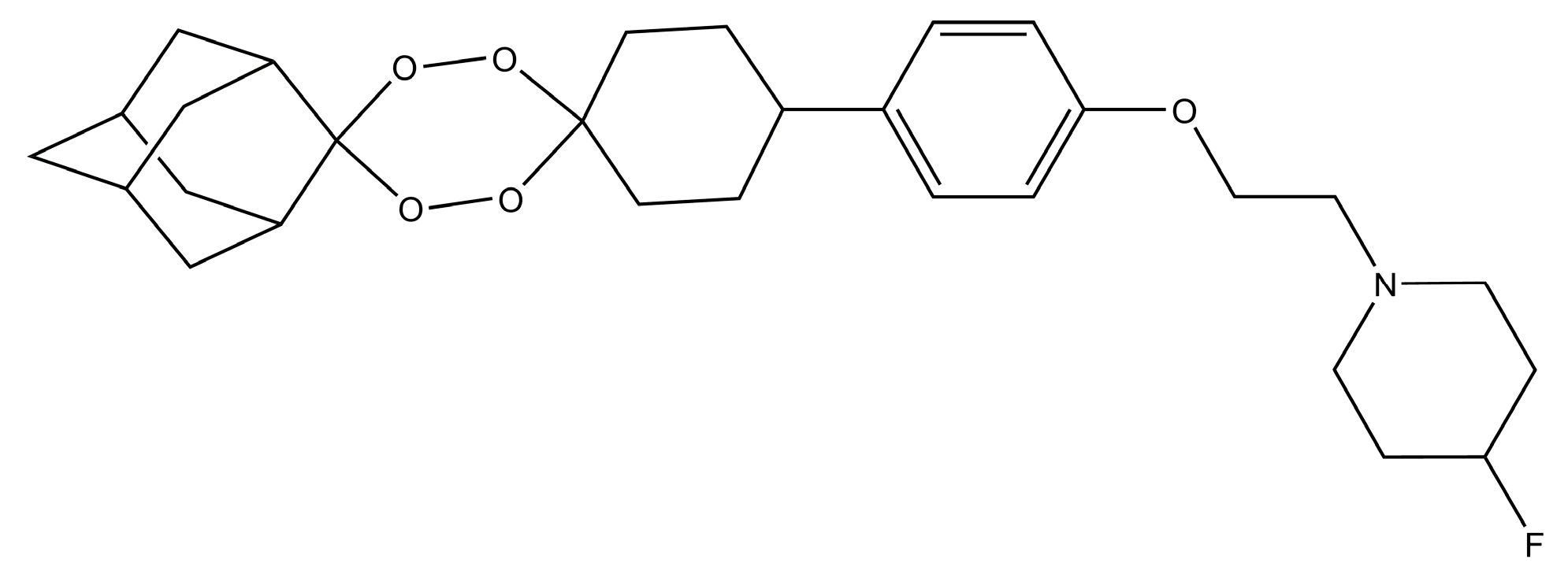
- O’Neill, P.M.; Amewu, R.K.; Charman, S.A.; Sabbani, S.; Gnädig, N.F.; Straimer, J.; Fidock, D.A.; Shore, E.R.; Roberts, N.L.; Wong, M.H.-L.; et al. A tetraoxane-based antimalarial drug candidate that overcomes PfK13-C580Y dependent artemisinin resistance. Nat. Commun. 2017, 8, 15159. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Hammill, J.T.; Guy, R.K. Seeking the Elusive Long-Acting Ozonide: Discovery of Artefenomel (OZ439). J. Med. Chem. 2017, 60, 2651–2653. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.; Amin, H.; Ullah, A.; Saba, S.; Rafique, J.; Khan, K.; Ahmad, N.; Badshah, S.L. Antioxidant and Antiplasmodial Activities of Bergenin and 11-O-Galloylbergenin Isolated from Mallotus philippensis. Oxid. Med. Cell. Longev. 2016, 2016, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Phyo, A.P.; Jittamala, P.; Nosten, F.H.; Pukrittayakamee, S.; Imwong, M.; White, N.J.; Duparc, S.; Macintyre, F.; Baker, M.; Möhrle, J.J. Antimalarial activity of artefenomel (OZ439), a novel synthetic antimalarial endoperoxide, in patients with Plasmodium falciparum and Plasmodium vivax malaria: An open-label phase 2 trial. Lancet Infect. Dis. 2016, 16, 61–69. [Google Scholar] [CrossRef]
- Patil, C.; Baig, M.; Doifode, S.; Katare, S. Fixed dose combination of arterolane and piperaquine: A newer prospect in antimalarial therapy. Ann. Med. Health Sci. Res. 2014, 4, 466. [Google Scholar] [CrossRef] [PubMed]
- Charman, S.A.; Arbe-Barnes, S.; Bathurst, I.C.; Brun, R.; Campbell, M.; Charman, W.N.; Chiu, F.C.K.; Chollet, J.; Craft, J.C.; Creek, D.J.; et al. Synthetic ozonide drug candidate OZ439 offers new hope for a single-dose cure of uncomplicated malaria. Proc. Natl. Acad. Sci. USA 2011, 108, 4400–4405. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Xie, S.C.; Cao, P.; Giannangelo, C.; McCaw, J.; Creek, D.J.; Charman, S.A.; Klonis, N.; Tilley, L. Comparison of the exposure time dependence of the activities of synthetic ozonide antimalarials and dihydroartemisinin against K13 wild-type and mutant Plasmodium falciparum Strains. Antimicrob. Agents Chemother. 2016, 60, 4501–4510. [Google Scholar] [CrossRef] [PubMed]
- Straimer, J.; Gnädig, N.F.; Stokes, B.H.; Ehrenberger, M.; Crane, A.A.; Fidock, D.A. Plasmodium falciparum K13 mutations differentially impact ozonide susceptibility and parasite fitness in vitro. mBio 2017, 8, e00172-17. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Wang, X.; Kamaraj, S.; Bulbule, V.J.; Chiu, F.C.K.; Chollet, J.; Dhanasekaran, M.; Hein, C.D.; Papastogiannidis, P.; Morizzi, J.; et al. Structure-Activity Relationship of the Antimalarial Ozonide Artefenomel (OZ439). J. Med. Chem. 2017, 60, 2654–2668. [Google Scholar] [CrossRef] [PubMed]
- Jourdan, J.; Matile, H.; Reift, E.; Biehlmaier, O.; Dong, Y.; Wang, X.; Mäser, P.; Vennerstrom, J.L.; Wittlin, S. Monoclonal Antibodies That Recognize the Alkylation Signature of Antimalarial Ozonides OZ277 (Arterolane) and OZ439 (Artefenomel). ACS Infect. Dis. 2016, 2, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.F.; Drown, B.S.; Riley, A.P.; Garcia, A.; Shirai, T.; Svec, R.L.; Hergenrother, P.J. Predictive compound accumulation rules yield a broad-spectrum antibiotic. Nature 2017, 545, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Blank, B.R.; Gut, J.; Rosenthal, P.J.; Renslo, A.R. Enantioselective Synthesis and in Vivo Evaluation of Regioisomeric Analogues of the Antimalarial Arterolane. J. Med. Chem. 2017, 60, 6400–6407. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badshah, S.L.; Ullah, A.; Ahmad, N.; Almarhoon, Z.M.; Mabkhot, Y. Increasing the Strength and Production of Artemisinin and Its Derivatives. Molecules 2018, 23, 100. https://doi.org/10.3390/molecules23010100
Badshah SL, Ullah A, Ahmad N, Almarhoon ZM, Mabkhot Y. Increasing the Strength and Production of Artemisinin and Its Derivatives. Molecules. 2018; 23(1):100. https://doi.org/10.3390/molecules23010100
Chicago/Turabian StyleBadshah, Syed Lal, Asad Ullah, Nasir Ahmad, Zainab M. Almarhoon, and Yahia Mabkhot. 2018. "Increasing the Strength and Production of Artemisinin and Its Derivatives" Molecules 23, no. 1: 100. https://doi.org/10.3390/molecules23010100
APA StyleBadshah, S. L., Ullah, A., Ahmad, N., Almarhoon, Z. M., & Mabkhot, Y. (2018). Increasing the Strength and Production of Artemisinin and Its Derivatives. Molecules, 23(1), 100. https://doi.org/10.3390/molecules23010100