β-Glucans: Relationships between Modification, Conformation and Functional Activities
Abstract
:1. Introduction
2. Effects of Physical Modification on the Conformation of β-Glucans
2.1. Ultrasonic Modification
2.2. Irradiation Modification
2.3. Microwave Modification
2.4. Other Physical Modification Methods
3. Effects of Chemical Modification on the Conformation of β-Glucan
3.1. Carboxymethylated Modification
3.2. Sulfated Modification
3.3. Phosphorylated Modification
4. Effects of Biological Modification on the Conformation of β-Glucan
5. Effects of Conformation Transformation on the Functional Activity of β-Glucan
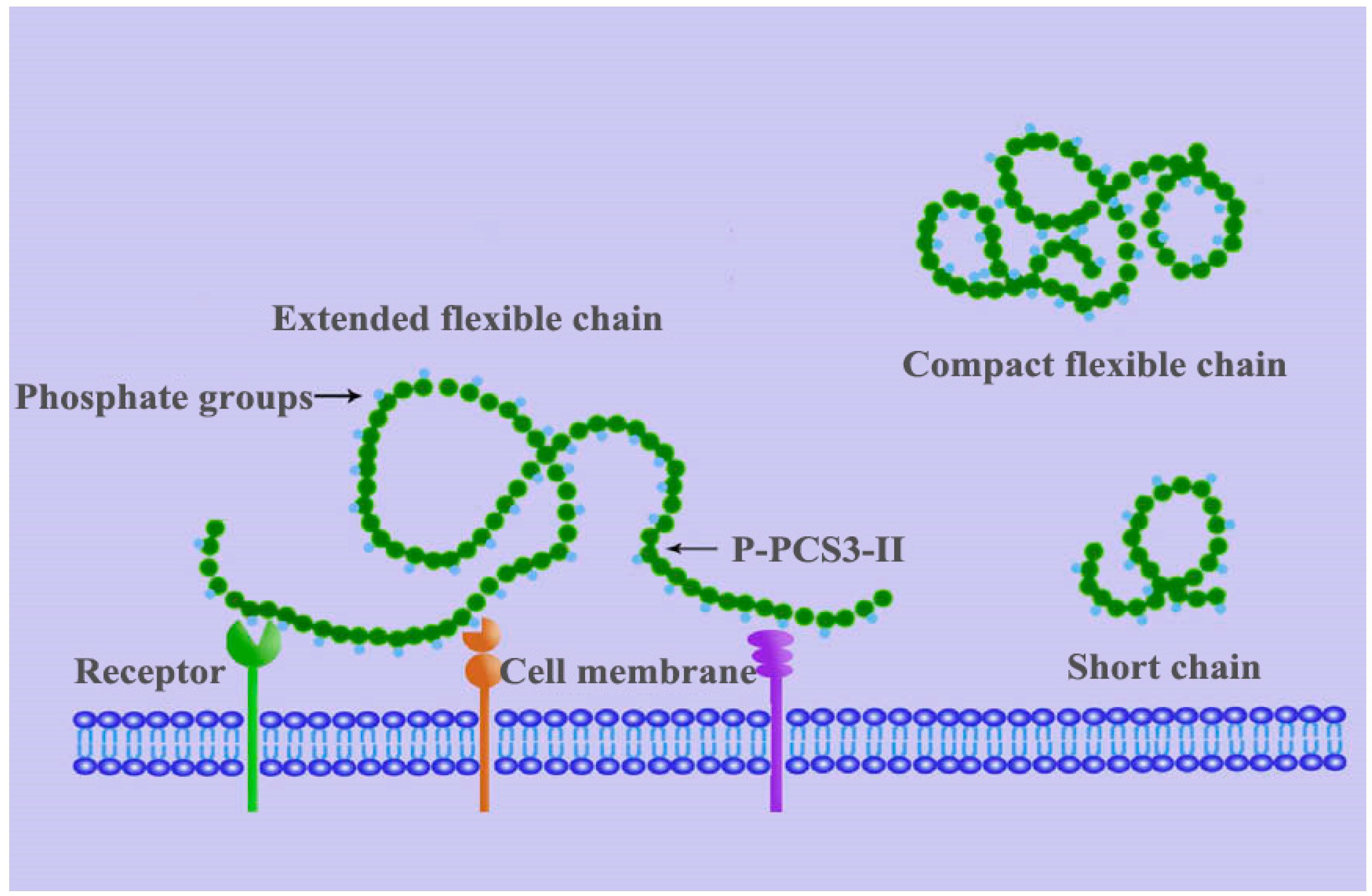
5.1. Effects of Backbone Flexibility on the Function Activity
5.2. Effects of Helix Conformation on the Function Activity
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Q.; Liu, H.Z.; Zhong, K. Review on the relationship of polysaccharide molecular chain conformation changes and bioactivities. Curr. Biotechnol. 2011, 318–326. [Google Scholar] [CrossRef]
- Li, S.; Xu, S.; Zhang, L.N. Advances in conformations and characterizations of fungi polysaccharides. Acta Polym. Sin. 2010, 1359–1375. [Google Scholar] [CrossRef]
- McIntosh, M.; Stone, B.A.; Stanisich, V.A. Curdlan and other bacterial (1→3)-d-glucanss. Appl. Microbiol. Biotechnol. 2005, 68, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Todaro, S.; Sabatinoa, M.A.; Mangione, M.R.; Picone, P.; Giacinto, M.L.D.; Bulone, D.; Dispenza, C. Temporal control of xyloglucan self-assembly into layered structures by radiation-induced degradation. Carbohydr. Polym. 2016, 152, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, X.; Zhang, L. Thermally Induced Conformation Transition of Triple-Helical Lentinan in NaCl Aqueous Solution. J. Phys. Chem. B 2008, 112, 10343–10351. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, S.; Wang, X.; Zhang, L.; Cheung, P.C.K. Advances in Lentinan: Isolation, structure, chain conformation and bioactivities. Food Hydrocoll. 2011, 25, 196–206. [Google Scholar] [CrossRef]
- Zhang, M.; Kim, J.A. Effect of molecular size and modification pattern on the internalization of water soluble β-(1→3)-(1→4)-glucan by primary murine macrophages. Int. J. Biochem. Cell Biol. 2012, 44, 914–927. [Google Scholar] [CrossRef] [PubMed]
- Jagodzinski, P.P.; Wiaderkiewicz, R.; Kurzawski, G.; Kloczewiak, M.; Nakashima, H.; Hyjek, E.; Yamamoto, N.; Uryu, T.; Kaneko, Y.; Posner, M.R.; et al. Mechanism of the Inhibitory Effect of Curdlan Sulfate on HIV-1 infection in vitro. Virology 1994, 202, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Li, J.B.; Li, L.; Li, B.; Chen, L.; Huang, G.X. Advances in ultrasonic degradation of polysaccharide. Sci. Technol. Food Ind. 2006, 181–184. [Google Scholar] [CrossRef]
- Liu, X.Y. Studies on the Preparation, Conformation and Immunobiological Activity of β-d-Glucan in Saccharomyces cerevisiae. Ph.D Thesis, Jiangnan University, Wuxi, China, 2007. [Google Scholar]
- Zhang, L.; Li, X.; Xu, X.; Zeng, F. Correlation between antitumor activity, molecular weight, and conformation of Lentinan. Carbohydr. Res. 2005, 340, 1515–1521. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.J.; Ha, Y.M.; Fan, P.; Wang, F.; Li, A. Application of radiation technology in molecular modification of polysaccharides: A review. Food Sci. 2009, 30, 403–408. [Google Scholar]
- Methacanon, P.; Weerawatsophon, U.; Tanjak, P.; Rachtawee, P.; Prathumpai, W. Interleukin-8 stimulating activity of low molecular weight β-glucan depolymerized by γ-irradiation. Carbohydr. Polym. 2011, 86, 574–580. [Google Scholar] [CrossRef]
- Tao, Y.; Xu, W. Microwave-assisted solubilization and solution properties of hyperbranched polysaccharide. Carbohydr. Res. 2008, 343, 3071–3078. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Xu, X.; Zhang, L.; Zeng, F. Chain conformation and anti-tumor activities of phosphorylated (1→3)-β-d-glucan from Poria cocos. Carbohydr. Polym. 2009, 78, 581–587. [Google Scholar] [CrossRef]
- Huang, Q.; Zhang, L. Preparation, chain conformation and anti-tumor activities of water-soluble phosphated (1→3)-α-d-glucan from Poria cocos mycelia. Carbohydr. Polym. 2011, 83, 1363–1369. [Google Scholar] [CrossRef]
- Roubroeks, J.P.; Andersson, R.; Mastromauro, D.I.; Christensen, B.E.; Åman, P. Molecular weight, structure and shape of oat (1→3),(1→4)-β-d-glucan fractions obtained by enzymatic degradation with (1→4)-β-d-glucan 4-glucanohydrolase from Trichoderma reesei. Carbohydr. Polym. 2001, 46, 275–285. [Google Scholar] [CrossRef]
- Zhang, M. Heating-induced conformational change of a novel β-(1→3)-d-glucan from Pleurotus geestanus. Biopolymers 2010, 93, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Kasaai, M.R.; Charlet, G.; Paquin, P.; Arul, J. Fragmentation of chitosan by microfluidization process. Innov. Food Sci. Emerg. Technol. 2003, 4, 403–413. [Google Scholar] [CrossRef]
- Liu, H.Z.; Li, Y.N.; Gao, J.; Shi, A.M.; Liu, L.; Hu, H.; Putri, N.; Yu, H.W.; Fan, W.; Wang, Q. Effects of microfluidization with ionic liquids on the solubilization and structure of β-d-glucan. Int. J. Biol. Macromol. Carbohydr. Polym. 2006, 64, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Kan, M.; Zhang, Y.; Liu, C.M.; Liu, W.; Wang, J. Effect of Dynamic High Pressure Microfluidization on Molecular Weight Distribution and Functional Groups of Hemicellulose B Fractions Purified from Soybean Dregs Dietary Fibers. Food Sci. 2010, 13, 15–19. [Google Scholar]
- Lagoueyte, N.; Paquin, P. Effects of microfluidization on the functional properties of xanthan gum. Food Hydrocoll. 1998, 12, 365–371. [Google Scholar] [CrossRef]
- Wang, Y.; Li, D.; Wang, L.J.; Xue, J. Effects of high pressure homogenization on rheological properties of flaxseed gum. Carbohydr. Polym. 2011, 83, 489–494. [Google Scholar] [CrossRef]
- Bonilla, J.; Atarés, L.; Vargas, M.; Chiralt, A. Effect of essential oils and homogenization conditions on properties of chitosan-based films. Food Hydrocoll. 2012, 26, 9–16. [Google Scholar] [CrossRef]
- Zhang, M.; Cheung, P.C.K.; Zhang, L.; Chiu, C.M.; Ooi, V.E.C. Carboxymethylated β-glucan from mushroom sclerotium of Pleurotus tuber-regium as novel water-soluble anti-tumor agent. Carbohydr. Polym. 2004, 57, 319–325. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, L. Chain conformation of carboxymethylated derivatives of (1→3)-β-d-glucan from Poria cocos sclerotium. Carbohydr. Polym. 2006, 65, 504–509. [Google Scholar] [CrossRef]
- Wang, M.; Ding, X. The physiochemical properties and the conformation in the solution of modified yeast glucan—CMG. Chin. J. Biochem. Mol. Biol. 1998, 14, 636–640. [Google Scholar]
- Magnani, M.; Calliari, C.M.; de Macedo, F.C., Jr.; Mori, M.P.; de Syllos Cólus, I.M.; Castro-Gomez, R.J.H. Optimized methodology for extraction of (1→3)(1→6)-β-d-glucan from Saccharomyces cerevisiae and in vitro evaluation of the cytotoxicity and genotoxicity of the corresponding carboxymethyl derivative. Carbohydr. Polym. 2009, 78, 658–665. [Google Scholar] [CrossRef]
- Wang, J.G.; Zhang, L. Structure and chain conformation of five water-soluble derivatives of a β-d-glucan isolated from Ganoderma lucidum. Carbohydr. Res. 2009, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, W.A.J.P.; Jeon, Y.J. Biological activities and potential industrial applications of fucose rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Cong, C.; Wu, W.; Xu, X. Chain conformation and anti-tumor activity of derivatives of polysaccharide from Rhizoma Panacis Japonici. Carbohydr. Polym. 2014, 105, 308–316. [Google Scholar]
- Williams, D.L.; Pretus, H.A.; McNamee, R.B.; Jones, E.L.; Ensley, H.E.; William Browder, I.; Di Luzio, N.R. Development, physicochemical characterization and preclinical efficacy evaluation of a water soluble glucan sulfate derived from Saccharomyces cerevisiae. Immunopharmacology 1991, 22, 139–156. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, L.; Wang, Y.; Cheung, P.C.K. Chain conformation of sulfated derivatives of β-glucan from sclerotia of Pleurotus tuber-regium. Carbohydr. Res. 2003, 338, 2863–2870. [Google Scholar] [CrossRef] [PubMed]
- Zjawiony, J.K. Biologically active compounds from Aphyllophorales (Polypore) fungi. J. Nat. Prod. 2004, 67, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.L.; McNamee, R.B.; Jones, E.L.; Pretus, H.A.; Ensley, H.E.; Browder, I.W.; Di Luzio, N.R. A method for the solubilization of a (1→3)-β-d-glucan isolated from Saccharomyces cerevisiae. Carbohydr. Res. 1991, 219, 203–213. [Google Scholar] [CrossRef]
- Müller, A.; Ensley, H.; Pretus, H.; McNamee, R.; Jones, E.; McLaughlin, E.; Chandley, W.; Browder, W.; Lowman, D.; Williams, D. The application of various protic acids in the extraction of (1→3)-β-d-glucan from Saccharomyces cerevisiae. Carbohydr. Res. 1997, 299, 203–208. [Google Scholar] [CrossRef]
- Duan, H.K. Anti-Tumor Activity of Enzymatic Hydrolysates of β-1,3-Glucan from Saccharomyces cerevisiae. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2007. [Google Scholar]
- Carlucci, M.J.; Ciancia, M.; Matulewicz, M.C.; Cerezo, A.S.; Damonte, E.B. Antiherpetic activity and mode of action of natural carrageenans of diverse structural types. Antivir. Res. 1999, 43, 93–102. [Google Scholar] [CrossRef]
- Mueller, A.; Raptis, J.; Rice, P.J.; Kalbfleisch, J.H.; Stout, R.D.; Ensley, H.E.; Browder, W.; Williams, D.L. The influence of glucan polymer structure and solution conformation on binding to (1→3)-d-glucan receptors in a human monocyte-like cell line. Glycobiology 2000, 10, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Rice, P.J.; Kelley, J.L.; Kogan, G.; Ensley, H.E.; Kalbfleisch, J.H.; Browder, I.W.; Williams, D.L. Human monocyte scavenger receptors are pattern recognition receptors for (1→3)-β-d-glucan. J. Leukoc. Biol. 2002, 72, 140–146. [Google Scholar] [PubMed]
- Thornton, B.P.; Vĕtvicka, V.; Pitman, M.; Goldman, R.C.; Ross, G.D. Analysis of the sugar specificity and molecular location of the beta-glucan-binding lectin site of complement receptor type 3 (CD11b/CD18). J. Immunol. 1996, 156, 1235–1246. [Google Scholar] [PubMed]
- Vetvicka, V.; Thornton, B.P.; Ross, G.D. Soluble beta-glucan polysaccharide binding to the lectin site of neutrophil or natural killer cell complement receptor type 3 (CD11b/CD18) generates a primed state of the receptor capable of mediating cytotoxicity of iC3b-opsonized target cells. J. Clin. Investig. 1996, 98, 50. [Google Scholar] [CrossRef] [PubMed]
- Misaki, A.; Kakuta, M.; Sasaki, T.; Tanaka, M.; Miyaji, H. Studies on interrelation of structure and antitumor effects of polysaccharides: Antitumor action of periodate-modified, branched (1→3)-β-d-glucan of auricularia auricula-judae, and other polysaccharides containing (1→3)-glycosidic linkages. Carbohydr. Res. 1981, 92, 115–129. [Google Scholar] [CrossRef]
- Yu, Z.; Ming, G.; Kaiping, W.; Zhixiang, C.; Liquan, D.; Jingyu, L.; Fang, Z. Structure, chain conformation and antitumor activity of a novel polysaccharide from Lentinus edodes. Fitoterapia 2010, 81, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Saitô, H.; Yoshioka, Y.; Uehara, N.; Aketagawa, J.; Tanaka, S.; Shibata, Y. Relationship between conformation and biological response for (1→3)-β-d-glucan in the activation of coagulation factor G from limulus amebocyte lysate and host-mediated antitumor activity. Demonstration of single-helix conformation as a stimulant. Carbohydr. Res. 1991, 217, 181–190. [Google Scholar] [CrossRef]
- Kagimura, F.Y.; da Cunha, M.A.; Barbosa, A.M.; Dekker, R.F.; Malfatti, C.R. Biological activities of derivatized d-glucans: A review. Int. J. Biol. Macromol. 2015, 72, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L. Correlation of Conformation Transition to Bioactivities of Lentinan from Lentinusedoes. Master’s Thesis, Wuhan University, Wuhan, China, 2004. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
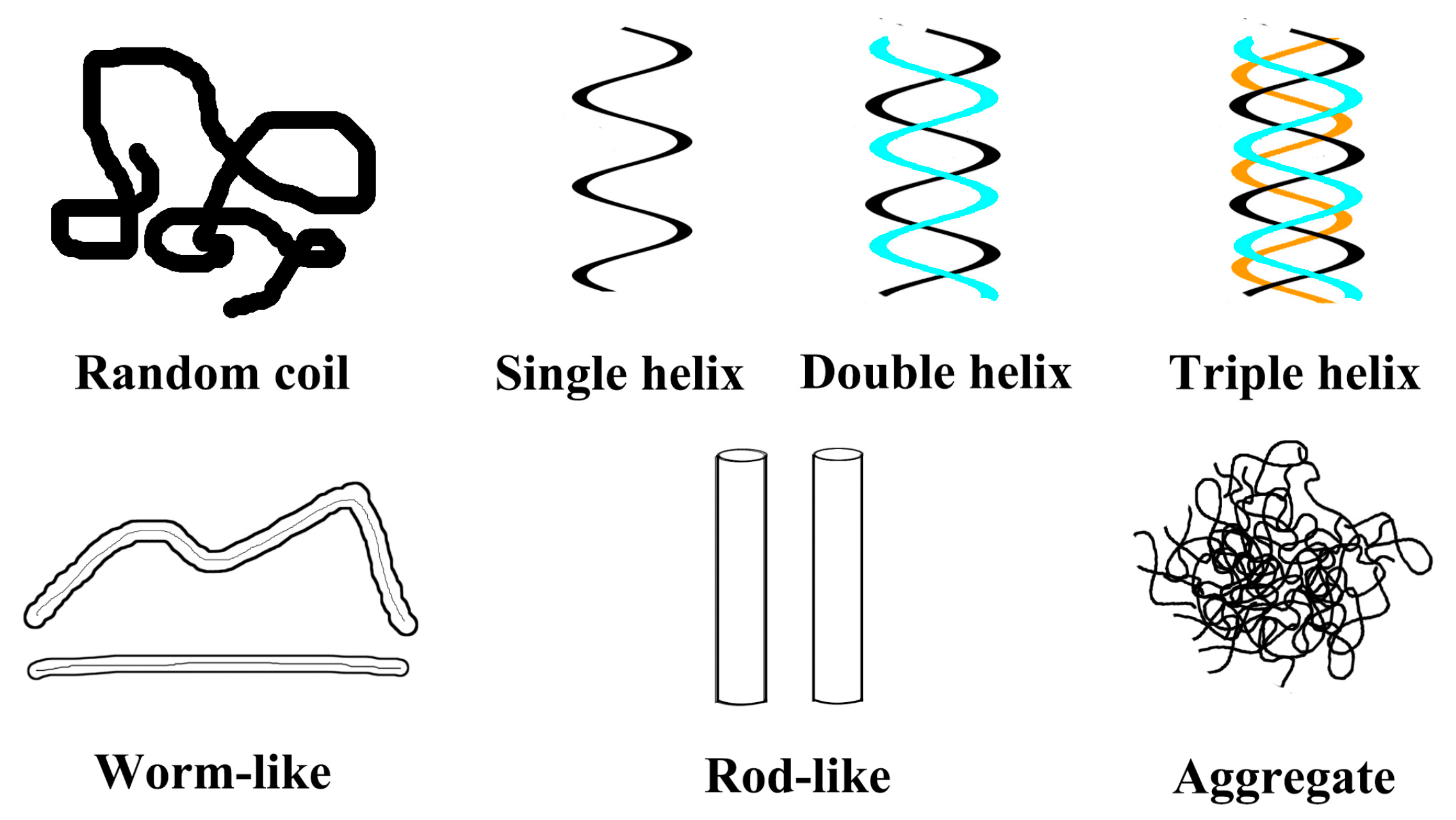
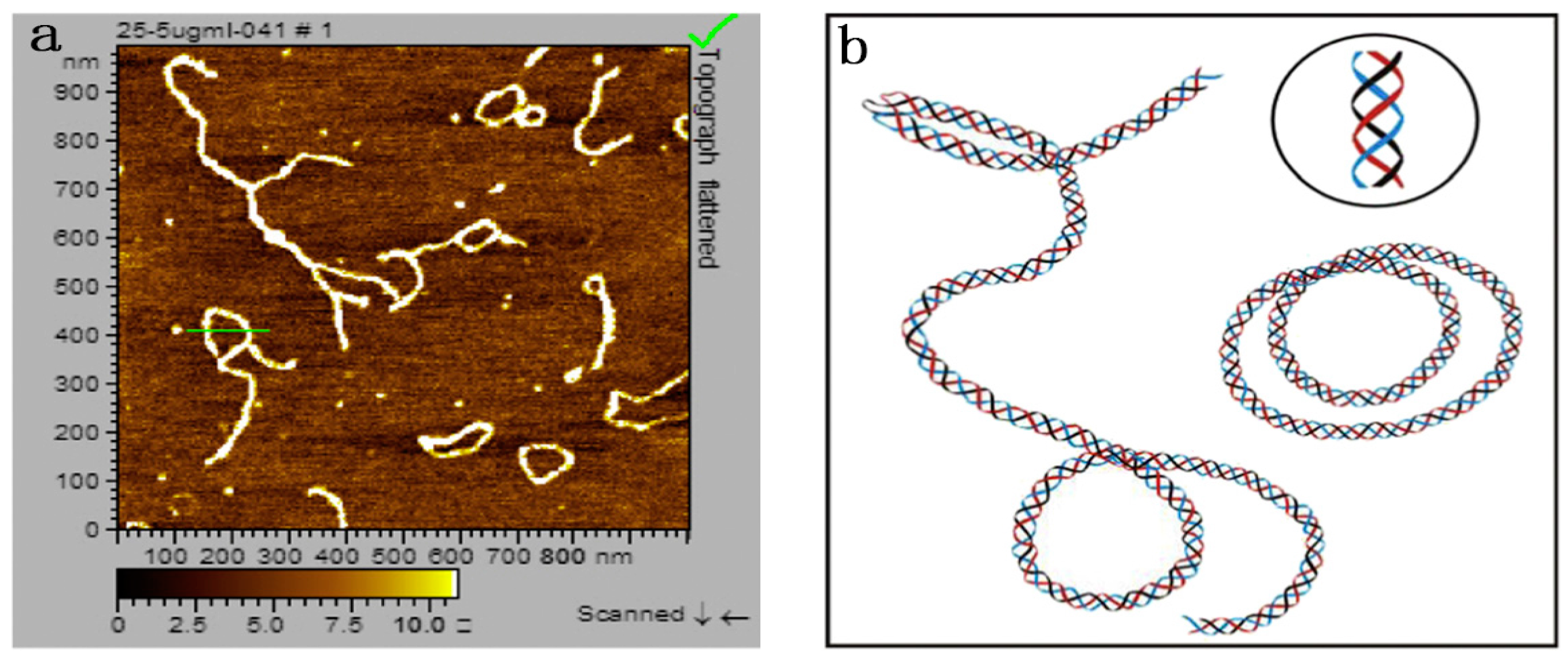
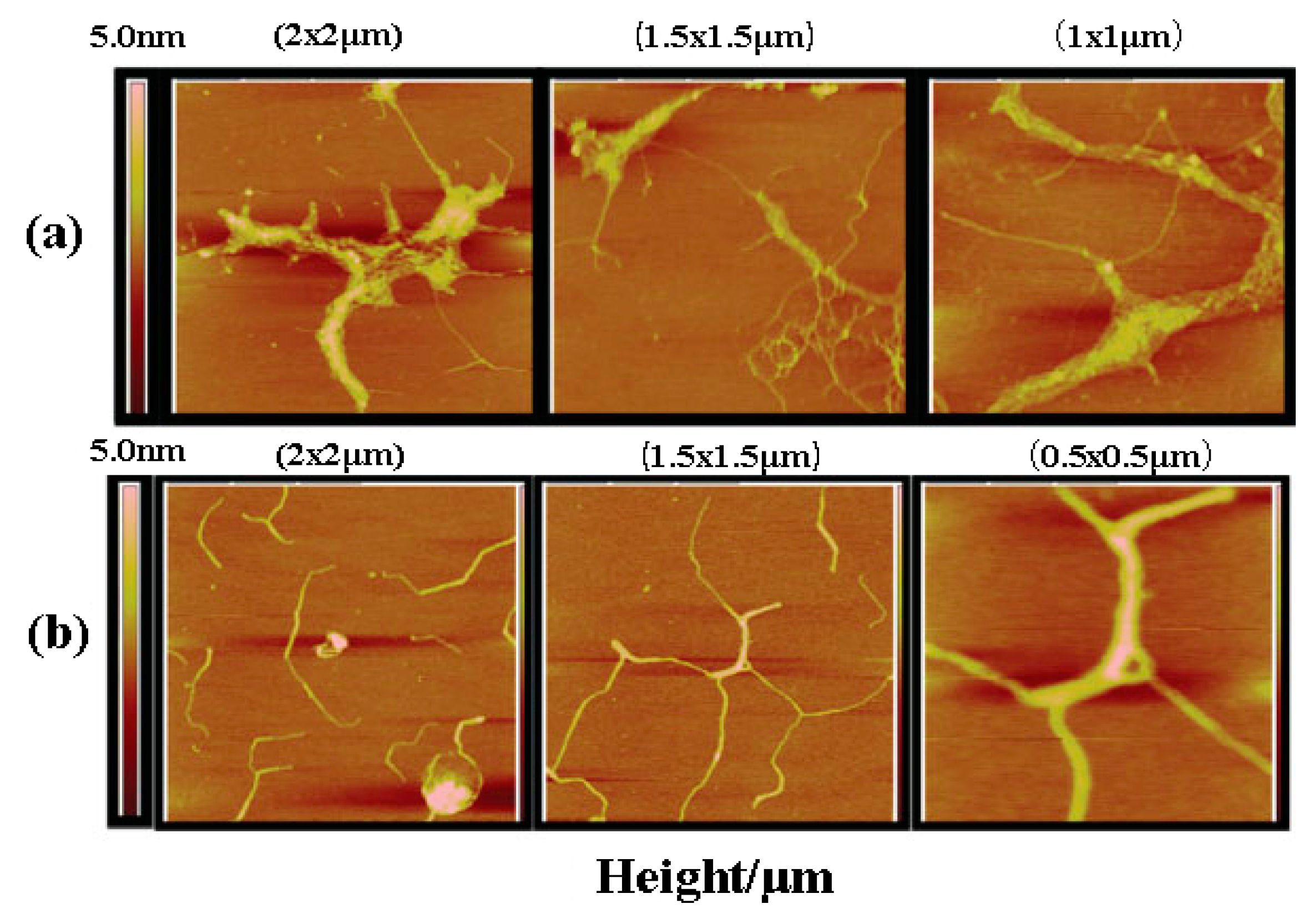
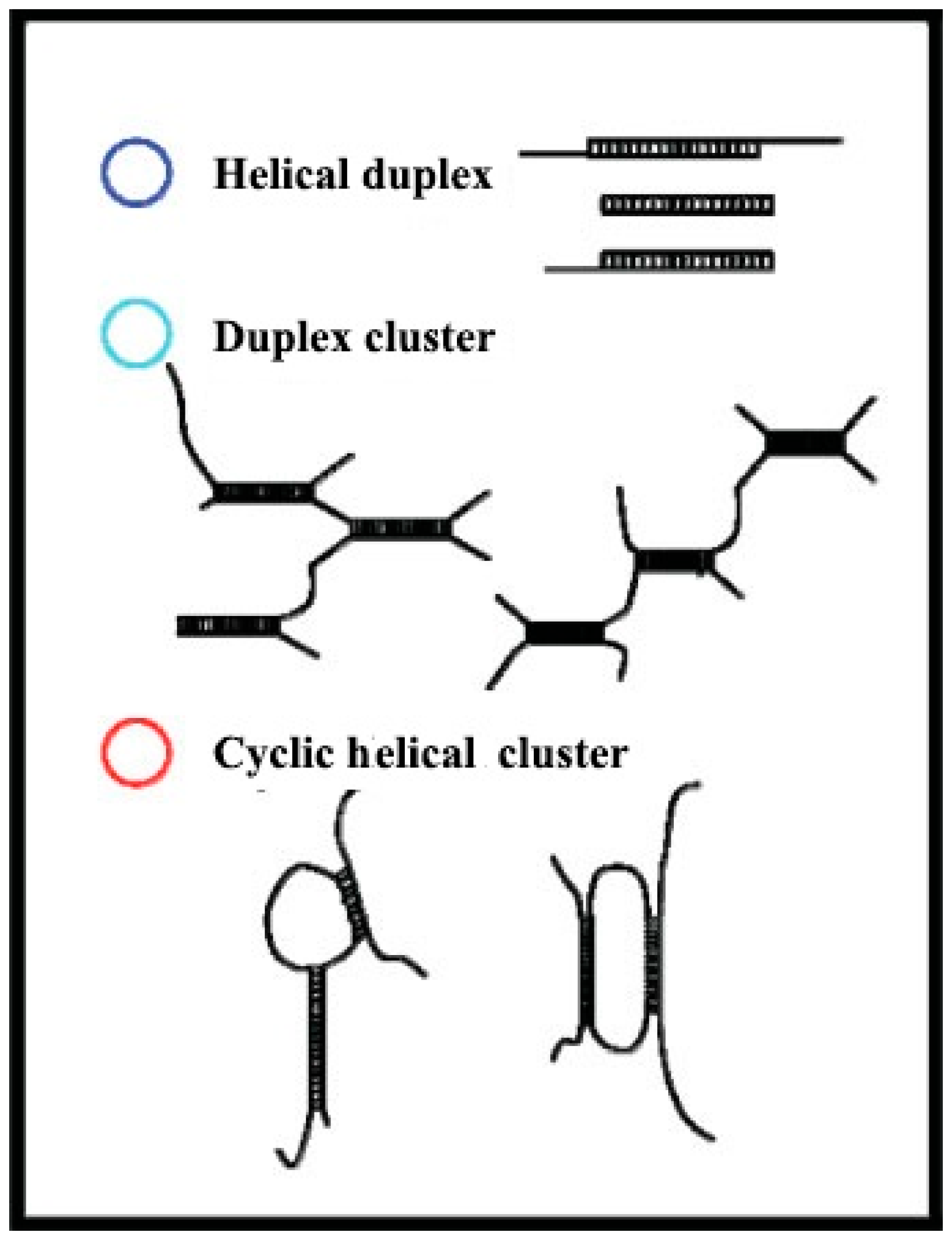
| Glucan | Modification Methods | Solution | Mw Range | Conformations and Correlation Parameters | Ref. |
|---|---|---|---|---|---|
| Pleurotus tuber-regium β-glucan | Microwave | 0.02% NaN3 | 8.20 × 105~4.88 × 106 | Sphere-like chain. [η] = 3.02 × 10−2 Mw0.44 ± 0.1 (cm3·g−1). <S2>z1/2 = 8.32 × 10−2 Mw0.38 ± 0.2(nm) | [14] |
| Yeast β-glucan | Sulfate | 0.2 M·NaCl | 2.22~8.78 × 104/5.06~8.78 × 104 | Semi-stiff chain. [η] = (5.03 ± 0.31) × 10−3 Mw0.84 ± 0.03 (cm3·g−1), Yamakawa-Fujii-Yoshizaki (YFY) worm-like cylinder model, ML = 646 nm−1, q = 5.1 nm, C∞ = 16.33. <S2>z1/2 = (2.33 ± 0.40) × 10−3 Mw0.62 ± 0.02 (nm), Kratky-Porod (KP) worm-like chain model, ML = 596 nm−1, q = 8.7 nm | [10] |
| Poria cocos β-glucan | Phosphory-lated | 0.15 M·NaCl | 2.6 × 104~26.9 × 104 | Relatively extended flexible. [η] = 6.61 × 10−3 Mw0.73 (cm3·g−1), worm-like cylinder model, ML = 818 nm−1, q = 3.0 nm, C∞ = 5.2 | [15] |
| Poria cocos mycelia α-glucan | Phosphory-lated | 0.15 M·NaCl | 1.96 × 104~15.6 × 104 | Semi-stiff chain. [η] = 2.87 × 10−3 Mw0.86 ± 0.02 (mL·g−1), ML = 780 ± 40 nm−1, q = 6.0 ± 1 nm, C∞ = 15.1 ± 1 | [16] |
| Oat β-glucan | Enzyment | 2.2 × 103~2.1 × 105 | Random coil. [η] = 1.06 × 10−2 Mw0.86 | [17] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Sheng, X.; Shi, A.; Hu, H.; Yang, Y.; Liu, L.; Fei, L.; Liu, H.
β-Glucans: Relationships between Modification, Conformation and Functional Activities
. Molecules 2017, 22, 257.
https://doi.org/10.3390/molecules22020257
Wang Q, Sheng X, Shi A, Hu H, Yang Y, Liu L, Fei L, Liu H.
β-Glucans: Relationships between Modification, Conformation and Functional Activities
. Molecules. 2017; 22(2):257.
https://doi.org/10.3390/molecules22020257
Wang, Qiang, Xiaojing Sheng, Aimin Shi, Hui Hu, Ying Yang, Li Liu, Ling Fei, and Hongzhi Liu.
2017. "β-Glucans: Relationships between Modification, Conformation and Functional Activities
" Molecules 22, no. 2: 257.
https://doi.org/10.3390/molecules22020257
Wang, Q., Sheng, X., Shi, A., Hu, H., Yang, Y., Liu, L., Fei, L., & Liu, H.
(2017). β-Glucans: Relationships between Modification, Conformation and Functional Activities
. Molecules, 22(2), 257.
https://doi.org/10.3390/molecules22020257





