Screening for Antibacterial and Antioxidant Activities and Phytochemical Analysis of Oroxylum indicum Fruit Extracts
Abstract
:1. Introduction
2. Results and Discussion
2.1. In Vitro Antibacterial Activity of O. indicum Extracts Using a Disc Diffusion Assay
2.2. In Vitro Antioxidant Activity of O. indicum Extracts by DPPH Scavenging Method
2.3. Determination Oftotal Phenolic Content in O. indicum Extracts Using the Folin-Ciocalteu Method
2.4. Determination of Total Flavonoid Content in O. indicum Extracts Using the Aluminium Chloride Method
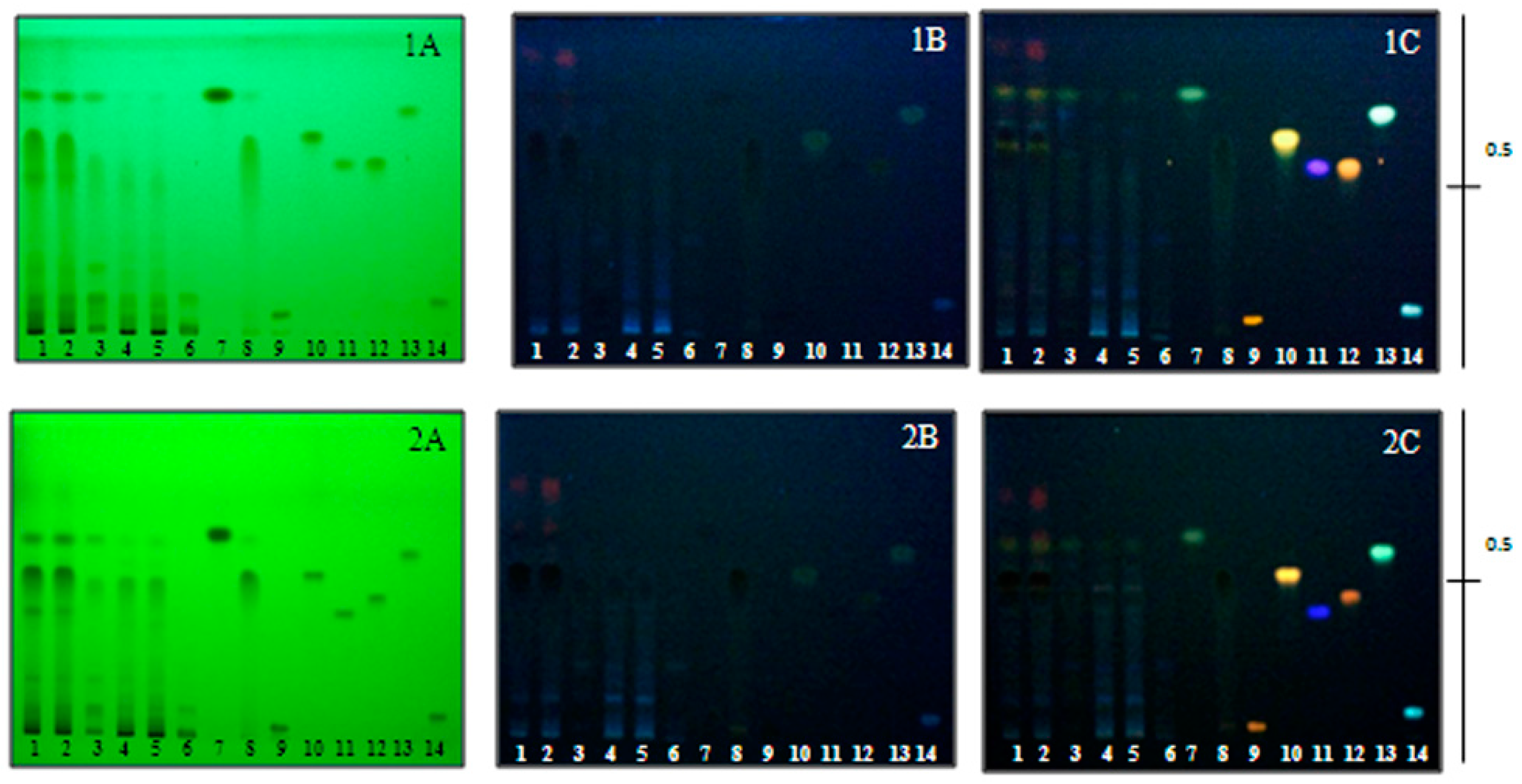
2.5. Thin Layer Chromatography (TLC) Analysis
3. Materials and Methods
3.1. Preparation of Oroxylum Indicum Fruit Extracts
3.1.1. Plant Material Preparation
3.1.2. Plant Extract Preparations
3.2. Determination of in Vitro Antibacterial Activity of O. indicum Extracts Using Disc Diffusion Assay
3.3. Determination of in Vitro Antioxidant Activity of O. indicum Extracts Using DPPH Scavenging Method
3.4. Phytochemical Analysis
3.4.1. Determination for Total Phenolic Content in O. indicum Extracts Using the Folin-Ciocalteu Method
3.4.2. Determination of Total Flavonoid Content in O. indicum Extracts Using the Aluminium Chloride Method
3.4.3. Thin Layer Chromatography (TLC) Analysis
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McGaw, L.J.; Van der, M.D.; Eloff, J.N. In vitro anthelmintic, antibacterial and cytotoxic effects of extracts from plants used in South African ethnoveterinary medicine. Vet. J. 2007, 173, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Nazir, M.; Ali, S.M.; Hussain, H.; Lee, S.Y.; Riaz, N.; Jabbar, A. Antimicrobial natural products: an update on future antibiotic drug candidates. R. Soc. Chem. 2010, 27, 238–254. [Google Scholar]
- Harminder, S.V.; Chaudhary, A.K. A review on the taxonomy, ethnobotany, chemistry and pharmacology of Oroxylum indicum Vent. Indian J. Pharm. Sci. 2011, 73, 483–490. [Google Scholar] [PubMed]
- Warrier, P.K.; Nambiar, V.P.K.; Ramankutty, C. Indian Medicinal Plant: A Compendium of 500 Species (4); Orient Longman: Hyderabad, India, 1993; pp. 186–190. [Google Scholar]
- Chauhan, S.N. Medicinal and Aromatic Plants of Himachal Pradesh; Indus Publishing: New Dehli, India, 1999; pp. 296–298. [Google Scholar]
- Subramaniam, S.S.; Nair, A.G. Flavonoids of the stem bark of Oroxylum indicum. Curr. Sci. 1972, 41, 62–63. [Google Scholar]
- Jayaram, K.; Prasad, M.N. Genetic diversity in Oroxylum indicum (L.) Vent. (Bignoniaceae), a vulnerable medicinal plant by random amplified polymorphic DNA marker. Afr. J. Biotechnol. 2008, 7, 254–262. [Google Scholar]
- Bhattacharje, A.K.; Das, A.K. Phytochemical screening of some Indian plants. Q. J. Crude Drug Res. 1969, 9, 1408–1412. [Google Scholar] [CrossRef]
- Chen, L.J.; Games, D.E.; Jones, J. Isolation and identification of four flavonoid constituents from the seeds of Oroxylum indicum by high-speed counter-current chromatography. J. Chromatogr. A 2003, 988, 95–105. [Google Scholar] [CrossRef]
- Chen, L.J.; Song, H.; Lan, X.Q.; Games, D.E.; Sutherland, I.A. Comparison of high-speed counter-current chromatography instruments for the separation of the extracts of the seeds of Oroxylum indicum. J. Chromatogr. A 2005, 1063, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Zaveri, M.; Khandhar, A.; Jain, S. Quantification of baicalein, chrysin, biochanin-A and ellagic acid in root bark of Oroxylum indicum by RP-HPLC with UV detection. Eurasian J. Anal. Chem. 2008, 3, 245–257. [Google Scholar]
- Nijveldt, R.J.; van Nood, E.; van Hoorn, D.E.C.; Boelens, P.G.; van Norren, K.; van Leeuwen, P.A.M. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar] [PubMed]
- Sithisarn, P.; Supabphol, R.; Gritsanapan, W. Comparison of free radical scavenging activity of Siamese neem tree (Azadirachta indica A. Juss var. siamensis Valeton) leaf extracts prepared by different methods of extraction. Med. Princ. Pract. 2006, 15, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Hosein, H.K.M.; Zinab, D. Phenolic compounds and antioxidant activity of henna leaves extracts (Lawsonia inermis). World J. Dairy Food Sci. 2007, 2, 38–41. [Google Scholar]
- Samatha, T.; Shyamsundarachary, R.; Srinivas, P.; Swamy, N.R. Quantification of total phenolic and total flavonoid contents in extracts of Oroxylum indicum L. Kurz. Asian J. Pharm. Clin. Res. 2012, 5, 177–179. [Google Scholar]
- Moirangthem, D.S.; Talukdar, N.C.; Bora, U.; Kasoju, N.; Das, R.K. Differential effects of Oroxylum indicum bark extracts: Antioxidant, antimicrobial, cytotoxic and apoptotic study. Cytotechnology 2013, 65, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.Y.; Cao, Y.Y.; Chen, C.Y.; Dai, H.Q.; Yu, S.X.; Wei, J.J.; Li, H.; Yang, B. Antioxidant flavonoids from the seed of Oroxylum indicum. Fitoterapia 2011, 82, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Babu, T.H.; Manjulatha, K.; Kumar, G.S.; Hymavathi, A.; Tiwari, A.K.; Purohit, M.; Rao, J.M.; Babu, K.S. Gastroprotective flavonoid constituents from Oroxylum indicum Vent. Bioorg. Med. Chem. Lett. 2010, 20, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.R.; Houghton, P.J.; Raman, A.; Hoult, J.R.S. Antimicrobial and anti-inflammatory activities of extracts and constituents of Oroxylum indicum (L.) Vent. Phytomedicine 1998, 5, 375–381. [Google Scholar]
- Sithisarn, P.; Jarikasem, S. Antioxidant Activity of Acanthopanax trifoliatus. Med. Princ. Pract. 2009, 18, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Herald, J.T.; Gadgil, P.; Tilley, M. High-throughput microplate assays for screening flavonoid content and DPPH-scavenging activity in sorghum bran and flour. J. Food Agric. 2012, 23, 26–31. [Google Scholar]
- Mayur, B.; Sandesh, S.; Shruti, S.; Yum, S.S. Antioxidant and alpha glucosidase inhibitory properties of Carpesium abrotanoides I. J. Med. Plant Res. 2010, 15, 47–53. [Google Scholar]
- Sample Availability: Samples of the compounds are not available from the authors.
| Bacterial Strain | Concentration (mg/mL) | Zone of Inhibition (mm) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OPCD | OPCE | OPND | OPNE | OSBD | OSBE | AMC 30 µg | CN 10 µg | SXT 25 µg | ||
| Staphylococcus intermedius | 1.5625 | 0 | 0 | 0 | 0 | 0 | 0 | 24.44 ± 0.73 | 15.00 ± 0.50 | 0 |
| 6.25 | 0 | 0 | 0 | 0 | 0 | 6.78 ± 1.54 | ||||
| 25 | 0 | 0 | 0 | 0 | 0 | 7.34 ± 1.87 | ||||
| 100 | 0 | 8.33 ± 0.71 b | 0 | 8.56 ± 0.73 d | 7.83 ± 0.98 | 7.89 ± 4.89 | ||||
| 125 | 0 | 9.28 ± 0.36 b | 0 | 10.22 ± 0.83 | - | - | ||||
| 250 | 6.22 ± 0.44 a,1,2 | 10.33 ± 0.43 b,1,3 | 6.72 ± 0.51 c,3,4 | 11.33 ± 0.83 2,4 | - | - | ||||
| 500 | 8.72 ± 0.44 a,5,6 | 11.56 ± 0.46 b,5,7 | 8.67 ± 0.50 c,7,8 | 13.50 ± 1.32 5,6,8 | - | - | ||||
| 1000 | 11.17 ± 0.35 a,9 | 12.67 ± 0.35 b | 11.17 ± 0.61 c,10 | 15.11 ± 2.10 d,9,10 | - | - | ||||
| Streptococcus suis | 1.5625 | 0 | 0 | 0 | 0 | 0 | 8.22 ± 0.97 | 32.56 ± 0.53 | 0 | 0 |
| 6.25 | 0 | 0 | 0 | 0 | 0 | 8.56 ± 1.34 | ||||
| 25 | 0 | 0 | 0 | 0 | 0 | 8.78 ± 1.86 | ||||
| 100 | 0 | 0 | 0 | 0 | 0 | 7.56 ± 2.69 | ||||
| 125 | 0 | 0 | 0 | 0 | - | - | ||||
| 250 | 0 | 7.28 ± 0.44 f | 0 | 8.06 ± 0.39 h | - | - | ||||
| 500 | 7.00 ± 1.00 e,11 | 8.67 ± 1.00 | 7.72 ± 0.75 g,12 | 11.33 ± 1.00 11,12 | - | - | ||||
| 1000 | 10.78 ± 0.67 e | 10.61 ± 0.99 f | 10.28 ± 0.91 g | 14.39 ± 2.47 h | - | - | ||||
| Extracts | DPPH Assay EC50 (µg/mL) | Total Phenolic Content (g% GAE) | Total Flavonoid Content (g% QE) |
|---|---|---|---|
| OPCD | 171.30 ± 8.46 e | 3.22 ± 0.10 b | 1.68 ± 0.13 a |
| OPCE | 65.89 ± 5.48 c | 4.57 ± 0.45 c | 4.68 ± 0.12 c |
| OPND | 220.50 ± 14.50 f | 2.70 ± 0.05 a | 1.49 ± 0.18 a |
| OPNE | 84.29 ± 5.16 d | 4.39 ± 0.19 c | 3.76 ± 0.67 b,c |
| OSBD | 38.87 ± 3.90 b | 8.21 ± 0.62 d | 3.94 ± 0.22 b |
| OSBE | 26.33 ± 0.84 a | 10.66 ± 0.27 e | 7.16 ± 0.06 d |
| Ascorbic acid | 3.86 ± 0.12 | - | - |
| Baicalein | 3.17 ± 0.05 | - | - |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sithisarn, P.; Nantateerapong, P.; Rojsanga, P.; Sithisarn, P. Screening for Antibacterial and Antioxidant Activities and Phytochemical Analysis of Oroxylum indicum Fruit Extracts. Molecules 2016, 21, 446. https://doi.org/10.3390/molecules21040446
Sithisarn P, Nantateerapong P, Rojsanga P, Sithisarn P. Screening for Antibacterial and Antioxidant Activities and Phytochemical Analysis of Oroxylum indicum Fruit Extracts. Molecules. 2016; 21(4):446. https://doi.org/10.3390/molecules21040446
Chicago/Turabian StyleSithisarn, Patchima, Petcharat Nantateerapong, Piyanuch Rojsanga, and Pongtip Sithisarn. 2016. "Screening for Antibacterial and Antioxidant Activities and Phytochemical Analysis of Oroxylum indicum Fruit Extracts" Molecules 21, no. 4: 446. https://doi.org/10.3390/molecules21040446
APA StyleSithisarn, P., Nantateerapong, P., Rojsanga, P., & Sithisarn, P. (2016). Screening for Antibacterial and Antioxidant Activities and Phytochemical Analysis of Oroxylum indicum Fruit Extracts. Molecules, 21(4), 446. https://doi.org/10.3390/molecules21040446





