2. Current Data Regarding the Biosynthesis of Xanthones within Lichens
As with most lichen metabolites [
6], the biosynthesis of xanthones proceeds through the polyacetate/polymalonate pathway, featuring the internal cyclization of a single folded polyketide chain. Two distinct series of xanthones are obtained, depending on this folding pattern.
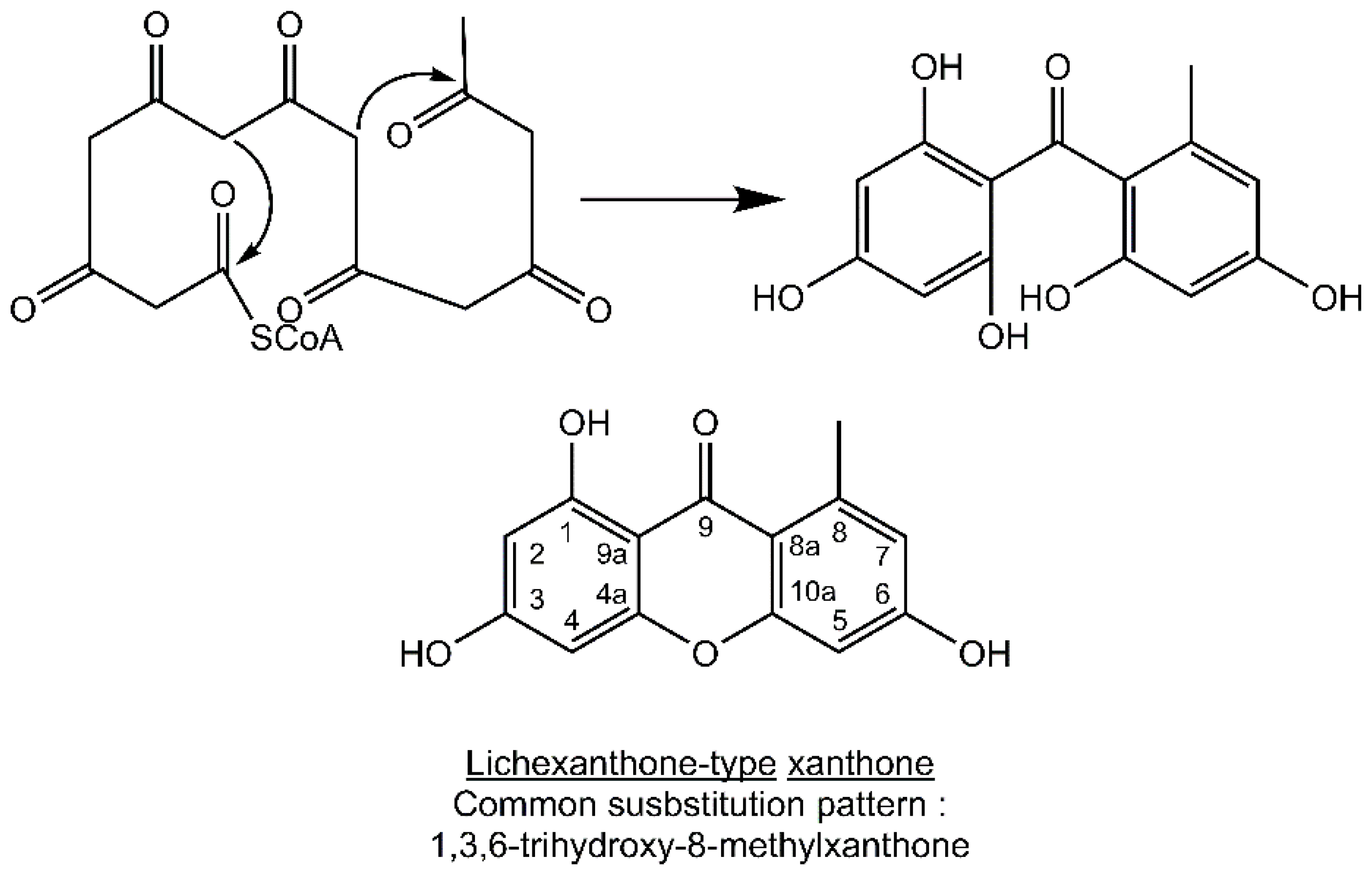
Most lichen xanthones arise through the folding of a polyketide intermediate as described by Cacho
et al. [
7], resulting in structures displaying a methyl group in position 8 (
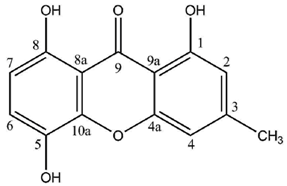
Figure 1). Aldol condensation and Claisen-type cyclization and release and yield a benzophenone intermediate that might spontaneously dehydrate to obtain the central pyrone core. This biosynthetic scheme gives rise to the common oxygen substitution pattern of lichexanthone and norlichexanthone (1,3,6-trihydroxy-8-methylxanthone).
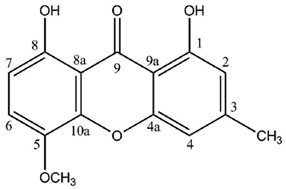
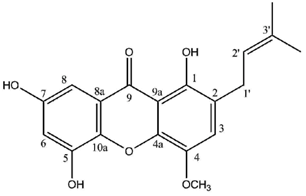
In contrast, a limited number of structures arise via a distinct biosynthetic pathway that leads to the ravenelin skeleton, with the methyl group in position 3. This biosynthetic scheme begins with the widespread anthraquinone emodin as a precursor [
8] (
Figure 2). To begin with, the hydroxyl group on C-6 of the emodin disappears (yielding chrysophanol) as it was observed in cell-free preparations of the fungus
Pyrenochaeta terrestris [
9,
10]. The hydroxyl group on C-4 is then incorporated after the oxidative ring opens [
11]. Deeper insights into this latter biosynthetic event were discussed by Henry and Townsend [
12], who proposed an aryl epoxidation across an A-ring edge of chrysophanol to yield an intermediate that lost its A-ring aromaticity. Under this scheme, this intermediate, stabilized by a hydrogen bond between its newly formed phenol group and the neighboring quinone group, recovers its A-ring aromaticity to grant islandicin as a shunt product. An alternative for this intermediate is to undergo a second oxidation, most likely by the same P450 oxygenase, to afford a Baeyer-Villiger cleavage of the central quinone ring to yield an
ortho carboxybenzophenone that might follow several metabolic fates. A first possibility is the 1,4-addition of a B-ring phenol to the A-ring dienone followed by dehydration and decarboxylation to access ravenelin-like xanthones after a final oxidation [
12]. This results in xanthones displaying an archetypical 1,4,8-trihydroxy-3-methylxanthone skeleton. A second metabolic pathway, granting access to eumitrins and secalonic acids, is assumed to include a methylation of the carboxy group to prevent its subsequent elimination after a similar 1,4-addition. Finally, a subsequent 1,2-addition to the benzophenone intermediate leads to further cores similar to that of tajixanthone produced by
Aspergillus variecolor, a skeleton thus far unknown from lichens.
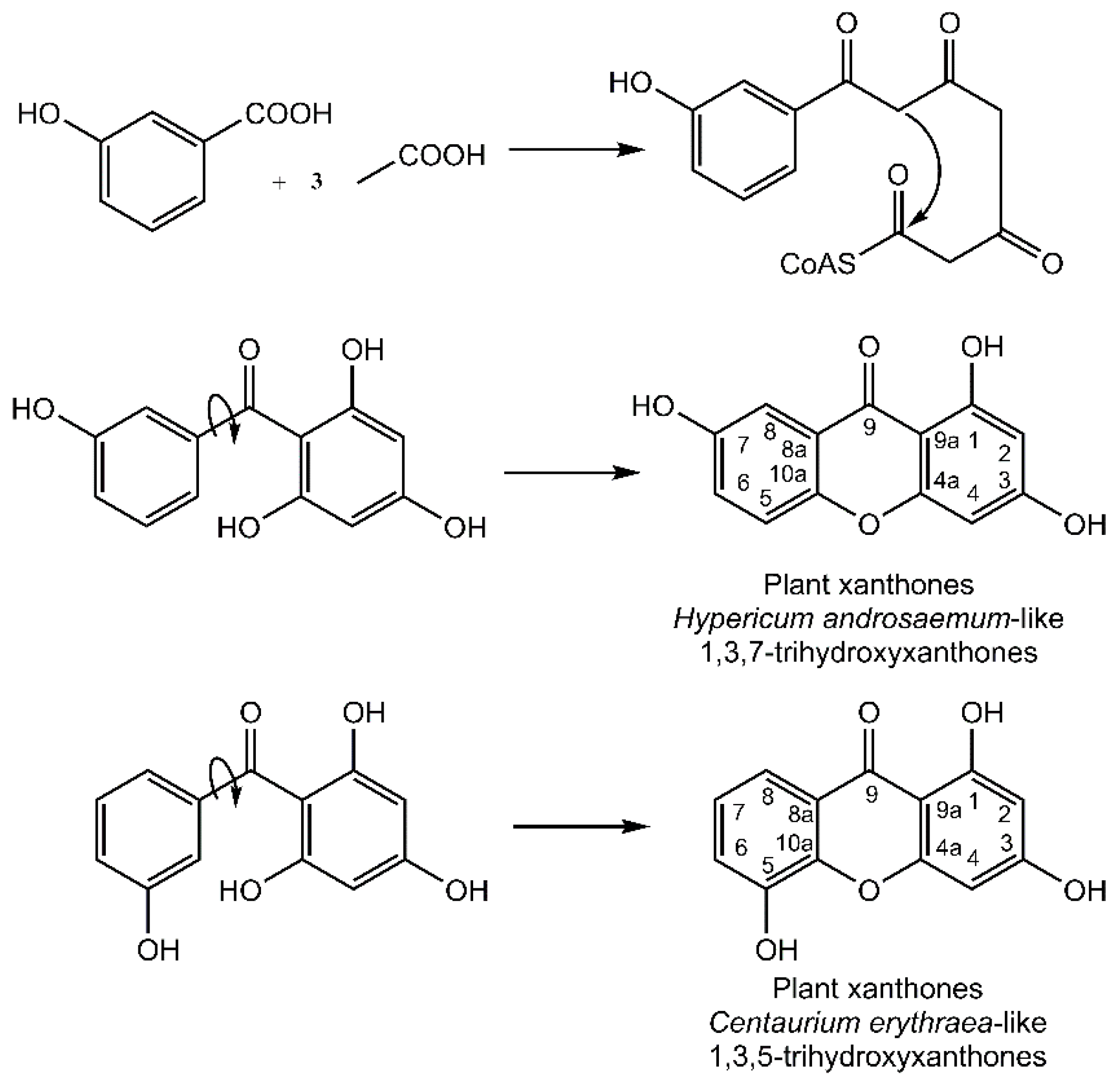
It is noteworthy that the xanthone nucleus of plants is of mixed biosynthetic origin with the A-ring being acetate-derived whereas shikimic acid pathway–derived 3-hydroxybenzoic acid gives rise to the C-ring (
Figure 3). Aromatization of the side chain leads to a freely rotating benzophenone intermediate that can further cyclize to yield the xanthone core. Regioselective oxidative coupling can then yield a 1,3,7-trihydroxyxanthone (
Hypericum androsaemum) or 1,3,5-trihydroxyxanthone (
Centaurium erythraea) [
4].
Given these different biosynthetic pathways, only few lichen xanthones are known from non-lichenized organisms. However, some can be produced by higher plants, such as lichexanthone (e.g.,
Anthocleista djalonensis [
13],
Croton cuneatus [
14]
, Cupania cinerea [
15]
, Feroniella lucida [
16]
, Minquartia guianensis [
17],
Zanthoxylum microcarpum [
18],
Z. valens [
18]), vinetorin (
Hypericum ascyron [
19]). Likewise, non-lichenized fungi have the ability to synthesize some lichen xanthones, such as lichexanthone from various
Penicillium [
20], norlichexanthone from
Penicillium patulum [
21] and the endolichenic fungus
Ulocladium [
22]; 1,3,6-trihydroxy-8-methylxanthone (also known as griseoxanthone C) is a precursor of aflatoxins—a group of significant environmental mycotoxins—first reported from
Penicillium patulum [
23]. Secalonic acids are also mycotoxins produced by a wide array of fungi [
24].
Likewise, very few xanthones are common to higher plants and fungi, with some such examples being 1,7-dihydroxyxanthone (known from the plants
Vismia parviflora [
25] and
Weddelina squamulosa [
26] while also being produced by a
Penicillium strain [
27]) 1,8-dihydroxy-3-methoxy-6-methylxanthone (plant
Cassia obtusifolia [
28] and fungus
Astrocystis sp. BCC 22166 [
29]), pinselin (plant
Cassia occidentalis [
30] and several fungal strains including an endophytic
Phomopsis sp. [
31],
Talaromyces bacillosporus [
32] and the marine-derived
Engyodontium album [
33]), 6-
O-methyl-2-deprenylrheediaxanthone B (plant
Garcinia vieillardii [
34] and fungus
Phomopsis sp. [
31]), as well as 8-desoxygartanin (produced by both
Garcinia mangostana and
Streptomyces rishiriensis 265-P5921 (according to the Dictionary of Natural Products)). It can therefore be stated that xanthones are highly unique to each realm, legitimating joint efforts on higher plants, non-lichenized fungi and lichens to widen the chemical diversity of these privileged structures.
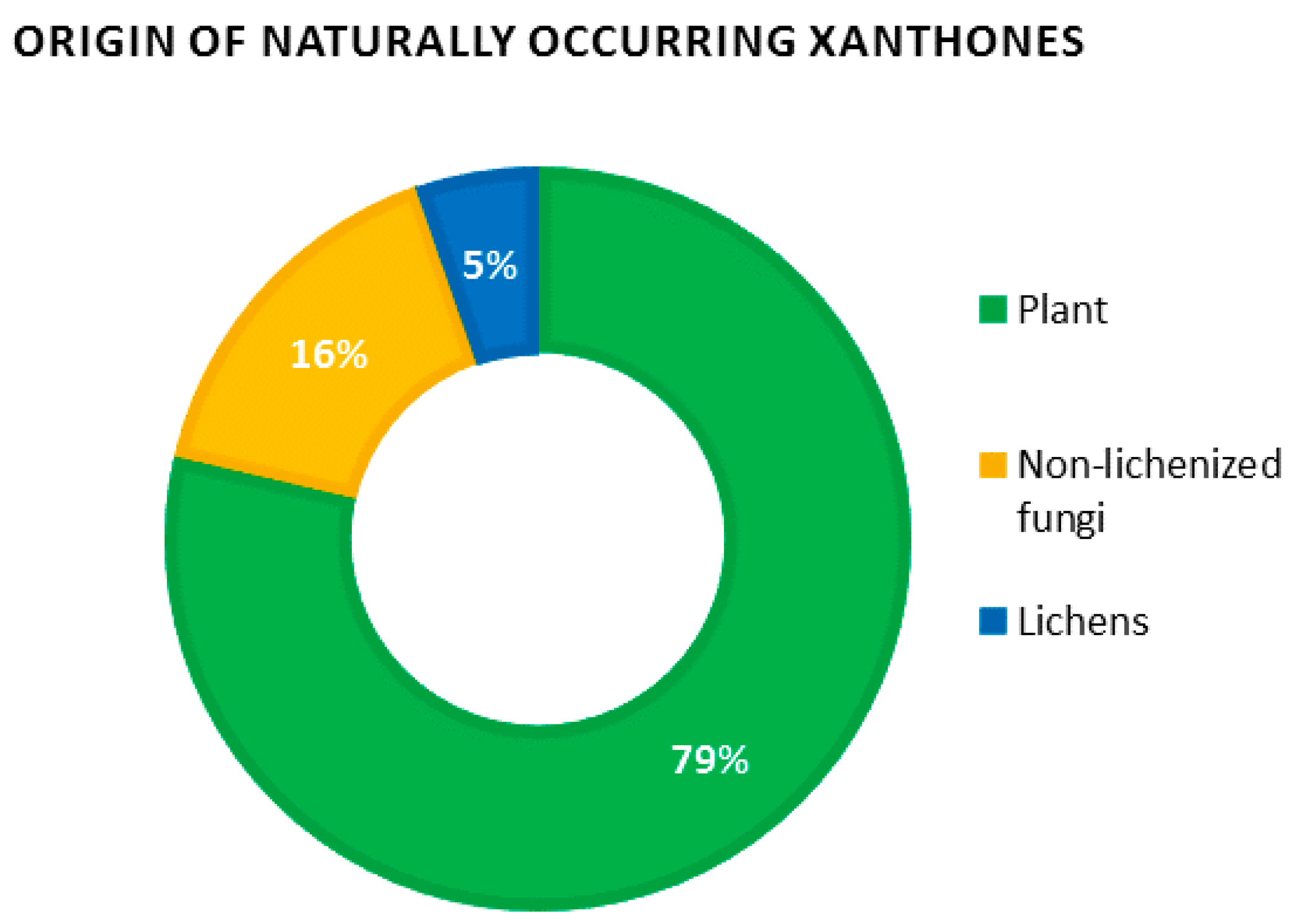
Recent data regarding the number of naturally occurring xanthones are scarce, with the last numbered record of 278 xanthones listed by Vieira and Kijjoa more than 10 years ago [
3]. By January 2016, the Dictionary of Natural Products revealed a dramatic increase in the number of natural xanthones with
ca. 2000 occurrences of xanthones
sensu lato (
i.e., including their reduced derivatives di-, tetra- and hexahydroxanthones). These considerable efforts might be explained by the discovery of promising leads such as the anti-angiogenic molecule Vadimezan (AS 404) which is currently undergoing phase III clinical trials as a tumor vascular-disrupting agent [
35,
36], as well as the pleiotropic pharmacological activities of mangosteen xanthones which have garnered considerable interest from the scientific community [
37,
38]. Obviously, plants remain the prevalent source of xanthones, concentrating almost 80% of natural xanthones. Non-lichenized fungi represent a further 15% while lichens account for the remaining 5% (
Figure 4).
Structural diversity of lichen xanthones mostly stems from variations in the orientation and degree of chlorination of the norlichexanthone or ravenelin core, as well as from the position and extent of methylation of the phenolic groups. Elix and Crook tremendously advanced the understanding of chlorination and methylation processes affecting the xanthone core. Through the example of 40 different species of lichens, theoretical biosynthetic schemes mirroring the sequence of biogenetic events leading to the joint occurrence of xanthones among lichens were constructed [
39]. This results in chemosyndromes in which the biosynthetic intermediates can be expected alongside the final xanthone. As an example, isoarthothelin (=2,5,7-trichloronorlichexanthone) might be biosynthesized from 2,4-dichloronorlichexanthone and 2,5-dichloronorlichexanthone. Therefore, the joint occurrence of these species is referred to as the isoarthothelin chemosyndrome [
40].
Further structural modifications might be taken into account to extend the chemodiversity of lichen xanthones. Important contributors to the chemical diversity of xanthones are prenyl groups which are most often incorporated as dimethylallyl moieties [
41]. Generally speaking, prenylated secondary metabolites are of paramount interest since their bioactivities are often distinct from those of their non-prenylated precursors [
42]. Even though prenylxanthones have been known to come from fungi for a long time [
43], no reports of such xanthones were made by the time Huneck and Yoshimura published their compendium of lichen substances [
5] since the first lichen prenylxanthones were reported by Rezanka
et al. in 2002 [
44]. Underlying biosynthetic pathways were recently outlined within the prenylxanthone-producing fungus
Aspergillus nidulans, through the identification of prenyltransferase genes belonging to the fungal indole prenyltransferases, so far known for their involvement in the prenylation of amino acids [
45]. Glycosylations represent another kind of functionalization recently evidenced from lichen xanthones through the examples of umbilicaxanthosides A and B [
44] and hirtusneanoside [
46].
The diversity of lichen xanthones is also extended through some dimeric xanthones. Although the identification of key dimerization processes still warrants further investigation, xanthone dimers are most likely obtained after the biaryl linkage of monomers. This hypothesis is preferred to that of a tandem biosynthetic pathway which would involve a side-by-side cyclization of a double-length polyketide, especially since the discovery of the long-sought-after monomeric units of secalonic acids (the so-called blennolides) within the fungus
Blennoria sp. [
47]. Enzymatically mediated or not, it is admitted that the dimerization would involve a xanthonyl radical that subsequently couples to electron donors. Resonance contributors of this delocalized aryl radical might then account for the reactivity of
ortho and
para C positions [
24].
Reductive dearomatizations are sometimes observed on xanthones to yield dihydro-, tetrahydro- or hexahydroxanthones. To date, in lichens, such reduced species were only observed from dimeric xanthones: secalonic acids, hirtusneanoside and eumitrin A1 (bis tetrahydroxanthones) and eumitrin A2, B and T (unsymmetrical tetrahydro- and hexahydroxanthones). Overall, a vast majority of xanthones reported from lichens have a monomeric and fully aromatized structure.
5. Structures and Bioactivities of Lichen Xanthones
It is noteworthy that lichen xanthones described up to 1996 most often belonged to the two basic cores described earlier with canonical substitution patterns. Today, 62 molecules displaying the lichexanthone scaffold and 19 molecules following the thiomelin substitution pattern (
Figure 2) have been described. However, the logical biosynthetic sequences delineated by Elix and Crook [
39] enable the prediction of putative intermediates that have not yet been reported from lichens. These putative intermediates might represent eight additional lichexanthone derivatives and three other thiomelin-type xanthones. Interestingly, a number of xanthone species described as putative in 1996 [
5] have been purified since then. Such species include 7-chlorolichexanthone (
Lecanora schofieldii [
88]), 2,4-dichloro-3-
O-methylnorlichexanthone (
Pertusaria aceroae and
P. calderae [
89]), 2,5-dichloro-3-
O-methylnorlichexanthone (obtained from
Calopadia fusca [
90] among others), 3,6-di-
O-mehtylthiophanic acid from
Calopadia subcoerulescens [
90] and
Phyllopsora chodatinica [
91]), 4,5,7-trichlorolichexanthone (
Sporopodium leprosum [
92]) and 2,4,7-trichloro-3-
O-methylnorlichexanthone (
Calopadia perpallida [
90]). Chlorinations and methylations from these cores can directly account for the structures of most lichen xanthones described by Huneck and Yoshimura. This diversity of lichen xanthones has to be extended to the vast array of secalonic acid and eumitrin derivatives identified by their chromatographic data but for which their minute amounts have so far hampered their NMR structure elucidation. Such tetrahydroxanthone dimers might represent privileged metabolites given their structural homology with the promising anti-tumor compound phomoxanthone A [
93]. One might also take into account the putative monomeric units of secalonic acids and eumitrins, none of them being identified from lichen sources so far.
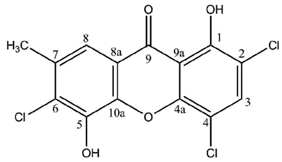
Besides these fully rationalized structures, a limited number of metabolites exhibit structural variations that are more or less difficult to explain. As an example, demethylchodatin displays a methoxy group in position 4 that might be introduced by a xanthone oxidase on this trichlorinated norlichexanthone derivative. Another unusual modification of the lichexanthone series is the occurrence of two acetyl groups in erythrommone. The chemical structures of xanthones with trivial names discussed in this manuscript are enlisted in
Figure 6.
However, 22 lichen xanthones display unusual structural features that make their biosynthetic intermediates tricky to unravel (including a series of 16 related species described by Rezanka and colleagues [
44,
94], see further). Three such species were already mentioned in the compendium established by Huneck and Yoshimura [
5]: the absence of a methyl substituent in 1,8-dihydroxy-3,6-dimethoxyxanthone is biogenetically difficult to explain, as are the two methyl substituents of 1,7-dihydroxy-2,4-dichloro-6,8-dimethylxanthone and of 1-hydroxy-2,4-dichloro-6,8-dimethylxanthone, obtained from
Rinodina thiomela.
As of 2016, a total of 103 xanthones were isolated from lichens, implying the presence of 11 further species as biosynthetic intermediates. This chemodiversity will soon be enriched with an extensive array of unidentified tetrahydrobisxanthones and their associated monomeric units.
Very few lichen xanthones have been investigated to date for their bioactivities. Lichexanthone exerts a weak activity against
Mycobacterium tuberculosis [
95] and
M. aurum [
96]. Despite this mild activity, a dihydropyrane xanthone derivative of lichexanthone revealed an antimycobacterial activity comparable to that of drugs commonly used to treat tuberculosis [
97]. Lichexanthone and griseoxanthone C exhibit strong antibiotic effects towards
Bacillus subtilis with respective IC
50 values of 2.25 and 1.29 μM, but only the former inhibited the growth of methicillin-resistant
Staphylococcus aureus (IC
50: 21 μM) [
22]. No anti-parasitic activity of lichexanthone could be observed towards
Plasmodium falciparum and
Trypanosoma brucei [
15]. Lichexanthone also displays sperm mobility–enhancing properties [
98] and induces the production of NO by murine macrophages which might reveal their activation [
99]. Lichexanthone revealed no cytotoxic activity against murine melanoma B16F10, human melanoma UACC-62 and fibroblast cells NIH/3T3 [
100]. At last, lichexanthone was found to be effective against the dengue vector
Aedes aegypti [
98].
Norlichexanthone was shown to display promising cytotoxic activities [
101]. Indeed, norlichexanthone caused 100% inhibition of p56
lck tyrosine kinase at 200 μg/mL [
102] and inhibited the activity of the protein kinases aurora-B, PIM1 and VEGF-R2 with mean IC
50 values ranging from 0.3 to 12 μM [
103]. Norlichexanthone also promotes the expression of the insulin-sensitizing, anti-diabetic and anti-atherogenic protein adiponectin within cultured ST-13 adipocytes [
104]. Dayan and Romagni also reported on the fungicidal effect of thiophanic (
Figure 5) and thiophaninic acids [
105]. Alongside many other lichen compounds, Huneck and Schreiber reported on the allelopathic effect of thiophanic acid towards a wide array of higher plants [
106].
As most structural classes of lichen metabolites, xanthones display strong UV-absorbing properties, predominantly in the wavelength range of UVA [
107]. Indeed, lichexanthone synthesis could be triggered in juvenile mycelia of
Haematomma fluorescens as a response to a 365 nm UV light exposure, emphasizing its protective role as a light filter [
108]. Deposition of xanthones in the cortical layers further suggests their involvement in the protection of the UV-sensitive algal layer [
109,
110]. Such properties might be of utmost interest when urgently needing new materials to outperform the currently used UV-photoprotective products [
111]. A Time-Dependent Density Functional Theory (TD-DFT) modelization recently delineated the electronic transitions accounting for the UV/Vis spectra of secalonic acids. This revealed that absorption wavelengths and molecular extinction coefficients obtained from these bisxanthones were comparable to that of UVA-referent sunscreens, which might stem from the structural homologies shared by the commercially available avobenzone and the phenyl-β-diketo moiety of xanthones [
112].
6. New Lichen Xanthones Described since 1996: Discussion, Physico-Chemical Properties (Including NMR Data) and Bioactivities
The past 20 years witnessed the isolation of 23 further lichen xanthones. It is noteworthy that those new xanthones displayed original substitution patterns and revealed original moieties compared to the previous state of the art.
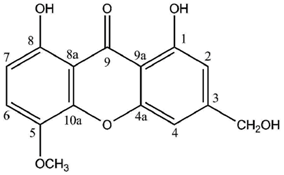
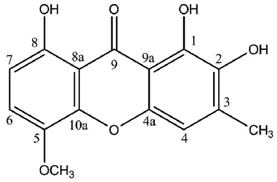
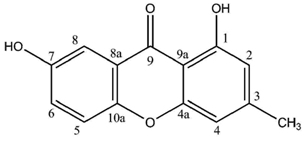
Cultures of the spore-derived mycobionts of
Pyrenula japonica and
P. pseudobufonia afforded a series of five new ravenelin-type lichen xanthones, the first reports of such xanthones from aposymbiotically grown lichen mycobionts. A first report elucidated the structures of 1,5,8-trihydroxy-3-methylxanthone, 1,8-dihydroxy-5-methoxy-3-methylxanthone and 1,7-dihydroxy-3-methylxanthone [
51] while a second one established the occurrence of 1,8-dihydroxy-3-hydroxymethyl-5-methoxyxanthone and 1,4,8-trihydroxy-5-methoxy-3-methylxanthone, typically associated with the thiomelin pathway [
52]. Notably, 1,7-dihydroxy-3-methylxanthone stands among the rare xanthones also described from plants (
Cassia occidentalis [
113]) and also from an Ascomycete fungus [
114]. Surprisingly, all molecules described from this series lack the 4-hydroxy group with four of them displaying an unusual oxygenated substituent at C5 instead. In one case, position 2 is oxygenated, which is a unique structural feature among the lichen xanthones described so far. One will note that 1,7-dihydroxy-3-methylxanthone also lacks the oxygenated substituent on C8 which is replaced by a unique hydroxyl substituent on C7, an unprecedented structural peculiarity for a lichen xanthone. Finally, the oxidation of the methyl group into a hydroxymethyl for one of these molecules is also new to lichen xanthones. Of special interest is that these compounds could not be identified from the whole lichen, whereas they are constantly isolated from mycobionts harvested in highly unrelated sampling sites (one in Japan and one in the USA), which might point to their biological significance in the prelichenized condition. Notably, 1,5,8-trihydroxy-3-methylxanthone and 1,2,8-trihydroxy-5-methoxy-3-methylxanthone displayed stronger antioxidant activities than α-tocopherol via the DPPH radical test [
52]. Besides, 1,7-dihydroxy-3-methylxanthone also exerts a moderate Mono Amine Oxidase-inhibiting activity [
114].
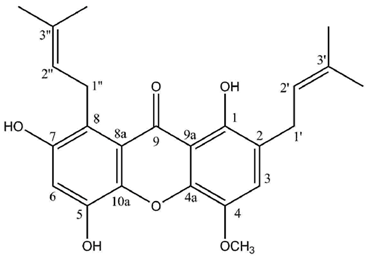
Two highly unusual glycosylated prenylxanthones were subsequently identified from the Ural lichen
Umbilicaria proboscidea [
44]. Umbilicaxanthoside A is a C2-monoprenyl xanthone having a β-
d-glucopyranose unit anchored at position O-7, whereas umbilicaxanthoside B stands for a C2,C8-diprenylxanthone with a disaccharide β-
d-glucopyranose-(1→4)-β-
d-glucopyranose moiety at
O-7. These compounds were the first prenylated xanthones to be isolated from a lichen source and glycosides are not frequently encountered in lichens [
84]. Besides, both molecules reveal a highly unusual oxygenation pattern, rendering their biosynthetic pathway tricky to delineate. A follow-up study carried out on the same lichen identified 14 acylated xanthone
O-glycosides corresponding to linolenoyl, lineoyl, palmitoleoyl, oleoyl, palmitoyl, eicosenoyl and stearoyl esters of both umbilicaxanthosides A and B [
94]. Fragmentation patterns obtained by LC-APCI-MS were highly informative and could establish the nature of the esterified fatty acid. None of those compounds were investigated for their biological properties.
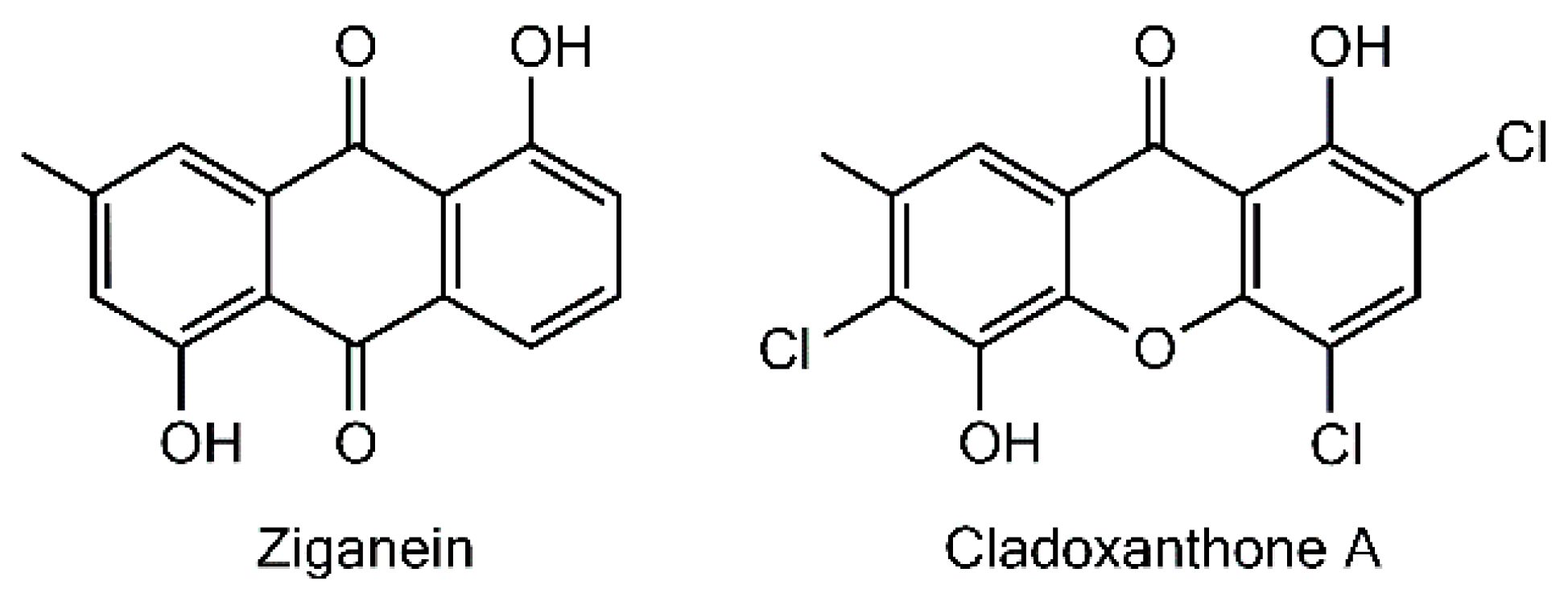
The last monomeric xanthone isolated so far from a lichen source is cladoxanthone A (=1,5-dihydroxy-2,4,6-trichloro-7-methylxanthone), obtained from
Cladonia incrassata [
115]. The occurrence of a methyl substituent on C-7 is an unprecedented structural feature among lichen xanthones, thus questioning the underlying biogenetic schemes. A possible hypothesis to account for the structure of this xanthone is to consider ziganein as a possible anthraquinone precursor [
116]. It is noteworthy that this metabolite presents a substitution pattern identical to that of cladoxanthone A and it was isolated from the endophytic fungus
Sporormiella minimoides [
116] (
Figure 7). Cladoxanthone A revealed an antibacterial effect towards
Staphylococcus aureus even though the paucity of the compound precluded the determination of a minimal inhibitory concentration [
115].
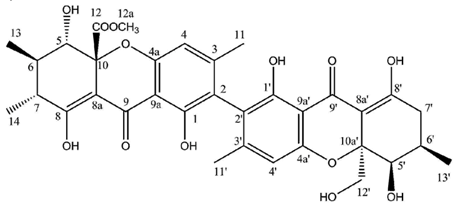
Regarding dimeric xanthones, known metabolites of the secalonic acid series were first isolated from lichen sources in 2009,
i.e., secalonic acids B, D and F were purified from
Diploicia canescens [
117]. The joint occurrence of these isomers in this lichen is not surprising because of their biosynthetic relationship. Indeed, the association of the two monomeric tetrahydroxanthone blennolides A and B, respectively, yields secalonic acids B and D [
47]. Secalonic acid F then represents a hybrid dimer of blennolides A and B. It is noteworthy that each of these compounds displays an identical configuration at C-10a, suggesting that the cyclization is catalyzed by a specific enzyme giving rise to a unique type of product [
117]. Secalonic acid is a major environmental mycotoxin essentially known for its production by the microbial
Penicillium oxalicum, a notorious foodstuff contaminant. Such a presence represents an alarming health issue given the acute toxicity and the teratogenic effects of secalonic acid D [
118,
119], warranting extended investigations of its bioactivities which highlighted its pleiotropic pharmacological activities. Secalonic acids D and F revealed antimicrobial activities against
Bacillus megaterium while secalonic acid A displayed activity against
Bacillus subtilis and
Piricularia oryzae [
120]. Likewise, secalonic acid B is an effective antimicrobial (
Bacillus megaterium and
Escherichia coli) as well as antifungal (
Microbotryum violaceum) and antialgal agent (
Chlorella fusca) [
47]. Several reports suggested the cytotoxic activity of secalonic acid derivatives [
117]. Bioassay-guided fractionation of the extracts of the marine lichen–derived
Gliocladium sp. T 31 streamlined the isolation of secalonic acid D as the cytotoxic metabolite against four cell lines in a highly variable range of 0.03–15 μM, suggesting selective pharmacodynamic properties [
121]. Further studies revealed a submicromolar IC
50 of secalonic acid D on the carcinoma KB cells and an inhibition of human topoisomerase 1 with a promising IC
50 of 0.16 μg/mL [
122]. More recently, secalonic acid D was shown to down-regulate the expression of efflux pump ABCG2 which is known to confer the Multidrug Resistance phenotype [
123]. As a last contribution to its cytotoxic activity, secalonic acid D exhibits an anti-angiogenic activity via the Akt/mTor/P70S6K pathway [
124]. Another feature of the pleiotropic activities of secalonic acid D depends on the inhibition of protein kinase C and several other Ca
++-dependent enzymes through competitive inhibition [
125]. Subsequent disruptions of cell signaling might account for the teratogenicity of these compounds. Indeed, it was demonstrated on a murine model that secalonic acid D decreases the levels of Epidermal Growth Factor (EGF) as well as its associated signal transduction [
126]. As a consequence, inhibition and alteration of transcription factors in the developing murine plate following an exposition to the toxin at normal human dietary levels were observed, resulting in the cleft palate condition [
127,
128].
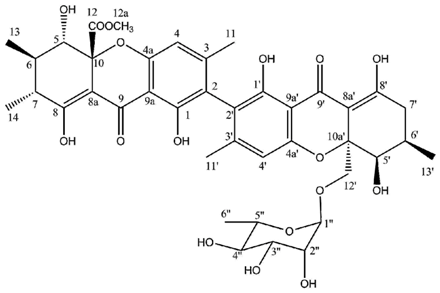
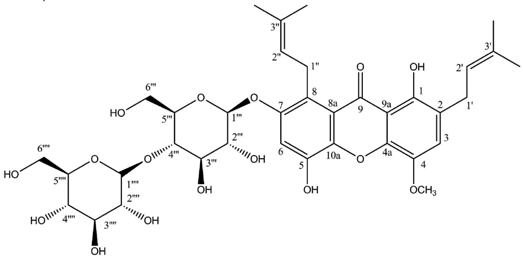
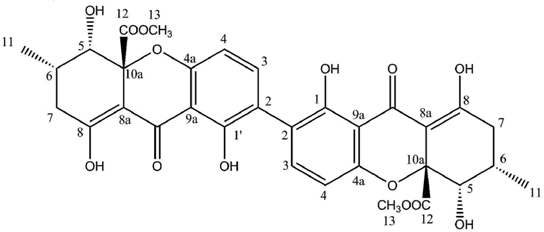
Hirtusneanoside is a new xanthone dimer isolated from
Usnea hirta corresponding to a rhamnoside of an unsymmetrical dimeric tetrahydroxanthone [
46]. If the oxygenation pattern can be fully rationalized according to the biosynthetic pathway discussed earlier, this structure contains several additional methyl groups of unknown origin. Hirtusneanoside was proven to be active against
Staphylococcus aureus and
Bacillus subtilis [
46]. The enzymatic hydrolysis of hirtusneanoside yielded α-
l-rhamnose and the aglycone named hirtusneanin, the physico-chemical data of which were also reported by Rezanka and Sigler [
46].
Available physico-chemical properties regarding the newly described lichen compounds are listed below.
Cladoxanthone A
Yellow powder
UV λmax (MeOH)(log ε): 381 (3.24), 323 (3.50), 259 (4.11) nm
IR νmax (CHCl3): 3353, 3056, 1642, 1597, 1450, 800 cm−1
Sources:
Cladonia incrassata [
115].
| Cladoxanthone A (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 156.2 | - |
| –OH 1 | - | 13.16 (s) (1H) |
| 2 | 115.2 | - |
| 3 | 136.6 | 7.80 (s) (1H) |
| 4 | 111.2 | - |
| 4a | 149.5 | - |
| 5 | 141.2 | - |
| –OH 5 | - | 6.09 (s) (1H) |
| 6 | 127.9 | - |
| 7 | 133.7 | - |
| CH3–7 | 20.2 | 2.51 (s) |
| 8 | 117.0 | 7.73 (s) |
| 8a | 118.4 | - |
| 9a | 109.7 | - |
| 10a | 143.0 | - |
1,8-Dihydroxy-3-hydroxymethyl-5-methoxyxanthone
Yellow needles, mp 221–222 °C
UV λmax (MeOH)(log ε): 387.5 (3.27), 341 (3.84), 271 (4.16), 263 (4.24), 255 (4.29), 236 (4.17)
IR νmax (KBr): 3533, 1659, 1634, 1609, 1589, 1493 cm−1
Sources: Mycobiont of
Pyrenula japonica [
52].
| 1,8-Dihydroxy-3-hydroxymethyl-5-methoxyxanthone (DMSO-d6) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 160.0 | - |
| –OH 1 | - | 11.10 (br s) a |
| 2 | 107.7 | 6.98 (br s) (1H) |
| 3 | 154.8 | - |
| 3–CH2OH | 62.2 | 4.57 (br s) (2H) |
| 3–CH2OH | - | 5.58 (br s) (1H) |
| 4 | 104.2 | 6.75 (br s) (1H) |
| 4a | 155.6 | - |
| 5 | 139.7 | - |
| 5–OCH3 | 56.7 | 3.86 (s) (3H) |
| 6 | 121.1 | 7.45 (d, 9.0) (1H) |
| 7 | 108.9 | 6.71 (d, 9.0) (1H) |
| 8 | 152.7 | - |
| 8–OH | - | 11.60 (br s) a |
| 8a | 107.8 | - |
| 9 | 184.9 | - |
| 9a | 106.1 | - |
| 10a | 144.8 | - |
1,2,8-Trihydroxy-5-methoxy-3-methylxanthone
Yellow needles, mp 214–215 °C
UV λmax (MeOH) (log ε): 421 (3.42), 352 (3.58), 280 (4.27), 263 (4.18), 243 (4.17), 206 (4.23)
IR νmax (KBr): 3487, 1663, 1636, 1607, 1576, 1489 cm−1
Sources: Mycobiont of
Pyrenula japonica [
52].
| 1,2,8-Trihydroxy-5-methoxy-3-methylxanthone (DMSO-d6) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 145.8 | - |
| 2 | 138.4 | - |
| –OH 2 | - | |
| 3 | 137.0 | - |
| 3–CH3 | 17.1 | 2.30 (s) (3H) |
| 4 | 107.6 | 6.95 (br s) (1H) |
| 4a | 147.7 | - |
| 5 | 139.7 | - |
| 5–OCH3 | 56.7 | 3.88 (s) (3H) |
| 6 | 120.5 | 7.45 (d, 9.0) (1H) |
| 7 | 108.2 | 6.70 (d, 9.0) (1H) |
| 8 | 152.6 | - |
| 8a | 107.6 | - |
| 9 | 185.1 | - |
| 9a | 105.9 | - |
| 10a | 145.0 | - |
1,7-Dihydroxy-3-methylxanthone
Yellow needles, mp 259–260 °C
UV λmax (MeOH) (log ε): 384 (3.84), 290 (3.98), 261 (4.58), 235 (4.45)
IR νmax (KBr): 3285, 1653, 1607, 1585, 1483 cm−1
Sources: Mycobiont of
Pyrenula japonica [
52].
| 1,7-Dihydroxy-3-methylxanthone (CDCl3-CD3OD) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 161.7 | - |
| 2 | 111.1 | 6.57 (br s) (1H) |
| 3 | 149.5 | - |
| 3–CH3 | 22.6 | 2.42 (s) (3H) |
| 4 | 108.1 | 6.75 (br s) (1H) |
| 4a | 157.1 | - |
| 5 | 119.6 | 7.37 (d, 9.0) (1H) |
| 6 | 125.5 | 7.29 (dd, 9.0, 2.5) (1H) |
| 7 | 154.5 | 6.70 (d, 9.0) (1H) |
| 8 | 109.0 | 7.52 (d, 2.5) (1H) |
| 8a | 121.6 | - |
| 9 | 182.5 | - |
| 9a | 107.1 | - |
| 10a | 150.8 | - |
1,5,8-Trihydroxy-3-methylxanthone
Yellow needles, mp 277–278 °C
UV λmax (MeOH) (log ε): 403 (3.54), 341 (4.01), 272 (4.58), 263 (4.40), 255 (4.47), 236 (4.32)
IR νmax (KBr): 3461, 1661, 1633, 1606, 1591, 1497 cm−1
Sources: Mycobiont of
Pyrenula japonica and
Pyrenula pseudobufonia [
52].
| 1,5,8-Trihydroxy-3-methylxanthone (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 161.4 | - |
| 1–OH | - | 11.85 (s) (1H) a |
| 2 | 111.9 | 6.62 (m) (1H) |
| 3 | 150.6 | - |
| 3–CH3 | 22.7 | 2.45 (s) (3H) |
| 4 | 108.5 | 6.89 (m) (1H) |
| 4a | 156.9 | - |
| 5 | 137.8 | - |
| 6 | 124.6 | 7.24 (d, 9.0) (1H) |
| 7 | 110.0 | 6.64 (d, 9.0) (1H) |
| 8 | 153.6 | - |
| 8–OH | - | 11.27 (br s) (1H) a |
| 8a | 108.6 | - |
| 9 | 186.5 | - |
| 9a | 106.4 | - |
| 10a | 144.7 | - |
1,8-Dihydroxy-5-methoxy-3-methylxanthone
Yellow needles, mp 214–215 °C
UV λmax (MeOH) (log ε): 393 (3.54), 340 (4.00), 272 (4.30), 254 (4.49), 235 (4.36)
IR νmax (KBr): 3445, 1661, 1631, 1609, 1585, 1489 cm−1
Sources: Mycobiont of
Pyrenula japonica and
Pyrenula pseudobufonia [
52].
| 1,8-Dihydroxy-5-methoxy-3-methylxanthone (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 161.0 | - |
| 1–OH | - | 11.73 (s) (1H) a |
| 2 | 111.7 | 6.62 (br s) (1H) |
| 3 | 149.9 | - |
| 3–CH3 | 22.6 | 2.42 (s) (3H) |
| 4 | 108.0 | 6.84 (br s) (1H) |
| 4a | 156.0 | - |
| 5 | 140.0 | - |
| 5–OCH3 | 57.4 | 3.94 (s) (3H) |
| 6 | 120.8 | 7.23 (d, 9.0) (1H) |
| 7 | 109.1 | 6.70 (d, 9.0) (1H) |
| 8 | 154.2 | - |
| 8–OH | - | 11.33 (br s) (1H) a |
| 8a | 108.3 | - |
| 9 | 185.6 | - |
| 9a | 105.9 | - |
| 10a | 145.6 | - |
Hirtusneanoside
Faint yellow crystals, mp 231–232 °C
= −251 (c = 0.02 in MeOH)
UV λmax (MeOH)(log ε): 340 (3.24), 275 (4.01), 230 (4.52) nm
IR νmax (CHCl3): 3290, 1735, 1620, 1590, 870 cm−1
Sources:
Usnea hirta [
46].
| Hirtusneanoside (DMSO-d6) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 159.2 | - |
| –OH 1 | - | 11.5 (br s) (1H) |
| 2 | 116.7 | - |
| 3 | 149.6 | - |
| 4 | 109.2 | 6.66 (s) (1H) |
| 4a | 156.8 | |
| 5 | 68.8 | 4.02 (d, 9.5) (1H) |
| 6 | 31.3 | 2.05 (ddq, 10.3, 9.5, 6.7) (1H) |
| 7 | 36.7 | 2.36 (dq, 10.3, 6.4) (1H) |
| 8 | 177.8 | - |
| –OH 8 | - | 13.7 (br s) (1H) |
| 8a | 101.3 | - |
| 9 | 186.8 | - |
| 9a | 105.7 | - |
| 10a | 85.1 | - |
| 11 | 20.7 | 1.94 (s) (3H) |
| 12 | 171.3 | - |
| 12a | 54.3 | 3.73 (s) (3H) |
| 13 | 15.6 | 1.09 (d, 6.7) (3H) |
| 14 | 17.3 | 1.01 (d, 6.4) (3H) |
| 1′ | 159.6 | - |
| 1′–OH | - | 11.5 (br s) (1H) |
| 2′ | 118.1 | - |
| 3′ | 150.2 | - |
| 4′ | 109.3 | 6.69 (s) (1H) |
| 4a′ | 156.7 | - |
| 5′ | 68.9 | 4.07 (d, 1.3) (1H) |
| 6′ | 28.5 | 2.28 (dddq, 11.3, 6.7, 6.5, 1.3) (1H) |
| 7′ | 33.6 | 2.48 (dd, 19.2, 11.3) (1H) |
| | | 2.35 (dd, 19.2, 6.5) (1H) |
| 8′ | 177.6 | - |
| 8′–OH | - | 13.7 (br s) (1H) |
| 8a′ | 101.8 | - |
| 9′ | 186.6 | - |
| 9a′ | 106.3 | - |
| 10a′ | 84.4 | - |
| 11′ | 20.6 | 1.96 (s) (3H) |
| 12′ | 64.5 | 3.89 (d, 13.0) (1H) |
| | | 3.51 (d, 13.0) (1H) |
| 13′ | 17.7 | 1.04 (d, 6.7) (3H) |
| 1″ | 101.1 | 5.01 (d, 1.5) (1H) |
| 2″ | 72.2 | 3.89 (dd, 2.5, 1.5) (1H) |
| 3″ | 70.9 | 3.71 (dd, 9.5, 2.5) (1H) |
| 4″ | 74.5 | 4.32 (t, 9.5) (1H) |
| 5″ | 69.7 | 4.13 (dq, 9.5, 6.5) (1H) |
| 6″ | 18.6 | 1.31 (d, 6.5) (3H) |
Hirtusneanine
Pale yellow crystals, mp 251–253 °C
= −232 (c = 0.01 in MeOH)
UV λmax (MeOH)(log ε): 338 (3.97), 275 (4.04), 231 (4.43) nm
IR νmax (CHCl3): 3290, 1734, 1608, 1590, 870 cm−1
Sources:
Usnea hirta [
46].
| | Hirtusneanine (DMSO-d6) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 159.2 | - |
| –OH 1 | - | 11.5 (br s) (1H) |
| 2 | 116.7 | - |
| 3 | 149.6 | - |
| 4 | 109.2 | 6.66 (s) (1H) |
| 4a | 156.8 | |
| 5 | 68.8 | 4.02 (d, 9.5) (1H) |
| 6 | 31.3 | 2.05 (ddq, 10.3, 9.5, 6.7) (1H) |
| 7 | 36.7 | 2.36 (dq, 10.3, 6.4) (1H) |
| 8 | 177.8 | - |
| –OH 8 | - | 13.7 (br s) (1H) |
| 8a | 101.3 | - |
| 9 | 186.8 | - |
| 9a | 105.7 | - |
| 10a | 85.1 | - |
| 11 | 20.7 | 1.94 (s) (3H) |
| 12 | 171.3 | - |
| 12a | 54.3 | 3.73 (s) (3H) |
| 13 | 15.6 | 1.09 (d, 6.7) (3H) |
| 14 | 17.3 | 1.01 (d, 6.4) (3H) |
| 1′ | 159.6 | - |
| 1′–OH | - | 11.5 (br s) (1H) |
| 2′ | 118.1 | - |
| 3′ | 150.2 | - |
| 4′ | 109.3 | 6.69 (s) (1H) |
| 4a′ | 156.7 | - |
| 5′ | 68.7 | 4.07 (d, 1.3) (1H) |
| 6′ | 28.5 | 2.28 (dddq, 11.3, 6.7, 6.5, 1.3) (1H) |
| 7′ | 33.6 | 2.48 (dd, 19.2, 11.3) (1H) |
| | | 2.35 (dd, 19.2, 6.5) (1H) |
| 8′ | 177.6 | - |
| 8′–OH | - | 13.7 (br s) (1H) |
| 8a′ | 102.1 | - |
| 9′ | 186.6 | - |
| 9a′ | 106.3 | - |
| 10a′ | 84.1 | - |
| 11′ | 20.6 | 1.96 (s) (3H) |
| 12′ | 68.7 | 4.14 (dd, 13.0, 7.0) (1H) |
| | | 3.75 (dd, 13.0, 4.7) (1H) |
| 12′–OH | | 3.28 (m) (1H) |
| 13′ | 17.7 | 1.04 (d, 6.7) (3H) |
Umbilicaxanthoside A
Yellow needles, mp 114 °C
= −35
UV λmax (MeOH) (log ε): 345 (3.90), 310 (4.15), 270 (4.05), 245 (4.50).
IR νmax (KBr): 3320, 2945, 2905, 1643 cm−1
Sources:
Umbilicaria proboscidea [
44].
| | Umbilicaxanthoside A (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 150.9 | - |
| 1–OH | - | 13.25 (s) (1H) |
| 2 | 118.2 | - |
| 3 | 120.1 | 6.51 (s) (1H) |
| 4 | 142.6 | - |
| 4–OCH3 | 56.4 | 3.75 (s) (3H) |
| 4a | 140.9 | - |
| 5 | 146.4 | - |
| 6 | 104.8 | 6.39 (d, 2.1) (1H) |
| 7 | 153.0 | - |
| 8 | 108.1 | 6.73 (d, 2.1) (1H) |
| 8a | 128.9 | - |
| 9 | 179.8 | - |
| 9a | 116.1 | - |
| 10a | 136.5 | - |
| 1′ | 22.0 | 3.19 (d, 6.6) (2H) |
| 2′ | 123.5 | 5.92 (td, 6.6, 1.3) (1H) |
| 3′ | 131.3 | - |
| 4′ | 25.9 | 1.64 (s) (3H) |
| 5′ | 17.9 | 1.57 (s) (3H) |
| 1″ | 99.8 | 4.80 (d, 7.3) (1H) |
| 2″ | 74.7 | 3.52 (dd, 8.9, 7.3) (1H) |
| 3″ | 77.2 | 3.58 (t, 8.9) (1H) |
| 4″ | 71.4 | 3.41 (t, 8.9) (1H) |
| 5″ | 78.6 | 3.45 (m) (1H) |
| 6″ | 62.6 | 3.93 (dd, 11.8, 2.3) (1H) |
| | | 3.72 (dd, 12.1, 5.2) (1H) |
Umbilicaxanthone A
Sources:
Umbilicaria proboscidea [
44].
| | Umbilicaxanthone A (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 150.9 | - |
| 1–OH | - | 13.25 (s) (1H) |
| 2 | 118.2 | - |
| 3 | 120.1 | 6.51 (s) (1H) |
| 4 | 142.6 | - |
| 4–OCH3 | 56.4 | 3.75 (s) (3H) |
| 4a | 140.9 | - |
| 5 | 146.4 | - |
| 6 | 102.1 | 6.15 (d, 2.1) (1H) |
| 7 | 156.9 | - |
| 8 | 106.3 | a |
| 8a | 128.9 | - |
| 9 | 179.8 | - |
| 9a | 116.1 | - |
| 10a | 136.5 | - |
| 1′ | 22.0 | 3.19 (d, 6.6) (2H) |
| 2′ | 123.5 | 5.92 (td, 6.6, 1.3) (1H) |
| 3′ | 131.3 | - |
| 4′ | 25.9 | 1.64 (s) (3H) |
| 5′ | 17.9 | 1.57 (s) (3H) |
Umbilicaxanthoside B
Pale yellow needles, mp 133 °C
UV λmax (MeOH) (log ε): 355 (4.06), 319 (4.01), 271 (3.96), 244 (4.08)
IR νmax (KBr): 3350, 2950, 2900, 1640 cm−1
Sources:
Umbilicaria proboscidea [
44].
| | Umbilicaxanthoside B (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 151.0 | - |
| 1–OH | - | 13.30 (s) (1H) |
| 2 | 117.8 | - |
| 3 | 119.4 | 6.47 (s) (1H) |
| 4 | 143.2 | - |
| 4–OCH3 | 56.3 | 3.75 (s) (3H) |
| 4a | 141.3 | - |
| 5 | 147.1 | - |
| 6 | 103.7 | 6.23 (s) (1H) |
| 7 | 153.2 | - |
| 8 | 118.4 | - |
| 8a | 129.6 | - |
| 9 | 183.1 | - |
| 9a | 114.6 | - |
| 10a | 135.7 | - |
| 1′ | 25.4 | 3.38 (d, 7.2) (2H) |
| 2′ | 123.8 | 5.39 (td, 7.2, 1.2) (1H) |
| 3′ | 130.9 | - |
| 4′ | 26.2 | 1.65 (s) (3H) |
| 5′ | 18.4 | 1.79 (s) (3H) |
| 1″ | 26.1 | 4.19 (d, 6.8) (2H) |
| 2″ | 124.8 | 5.37 (td, 6.8, 1.5) (1H) |
| 3″ | 132.0 | - |
| 4″ | 25.9 | 1.67 (s) (3H) |
| 5″ | 18.0 | 1.84 (s) (3H) |
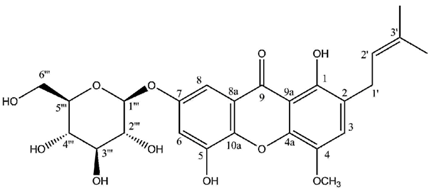
| 1‴ | 99.8 | 5.05 (d, 8.1) (1H) |
| 2‴ | 74.2 | 3.52 (dd, 8.9, 8.1) (1H) |
| 3‴ | 76.9 | 3.58 (t, 8.9) (1H) |
| 4‴ | 78.2 | 3.12 (dd, 9.3, 8.9) (1H) |
| 5‴ | 78.7 | 3.45 (m) (1H) |
| 6‴ | 62.6 | 3.93 (dd, 12.1, 2.3) (1H) |
| | | 3.72 (dd, 12.1, 5.2) (1H) |
| 1’’’’ | 104.1 | 5.15 (d, 8.0) (1H) |
| 2’’’’ | 73.8 | 3.52 (dd, 9.0, 8.0) (1H) |
| 3’’’’ | 76.5 | 3.58 (t, 9.0) (1H) |
| 4’’’’ | 69.7 | 3.41 (t, 9.0) (1H) |
| 5’’’’ | 76.9 | 3.45 (m) (1H) |
| 6’’’’ | 62.6 | 3.93 (dd, 12.1, 2.2) (1H) |
| | | 3.72 (dd, 12.1, 5.2) (1H) |
Umbilicaxanthone B
Sources:
Umbilicaria proboscidea [
44].
| | Umbilicaxanthone B (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 151.0 | - |
| 1–OH | - | 13.30 (s) (1H) |
| 2 | 117.8 | - |
| 3 | 119.4 | 6.49 (s) (1H) |
| 4 | 143.2 | - |
| 4–OCH3 | 56.3 | 3.75 (s) (3H) |
| 4a | 141.3 | - |
| 5 | 146.2 | - |
| 6 | 102.4 | 6.27 (s) (1H) |
| 7 | 157.2 | - |
| 8 | 117.6 | - |
| 8a | 129.6 | - |
| 9 | 183.1 | - |
| 9a | 114.6 | - |
| 10a | 135.7 | - |
| 1′ | 25.4 | 3.38 (d, 7.2) (2H) |
| 2′ | 123.8 | 5.39 (td, 7.2, 1.2) (1H) |
| 3′ | 130.9 | - |
| 4′ | 26.2 | 1.65 (s) (3H) |
| 5′ | 18.4 | 1.79 (s) (3H) |
| 1″ | 26.1 | 4.19 (d, 6.8) (2H) |
| 2″ | 124.8 | 5.37 (td, 6.8, 1.5) (1H) |
| 3″ | 132.0 | - |
| 4″ | 25.9 | 1.67 (s) (3H) |
| 5″ | 18.0 | 1.84 (s) (3H) |
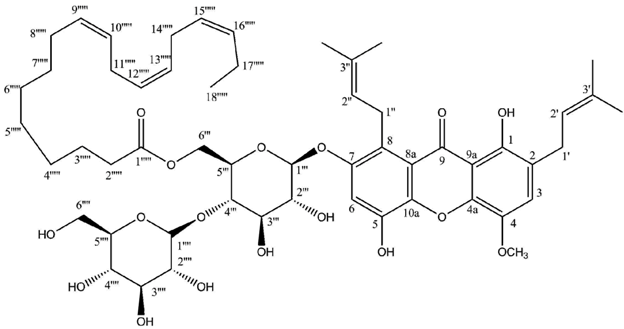
Linolenoylumbilicaxanthoside B
Pale yellow needles, mp 133 °C
UV λmax (MeOH) (log ε): 355 (4.06), 319 (4.01), 271 (3.96), 244 (4.08)
IR νmax (KBr): 3350, 2950, 2900, 1640 cm−1
Sources:
Umbilicaria proboscidea [
94].
| | Linolenoylumbilicaxanthoside B (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1 | 151.0 | - |
| 1–OH | - | 13.30 (s) (1H) |
| 2 | 117.8 | - |
| 3 | 119.4 | 6.47 (s) (1H) |
| 4 | 143.2 | - |
| 4–OCH3 | 56.3 | 3.75 (s) (3H) |
| 4a | 141.3 | - |
| 5 | 147.1 | - |
| 6 | 103.7 | 6.42 (s) (1H) |
| 7 | 153.2 | - |
| 8 | 118.4 | - |
| 8a | 129.6 | - |
| 9 | 183.1 | - |
| 9a | 114.6 | - |
| 10a | 135.7 | - |
| 1′ | 25.4 | 3.38 (d, 7.2) (2H) |
| 2′ | 123.8 | 5.39 (dd, 7.2, 1.2) (1H) |
| 3′ | 130.9 | - |
| 4′ | 26.2 | 1.65 (s) (3H) |
| 5′ | 18.4 | 1.79 (s) (3H) |
| 1″ | 26.1 | 4.19 (d, 6.8) (2H) |
| 2″ | 124.8 | 5.37 (dd, 6.8, 1.5) (1H) |
| 3″ | 132.0 | - |
| 4″ | 25.9 | 1.67 (s) (3H) |
| 5″ | 18.0 | 1.84 (s) (3H) |
| 1‴ | 99.8 | 5.05 (d, 8.1) (1H) |
| 2‴ | 74.2 | 3.50 (dd, 8.9, 8.1) (1H) |
| 3‴ | 77.1 | 3.58 (t, 8.9) (1H) |
| 4‴ | 76.7 | 3.05 (dd, 9.3, 8.9) (1H) |
| 5‴ | 76.1 | 3.54 (m) (1H) |
| 6‴ | 65.3 | 4.51 (dd, 12.1, 2.3) (1H) |
| | | 4.03 (dd, 12.1, 5.2) (1H) |
| 1’’’’ | 104.1 | 5.15 (d, 8.0) (1H) |
| 2’’’’ | 73.8 | 3.52 (dd, 9.0, 8.0) (1H) |
| 3’’’’ | 76.5 | 3.61 (t, 9.0) (1H) |
| 4’’’’ | 69.7 | 3.41 (t, 9.0) (1H) |
| 5’’’’ | 76.9 | 3.45 (m) (1H) |
| 6’’’’ | 62.6 | 3.93 (dd, 12.1, 2.2) (1H) |
| | | 3.72 (dd, 12.1, 5.2) (1H) |
| 1’’’’’ | 172.0 | - |
| 2’’’’’ | 34.6 | 2.25 (m) (2H) |
| 3’’’’’ | 25.0 | 1.68 (m) (2H) |
| 4’’’’' | 29.2 | 1.29 (m) (6H) |
| 5’’’’’ | 29.3 | 1.29 (m) (6H) |
| 6’’’’’ | 29.1 | 1.29 (m) (6H) |
| 7’’’’’ | 30.3 | 1.33 (m) (2H) |
| 8’’’’’ | 27.3 | 1.96 (m) (2H) |
| 9’’’’’ | 131.8 | 5.37 (m) (1H) |
| 10’’’’’ | 131.7 | 5.42 (m) (1H) |
| 11’’’’’ | 25.7 | 2.68 (m) (2H) |
| 12’’’’’ | 128.3 | 5.35 (m) (2H) |
| 13’’’’’ | 128.4 | 5.35 (m) (2H) |
| 14’’’’’ | 25.6 | 2.73 (m) (2H) |
| 15’’’’’ | 127.1 | 5.43 (m) (1H) |
| 16’’’’’ | 131.9 | 5.36 (m) (1H) |
| 17’’’’’ | 20.6 | 1.93 (m) (2H) |
| 18’’’’’ | 14.2 | 1.06 (t, 6.8) (3H) |
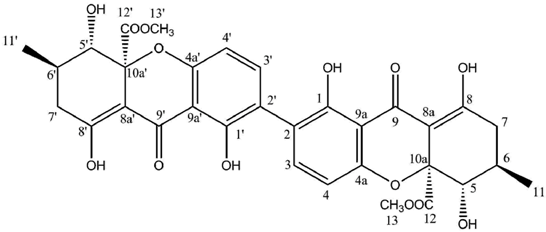
Secalonic acid A
Faint yellow crystals, mp 231–232 °C
= −64.1 (c = 0.1 in CHCl3)
UV λmax (MeOH)(log ε): 338 (4.52), 260 (4.12), 228 (4.32) nm
IR νmax (CHCl3): 3464, 1726, 1610, 1585, 1566, 1436, 1233, 1067 cm−1
Sources:
Diploicia canescens [
117].
Enantiomeric secalonic acid D: similar physicochemical data except = + 64.1 (c = 0.14 in CHCl3)
| | Secalonic Acid A (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1, 1′ | 159.4 | - |
| –OH 1,1′ | - | 11.75 (s) (2H) |
| 2, 2′ | 118.2 | - |
| 3, 3′ | 140.2 | 7.46 (d, 8.5) (2H) |
| 4, 4′ | 107.6 | 6.63 (d, 8.5) (2H) |
| 4a, 4a′ | 158.3 | - |
| 5, 5′ | 76.9 | 3.93 (dd, 11.3, 1.6) (2H) |
| –OH 5,5′ | - | 2.81 (d, 2.2) (2H) |
| 6, 6′ | 29.2 | 2.41 (m) (2H) |
| 7, 7′ | 36.3 | 2.74 (dd, 19.0, 6.1) (2H) |
| | | 2.32 (dd, 19.0, 11.5) (2H) |
| 8, 8′ | 177.5 | - |
| –OH 8,8′ | - | 13.78 (s) (2H) |
| 8a, 8a′ | 101.5 | - |
| 9, 9′ | 187.2 | - |
| 9a, 9a′ | 106.9 | - |
| 10a, 10a′ | 84.7 | - |
| 11, 11′ | 18.0 | 1.17 (d, 6.4) (6H) |
| 12, 12′ | 170.3 | - |
| 13, 13′ | 53.3 | 3.73 (s) (6H) |
Secalonic acid B
Faint yellow crystals, mp 231–232 °C
= + 133.7 (c = 0.38 in CHCl3)
UV λmax (MeOH)(log Ɛ): 338 (4.52), 260 (4.12), 228 (4.32) nm
IR νmax (CHCl3): 3582, 3014, 1747, 1611, 1589, 1214, 1058, 796, 726 cm−1
Sources:
Diploicia canescens [
117].
| | Secalonic acid B (CDCl3) |
|---|
| Position | δC | δH (J in Hz) |
| 1, 1′ | 159.4 | - |
| –OH 1,1′ | - | 11.85 (s) (2H) |
| 2, 2′ | 118.7 | - |
| 3, 3′ | 139.7 | 7.42 (d, 8.5) (2H) |
| 4, 4′ | 107.5 | 6.57 (d, 8.5) (2H) |
| 4a, 4a′ | 157.2 | - |
| 5, 5′ | 71.4 | 4.12 (d, 1.4) (2H) |
| –OH 5,5′ | - | 2.57 (s) (2H) |
| 6, 6′ | 28.5 | 2.12 (m) (2H) |
| 7, 7′ | 32.6 | 7, 7′ α 2.52 (dd, 19.0, 11.5) (2H) |
| | | 7, 7′ β 2.41 (dd, 19.0, 6.1) (2H) |
| 8, 8′ | 179.8 | - |
| –OH 8,8′ | - | 13.96 (s) (2H) |
| 8a, 8a′ | 99.9 | - |
| 9, 9′ | 187.7 | - |
| 9a, 9a′ | 107.0 | - |
| 10a, 10a′ | 84.8 | - |
| 11, 11′ | 17.5 | 1.18 (d, 6.8) (6H) |
| 12, 12′ | 171.2 | - |
| 13, 13′ | 53.4 | 3.72 (s) (6H) |