Azaphenanthrene Alkaloids with Antitumoral Activity from Anaxagorea dolichocarpa Sprague & Sandwith (Annonaceae)
Abstract
:1. Introduction
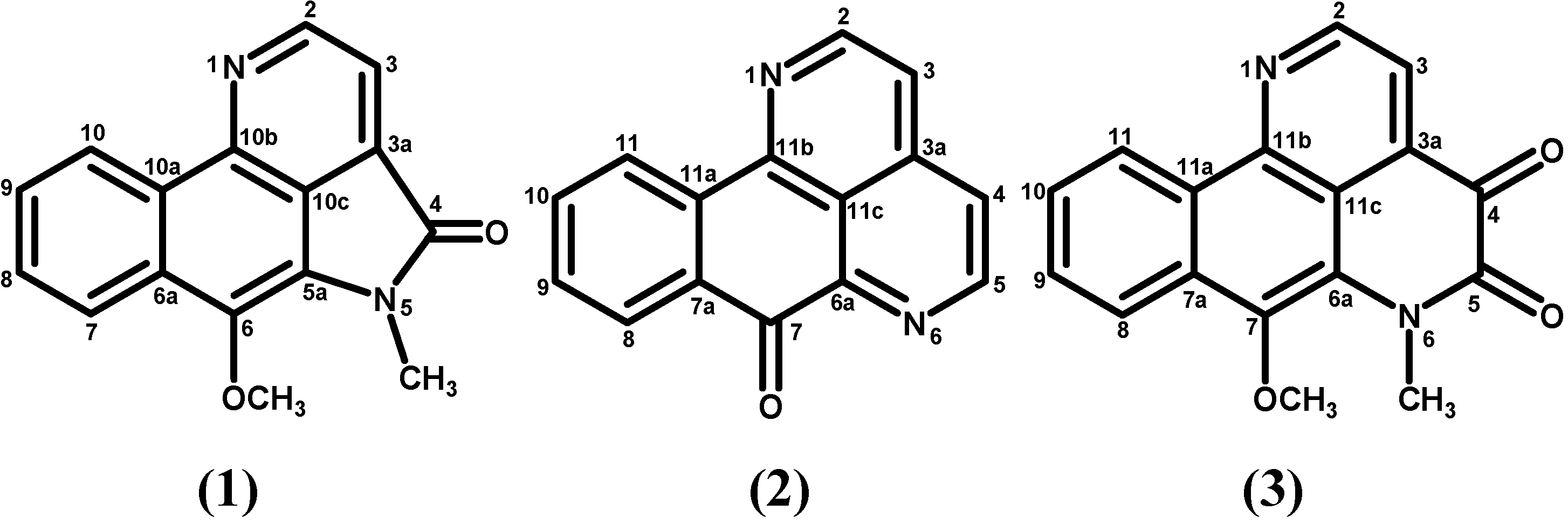
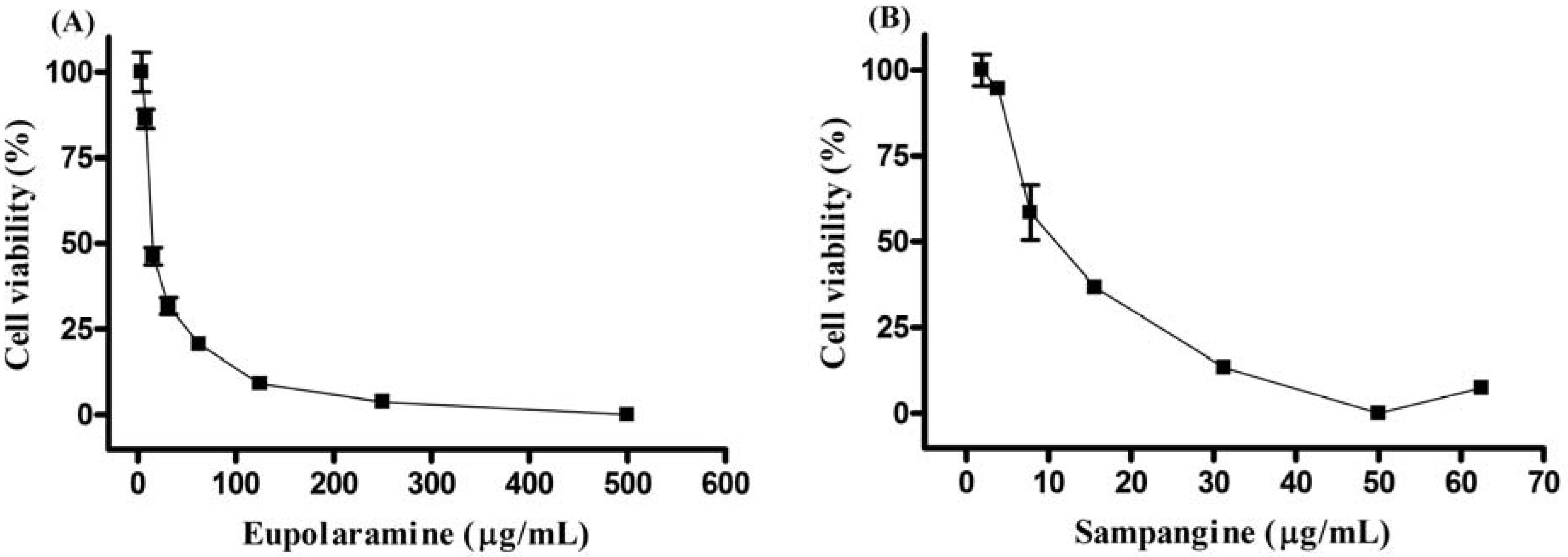
2. Results and Discussion
3. Experimental
3.1. General
3.2. Plant material
3.3. Extraction and isolation procedures
3.4. Evaluation of “in vitro” antitumor activity
4. Conclusions
Acknowledgements
References
- Chatrou, L.W.; Rainer, H.; Maas, P.J.M. Annonaceae (Soursop Family). In Flowering Plants of Neotropics; Smith, N., Mori, S.A., Henderson, A., Stevenson, D.W., Heald, S.V., Eds.; Botanical Garden: New York, NY, USA, 2004; pp. 18–20. [Google Scholar]
- Champy, P.; Guérineau, V.; Laprévote, O. MALDI-TOF MS profiling of Annonaceous acetogenins in Annona muricata products for human. Molecules 2009, 14, 5235–5246. [Google Scholar] [CrossRef]
- Melot, A.; Fall, D.; Gleye, C.; Champy, P. Apolar Annonaceous acetogenins from the fruit pulp of Annona muricata. Molecules 2009, 14, 4387–4395. [Google Scholar] [CrossRef]
- Fechine, I.M.; Vanessa, R.; Navarro, V.R.; Cunha, E.V.L.; Silva, M.S.; Maia, J.G.S.; Barbosa-Filho, J.M. Alkaloids and volatile constituents from Duguetia flagellaris. Biochem. Syst. Ecol. 2002, 30, 267–269. [Google Scholar] [CrossRef]
- Alias, A.; Hazni, H.; Jaafar, F.M.; Awang, K.; Ismail, N.H. Alkaloids from Fissistigma latifolium (Dunal) Merr. Molecules 2010, 15, 4583–4588. [Google Scholar] [CrossRef]
- Tavares, J.F.; Silva, M.V.B.; Queiroga, K.F.; Diniz, M.F.F.M.; Barbosa-Filho, J.M.; Haun, M.; Melo, P.S.; Silva, M.S. Xylodiol, a new atisane diterpenoid from Xylopia langsdorffiana St.-Hil. & Tul. (Annonaceae). Z. Naturforsch. 2007, 62b, 742–744. [Google Scholar]
- Cavé, A.; Leboeuf, M.; Waterman, P.G. The aporphinoid alkaloids of the Annonaceae. In Alkaloids: Chemical and Biological Perspectives; Pelletier, S.W., Ed.; Wiley: New York, NY, USA, 1987; Volume 5, pp. 134–270. [Google Scholar]
- Maas, P.J.M.; Westra, L.Y.T. Studies in Annonaceae II. A monograph of the genus Anaxagorea A. St. Hil. Part 2. Bot. Jahrb. Syst. 1985, 105, 145–204. [Google Scholar]
- Diaz, A.M.P. Neolignans from Anaxagorea clavata. Phytochemistry 1997, 44, 345–346. [Google Scholar] [CrossRef]
- Gonda, R.; Takeda, T.; Akiyama, T. Studies on the constituents of Anaxagorea luzonensis A. Gray III. Nat. Med. 2002, 56, 10–12. [Google Scholar]
- Gonda, R.; Takeda, T.; Akiyama, T. Studies on the constituents of Anaxagorea luzonensis A. Gray. Chem. Pharm. Bull. 2000, 48, 1219–1222. [Google Scholar] [CrossRef]
- Hocquemiller, R.; Rasamizafy, S.; Moretti, C.; Jacquemin, H.; Cavé, A. Anaxagoreine, a new alkaloid isolated from two species of the genus Anaxagorea. Planta Med. 1981, 41, 48–50. [Google Scholar] [CrossRef]
- Lobão, A.Q.; Araújo, D.S.D.; Kurtz, B.C. Annonaceae das restingas do estado do Rio de Janeiro, Brasil. Rodriguésia 2005, 56, 85–96. [Google Scholar]
- Andrade, E.H.A.; Oliveira, J.; Zoghbi, M.G. Volatiles of Anaxagorea dolichocarpa Spreng. & Sandw. and Annona densicoma Mart. growing wild in the state of Pará, Brazil. Flavour Frag. J. 2007, 22, 158–160. [Google Scholar] [CrossRef]
- Chiappeta, A.D.A.; Mello, J.F. Higher plants with biological activity: Plants of Pernambuco. Rev. Inst. Antibiot. UFPE 1984, 11, 99–111. [Google Scholar]
- Muhammad, I.; Dunbar, D.C.; Takamatsu, S.; Walker, L.A.; Clark, A.M. Antimalarial, cytotoxic and antifungal alkaloids from Duguetia hadrantha. J. Nat. Prod. 2001, 64, 559–562. [Google Scholar] [CrossRef]
- Rys, A.; Couture, A.; Deniau, E.; Lebrun, S.; Grandclaudon, P. A brief total synthesis of eupolauramine. Synlett 2004, 12, 2233–2235. [Google Scholar]
- Orabi, K.Y.; Li, E.; Clark, A.M.; Huffird, C.D. Microbial transformation of sampangine. J. Nat. Prod. 1999, 62, 988–992. [Google Scholar] [CrossRef]
- Kitahara, Y.; Mochii, M.; Mori, M.; Kubo, A. Synthetic studies of imbiline 1, a constituent of Eupomatia species. Tetrahedron 2003, 59, 2885–2891. [Google Scholar] [CrossRef]
- Carroll, A.R.; Taylor, W.C. Constituents of Eupomatia species. XII. Isolation of constituents of the tubers and aerial parts of Eupomatia bennettii and determination of the structures of new alkaloids from the aerial parts of E. bennettii and minor alkaloids of E. laurina. Aust. J. Chem. 1991, 44, 1615–1626. [Google Scholar] [CrossRef]
- Agarwal, A.K.; Xu, T.; Jacob, M.R.; Feng, Q.; Lorenz, M.C.; Walker, L.A.; Clark, A.M. Role of Heme in the antifungal activity of the azaoxoaporphine alkaloids sampangine. Eukaryot. Cell 2008, 7, 387–400. [Google Scholar] [CrossRef]
- Peterson, J.R.; Zjawiony, J.K.; Liu, S.; Hufford, C.D.; Clark, A.M.; Rogers, R.D. Copyrine alkaloids: Synthesis, spectroscopic characterization, and antimycotic/antimycobacterial activity of A- and B-ring-functionalized sampangines. J. Med. Chem. 1992, 35, 4069–4077. [Google Scholar] [CrossRef]
- Kluza, J.; Clark, A.M.; Bailly, C. Apoptosis induced by the alkaloid sampangine in HL-60 leukemia cells: Correlation between the effects on the cell cycle progression and changes of mitochondrial potential. Ann. N. Y. Acad. Sci. 2003, 1010, 331–334. [Google Scholar] [CrossRef]
- Costa-Lotufo, L.V.; Khan, M.T.H.; Ather, A.; Wilke, D.V.; Jimenez, P.C.; Pessoa, C.; Moraes, M.E.A.; Moraes, M.O. Studies of the anticancer potential of plants used in Bangladeshi folk medicine. J. Ethnopharmacol. 2005, 99, 21–30. [Google Scholar] [CrossRef]
- Bezerra, D.P.; De Castro, F.O.; Alves, A.P.; Pessoa, C.; De Moraes, M.O.; Silveira, E.R.; Lima, M.A.; Elmiro, F.J.; De Alencar, N.M.; Mesquita, R.O.; Lima, M.W.; Costa-Lotufo, L.V. In vitro and in vivo antitumor effect of 5-FU combined with piplartine and piperine. J. Appl. Toxicol. 2008, 28, 156–163. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1-3 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lúcio, A.S.S.C.; Da Silva Almeida, J.R.G.; Barbosa-Filho, J.M.; Pita, J.C.L.R.; Branco, M.V.S.C.; De Fátima Formiga Melo Diniz, M.; De Fátima Agra, M.; Da-Cunha, E.V.L.; Da Silva, M.S.; Tavares, J.F. Azaphenanthrene Alkaloids with Antitumoral Activity from Anaxagorea dolichocarpa Sprague & Sandwith (Annonaceae). Molecules 2011, 16, 7125-7131. https://doi.org/10.3390/molecules16087125
Lúcio ASSC, Da Silva Almeida JRG, Barbosa-Filho JM, Pita JCLR, Branco MVSC, De Fátima Formiga Melo Diniz M, De Fátima Agra M, Da-Cunha EVL, Da Silva MS, Tavares JF. Azaphenanthrene Alkaloids with Antitumoral Activity from Anaxagorea dolichocarpa Sprague & Sandwith (Annonaceae). Molecules. 2011; 16(8):7125-7131. https://doi.org/10.3390/molecules16087125
Chicago/Turabian StyleLúcio, Ana Silvia Suassuna Carneiro, Jackson Roberto Guedes Da Silva Almeida, José Maria Barbosa-Filho, João Carlos Lima Rodrigues Pita, Marianna Vieira Sobral Castello Branco, Margareth De Fátima Formiga Melo Diniz, Maria De Fátima Agra, Emidio V.L. Da-Cunha, Marcelo Sobral Da Silva, and Josean Fechine Tavares. 2011. "Azaphenanthrene Alkaloids with Antitumoral Activity from Anaxagorea dolichocarpa Sprague & Sandwith (Annonaceae)" Molecules 16, no. 8: 7125-7131. https://doi.org/10.3390/molecules16087125
APA StyleLúcio, A. S. S. C., Da Silva Almeida, J. R. G., Barbosa-Filho, J. M., Pita, J. C. L. R., Branco, M. V. S. C., De Fátima Formiga Melo Diniz, M., De Fátima Agra, M., Da-Cunha, E. V. L., Da Silva, M. S., & Tavares, J. F. (2011). Azaphenanthrene Alkaloids with Antitumoral Activity from Anaxagorea dolichocarpa Sprague & Sandwith (Annonaceae). Molecules, 16(8), 7125-7131. https://doi.org/10.3390/molecules16087125



