Alkaloids from Hippeastrum papilio
Abstract
:1. Introduction
2. Results and Discussion
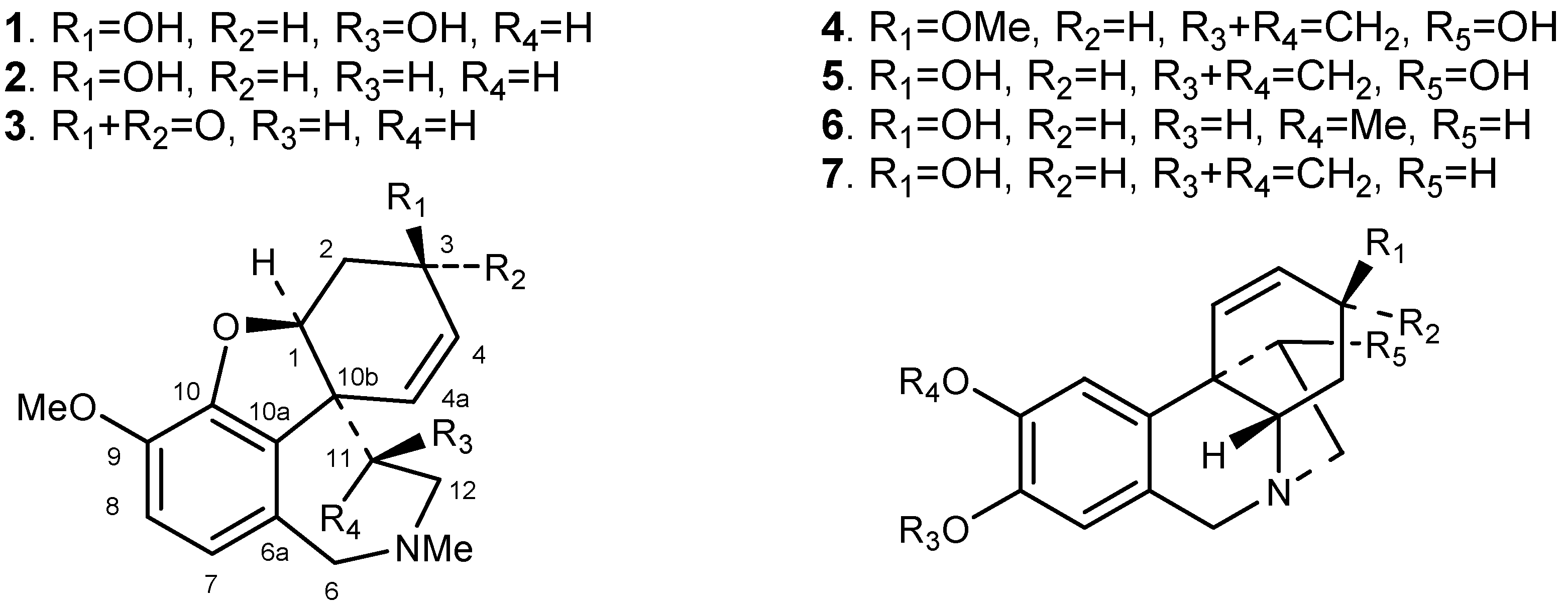
| Position | H δ | H δ (J in Hz) | COSY | NOESY | HSQC | HMBC | ||
|---|---|---|---|---|---|---|---|---|
| 1 | 4.88 | br s | H-2α, H-2β | H-2α, H-2β, H-11α | 89.0 | d | C-3, C-4a, C-11 | |
| 2α | 2.70 | ddd (15.6, 5.2, 2.8) | H-1, H-2β | H-1, H-2β, H-3 | 32.3 | t | C-1, C-3, C-4, C-10b | |
| 2β | 2.37 | br dt (15.6, 1.6) | H-1, H-2α, H-3 | H-1, H-2α, H-3 | -- | |||
| 3 | 4.20 | br t (4.8) | H-2β, H-4 | H-2α, H-2β, H-4 | 62.2 | d | C-1, C-4, C-4a | |
| 4 | 6.29 | dd (10.4, 4.8) | H-3, H-4a | H-3, H-4a | 133.1 | d | C-2, C-10b | |
| 4a | 5.96 | d (10.0) | H-4 | H-4, H-6β, H-12β | 122.6 | d | C-1, C-3, C-10b | |
| 6α | 3.60 | d (15.2) | H-6β | H-6β, H-7, NMe | 59.5 | t | C-6a, C-7, C-10a, C-12, NMe | |
| 6β | 3.93 | d (14.8) | H-6α | H-4a, H-6α, H-12β | C-6a, C-7, C-10a, C-12, NMe | |||
| 6a | 129.9 | s | ||||||
| 7 | 6.61 | d (8.0) | H-8 | H-6α, H-8 | 122.4 | d | C-6, C-9, C-10a | |
| 8 | 6.68 | d (8.0) | H-7 | H-7, OMe | 111.8 | d | C-6a, C-10 | |
| 9 | 144.6 | s | ||||||
| 10 | 146.9 | s | ||||||
| 10a | 128.9 | s | ||||||
| 10b | 53.4 | s | ||||||
| 11α | 4.05 | dd (10.8, 4.0) | H-12α, H-12β | H-1, H-12α, H-12β, NMe | 72.0 | d | C-1, C-4a | |
| 12α | 3.02 | dd (14.0, 3.2) | H-11α, H-12β | H-11α, H-12β, NMe | 62.1 | t | C-6, C-10b, C-11 | |
| 12β | 3.17 | dd (13.6, 10.8) | H-11α, H-12α | H-4a, H-6β, H-11α, H-12α | C-6, C-11, NMe | |||
| OMe | 3.84 | s (3H) | H-8 | 56.2 | q | C-9 | ||
| NMe | 2.45 | s (3H) | H-6α, H-11α, H-12α | 43.2 | q | C-6, C-12 | ||
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation of Alkaloids
3.4. Microplate AChE Assay
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Bastida, J.; Lavilla, R.; Viladomat, F. Chemical and biological aspects of Narcissus alkaloids. In The Alkaloids; Cordell, G.A., Ed.; Elsevier Scientific Publishing: Amsterdam, The Netherlands, 2006; Volume 63, pp. 87–179. [Google Scholar]
- Sener, B.; Orhan, I.; Satayavivad, J. Antimalarial activity screening of some alkaloids and the plant extracts from Amaryllidaceae. Phytother. Res. 2003, 17, 1220–1223. [Google Scholar] [CrossRef]
- McNulty, J.; Nair, J.J.; Bastida, J.; Pandey, S.; Griffin, C. Structure-activity studies on the lycorine pharmacophore: a potent inducer of apoptosis in human leukemia cells. Phytochemistry 2009, 70, 913–919. [Google Scholar] [CrossRef]
- Berkov, S.; Codina, C.; Viladomat, F.; Bastida, J. N-alkylated galanthamine derivatives: potent acetylcholinesterase inhibitors from Leucojum aestivum. Bioorg. Med. Chem. Lett. 2008, 18, 2263–2266. [Google Scholar] [CrossRef]
- Maelicke, A.; Samochocki, M.; Jostock, R.; Fehrenbacher, A.; Ludwig, J.; Albuquerque, E.X.; Zerlin, M. Allosteric sensitization of nicotinic receptors by galantamine, a new treatment strategy for Alzheimer’s Disease. Biol. Psychiatry 2001, 49, 279–288. [Google Scholar] [CrossRef]
- Berkov, S.; Bastida, J.; Viladomat, F.; Codina, C. Development and validation of a GC-MS method for rapid determination of galanthamine in Leucojum aestivum and Narcissus ssp.: A metabolomic approach. Talanta 2011, 83, 1455–1465. [Google Scholar] [CrossRef]
- da Silva, A.F.S.; de Andrade, J.P.; Bevilaqua, L.R.M.; de Souza, M.M.; Izquierdo, I.; Henriques, A.T.; Zuanazzi, J.A.S. Anxiolytic-, antidepressant- and anticonvulsant-like effects of the alkaloid montanine isolated from Hippeastrum vittatum. Pharmacol. Biochem. Behav. 2006, 85, 148–154. [Google Scholar] [CrossRef]
- Pagliosa, L.B.; Monteiro, S.C.; Silva, K.B.; de Andrade, J.P.; Dutilh, J.; Bastida, J.; Cammarota, M.; Zuanazzi, J.A.S. Effect of isoquinoline alkaloids from two Hippeastrum species on in vitro acetylcholinesterase activity. Phytomedicine 2010, 17, 698–701. [Google Scholar] [CrossRef]
- Giordani, R.B.; Vieira, P.B.; Weizenmann, M.; Rosemberg, D.B.; Souza, A.P.; Bonorino, C.; de Carli, G.A.; Bogo, M.R.; Zuanazzi, J.A.S.; Tasca, T. Candimine-induced cell death of the Amitochondriate Parasite Trichomonas vaginalis. J. Nat. Prod. 2010, 73, 2019–2023. [Google Scholar] [CrossRef]
- Meerow, A.W.; Guy, C.L.; Li, Q.B.; Yang, S.L. Phylogeny of the American Amaryllidaceae based on nrDNA ITS sequences. Syst. Bot. 2000, 25, 708–726. [Google Scholar] [CrossRef]
- Hesse, M.; Berhard, H.O. Amaryllidaceae alkaloids. In Progress in Mass Spectrometry; von Budzikiewicz, H., Ed.; Verlag Chemie: Weinheim, Germany, 1975; Volume 3, pp. 164–184. [Google Scholar]
- Wildman, W.C.; Brown, C.L. The structure of habranthine. Tetrahedron Lett. 1968, 43, 4573–4576. [Google Scholar] [CrossRef]
- Tato, M.P.V.; Castedo, L.; Riguera, R. New alkaloids from Pancratium maritimum L. Heterocycles 1988, 27, 2833–2838. [Google Scholar] [CrossRef]
- López, S.; Bastida, J.; Viladomat, F.; Codina, C. Acetylcholinesterase inhibitory activity of some Amaryllidaceae alkaloids and Narcissus extracts. Life Sci. 2002, 71, 2521–2529. [Google Scholar] [CrossRef]
- Bastida, J.; Viladomat, F.; Llabrés, J.M.; Codina, C.; Feliz, M.; Rubiralta, M. Alkaloids from Narcissus confusus. Phytochemistry 1987, 26, 1519–1524. [Google Scholar] [CrossRef]
- Bastida, J.; Contreras, J.L.; Codina, C.; Wright, C.W.; Phillipson, J.D. Alkaloids from Narcissus cantabricus. Phytochemistry 1995, 40, 1549–1551. [Google Scholar] [CrossRef]
- Berkov, S.; Bastida, J.; Tsvetkova, R.; Viladomat, F.; Codina, C. Alkaloids from Sternbergia colchiciflora. Z. Naturforsch. C 2009, 64, 311–316. [Google Scholar]
- Bastida, J.; Bergoñón, S.; Viladomat, F.; Codina, C. Alkaloids from Narcissus primigenius. Planta Med. 1994, 60, 95–96. [Google Scholar]
- Matharu, B.; Gibson, G.; Parsons, R.; Huckerby, T.N.; Moore, S.A.; Cooper, L.J.; Millichamp, R.; Allsop, D.; Austen, B. Galantamine inhibits β-amyloid aggregation and cytotoxicity. J. Neurol. Sci. 2009, 280, 49–58. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Andrade, J.P.d.; Berkov, S.; Viladomat, F.; Codina, C.; Zuanazzi, J.A.S.; Bastida, J. Alkaloids from Hippeastrum papilio. Molecules 2011, 16, 7097-7104. https://doi.org/10.3390/molecules16087097
Andrade JPd, Berkov S, Viladomat F, Codina C, Zuanazzi JAS, Bastida J. Alkaloids from Hippeastrum papilio. Molecules. 2011; 16(8):7097-7104. https://doi.org/10.3390/molecules16087097
Chicago/Turabian StyleAndrade, Jean Paulo de, Strahil Berkov, Francesc Viladomat, Carles Codina, José Angelo S. Zuanazzi, and Jaume Bastida. 2011. "Alkaloids from Hippeastrum papilio" Molecules 16, no. 8: 7097-7104. https://doi.org/10.3390/molecules16087097
APA StyleAndrade, J. P. d., Berkov, S., Viladomat, F., Codina, C., Zuanazzi, J. A. S., & Bastida, J. (2011). Alkaloids from Hippeastrum papilio. Molecules, 16(8), 7097-7104. https://doi.org/10.3390/molecules16087097






