Consumption of Dairy Products in Relation to Type 2 Diabetes Mellitus in Chinese People: The Henan Rural Cohort Study and an Updated Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. The Henan Rural Cohort Study
2.1.1. Participants
2.1.2. Dietary Assessment
2.1.3. Ascertainment of T2DM
2.1.4. Non-Dietary Covariates
2.1.5. Statistical Analysis
2.2. Meta-Analysis
2.2.1. Literature Search and Study Design
2.2.2. Study Selection, Data Extraction and Quality Assessment
2.2.3. Statistical Methods
3. Results
3.1. The Henan Rural Cohort Study
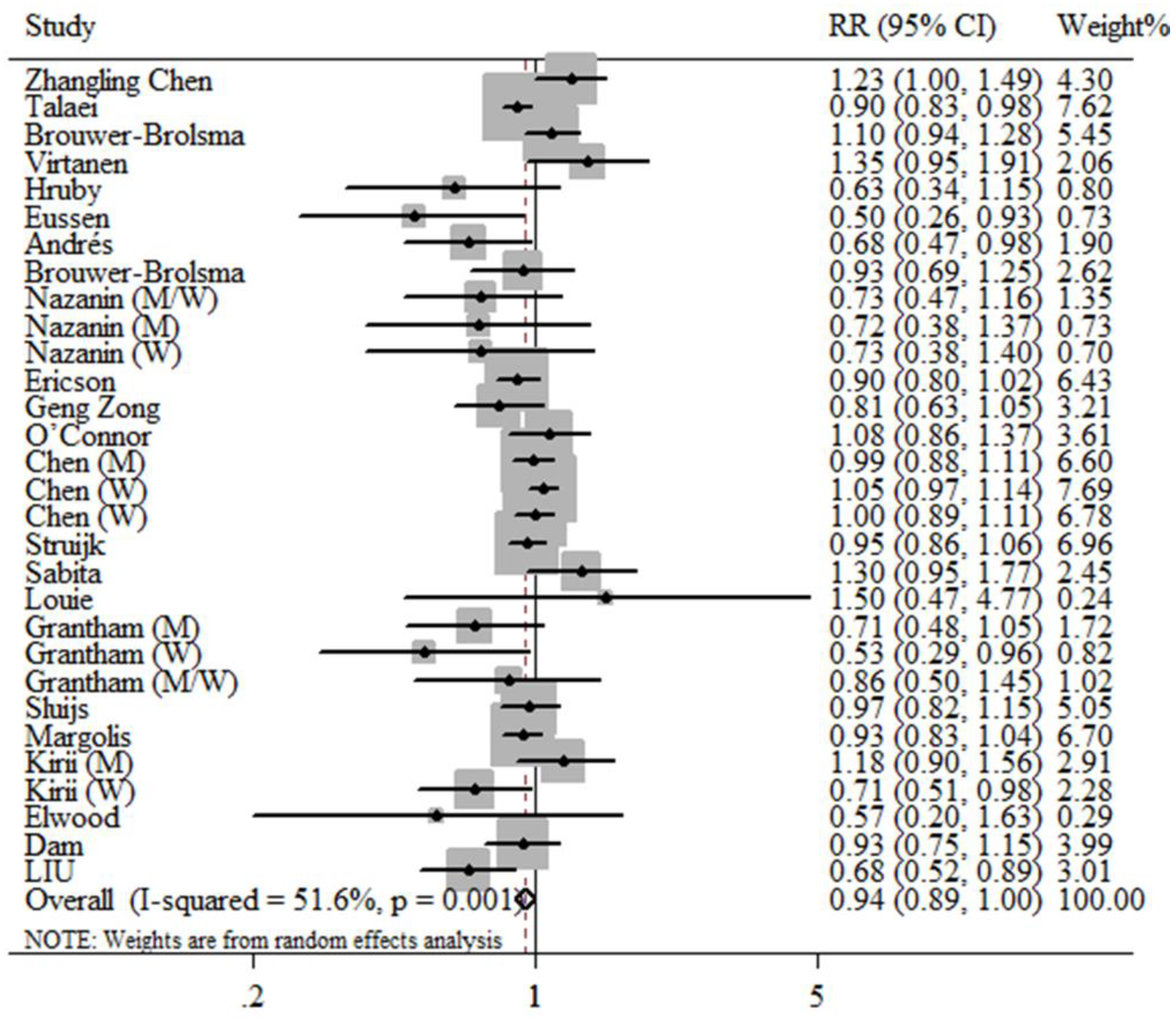
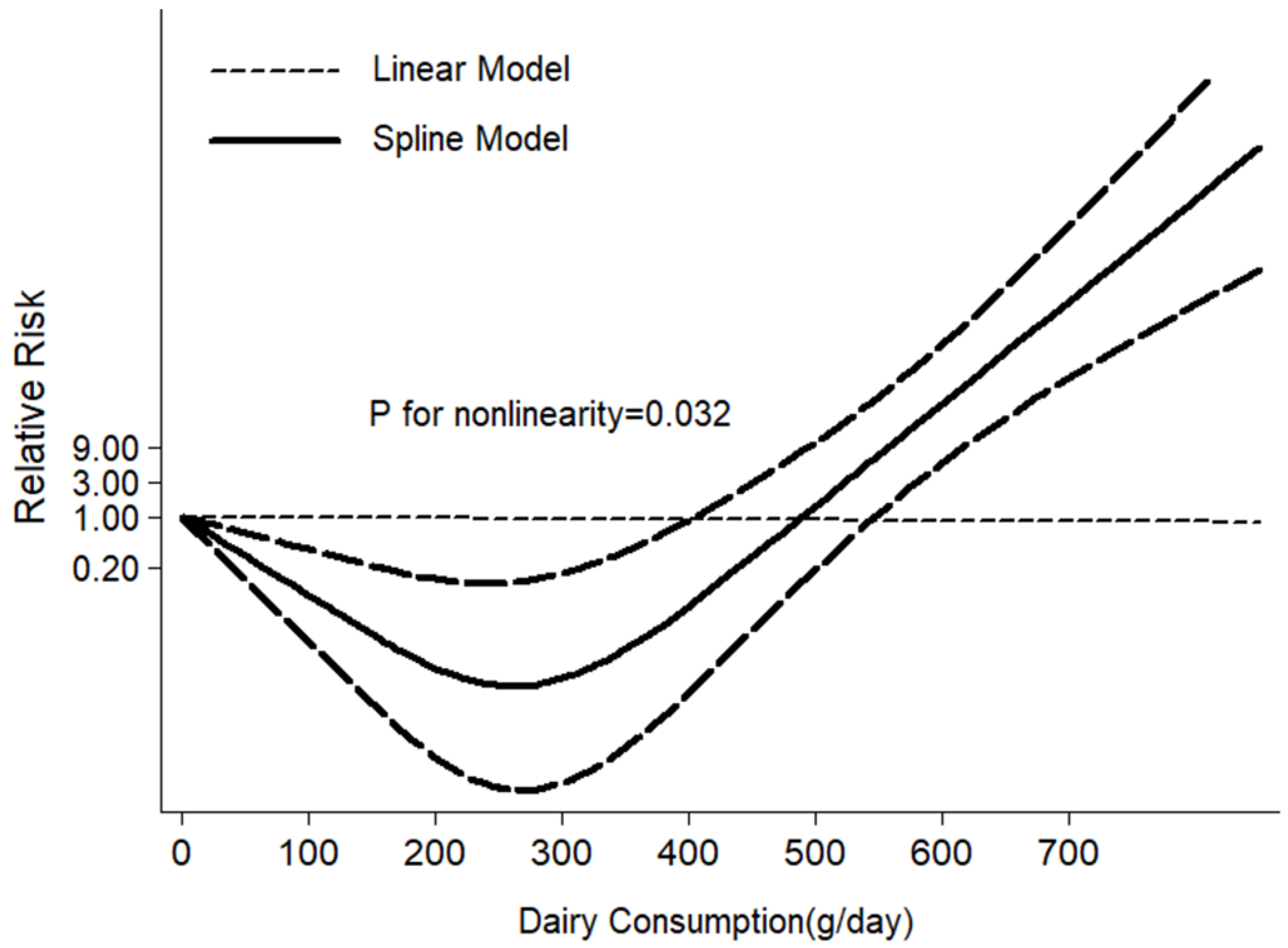
3.2. Meta-Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| T2DM | type 2 diabetes mellitus |
| FFQ | food frequency questionnaire |
| ADA | American Diabetes Association |
| FPG | fasting blood glucose |
| BMI | body mass index |
| TC | total cholesterol |
| TG | triglyceride |
| HDL-C | high-density lipoprotein cholesterol |
| LDL-C | low-density lipoprotein cholesterol |
| NOS | Newcastle Ottawa Scale |
| ANOVA | analysis of variance |
| OR | odds ratio |
| CI | confidence intervals |
| RR | relative risk |
| HR | hazard ratio |
References
- Wang, L.; Gao, P.; Zhang, M.; Huang, Z.; Zhang, D.; Deng, Q.; Li, Y.; Zhao, Z.; Qin, X.; Jin, D.; et al. Prevalence and Ethnic Pattern of Diabetes and Prediabetes in China in 2013. JAMA 2017, 317, 2515–2523. [Google Scholar] [CrossRef]
- National Diabetes Research Group. A mass survey of diabetes mellitus in a population of 300,000 in 14 provinces and municipalities in China. Zhonghua Nei Ke Za Zhi 1981, 20, 678–683. [Google Scholar]
- Yang, W.; Lu, J.; Weng, J.; Jia, W.; Ji, L.; Xiao, J.; Shan, Z.; Liu, J.; Tian, H.; Ji, Q.; et al. Prevalence of diabetes among men and women in China. N. Engl. J. Med. 2010, 362, 1090–1101. [Google Scholar] [CrossRef] [PubMed]
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.C.W. Epidemiology of diabetes and diabetic complications in China. Diabetologia 2018, 61, 1249–1260. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, D.D.; Ley, S.H.; Vasanti, M.; Howard, A.G.; He, Y.; Hu, F.B. Time Trends of Dietary and Lifestyle Factors and Their Potential Impact on Diabetes Burden in China. Diabetes Care 2017, 40, 1685–1694. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, K.; Maisonet, M.; Wang, L.; Zheng, S. Associations of lifestyle factors (smoking, alcohol consumption, diet and physical activity) with type 2 diabetes among American adults from National Health and Nutrition Examination Survey (NHANES) 2005–2014. J. Diabetes 2017, 9, 846–854. [Google Scholar] [CrossRef]
- Zhou, M.; Wang, H.; Zeng, X.; Yin, P.; Zhu, J.; Chen, W.; Li, X.; Wang, L.; Wang, L.; Liu, Y.; et al. Mortality, morbidity, and risk factors in China and its provinces, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 394, 1145–1158. [Google Scholar] [CrossRef]
- Ortega, R.M.; Jiménez Ortega, A.I.; Perea Sánchez, J.M.; Cuadrado Soto, E.; Aparicio Vizuete, A.; López-Sobaler, A.M. Nutritional value of dairy products and recommended daily consumption. Nutr. Hosp. 2019, 36, 25–29. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G.; Schwedhelm, C.; Kalle-Uhlmann, T.; Missbach, B.; Knuppel, S.; Boeing, H. Consumption of Dairy Products in Relation to Changes in Anthropometric Variables in Adult Populations: A Systematic Review and Meta-Analysis of Cohort Studies. PLoS ONE 2016, 11, e0157461. [Google Scholar] [CrossRef]
- Chen, M.; Sun, Q.; Giovannucci, E.; Mozaffarian, D.; Manson, J.E.; Willett, W.C.; Hu, F.B. Dairy consumption and risk of type 2 diabetes: 3 cohorts of US adults and an updated meta-analysis. BMC Med. 2014, 12, 215. [Google Scholar] [CrossRef] [PubMed]
- Mitri, J.; Mohd Yusof, B.N.; Maryniuk, M.; Schrager, C.; Hamdy, O.; Salsberg, V. Dairy intake and type 2 diabetes risk factors: A narrative review. Diabetes Metab. Syndr. 2019, 13, 2879–2887. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Xu, Q.; Jiang, R.; Han, T.; Sun, C.; Na, L. Dietary Protein Consumption and the Risk of Type 2 Diabetes: A Systematic Review and Meta-Analysis of Cohort Studies. Nutrients 2017, 9, 982. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G.; Lampousi, A.M.; Knuppel, S.; Iqbal, K.; Schwedhelm, C.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of type 2 diabetes mellitus: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2017, 32, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Cao, H.; King, I.B.; Lemaitre, R.N.; Song, X.; Siscovick, D.S.; Hotamisligil, G.S. Trans-palmitoleic acid, metabolic risk factors, and new-onset diabetes in U.S. adults: A cohort study. Ann. Intern. Med. 2010, 153, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Franco, O.H.; Lamballais, S.; Ikram, M.A.; Schoufour, J.D.; Muka, T.; Voortman, T. Associations of specific dietary protein with longitudinal insulin resistance, prediabetes and type 2 diabetes: The Rotterdam Study. Clin. Nutr. 2020, 39, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Talaei, M.; Pan, A.; Yuan, J.M.; Koh, W.P. Dairy intake and risk of type 2 diabetes. Clin. Nutr. 2018, 37, 712–718. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; Sluik, D.; Singh-Povel, C.M.; Feskens, E.J.M. Dairy product consumption is associated with pre-diabetes and newly diagnosed type 2 diabetes in the Lifelines Cohort Study. Br. J. Nutr. 2018, 119, 442–455. [Google Scholar] [CrossRef]
- Virtanen, H.E.K.; Koskinen, T.T.; Voutilainen, S.; Mursu, J.; Tuomainen, T.P.; Kokko, P.; Virtanen, J.K. Intake of different dietary proteins and risk of type 2 diabetes in men: The Kuopio Ischaemic Heart Disease Risk Factor Study. Br. J. Nutr. 2017, 117, 882–893. [Google Scholar] [CrossRef]
- Hruby, A.; Ma, J.; Rogers, G.; Meigs, J.B.; Jacques, P.F. Associations of Dairy Intake with Incident Prediabetes or Diabetes in Middle-Aged Adults Vary by Both Dairy Type and Glycemic Status. J. Nutr. 2017, 147, 1764–1775. [Google Scholar] [CrossRef]
- Eussen, S.J.; van Dongen, M.C.; Wijckmans, N.; den Biggelaar, L.; Oude Elferink, S.J.; Singh-Povel, C.M.; Schram, M.T.; Sep, S.J.; van der Kallen, C.J.; Koster, A.; et al. Consumption of dairy foods in relation to impaired glucose metabolism and type 2 diabetes mellitus: The Maastricht Study. Br. J. Nutr. 2016, 115, 1453–1461. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Lopez, A.; Bullo, M.; Martinez-Gonzalez, M.A.; Corella, D.; Estruch, R.; Fito, M.; Gomez-Gracia, E.; Fiol, M.; Garcia de la Corte, F.J.; Ros, E.; et al. Dairy product consumption and risk of type 2 diabetes in an elderly Spanish Mediterranean population at high cardiovascular risk. Eur. J. Nutr. 2016, 55, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Brouwer-Brolsma, E.M.; van Woudenbergh, G.J.; Oude Elferink, S.J.; Singh-Povel, C.M.; Hofman, A.; Dehghan, A.; Franco, O.H.; Feskens, E.J. Intake of different types of dairy and its prospective association with risk of type 2 diabetes: The Rotterdam Study. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Moslehi, N.; Shab-Bidar, S.; Mirmiran, P.; Sadeghi, M.; Azizi, F. Associations between dairy products consumption and risk of type 2 diabetes: Tehran lipid and glucose study. Int. J. Food Sci. Nutr. 2015, 66, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Ericson, U.; Hellstrand, S.; Brunkwall, L.; Schulz, C.A.; Sonestedt, E.; Wallstrom, P.; Gullberg, B.; Wirfalt, E.; Orho-Melander, M. Food sources of fat may clarify the inconsistent role of dietary fat intake for incidence of type 2 diabetes. Am. J. Clin. Nutr. 2015, 101, 1065–1080. [Google Scholar] [CrossRef] [PubMed]
- Zong, G.; Sun, Q.; Yu, D.; Zhu, J.; Sun, L.; Ye, X.; Li, H.; Jin, Q.; Zheng, H.; Hu, F.B.; et al. Dairy consumption, type 2 diabetes, and changes in cardiometabolic traits: A prospective cohort study of middle-aged and older Chinese in Beijing and Shanghai. Diabetes Care 2014, 37, 56–63. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, L.M.; Lentjes, M.A.; Luben, R.N.; Khaw, K.T.; Wareham, N.J.; Forouhi, N.G. Dietary dairy product intake and incident type 2 diabetes: A prospective study using dietary data from a 7-day food diary. Diabetologia 2014, 57, 909–917. [Google Scholar] [CrossRef]
- Struijk, E.A.; Heraclides, A.; Witte, D.R.; Soedamah-Muthu, S.S.; Geleijnse, J.M.; Toft, U.; Lau, C.J. Dairy product intake in relation to glucose regulation indices and risk of type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 822–828. [Google Scholar] [CrossRef]
- Soedamah-Muthu, S.S.; Masset, G.; Verberne, L.; Geleijnse, J.M.; Brunner, E.J. Consumption of dairy products and associations with incident diabetes, CHD and mortality in the Whitehall II study. Br. J. Nutr. 2013, 109, 718–726. [Google Scholar] [CrossRef]
- Louie, J.C.; Flood, V.M.; Rangan, A.M.; Burlutsky, G.; Gill, T.P.; Gopinath, B.; Mitchell, P. Higher regular fat dairy consumption is associated with lower incidence of metabolic syndrome but not type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 816–821. [Google Scholar] [CrossRef]
- Grantham, N.M.; Magliano, D.J.; Hodge, A.; Jowett, J.; Meikle, P.; Shaw, J.E. The association between dairy food intake and the incidence of diabetes in Australia: The Australian Diabetes Obesity and Lifestyle Study (AusDiab). Public Health Nutr. 2013, 16, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Sluijs, I.; Forouhi, N.G.; Beulens, J.W.; van der Schouw, Y.T.; Agnoli, C.; Arriola, L.; Balkau, B.; Barricarte, A.; Boeing, H.; Bueno-de-Mesquita, H.B.; et al. The amount and type of dairy product intake and incident type 2 diabetes: Results from the EPIC-InterAct Study. Am. J. Clin. Nutr. 2012, 96, 382–390. [Google Scholar] [PubMed]
- Margolis, K.L.; Wei, F.; de Boer, I.H.; Howard, B.V.; Liu, S.; Manson, J.E.; Mossavar-Rahmani, Y.; Phillips, L.S.; Shikany, J.M.; Tinker, L.F.; et al. A diet high in low-fat dairy products lowers diabetes risk in postmenopausal women. J. Nutr. 2011, 141, 1969–1974. [Google Scholar] [PubMed]
- Kirii, K.; Mizoue, T.; Iso, H.; Takahashi, Y.; Kato, M.; Inoue, M.; Noda, M.; Tsugane, S.; Japan Public Health Center-based Prospective Study Group. Calcium, vitamin D and dairy intake in relation to type 2 diabetes risk in a Japanese cohort. Diabetologia 2009, 52, 2542–2550. [Google Scholar] [CrossRef] [PubMed]
- Elwood, P.C.; Pickering, J.E.; Fehily, A.M. Milk and dairy consumption, diabetes and the metabolic syndrome: The Caerphilly prospective study. J. Epidemiol. Community Health 2007, 61, 695–698. [Google Scholar] [CrossRef] [PubMed]
- van Dam, R.M.; Hu, F.B.; Rosenberg, L.; Krishnan, S.; Palmer, J.R. Dietary calcium and magnesium, major food sources, and risk of type 2 diabetes in U.S. black women. Diabetes Care 2006, 29, 2238–2243. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Choi, H.K.; Ford, E.; Song, Y.; Klevak, A.; Buring, J.E.; Manson, J.E. A prospective study of dairy intake and the risk of type 2 diabetes in women. Diabetes Care 2006, 29, 1579–1584. [Google Scholar] [CrossRef]
- Liu, X.; Mao, Z.; Li, Y.; Wu, W.; Zhang, X.; Huo, W.; Yu, S.; Shen, L.; Li, L.; Tu, R.; et al. Cohort Profile: The Henan Rural Cohort: A prospective study of chronic non-communicable diseases. Int. J. Epidemiol. 2019, 48, 1756–1756j. [Google Scholar] [CrossRef]
- Xue, Y.; Yang, K.; Wang, B.; Liu, C.; Mao, Z.; Yu, S.; Li, X.; Wang, Y.; Sun, H.; Wang, C.; et al. Reproducibility and validity of an FFQ in the Henan Rural Cohort Study. Public Health Nutr. 2020, 23, 34–40. [Google Scholar] [CrossRef]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2009, 32 (Suppl. 1), S62–S67. [Google Scholar] [CrossRef]
- Lo, C.K.; Mertz, D.; Loeb, M. Newcastle-Ottawa Scale: Comparing reviewers’ to authors’ assessments. BMC Med. Res. Methodol. 2014, 14, 45. [Google Scholar] [CrossRef]
- Bagos, P.G.; Nikolopoulos, G.K. Generalized least squares for assessing trends in cumulative meta-analysis with applications in genetic epidemiology. J. Clin. Epidemiol. 2009, 62, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Orsini, N.; Li, R.; Wolk, A.; Khudyakov, P.; Spiegelman, D. Meta-analysis for linear and nonlinear dose-response relations: Examples, an evaluation of approximations, and software. Am. J. Epidemiol. 2012, 175, 66–73. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials revisited. Contemp. Clin. Trials 2015, 45, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Benedetti, A. Quantifying heterogeneity in individual participant data meta-analysis with binary outcomes. Syst. Rev. 2017, 6, 243. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Forouhi, N.G.; Koulman, A.; Sharp, S.J.; Imamura, F.; Kröger, J.; Schulze, M.B.; Crowe, F.L.; Huerta, J.M.; Guevara, M.; Beulens, J.W.J.; et al. Differences in the prospective association between individual plasma phospholipid saturated fatty acids and incident type 2 diabetes: The EPIC-InterAct case-cohort study. Lancet Diabetes Endocrinol. 2014, 2, 810–818. [Google Scholar] [CrossRef]
- Taheri, E.; Saedisomeolia, A.; Djalali, M.; Qorbani, M.; Madani Civi, M. The relationship between serum 25-hydroxy vitamin D concentration and obesity in type 2 diabetic patients and healthy subjects. J. Diabetes Metab. Disord. 2012, 11, 16. [Google Scholar] [CrossRef]
- Onakpoya, I.J.; Perry, R.; Zhang, J.; Ernst, E. Efficacy of calcium supplementation for management of overweight and obesity: Systematic review of randomized clinical trials. Nutr. Rev. 2011, 69, 335–343. [Google Scholar] [CrossRef]
- Yanovski, J.A.; Parikh, S.J.; Yanoff, L.B.; Denkinger, B.I.; Calis, K.A.; Reynolds, J.C.; Sebring, N.G.; McHugh, T. Effects of calcium supplementation on body weight and adiposity in overweight and obese adults: A randomized trial. Ann. Intern. Med. 2009, 150, 821–829. [Google Scholar] [CrossRef]
- Ejtahed, H.S.; Mohtadi-Nia, J.; Homayouni-Rad, A.; Niafar, M.; Asghari-Jafarabadi, M.; Mofid, V. Probiotic yogurt improves antioxidant status in type 2 diabetic patients. Nutrition 2012, 28, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Li, Y.; Wang, C.; Mao, Z.; Zhou, W.; Zhang, L.; Yang, X.; Cui, S.; Li, L. Dietary Protein Consumption and the Risk of Type 2 Diabetes: ADose-Response Meta-Analysis of Prospective Studies. Nutrients 2019, 11, 2783. [Google Scholar] [CrossRef] [PubMed]
- Lean, M.E.; Te Morenga, L. Sugar and Type 2 diabetes. Br. Med. Bull. 2016, 120, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Hossain, A.; Yamaguchi, F.; Matsuo, T.; Tsukamoto, I.; Toyoda, Y.; Ogawa, M.; Nagata, Y.; Tokuda, M. Rare sugar D-allulose: Potential role and therapeutic monitoring in maintaining obesity and type 2 diabetes mellitus. Pharmacol. Ther. 2015, 155, 49–59. [Google Scholar] [CrossRef]
- Wang, Y.; Li, S. Worldwide trends in dairy production and consumption and calcium intake: Is promoting consumption of dairy products a sustainable solution for inadequate calcium intake? Food Nutr. Bull. 2008, 29, 172–185. [Google Scholar] [CrossRef]
- He, Y.; Yang, X.; Xia, J.; Zhao, L.; Yang, Y. Consumption of meat and dairy products in China: A review. Proc. Nutr. Soc. 2016, 75, 385–391. [Google Scholar] [CrossRef]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Dairy products and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Am. J. Clin. Nutr. 2013, 98, 1066–1083. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Lay, S.; Yu, H.N.; Shen, S.R. Dietary Guidelines for Chinese Residents (2016): Comments and comparisons. J. Zhejiang Univ. Sci. B 2016, 17, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Yu, Q.; Mai, W.; Liang, P.; Liu, X.; Wang, Y. Dietary protein intake and subsequent risk of type 2 diabetes: A dose-response meta-analysis of prospective cohort studies. Acta Diabetol. 2019, 56, 851–870. [Google Scholar] [CrossRef] [PubMed]
| Intake (g/day) of Dairy Products | ||||
|---|---|---|---|---|
| 0 | 0–53.68 | ≥53.68 | p-Value a | |
| No. subjects | 26,466 (68.33) | 5922 (15.29) | 6347 (16.62) | |
| Male sex | 10,382 (39.23) | 2456 (41.47) | 2496 (39.33) | 0.005 |
| Age (year) | 56.37 ± 11.12 | 54.20 ± 13.01 | 53.49 ± 15.04 | <0.01 |
| Smoking status | 0.002 | |||
| Never | 19,262 (72.78) | 4228 (72.41) | 4675 (72.63) | |
| Ever | 2068 (7.81) | 538 (9.08) | 525 (8.16) | |
| Current | 5136 (19.41) | 1156 (18.51) | 1147 (19.21) | |
| Drinking status | <0.01 | |||
| Never | 20,667 (78.09) | 4392 (74.16) | 4855 (76.49) | |
| Ever | 1136 (4.29) | 298 (5.03) | 359 (5.66) | |
| Current | 4663 (17.62) | 1232 (20.80) | 1133 (17.85) | |
| Physical activity | <0.01 | |||
| Low | 8131 (30.72) | 1906 (32.19) | 2466 (38.85) | |
| Moderate | 9997 (37.77) | 2303 (38.89) | 2314 (36.46) | |
| High | 8338 (31.50) | 1713 (28.93) | 1567 (24.69) | |
| Education level | <0.01 | |||
| Illiterate | 4891 (18.48) | 820 (13.85) | 738 (11.63) | |
| Primary school | 7977 (30.14) | 1491 (25.18) | 1420 (22.37) | |
| Middle school | 10,422 (39.38) | 2514 (42.45) | 2502 (39.42) | |
| High school | 2767 (10.45) | 863 (14.57) | 1191 (18.76) | |
| University or higher | 409 (1.55) | 234 (3.95) | 496 (7.81) | |
| Per capita monthly income | <0.01 | |||
| <500 RMB | 9978 (37.70) | 1941 (32.78) | 1891 (29.79) | |
| 500–999 RMB | 8851 (33.44) | 1967 (33.22) | 1938 (30.53) | |
| 1000–1999 RMB | 6041 (22.83) | 1517 (25.62) | 1730 (27.26) | |
| 2000–2999 RMB | 1094 (4.13) | 304 (5.13) | 497 (7.83) | |
| ≥3000 RMB | 502 (1.90) | 193 (3.26) | 291 (4.58) | |
| Variable | Total Dairy (g/day) a | |||
|---|---|---|---|---|
| 0 | 0–53.68 | ≥53.68 | p-value b | |
| Staple food (g/day) c | 425.19 ± 152.93 | 432.82 ± 160.32 | 405.95 ± 155.60 | <0.001 |
| Livestock (g/day) d | 14.29 (5–35.71) | 16.67 (7.14–42.86) | 21.43 (8.33–50.00) | <0.001 |
| Poultry (g/day) d | 6.67 (1.37–16.67) | 7.14 (3.33–16.67) | 8.33 (3.33–28.57) | <0.001 |
| Fish (g/day) d | 0.82 (0–3.33) | 1.67 (0.27–5.48) | 2.74 (0.27–8.33) | <0.001 |
| Egg (g/day) d | 52.60 ± 44.82 | 55.29 ± 43.51 | 67.64 ± 44.91 | <0.001 |
| Fruit (g/day) d | 129.23 ± 132.68 | 152.84 ± 141.33 | 175.32 ± 144.07 | <0.001 |
| Vegetable (g/day) d | 316.93 ± 179.21 | 320.80 ± 182.12 | 318.60 ± 184.41 | 0.388 |
| Bean (g/day) d | 14.29 (3.33–35.71) | 16.67 (7.14–42.86) | 28.57 (10.96–57.14) | <0.001 |
| Nut (g/day) d | 5.48 (0–16.67) | 7.14 (1.67–21.43) | 8.33 (1.67–33.33) | <0.001 |
| Cereal (g/day) d | 35.00 (10.96–85.71) | 33.33 (13.33–71.43) | 50.00 (16.67–100.00) | <0.001 |
| BMI (kg/m2) e | <0.001 | |||
| Underweight | 560 (2.12) | 162 (2.74) | 215 (3.39) | |
| Normal | 10,209 (38.57) | 2503 (42.27) | 2814 (44.34) | |
| Overweight | 10,612 (40.10) | 2294 (38.74) | 2370 (37.34) | |
| Obesity | 4998 (18.88) | 947 (15.99) | 923 (14.54) | |
| T2DM e | 2502 (9.45) | 481 (8.12) | 671 (10.57) | <0.001 |
| Family history of diabetes e | 1029 (3.89) | 282 (4.76) | 303 (4.77) | <0.001 |
| Intake (g/day) of Dairy Products | ||||
|---|---|---|---|---|
| 0 | 0–53.68 | ≥53.68 | p-Value for Trend | |
| All Cases/n | 2502/26,466 | 481/5922 | 671/6347 | |
| Model 1 | 1.00 | 0.90 (0.81,0.99) | 1.20 (1.10,1.32) | <0.01 |
| Model 2 | 1.00 | 0.96 (0.86,1.07) | 1.31 (1.19,1.45) | <0.01 |
| Model 3 | 1.00 | 0.97 (0.87,1.08) | 1.34 (1.21,1.48) | <0.01 |
| Men Cases/n | 905/10,382 | 194/2456 | 290/2496 | |
| Model 1 | 1.00 | 0.92 (0.78,1.08) | 1.38 (1.20,1.59) | <0.01 |
| Model 2 | 1.00 | 0.98 (0.83,1.16) | 1.41 (1.21,1.64) | <0.01 |
| Model 3 | 1.00 | 0.97 (0.82,1.15) | 1.44 (1.24,1.68) | <0.01 |
| Women Cases/n | 1597/16,084 | 287/3466 | 381/3851 | |
| Model 1 | 1.00 | 0.90 (0.79,1.03) | 1.12 (0.99,1.26) | 0.16 |
| Model 2 | 1.00 | 0.97 (0.85,1.12) | 1.30 (1.14,1.47) | <0.01 |
| Model 3 | 1.00 | 0.99 (0.86,1.14) | 1.33 (1.17,1.51) | <0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, M.; Li, Y.; Wang, C.; Mao, Z.; Zhang, L.; Yang, X.; Cui, S.; Li, L. Consumption of Dairy Products in Relation to Type 2 Diabetes Mellitus in Chinese People: The Henan Rural Cohort Study and an Updated Meta-Analysis. Nutrients 2020, 12, 3827. https://doi.org/10.3390/nu12123827
Fan M, Li Y, Wang C, Mao Z, Zhang L, Yang X, Cui S, Li L. Consumption of Dairy Products in Relation to Type 2 Diabetes Mellitus in Chinese People: The Henan Rural Cohort Study and an Updated Meta-Analysis. Nutrients. 2020; 12(12):3827. https://doi.org/10.3390/nu12123827
Chicago/Turabian StyleFan, Mengying, Yuqian Li, Chongjian Wang, Zhenxing Mao, Lulu Zhang, Xiu Yang, Songyang Cui, and Linlin Li. 2020. "Consumption of Dairy Products in Relation to Type 2 Diabetes Mellitus in Chinese People: The Henan Rural Cohort Study and an Updated Meta-Analysis" Nutrients 12, no. 12: 3827. https://doi.org/10.3390/nu12123827
APA StyleFan, M., Li, Y., Wang, C., Mao, Z., Zhang, L., Yang, X., Cui, S., & Li, L. (2020). Consumption of Dairy Products in Relation to Type 2 Diabetes Mellitus in Chinese People: The Henan Rural Cohort Study and an Updated Meta-Analysis. Nutrients, 12(12), 3827. https://doi.org/10.3390/nu12123827






