Caffeine Supplementation for 4 Days Does Not Induce Tolerance to the Ergogenic Effects Promoted by Acute Intake on Physiological, Metabolic, and Performance Parameters of Cyclists: A Randomized, Double-Blind, Crossover, Placebo-Controlled Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
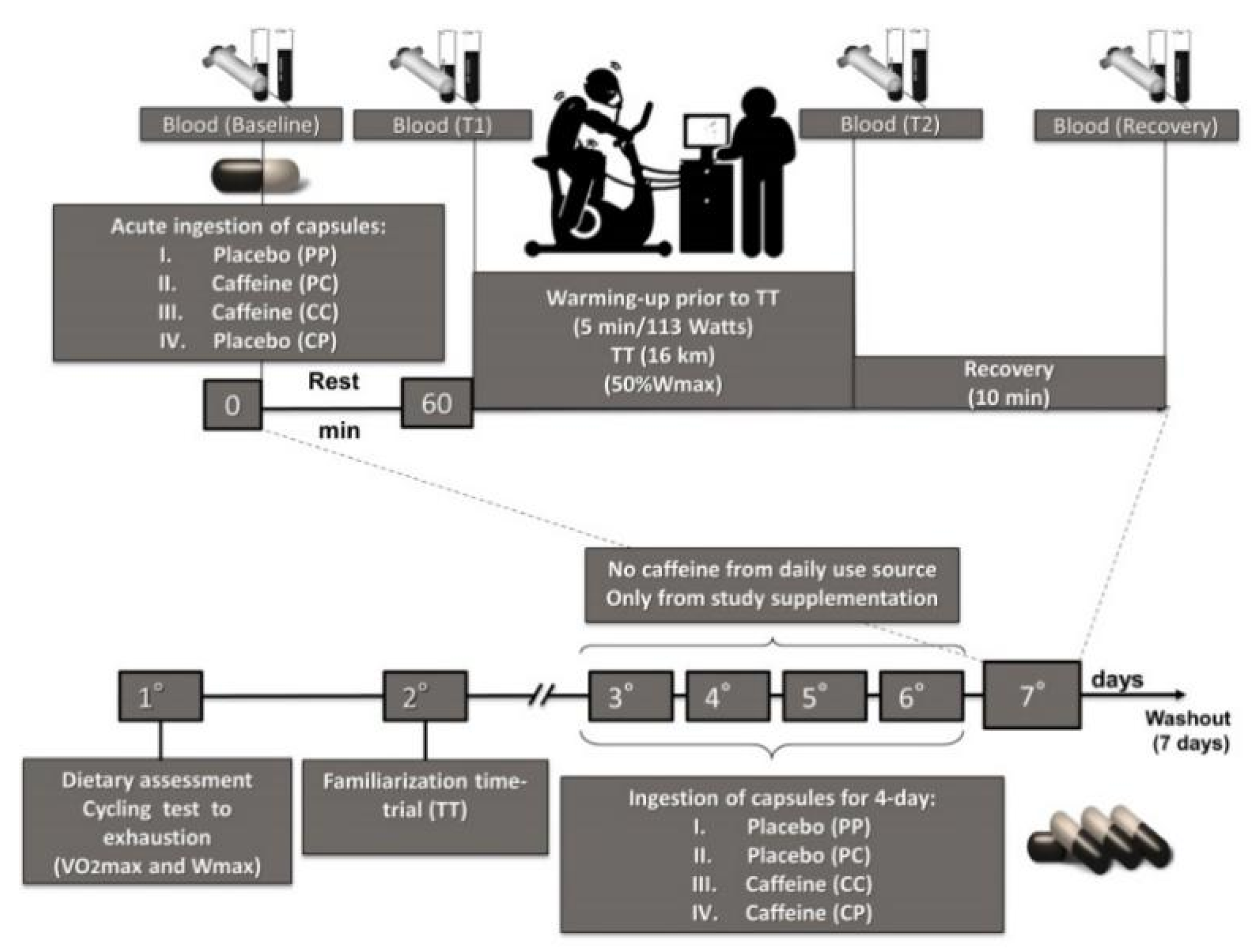
2.2. Study Design
2.3. Habitual Food Intake Recording and Caffeine-Containing Foods
2.4. VO2max and Workload Capacity
2.5. Simulated Cycling TT Performance
2.6. Nutritional Intervention
2.7. Blood Caffeine, Cortisol, Lactate, and Glucose Concentrations Analysis
2.8. Statistical Analysis
3. Results
3.1. Habitual Food Intake Recording
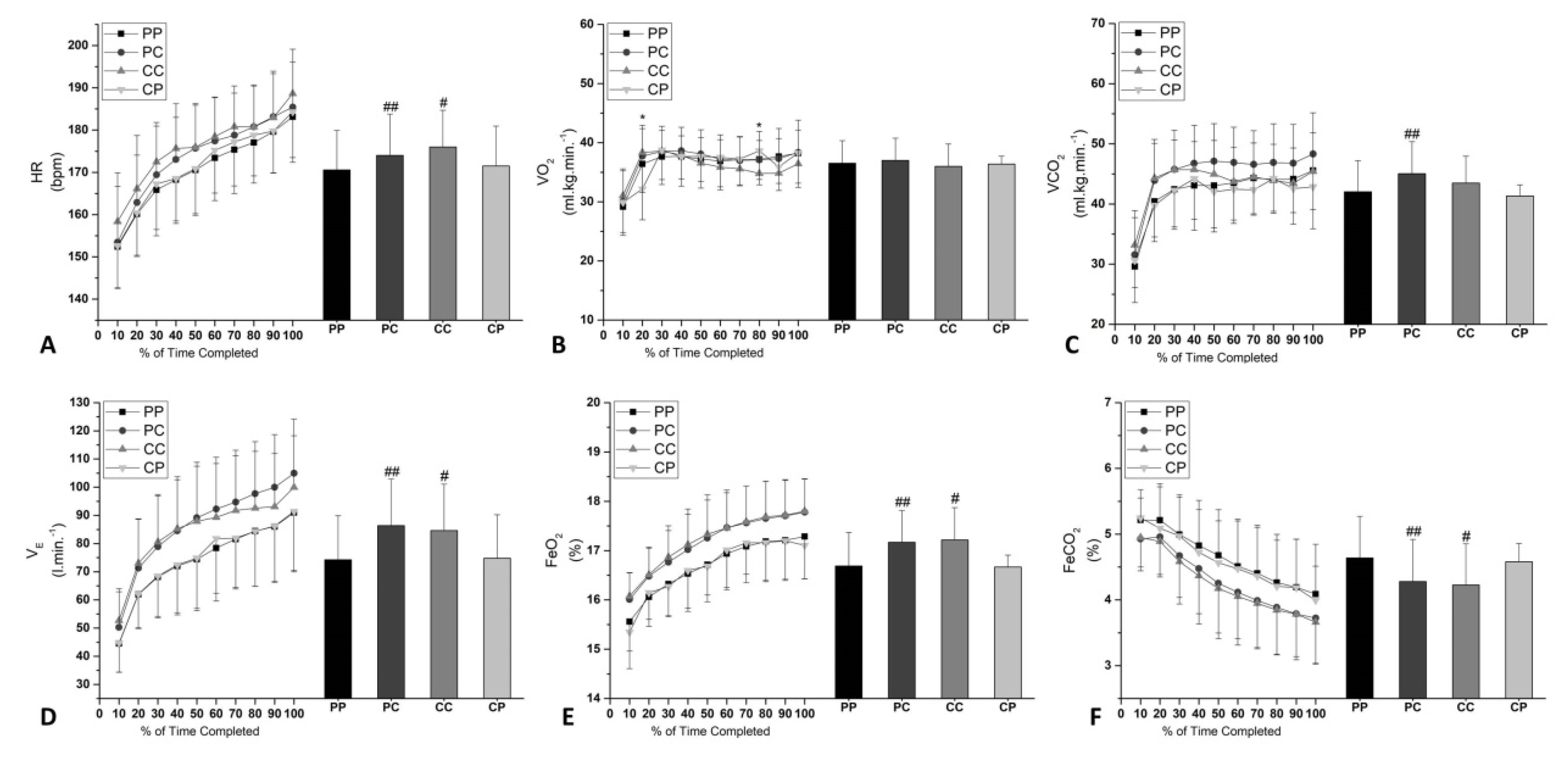
3.2. RPE and Cardiorespiratory Parameters
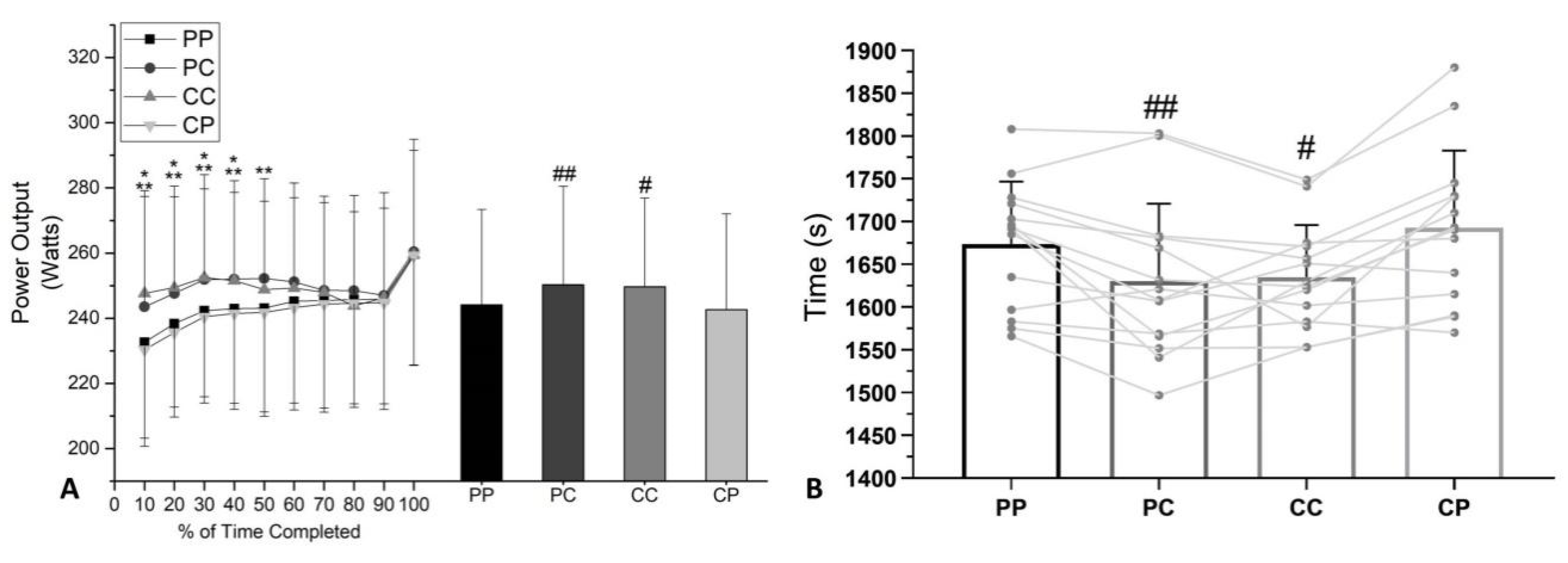
3.3. Power Output and Cycling TT Performance
3.4. Blood Caffeine, Cortisol, Lactate and Glucose Concentrations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ribeiro, B.G.; Morales, A.P.; Sampaio-Jorge, F.; Tinoco, F.S.; Matos, A.A.; Leite, T.C. Acute effects of caffeine intake on athletic performance: A systematic review and meta-analysis. Rev. Chil. Nutr. 2017, 44, 283–291. [Google Scholar] [CrossRef]
- Ribeiro, B.G.; Morales, A.P.; Sampaio-Jorge, F.; Barth, T.; De Oliveira, M.B.; Coelho, G.M.; Leite, T.C. Caffeine attenuates decreases in leg power without increased muscle damage. J. Strength Cond. Res. 2016, 30, 2354–2360. [Google Scholar] [CrossRef]
- Potgieter, S.; Wright, H.H.; Smith, C. Caffeine improves triathlon performance: A field study in males and females. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 228–237. [Google Scholar] [CrossRef]
- Lopes, J.P.; Pliássova, A.; Cunha, R.A. The physiological effects of caffeine on synaptic transmission and plasticity in the mouse hippocampus selectively depend on adenosine A1 and A2A receptors. Biochem. Pharmacol. 2019, 166, 313–321. [Google Scholar] [CrossRef]
- Cox, G.R.; Desbrow, B.; Montgomery, P.G.; Anderson, M.E.; Bruce, C.R.; Macrides, T.A.; Martin, D.T.; Moquin, A.; Roberts, A.; Hawley, J.A.; et al. Effect of different protocols of caffeine intake on metabolism and endurance performance. J. Appl. Physiol. (1985) 2002, 93, 990–999. [Google Scholar] [CrossRef]
- Bell, D.G.; McLellan, T.M. Exercise endurance 1, 3, and 6 h after caffeine ingestion in caffeine users and nonusers. J. Appl. Physiol. (1985) 2002, 93, 1227–1234. [Google Scholar] [CrossRef]
- Irwin, C.; Desbrow, B.; Ellis, A.; O’Keeffe, B.; Grant, G.; Leveritt, M. Caffeine withdrawal and high-intensity endurance cycling performance. J. Sports Sci. 2011, 29, 509–515. [Google Scholar] [CrossRef]
- Gonçalves, L.S.; Painelli, V.S.; Yamaguchi, G.; Oliveira, L.F.; Saunders, B.; Da Silva, R.P.; Maciel, E.; Artioli, G.G.; Roschel, H.; Gualano, B. Dispelling the myth that habitual caffeine consumption influences the performance response to acute caffeine supplementation. J. Appl. Physiol. (1985) 2017, 123, 213–220. [Google Scholar] [CrossRef]
- Beaumont, R.; Cordery, P.; Funnell, M.; Mears, S.; James, L.; Watson, P. Chronic ingestion of a low dose of caffeine induces tolerance to the performance benefits of caffeine. J. Sports Sci. 2017, 35, 1920–1927. [Google Scholar] [CrossRef]
- Lara, B.; Ruiz-Moreno, C.; Salinero, J.J.; Del Coso, J. Time course of tolerance to the performance benefits of caffeine. PLoS ONE 2019, 14, e0210275. [Google Scholar] [CrossRef]
- Wilk, M.; Krzysztofik, M.; Maszczyk, A.; Chycki, J.; Zając, A. The acute effects of caffeine intake on time under tension and power generated during the bench press movement. J. Int. Soc. Sports Nutr. 2019, 16, 8. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef]
- Landrum, R.E. College students use of caffeine and its relationship to personality. Coll. Stud. J. 1992, 26, 151–155. [Google Scholar]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Van Soeren, M.H.; Graham, T.E. Effect of caffeine on metabolism, exercise endurance, and catecholamine responses after withdrawal. J. Appl. Physiol. (1985) 1998, 85, 1493–1501. [Google Scholar] [CrossRef] [PubMed]
- Graham, T.E.; Spriet, L.L. Metabolic, catecholamine, and exercise performance responses to various doses of caffeine. J. Appl. Physiol. (1985) 1995, 78, 867–874. [Google Scholar] [CrossRef]
- Guest, N.; Corey, P.; Vescovi, J.; El-Sohemy, A. Caffeine, CYP1A2 genotype, and endurance performance in athletes. Med. Sci. Sports Exerc. 2018, 50, 1570–1578. [Google Scholar] [CrossRef] [PubMed]
- Pickering, C.; Kiely, J. Are the current guidelines on caffeine use in sport optimal for everyone? Inter-individual variation in caffeine ergogenicity, and a move towards personalised sports nutrition. Sports Med. 2018, 48, 7–16. [Google Scholar] [CrossRef]
- Svenningsson, P.; Nomikos, G.G.; Fredholm, B.B. The stimulatory action and the development of tolerance to caffeine is associated with alterations in gene expression in specific brain regions. J. Neurosci. 1999, 19, 4011–4022. [Google Scholar] [CrossRef]
- Hearris, M.A.; Hammond, K.M.; Fell, J.M.; Morton, J.P. Regulation of muscle glycogen metabolism during exercise: Implications for endurance performance and training adaptations. Nutrients 2018, 10, E298. [Google Scholar] [CrossRef]
- Lovallo, W.R.; al’Absi, M.; Blick, K.; Whitsett, T.L.; Wilson, M.F. Stress-like adrenocorticotropin responses to caffeine in young healthy men. Pharmacol. Biochem. Behav. 1996, 55, 365–369. [Google Scholar] [CrossRef]
- Hill, E.E.; Zacki, E.; Battaglini, C.; Viru, M.; Viru, A.; Hackney, A.C. Exercise and circulating cortisol levels: The intensity threshold effect. J. Endocrinol. Investig. 2008, 31, 587–591. [Google Scholar] [CrossRef]
- Jammes, Y.; Joulia, F.; Steinberg, J.G.; Ravailhe, S.; Delpierre, S.; Condo, J.; Guieu, R.; Delliaux, S. Endogenous adenosine release is involved in the control of heart rate in rats. Can. J. Physiol. Pharmacol. 2015, 675, 667–675. [Google Scholar] [CrossRef]
- Amann, M. Significance of group III and IV muscle afferents for the endurance exercising human. Clin. Exp. Pharmacol. Physiol. 2012, 39, 831–835. [Google Scholar] [CrossRef]
- Passfield, L.; Coakley, S. Comparing time-trial and time to exhaustion performance. J. Sci. Cycling 2014, 3, 39. [Google Scholar]
- Smirmaul, B.P.; de Moraes, A.C.; Angius, L.; Marcora, S.M. Effects of caffeine on neuromuscular fatigue and performance during high-intensity cycling exercise in moderate hypoxia. Eur. J. Appl. Physiol. 2017, 117, 27–38. [Google Scholar] [CrossRef]
- Jones, A.M.; Wilkerson, D.P.; Vanhatalo, A.; Burnley, M. Influence of pacing strategy on O2 uptake and exercise tolerance. Scand. J. Med. Sci. Sports 2008, 18, 615–626. [Google Scholar] [CrossRef]
- Chapman, R.F.; Mickleborough, T.D. The effects of caffeine on ventilation and pulmonary function during exercise: An often-overlooked response. Physician Sportsmed. 2009, 37, 97–103. [Google Scholar] [CrossRef]
- Grgic, J.; Diaz-Lara, F.J.; Del Coso, J.; Duncan, M.J.; Tallis, J.; Pickering, C.; Schoenfeld, B.J.; Mikulic, P. The effects of caffeine ingestion on measures of rowing performance: A systematic review and meta-analysis. Nutrients 2020, 12, 434. [Google Scholar] [CrossRef]
- Saunders, B.; de Oliveira, L.F.; da Silva, R.P.; de Salles Painelli, V.; Gonçalves, L.S.; Yamaguchi, G.; Gualano, B. Placebo in sports nutrition: A proof-of-principle study involving caffeine supplementation. Scand. J. Med. Sci. Spor. 2016, 27, 1240–1247. [Google Scholar] [CrossRef]
- Ribeiro, J.A.; Sebastião, A.M. Caffeine and adenosine. J. Alzheimers Dis. 2010, 20 S1, S3–S15. [Google Scholar] [CrossRef]
- Grgic, J.; Grgic, I.; Pickering, C.; Schoenfeld, B.J.; Bishop, D.J.; Pedisic, Z. Wake up and smell the coffee: Caffeine supplementation and exercise performance-an umbrella review of 21 published meta-analyses. Br. J. Sports Med. 2020, 54, 681–688. [Google Scholar] [CrossRef] [PubMed]
| PP (n = 14) | PC (n = 14) | CC (n = 14) | CP (n = 14) | |
|---|---|---|---|---|
| Energy (kcal) | 2132.93 ± 743.67 | 2056.90 ± 389.76 | 2181.42 ± 452.35 | 2261.41 ± 525.69 |
| Carbohydrates (g∙day−1) | 250.92 ± 67.58 | 242.06 ± 67.23 | 276.70 ± 80.51 | 291.61 ± 67.58 |
| Protein (g∙day−1) | 121.62 ± 45.38 | 114.38 ± 42.55 | 126.62 ± 47.25 | 131.63 ± 45.38 |
| Lipids (g∙day−1) | 78.50 ± 45.69 | 69.23 ± 25.28 | 62.01 ± 27.92 | 76.35 ± 27.62 |
| Distance | ||||||||
|---|---|---|---|---|---|---|---|---|
| Treatment | 2 km | 4 km | 6 km | 8 km | 10 km | 12 km | 14 km | 16 km |
| PP | 3.56 ± 1.01 | 4.67 ± 1.22 a | 5.67 ± 1.32 a | 6.44 ± 1.13 a | 6.89 ± 1.05 a | 8.11 ± 0.93 a | 9.00 ± 1.00 a | 9.11 ± 0.78 a |
| PC | 3.44 ± 1.51 | 4.67 ± 1.32 a | 5.78 ± 1.48 a | 6.56 ± 1.42 a | 7.11 ± 1.05 a | 8.00 ± 1.12 a | 8.78 ± 0.97 a | 9.00 ± 1.0 a |
| CC | 4.00 ± 1.32 | 4.67 ± 1.12 a | 5.56 ± 1.42 a | 6.67 ± 1.22 a | 7.22 ± 0.83 a | 8.00 ± 1.00 a | 8.67 ± 1.32 a | 8.78 ± 1.30 a |
| CP | 4.11 ± 1.54 | 5.22 ± 1.72 a | 6.22 ± 1.64 a | 7.00 ± 1.41 a | 7.44 ± 1.13 a | 8.67 ± 1.12 a | 9.00 ± 1.00 a | 9.11 ± 0.93 a |
| Baseline | T1 | T2 | Recovery | |
|---|---|---|---|---|
| Caffeine (µg·mL−1) | ||||
| PP | 0.34 (0.14) | 0.37 (0.22) | 0.36 (0.16) | 0.37 (0.11) |
| PC | 0.00 (0.32) | 7.60 (1.32) a,b | 7.61 (1.33) a,b | 7.80 (0.56) a,b |
| CC | 0.45 (0.41) | 8.19 (0.76) a,c | 8.35 (0.76) a,c | 8.18 (1.11) a,c |
| CP | 0.33 (0.36) | 0.30 (0.38) | 0.21 (0.31) | 0.27 (0.44) |
| Cortisol (µg·mL−1) | ||||
| PP | 10.74 ± 3.45 | 10.98 ± 3.34 | 14.90 ± 4.2 a | 15.33 ± 4.20 a |
| PC | 9.64 ± 3.66 | 9.70 ± 3.67 | 16.60 ± 5.2 a | 16.84 ± 5.06 a |
| CC | 10.73 ± 3.58 | 10.90 ± 3.39 | 16.02 ± 4.28 a | 16.44 ± 4.23 a |
| CP | 11.49 ± 2.14 | 11.62 ± 2.26 | 15.76 ± 2.61 a | 16.17 ± 2.80 a |
| Lactate (mmol·L−1) | ||||
| PP | 1.09 ± 0.42 | 1.02 ± 0.50 | 12.58 ± 4.59 a | 9.16 ± 3.31 a |
| PC | 1.04 ± 0.60 | 1.38 ± 0.54 | 16.13 ± 5.06 a,b | 12.02 ± 3.68 a,b |
| CC | 1.11 ± 0.48 | 1.30 ± 0.53 | 15.14 ± 4.34 a,c | 12.01 ± 4.07 a,c |
| CP | 1.14 ± 0.77 | 1.01 ± 0.64 | 11.69 ± 4.43 a | 8.83 ± 3.35 a |
| Glucose (mmol·L−1) | ||||
| PP | 5.84 (1.62) | 5.67 (1.35) | 9.80 (4.25) a | 9.87 (3.31) a |
| PC | 5.41 (1.79) | 5.72 (1.33) | 12.10 (6.05) a | 12.55 (6.07) a |
| CC | 5.97 (1.37) | 5.97 (1.46) | 10.82 (2.45) a | 10.80 (4.33) a |
| CP | 6.44 (1.76) | 5.94 (1.33) | 9.54 (4.34) a | 9.66 (3.31) a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morales, A.P.; Sampaio-Jorge, F.; Barth, T.; Pierucci, A.P.T.R.; Ribeiro, B.G. Caffeine Supplementation for 4 Days Does Not Induce Tolerance to the Ergogenic Effects Promoted by Acute Intake on Physiological, Metabolic, and Performance Parameters of Cyclists: A Randomized, Double-Blind, Crossover, Placebo-Controlled Study. Nutrients 2020, 12, 2101. https://doi.org/10.3390/nu12072101
Morales AP, Sampaio-Jorge F, Barth T, Pierucci APTR, Ribeiro BG. Caffeine Supplementation for 4 Days Does Not Induce Tolerance to the Ergogenic Effects Promoted by Acute Intake on Physiological, Metabolic, and Performance Parameters of Cyclists: A Randomized, Double-Blind, Crossover, Placebo-Controlled Study. Nutrients. 2020; 12(7):2101. https://doi.org/10.3390/nu12072101
Chicago/Turabian StyleMorales, Anderson Pontes, Felipe Sampaio-Jorge, Thiago Barth, Anna Paola Trindade Rocha Pierucci, and Beatriz Gonçalves Ribeiro. 2020. "Caffeine Supplementation for 4 Days Does Not Induce Tolerance to the Ergogenic Effects Promoted by Acute Intake on Physiological, Metabolic, and Performance Parameters of Cyclists: A Randomized, Double-Blind, Crossover, Placebo-Controlled Study" Nutrients 12, no. 7: 2101. https://doi.org/10.3390/nu12072101
APA StyleMorales, A. P., Sampaio-Jorge, F., Barth, T., Pierucci, A. P. T. R., & Ribeiro, B. G. (2020). Caffeine Supplementation for 4 Days Does Not Induce Tolerance to the Ergogenic Effects Promoted by Acute Intake on Physiological, Metabolic, and Performance Parameters of Cyclists: A Randomized, Double-Blind, Crossover, Placebo-Controlled Study. Nutrients, 12(7), 2101. https://doi.org/10.3390/nu12072101





