New Terpenoids from Chamaecyparis formosensis (Cupressaceae) Leaves with Modulatory Activity on Matrix Metalloproteases 2 and 9
Abstract
1. Introduction
2. Results and Discussion
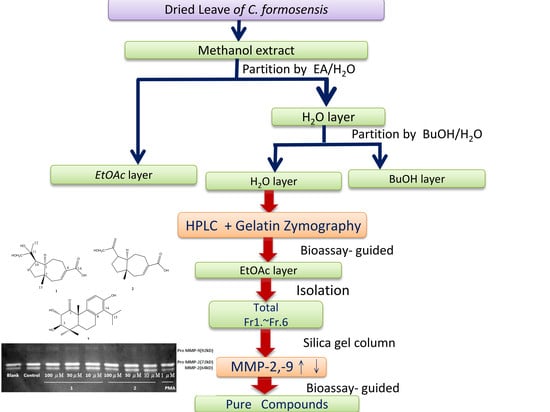
2.1. Compounds Isolated from the Leaves of C. formosensis
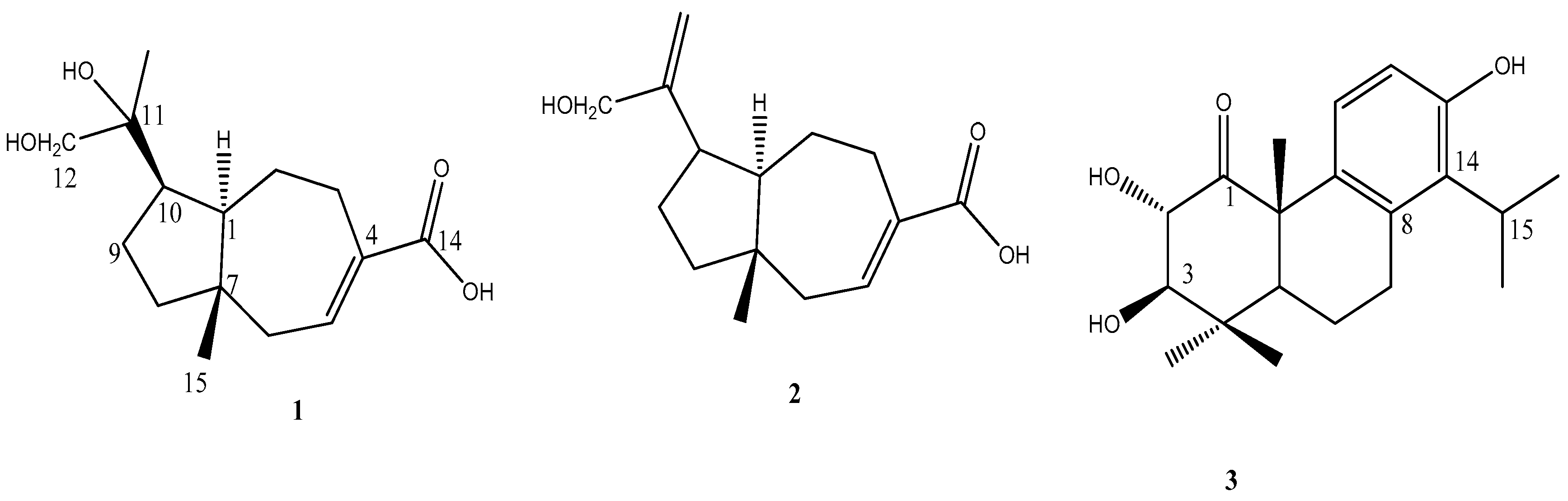
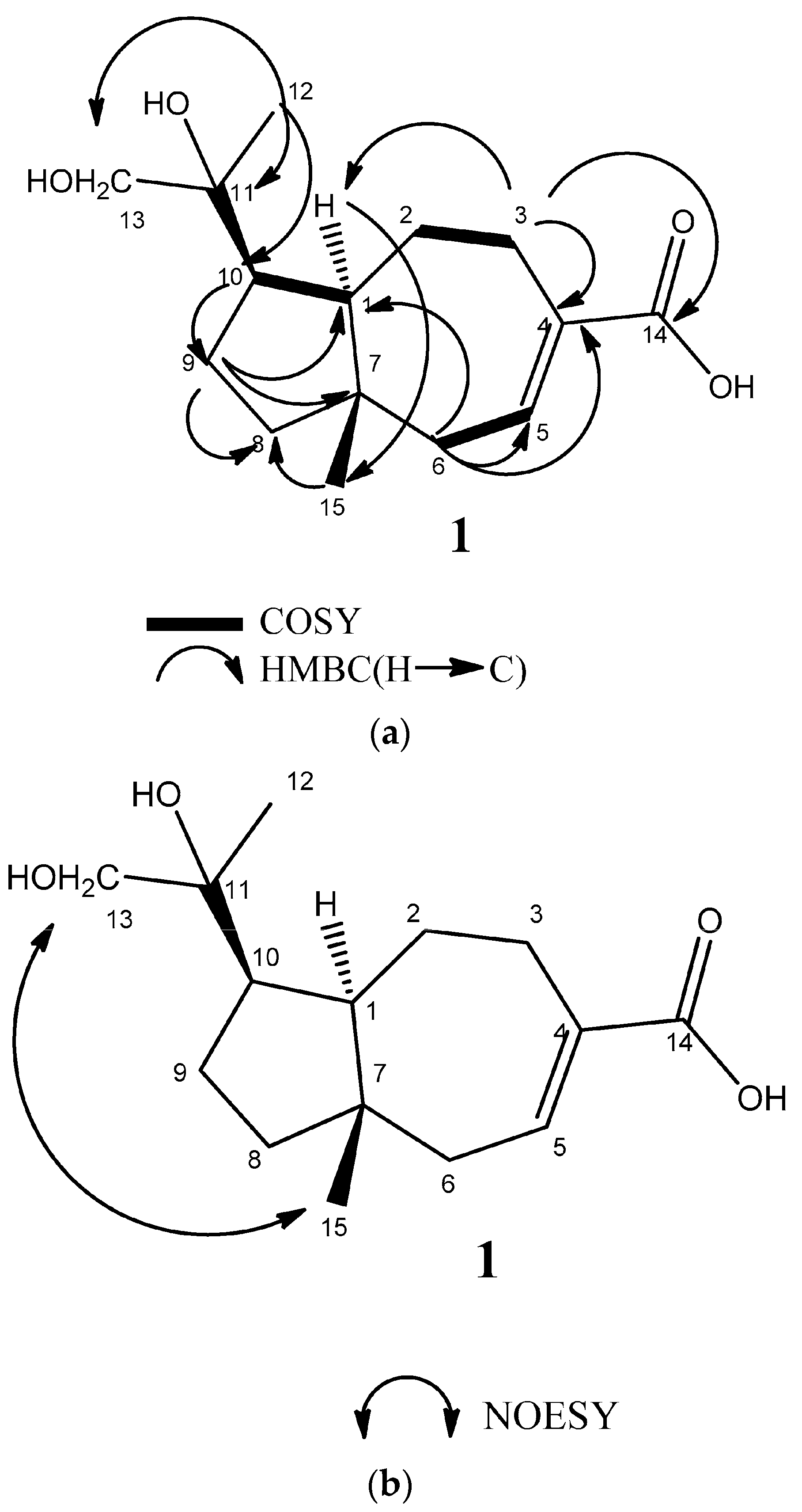
2.2. Structural Elucidation of Compounds 1–3
2.3. Evaluation of Modulatory Effects of Compounds on MMP-2 and MMP-9 Expression in HT-1080 Cells
3. Materials and Methods
3.1. General Methods
3.2. Plant Material
3.3. Extraction and Isolation
3.4. 11,12-Dihydroxyisodaucenoic Acid (1)
3.5. 12-Hydroxyisodaucenoic Acid (2)
3.6. 1-Oxo-2α,3β-dihydroxytotarol (3)
3.7. Cell Culture
3.8. Gelatin Zymography
3.9. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, W.P.; Hwang, C.Y.; Lin, T.P.; Hwang, S.Y. Historical biogeography and phylogenetic relationships of the genus Chamaecyparis (Cupressaceae) inferred from chloroplast DNA polymorphism. Plant Syst. Evol. 2003, 241, 13–28. [Google Scholar] [CrossRef]
- Yang, J.K.; Choi, M.S.; Seo, W.T.; Rinker, D.L.; Han, S.W.; Cheong, G.W. Chemical composition and antimicrobial activity of Chamaecyparis obtusa leaf essential oil. Fitoterapia 2007, 78, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Park, I.K.; Lee, S.G.; Choi, D.H.; Park, J.D.; Ahn, Y.J. Insecticidal activities of constituents identified in the essential oil from leaves of Chamaecyparis obtusa against Callosobruchus chinensis (L.) and Sitophilus oryzae (L.). J. Stored Prod. Res. 2003, 39, 375–384. [Google Scholar] [CrossRef]
- Hsieh, C.C.; Kuo, Y.H.; Kuo, C.C.; Chen, L.T.; Cheung, C.H.; Chao, T.Y.; Lin, C.H.; Pan, W.Y.; Chang, C.Y.; Chien, S.C. Chamaecypanone C, a novel skeleton microtubule inhibitor, with anticancer activity by trigger caspase 8-Fas/FasL dependent apoptotic pathway in human cancer cells. Biochem. Pharmacol. 2010, 79, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Lu, F.Y.; Ou, C.H. Trees of Taiwan. Monographic Publication No. 7; College of Agriculture, National Chung Hsing University: Taichung, Taiwan, 1988; pp. 91–92. [Google Scholar]
- Li, H.-L.; Keng, H. Flora of Taiwan, 2nd ed.; Huang, T.-C., Ed.; Epoch Publ. Co.: Taipei, Taiwan, 1994. [Google Scholar]
- Ho, C.L.; Hua, K.F.; Hsu, K.P.; Wang, E.I.C.; Su, Y.C. Composition and antipathogenic activities of the twig essential oil of Chamaecyparis formosensis from Taiwan. Nat. Prod. Commun. 2012, 7, 933–936. [Google Scholar] [PubMed]
- Wang, S.Y.; Wu, C.L.; Chu, F.H.; Chien, S.C.; Kuo, Y.H.; Shyur, L.F.; Chang, S.T. Chemical composition and antifungal activity of essential oil isolated from Chamaecyparis formosensis Matsum Wood. Holzforschung 2005, 59, 295–299. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Lin, C.Y.; Chang, S.T. Antitermitic activities of wood essential oil and its constituents from Chamaecyparis formosensis. Wood Sci. Technol. 2016, 50, 663–676. [Google Scholar] [CrossRef]
- Kafuka, K.; Ichikawa, N. The volatile compounds from leaves of Chamaecyparis formosensis. Nippon Kagaku Kaishi 1931, 52, 222–228. [Google Scholar] [CrossRef]
- Nozoe, T.; Chen, Y.S.; Toda, T. The structure of chamaecynone—A novel norsesquitenoid from Chamaecyparis formosensis Matsum. Tetrahedron Lett. 1966, 31, 3663–3669. [Google Scholar] [CrossRef]
- Fang, J.M.; Lai, L.J.; Cheng, Y.S. The constituents of the leaves of Chamaecyparis formosensis. J. Chin. Chem. Soc. 1986, 33, 265–266. [Google Scholar] [CrossRef]
- Fang, J.M.; Sheu, C.M.; Cheng, Y.S. A study of the constituents of the bark of Chamaecyparis formosensis. J. Chin. Chem. Soc. 1986, 33, 245–249. [Google Scholar] [CrossRef]
- Hsu, K.C.; Fang, J.M.; Cheng, Y.S. Diterpenes from pericarps of Chamaecyparis formosensis. J. Nat. Prod. 1995, 58, 1592–1595. [Google Scholar] [CrossRef]
- Zitka, O.; Kukacka, J.; Krizkov, S.; Haska, D.; Adam, V.; Masarik, M.; Prusa, R.; Kizek, R. Matrix metalloproteinases. Curr. Med. Chem. 2010, 17, 3751–3768. [Google Scholar] [CrossRef] [PubMed]
- Ra, H.J.; Parks, W.C. Control of Matrix Metalloproteinase Catalytic Activity Matrix Biology. J. Int. Soc. Matrix Biol. 2007, 26, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Kessenbrock, K.; Plaks, V.; Werb, Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell 2010, 141, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.M.; Gaggar, A.; Blalock, J.E. MMP generated matrikines. Matrix Biol. 2015, 44–46, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Farina, A.R.; Mackay, A.R. Gelatinase B/MMP-9 in tumour pathogenesis and progression. Cancers 2014, 6, 240–296. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Manuel, J.A.; Gawronska-Kozak, B. Matrix metalloproteinase 9 (MMP-9) is upregulated during scarless wound healing in athymic nude mice. Matrix Biol. 2006, 25, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Quan, T.; Purohit, T.; Shao, Y.; Cho, M.K.; He, T.; Varani, J.; Kang, S.; Voorhees, J.J. Collagen fragmentation promotes oxidative stress and elevates matrix metalloproteinase-1 in fibroblasts in aged human skin. Am. J. Pathol. 2009, 174, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.C. Study on the Constituents of the Leaves of Chamecyparis formosensis Matsum. Ph.D. Thesis, Department of Chemistry, National Taiwan University, Taipei, Taiwan, 1997. [Google Scholar]
- Knapp, H.; Weigand, C.; Gloser, J.; Winterhalter, P. 2-Hydroxy-2,6,10,10-tetramethyl-1-oxaspiro[4.5]dec-6-en-8-one: Precursor of 8,9-Dehydrotheaspirone in White-Fleshed Nectarines. J. Agric. Food Chem. 1997, 45, 1309–1313. [Google Scholar] [CrossRef]
- Ulubelen, A.; Topcu, G.; Chen, S.; Cai, P.; Snyder, J.K. A new abietane diterpene from Salvia wiedemannii Boiss. J. Org. Chem. 1991, 56, 7354–7356. [Google Scholar] [CrossRef]
- Sanchez, A.J.; Konopelski, J.P. Phenol benzylic epoxide to quinone methide electron reorganization: Synthesis of (f)-taxodone. J. Org. Chem. 1994, 59, 5445–5452. [Google Scholar] [CrossRef]
- Lin, T.C.; Fang, J.M.; Cheng, Y.S. Terpenes and lignans from leaves of Chamaecyparis formosensis. Phytochemistry 1999, 51, 793–801. [Google Scholar] [CrossRef]
- Barrero, A.F.; Sanchez, J.F.; Alvarez-Manzaneda, E.J.; Mu˜noz, M.; Haidour, A. Diterpenoids and cyclolanostanolides from Abies marocana. Phytochemistry 1992, 31, 615–620. [Google Scholar] [CrossRef]
- Kashiwada, Y.; Nishimura, K.; Kurimoto, S.; Takaishi, Y. New 29-nor-cycloartanes with a 3,4-seco- and a novel 2,3-seco-structure from the leaves of Sinocalycanthus chinensis. Bioorg. Med. Chem. 2011, 19, 2790–2796. [Google Scholar] [CrossRef] [PubMed]
- Estévez-Braun, A.; Estévez-Reyes, R.; González-Pérez, J.A.; González, A.G. Busaliol and busalicifol, two new tetrahydrofuran liganans from Bupleurum Salicifolium. J. Nat. Prod. 1995, 58, 887–892. [Google Scholar] [CrossRef][Green Version]
- Nishibe, S.; Chiba, M.; Sakushima, A.; Hisada, S.; Yamanouchi, S.; Takido, M.; Sankawa, U.; Sakakibara, A. Introduction of an alcoholic hydroxyl group into 2,3-dibenzylbutyrolactone lignans with oxidizing agents and carbon-13 nuclear magnetic resonance spectra of the oxidation products. Chem. Pharm. Bull. 1980, 28, 850–860. [Google Scholar] [CrossRef]
- Sena Filho, J.G.; Nimmo, S.L.; Xavier, H.S.; Barbosa-Filho, J.M.; Cichewicz, R.H. Phenylethanoid and lignan glycosides from polar extracts of Lantana, a genus of verbenaceous plants widely used in traditional herbal therapies. J. Nat. Prod. 2009, 72, 1344–1347. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.L.; Li, J.; Wang, N.L.; Yao, X.S. Flavonoids and a new polyacetylene from Bidens parviflora Willd. Molecules 2008, 13, 1931–1941. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.F.; Lee, S.S. Flavonoid composition in the leaves of Twelve Litsea and Neolitsea Plants. J. Chin. Chem. Soc. 2011, 58, 376–383. [Google Scholar] [CrossRef]
- Marcus, V.B.; dos Santos, J.B.; Juceni, P.D.; Jorge, M.D. Biflavonoids and other phenolics from Caesalpinia pyramidalis (Fabaceae). J. Braz. Chem. Soc. 2005, 16, 1402–1405. [Google Scholar]
- Velandia, J.R.; de Carvalho, M.G.; Braz-Filho, R.; Werle, A.A. Biflavonoids and a Glucopyranoside Derivative from Ouratea semiserrata. Phytochem. Anal. 2002, 13, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Garg, H.S.; Mitra, C.R. Putraflavone, a new biflavonoid from Putranjiva roxburghii. Phytochemistry 1971, 10, 2787–2791. [Google Scholar] [CrossRef]
- Li, S.H.; Zhang, H.J.; Niu, X.M.; Yao, P.; Sun, H.D.; Fong, H.S. Chemical Constituents from Amentotaxus yunnanensis and Torreya yunnanensis. J. Nat. Prod. 2003, 66, 1002–1005. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.A.; Harborne, J.B.; Tomas-Barberan, F.A. Biflavonoids in the primitive monocots Isophysis tasmanica and Xerophyta plicata. Phyrochemistry 1987, 26, 2553–2555. [Google Scholar] [CrossRef]
- Pakrashi, A.; Kabir, S.N.; Ray, H. 3-(4-Hydroxyphenyl)-2-propenoicacid-A reproductive inhibitor in male rat. Contraception 1981, 23, 677–686. [Google Scholar] [CrossRef]
- Matsumoto, K.; Feng, C.; Handa, S.; Oguma, T.; Katsuki, T. Asymmetric epoxidation of (Z)-enol esters catalyzed by titanium(salalen) complex with aqueous hydrogen peroxide. Tetrahedron 2011, 67, 6474–6478. [Google Scholar] [CrossRef]
- Zeng, B.B.; Wu, Y.; Jiang, S.; Yu, Q.; Yao, Z.J.; Liu, Z.H.; Li, H.Y.; Li, Y.; Chen, X.G.; Wu, Y.L. Studies on mimicry of naturally occurring annonaceous acetogenins: Non-THF analogues leading to remarkable selective cytotoxicity against human tumor cells. Chem. Eur. J. 2003, 9, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Li, Y.; Jiang, M.; Wang, J.; Fu, H. General copper-catalyzed transformations of functional groups from arylboronic acids in water. Chem. Eur. J. 2011, 17, 5652–5660. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, M.A.; Ahmad, A.S.; Nafady, A.M.; Mansour, A.I. Chemical composition of the stem bark and leaves of Ficus pandurata Hance. Nat. Prod. Res. 2009, 23, 1218–1230. [Google Scholar] [CrossRef] [PubMed]
- Hashidoko, Y.; Tashara, S.; Mizutani, J. Carotanoids and an Acoranoid from Rosa rugosa Leaves. Phytochemistry 1991, 30, 3729–3739. [Google Scholar] [CrossRef]
- Ma, W.H.; Huang, H.; Zhou, P.; Chen, D.F. Schisanwilsonenes A-C, anti-HBV carotane sesquiterpenoids from the fruits of Schisandra wilsoniana. J. Nat. Prod. 2009, 72, 676–678. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.T.; Lin, J.K. EGCG inhibits the invavion of highly invasive CL1-5 lung cancer cells through suppressing MMP-2 expression via JNK signaling and induces G2/Marrest. J. Agric. Food Chem. 2011, 59, 13318–13327. [Google Scholar] [CrossRef] [PubMed]
- Tseng, H.H.; Chen, P.N.; Kuo, W.H.; Wang, J.W.; Chu, S.C.; Hsieh, Y.S. Antimetastatic potentials of phyllanthusurinaria L on A549 and Lewis lung carcinoma cells via repression of matrix-degrading proteases. Integr. Cancer Ther. 2011, 10, 341–349. [Google Scholar]
- Lee, C.W.; Choi, H.J.; Kim, H.S.; Kim, D.H.; Chang, I.S.; Moon, H.T.; Lee, S.Y.; Oh, W.K.; Woo, E.R. Biflavonoids isolated from Selaginella tamariscina regulate the expression of matrix metalloproteinase in human skin fibroblasrs. Bioorg. Med. Chem. 2008, 16, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Berg, G.; Miksztowicz, V.; Schreier, L. Metalloproteinases in metabolic syndrome. Clin. Chim. Acta 2011, 412, 1731–1739. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Position | 1 | 2 | ||
|---|---|---|---|---|
| δH | δC | δH | δC | |
| 1 | 1.99 (1H, m) | 58.0 | 1.89 (1H, m) | 57.2 |
| 2 | 1.48 (1H, m) 2.54 (1H, m) | 24.2 | 1.25 (1H, m) 1.54 (1H, m) | 24.0 |
| 3 | 1.80 (1H, m) 3.08 (1H, m) | 30.9 | 3.03 (1H, m) 1.90 (1H, m) | 28.4 |
| 4 | 136.4 | 134.5 | ||
| 5 | 7.10 (1H, m) | 142.2 | 7.20 (1H, m) | 144.8 |
| 6 | 2.40 (1H, m) 2.10 (1H, m) | 43.3 | 2.45 (1H, dd, J = 14.0, 9.6) 2.03 (1H, m) | 43.0 |
| 7 | 42.7 | 42.9 | ||
| 8 | 1.38 (1H, m) 1.48 (1H, m) | 42.4 | 1.58 (1H, br dd, J = 11.6, 6.7) 1.46 (1H, br dd, J = 11.6, 6.7) | 42.5 |
| 9 | 1.65 (1H, m) 1.78 (1H, m) | 27.7 | 1.70 (2H, m) | 30.1 |
| 10 | 2.59 (1H, m) | 48.6 | 2.91 (1H, m) | 45.6 |
| 11 | 75.9 | 151.7 | ||
| 12 | 3.28 (1H, d, J = 10.4 Hz) 3.33 (1H, d, J = 10.4 Hz) | 70.9 | 4.06 (1H, d, J = 14) 3.98 (1H, d, J = 14) | 67.7 |
| 13 | 1.10 (3H, s) | 22.0 | 4.93 (1H, br s), 5.16 (1H, br s) | 111.4 |
| 14 | 169.6 | 172.8 | ||
| 15 | 0.80 (3H, s) | 19.7 | 0.81 (3H, s, CH3) | 19.9 |
| Compound | MMP-2 | MMP-9 |
|---|---|---|
| PMA | 2.5 * (Pro MMP-9 **, 1 μM ***) | |
| EGCG | 0.2–0.4 (Pro MMP-2, 100 μM) | |
| 13-Hydroxyisodaucenoic acid (2) | 1.26 (50 µM) 1.32 (10 µM) | |
| Nortrachelogenin (18) | 1.30 (50 µM) 1.59 (10 µM) | 1.54 (10 µM) |
| 8′β-hydroxynortrachelogenin (19) | 1.23 (50 µM) 1.34 (10 µM) | |
| Epicatechin (23) | 1.31 (50 µM) 1.40 (10 µM) | |
| Catechin (24) | 1.29 (50 µM) 1.17 (10 µM) | |
| 4′′′-O-methylametoflavone (26) | 1.42 (Pro MMP-9 ***, 10 µM) | |
| 4′′′,7-dimethoxyametoflavone (27) | 1.18 (Pro-MMP-9, 50 µM) |
| Position | δH (mult., J in Hz) | δC |
|---|---|---|
| 1 | 210.4 | |
| 2 | 4.70 (1H, dd, J = 10.4, 5.5 Hz) | 75.4 |
| 3 | 3.05 (1H, d, J = 10.4 Hz) | 85.5 |
| 4 | 38.6 | |
| 5 | 1.45 (1H, dd, J = 12.1, 1.2 Hz) | 49.8 |
| 6 | 1.55 (1H, m) | 18.7 |
| 2.02 (1H, m) | ||
| 7 | 2.67 (1H, m) | 29.9 |
| 2.95 (1H, m) | ||
| 8 | 132.5 | |
| 9 | 131.3 | |
| 10 | 53.5 | |
| 11 | 6.98 (1H, d, J = 8.5 Hz) | 128.9 |
| 12 | 6.57 (1H, d, J = 8.5 Hz) | 114.4 |
| 13 | 153.1 | |
| 14 | 132.5 | |
| 15 | 3.20 (1H, sept) | 29.6 |
| 16 | 1.30 (3H, d, J = 6.7 Hz) | 20.5 |
| 17 | 1.31 (3H, d, J = 6.7 Hz) | 20.4 |
| 18 | 1.14 (3H, s) | 29.0 |
| 19 | 1.15 (3H, s) | 17.0 |
| 20 | 1.61 (3H, s) | 25.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, M.-L.; Mei, H.-C.; Kuo, I.-C.; Hsiao, G.; Kuo, Y.-H.; Lee, C.-K. New Terpenoids from Chamaecyparis formosensis (Cupressaceae) Leaves with Modulatory Activity on Matrix Metalloproteases 2 and 9. Molecules 2018, 23, 604. https://doi.org/10.3390/molecules23030604
Chang M-L, Mei H-C, Kuo I-C, Hsiao G, Kuo Y-H, Lee C-K. New Terpenoids from Chamaecyparis formosensis (Cupressaceae) Leaves with Modulatory Activity on Matrix Metalloproteases 2 and 9. Molecules. 2018; 23(3):604. https://doi.org/10.3390/molecules23030604
Chicago/Turabian StyleChang, Meng-Lun, Hui-Ching Mei, I-Chih Kuo, George Hsiao, Yueh-Hsiung Kuo, and Ching-Kuo Lee. 2018. "New Terpenoids from Chamaecyparis formosensis (Cupressaceae) Leaves with Modulatory Activity on Matrix Metalloproteases 2 and 9" Molecules 23, no. 3: 604. https://doi.org/10.3390/molecules23030604
APA StyleChang, M.-L., Mei, H.-C., Kuo, I.-C., Hsiao, G., Kuo, Y.-H., & Lee, C.-K. (2018). New Terpenoids from Chamaecyparis formosensis (Cupressaceae) Leaves with Modulatory Activity on Matrix Metalloproteases 2 and 9. Molecules, 23(3), 604. https://doi.org/10.3390/molecules23030604