Transforming By-Products into Functional Resources: The Potential of Cucurbitaceae Family Seeds in Cosmetics
Abstract
1. Introduction
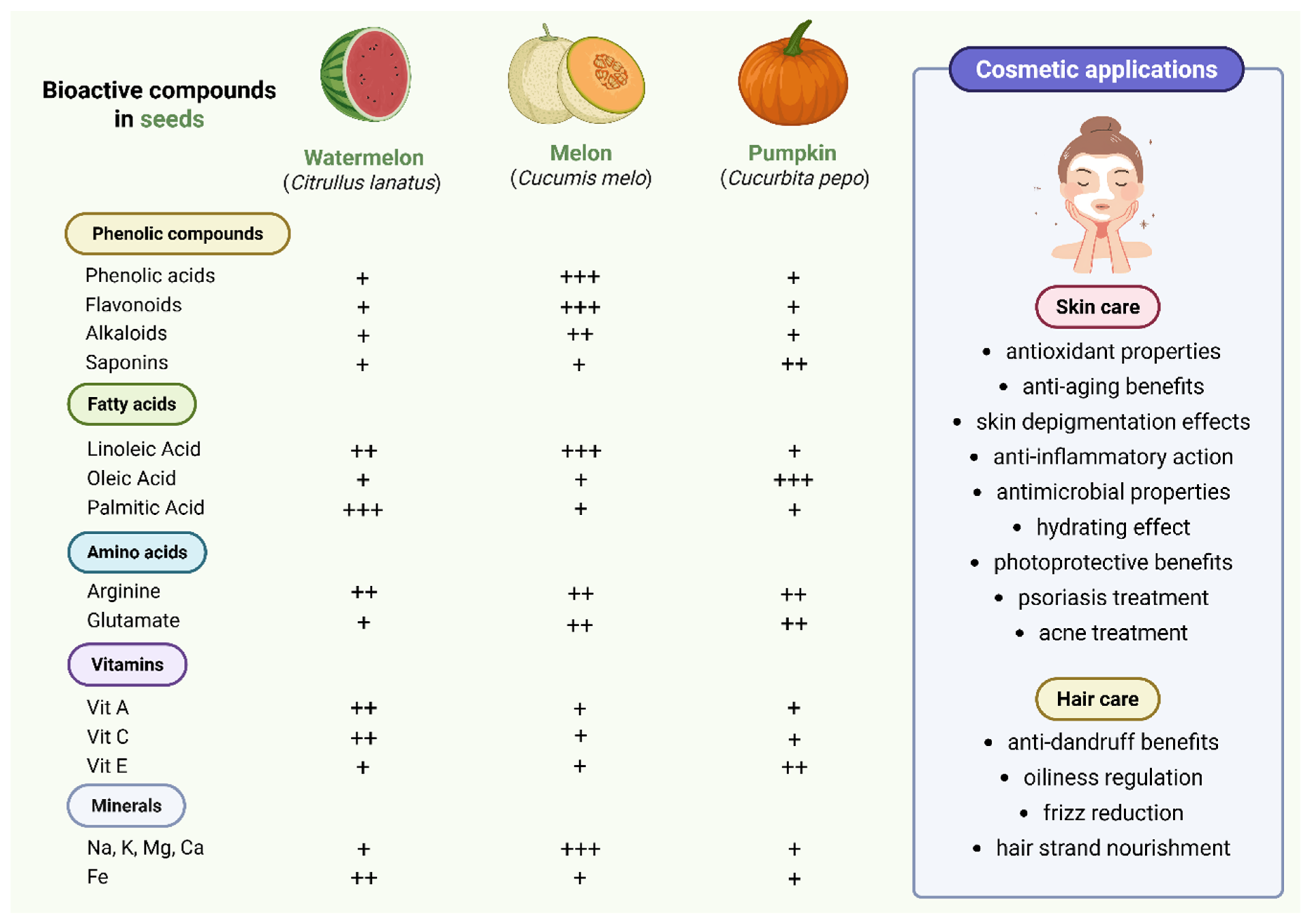
2. Chemical Composition of Cucurbitaceae Seeds and Their Application in Cosmetics
2.1. Phenolic Compounds
2.2. Fatty Acids, Tocopherols, and Phytosterols
2.3. Aminoacids
| Amino Acid | C. lanatus | C. melo | C. pepo | Properties/Applications |
|---|---|---|---|---|
| Alanine | 3.786–5.05 [99,100] | 4.028–4.45 [45,101] | 3.7–4.210 [102,103] | Used in conditioners to improve hair surface hydrophobicity of bleach-damaged hair [115] |
| Arginine | 4.977–15.21 [99,100] | 13.044–13.4 [45,101] | 3.182–16.4 [102,103] | Antioxidant, supports collagen production [29], increasing collagen deposition [116] |
| Aspartate | 6.09–10.39 [99] | 7.318–8.98 [45,101] | 9.0 [102] | Antioxidant, collagen production-promoting effect [116] |
| Cysteine | 0.043–6.09 [99] | 5.32 [101] | 1.1 [102] | Used in hair waving and straightening formulations; fragrance ingredients [97] |
| Glutamate | 4.462–16.75 [99,100] | 19.7–20.523 [45,101] | 17.9 [102] | Conditioning, cleansing, and emulsifying properties [96]; wrinkle formation and skin roughness reduction [111] |
| Glycine | 1.20–5.04 [99] | 1.983–4.94 [45,101] | 4.2–5.912 [102,103] | Improvement in moisture retention and collagen production and strengthens the skin; skin repair and regeneration [29] |
| Histidine | 2.15–2.887 [99,100] | 1.58 [101] | 2.312–2.4 [102,103] | Improve hair tensile strength [115] |
| Isoleucine | 3.48–5.21 [99] | 4.631–5.01 [45,101] | 2.141–3.9 [102,103] | Repair and/or prevention of split ends in hair (in combination with lysine) [113] |
| Leucine | 1.723–7.19 [99,100] | 7.36–9.640 [45,101] | 6.9–8.512 [102,103] | Minimize muscular flaccidity and decreased muscle tone; improving intramuscular protein synthesis, potentiating the increase in tonus and the contractile response of the muscle during daily living activities [117] |
| Lysine | 0.315–3.05 [99,100] | 2.81–3.369 [45,101] | 3.012–3.8 [102,103] | Repair and/or prevention of split ends in hair (in combination with isoleucine) [113] |
| Methionine | 0.88–2.17 [99] | 0.82–2.205 [45,101] | 2.1–7.217 [102,103] | Skin-conditioning agent, softener, and conditioner [118] |
| Phenylalanine | 4.35–5.83 [99] | 4.74–6.335 [45,101] | 5.5–7.217 [102,103] | Production of the pigment melanin; treatment of vitiligo [119]; improvement in hair tensile strength [115] |
| Proline | 3.05–4.142 [99,100] | 3.86 [101] | 3.4–4.126 [102,103] | Collagen biosynthesis [116] |
| Serine | 3.91–4.96 [99] | 2.381–4.86 [45,101] | 2.013–5.2 [102,103] | Ultraviolet damage reduction [111] |
| Threonine | 2.61–4.249 [99,100] | 3.58–4.741 [45,101] | 3.1–1.963 [102,103] | Makes up collagen and elastin; skin-conditioning agent; hair styling and care, hair conditioner, hair waving or straightening [120] |
| Tryptophan | 1.037–1.17 [99,100] | 12.991 [45] | 0.517–2.7 [102,103] | Improve the strength and condition of the hair [112] |
| Tyrosine | 3.298–3.96 [99,100] | 2.40–4.06 [45,101] | 3.012–3.9 [102,103] | Anti-melanogenic effect [121] |
| Valine | 3.48–4.572 [99,100] | 1.430–4.34 [45,101] | 4.021–4.7 [102,103] | Make-up (eyeliners or brow-coloring products); flavoring [122] |
2.4. Vitamins
| Vitamin | C. lanatus | C. melo | C. pepo | Properties/Applications |
|---|---|---|---|---|
| Vitamin A | 701 [131] | 1.513 [132] | 0.048–0.19 [133,134] | Promote a normal keratinization cycle; control sebum production in acne; reverse and treat damage from sun exposure, striae, and cellulite [28] |
| Vitamin B1 | 0.2–2.14 [130,131] | 0.013 [132] | 0.34–2.72 [133,134] | Antipruritic action, rosacea, and seborrheic diseases treatment [124] |
| Vitamin B2 | 0.796–2.54 [110,131] | Not found | 0.52–1.5 [133,134] | Anti-inflammatory action, rosacea, and seborrheic disease treatment [124] |
| Vitamin B3 | 0.81–33.2 [130,131] | Not found | 2.86–48 [133,134] | Antimicrobial; anti-inflammatory; antipruritic; vasoactive; lightening; photoprotective; sebostatic effects, acne, atopic dermatitis, skin aging, melasma, light damage of the skin, hyperpigmentation treatment [124] |
| Vitamin B6 | 0.053–125.50 [110,131] | Not found | 0.37–1.4 [133,134] | Increase skin barrier’s integrity and function; atopic dermatitis treatment [124] |
| Vitamin B9 | 1.843 [110] | 0.0043 [132] | 0.09–0.58 [133,134] | Cell replication, gene regulation, skin renewal, photodamaged treatment, anti-aging [124] |
| Vitamin B12 | 1.07–1.092 [110,131] | Not found | Not found | Pro-inflammatory cytokine production suppression; cell proliferation; lymphocyte activity enhancement; atopic dermatitis and childhood eczema treatment [124] |
| Vitamin C | 19.450–372.90 [110,131] | 1.636 [132] | 2.72–3 [133,134] | Antioxidant; regulation of collagen synthesis; formation of a hydrolipidic mantle in the stratum corneum; vitamin E regeneration; photoprotection (when combined with vitamin E) [28] |
| Vitamin D | 8.760–13.98 [110,131] | Not found | Not found | Control of cutaneous immune system and epithelial proliferation stimulates differentiation [28]; photoprotection and prevention of skin aging [125]; atopic dermatitis, vitiligo, acne, and rosacea treatment [126] |
| Vitamin E | 3.533–8.86 [110,131] | 1.5 × 102 [132] | 2.22–351 [133,135] | Antioxidant, protection of the integrity of cell membranes against oxidative damage; photoprotection (when combined with vitamin C) [28] |
| Vitamin K | 1.437 [110] | Not found | 0.071 [134] | Treatment of under-eye circles, anti-aging, anti-wrinkle [136] |
2.5. Minerals
| Mineral | C. lanatus | C. melo | C. pepo | Properties/Applications |
|---|---|---|---|---|
| Al | 0.0015 [100] | Not found | 0.921 [102] | Skin care products, such as creams; inflammatory skin condition ulcers, pimples, and other types of skin rash treatments; active base in face masks [30] |
| Ca | 0.10–30.8 [141,142] | 8.34–806.4 [143] | 3.76–141.00 [103,144] | Texture enhancer, bulking agent, and opacifying agent; oral care (whitening agents, bleaching agents, abrasives); skin care (sunscreens, pigments, foundations, sebum adsorbers, delivery of nutrients, cleansers); hair care (dyes and solid hair products); deodorants (antibacterials, antiperspirants, odor control) [137] |
| Cr | 0.0683 [100] | Not found | 0.284 [102] | Eyeshadow, face paint, lipstick, make-up powder, skin cream and emulsion, soap [145] |
| Cu | 0.243–2.53 [100,141] | 0.53–15.9 [143] | 0.21–89.84 [103,144] | Collagen maturation stimulation and skin elasticity improvement [123] |
| Fe | 1.5190–144.70 [100,141] | 2.69–81.17 [143] | 3.75–33.14 [144] | Colorants in skin, hair, and nail cosmetic products [146]; eyeshadow, blusher, powder, lipstick, and mineral make-up [147] |
| K | 1.8189–236.7 [100,141] | 309.1–9548.33 [142,143] | 103.12–4300.00 [144] | White clays [30] |
| Mg | 1.0637–25.0 [140] | 101.71–3299.27 [143] | 4.20–2385.00 [103,144] | White clays [30] |
| Mn | 0.0301–22.73 [100,141] | 1.25–15.20 [143] | 0.06–8.90 [144] | Lipstick, lip gloss/lip balm, face powder, eyeliner, eyeshadow/pencil, blush, mascara [145] |
| Na | 0.21–98.6 [140,141] | 41.22–386.13 [143] | 0.69–189.81 [102,144] | White clays [30] |
| Zn | 0.0905–21.05 [100,141] | 2.34–24.6 [142,143] | 1.24–14.14 [144] | UV irradiation skin protection [123]; acne and seborrheic dermatitis treatment [138] |
3. Nanoencapsulation Techniques for Seed Oils in Cosmetic Applications
Nanoencapsulation of Cucurbitaceae Seed Oils: Applications and Advancements in Cosmetics
4. Perspectives and Current Limitations in the Cosmetic Use of Cucurbitaceae Seed Oils
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Borecka, M.; Karaś, M. A comprehensive review of the nutritional and health-promoting properties of edible parts of selected Cucurbitaceae plants. Foods 2025, 14, 1200. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Singha, S.; Kar, A.; Chanda, J.; Banerjee, S.; Dasgupta, B.; Aldar, P.K.; Sharma, N. Therapeutic Importance of Cucurbitaceae: A Medicinally Important Family. J. Ethnopharmacol. 2022, 282, 114599. [Google Scholar] [CrossRef] [PubMed]
- Rolnik, A.; Olas, B. Vegetables from the Cucurbitaceae Family and Their Products: Positive Effect on Human Health. Nutrition 2020, 78, 110788. [Google Scholar] [CrossRef] [PubMed]
- Rico, X.; Gullón, B.; Alonso, J.L.; Yáñez, R. Recovery of high value-added compounds from pineapple, melon, watermelon and pumpkin processing by-products: An overview. Food Res. Int. 2020, 132, 109086. [Google Scholar] [CrossRef]
- Gómez-García, R.; Campos, D.A.; Aguilar, C.N.; Madureira, A.R.; Pintado, M. Valorization of melon fruit (Cucumis melo L.) by-products: Phytochemical and biofunctional properties with emphasis on recent trends and advances. Trends Food Sci. Technol. 2020, 99, 507–519. [Google Scholar] [CrossRef]
- Nissar, J.; Sidiqi, U.S.; Hussain, A.; Akbar, U. Nutritional composition and bioactive potential of watermelon seeds: A pathway to sustainable food and health innovation. Sustain. Food Technol. 2025, 3, 375–395. [Google Scholar] [CrossRef]
- Górnaś, P.; Rudzińska, M. Seeds recovered from industry by-products of nine fruit species with a high potential utility as a source of unconventional oil for biodiesel and cosmetic and pharmaceutical sectors. Ind. Crops Prod. 2016, 83, 329–338. [Google Scholar] [CrossRef]
- Vermaak, I.; Kamatou, G.P.P.; Komane-Mofokeng, B.; Viljoen, A.M.; Beckett, K. African seed oils of commercial importance—Cosmetic applications. S. Afr. J. Bot. 2011, 77, 920–933. [Google Scholar] [CrossRef]
- Sagbo, I.J.; Mbeng, W.O. Are plants used in the Eastern Cape province for cosmetics fully commercialized? Indian J. Pharmacol. 2019, 51, 140–149. [Google Scholar] [CrossRef]
- Cheikhyoussef, N.; Kandawa-Schulz, M.; Böck, R.; de Koning, C.; Cheikhyoussef, A.; Hussein, A.A. Characterization of Acanthosicyos horridus and Citrullus lanatus seed oils: Two melon seed oils from Namibia used in food and cosmetics applications. 3 Biotech. 2017, 7, 297. [Google Scholar] [CrossRef]
- Sripisut, T.; Teja, A.; Pengsuwan, N.; Tree-Udom, T.; Chanpirom, S. Pectin-like polysaccharide extracted from Cucumis melo pulp: Physicochemical, antioxidant, and skin hydration efficacy. Nat. Prod. Res. 2024, 1–9. [Google Scholar] [CrossRef]
- Ge, H.; Yang, L.; Yao, Q.; Gu, Y.; Zhang, C.; Liu, X.; Huang, F.; Zhang, Y. Carotenes from pumpkin flesh and peel: A review on their physicochemical properties, preparation techniques, detection methods and antioxidant potential. ACS Food Sci. Technol. 2025, 5, 898–908. [Google Scholar] [CrossRef]
- Xie, Y.; Zhu, G.; Yi, J.; Ji, Y.; Xia, Y.; Zheng, Y.; Ye, C. A new product of multi-plant extracts improved skin photoaging: An oral intake in vivo study. J. Cosmet. Dermatol. 2022, 21, 3406–3415. [Google Scholar] [CrossRef] [PubMed]
- Sasounian, R.; Martinez, R.M.; Lopes, A.M.; Giarolla, J.; Rosado, C.; Magalhães, W.V.; Velasco, M.V.R.; Baby, A.R. Innovative approaches to an eco-friendly cosmetic industry: A review of sustainable ingredients. Clean Technol. 2024, 6, 176–198. [Google Scholar] [CrossRef]
- Rocca, R.; Acerbi, F.; Fumagalli, L.; Taisch, M. Sustainability paradigm in the cosmetics industry: State of the art. Clean. Waste Syst. 2022, 3, 100057. [Google Scholar] [CrossRef]
- Wei, P.; Zhao, F.; Wang, Z.; Wang, Q.; Chai, X.; Hou, G.; Meng, Q. Sesame (Sesamum indicum L.): A comprehensive review of nutritional value, phytochemical composition, health benefits, development of food, and industrial applications. Nutrients 2022, 14, 4079. [Google Scholar] [CrossRef] [PubMed]
- Ouassor, I.; Aqil, Y.; Belmaghraoui, W.; El Hajjaji, S. Characterization of two Moroccan watermelon seeds oil varieties by three different extraction methods. OCL 2020, 27, 13. [Google Scholar] [CrossRef]
- Brah, A.S.; Obuah, C.; Adokoh, C.K. Innovations and modifications of current extraction methods and techniques of citrus essential oils: A review. Discov. Appl. Sci. 2024, 6, 460. [Google Scholar] [CrossRef]
- Ahmed, I.A.; Mikail, M.A.; Zamakshshari, N.; Abdullah, A.H. Natural anti-aging skincare: Role and potential. Biogerontology 2020, 21, 293–310. [Google Scholar] [CrossRef]
- Gad, H.A.; Roberts, A.; Hamzi, S.H.; Gad, H.A.; Touiss, I.; Altyar, A.E.; Kensara, O.A.; Ashour, M.L. Jojoba oil: An updated comprehensive review on chemistry, pharmaceutical uses, and toxicity. Polymers 2021, 13, 1711. [Google Scholar] [CrossRef]
- Chakrabarty, S.; Jigdrel, K.; Mukherjee, P.; Paul, T.; Drakpa, D.; Gupta, J. Bioactivities of jojoba oil beyond skincare. J. Med. Food 2024, 27, 579–588. [Google Scholar] [CrossRef]
- Monika, M.; Anna, K.D. Nut oils and their dietetic and cosmetic significance: A review. J. Oleo Sci. 2019, 68, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Li, Z.; Fu, M. Comparison of quality and oxidative stability of pumpkin seed (Cucurbita maxima) oil between conventional and enzymatic extraction methods. Sustain. Food Technol. 2024, 2, 1033. [Google Scholar] [CrossRef]
- Somkuwar, P.S.; Samarth, O.N.; Dahake, P.P.; Bhadange, J.A. Nature’s secret weapon: Watermelon seed oil as petroleum jelly alternative for oily skin care moisturizer. World J. Biol. Pharm. Health Sci. 2024, 20, 136–153. [Google Scholar] [CrossRef]
- Lakhe, P.; Chopde, M.; Borikar, M. Watermelon seed oil: A potential skin hydrating agent. Int. J. Res. Biol. Appl. Technol. 2022, 2, 229–233. [Google Scholar]
- Petchsomrit, A.; McDermott, M.I.; Chanroj, S.; Choksawangkarn, W. Watermelon seeds and peels: Fatty acid composition and cosmeceutical potential. OCL 2020, 27, 54. [Google Scholar] [CrossRef]
- Serra, M.; Casas, A.; Teixeira, J.A.; Barros, A.N. Revealing the Beauty Potential of Grape Stems: Harnessing Phenolic Compounds for Cosmetics. Int. J. Mol. Sci. 2023, 24, 11751. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Saliou, C. Role of vitamins in skin care. Nutrition 2001, 17, 839–844. [Google Scholar] [CrossRef]
- Diaz, I.; Namkoong, J.; Wu, J.Q.; Giancola, G. Amino acid complex (AAComplex) benefits in cosmetic products: In vitro and in vivo clinical studies. J. Cosmet. Dermatol. 2022, 21, 3046–3052. [Google Scholar] [CrossRef]
- Wargala, E.; Sławska, M.; Zalewska, A.; Toporowska, M. Health Effects of Dyes, Minerals, and Vitamins Used in Cosmetics. Women 2021, 1, 223–237. [Google Scholar] [CrossRef]
- Gupta, M.; Mahajan, V.K.; Mehta, K.S.; Chauhan, P.S. Zinc therapy in dermatology: A review. Dermatol. Res. Pract. 2014, 2014, 709152. [Google Scholar] [CrossRef]
- Jesus, A.; Ratanji, S.; Cidade, H.; Sousa, E.; Cruz, M.T.; Oliveira, R.; Almeida, I.F. Phenolics as Active Ingredients in Skincare Products: A Myth or Reality? Molecules 2025, 30, 1423. [Google Scholar] [CrossRef] [PubMed]
- Stępniowska, A.; Cieplińska, P.; Fac, W.; Górska, J. Selected Alkaloids Used in the Cosmetics Industry. J. Cosmet. Sci. 2021, 72, 229–245. [Google Scholar] [PubMed]
- Nunes, K.C.; Lazarin-Bidoia, D.; Ueda-Nakamura, T.; Lautenschlager, S.O.S.; Michel, R.; Auzély-Velty, R.; Nakamura, C.V. Syringic acid protective role: Combatting oxidative stress induced by UVB radiation in L-929 fibroblasts. J. Photochem. Photobiol. B Biol. 2025, 264, 113104. [Google Scholar] [CrossRef] [PubMed]
- Girsang, E.; Ginting, C.N.; Ehrich Lister, I.N.; Widowati, W. Anti-inflammatory and antiaging properties of chlorogenic acid on UV-induced fibroblast cell. PeerJ 2021, 9, e11419. [Google Scholar] [CrossRef] [PubMed]
- Przybylska-Balcerek, A.; Stuper-Szablewska, K. Phenolic acids used in the cosmetics industry as natural antioxidants. Eur. J. Med. Technol. 2019, 4, 24–32. [Google Scholar]
- Boo, Y.C. p-Coumaric Acid as An Active Ingredient in Cosmetics: A Review Focusing on its Antimelanogenic Effects. Antioxidants 2019, 8, 275. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-L.; Lin, C.-P.; Gowrisankar, Y.V.; Huang, P.-J.; Chang, W.-L.; Shrestha, S.; Hseu, Y.-C. The anti-melanogenic effects of ellagic acid through induction of autophagy in melanocytes and suppression of UVA-activated α-MSH pathways via Nrf2 activation in keratinocytes. Biochem. Pharmacol. 2021, 185, 114454. [Google Scholar] [CrossRef]
- Andrabi, N.I.; Kumar, D.; Sawhney, G. Flavonoids: A Key Component of Cosmeceuticals. In Specialized Plant Metabolites as Cosmeceuticals; Kathuria, D., Sharma, A., Simal-Gandara, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2024; pp. 43–63. [Google Scholar] [CrossRef]
- Yoon, J.H.; Kim, Y.; Cho, J.Y. Apigenin: A Therapeutic Agent for Treatment of Skin Inflammatory Diseases and Cancer. Int. J. Mol. Sci. 2023, 24, 1498. [Google Scholar] [CrossRef]
- Čižmárová, B.; Hubková, B.; Tomečková, V.; Birková, A. Flavonoids as Promising Natural Compounds in the Prevention and Treatment of Selected Skin Diseases. Int. J. Mol. Sci. 2023, 24, 6324. [Google Scholar] [CrossRef]
- Seidu, K.T.; Otutu, O.L. Phytochemical Composition and Radical Scavenging Activities of Watermelon (Citrullus lanatus) Seed Constituents. Croat. J. Food Sci. Technol. 2016, 8, 83–89. [Google Scholar] [CrossRef]
- Saleh, A.S.; El-Newary, S.A.; Mohamed, W.A.; Elgamal, A.M.; Farah, M.A. Pumpkin Seeds (Cucurbita pepo subsp. ovifera) Decoction Promotes Trichinella spiralis Expulsion During Intestinal Phase via “Weep and Sweep” Mechanism. Sci. Rep. 2024, 14, 1548. [Google Scholar] [CrossRef] [PubMed]
- Jolly, A.; Kim, H.; Moon, J.-Y.; Mohan, A.; Lee, Y.-C. Exploring the imminent trends of saponins in personal care product development: A review. Ind. Crops Prod. 2023, 205, 117489. [Google Scholar] [CrossRef]
- Mallek-Ayadi, S.; Bahloul, N.; Kechaou, N. Phytochemical Profile, Nutraceutical Potential and Functional Properties of Cucumis melo L. Seeds. J. Sci. Food Agric. 2019, 99, 1294–1301. [Google Scholar] [CrossRef]
- Na, E.J.; Ryu, J.Y. Anti-Inflammatory Effects of Prunin on UVB-Irradiated Human Keratinocytes. Biomed. Dermatol. 2018, 2, 14. [Google Scholar] [CrossRef]
- Jimoh, T.O.; Ademiluyi, A.O.; Oboh, G.; Boligon, A.A. Phenolic Extracts and Amino Acids Content from Cucumeropsis mannii Naudin and Citrullus lanatus Inhibit Relevant Enzymes of Erectile Dysfunction in Rat’s Penile Tissue. Biochem. Biophys. Rep. 2017, 12, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Andjelkovic, M.; Camp, J.V.; Trawka, A.; Verhé, R. Phenolic Compounds and Some Quality Parameters of Pumpkin Seed Oil. Eur. J. Lipid Sci. Technol. 2010, 112, 208–217. [Google Scholar] [CrossRef]
- Jaroszewska, A.; Jedrejek, D.; Sobolewska, M.; Kowalska, I.; Dzięcioł, M. Mineral, Nutritional, and Phytochemical Composition and Baking Properties of Teff and Watermelon Seed Flours. Molecules 2023, 28, 3255. [Google Scholar] [CrossRef]
- Wahid, M.; Saqib, F. Scientific Basis for Medicinal Use of Citrullus lanatus (Thunb.) in Diarrhea and Asthma: In Vitro, In Vivo and In Silico Studies. Phytomedicine 2022, 98, 153978. [Google Scholar] [CrossRef]
- Lee, H.; Kim, S.-Y.; Lee, S.W.; Kwak, S.; Li, H.; Piao, R.; Park, H.-Y.; Choi, S.; Jeong, T.-S. Amentoflavone-Enriched Selaginella rossii Protects against Ultraviolet- and Oxidative-Stress-Induced Aging in Skin Cells. Life 2022, 12, 2106. [Google Scholar] [CrossRef]
- Bonesi, M.; Loizzo, M.R.; Menichini, F.; Tundis, R. Flavonoids in Treating Psoriasis. In Immunity and Inflammation in Health and Disease; Chatterjee, S., Jungraithmayr, W., Bagchi, D., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 281–294. [Google Scholar] [CrossRef]
- Rodrigues, R.; Alves, R.C.; Oliveira, M.B. Exploring Olive Pomace for Skincare Applications: A Review. Cosmetics 2023, 10, 35. [Google Scholar] [CrossRef]
- Cádiz-Gurrea, M.d.l.L.; Pinto, D.; Delerue-Matos, C.; Rodrigues, F. Olive Fruit and Leaf Wastes as Bioactive Ingredients for Cosmetics—A Preliminary Study. Antioxidants 2021, 10, 245. [Google Scholar] [CrossRef] [PubMed]
- Ratz-Łyko, A.; Arct, J. Resveratrol as an Active Ingredient for Cosmetic and Dermatological Applications: A Review. J. Cosmet. Laser Ther. 2019, 21, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ni, C.; Huang, Y.; Tang, Y.; Yang, K.; Shi, X.; Zhang, Y.; Li, Z.; Wang, J.; Zhu, Y.; et al. Hair Growth-Promoting Effect of Resveratrol in Mice, Human Hair Follicles and Dermal Papilla Cells. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1805–1814. [Google Scholar] [CrossRef]
- Zeb, A. Phenolic Profile and Antioxidant Activity of Melon (Cucumis melo L.) Seeds from Pakistan. Foods 2016, 5, 67. [Google Scholar] [CrossRef] [PubMed]
- Peng, M.; Lu, D.; Liu, J.; Jiang, B.; Chen, J. Effect of Roasting on the Antioxidant Activity, Phenolic Composition, and Nutritional Quality of Pumpkin (Cucurbita pepo L.) Seeds. Front. Nutr. 2021, 8, 647354. [Google Scholar] [CrossRef]
- Ariede, M.B.; Gomez Junior, W.A.; Cândido, T.M.; de Aguiar, M.M.G.B.; Rosado, C.; Rangel-Yagui, C.d.O.; Pessoa, F.V.L.S.; Velasco, M.V.R.; Baby, A.R. Would Rutin Be a Feasible Strategy for Environmental-Friendly Photoprotective Samples? A Review from Stability to Skin Permeability and Efficacy in Sunscreen Systems. Cosmetics 2024, 11, 141. [Google Scholar] [CrossRef]
- Zhang, G.; Li, Z.; Charalampopoulos, D. Upcycling of Melon Seed (Cucumis melo L.) Oil Processing By-Product: Evaluation of Functional Properties and Nutritional Components as Novel Ingredient. Chem. Biol. Technol. Agric. 2024, 11, 101. [Google Scholar] [CrossRef]
- Rolim, P.M.; Fidelis, G.P.; Padilha, C.E.A.; Santos, E.S.; Rocha, H.A.O.; Macedo, G.R. Phenolic Profile and Antioxidant Activity from Peels and Seeds of Melon (Cucumis melo L. var. reticulatus) and Their Antiproliferative Effect in Cancer Cells. Braz. J. Med. Biol. Res. 2018, 51, e6069. [Google Scholar] [CrossRef]
- Mita, S.R.; Husni, P.; Putriana, N.A.; Maharani, R.; Hendrawan, R.P.; Dewi, D.A. A Recent Update on the Potential Use of Catechins in Cosmeceuticals. Cosmetics 2024, 11, 23. [Google Scholar] [CrossRef]
- Macedo, C.; Silva, A.M.; Ferreira, A.S.; Moreira, M.M.; Delerue-Matos, C.; Rodrigues, F. Microwave- and Ultrasound-Assisted Extraction of Cucurbita pepo Seeds: A Comparison Study of Antioxidant Activity, Phenolic Profile, and In-Vitro Cells Effects. Appl. Sci. 2022, 12, 1763. [Google Scholar] [CrossRef]
- Khan, B.A.; Mahmood, T.; Menaa, F.; Shahzad, Y.; Yousaf, A.M.; Hussain, T.; Ray, S.D. New Perspectives on the Efficacy of Gallic Acid in Cosmetics & Nanocosmeceuticals. Curr. Pharm. Des. 2018, 24, 5181–5187. [Google Scholar] [CrossRef]
- Saini, U.; Sharma, A.; Mittal, V. Gallic Acid: A Potent Antioxidant and Anti-Inflammatory Agent in Modern Cosmeceuticals. Recent Adv. Drug Deliv. Formul. 2025; in press. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Cho, S.H.; Park, D.; Jung, E. Anti-Skin Aging Properties of Protocatechuic Acid In Vitro and In Vivo. J. Cosmet. Dermatol. 2020, 19, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Son, J.H.; Kim, S.Y.; Jang, H.H.; Lee, J.E.; Moon, D.I.; Kim, B.K.; Oh, C.H.; Yeo, J.H. Protective Effect of Protocatechuic Acid Against Inflammatory Stress Induced in Human Dermal Fibroblasts. Biomed. Dermatol. 2018, 2, 9. [Google Scholar] [CrossRef]
- Shahein, M.R.; Atwaa, E.S.; Magawry, K.; Elmaadawy, A.A.; Hijazy, H.H.; Sitohy, M.Z.; Albrakati, A.; Elmahallawy, E.K. Remedial Action of Yoghurt Enriched with Watermelon Seed Milk on Renal-Injured Hyperuricemic Rats. Fermentation 2022, 8, 41. [Google Scholar] [CrossRef]
- Peričin, D.; Krimer, V.; Trivić, S.; Radulović, L. The Distribution of Phenolic Acids in Pumpkin’s Hull-less Seed, Skin, Oil Cake Meal, Dehulled Kernel and Hull. Food Chem. 2009, 113, 450–456. [Google Scholar] [CrossRef]
- García-García, R.; Searle, S.S. Preservatives: Food Use. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 505–509. [Google Scholar] [CrossRef]
- Girawale, S.D.; Meena, S.N.; Nandre, V.S.; Waghmode, S.B.; Kodam, K.M. Biosynthesis of vanillic acid by Ochrobactrum anthropi and its applications. Bioorg. Med. Chem. 2022, 72, 117000. [Google Scholar] [CrossRef]
- Howes, M.-J.R. Phytochemicals as Anti-Inflammatory Nutraceuticals and Phytopharmaceuticals. In Immunity and Inflammation in Health and Disease; Chatterjee, S., Jungraithmayr, W., Bagchi, D., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 363–388. [Google Scholar] [CrossRef]
- Fehér, J.; Červeňanský, I.; Václavík, L.; Markoš, J. Electrodialysis applied for phenylacetic acid separation from organic impurities: Increasing the recovery. Sep. Purif. Technol. 2020, 235, 116222. [Google Scholar] [CrossRef]
- Markert, T.; Moussou, P.; Danoux, L.; Rathjens, A. Use of Sinapic Acid in Non-Therapeutic Cosmetic and/or Topical Compositions. European Patent EP2117499B1, 26 February 2008. Available online: https://patentimages.storage.googleapis.com/ea/3a/cd/4e634638f0ff7b/EP2117499B1.pdf (accessed on 9 May 2025).
- Jokubaite, M.; Ramanauskiene, K. Potential Unlocking of Biological Activity of Caffeic Acid by Incorporation into Hydrophilic Gels. Gels 2024, 10, 794. [Google Scholar] [CrossRef] [PubMed]
- Bastianini, M.; Faffa, C.; Sisani, M.; Petracci, A. Caffeic Acid-layered Double Hydroxide Hybrid: A New Raw Material for Cosmetic Applications. Cosmetics 2018, 5, 51. [Google Scholar] [CrossRef]
- Silva, M.A.; Albuquerque, T.G.; Alves, R.C.; Oliveira, M.B.P.P.; Costa, H.S. Cucumis melo L. seed oil components and biological activities. In Multiple Biological Activities of Unconventional Seed Oils; Mariod, A.A., Ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 125–138. [Google Scholar] [CrossRef]
- Kelm, G.R.; Wickett, R.R. Chapter 12—The Role of Fatty Acids in Cosmetic Technology. In Fatty Acids; Ahmad, M.U., Ed.; Academic Press: Cambridge, MA, USA; AOCS Press: Urbana, IL, USA, 2017; pp. 385–404. ISBN 9780128095218. [Google Scholar] [CrossRef]
- Fiume, M.M.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G., Jr.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; et al. Safety Assessment of Tocopherols and Tocotrienols as Used in Cosmetics. Int. J. Toxicol. 2018, 37 (Suppl. S2), 61S–94S. [Google Scholar] [CrossRef]
- Silva, S.; Ferreira, M.; Oliveira, A.S.; Magalhães, C.; Sousa, M.E.; Pinto, M.; Sousa Lobo, J.M.; Almeida, I.F. Evolution of the use of antioxidants in anti-ageing cosmetics. Int. J. Cosmet. Sci. 2019, 41, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, P.; Cabral, J.M. Phytosterols: Applications and recovery methods. Bioresour. Technol. 2007, 98, 2335–2350. [Google Scholar] [CrossRef] [PubMed]
- Grether-Beck, S.; Mühlberg, K.; Brenden, H.; Krutmann, J. Topische Applikation von Vitaminen, Phytosterolen und Ceramiden. Hautarzt 2008, 59, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Haiyuan, Y.U.; Shen, X.; Liu, D.; Hong, M.; Lu, Y. The protective effects of β-sitosterol and vermicularin from Thamnolia vermicularis (Sw.) Ach. against skin aging in vitro. An. Acad. Bras. Cienc. 2019, 91, e20181088. [Google Scholar] [CrossRef]
- Wang, X.; Jia, Y.; He, H. The Role of Linoleic Acid in Skin and Hair Health: A Review. Int. J. Mol. Sci. 2024, 26, 246. [Google Scholar] [CrossRef]
- Notman, R.; Anwar, J. Breaching the skin barrier—Insights from molecular simulation of model membranes. Adv. Drug Deliv. Rev. 2013, 65, 237–250. [Google Scholar] [CrossRef] [PubMed]
- Atef, B.R.; Ishak, R.A.H.; Badawy, S.S.; Osman, R. Exploring the potential of oleic acid in nanotechnology-mediated dermal drug delivery: An up-to-date review. J. Drug Deliv. Sci. Technol. 2022, 67, 103032. [Google Scholar] [CrossRef]
- Rabadán, A.; Nunes, M.A.; Bessada, S.M.; Pardo, J.E.; Oliveira, M.B. From By-Product to the Food Chain: Melon (Cucumis melo L.) Seeds as Potential Source for Oils. Foods 2020, 9, 1341. [Google Scholar] [CrossRef]
- Schoss, K.; Glavač, N.K. Supercritical CO2 Extraction vs. Hexane Extraction and Cold Pressing: Comparative Analysis of Seed Oils from Six Plant Species. Plants 2024, 13, 3409. [Google Scholar] [CrossRef]
- Yarova, G.; Lathrop, W.; Nip, J.; Bajor, J.; Hawkins, S.S.; Dasgupta, B.R.; Hermanson, K.D.; Mayes, A. Topically applied skin natural fatty acids and 12-hydroxystearic acid boosts barrier lipids. J. Am. Acad. Dermatol. 2022, 87 (Suppl. S3), AB214. [Google Scholar] [CrossRef]
- Shashkina, V.G.; Neimane, G.Y.; Belkevich, P.I.; Kaganovich, F.L.; Dolidovich, E.F.; Sheremet, L.S.; Yachnik, V.J. Cleansing Agents Containing Oleic Acid, Isopropanol and Ethylacetate. U.S. Patent 4,212,758, 15 July 1980. Available online: https://patentimages.storage.googleapis.com/03/59/5a/26d1b06b62dc4f/US4212758.pdf (accessed on 9 May 2025).
- Broznić, D.; Jurešić, G.Č.; Milin, Č. Involvement of α-, γ- and δ-Tocopherol Isomers from Pumpkin (Cucurbita pepo L.) Seed Oil or Oil Mixtures in the Biphasic DPPH Disappearance Kinetics. Food Technol. Biotechnol. 2016, 54, 200–210. [Google Scholar] [CrossRef] [PubMed]
- François, G.; Nathalie, B.; Jean-Pierre, V.; Daniel, P.; Didier, M. Effect of roasting on tocopherols of gourd seeds (Cucurbita pepo). Grasas Aceites 2006, 57, 409–414. Available online: https://grasasyaceites.revistas.csic.es/index.php/grasasyaceites/article/view/67 (accessed on 14 May 2025). [CrossRef]
- Zhang, G.; Li, Z.; Liu, L.; Xiang, Q. Road to valorisation of melon seeds (Cucumis melo L.): A comprehensive review of nutritional profiles, biological activities, and food applications. Sustain. Food Technol. 2024, 2, 1166. [Google Scholar] [CrossRef]
- Fernandes, T.; Cordeiro, N. Microalgae as Sustainable Biofactories to Produce High-Value Lipids: Biodiversity, Exploitation, and Biotechnological Applications. Mar. Drugs 2021, 19, 573. [Google Scholar] [CrossRef] [PubMed]
- Burnett, C.L.; Heldreth, B.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G., Jr.; Shank, R.C.; Slaga, T.J.; et al. Safety Assessment of α-Amino Acids as Used in Cosmetics. Int. J. Toxicol. 2013, 32 (Suppl. S6), 41S–64S. [Google Scholar] [CrossRef] [PubMed]
- Pralong, P.; Dendooven, E.; Aerts, O. Sodium Stearoyl Glutamate: Another Amino Acid Alkyl Amide Sensitizer in Cosmetics. Contact Dermat. 2022, 87, 453–454. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Additives and Products or Substances Used in Animal Feed (FEEDAP). Scientific Opinion on the Safety and Efficacy of L-Threonine Produced by Escherichia coli for All Animal Species Based on a Dossier Submitted by HELM AG on Behalf of Meihua Holdings Group Co. Ltd. EFSA J. 2015, 13, 4051. [Google Scholar] [CrossRef]
- Williams, J.Z.; Abumrad, N.; Barbul, A. Effect of a Specialized Amino Acid Mixture on Human Collagen Deposition. Ann. Surg. 2002, 236, 369–374. [Google Scholar] [CrossRef]
- Benmeziane, F.; Derradji. Composition, Bioactive Potential and Food Applications of Watermelon (Citrullus lanatus) Seeds—A Review. J. Food Meas. Charact. 2023, 17, 5045–5061. [Google Scholar] [CrossRef]
- Enemor, V.H.A.; Oguazu, C.E.; Odiakosa, A.U.; Okafor, S.C. Evaluation of the Medicinal Properties and Possible Nutrient Composition of Citrullus lanatus (Watermelon) Seed. Res. J. Med. Plants 2019, 13, 129–135. [Google Scholar] [CrossRef]
- de Mello, M.L.S.; Bora, P.S.; Narain, N. Fatty and Amino Acids Composition of Melon (Cucumis melo var. saccharinus) Seeds. J. Food Compos. Anal. 2001, 14, 69–74. [Google Scholar] [CrossRef]
- Glew, R.H.; Glew, R.S.; Chuang, L.T.; Huang, Y.S.; Millson, M.; Constans, D.; VanderJagt, D.J. Amino Acid, Mineral and Fatty Acid Content of Pumpkin Seeds (Cucurbita spp.) and Cyperus esculentus Nuts in the Republic of Niger. Plant Foods Hum. Nutr. 2006, 61, 51–56. [Google Scholar] [CrossRef]
- Habib, M.; Singh, S.; Sagar, N.A.; Ahmad, S.; Qureshi, I.; Jan, S.; Jana, K.; Bashir, K. Physicochemical and Functional Characterization of Pumpkin Seed Protein Isolate. Sustain. Food Technol. 2025, 3, 445. [Google Scholar] [CrossRef]
- Ozuna, C.; León-Galván, M.F. Cucurbitaceae seed protein hydrolysates as a potential source of bioactive peptides with functional properties. Biomed. Res. Int. 2017, 2017, 2121878. [Google Scholar] [CrossRef]
- Oyeleke, A.; Oluwajuyitan, T.; Oluwamukomi, M.; Enujiugha, V. Amino acid profile, functional properties and in-vitro antioxidant capacity of Cucurbita maxima and Cucurbita mixta fruit pulps and seeds. Eur. J. Nutr. Food Saf. 2019, 10, 224–241. [Google Scholar] [CrossRef]
- Gavril, R.N.; Stoica, F.; Lipșa, F.D.; Constantin, O.E.; Stănciuc, N.; Aprodu, I.; Râpeanu, G. Pumpkin and pumpkin by-products: A comprehensive overview of phytochemicals, extraction, health benefits, and food applications. Foods 2024, 13, 2694. [Google Scholar] [CrossRef] [PubMed]
- McKay, T.B.; Priyadarsini, S.; Rowsey, T.; Karamichos, D. Arginine Supplementation Promotes Extracellular Matrix and Metabolic Changes in Keratoconus. Cells 2021, 10, 2076. [Google Scholar] [CrossRef] [PubMed]
- Jerônimo, M.S.; Barros, A.P.; Morita, V.E.Z.; Alves, E.O.; de Souza, N.L.B.; de Almeida, R.M.; Nóbrega, Y.K.M.; Cavalcanti Neto, F.F.; Amorin, R.; Borin, M.F.; et al. Oral or Topical Administration of L-Arginine Changes the Expression of TGF and iNOS and Results in Early Wound Healing. Acta Cir. Bras. 2016, 31, 586–596. [Google Scholar] [CrossRef]
- Kwon, K.C.; Won, J.G.; Seo, J.H.; Kwon, O.S.; Kim, E.H.; Kim, M.S.; Park, S.W. Effects of Arginine Glutamate (RE:pair) on Wound Healing and Skin Elasticity Improvement after CO2 Laser Irradiation. J. Cosmet. Dermatol. 2022, 21, 5037–5048. [Google Scholar] [CrossRef] [PubMed]
- Kay, E.J.; Koulouras, G.; Zanivan, S. Regulation of Extracellular Matrix Production in Activated Fibroblasts: Roles of Amino Acid Metabolism in Collagen Synthesis. Front. Oncol. 2021, 11, 719922. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Ma, Q.; Zhu, H. Distribution, Industrial Applications, and Enzymatic Synthesis of D-Amino Acids. Appl. Microbiol. Biotechnol. 2015, 99, 3341–3349. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, E.; Schofield, S.R.; Meredith, P.; Gummer, C.L. Hair Care Compositions Comprising One Tryptophan Compound and Another Amino Acid Compound. WO 2000/051555 A1, 8 September 2000. Available online: https://patents.google.com/patent/WO2000051555A1/en (accessed on 11 June 2025).
- Tuccori, S.R. Use of Amino Acids in Hair Treatment. U.S. Patent 2001/0051141A1, 13 December 2001. Available online: https://patents.google.com/patent/US20010051141A1 (accessed on 11 June 2025).
- Gowda, D.; Premalatha, V.; Imtiyaz, D.B. Prevalence of Nutritional Deficiencies in Hair Loss among Indian Participants: Results of a Cross-Sectional Study. Int. J. Trichol. 2017, 9, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Oshimura, E.; Abe, H.; Oota, R. Hair and Amino Acids: The Interactions and the Effects. J. Cosmet. Sci. 2007, 58, 347–357. [Google Scholar]
- Karna, E.; Szoka, L.; Huynh, T.Y.L.; Palka, J.A. Proline-Dependent Regulation of Collagen Metabolism. Cell Mol. Life Sci. 2020, 77, 1911–1918. [Google Scholar] [CrossRef]
- da Silva, C.S.M.; Costa, G.A.N.; Aguiar, A.F.; Camargo, M.Z.; Fernandes, K.B.P.; Oliveira, M.R.; da Silva, R.A. Effect of the Use of a Cream with Leucine and Lactic Acid Associated with Electrostimulation in Contouring and Facial Tonus: A Randomized Clinical Controlled Trial. Cosmetics 2022, 9, 36. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information (NCBI). PubChem Compound Summary for CID 6137, Methionine; PubChem Database: Bethesda, MD, USA, 2025. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Methionine (accessed on 17 April 2025).
- Nordlund, J.J. The Medical Treatment of Vitiligo: An Historical Review. Dermatol. Clin. 2017, 35, 107–116. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information (NCBI). PubChem Compound Summary for CID 6288, Threonine; PubChem Database: Bethesda, MD, USA, 2025. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Threonine (accessed on 7 June 2025).
- Park, J.; Jung, H.; Jang, B.; Song, H.K.; Han, I.O.; Oh, E.S. D-Tyrosine Adds an Anti-Melanogenic Effect to Cosmetic Peptides. Sci. Rep. 2020, 10, 262. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information (NCBI). PubChem Compound Summary for CID 6287, Valine; PubChem Database: Bethesda, MD, USA, 2025. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Valine (accessed on 17 April 2025).
- Bjørklund, G.; Shanaida, M.; Lysiuk, R.; Butnariu, M.; Peana, M.; Sarac, I.; Strus, O.; Smetanina, K.; Chirumbolo, S. Natural Compounds and Products from an Anti-Aging Perspective. Molecules 2022, 27, 7084. [Google Scholar] [CrossRef] [PubMed]
- Temova Rakuša, Ž.; Šenk, A.; Roškar, R. Content and Stability of B-Complex Vitamins in Commercial Cosmetic Products. J. Cosmet. Dermatol. 2023, 22, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Bocheva, G.; Slominski, R.M.; Slominski, A.T. The Impact of Vitamin D on Skin Aging. Int. J. Mol. Sci. 2021, 22, 9097. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, S.N.; Anastasiou, E.A.; Adamantidi, T.; Ofrydopoulou, A.; Letsiou, S.; Tsoupras, A. A Comprehensive Review on the Beneficial Roles of Vitamin D in Skin Health as a Bio-Functional Ingredient in Nutricosmetic, Cosmeceutical, and Cosmetic Applications. Appl. Sci. 2025, 15, 796. [Google Scholar] [CrossRef]
- Rezanejad Gatabi, Z.; Rahimnia, S.M.; Morteza-Semnani, K.; Yazdian-Robati, R.; Hashemi, S.M.H.; Saeedi, M. Vitamin K (Menadione)-Incorporated Chitosan/Alginate Hydrogel as a Novel Product for Periorbital Hyperpigmentation. J. Biomater. Sci. Polym. Ed. 2024, 35, 967–988. [Google Scholar] [CrossRef]
- Cohen, J.L.; Bhatia, A.C. The Role of Topical Vitamin K Oxide Gel in the Resolution of Post-Procedural Purpura. J. Drugs Dermatol. 2009, 8, 1020–1024. [Google Scholar]
- Pazyar, N.; Houshmand, G.; Yaghoobi, R.; Hemmati, A.A.; Zeineli, Z.; Ghorbanzadeh, B. Wound-Healing Effects of Topical Vitamin K: A Randomized Controlled Trial. Indian J. Pharmacol. 2019, 51, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Polyzos, N.; Fernandes, Â.; Calhelha, R.C.; Petrović, J.; Soković, M.; Ferreira, I.C.F.R.; Barros, L.; Petropoulos, S.A. Biochemical Composition of Pumpkin Seeds and Seed By-Products. Plants 2024, 13, 2395. [Google Scholar] [CrossRef] [PubMed]
- Egbuonu, A.C.C. Comparative Assessment of Some Mineral, Amino Acid and Vitamin Compositions of Watermelon (Citrullus lanatus) Rind and Seed. Asian J. Biochem. 2015, 10, 230–236. [Google Scholar] [CrossRef]
- Opara, I.J.; Ushie, O.A.; Aondoyima, I.; Onudibia, M.E. Phytochemical Screening, Proximate and Vitamin Composition of Cucumis melo Seeds (Sweet Melon). Int. J. Res. Inf. Sci. Appl. Tech. 2019, 3, 22–28. [Google Scholar] [CrossRef]
- Niazi, M.K.; Hassan, F.; Hassan Zaidi, S.Z.ul.H.; Sahi, A.A.; Ashfaq, J.; Ejaz, F.; Aamir, Z.; Imran, S. The Nutritional and Potential Health Effect of Pumpkin seeds: Health Effect of Pumpkin seeds. PBMJ 2022, 5, 17–21. [Google Scholar] [CrossRef]
- Batool, M.; Ranjha, M.M.A.N.; Roobab, U.; Manzoor, M.F.; Farooq, U.; Nadeem, H.R.; Nadeem, M.; Kanwal, R.; AbdElgawad, H.; Al Jaouni, S.K.; et al. Nutritional Value, Phytochemical Potential, and Therapeutic Benefits of Pumpkin (Cucurbita sp.). Plants 2022, 11, 1394. [Google Scholar] [CrossRef]
- Razis, A.F.A.; Shehzad, M.M.; Usman, S.; Ali, N.B.; Iqbal, S.Z.; Naheed, N.; Asi, M.R. Seasonal Variation in Aflatoxin Levels in Edible Seeds, Estimation of Its Dietary Intake and Vitamin E Levels in Southern Areas of Punjab, Pakistan. Int. J. Environ. Res. Public Health 2020, 17, 8964. [Google Scholar] [CrossRef]
- Ahmadraji, F.; Shatalebi, M.A. Evaluation of the Clinical Efficacy and Safety of an Eye Counter Pad Containing Caffeine and Vitamin K in Emulsified Emu Oil Base. Adv. Biomed. Res. 2015, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Carella, F.; Degli Esposti, L.; Adamiano, A.; Iafisco, M. The Use of Calcium Phosphates in Cosmetics: State of the Art and Future Perspectives. Materials 2021, 14, 6398. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, J.; Eber, A.E.; Perper, M.; Nascimento, V.M.; Nouri, K.; Keri, J.E. The Role of Zinc in the Treatment of Acne: A Review of the Literature. Dermatol. Ther. 2018, 31, e12576. [Google Scholar] [CrossRef] [PubMed]
- Borowska, S.; Brzóska, M.M. Metals in Cosmetics: Implications for Human Health. J. Appl. Toxicol. 2015, 35, 551–572. [Google Scholar] [CrossRef] [PubMed]
- Bersntein, E.F.; Sarkas, H.W.; Boland, P. Iron oxides in novel skin care formulations attenuate blue light for enhanced protection against skin damage. J. Cosmet. Dermatol. 2021, 20, 532–537. [Google Scholar] [CrossRef]
- Jacob, A.G.; Etong, D.I.; Tijjani, A. Proximate, Mineral and Anti-nutritional Compositions of Melon (Citrullus lanatus) Seeds. BJR 2015, 2, 142–151. [Google Scholar]
- Saeed, F.; Afzaal, M.; Niaz, B.; Hussain, M.; Rasheed, A.; Raza, M.A.; Umar, M.; Khan, M.A.; Suleria, H.; Tufail, T.; et al. Comparative study of nutritional composition, antioxidant activity and functional properties of Cucumis melo and Citrullus lanatus seeds powder. Cogent Food Agric. 2024, 10, 2293517. [Google Scholar] [CrossRef]
- Silva, M.A.; Albuquerque, T.G.; Alves, R.C.; Oliveira, M.B.P.P.; Costa, H.S. Melon (Cucumis melo L.) By-Products: Potential Food Ingredients for Novel Functional Foods? Trends Food Sci. Technol. 2020, 98, 181–189. [Google Scholar] [CrossRef]
- Singh, A.; Kumar, V. Pumpkin Seeds as Nutraceutical and Functional Food Ingredient for Future: A Review. Grain Oil Sci. Technol. 2024, 7, 12–29. [Google Scholar] [CrossRef]
- Iwegbue, C.M.A.; Bassey, F.I.; Obi, G.; Tesi, G.O.; Martincigh, B.S. Concentrations and exposure risks of some metals in facial cosmetics in Nigeria. Toxicol. Rep. 2016, 3, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Lansdown, A.B. Iron: A cosmetic constituent but an essential nutrient for healthy skin. Int. J. Cosmet. Sci. 2001, 23, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Teklay, M.; Thole, J.T.; Ndumbu, N.; Vries, J.; Mezger, K. Mineralogical and chemical characterization of ochres used by the Himba and Nama people of Namibia. J. Archaeol. Sci. Rep. 2023, 47, 103690. [Google Scholar] [CrossRef]
- Javed, S.; Mangla, B.; Salawi, A.; Sultan, M.H.; Almoshari, Y.; Ahsan, W. Essential oils as dermocosmetic agents, their mechanism of action and nanolipidic formulations for maximized skincare. Cosmetics 2024, 11, 210. [Google Scholar] [CrossRef]
- Yukuyama, M.N.; Ghisleni, D.D.M.; Pinto, T.J.A.; Bou-Chacra, N.A. Nanoemulsion: Process selection and application in cosmetics—A review. Int. J. Cosmet. Sci. 2016, 38, 13–24. [Google Scholar] [CrossRef]
- Ashaolu, T.J. Nanoemulsions for health, food, and cosmetics: A review. Environ. Chem. Lett. 2021, 19, 3381–3395. [Google Scholar] [CrossRef]
- Kowalczyk, T.; Merecz-Sadowska, A.; Ghorbanpour, M.; Szemraj, J.; Piekarski, J.; Bijak, M.; Śliwiński, T.; Zajdel, R.; Sitarek, P. Enhanced natural strength: Lamiaceae essential oils and nanotechnology in in vitro and in vivo medical research. Int. J. Mol. Sci. 2023, 24, 15279. [Google Scholar] [CrossRef]
- Joye, I.J.; McClements, D.J. Production of nanoparticles by anti-solvent precipitation for use in food systems. Trends Food Sci. Technol. 2013, 34, 109–123. [Google Scholar] [CrossRef]
- Tiwari, N.; Osorio-Blanco, E.R.; Sonzogni, A.; Esporrín-Ubieto, D.; Wang, H.; Calderón, M. Nanocarriers for skin applications: Where do we stand? Angew. Chem. Int. Ed. 2022, 61, e202107960. [Google Scholar] [CrossRef]
- Pardeike, J.; Hommoss, A.; Müller, R.H. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int. J. Pharm. 2009, 366, 170–184. [Google Scholar] [CrossRef] [PubMed]
- Solans, C.; Solé, I. Nano-emulsions: Formation by low-energy methods. Curr. Opin. Colloid Interface Sci. 2012, 17, 246–254. [Google Scholar] [CrossRef]
- Lozada, M.I.O.; Rodrigues Maldonade, I.; Bobrowski Rodrigues, D.; Silva Santos, D.; Ortega Sanchez, B.A.; Narcizo de Souza, P.E.; Longo, J.P.; Bernardo Amaro, G.; de Lacerda de Oliveira, L. Physicochemical characterization and nano-emulsification of three species of pumpkin seed oils with focus on their physical stability. Food Chem. 2021, 343, 128512. [Google Scholar] [CrossRef] [PubMed]
- Infante, V.H.; Campos, P.M.M.; Darvin, M.; Lohan, S.; Schleusener, J.; Schanzer, S.; Lademann, J.; Meinke, M. Cosmetic formulations with Melaleuca alternifolia essential oil for the improvement of photoaged skin: A double-blind, randomized, placebo-controlled clinical study. Photochem. Photobiol. 2023, 99, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Ashagrie, Y.N.; Tadesse, M.G.; Bachheti, R.K.; Nijhawan, G.; Tyagi, S.; Bachheti, A. Formulation and characterization of Caesalpinia decapetala seed oil nanoemulsion: Physicochemical properties, stability, and antibacterial activity. Sci. Rep. 2025, 15, 14598. [Google Scholar] [CrossRef] [PubMed]
- Baykara, T.; Taylan, G. Coaxial electrospinning of PVA/Nigella seed oil nanofibers: Processing and morphological characterization. Mater. Sci. Eng. B 2021, 265, 115012. [Google Scholar] [CrossRef]
- Rezk, M.Y.; Ibrahim, S.; Khalil, E.A.; Saba, D.; Abdellatif, M.; Abdellatif, A.; Allam, N.K. Pumpkin seed oil-loaded chitosan/polyvinyl alcohol electrospun nanofiber scaffold for dermal and oral wound dressing. Chem. Select 2023, 8, e202300722. [Google Scholar] [CrossRef]
- Lin, M.; Ji, Q.; Jin, Z.; Guo, T.; Yu, K.; Ding, W.; Liu, C.; Wu, Y.; Zhang, N. Nano-microencapsulation of tea seed oil via modified complex coacervation with propolis and phosphatidylcholine for improving antioxidant activity. LWT 2022, 163, 113550. [Google Scholar] [CrossRef]
- Viana, A.R.; Martins, E.S.R.; da Silva, S.C.; Levy, B.S.; Wagner, R.; Klein, B.; Krause, L.M.F.; de Souza, D.; Boeck, C.R. Nanocapsules prepared by nanoprecipitation protect chia seed oil (Salvia hispanica L.) against thermal degradation. ACS Food Sci. Technol. 2024, 4, 2824–2834. [Google Scholar] [CrossRef]
- Menin, A.; Zanoni, F.; Vakarelova, M.; Chignola, R.; Donà, G.; Rizzi, C.; Mainente, F.; Zoccatelli, G. Effects of microencapsulation by ionic gelation on the oxidative stability of flaxseed oil. Food Chem. 2018, 269, 293–299. [Google Scholar] [CrossRef]
- Alkhatib, H.; Doolaanea, A.A.; Assadpour, E.; Mohmad Sabere, A.S.; Mohamed, F.; Jafari, S.M. Optimizing the encapsulation of black seed oil into alginate beads by ionic gelation. J. Food Eng. 2022, 328, 111065. [Google Scholar] [CrossRef]
- Azad, A.K.; Al-Mahmood, S.M.A.; Chatterjee, B.; Wan Sulaiman, W.M.A.; Elsayed, T.M.; Doolaanea, A.A. Encapsulation of black seed oil in alginate beads as a pH-sensitive carrier for intestine-targeted drug delivery: In vitro, in vivo and ex vivo study. Pharmaceutics 2020, 12, 219. [Google Scholar] [CrossRef] [PubMed]
- Alkhatib, H.; Mohamed, F.; Alfatama, M.; Assadpour, E.; Kharazmi, M.S.; Jafari, S.M.; Sarker, M.Z.I.; Sadasivuni, K.; Mohmad Sabere, A.S.; Doolaanea, A.A. Pilot scale manufacturing of black seed oil-loaded alginate beads; process development, and stability of thymoquinone. Heliyon 2024, 10, e37630. [Google Scholar] [CrossRef] [PubMed]
- Negi, P.; Sharma, I.; Hemrajani, C.; Rathore, C.; Bisht, A.; Raza, K.; Katare, O.P. Thymoquinone-loaded lipid vesicles: A promising nanomedicine for psoriasis. BMC Complement. Altern. Med. 2019, 19, 334. [Google Scholar] [CrossRef] [PubMed]
- Karaaslan, A. Nano- and micro-encapsulation of long-chain-fatty-acid-rich melon seed oil and its release attributes under in vitro digestion model. Foods 2023, 12, 2371. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Hong, L.; Li, Y.; Ngai, T. All-silica submicrometer colloidosomes for cargo protection and tunable release. Angew. Chem. Int. Ed. 2018, 57, 11662–11666. [Google Scholar] [CrossRef]
- Jiang, H.; Sheng, Y.; Ngai, T. Pickering emulsions: Versatility of colloidal particles and recent applications. Curr. Opin. Colloid Interface Sci. 2020, 49, 1–15. [Google Scholar] [CrossRef]
- Slavova, T.; Radulova, G.; Kralchevsky, P.; Danov, K. Encapsulation of fragrances and oils by core-shell structures from silica nanoparticles, surfactant and polymer: Effect of particle size. Colloids Surf. A Physicochem. Eng. Asp. 2020, 606, 125558. [Google Scholar] [CrossRef]
- Müller, R.H.; Petersen, R.D.; Hommoss, A.; Pardeike, J. Nanostructured lipid carriers (NLC) in cosmetic dermal products. Adv. Drug Deliv. Rev. 2007, 59, 522–530. [Google Scholar] [CrossRef]
- Krasodomska, O.; Paolicelli, P.; Cesa, S.; Casadei, M.A.; Jungnickel, C. Protection and viability of fruit seeds oils by nanostructured lipid carrier (NLC) nanosuspensions. J. Colloid Interface Sci. 2016, 479, 25–33. [Google Scholar] [CrossRef]
- AlZahabi, S.; Sakr, O.S.; Ramadan, A.A. Nanostructured lipid carriers incorporating prickly pear seed oil for the encapsulation of vitamin A. J. Cosmet. Dermatol. 2019, 18, 1875–1884. [Google Scholar] [CrossRef]
- Chu, C.C.; Tan, C.P.; Nyam, K.L. Development of nanostructured lipid carriers (NLCs) using pumpkin and kenaf seed oils with potential photoprotective and antioxidative properties. Eur. J. Lipid Sci. Technol. 2019, 121, 1900082. [Google Scholar] [CrossRef]
- Fathi, N.; Lotfipour, F.; Dizaj, S.M.; Hamishehkar, H.; Mohammadi, M. Antimicrobial activity of nanostructured lipid carriers loaded Punica granatum seed oil against Staphylococcus epidermidis. Pharm. Nanotechnol. 2020, 8, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Saporito, F.; Sandri, G.; Bonferoni, M.C.; Rossi, S.; Boselli, C.; Cornaglia, A.I.; Mannucci, B.; Grisoli, P.; Vigani, B.; Ferrari, F. Essential oil-loaded lipid nanoparticles for wound healing. Int. J. Nanomed. 2017, 13, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Klopcevska, J.; Kavrakovski, Z.; Srbinoska, M.; Rafajlovska, V. Nanoemulsions of pumpkin seed oil with turmeric extract. In Proceedings of the International Scientific and Professional Conference POLITEHNIKA 2023, Belgrade, Serbia, 15 December 2023; pp. 861–866. Available online: https://www.researchgate.net/publication/376957967_nanoemulsions_of_pumpkin_seed_oil_with_turmeric_extract (accessed on 11 June 2025).
- Al-Bayati, M.A.; Hasan, W.A.; Abdulwahab, W.M. Burned wound healing effect of prepared pumpkin seed oil nano phytosome loaded lidocaine in rabbit. Adv. Anim. Vet. Sci. 2024, 12, 723–731. [Google Scholar] [CrossRef]
- Akhter, A.; Shirazi, J.H.; Khan, H.M.S.; Hussain, M.D.; Kazi, M. Development and evaluation of nanoemulsion gel loaded with bioactive extract of Cucumis melo var. agrestis: A novel approach for enhanced skin permeability and antifungal activity. Heliyon 2024, 10, e35069. [Google Scholar] [CrossRef] [PubMed]
- Teeranachaideekul, V.; Parichatikanond, W.; Junyaprasert, V.B.; Morakul, B. Pumpkin seed oil-loaded niosomes for topical application: 5α-reductase inhibitory, anti-inflammatory, and in vivo anti-hair loss effects. Pharmaceuticals 2022, 15, 930. [Google Scholar] [CrossRef]
- Silva, M.A.; Albuquerque, T.G.; Ferreira, D.M.; Alves, R.C.; Oliveira, M.B.; Costa, H.S. Nutritional and Bioactive Profiling of Cucumis melo L. By-Products: Towards a Circular Food Economy. Molecules 2025, 30, 1287. [Google Scholar] [CrossRef]
- Azwanida, N.N. A review on the extraction methods use in medicinal plants, principle, strength and limitation. Med. Aromat. Plants 2015, 4, 1000196. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkar, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64–73. [Google Scholar] [CrossRef]
- Lane, M.E. Skin penetration enhancers. Int. J. Pharm. 2013, 447, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Characteristics of the Aging Skin. Adv. Wound Care 2013, 2, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ganguli-Indra, G.; Indra, A.K. Lipidomic analysis of epidermal lipids: A tool to predict progression of inflammatory skin disease in humans. Expert Rev. Proteom. 2016, 13, 451–456. [Google Scholar] [CrossRef]
- Liu, J.K. Natural products in cosmetics. Nat. Prod. Bioprospect. 2022, 12, 40. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Tarafdar, A.; Kumar, V.; Prabakaran, G.; Rajendran, K.; Giri, B.; Gómez-García, R.; Li, H.; Zhang, Z.; Raveendran, S.; et al. Sustainable biorefinery approaches towards circular economy for conversion of biowaste to value added materials and future perspectives. Fuel 2022, 325, 124846. [Google Scholar] [CrossRef]
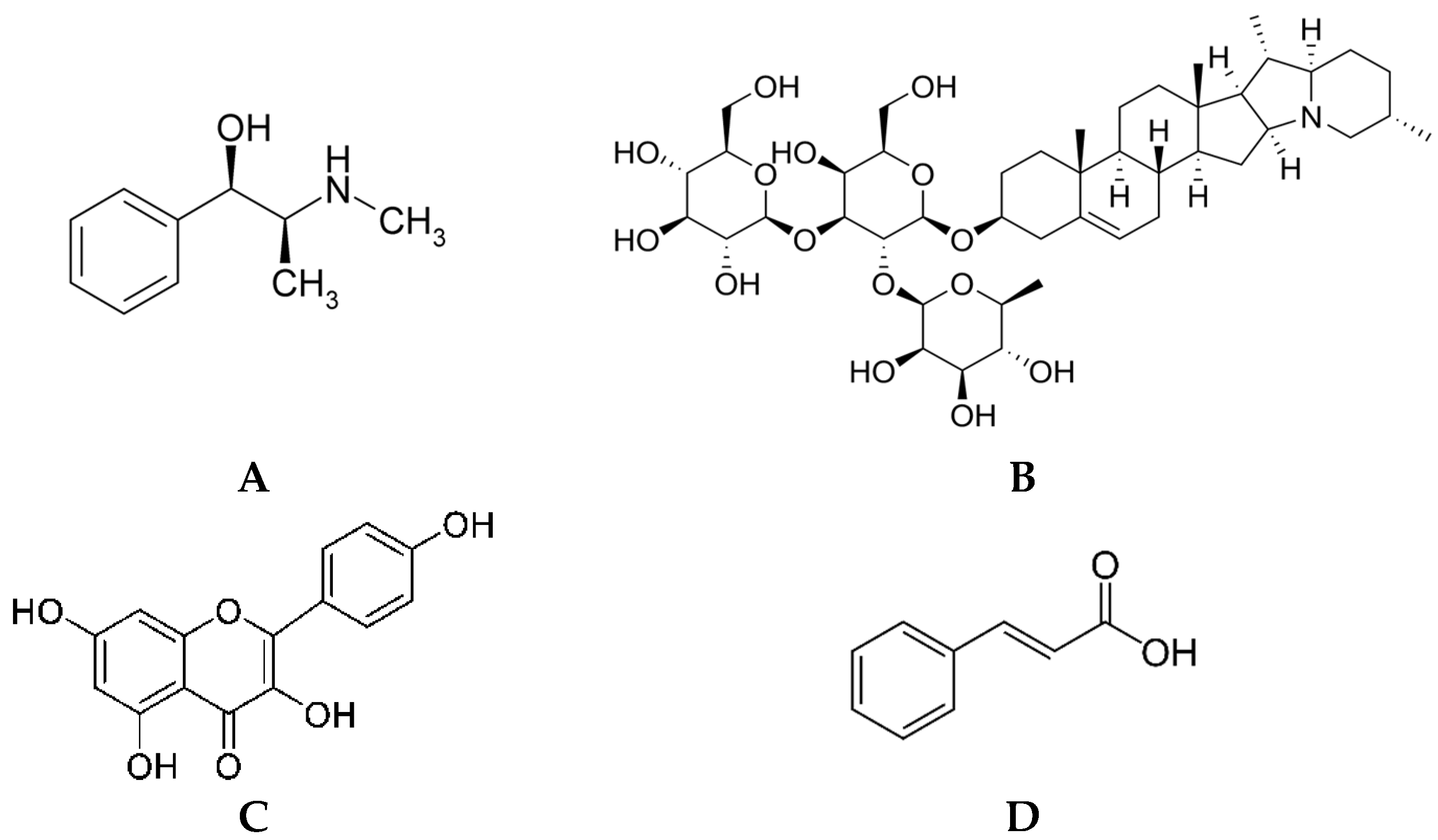
| Group | Compound | C. lanatus | C. melo | C. pepo | Properties/Applications |
|---|---|---|---|---|---|
| Alkaloids | 28.33–95.8 mg/g [1,42] | 2.54–304.12 mg/g [1,42] | 41.47 mg/g [43] | Anticellulitis and anti-aging products; tonics, creams, lotions, face masks; hair masks and discoloration [33] | |
| Saponins | 11.62–16.87 mg/g [1,42] | 13.25 mg/g [42] | 33.05 mg/g [43] | Skin, hair and oral care products; emulsifiers; antioxidants; surface-active activities [44] | |
| Flavonoids | Naringenin-7-O-glycoside | Not found | 4.30 mg/100 g [45] | Not found | Anti-inflammatory effects in cells exposed to ultraviolet B (UVB) radiation [46] |
| Luteolin | 7.19 mg/mL [47] | 2.10 mg/100 g [45] | Detected; not quantified [48] | Anti-inflammatory, signs of skin diseases (psoriasis and dermatitis) attenuation, wound healing; photoprotection and anti-aging [46] | |
| Apigenin | Detected; not quantified [49,50] | 0.50 mg/100 g [45] | Not found | Symptoms of skin inflammatory diseases attenuation, psoriasis combat, and pruritus alleviation [40] | |
| Amentoflavone | Not found | 1.78 mg/100 g [45] | Not found | Anti-wrinkle [51]; anti-inflammatory; improvement of psoriasis-like lesions [52] | |
| Hydroxytyrosol | Not found | 1.28 mg/100 g [45] | Not found | Antioxidant; antimicrobial; anti-inflammatory; anti-aging; photoprotector; depigmenting [53] | |
| Oleuropein | Not found | 3.03 mg/100 g [45] | Not found | Antioxidant; antimicrobial; anti-inflammatory; anti-aging; hydration effect [54] | |
| Resveratrol | Not found | 2.92 mg/100 g [45] | Not found | Antioxidant; antimicrobial; anti-inflammatory; anti-aging; photoprotector [55]; hair growth promotion [56] | |
| Quercetin-3-rutinoside | 0.46 mg/mL [47] | 3.91 mg/100 g [57] | Detected; not quantified [58] | Antioxidant, anti-inflammatory, photoprotector [59] | |
| Catechin | Not found | 4.33–7.4 mg/100 g [57,60] 0.90–1.77 μg/mL [61] | 9.7 mg/100 g [60] | Component of soaps, sunscreens, creams; antioxidant; antibacterial; anti-inflammatory; anti-acne; anti-aging; photoprotector [62] | |
| Phenolic acids | Gallic acid | 3.765 μg/100 g [47] 2.37 mg/mL [44] | 4.24–8.4 mg/100 g [45] | 945–5.4 × 103 μg/100 g [60,63] | Antioxidant; anti-inflammatory; anti-skin aging; atopic dermatitis and hyperpigmentation combat; wound-healing improvement [64,65] |
| Protocatechuic acid | Not found | 0.93–4.9 mg/100 g [45,60] | 158–5.2 × 103 μg/100 g [1,60] | Antioxidant; anti-inflammatory; environmental damage protection; anti-wrinkle and anti-aging effects [66,67] | |
| Chlorogenic acid | Not found | 1.25–5.3 mg/100 g [45,60] | 79–1490 μg/100 g [63] | Antioxidant; anti-inflammatory; anti-aging effects [35] | |
| p-hydroxybenzoic acid | 52.84 μg/100 g [68] | 3.28 mg/100 g [45] | 2.70 × 10−3–364 μg/100 g [1,69] | Antimicrobial; cosmetic preservative [70] | |
| Vanillic acid | 28.42 μg/100 g [68] | 3.87 mg/100 g [45] 0.58–1.02 μg/mL [61] | 224–835 μg/100 g [63] | Anti-aging; photoprotector [71] | |
| Rosmarinic acid | Not found | 1.87 mg/100 g [45] | Not found | Atopic dermatitis symptoms combat [72]; skin hydration, elasticity, and firmness [36] | |
| Phenylacetic acid | Not found | 1.35 mg/100 g [45] | Not found | Applied in cosmetics due to its honey-like scent characteristic [73] | |
| Sinapic acid | 138.41 μg/100 g [68] | Not found | 62–155 μg/100 g [63] | Skin whitening; anti-aging [74] | |
| Caffeic acid | 26.33–133 μg/100 g [1,68] 0.45 mg/mL [47] | 1.9–66.0 mg/100 g [57,60] | 32–184 μg/100 g [63] | Antimicrobial; antioxidant; anti-inflammatory; anti-skin aging; photoprotector [75]; anti-wrinkle [76] | |
| Syringic acid | 15.11 μg/100 g [68] | 1.5 mg/100 g [60] | 47–1.0 × 103 μg/100 g [60,63] | Antioxidant; photoprotector; wound-healing improvement [34] | |
| p-coumaric acid | 28.52 μg/100 g [68] 2.29 mg/mL [47] | 1.2 mg/100 g [60] | 123–504 μg/100 g [1,63] | Antioxidant; antimelanogenic; UV-induced erythema attenuation [37] | |
| Ellagic acid | Not found | 6.52 mg/100 g [57] 0.06–0.07 μg/mL [61] | 7.191 mg/100 g [66] | Skin whitening agent; UV photoprotector and anti-photoaging [38] | |
| Ferulic acid | 74.12 μg/100 g [68] | 13.483 mg/100 g [77] | 44–132 μg/100 g [63] | Antioxidant; skin photoaging protection; collagen and elastin fibers synthesis stimulation; anti-wrinkle creams, sunscreens, body scrubs, skin moisturizing, anti-seborrheic, and anti-acne preparations [36] |
| Compound | C. lanatus | C. melo | C. pepo | Properties/Applications |
|---|---|---|---|---|
| Palmitic acid | 14.84–16.58 [10] | 7.19–9.74 [87] | 9.8–15.3 [88] | Lotions and cleansers; essential for skin barrier health [89] |
| Stearic acid | 13.83–14.58 [10] | 4.57–5.86 [87] | 6.9–9.1 [88] | Lotions and cleansers; essential for skin barrier health [89] |
| Oleic acid | 10.51–17.73 [10] | 15.23–33.96 [87] | 37.9–45.5 [88] | Cleansing agent [90]; effective penetration enhancer in topical application [85]; used in nanoparticle-mediated delivery systems [86] |
| Linoleic acid | 52.57–56.94 [10] | 50.69–69.22 [87] | 32.6–35.4 [88] | Repair of the skin barrier; skin whitening; photoprotection; stimulation of hair growth [84] |
| α-Tocopherol | 6.22 × 10−3–9.23 × 10−3 [10] | 3.74 × 10−3–7.47 × 10−3 [87] | 1.01 × 10−3–1.15 × 10−3 [91] | Lipsticks, sunscreens, hair and nail cosmetics, body lotions, and anti-wrinkle creams [30]; antioxidants and skin-conditioning agents [79] |
| β-Tocopherol | 0.73 × 10−3–0.78 × 10−3 [10] | 1.15 × 10−3–2.13 × 10−3 [87] | 1.35 × 10−3 [92] | |
| γ-Tocopherol | 16.33 × 10−3–59.99 × 10−3 [10] | 9.99 × 10−3–45.67 × 10−3 [87] | 25.10 × 10−3–96.4 × 10−3 [91,92] | |
| δ-Tocopherol | 4.28 × 10−3–4.54 × 10−3 [10] | 0.93 × 10−3–2.72 × 10−3 [87] | 2.2 × 10−4–1.99 × 10−3 [91,92] | |
| Stigmasterol/β -Sitosterol | 28.30 × 10−3–58.05 × 10−3 [10] | 1.29 × 10−3–324.84 × 10−3 [93] | 147.7 × 10−3–208.2 × 10−3 [16] | Creams and lipstick [94]; anti-aging products, promote collagen and hyaluronic acid biosynthesis; retain moisture within skin [83] |
| Technique/Delivery System | Encapsulating Material | Particle Size | Encapsulation Efficiency | Stability | Description | Cosmetic Application | Seed Oil | References |
|---|---|---|---|---|---|---|---|---|
| Nanoemulsification | Tween 80, lecithin, PEG-40 hydrogenated castor oil | 20–200 nm | 80–95% | Stable for up to 3 months (RT) | Oil-in-water or water-in-oil systems stabilized by surfactants. Enhance dispersion of hydrophobic oils. Increase bioavailability and stability of lipophilic compounds. | Provides light and non-greasy texture. Increased skin absorption. Used in moisturizers, sunscreens, and anti-aging creams. | Caesalpinia decapetala, Melaleuca alternifolia, Nigella sativa. Cucurbita pepo, Cucurbita maxima, Cucurbita moschata | [149,150,155,156,157,158] |
| Electrospinning | Chitosan and polyvinyl alcohol (PVA) | ~200–700 nm (fiber diameter) | Moderate (~60%) | Stable in dry conditions; moisture-sensitive | High-voltage technique producing nanofibers embedding oils. | Applied in dermal masks, wound dressings, and anti-inflammatory pads. Biodegradable and high-surface release systems. | Nigella sativa, Cucurbita pepo | [159,160] |
| Nanoprecipitation | Propolis–phosphatidylcholine complex and Eudragit | 100–250 nm | 80–90% | Stable for >30 days at RT or 4 °C | Solvent displacement process creating polymer nanoparticles. Improves oxidative stability and dispersibility of seed oils. Rapid and energy-efficient process. | Incorporated in antioxidant gels and anti-aging emulsions. Employed in cosmetic formulations containing heat-sensitive lipophilic bioactives. | Camellia sinensis, Salvia hispanica | [152,161,162] |
| Ionic gelation | Chitosan and sodium tripolyphosphate (TPP) | ~50–800 nm | ~70–85% | Stable in acidic pH; sensitive to multivalent ions | Nanoparticles formed by ionic crosslinking of oppositely charged biopolymers. Carried out under mild conditions without organic solvents. Suitable for encapsulating heat- and pH-sensitive seed oils. | Used in moisturizing gels, anti-inflammatory creams, and sensitive skin formulations. For controlled release of antioxidants and enhanced dermal delivery. | Citrus sinensis, Linalool oil | [163,164,165,166] |
| Liposomes | Phosphatidylcholine and cholesterol | 50–300 nm | Moderate to high (60–85%) | Physically unstable over time (risk of fusion/leakage); improved with ethosomes or gel-thickened systems | Spherical vesicles with phospholipid bilayers encapsulating aqueous and lipophilic phases. Mimic skin lipid structure, enhancing bioavailability and hydration. Flexible formulations include ethosomes, niosomes, transfersomes, and gelosomes. | Used in anti-aging serums, eye creams, and skin-repair gels. Enhance skin penetration, reduce irritation, and provide deep moisturization. | Ribes nigrum, Cucurbita pepo, Cucumis melo; Punica granatum | [148,151,167,168] |
| Colloidosomes | Silica nanoparticles, chitosan, polymeric shells | 200 nm–2 μm | Moderate (~60–75%) | Stable under controlled pH and ionic conditions; tunable release via shell permeability | Microcapsules formed by self-assembly of colloidal particles at oil–water interfaces. Can be designed to respond to pH or temperature for controlled release. Limited commercial application but promising for targeted delivery. | Potential use in serums or targeted release patches. Offers protection of sensitive compounds and gradual release of antioxidants or fragrances. | Punica granatum, Linum usitatissimum | [169,170,171] |
| Nanostructured lipid carriers (NLCs) | Mixture of solid (e.g., glyceryl behenate) and liquid lipids (e.g., oleic acid), stabilized with surfactants | 80–200 nm | High (70–95%) | Good physical and chemical stability; resistant to lipid polymorphism if optimized | Lipid matrix combining solid and liquid lipids forms imperfect crystalline structure. Enables high loading and controlled release of bioactives. Suitable for hydrophobic seed oils needing enhanced penetration and shelf-life. | Widely used in anti-aging creams, UV-protective lotions, and intensive moisturizers. Improves skin retention, hydration, and antioxidant protection. | Opuntia ficus-indica, Punica granatum, Cucurbita pepo, Ribes nigrum, Morus nigra, Rubus idaeus, Fragaria × ananassa, Prunus domestica | [148,154,172,173,174,175,176] |
| Solid lipid nanoparticles (SLNs) | Solid lipids (e.g., stearic acid, glyceryl stearate) and surfactants | 50–300 nm | Moderate to high (60–85%) | High oxidative and photostability; crystallization may lead to bioactive expulsion over time | Nanoparticles composed entirely of solid lipids forming a crystalline matrix. Protects oils from oxidation and light degradation. | Used in skin barrier-repair creams, antioxidant serums, and wound-healing ointments. Improves bioactive stability, prolongs skin contact and moisturizing effect. | Cucurbita pepo, Vitis vinifera | [177] |
| Seed Oil | Encapsulation System | Cosmetic Application | Observed Benefits | Reference |
|---|---|---|---|---|
| C. pepo | NLCs | Sunscreens; photoprotective lotions | UV protection and photostability increasement | [175] |
| Niosomes | Topical anti-inflammatory creams; anti-hair loss therapy | Enhanced skin permeation; inhibition of 5α-reductase; hair loss reduction of 44.42% in vivo | [181] | |
| Chitosan/PVA-based electrospun nanofiber | Wound dressing; dermal regeneration | Promotes skin regeneration; provides sustained release; protective moist environment for wound healing | [160] | |
| Nanoemulsion | Moisturizing creams; anti-aging products | Enhanced skin hydration; improved antioxidant stability; formation of stable O/W emulsions | [178] | |
| Chitosan nanoparticles (phytosome via ionic gelation) | Wound-healing gels; skin care formulations | Improved skin penetration; controlled release of bioactives; accelerated wound healing in vivo | [179] | |
| C. melo | Nanoemulsion | Topical delivery with antifungal activity | Improved skin permeability; enhanced antifungal activity (vs. Candida albicans and Trichophyton rubrum) | [180] |
| C. lanatus | Nanoemulsion | Cosmeceutical formulations; moisturizing creams | Enhanced antioxidant activity; improved skin hydration; high PUFA content contributes to barrier repair | [26] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousa, C.; Moutinho, C.G.; Carvalho, M.; Matos, C.; Vinha, A.F. Transforming By-Products into Functional Resources: The Potential of Cucurbitaceae Family Seeds in Cosmetics. Seeds 2025, 4, 36. https://doi.org/10.3390/seeds4030036
Sousa C, Moutinho CG, Carvalho M, Matos C, Vinha AF. Transforming By-Products into Functional Resources: The Potential of Cucurbitaceae Family Seeds in Cosmetics. Seeds. 2025; 4(3):36. https://doi.org/10.3390/seeds4030036
Chicago/Turabian StyleSousa, Carla, Carla Guimarães Moutinho, Márcia Carvalho, Carla Matos, and Ana Ferreira Vinha. 2025. "Transforming By-Products into Functional Resources: The Potential of Cucurbitaceae Family Seeds in Cosmetics" Seeds 4, no. 3: 36. https://doi.org/10.3390/seeds4030036
APA StyleSousa, C., Moutinho, C. G., Carvalho, M., Matos, C., & Vinha, A. F. (2025). Transforming By-Products into Functional Resources: The Potential of Cucurbitaceae Family Seeds in Cosmetics. Seeds, 4(3), 36. https://doi.org/10.3390/seeds4030036







