Environmental Determinates of Distribution for Dragonfly Nymphs (Odonata: Anisoptera) in Urban and Non-Urban East Texas Streams, USA
Abstract
:1. Introduction
2. Materials and Methods
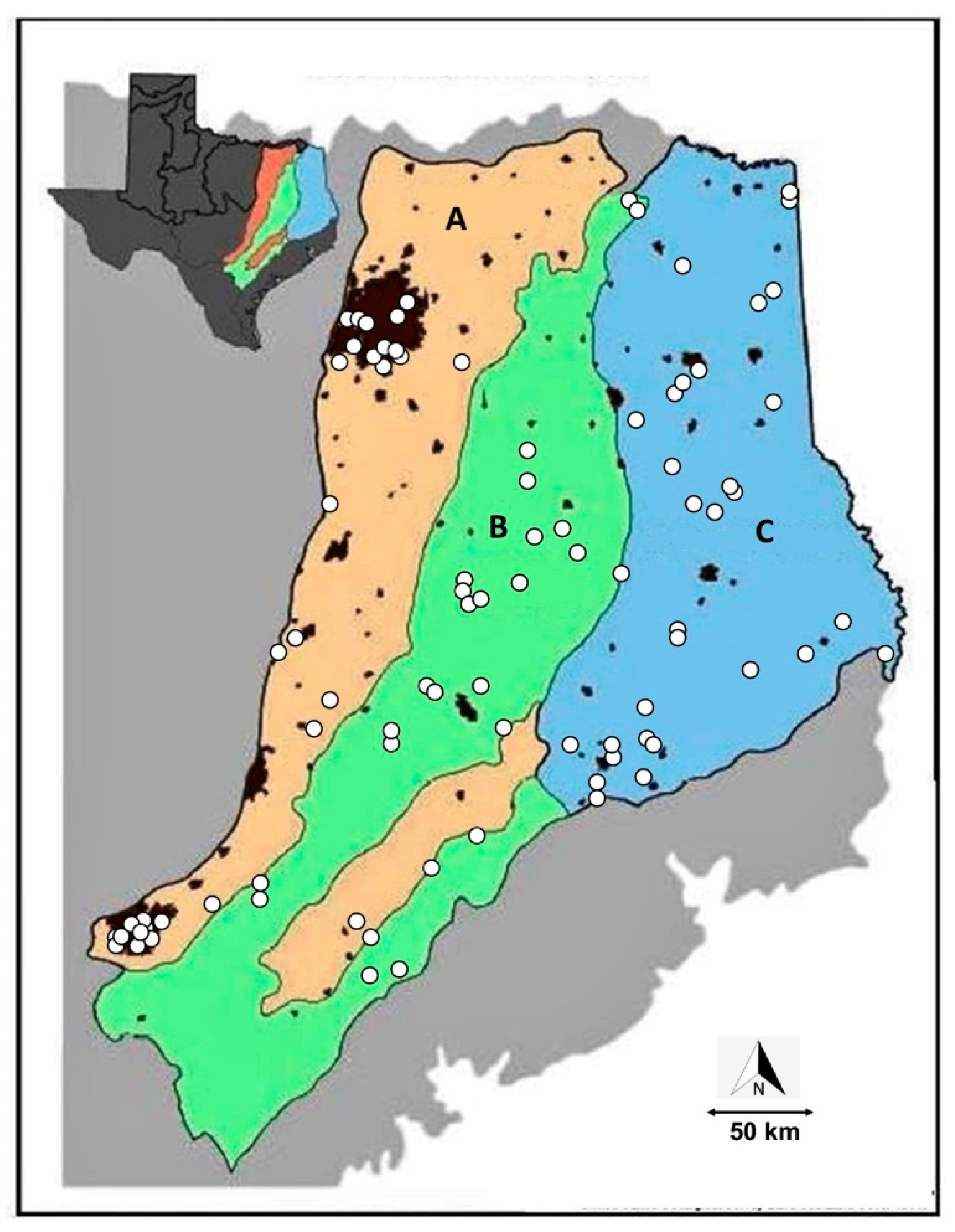
2.1. Sampling Sites
2.2. Sample Collections
2.3. Specimen Identification
2.4. Tolerance Values
2.5. Statistical Analyses
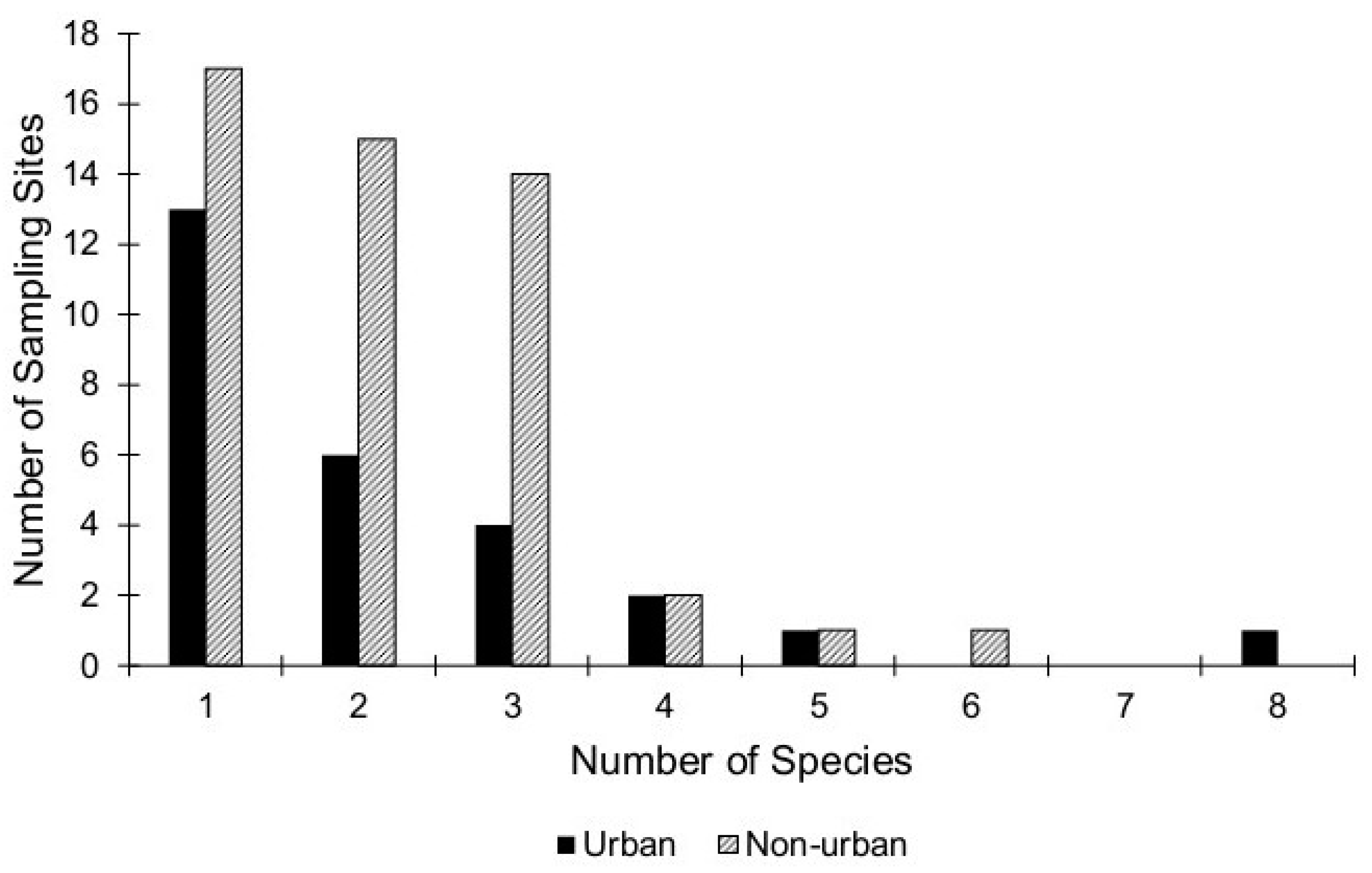
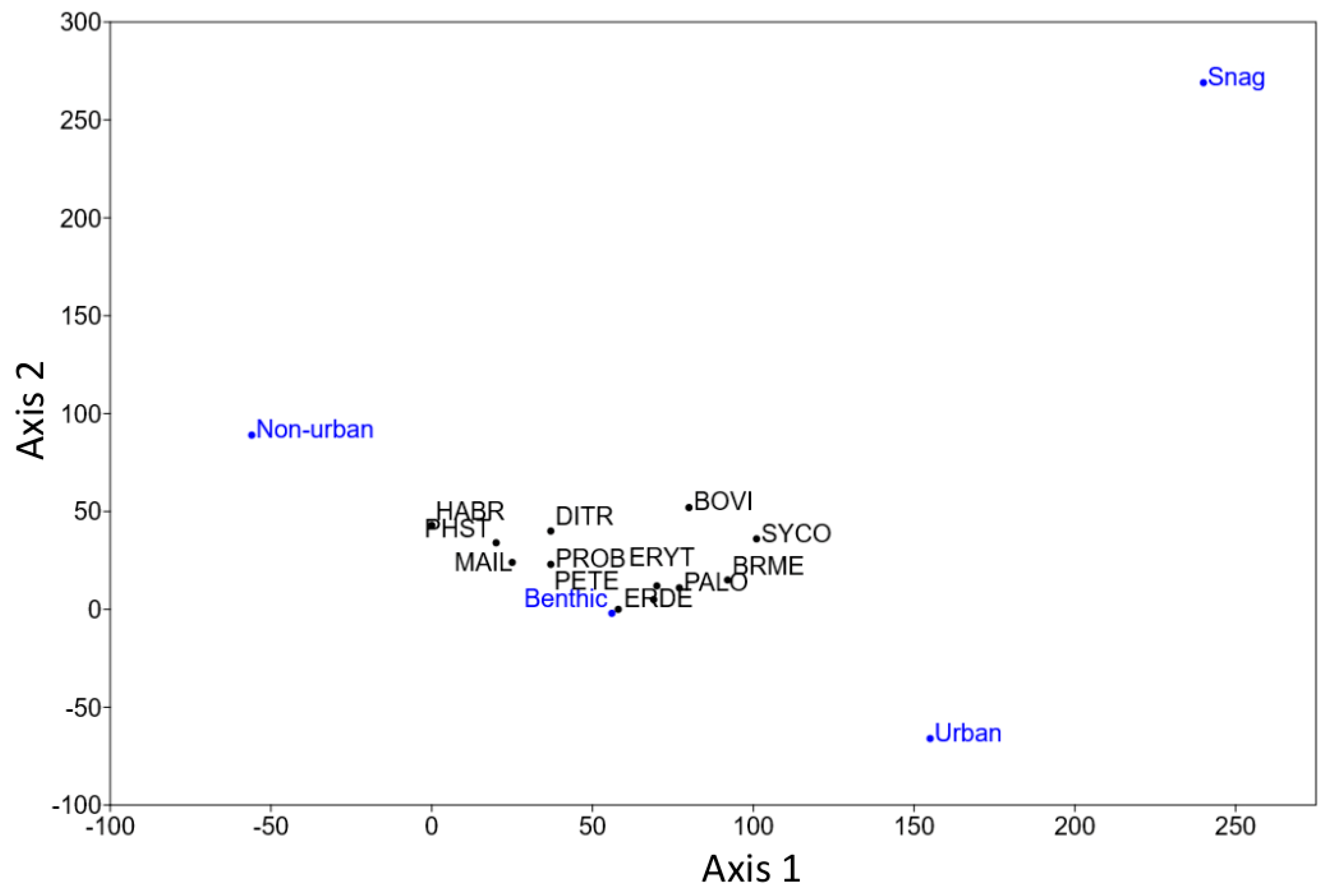
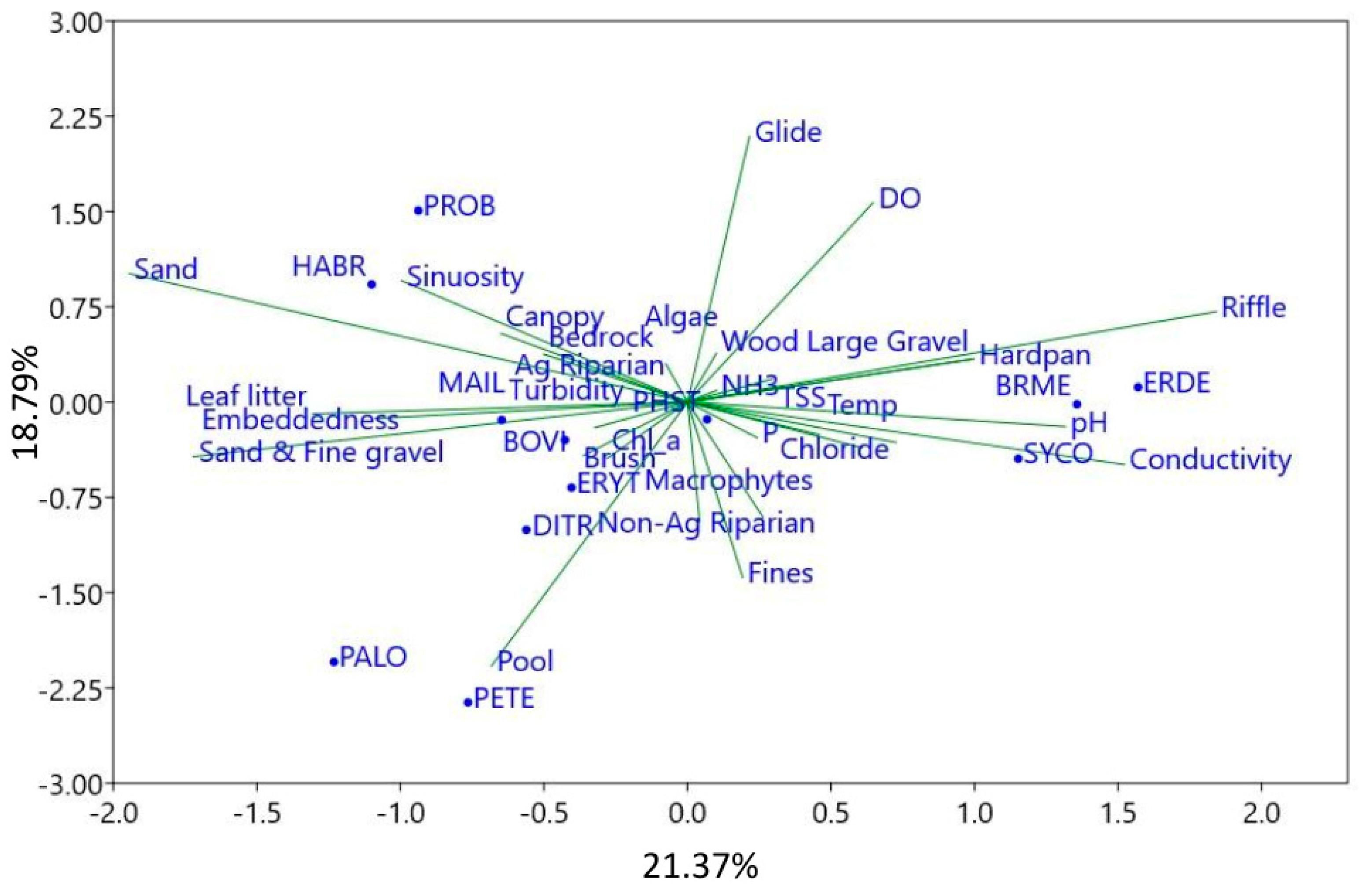
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hassall, C.; Hollinshead, J.; Hull, A. Environmental correlates of plant and invertebrate species richness in ponds. Biodivers. Conserv. 2011, 20, 3189–3222. [Google Scholar] [CrossRef]
- Monteiro-Júnior, C.S.; Juen, L.; Hamada, N. Analysis of urban impacts on aquatic habitats in the central Amazon basin: Adult odonates as bioindicators of environmental quality. Ecol. Indic. 2015, 48, 303–311. [Google Scholar] [CrossRef]
- Monteiro-Júnior, C.S.; Juen, L.; Hamada, N. Effects of urbanization on stream habitats and associated adult dragonfly and damselfly communities in central Brazilian Amazonia. Landsc. Urban Plan. 2014, 127, 28–40. [Google Scholar] [CrossRef]
- Oliveira-Junior, J.M.B.; Shimano, Y.; Gardner, T.A.; Hughes, R.M.; de Marco Júnior, P.; Juen, L. Neotropical dragonflies (Insecta: Odonata) as indicators of ecological condition of small streams in the eastern Amazon. Austral Ecol. 2015, 40, 733–744. [Google Scholar] [CrossRef]
- Oliveira-Junior, J.M.B.; Juen, L. The Zygoptera/Anisoptera ratio (Insecta: Odonata): A new tool for habitat alterations assessment in Amazonian streams. Neotrop. Entomol. 2019, 48, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Samways, M.J.; Simaika, J.P. Dragonfly biotic index. In Manual of Freshwater Assessment for South Africa; South African National Biodiversity Institute: Pretoria, South Africa, 2016; p. 224. [Google Scholar]
- Seidu, I.C.; Nsor, A.; Danquah, E.; Lancaster, J.T. Odonata assemblages along an anthropogenic disturbance gradient in Ghana’s Eastern Region. Odonatologica 2018, 47, 73–100. [Google Scholar]
- Silva, L.F.R.; Machado, F.S.; Resende, D.L.M.C.; Neiss, U.G. Immature Odonata community in streams: Diversity, season variation and habitat preference in different levels of degradation. North-West. J. Zool. 2018, 14, 232–236. [Google Scholar]
- Šigutová, H.; Šipošb, J.; Dolný, A. A novel approach involving the use of Odonata as indicators of tropical forest degradation: When family matters. Ecol. Indic. 2019, 104, 229–236. [Google Scholar] [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.D.; Stribling, J.B. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrate, and Fish; EPA 841-B-99-002, 2nd ed.; U.S. Environmental Protection Agency: Washington, DC, USA, 1999; p. 339.
- Edwards, R.J.; Longley, G.; Moss, R.; Ward, J.; Matthews, R.; Stewart, B. A classification of Texas aquatic communities with special consideration toward the conservation of endangered and threatened taxa. Tex. J. Sci. 1989, 41, 231–240. [Google Scholar]
- Bowles, D.E.; Kleinsasser, L.J.; Jurgensen, T.A. Environmental determinates of stream caddisfly (Trichoptera) diversity in eastern Texas, USA. Trans. Kans. Acad. Sci. 2016, 119, 281–298. [Google Scholar] [CrossRef]
- Arrowsmith, J.; Shivaprakash, K.N.; Larrivée, M.; Turgeon, J.; Lessard, J.P. Environmental filtering along a broad-scale acidity gradient shapes the structure of odonate communities. Ecosphere 2018, 9, e02473. [Google Scholar] [CrossRef]
- Leland, H.V.; Fend, S.V. Benthic invertebrate distributions in the San Joaquin River, California, in relation to physical and chemical factors. Can. J. Fish. Aquat. Sci. 1998, 55, 1051–1067. [Google Scholar] [CrossRef]
- Urban, M.C.; Skelly, D.K.; Burchstead, D.; Price, W.; Lowry, S. Stream communities across a rural-urban landscape gradient. Divers. Distrib. 2006, 12, 337–350. [Google Scholar] [CrossRef]
- Lassettre, N.S.; Kondolf, G.M. Large woody debris in urban stream channels: Redefining the problem. River Res. Appl. 2011, 28, 1477–1487. [Google Scholar] [CrossRef]
- Valente-Neto, F.; Koroiva, R.; Fonseca-Gessner, A.A.; de Oliveira Roque, F. The effect of riparian deforestation on macroinvertebrates associated with submerged woody debris. Aquat. Ecol. 2015, 49, 115–125. [Google Scholar] [CrossRef]
- Balinsky, B.I. On some intrinsic and environmental factors controlling the distribution of dragonflies (Odonata), with redescription and a new name for a little known species. J. Entomol. Soc. S. Afr. 1967, 29, 3–22. [Google Scholar]
- Schindler, M.; Christian, F.; Chovanec, A. Dragonfly associations (Insecta: Odonata) in relation to habitat variables: A multivariate approach. Hydrobiologia 2003, 497, 169–180. [Google Scholar] [CrossRef]
- Remsburg, A.J.; Turner, M.G. Aquatic and terrestrial drivers of dragonfly (Odonata) assemblages within and among north-temperate lakes. J. N. Am. Benthol. Soc. 2009, 28, 44–56. [Google Scholar] [CrossRef]
- Carvalho, F.G.; Pinto, N.S.; Oliveira-Júnior, J.M.B.; Juen, L. Effects of marginal vegetation removal on Odonata communities. Acta Limnol. Bras. 2013, 25, 10–18. [Google Scholar] [CrossRef] [Green Version]
- Koparde, P.; Mhaske, P.; Patwardhan, A. Habitat correlates of Odonata species diversity in the northern Western Ghats, India. Odonatologica 2015, 44, 21–43. [Google Scholar]
- Oliveira-Junior, J.M.B.; de Junior, P.; Dias-Silva, K.; Leitão, R.P.C.G.; Pompeu, P.S.; Gardner, T.A.; Hughes, T.A.; Juen, L. Effects of human disturbance and riparian conditions on Odonata (Insecta) assemblages in eastern Amazon basin streams. Limnologica 2017, 66, 31–39. [Google Scholar] [CrossRef]
- Brito, J.S.; Michelan, T.S.; Juen, L. Aquatic macrophytes are important substrates for Libellulidae (Odonata) larvae and adults. Limnology 2020, 22, 139–149. [Google Scholar] [CrossRef]
- Wildermuth, H. Habitat requirements of Orthetrum coerulescens and management of a secondary habitat in a highly man-modified landscape (Odonata: Libellulidae). Int. J. Odonatol. 2008, 11, 261–276. [Google Scholar] [CrossRef]
- Balzan, M.V. Associations of dragonflies (Odonata) to habitat variables within the Maltese Islands: A spatiotemporal approach. J. Insect Sci. 2012, 12, 87. [Google Scholar] [CrossRef] [Green Version]
- Cuevas-Yánez, K.; Benítez, M.; Rocha, M.; Córdoba-Aguilara, A. Large-scale human environmental intervention is related to a richness reduction in Mexican odonates. Rev. Mex. Biodivers. 2017, 88, 664–673. [Google Scholar] [CrossRef]
- Oliveira-Junior, J.M.B.; Dias-Silva, K.; Teodósio, M.A.; Juen, L. The response of Neotropical dragonflies (Insecta: Odonata) to local and regional abiotic factors in small streams of the Amazon. Insects 2019, 10, 446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benchalel, W.; Bouziane, A.; Bouslama, Z.; Ramdani, M.; Elmsellem, H.; Flower, R.; Mohammed, M.R. Odonata of Wadi Bouarroug (northeastern Algeria) and environmental determinants of their distribution. Mor. J. Chem. 2018, 6, 78–91. [Google Scholar]
- Prescott, V.A.; Eason, P.K. Lentic and lotic odonate communities and the factors that influence them in urban versus rural landscapes. Urban Ecosyst. 2018, 21, 737–750. [Google Scholar] [CrossRef]
- Nagy, H.B.; László, Z.; Szabó, F.; Szőcs, L.; Dévai, G.; Tóthmérész, B. Landscape-scale terrestrial factors are also vital in shaping Odonata assemblages of watercourses. Sci. Rep. 2019, 20199, 18196. [Google Scholar] [CrossRef]
- Oliveira-Junior, J.M.B.; Juen, L. Structuring of dragonfly communities (Insecta: Odonata) in eastern Amazon: Effects of environmental and spatial factors in preserved and altered streams. Insects 2019, 10, 322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moring, J.B. Influence of stream habitat and land use on benthic macroinvertebrate indictors of stream quality of selected above-tidal streams in the Houston-Galveston area council service area, Texas, 1997–1998. In Water-Resources Investigations Report 01-4010; U.S. Geological Survey: Washington, DC, USA, 2001; p. 22. [Google Scholar]
- Kleinsasser, L.J.; Jurgensen, T.A.; Bowles, D.E.; Boles, S.; Aziz, K.; Saunders, K.S.; Linam, G.W.; Trungale, J.F.; Mayes, K.B.; Rector, J.; et al. Status of biotic integrity, water quality, and physical habitat in wadeable East Texas streams. In River Studies Report No. 19; Texas Parks and Wildlife Department: Austin, TX, USA, 2004; p. 80. [Google Scholar]
- Hernandez, K.M.; Reece, B.A.; McIntyre, N.E. Effects of anthropogenic land use on Odonata in playas of the southern high plains. West. N. Am. Nat. 2006, 66, 273–278. [Google Scholar] [CrossRef] [Green Version]
- Reece, B.A.; McIntyre, N.E. Community assemblage patterns of odonates inhabiting a wetland complex influenced by anthropogenic disturbance. Insect Conserv. Divers. 2009, 2, 73–80. [Google Scholar] [CrossRef]
- Husband, D.M.; McIntyre, N.E. Urban areas create refugia for odonates in a semi-arid region. Insects 2021, 12, 431. [Google Scholar] [CrossRef]
- Phillips, E.C. Habitat preference of aquatic macroinvertebrates in an east Texas sandy stream. J. Freshw. Ecol. 2003, 18, 1–11. [Google Scholar] [CrossRef]
- Horton, R.E. Erosional development of streams and their drainage basins; hydrophysical approach to quantitative morphology. Bull. Geol. Soc. Amer. 1945, 56, 275–370. [Google Scholar] [CrossRef] [Green Version]
- Omernik, J.M. Ecoregions of the conterminous United States. Ann. Am. Assoc. Geogr. 1987, 77, 118–125. [Google Scholar] [CrossRef]
- Environmental Monitoring and Assessment Program: Surface Waters and Region 3 Regional Environmental Monitoring and Assessment Program: 1994 Pilot Field Operations and Methods Manual, EPA/620/R-94/004; Klemm, D.J.; Lazorchak, J.M. (Eds.) U.S. Environmental Protection Agency: Cincinnati, OH, USA, 1994; p. 309.
- Needham, J.G.; Westfall, M.J., Jr.; May, M.L. Dragonflies of North America, revised edition; Scientific Publishers: Gainesville, FL, USA, 2000; p. 939. [Google Scholar]
- Hilsenhoff, W.L. An improved biotic index of organic stream pollution. Gt. Lakes Entomol. 1987, 20, 31–39. [Google Scholar]
- Hilsenhoff, W.L. Rapid field assessment of organic pollution with a family-level biotic index. J. N. Am. Benthol. Soc. 1988, 7, 65–68. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology, 3rd ed.; Elsevier: New York, NY, USA, 2012; p. 1006. [Google Scholar]
- Farhangfar, A.; Kurgan, L.; Dy, J. Impact of imputation of missing values on classification error for discrete data. Pattern Recognit. 2008, 41, 3692–3705. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- ter Braak, C.J.E.; Verdonschot, P.E.M. Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquat. Sci. 1995, 57, 255–289. [Google Scholar] [CrossRef]
- Stewart, J.S.; Wang, L.Z.; Lyons, J.; Horwatich, J.A.; Bannerman, R. Influences of watershed, riparian-corridor, and reach-scale characteristics on aquatic biota in agricultural watersheds. J. Am. Water Resour. Assoc. 2001, 37, 1475–1487. [Google Scholar] [CrossRef]
- Wang, L.; Lyons, J.; Kanehl, P.; Bannerman, R. Impacts of urbanization on stream habitat and fish across multiple spatial scales. Environ. Manage. 2001, 28, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Bressler, D.W.; Stribling, J.B.; Paul, M.J.; Hicks, M.B. Stressor tolerance values for benthic macroinvertebrates in Mississippi. Hydrobiologia 2006, 57, 155–172. [Google Scholar] [CrossRef]
- Lenat, D.R. A biotic index for the Southeastern United States: Derivation and list of tolerance. J. N. Am. Benthol. Soc. 1993, 12, 279–290. [Google Scholar] [CrossRef]
- Texas Commission on Environmental Quality (TCEQ). Surface water quality monitoring procedures, volume 2. In Methods for Collecting and Analyzing Biological Assemblage and Habitat Data; TCEQ: Austin, TX, USA, 2014. Available online: https://www.tceq.texas.gov/publications/rg/rg-416 (accessed on 1 March 2021).
- Bogan, M.T.; Eppenhimer, D.; Hamdhani, H.; Hollien, K. If you build it, they will come: Rapid colonization by dragonflies in a new effluent-dependent river reach. PeerJ 2020, 8, e9856. [Google Scholar] [CrossRef]
- Oppel, S. Habitat associations of an Odonata community in a lower montane rainforest in Papua New Guinea. Int. J. Odonatol. 2005, 8, 243–257. [Google Scholar] [CrossRef]
- Burcher, C.L.; Smock, L.A. Habitat distribution, dietary composition and life history characteristics of odonate nymphs in a blackwater Coastal Plain Stream. Am. Midl. Nat. 2002, 148, 75–89. [Google Scholar] [CrossRef]
- Interlandi, S.; Crockett, C.S. Recent water quality trends in Schuylkill River, Pennsylvania, USA: A preliminary assessment of the relative influences of climate, river discharge and suburban development. Water Res. 2003, 37, 1737–1748. [Google Scholar] [CrossRef]
- Paul, M.J.; Meyer, J.L. Streams in the urban landscape. Annu. Rev. Ecol. Evol. Syst. 2001, 32, 333–365. [Google Scholar] [CrossRef]
- Sandamini, P.M.M.A.; Gunarathna, S.D.; Chandana, E.P.S. Habitat quality and climate variability determine odonate species diversity and distribution patterns in selected habitats of southern Sri Lanka. Asian J. Conserv. Biol. 2019, 8, 47–57. [Google Scholar]
- Bastos, R.C.; Brito, J.; Cunhar, E.J.; Cruz, G.M.; Pereira, J.L.S.; Vieira, J.; Juen, L. Environmental impacts from human activities affect the diversity of the Odonata (Insecta) in the eastern Amazon. Intl. J. Odonatol. 2021, 24, 300–315. [Google Scholar] [CrossRef]
| Variable | Abbreviation |
|---|---|
| 24-h mean dissolved oxygen (mg/L) | DO |
| Mean temperature (Celsius) | Temp |
| pH | pH |
| Specific conductance (µm/cm) | Conductivity |
| Turbidity (NTU) | Turbidity |
| Total suspended solids (mg/L) | TSS |
| Chlorophyll a (mg/L) | CHL-a |
| Chloride (mg/L) | Chloride |
| Ammonia (mg/L) | NH3 |
| Total phosphorus (mg/L) | Phosphorus |
| Leaf litter (%) | Leaf litter |
| Filamentous algae (%) | Algae |
| Aquatic plants (%) | Macrophytes |
| Canopy, mid-channel (%) | Canopy |
| Substrate embeddedness (%) | Embeddedness |
| Large woody debris (%) | Wood |
| Brush (%) | Brush |
| Non-agricultural riparian zone (%) | Non-ag riparian |
| Agricultural riparian zone (%) | Ag riparian |
| Substrate, fines (%) | Fines |
| Substrate, sand (%) | Sand |
| Substrate ≤ sand/fine gravel (%) | Sand and fine gravel |
| Substrate, ≥large gravel (%) | Large gravel |
| Substrate, hardpan clay (%) | Hardpan |
| Bedrock substrate (%) | Bedrock |
| Channel sinuosity | Sinuosity |
| Glide habitat (%) | Glide |
| Riffle habitat (%) | Riffle |
| Pool habitat (%) | Pool |
| Taxon | Acronym | Non-Urban | Urban | Benthic | Snag |
|---|---|---|---|---|---|
| Boyeria vinosa (Say) | BOVI | 8 | 7 | 10 | 5 |
| Brechmorhoga mendax (Hagen) | BRME | 3 | 7 | 8 | 2 |
| Didymops transversa (Say) | DITR | 6 | 2 | 7 | 1 |
| Erpetogomphus designatus Hagen in Selys | ERDE | 6 | 9 | 14 | 1 |
| Erythemis simplicicollis (Say) | ERYT | 8 | 11 | 17 | 2 |
| Hagenius brevistylus Selys | HABR | 5 | 0 | 5 | 0 |
| Macromia illinoensis Walsh | 31 | 10 | 41 | 0 | |
| Pachydiplax longipennis (Burmeister) | PALO | 3 | 5 | 7 | 1 |
| Perithemis tenera (Say) | PETE | 4 | 5 | 9 | 0 |
| Phyllogomphoides stigmatus (Say) | PHST | 25 | 5 | 29 | 1 |
| Progomphus obscurus (Rambur) | PROB | 17 | 8 | 24 | 1 |
| Sympetrum corruptum (Hagen) | SYCO | 1 | 2 | 2 | 1 |
| Mean | -- | 9.83 | 6.08 | 14.67 | 1.25 |
| Source * | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxon | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | Avg. |
| Family Aeshnidae | |||||||||||
| Boyeria vinosa | 6.3 | 2 | 3.5 | - | - | 5.4 | - | - | - | - | 4.3 |
| Nasiaeschna pentacantha | 8 | - | - | - | - | - | - | - | 8 | 8 | 8.0 |
| Family Corduliidae | |||||||||||
| Somatochlora spp. | 8.9 | 1 | - | 9 | 1 | 8.4 | 1 | - | 8.9 | 1 | 4.9 |
| Somatochlora linearis | - | - | - | - | - | - | - | - | - | - | |
| Family Gomphidae | |||||||||||
| Aphylla angustifolia | - | - | - | - | - | - | - | - | - | - | |
| Erpetogomphus spp. | - | - | - | 4 | - | - | - | - | - | 1 | 2.5 |
| Erpetogomphus designatus | - | - | - | - | - | - | - | - | - | - | |
| Hagenius brevistylus | 4 | 1 | - | - | - | - | 1 | - | 4 | - | 2.0 |
| Phyllogomphoides spp. | - | - | - | - | - | - | - | - | - | 1 | 1 |
| Phyllogomphoides stigmatus | - | - | - | - | - | - | - | - | - | - | |
| Progomphus obscurus | 8.7 | - | - | - | - | - | - | 8.7 | - | 8.7 | |
| Family Libellulidae | |||||||||||
| Brechmorhoga spp. | - | - | - | - | - | 10 | - | - | - | 6 | 8.0 |
| Brechmorhoga mendax | - | - | - | - | - | - | - | - | - | - | |
| Erythemis spp. | - | - | - | - | - | - | - | - | - | 5 | 5.0 |
| Erythemis simplicicollis | - | - | - | - | - | - | - | - | - | - | |
| Orthemis spp. | - | - | - | - | - | - | - | - | - | 9 | 9.0 |
| Orthemis ferruginea | - | - | - | - | - | - | - | - | - | - | |
| Pachydiplax longipennis | 9.6 | - | - | - | - | - | - | - | 9.6 | 10 | 9.7 |
| Perithemis spp. | 10 | - | - | - | 4 | - | 10 | - | - | 4 | 7.0 |
| Perithemis tenera (Say) | - | - | - | - | - | - | - | - | - | - | |
| Playthemis lydia (Drury) | - | - | - | - | - | - | - | - | - | - | |
| Sympetrum spp. | 7.3 | 10 | - | - | 4 | - | 10 | - | 7.3 | 7 | 7.6 |
| Sympetrum corruptum (Hagen) | - | - | - | - | - | - | - | - | - | - | - |
| Family Macromiidae | |||||||||||
| Didymops spp. | - | - | - | - | - | - | - | - | - | - | - |
| Didymops transversa (Say) | - | - | - | - | - | - | - | - | - | - | - |
| Macromia spp. | 6.7 | 2 | - | - | 2 | 4.9 | 2 | - | 6.7 | 3 | 3.9 |
| Macromia illinoensis Walsh | - | - | - | - | - | - | - | - | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bowles, D.E.; Kleinsasser, L.J. Environmental Determinates of Distribution for Dragonfly Nymphs (Odonata: Anisoptera) in Urban and Non-Urban East Texas Streams, USA. Hydrobiology 2022, 1, 76-88. https://doi.org/10.3390/hydrobiology1010006
Bowles DE, Kleinsasser LJ. Environmental Determinates of Distribution for Dragonfly Nymphs (Odonata: Anisoptera) in Urban and Non-Urban East Texas Streams, USA. Hydrobiology. 2022; 1(1):76-88. https://doi.org/10.3390/hydrobiology1010006
Chicago/Turabian StyleBowles, David E., and Leroy J. Kleinsasser. 2022. "Environmental Determinates of Distribution for Dragonfly Nymphs (Odonata: Anisoptera) in Urban and Non-Urban East Texas Streams, USA" Hydrobiology 1, no. 1: 76-88. https://doi.org/10.3390/hydrobiology1010006
APA StyleBowles, D. E., & Kleinsasser, L. J. (2022). Environmental Determinates of Distribution for Dragonfly Nymphs (Odonata: Anisoptera) in Urban and Non-Urban East Texas Streams, USA. Hydrobiology, 1(1), 76-88. https://doi.org/10.3390/hydrobiology1010006







