Advanced 3D/4D Bioprinting of Flexible Conductive Materials for Regenerative Medicine: From Bioinspired Design to Intelligent Regeneration
Abstract
1. Introduction
- High Reproducibility and Control. Ensuring batch-to-batch consistency in scaffold properties (e.g., conductivity, mechanical characteristics) and biocompatibility, meeting clinical application standards [32].
2. Classification and Properties of Key Conductive Materials
2.1. Conductive Polymer Systems
2.1.1. Polythiophene Derivatives (PEDOT:PSS)
2.1.2. Polyaniline (PANI)
2.1.3. Polypyrrole (PPy)
2.2. Carbon-Based Material Systems
2.2.1. Graphene
2.2.2. Carbon Nanotubes (CNTs)
2.3. Metal Materials
2.3.1. Gold (Au)
2.3.2. Silver (Ag)
3. 3D/4D Printing Technologies and Compatibility
3.1. Vat Photopolymerization (SLA/DLP)
- High-Precision Fabrication. Capable of micron-level resolution, enabling accurate replication of complex anatomical structures.
- Rapid Prototyping. Significantly improved manufacturing efficiency compared to extrusion-based bioprinting due to faster layer curing speeds [127].
- Enhanced Biocompatibility. Development using degradable resins (e.g., poly(d,l-lactide)—PDLLA) or novel bioinks (e.g., methacrylated silk fibroin—Sil-MA [126] (Figure 4a), glycidyl methacrylate-modified silk fibroin—Silk-GMA [128], dual-network methacrylated collagen-dimethylphenylphosphinate—CMA-DPPA [129]) significantly improves scaffold cytocompatibility, printability, and functionality [126,128,129].
3.2. Extrusion-Based Printing
3.2.1. Low-Temperature Extrusion
3.2.2. Adaptability of Cell-Laden Bioinks
3.2.3. Porous Structure Design
3.3. Inkjet Printing
3.3.1. Technical Characteristics
3.3.2. Material Compatibility
3.3.3. Application Challenges
3.4. Other Bioprinting Methods
3.5. Functionalized Printing Parameter Optimization
4. Advances in Regenerative Medicine Applications
4.1. Neural Tissue Regeneration
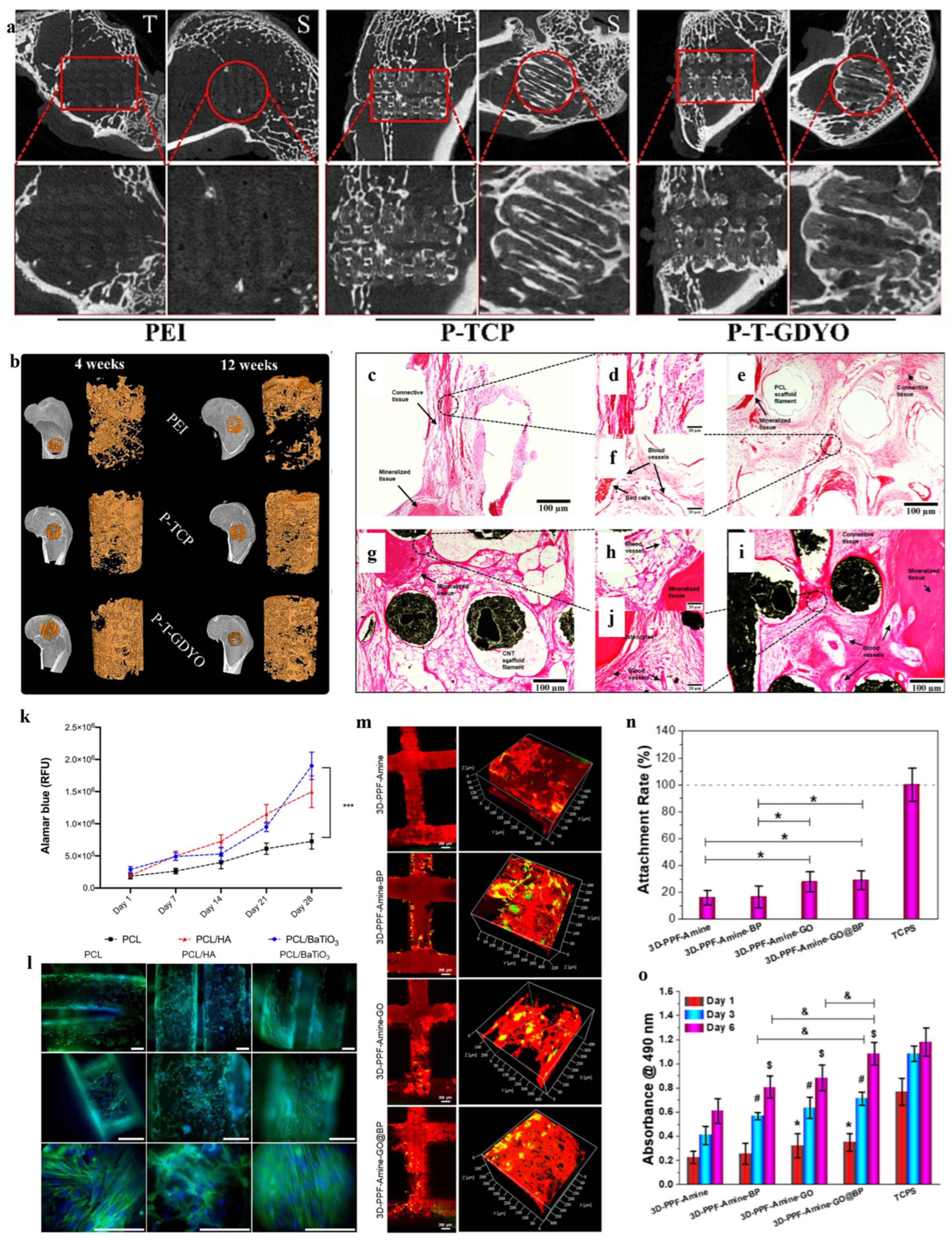
4.2. Cardiac Tissue Engineering
4.3. Bone and Cartilage Repair
5. Current Challenges and Future Directions
5.1. Balancing Material Properties
5.2. Limitations of Printing Technologies
5.3. Barriers to Clinical Translation
5.4. Emerging Research Directions
5.4.1. Four-Dimensional Printed Dynamic Responsive Materials
5.4.2. Multimodal Smart Scaffolds
5.4.3. Organ-on-a-Chip and Microfluidic Integration
6. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sinha, S.; Schreiner, A.J.; Biernaskie, J.; Nickerson, D.; Gabriel, V.A. Treating pain on skin graft donor sites: Review and clinical recommendations. J. Trauma Acute Care Surg. 2017, 83, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yeung, K.W.K. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact. Mater. 2017, 2, 224–247. [Google Scholar] [CrossRef] [PubMed]
- Ajith, A.; Mulloy, L.L.; Musa, M.A.; Bravo-Egana, V.; Horuzsko, D.D.; Gani, I.; Horuzsko, A. Humanized Mouse Model as a Novel Approach in the Assessment of Human Allogeneic Responses in Organ Transplantation. Front. Immunol. 2021, 12, 687715. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Egashira, N.; Masuda, S. Recent Topics on The Mechanisms of Immunosuppressive Therapy-Related Neurotoxicities. Int. J. Mol. Sci. 2019, 20, 3210. [Google Scholar] [CrossRef]
- Goldaracena, N.; Cullen, J.M.; Kim, D.-S.; Ekser, B.; Halazun, K.J. Expanding the donor pool for liver transplantation with marginal donors. Int. J. Surg. 2020, 82, 30–35. [Google Scholar] [CrossRef]
- Silva-López, M.S.; Alcántara-Quintana, L.E. The Era of Biomaterials: Smart Implants? ACS Appl. Bio Mater. 2023, 6, 2982–2994. [Google Scholar] [CrossRef]
- Busscher, H.J.; van der Mei, H.C.; Subbiahdoss, G.; Jutte, P.C.; van den Dungen, J.J.A.M.; Zaat, S.A.J.; Schultz, M.J.; Grainger, D.W. Biomaterial-Associated Infection: Locating the Finish Line in the Race for the Surface. Sci. Transl. Med. 2012, 4, 153rv10. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef]
- Carnicer-Lombarte, A.; Chen, S.-T.; Malliaras, G.G.; Barone, D.G. Foreign Body Reaction to Implanted Biomaterials and Its Impact in Nerve Neuroprosthetics. Front. Bioeng. Biotechnol. 2021, 9, 622524. [Google Scholar] [CrossRef]
- Langer, R.; Vacanti, J.P. Tissue Engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- Sikorski, P. Electroconductive scaffolds for tissue engineering applications. Biomater. Sci. 2020, 8, 5583–5588. [Google Scholar] [CrossRef]
- He, D.; Li, H. Biomaterials affect cell-cell interactions in vitro in tissue engineering. J. Mater. Sci. Technol. 2021, 63, 62–72. [Google Scholar] [CrossRef]
- McNamara, H.M.; Salegame, R.; Tanoury, Z.A.; Xu, H.; Begum, S.; Ortiz, G.; Pourquie, O.; Cohen, A.E. Bioelectrical domain walls in homogeneous tissues. Nat. Phys. 2020, 16, 357–364. [Google Scholar] [CrossRef]
- Kotwal, A.; Schmidt, C.E. Electrical stimulation alters protein adsorption and nerve cell interactions with electrically conducting biomaterials. Biomaterials 2001, 22, 1055–1064. [Google Scholar] [CrossRef]
- Lee, S.J.; Jeong, W.; Atala, A. 3D Bioprinting for Engineered Tissue Constructs and Patient-Specific Models: Current Progress and Prospects in Clinical Applications. Adv. Mater. 2024, 36, 2408032. [Google Scholar] [CrossRef]
- Zhang, Y.; Le Friec, A.; Zhang, Z.; Müller, C.A.; Du, T.; Dong, M.; Liu, Y.; Chen, M. Electroactive biomaterials synergizing with electrostimulation for cardiac tissue regeneration and function-monitoring. Mater. Today 2023, 70, 237–272. [Google Scholar] [CrossRef]
- da Silva, L.P.; Kundu, S.C.; Reis, R.L.; Correlo, V.M. Electric Phenomenon: A Disregarded Tool in Tissue Engineering and Regenerative Medicine. Trends Biotechnol. 2020, 38, 24–49. [Google Scholar] [CrossRef]
- Levin, M.; Pezzulo, G.; Finkelstein, J.M. Endogenous Bioelectric Signaling Networks: Exploiting Voltage Gradients for Control of Growth and Form. Annu. Rev. Biomed. Eng. 2017, 19, 353–387. [Google Scholar] [CrossRef]
- Liu, Z.; Wan, X.; Wang, Z.L.; Li, L. Electroactive Biomaterials and Systems for Cell Fate Determination and Tissue Regeneration: Design and Applications. Adv. Mater. 2021, 33, 2007429. [Google Scholar] [CrossRef]
- Kuzmenko, V.; Karabulut, E.; Pernevik, E.; Enoksson, P.; Gatenholm, P. Tailor-made conductive inks from cellulose nanofibrils for 3D printing of neural guidelines. Carbohydr. Polym. 2018, 189, 22–30. [Google Scholar] [CrossRef]
- Dvir, T.; Timko, B.P.; Brigham, M.D.; Naik, S.R.; Karajanagi, S.S.; Levy, O.; Jin, H.; Parker, K.K.; Langer, R.; Kohane, D.S. Nanowired three-dimensional cardiac patches. Nat. Nanotechnol. 2011, 6, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Vijayavenkataraman, S.; Kannan, S.; Cao, T.; Fuh, J.Y.H.; Sriram, G.; Lu, W.F. 3D-Printed PCL/PPy Conductive Scaffolds as Three-Dimensional Porous Nerve Guide Conduits (NGCs) for Peripheral Nerve Injury Repair. Front. Bioeng. Biotechnol. 2019, 7, 266. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Wang, K.; Liu, X.; Liu, X.; Wang, J.; Suo, M.; Wang, H.; Chen, S.; Chen, X.; Li, Z. Piezoelectric biomaterials for providing electrical stimulation in bone tissue engineering: Barium titanate. J. Orthop. Transl. 2025, 51, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Bassett, C.A.L.; Becker, R.O. Generation of Electric Potentials by Bone in Response to Mechanical Stress. Science 1962, 137, 1063–1064. [Google Scholar] [CrossRef]
- Senkoylu, A.; Cetinkaya, M.; Daldal, I.; Necefov, E.; Eren, A.; Samartzis, D. Personalized Three-Dimensional Printing Pedicle Screw Guide Innovation for the Surgical Management of Patients with Adolescent Idiopathic Scoliosis. World Neurosurg. 2020, 144, e513–e522. [Google Scholar] [CrossRef]
- Zhou, J.; See, C.W.; Sreenivasamurthy, S.; Zhu, D. Customized Additive Manufacturing in Bone Scaffolds—The Gateway to Precise Bone Defect Treatment. Research 2023, 6, 0239. [Google Scholar] [CrossRef]
- Mandrycky, C.; Wang, Z.; Kim, K.; Kim, D.-H. 3D bioprinting for engineering complex tissues. Biotechnol. Adv. 2016, 34, 422–434. [Google Scholar] [CrossRef]
- Jang, J.; Park, H.-J.; Kim, S.-W.; Kim, H.; Park, J.Y.; Na, S.J.; Kim, H.J.; Park, M.N.; Choi, S.H.; Park, S.H.; et al. 3D printed complex tissue construct using stem cell-laden decellularized extracellular matrix bioinks for cardiac repair. Biomaterials 2017, 112, 264–274. [Google Scholar] [CrossRef]
- Xue, W.; Shi, W.; Kong, Y.; Kuss, M.; Duan, B. Anisotropic scaffolds for peripheral nerve and spinal cord regeneration. Bioact. Mater. 2021, 6, 4141–4160. [Google Scholar] [CrossRef]
- Ho, L.; Hsu, S.-H. Cell reprogramming by 3D bioprinting of human fibroblasts in polyurethane hydrogel for fabrication of neural-like constructs. Acta Biomater. 2018, 70, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Hann, S.Y.; Cui, H.; Esworthy, T.; Zhou, X.; Lee, S.-J.; Plesniak, M.W.; Zhang, L.G. Dual 3D printing for vascularized bone tissue regeneration. Acta Biomater. 2021, 123, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Li, Q.; Tian, Z.; Yao, Q.; Zhang, M. Recent advances in 3D bioprinted cartilage-mimicking constructs for applications in tissue engineering. Mater. Today Bio 2023, 23, 100870. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Yuan, X.; Ma, L.; Bi, B.; Zhu, H.; Lei, Z.; Liu, W.; Pu, H.; Jiang, J.; Jiang, X.; et al. Mesenchymal stem cell-loaded thermosensitive hydroxypropyl chitin hydrogel combined with a three-dimensional-printed poly(ε-caprolactone)/nano-hydroxyapatite scaffold to repair bone defects via osteogenesis, angiogenesis and immunomodulation. Theranostics 2020, 10, 725–740. [Google Scholar] [CrossRef]
- Xiao, L.; Ye, M.; Fan, Y.; Liu, G.; He, H.; Xiao, J. Dual-network methacrylated collagen-magnesium hydrogels for enhanced biocompatibility and precision in DLP 3D bioprinting. Int. J. Biol. Macromol. 2025, 320, 145478. [Google Scholar] [CrossRef]
- Tamir, T.S.; Teferi, F.B.; Hua, X.; Leng, J.; Xiong, G.; Shen, Z.; Liu, Q. A review of advances in 3D and 4D bioprinting: Toward mass individualization paradigm. J. Intell. Manuf. 2024, 36, 5217–5246. [Google Scholar] [CrossRef]
- Mishra, A.; Singh, P.K.; Chauhan, N.; Roy, S.; Tiwari, A.; Gupta, S.; Tiwari, A.; Patra, S.; Das, T.R.; Mishra, P.; et al. Emergence of integrated biosensing-enabled digital healthcare devices. Sens. Diagn. 2024, 3, 718–744. [Google Scholar] [CrossRef]
- Park, J.Y.; Jang, J.; Kang, H.-W. 3D Bioprinting and its application to organ-on-a-chip. Microelectron. Eng. 2018, 200, 1–11. [Google Scholar] [CrossRef]
- Zahra, A.; Shayan, G.; Hossein, D.; Katayoun, D.; Seyed Mohammad, A.; Hadi, S. Conducting Polymers for Neural Tissue Engineering; CRC Press: Boca Raton, FL, USA, 2022; Volume 19, pp. 179–192. [Google Scholar]
- Guo, B.; Ma, P.X. Conducting Polymers for Tissue Engineering. Biomacromolecules 2018, 19, 1764–1782. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, C.; Cheng, S.; Li, Y.; Huang, Y.; Cao, X.; Zhang, Z.; Huang, J. 3D Bioprinting of Double-Layer Conductive Skin for Wound Healing. Adv. Healthc. Mater. 2025, 14, 2404388. [Google Scholar] [CrossRef]
- Spencer, A.R.; Shirzaei Sani, E.; Soucy, J.R.; Corbet, C.C.; Primbetova, A.; Koppes, R.A.; Annabi, N. Bioprinting of a Cell-Laden Conductive Hydrogel Composite. ACS Appl. Mater. Interfaces 2019, 11, 30518–30533. [Google Scholar] [CrossRef]
- Tomaskovic-Crook, E.; Zhang, P.; Ahtiainen, A.; Kaisvuo, H.; Lee, C.-Y.; Beirne, S.; Aqrawe, Z.; Svirskis, D.; Hyttinen, J.; Wallace, G.G.; et al. Human Neural Tissues from Neural Stem Cells Using Conductive Biogel and Printed Polymer Microelectrode Arrays for 3D Electrical Stimulation. Adv. Healthc. Mater. 2019, 8, 1900425. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.R.; Zihlmann, C.; Akbari, M.; Assawes, P.; Cheung, L.; Zhang, K.; Manoharan, V.; Zhang, Y.S.; Yüksekkaya, M.; Wan, K.-T.; et al. Reduced Graphene Oxide-GelMA Hybrid Hydrogels as Scaffolds for Cardiac Tissue Engineering. Small 2016, 12, 3677–3689. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.A.; Kryukov, O.; Bandela, A.K.; Muadi, H.; Ashkenasy, N.; Cohen, S.; Marks, R.S. Development of Covalently Functionalized Alginate–Pyrrole and Polypyrrole–Alginate Nanocomposites as 3D Printable Electroconductive Bioinks. Materials 2025, 18, 3120. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.-H.; Niu, C.-M.; Shi, J.-Q.; Wang, Y.-Y.; Yang, Y.-M.; Wang, H.-B. Novel conductive polypyrrole/silk fibroin scaffold for neural tissue repair. Neural Regen. Res. 2018, 13, 1455. [Google Scholar] [CrossRef]
- Zhou, Z.-F.; Zhang, F.; Wang, J.-G.; Chen, Q.-C.; Yang, W.-Z.; He, N.; Jiang, Y.-Y.; Chen, F.; Liu, J.-J. Electrospinning of PELA/PPY Fibrous Conduits: Promoting Peripheral Nerve Regeneration in Rats by Self-Originated Electrical Stimulation. ACS Biomater. Sci. Eng. 2016, 2, 1572–1581. [Google Scholar] [CrossRef]
- Kang, E.T.; Neoh, K.G.; Tan, K.L. Polyaniline: A polymer with many interesting intrinsic redox states. Prog. Polym. Sci. 1998, 23, 277–324. [Google Scholar] [CrossRef]
- Focke, W.W.; Wnek, G.E.; Wei, Y. Influence of oxidation state, pH, and counterion on the conductivity of polyaniline. J. Phys. Chem. 1987, 91, 5813–5818. [Google Scholar] [CrossRef]
- Angelo, M.; Andrea, N.; Giorgio, G.; Riccardo, P.; Dario, P. 3D printing of conductive organic polymers: Challenges and opportunities towards dynamic and electrically responsive materials. Mater. Today Chem. 2022, 26, 101135. [Google Scholar] [CrossRef]
- Sinha, S.; Bhadra, S.; Khastgir, D. Effect of dopant type on the properties of polyaniline. J. Appl. Polym. Sci. 2009, 112, 3135–3140. [Google Scholar] [CrossRef]
- Han, M.; Yildiz, E.; Kaleli, H.N.; Karaz, S.; Eren, G.O.; Dogru-Yuksel, I.B.; Senses, E.; Şahin, A.; Nizamoglu, S. Tissue-Like Optoelectronic Neural Interface Enabled by PEDOT:PSS Hydrogel for Cardiac and Neural Stimulation. Adv. Healthc. Mater. 2022, 11, 2102160. [Google Scholar] [CrossRef]
- Wang, J.; Li, Q.; Li, K.; Sun, X.; Wang, Y.; Zhuang, T.; Yan, J.; Wang, H. Ultra-High Electrical Conductivity in Filler-Free Polymeric Hydrogels Toward Thermoelectrics and Electromagnetic Interference Shielding. Adv. Mater. 2022, 34, 2109904. [Google Scholar] [CrossRef] [PubMed]
- Yuk, H.; Lu, B.; Lin, S.; Qu, K.; Xu, J.; Luo, J.; Zhao, X. 3D printing of conducting polymers. Nat. Commun. 2020, 11, 1604. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.D.; Kim, K.; Shin, M. Recent advances in 3D printable conductive hydrogel inks for neural engineering. Nano Converg. 2023, 10, 41. [Google Scholar] [CrossRef]
- Heo, D.N.; Lee, S.-J.; Timsina, R.; Qiu, X.; Castro, N.J.; Zhang, L.G. Development of 3D printable conductive hydrogel with crystallized PEDOT:PSS for neural tissue engineering. Mater. Sci. Eng. C 2019, 99, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Kamalesh, S.; Tan, P.; Wang, J.; Lee, T.; Kang, E.-T.; Wang, C.-H. Biocompatibility of electroactive polymers in tissues. J. Biomed. Mater. Res. 2000, 52, 467–478. [Google Scholar] [CrossRef]
- Wang, C.H.; Dong, Y.Q.; Sengothi, K.; Tan, K.L.; Kang, E.T. In-vivo tissue response to polyaniline. Synth. Met. 1999, 102, 1313–1314. [Google Scholar] [CrossRef]
- Bidez, P.R., 3rd; Li, S.; Macdiarmid, A.G.; Venancio, E.C.; Wei, Y.; Lelkes, P.I. Polyaniline, an electroactive polymer, supports adhesion and proliferation of cardiac myoblasts. J. Biomater. Sci. Polym. Ed. 2006, 17, 199–212. [Google Scholar] [CrossRef]
- Zhao, Y.; Liang, Y.; Ding, S.; Zhang, K.; Mao, H.-Q.; Yang, Y. Application of conductive PPy/SF composite scaffold and electrical stimulation for neural tissue engineering. Biomaterials 2020, 255, 120164. [Google Scholar] [CrossRef]
- Jakus, A.E.; Secor, E.B.; Rutz, A.L.; Jordan, S.W.; Hersam, M.C.; Shah, R.N. Three-Dimensional Printing of High-Content Graphene Scaffolds for Electronic and Biomedical Applications. ACS Nano 2015, 9, 4636–4648. [Google Scholar] [CrossRef]
- Bon, S.B.; Chiesa, I.; Degli Esposti, M.; Morselli, D.; Fabbri, P.; De Maria, C.; Morabito, A.; Coletta, R.; Calamai, M.; Pavone, F.S.; et al. Carbon Nanotubes/Regenerated Silk Composite as a Three-Dimensional Printable Bio-Adhesive Ink with Self-Powering Properties. ACS Appl. Mater. Interfaces 2021, 13, 21007–21017. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, E.M.; Oliveira, F.J.; Silva, R.F.; Neto, M.A.; Fernandes, M.H.; Amaral, M.; Vallet-Regí, M.; Vila, M. Three-dimensional printed PCL-hydroxyapatite scaffolds filled with CNTs for bone cell growth stimulation. J. Biomed. Mater. Res. B Appl. Biomater. 2016, 104, 1210–1219. [Google Scholar] [CrossRef] [PubMed]
- Rastin, H.; Zhang, B.; Mazinani, A.; Hassan, K.; Bi, J.; Tung, T.T.; Losic, D. 3D bioprinting of cell-laden electroconductive MXene nanocomposite bioinks. Nanoscale 2020, 12, 16069–16080. [Google Scholar] [CrossRef] [PubMed]
- Baei, P.; Jalili-Firoozinezhad, S.; Rajabi-Zeleti, S.; Tafazzoli-Shadpour, M.; Baharvand, H.; Aghdami, N. Electrically conductive gold nanoparticle-chitosan thermosensitive hydrogels for cardiac tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 63, 131–141. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, X.; Kim, H.-J.; Zhang, S.; Zhou, X.; Chen, Y.; Ling, H.; Xue, Y.; Chen, Z.; Qu, M.; et al. Flexible patch with printable and antibacterial conductive hydrogel electrodes for accelerated wound healing. Biomaterials 2022, 285, 121479. [Google Scholar] [CrossRef]
- Criado-Gonzalez, M.; Dominguez-Alfaro, A.; Lopez-Larrea, N.; Alegret, N.; Mecerreyes, D. Additive Manufacturing of Conducting Polymers: Recent Advances, Challenges, and Opportunities. ACS Appl. Polym. Mater. 2021, 3, 2865–2883. [Google Scholar] [CrossRef]
- Boara, G.; Sparpaglione, M. Synthesis of polyanilines with high electrical conductivity. Synth. Met. 1995, 72, 135–140. [Google Scholar] [CrossRef]
- Huang, W.-S.; Humphrey, B.D.; MacDiarmid, A.G. Polyaniline, a novel conducting polymer. Morphology and chemistry of its oxidation and reduction in aqueous electrolytes. J. Chem. Soc. Faraday Trans. 1 Phys. Chem. Condens. Phases 1986, 82, 2385–2400. [Google Scholar] [CrossRef]
- Hsiao, C.-W.; Bai, M.-Y.; Chang, Y.; Chung, M.-F.; Lee, T.-Y.; Wu, C.-T.; Maiti, B.; Liao, Z.-X.; Li, R.-K.; Sung, H.-W. Electrical coupling of isolated cardiomyocyte clusters grown on aligned conductive nanofibrous meshes for their synchronized beating. Biomaterials 2013, 34, 1063–1072. [Google Scholar] [CrossRef]
- Wehrle, B.; Limbach, H.-H.; Mortensen, J.; Heinze, J. Solid-state 15N CPMAS NMR study of the structure of polypyrrole. Synth. Met. 1990, 38, 293–298. [Google Scholar] [CrossRef]
- Vernitskaya, T.Y.V.; Efimov, O.N. Polypyrrole: A conducting polymer, its synthesis, properties and applications. Russ. Chem. Rev. 1997, 66, 443–457. [Google Scholar] [CrossRef]
- Nishio, K.; Fujimoto, M.; Ando, O.; Ono, H.; Murayama, T. Characteristics of polypyrrole chemically synthesized by various oxidizing reagents. J. Appl. Electrochem. 1996, 26, 425–429. [Google Scholar] [CrossRef]
- Subramanian, P.; Clark, N.B.; Spiccia, L.; MacFarlane, D.R.; Winther-Jensen, B.; Forsyth, C. Vapour phase polymerisation of pyrrole induced by iron(III) alkylbenzenesulfonate salt oxidising agents. Synth. Met. 2008, 158, 704–711. [Google Scholar] [CrossRef]
- Wu, Y.; Zou, J.; Tang, K.; Xia, Y.; Wang, X.; Song, L.; Wang, J.; Wang, K.; Wang, Z. From electricity to vitality: The emerging use of piezoelectric materials in tissue regeneration. Burn. Trauma 2024, 12, tkae013. [Google Scholar] [CrossRef]
- Quigley, A.F.; Razal, J.M.; Thompson, B.C.; Moulton, S.E.; Kita, M.; Kennedy, E.L.; Clark, G.M.; Wallace, G.G.; Kapsa, R.M.I. A Conducting-Polymer Platform with Biodegradable Fibers for Stimulation and Guidance of Axonal Growth. Adv. Mater. 2009, 21, 4393–4397. [Google Scholar] [CrossRef]
- Hong, X.; Liu, Y.; Li, Y.; Wang, X.; Fu, J.; Wang, X. Application Progress of Polyaniline, Polypyrrole and Polythiophene in Lithium-Sulfur Batteries. Polymers 2020, 12, 331. [Google Scholar] [CrossRef]
- Huang, Y.; Li, H.; Wang, Z.; Zhu, M.; Pei, Z.; Xue, Q.; Huang, Y.; Zhi, C. Nanostructured Polypyrrole as a flexible electrode material of supercapacitor. Nano Energy 2016, 22, 422–438. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, Y.; Liu, Z.; Ma, C.; Yan, H.; Xu, N.; Gang, F.; Wang, X.; Zhao, L.; Sun, X. Three-Dimensional Printing and Injectable Conductive Hydrogels for Tissue Engineering Application. Tissue Eng. Part B Rev. 2019, 25, 398–411. [Google Scholar] [CrossRef]
- Meng, J.; Song, J.; Zhang, X.; Wang, J.; Li, S. A highly stretchable carbon nanotube/reduced graphene oxide/poly(dimethylsiloxane) composite with high thermal conductivity as a flexible strain sensor. Compos. Commun. 2023, 42, 101693. [Google Scholar] [CrossRef]
- Ali, A.; Rahimian Koloor, S.S.; Alshehri, A.H.; Arockiarajan, A. Carbon nanotube characteristics and enhancement effects on the mechanical features of polymer-based materials and structures—A review. J. Mater. Res. Technol. 2023, 24, 6495–6521. [Google Scholar] [CrossRef]
- Zhao, X.; Ren, X.; Chen, Z.; Ge, C. Enhancement of mechanical and electrical properties of copper matrix composites by different types of carbon nanotubes. Mater. Charact. 2024, 218, 114575. [Google Scholar] [CrossRef]
- Charlier, J.-C.; Blase, X.; Roche, S. Electronic and transport properties of nanotubes. Rev. Mod. Phys. 2007, 79, 677–732. [Google Scholar] [CrossRef]
- Yadid, M.; Feiner, R.; Dvir, T. Gold Nanoparticle-Integrated Scaffolds for Tissue Engineering and Regenerative Medicine. Nano Lett. 2019, 19, 2198–2206. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Jang, C.H.; Kim, G.H. A Myoblast-Laden Collagen Bioink with Fully Aligned Au Nanowires for Muscle-Tissue Regeneration. Nano Lett. 2019, 19, 8612–8620. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Jiang, J.; Yuan, C.; Zhang, X.; Gu, L.; Wang, X. Omnidirectional anisotropic embedded 3D bioprinting. Mater. Today Bio 2024, 27, 101160. [Google Scholar] [CrossRef]
- Boularaoui, S.; Shanti, A.; Lanotte, M.; Luo, S.; Bawazir, S.; Lee, S.; Christoforou, N.; Khan, K.A.; Stefanini, C. Nanocomposite Conductive Bioinks Based on Low-Concentration GelMA and MXene Nanosheets/Gold Nanoparticles Providing Enhanced Printability of Functional Skeletal Muscle Tissues. ACS Biomater. Sci. Eng. 2021, 7, 5810–5822. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Walimbe, P.; Chaudhari, M. State-of-the-art advancements in studies and applications of graphene: A comprehensive review. Mater. Today Sustain. 2019, 6, 100026. [Google Scholar] [CrossRef]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the Elastic Properties and Intrinsic Strength of Monolayer Graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Li, J.; Liu, X.; Crook, J.M.; Wallace, G.G. 3D Printing of Cytocompatible Graphene/Alginate Scaffolds for Mimetic Tissue Constructs. Front. Bioeng. Biotechnol. 2020, 8, 824. [Google Scholar] [CrossRef]
- Silva, M.; Gomes, S.; Correia, C.; Peixoto, D.; Vinhas, A.; Rodrigues, M.T.; Gomes, M.E.; Covas, J.A.; Paiva, M.C.; Alves, N.M. Biocompatible 3D-Printed Tendon/Ligament Scaffolds Based on Polylactic Acid/Graphite Nanoplatelet Composites. Nanomaterials 2023, 13, 2518. [Google Scholar] [CrossRef] [PubMed]
- Yao, S.; Yang, Y.; Li, C.; Yang, K.; Song, X.; Li, C.; Cao, Z.; Zhao, H.; Yu, X.; Wang, X.; et al. Axon-like aligned conductive CNT/GelMA hydrogel fibers combined with electrical stimulation for spinal cord injury recovery. Bioact. Mater. 2024, 35, 534–548. [Google Scholar] [CrossRef] [PubMed]
- Nanot, S.; Thompson, N.A.; Kim, J.-H.; Wang, X.; Rice, W.D.; Hároz, E.H.; Ganesan, Y.; Pint, C.L.; Kono, J. Single-Walled Carbon Nanotubes. In Springer Handbook of Nanomaterials; Vajtai, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 105–146. [Google Scholar]
- Zare, Y. Modeling of tensile modulus in polymer/carbon nanotubes (CNT) nanocomposites. Synth. Met. 2015, 202, 68–72. [Google Scholar] [CrossRef]
- Fan, L.; Cai, X.; Wang, H.; Ye, J.; Hong, Y.; Ying, J. Toughening two-dimensional hybrid materials by integrating carbon nanotubes. Surf. Interf. 2023, 36, 102559. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, D. Separation methods of single-walled carbon nanotubes. Chin. Sci. Bull. 2014, 59, 3240–3263. [Google Scholar] [CrossRef]
- Smart, S.K.; Cassady, A.I.; Lu, G.Q.; Martin, D.J. The biocompatibility of carbon nanotubes. Carbon 2006, 44, 1034–1047. [Google Scholar] [CrossRef]
- Lee, S.-J.; Zhu, W.; Nowicki, M.; Lee, G.; Heo, D.N.; Kim, J.; Zuo, Y.Y.; Zhang, L.G. 3D printing nano conductive multi-walled carbon nanotube scaffolds for nerve regeneration. J. Neural Eng. 2018, 15, 016018. [Google Scholar] [CrossRef]
- Zips, S.; Grob, L.; Rinklin, P.; Terkan, K.; Adly, N.Y.; Weiß, L.J.K.; Mayer, D.; Wolfrum, B. Fully Printed μ-Needle Electrode Array from Conductive Polymer Ink for Bioelectronic Applications. ACS Appl. Mater. Interfaces 2019, 11, 32778–32786. [Google Scholar] [CrossRef]
- Liu, X.; George, M.N.; Park, S.; Miller, A.L., II; Gaihre, B.; Li, L.; Waletzki, B.E.; Terzic, A.; Yaszemski, M.J.; Lu, L. 3D-printed scaffolds with carbon nanotubes for bone tissue engineering: Fast and homogeneous one-step functionalization. Acta Biomater. 2020, 111, 129–140. [Google Scholar] [CrossRef]
- Cho, K.G.; Kwon, Y.K.; Jang, S.S.; Seol, K.H.; Park, J.H.; Hong, K.; Lee, K.H. Printable carbon nanotube-based elastic conductors for fully-printed sub-1 V stretchable electrolyte-gated transistors and inverters. J. Mater. Chem. C 2020, 8, 3639–3645. [Google Scholar] [CrossRef]
- Uřičář, J.; Veselý, P.; Pilnaj, D.; Sedláček, J.; Brožek, J. 3D printing conductive pastes based on polystyrene/graphite composite. In Proceedings of the 2024 47th International Spring Seminar on Electronics Technology (ISSE), Prague, Czech Republic, 15–19 May 2024; pp. 1–4. [Google Scholar]
- Venditti, I. Engineered Gold-Based Nanomaterials: Morphologies and Functionalities in Biomedical Applications. A Mini Review. Bioengineering 2019, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Husni, P.; Shin, Y.; Jeon, H.; Lee, E.S.; Youn, Y.S.; Poon, C.-D.; Lim, C.; Oh, K.T. Development and characterization of pH-responsive nanocarriers for chemo-photothermal combination therapy of acidic tumors. J. Control. Release 2023, 359, 52–68. [Google Scholar] [CrossRef] [PubMed]
- Sudhakar, S.; Santhosh, P.B. Chapter Six—Gold Nanomaterials: Recent Advances in Cancer Theranostics. In Advances in Biomembranes and Lipid Self-Assembly; Iglič, A., Garcia-Sáez, A., Rappolt, M., Eds.; Academic Press: Cambridge, MA, USA, 2017; Volume 25, pp. 161–180. [Google Scholar]
- Fathi-Achachelouei, M.; Knopf-Marques, H.; Ribeiro da Silva, C.E.; Barthès, J.; Bat, E.; Tezcaner, A.; Vrana, N.E. Use of Nanoparticles in Tissue Engineering and Regenerative Medicine. Front. Bioeng. Biotechnol. 2019, 7, 113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Lu, Q.; Wang, F.; Xiao, Z.; He, L.; He, D.; Deng, L. Gold–Platinum Nanodots with High-Peroxidase-like Activity and Photothermal Conversion Efficiency for Antibacterial Therapy. ACS Appl. Mater. Interfaces 2021, 13, 37535–37544. [Google Scholar] [CrossRef]
- Wu, S.; Li, A.; Zhao, X.; Zhang, C.; Yu, B.; Zhao, N.; Xu, F.-J. Silica-Coated Gold–Silver Nanocages as Photothermal Antibacterial Agents for Combined Anti-Infective Therapy. ACS Appl. Mater. Interfaces 2019, 11, 17177–17183. [Google Scholar] [CrossRef]
- Sesena-Rubfiaro, A.; Prajapati, N.J.; Lou, L.; Ghimire, G.; Agarwal, A.; He, J. Improving the development of human engineered cardiac tissue by gold nanorods embedded extracellular matrix for long-term viability. Nanoscale 2024, 16, 2983–2992. [Google Scholar] [CrossRef]
- Huang, W.-J.; Wang, J. Development of 3D-Printed, Biodegradable, Conductive PGSA Composites for Nerve Tissue Regeneration. Macromol. Biosci. 2023, 23, 2200470. [Google Scholar] [CrossRef]
- Campoccia, D.; Montanaro, L.; Arciola, C.R. A review of the clinical implications of anti-infective biomaterials and infection-resistant surfaces. Biomaterials 2013, 34, 8018–8029. [Google Scholar] [CrossRef]
- Sitnikova, N.A.; Solovieva, A.O.; Permyakova, E.S.; Sheveyko, A.N.; Shtansky, D.V.; Manakhov, A.M. Silver Ions Incorporation into Nanofibers for Enhanced hMSC Viability. Chemistry 2022, 4, 931–939. [Google Scholar] [CrossRef]
- Chota, A.; Abrahamse, H.; George, B.P. Green synthesis and characterization of AgNPs, liposomal loaded AgNPs and ZnPcS4 photosensitizer for enhanced photodynamic therapy effects in MCF-7 breast cancer cells. Photodiagn. Photodyn. Ther. 2024, 48, 104252. [Google Scholar] [CrossRef]
- Nandhini, J.; Karthikeyan, E.; Rajeshkumar, S. Nanomaterials for wound healing: Current status and futuristic frontier. Biomed. Technol. 2024, 6, 26–45. [Google Scholar] [CrossRef]
- Zhang, B.; Yan, W.; Zhu, Y.; Yang, W.; Le, W.; Chen, B.; Zhu, R.; Cheng, L. Nanomaterials in Neural-Stem-Cell-Mediated Regenerative Medicine: Imaging and Treatment of Neurological Diseases. Adv. Mater. 2018, 30, e1705694. [Google Scholar] [CrossRef]
- Damle, A.; Sundaresan, R.; Rajwade, J.M.; Srivastava, P.; Naik, A. A concise review on implications of silver nanoparticles in bone tissue engineering. Biomater. Adv. 2022, 141, 213099. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.; Zhao, D.; Zhang, C.; Liu, K.; He, Y.; Guan, F.; Yao, M. Nanocomposite multifunctional hyaluronic acid hydrogel with photothermal antibacterial and antioxidant properties for infected wound healing. Int. J. Biol. Macromol. 2023, 226, 870–884. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Feng, Z.; Lyu, Y.; Yang, J.; Lin, L.; Bai, H.; Li, Y.; Feng, Y.; Chen, Y. Electroactive injectable hydrogel based on oxidized sodium alginate and carboxymethyl chitosan for wound healing. Int. J. Biol. Macromol. 2023, 230, 123231. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Chu, L.; Zhuang, Y.; Qi, C.; Meng, S.; Liu, Z.; Kong, T. Multi-Material Digital Light Processing (DLP) Bioprinting of Heterogeneous Hydrogel Constructs with Perfusable Networks. Adv. Funct. Mater. 2024, 34, 2316456. [Google Scholar] [CrossRef]
- Melchels, F.P.W.; Feijen, J.; Grijpma, D.W. A review on stereolithography and its applications in biomedical engineering. Biomaterials 2010, 31, 6121–6130. [Google Scholar] [CrossRef]
- Yu, C.; Schimelman, J.; Wang, P.; Miller, K.L.; Ma, X.; You, S.; Guan, J.; Sun, B.; Zhu, W.; Chen, S. Photopolymerizable Biomaterials and Light-Based 3D Printing Strategies for Biomedical Applications. Chem. Rev. 2020, 120, 10695–10743. [Google Scholar] [CrossRef]
- Xu, X.; Awad, A.; Robles-Martinez, P.; Gaisford, S.; Goyanes, A.; Basit, A.W. Vat photopolymerization 3D printing for advanced drug delivery and medical device applications. J. Control. Release 2021, 329, 743–757. [Google Scholar] [CrossRef]
- Kuang, X.; Wu, J.; Chen, K.; Zhao, Z.; Ding, Z.; Hu, F.; Fang, D.; Qi, H.J. Grayscale digital light processing 3D printing for highly functionally graded materials. Sci. Adv. 2019, 5, eaav5790. [Google Scholar] [CrossRef]
- Pawar, A.A.; Saada, G.; Cooperstein, I.; Larush, L.; Jackman, J.A.; Tabaei, S.R.; Cho, N.-J.; Magdassi, S. High-performance 3D printing of hydrogels by water-dispersible photoinitiator nanoparticles. Sci. Adv. 2016, 2, e1501381. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Yeon, Y.K.; Lee, J.M.; Chao, J.R.; Lee, Y.J.; Seo, Y.B.; Sultan, M.T.; Lee, O.J.; Lee, J.S.; Yoon, S.-I.; et al. Precisely printable and biocompatible silk fibroin bioink for digital light processing 3D printing. Nat. Commun. 2018, 9, 1620. [Google Scholar] [CrossRef] [PubMed]
- Bernal, P.N.; Delrot, P.; Loterie, D.; Li, Y.; Malda, J.; Moser, C.; Levato, R. Volumetric Bioprinting of Complex Living-Tissue Constructs within Seconds. Adv. Mater. 2019, 31, e1904209. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Seo, Y.B.; Kim, D.Y.; Lee, J.S.; Lee, Y.J.; Lee, H.; Ajiteru, O.; Sultan, M.T.; Lee, O.J.; Kim, S.H.; et al. Digital light processing 3D printed silk fibroin hydrogel for cartilage tissue engineering. Biomaterials 2020, 232, 119679. [Google Scholar] [CrossRef]
- Xiao, L.; Ye, M.; Fan, Y.; Liu, G.; He, H.; Xiao, J. Dual-Cross-linked Methylacrylated Collagen-DPPA Bioinks for Precision DLP Bioprinting and Accelerated Skin Wound Healing. Biomacromolecules 2025, 26, 4308–4321. [Google Scholar] [CrossRef]
- Fantino, E.; Roppolo, I.; Zhang, D.; Xiao, J.; Chiappone, A.; Castellino, M.; Guo, Q.; Pirri, C.F.; Yang, J. 3D Printing/Interfacial Polymerization Coupling for the Fabrication of Conductive Hydrogel. Macromol. Mater. Eng. 2018, 303, 1700356. [Google Scholar] [CrossRef]
- Anindita, S.N.; Conti, R.; Zauchner, D.; Paunović, N.; Qiu, W.; Buzhor, M.G.; Krivitsky, A.; Luo, Z.; Müller, R.; Grützmacher, H.; et al. Tough PEG-only hydrogels with complex 3D structure enabled by digital light processing of “all-PEG” resins. Aggregate 2023, 4, e368. [Google Scholar] [CrossRef]
- Tao, J.; Zhu, S.; Liao, X.; Wang, Y.; Zhou, N.; Li, Z.; Wan, H.; Tang, Y.; Yang, S.; Du, T.; et al. DLP-based bioprinting of void-forming hydrogels for enhanced stem-cell-mediated bone regeneration. Mater. Today Bio 2022, 17, 100487. [Google Scholar] [CrossRef]
- Liu, D.; Jiang, P.; Wang, Y.; Lu, Y.; Wu, J.; Xu, X.; Ji, Z.; Sun, C.; Wang, X.; Liu, W. Engineering Tridimensional Hydrogel Tissue and Organ Phantoms with Tunable Springiness. Adv. Funct. Mater. 2023, 33, 2214885. [Google Scholar] [CrossRef]
- Garciamendez-Mijares, C.E.; Aguilar, F.J.; Hernandez, P.; Kuang, X.; Gonzalez, M.; Ortiz, V.; Riesgo, R.A.; Ruiz, D.S.R.; Rivera, V.A.M.; Rodriguez, J.C.; et al. Design considerations for digital light processing bioprinters. Appl. Phys. Rev. 2024, 11, 031314. [Google Scholar] [CrossRef]
- Lu, Z.; Gao, W.; Liu, F.; Cui, J.; Feng, S.; Liang, C.; Guo, Y.; Wang, Z.; Mao, Z.; Zhang, B. Vat photopolymerization based digital light processing 3D printing hydrogels in biomedical fields: Key parameters and perspective. Addit. Manuf. 2024, 94, 104443. [Google Scholar] [CrossRef]
- Bagheri, A.; Jin, J. Photopolymerization in 3D Printing. ACS Appl. Polym. Mater. 2019, 1, 593–611. [Google Scholar] [CrossRef]
- Bao, Y.; Paunović, N.; Leroux, J.-C. Challenges and Opportunities in 3D Printing of Biodegradable Medical Devices by Emerging Photopolymerization Techniques. Adv. Funct. Mater. 2022, 32, 2109864. [Google Scholar] [CrossRef]
- Li, H.; Dai, J.; Wang, Z.; Zheng, H.; Li, W.; Wang, M.; Cheng, F. Front Cover: Digital light processing (DLP)-based (bio)printing strategies for tissue modeling and regeneration. Aggregate 2023, 4, e270. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, X.; Gu, Z. Photoresist Development for 3D Printing of Conductive Microstructures via Two-Photon Polymerization. Adv. Mater. 2024, 36, 2409326. [Google Scholar] [CrossRef]
- Zhou, J.; Vijayavenkataraman, S. 3D-printable conductive materials for tissue engineering and biomedical applications. Bioprinting 2021, 24, e00166. [Google Scholar] [CrossRef]
- Liu, X.; George, M.N.; Li, L.; Gamble, D.; Miller, A.L., II; Gaihre, B.; Waletzki, B.E.; Lu, L. Injectable Electrical Conductive and Phosphate Releasing Gel with Two-Dimensional Black Phosphorus and Carbon Nanotubes for Bone Tissue Engineering. ACS Biomater. Sci. Eng. 2020, 6, 4653–4665. [Google Scholar] [CrossRef]
- Patel, P.; Dhal, K.; Gupta, R.; Tappa, K.; Rybicki, F.J.; Ravi, P. Medical 3D Printing Using Desktop Inverted Vat Photopolymerization: Background, Clinical Applications, and Challenges. Bioengineering 2023, 10, 782. [Google Scholar] [CrossRef]
- Hinton, T.J.; Jallerat, Q.; Palchesko, R.N.; Park, J.H.; Grodzicki, M.S.; Shue, H.-J.; Ramadan, M.H.; Hudson, A.R.; Feinberg, A.W. Three-dimensional printing of complex biological structures by freeform reversible embedding of suspended hydrogels. Sci. Adv. 2015, 1, e1500758. [Google Scholar] [CrossRef]
- Naghieh, S.; Chen, X. Printability–A key issue in extrusion-based bioprinting. J. Pharm. Anal. 2021, 11, 564–579. [Google Scholar] [CrossRef]
- Lian, M.; Sun, B.; Han, Y.; Yu, B.; Xin, W.; Xu, R.; Ni, B.; Jiang, W.; Hao, Y.; Zhang, X.; et al. A low-temperature-printed hierarchical porous sponge-like scaffold that promotes cell-material interaction and modulates paracrine activity of MSCs for vascularized bone regeneration. Biomaterials 2021, 274, 120841. [Google Scholar] [CrossRef] [PubMed]
- Skardal, A. Chapter 1—Bioprinting Essentials of Cell and Protein Viability. In Essentials of 3D Biofabrication and Translation; Atala, A., Yoo, J.J., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 1–17. [Google Scholar]
- Yue, Z.; Liu, X.; Coates, P.T.; Wallace, G.G. Advances in printing biomaterials and living cells: Implications for islet cell transplantation. Curr. Opin. Organ Transplant. 2016, 21, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.V.; Skardal, A.; Atala, A. Evaluation of hydrogels for bio-printing applications. J. Biomed. Mater. Res. Part A 2013, 101, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Panwar, A.; Tan, L.P. Current Status of Bioinks for Micro-Extrusion-Based 3D Bioprinting. Molecules 2016, 21, 685. [Google Scholar] [CrossRef]
- Lai, Y.; Li, Y.; Cao, H.; Long, J.; Wang, X.; Li, L.; Li, C.; Jia, Q.; Teng, B.; Tang, T.; et al. Osteogenic magnesium incorporated into PLGA/TCP porous scaffold by 3D printing for repairing challenging bone defect. Biomaterials 2019, 197, 207–219. [Google Scholar] [CrossRef]
- Gao, G.; Kim, B.S.; Jang, J.; Cho, D.-W. Recent Strategies in Extrusion-Based Three-Dimensional Cell Printing toward Organ Biofabrication. ACS Biomater. Sci. Eng. 2019, 5, 1150–1169. [Google Scholar] [CrossRef]
- Billiet, T.; Gevaert, E.; De Schryver, T.; Cornelissen, M.; Dubruel, P. The 3D printing of gelatin methacrylamide cell-laden tissue-engineered constructs with high cell viability. Biomaterials 2014, 35, 49–62. [Google Scholar] [CrossRef]
- Ouyang, L.; Yao, R.; Zhao, Y.; Sun, W. Effect of bioink properties on printability and cell viability for 3D bioplotting of embryonic stem cells. Biofabrication 2016, 8, 035020. [Google Scholar] [CrossRef]
- Hölzl, K.; Lin, S.; Tytgat, L.; Van Vlierberghe, S.; Gu, L.; Ovsianikov, A. Bioink properties before, during and after 3D bioprinting. Biofabrication 2016, 8, 032002. [Google Scholar] [CrossRef]
- Temirel, M.; Dabbagh, S.R.; Tasoglu, S. Shape Fidelity Evaluation of Alginate-Based Hydrogels through Extrusion-Based Bioprinting. J. Funct. Biomater. 2022, 13, 225. [Google Scholar] [CrossRef]
- Zhu, K.; Shin, S.R.; van Kempen, T.; Li, Y.-C.; Ponraj, V.; Nasajpour, A.; Mandla, S.; Hu, N.; Liu, X.; Leijten, J.; et al. Gold Nanocomposite Bioink for Printing 3D Cardiac Constructs. Adv. Funct. Mater. 2017, 27, 1605352. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Vyas, C.; Roberts, I.; Poutrel, Q.-A.; Chiang, W.-H.; Blaker, J.J.; Huang, Z.; Bártolo, P. Fabrication and characterisation of 3D printed MWCNT composite porous scaffolds for bone regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 98, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Mapili, G.; Suhali, G.; Chen, S.; Roy, K. A digital micro-mirror device-based system for the microfabrication of complex, spatially patterned tissue engineering scaffolds. J. Biomed. Mater. Res. Part A 2006, 77A, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.A.; Pfeil, A.; Alleyne, A.G.; Johnson, A.J.W. Process monitoring and control strategies in extrusion-based bioprinting to fabricate spatially graded structures. Bioprinting 2021, 21, e00126. [Google Scholar] [CrossRef]
- Mironov, A.V.; Grigoryev, A.M.; Krotova, L.I.; Skaletsky, N.N.; Popov, V.K.; Sevastianov, V.I. 3D printing of PLGA scaffolds for tissue engineering. J. Biomed. Mater. Res. Part A 2017, 105, 104–109. [Google Scholar] [CrossRef]
- Qu, H.; Han, Z.; Chen, Z.; Tang, L.; Gao, C.; Liu, K.; Pan, H.; Fu, H.; Ruan, C. Fractal Design Boosts Extrusion-Based 3D Printing of Bone-Mimicking Radial-Gradient Scaffolds. Research 2021, 2021, 9892689. [Google Scholar] [CrossRef]
- Li, X.; Liu, B.; Pei, B.; Chen, J.; Zhou, D.; Peng, J.; Zhang, X.; Jia, W.; Xu, T. Inkjet Bioprinting of Biomaterials. Chem. Rev. 2020, 120, 10793–10833. [Google Scholar] [CrossRef]
- Gonzalez, G.; Roppolo, I.; Pirri, C.F.; Chiappone, A. Current and emerging trends in polymeric 3D printed microfluidic devices. Addit. Manuf. 2022, 55, 102867. [Google Scholar] [CrossRef]
- Negro, A.; Cherbuin, T.; Lutolf, M.P. 3D Inkjet Printing of Complex, Cell-Laden Hydrogel Structures. Sci. Rep. 2018, 8, 17099. [Google Scholar] [CrossRef]
- Lee, D.K.; Sin, K.S.; Shin, C.; Kim, J.-H.; Hwang, K.-T.; Kim, U.-S.; Nahm, S.; Han, K.-S. Fabrication of 3D structure with heterogeneous compositions using inkjet printing process. Mater. Today Commun. 2023, 35, 105753. [Google Scholar] [CrossRef]
- Xu, T.; Zhao, W.; Zhu, J.-M.; Albanna, M.Z.; Yoo, J.J.; Atala, A. Complex heterogeneous tissue constructs containing multiple cell types prepared by inkjet printing technology. Biomaterials 2013, 34, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Li, J.; Pan, L.; Shi, Y. Inkjet printing for flexible and wearable electronics. APL Mater. 2020, 8, 120705. [Google Scholar] [CrossRef]
- Lee, Y.; Park, J.A.; Tuladhar, T.; Jung, S. Sonochemical Degradation of Gelatin Methacryloyl to Control Viscoelasticity for Inkjet Bioprinting. Macromol. Biosci. 2023, 23, 2200509. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, H.; Mo, X.; Wang, H. Reduced Graphene Oxide-Encapsulated Microfiber Patterns Enable Controllable Formation of Neuronal-Like Networks. Adv. Mater. 2020, 32, 2004555. [Google Scholar] [CrossRef]
- Qing, H.; Ji, Y.; Li, W.; Zhao, G.; Yang, Q.; Zhang, X.; Luo, Z.; Lu, T.J.; Jin, G.; Xu, F. Microfluidic Printing of Three-Dimensional Graphene Electroactive Microfibrous Scaffolds. ACS Appl. Mater. Interfaces 2020, 12, 2049–2058. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Zhang, S.; Thaharah, S.; Sriram, G.; Lu, W.F.; Fuh, J.Y. Electrohydrodynamic Jet 3D Printed Nerve Guide Conduits (NGCs) for Peripheral Nerve Injury Repair. Polymers 2018, 10, 753. [Google Scholar] [CrossRef]
- Cao, Y.; Tan, J.; Zhao, H.; Deng, T.; Hu, Y.; Zeng, J.; Li, J.; Cheng, Y.; Tang, J.; Hu, Z.; et al. Bead-jet printing enabled sparse mesenchymal stem cell patterning augments skeletal muscle and hair follicle regeneration. Nat. Commun. 2022, 13, 7463. [Google Scholar] [CrossRef]
- Guo, Y.; Patanwala, H.S.; Bognet, B.; Ma, A.W.K. Inkjet and inkjet-based 3D printing: Connecting fluid properties and printing performance. Rapid Prototyp. J. 2017, 23, 562–576. [Google Scholar] [CrossRef]
- Zhao, D.; Xu, H.; Ye, Z.; Yan, J.; Yang, H.; Wang, Y.; Yin, J. Sequential-crosslinking facilitated droplet-droplet collision inkjet 3D printing of soft biomaterials. Addit. Manuf. 2025, 106, 104809. [Google Scholar] [CrossRef]
- Mu, X.; Agostinacchio, F.; Xiang, N.; Pei, Y.; Khan, Y.; Guo, C.; Cebe, P.; Motta, A.; Kaplan, D.L. Recent advances in 3D printing with protein-based inks. Prog. Polym. Sci. 2021, 115, 101375. [Google Scholar] [CrossRef]
- Saunders, R.E.; Derby, B. Inkjet printing biomaterials for tissue engineering: Bioprinting. Int. Mater. Rev. 2014, 59, 430–448. [Google Scholar] [CrossRef]
- Genova, T.; Roato, I.; Carossa, M.; Motta, C.; Cavagnetto, D.; Mussano, F. Advances on Bone Substitutes through 3D Bioprinting. Int. J. Mol. Sci. 2020, 21, 7021. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Boland, T. Human microvasculature fabrication using thermal inkjet printing technology. Biomaterials 2009, 30, 6221–6227. [Google Scholar] [CrossRef] [PubMed]
- Bisht, G.S.; Canton, G.; Mirsepassi, A.; Kulinsky, L.; Oh, S.; Dunn-Rankin, D.; Madou, M.J. Controlled Continuous Patterning of Polymeric Nanofibers on Three-Dimensional Substrates Using Low-Voltage Near-Field Electrospinning. Nano Lett. 2011, 11, 1831–1837. [Google Scholar] [CrossRef]
- Sun, D.; Chang, C.; Li, S.; Lin, L. Near-field electrospinning. Nano Lett. 2006, 6, 839–842. [Google Scholar] [CrossRef]
- He, X.-X.; Zheng, J.; Yu, G.-F.; You, M.-H.; Yu, M.; Ning, X.; Long, Y.-Z. Near-Field Electrospinning: Progress and Applications. J. Phys. Chem. C 2017, 121, 8663–8678. [Google Scholar] [CrossRef]
- Qiu, Z.; Zhu, H.; Wang, Y.; Kasimu, A.; Li, D.; He, J. Functionalized alginate-based bioinks for microscale electrohydrodynamic bioprinting of living tissue constructs with improved cellular spreading and alignment. Bio-Des. Manuf. 2023, 6, 136–149. [Google Scholar] [CrossRef]
- Jakus, A.E.; Shah, R.N. Multi and mixed 3D-printing of graphene-hydroxyapatite hybrid materials for complex tissue engineering. J. Biomed. Mater. Res. Part A 2017, 105, 274–283. [Google Scholar] [CrossRef]
- Khan, S.B.; Chen, S.; Sun, X. Advancements in polymer nanocomposite manufacturing: Revolutionizing medical breakthroughs via additive manufacturing. Polym. Bull. 2024, 81, 9465–9517. [Google Scholar] [CrossRef]
- Rayhan, T.; Islam, A.; Khan, M.; Hasan, A.; Mobarak, H.; Rimon, I.H.; Hossain, N. Advances in additive manufacturing of nanocomposite materials fabrications and applications. Eur. Polym. J. 2024, 220, 113406. [Google Scholar] [CrossRef]
- Han, K.; Mao, M.; Fu, L.; Zhang, Y.; Kang, Y.; Li, D.; He, J. Multimaterial Printing of Serpentine Microarchitectures with Synergistic Mechanical/Piezoelectric Stimulation for Enhanced Cardiac-Specific Functional Regeneration. Small 2024, 20, 2401561. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Gimenez-Camino, N.; Xiao, M.; Bi, S.; DiVito, K.A. Technological advances in three-dimensional skin tissue engineering. Rev. Adv. Mater. Sci. 2023, 62, 20220289. [Google Scholar] [CrossRef]
- Lee, J., Jr.; Ng, H.Y.; Lin, Y.-H.; Liu, E.-W.; Lin, T.-J.; Chiu, H.-T.; Ho, X.-R.; Yang, H.-A.; Shie, M.-Y. The 3D printed conductive grooved topography hydrogel combined with electrical stimulation for synergistically enhancing wound healing of dermal fibroblast cells. Biomater. Adv. 2022, 142, 213132. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Sun, M.; Lu, X.; Xu, K.; Yu, M.; Yang, H.; Yin, J. A 3D printable gelatin methacryloyl/chitosan hydrogel assembled with conductive PEDOT for neural tissue engineering. Compos. Part B Eng. 2024, 273, 111241. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, H.; Wang, Y.; Xu, C.; Esworthy, T.J.; Hann, S.Y.; Boehm, M.; Shen, Y.-L.; Mei, D.; Zhang, L.G. 4D Printed Cardiac Construct with Aligned Myofibers and Adjustable Curvature for Myocardial Regeneration. ACS Appl. Mater. Interfaces 2021, 13, 12746–12758. [Google Scholar] [CrossRef]
- Chai, X.; Lou, Y.; Nie, L.; Shavandi, A.; Yunusov, K.E.; Sun, Y.; Jiang, G. A three-dimensional printable conductive composite dressing for accelerating wound healing under electrical stimulation. Colloids Surf. B Biointerfaces 2025, 245, 114264. [Google Scholar] [CrossRef]
- Dixon, D.T.; Landree, E.N.; Gomillion, C.T. 3D-Printed Demineralized Bone Matrix-Based Conductive Scaffolds Combined with Electrical Stimulation for Bone Tissue Engineering Applications. ACS Appl. Bio Mater. 2024, 7, 4366–4378. [Google Scholar] [CrossRef]
- Serafin, A.; Culebras, M.; Oliveira, J.M.; Koffler, J.; Collins, M.N. 3D printable electroconductive gelatin-hyaluronic acid materials containing polypyrrole nanoparticles for electroactive tissue engineering. Adv. Compos. Hybrid Mater. 2023, 6, 109. [Google Scholar] [CrossRef]
- Distler, T.; Polley, C.; Shi, F.; Schneidereit, D.; Ashton, M.D.; Friedrich, O.; Kolb, J.F.; Hardy, J.G.; Detsch, R.; Seitz, H.; et al. Electrically Conductive and 3D-Printable Oxidized Alginate-Gelatin Polypyrrole:PSS Hydrogels for Tissue Engineering. Adv. Healthc. Mater. 2021, 10, 2001876. [Google Scholar] [CrossRef]
- Serafin, A.; Murphy, C.; Rubio, M.C.; Collins, M.N. Printable alginate/gelatin hydrogel reinforced with carbon nanofibers as electrically conductive scaffolds for tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 122, 111927. [Google Scholar] [CrossRef]
- e Silva, E.P.; Huang, B.; Helaehil, J.V.; Nalesso, P.R.L.; Bagne, L.; de Oliveira, M.A.; Albiazetti, G.C.C.; Aldalbahi, A.; El-Newehy, M.; Santamaria, M., Jr.; et al. In vivo study of conductive 3D printed PCL/MWCNTs scaffolds with electrical stimulation for bone tissue engineering. Bio-Des. Manuf. 2021, 4, 190–202. [Google Scholar] [CrossRef]
- Wibowo, A.; Vyas, C.; Cooper, G.; Qulub, F.; Suratman, R.; Mahyuddin, A.I.; Dirgantara, T.; Bartolo, P. 3D Printing of Polycaprolactone–Polyaniline Electroactive Scaffolds for Bone Tissue Engineering. Materials 2020, 13, 512. [Google Scholar] [CrossRef] [PubMed]
- Zurita, F.; Grob, L.; Erben, A.; Del Duca, F.; Clausen-Schaumann, H.; Sudhop, S.; Hayden, O.; Wolfrum, B. Fully 3D-Printed Cuff Electrode for Small Nerve Interfacing. Adv. Mater. Technol. 2023, 8, 2200989. [Google Scholar] [CrossRef]
- Cheng, R.; Liu, Z.; Li, M.; Shen, Z.; Wang, X.; Zhang, J.; Sang, S. Peripheral nerve regeneration with 3D printed bionic double-network conductive scaffold based on GelMA/chitosan/polypyrrole. Int. J. Biol. Macromol. 2025, 304, 140746. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Thaharah, S.; Zhang, S.; Lu, W.F.; Fuh, J.Y.H. 3D-Printed PCL/rGO Conductive Scaffolds for Peripheral Nerve Injury Repair. Artif. Organs 2019, 43, 515–523. [Google Scholar] [CrossRef]
- Basara, G.; Saeidi-Javash, M.; Ren, X.; Bahcecioglu, G.; Wyatt, B.C.; Anasori, B.; Zhang, Y.; Zorlutuna, P. Electrically conductive 3D printed Ti3C2Tx MXene-PEG composite constructs for cardiac tissue engineering. Acta Biomater. 2022, 139, 179–189. [Google Scholar] [CrossRef]
- Lei, Q.; He, J.; Li, D. Electrohydrodynamic 3D printing of layer-specifically oriented, multiscale conductive scaffolds for cardiac tissue engineering. Nanoscale 2019, 11, 15195–15205. [Google Scholar] [CrossRef]
- Dutta, S.D.; Ganguly, K.; Randhawa, A.; Patil, T.V.; Patel, D.K.; Lim, K.-T. Electrically stimulated 3D bioprinting of gelatin-polypyrrole hydrogel with dynamic semi-IPN network induces osteogenesis via collective signaling and immunopolarization. Biomaterials 2023, 294, 121999. [Google Scholar] [CrossRef]
- Luque, G.C.; Picchio, M.L.; Daou, B.; Lasa-Fernandez, H.; Criado-Gonzalez, M.; Querejeta, R.; Filgueiras-Ramas, D.; Prato, M.; Mecerreyes, D.; Ruiz-Cabello, J.; et al. Printable Poly(3,4-ethylenedioxythiophene)-Based Conductive Patches for Cardiac Tissue Remodeling. ACS Appl. Mater. Interfaces 2024, 16, 34467–34479. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, C.; Liu, Z.; Ko, J.; Chen, L.; Zhang, T.; Xiong, Z.; Zhang, L.; Sun, W. 3D Printed Conductive Multiscale Nerve Guidance Conduit with Hierarchical Fibers for Peripheral Nerve Regeneration. Adv. Sci. 2023, 10, 2205744. [Google Scholar] [CrossRef]
- Zhang, Y.; Le Friec, A.; Sun, D.; Chen, M. Sinusoidal stretchable fibrous electrodes regulate cardiac contraction. Chem. Eng. J. 2023, 455, 140555. [Google Scholar] [CrossRef]
- Ghaziof, S.; Shojaei, S.; Mehdikhani, M.; Khodaei, M.; Jafari Nodoushan, M. Electro-conductive 3D printed polycaprolactone/gold nanoparticles nanocomposite scaffolds for myocardial tissue engineering. J. Mech. Behav. Biomed. Mater. 2022, 132, 105271. [Google Scholar] [CrossRef] [PubMed]
- Petcu, E.B.; Midha, R.; McColl, E.; Popa-Wagner, A.; Chirila, T.V.; Dalton, P.D. 3D printing strategies for peripheral nerve regeneration. Biofabrication 2018, 10, 032001. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; You, D.; Zhan, N.; Liu, C.; Zhang, X.; Lin, L.; Zhang, J.; Lou, Y.; Chen, Y.; Liu, C.; et al. 4D Oriented Dynamic Scaffold for Promoting Peripheral Nerve Regeneration and Functional Recovery. Adv. Funct. Mater. 2024, 34, 2305827. [Google Scholar] [CrossRef]
- Shin, S.R.; Li, Y.-C.; Jang, H.L.; Khoshakhlagh, P.; Akbari, M.; Nasajpour, A.; Zhang, Y.S.; Tamayol, A.; Khademhosseini, A. Graphene-based materials for tissue engineering. Adv. Drug Deliv. Rev. 2016, 105, 255–274. [Google Scholar] [CrossRef]
- Yu, J.; Wan, R.; Tian, F.; Cao, J.; Wang, W.; Liu, Q.; Yang, H.; Liu, J.; Liu, X.; Lin, T.; et al. 3D Printing of Robust High-Performance Conducting Polymer Hydrogel-Based Electrical Bioadhesive Interface for Soft Bioelectronics. Small 2024, 20, 2308778. [Google Scholar] [CrossRef]
- Ruggiero, A.; Criscuolo, V.; Grasselli, S.; Bruno, U.; Ausilio, C.; Bovio, C.L.; Bettucci, O.; Santoro, F. Two-photon polymerization lithography enabling the fabrication of PEDOT:PSS 3D structures for bioelectronic applications. Chem. Commun. 2022, 58, 9790–9793. [Google Scholar] [CrossRef]
- Heinrich, M.A.; Liu, W.; Jimenez, A.; Yang, J.; Akpek, A.; Liu, X.; Pi, Q.; Mu, X.; Hu, N.; Schiffelers, R.M.; et al. 3D Bioprinting: From Benches to Translational Applications. Small 2019, 15, 1805510. [Google Scholar] [CrossRef]
- Shevach, M.; Maoz, B.M.; Feiner, R.; Shapira, A.; Dvir, T. Nanoengineering gold particle composite fibers for cardiac tissue engineering. J. Mater. Chem. B 2013, 1, 5210–5217. [Google Scholar] [CrossRef]
- Song, C.; Zhang, X.; Wang, L.; Wen, F.; Xu, K.; Xiong, W.; Li, C.; Li, B.; Wang, Q.; Xing, M.M.Q.; et al. An Injectable Conductive Three-Dimensional Elastic Network by Tangled Surgical-Suture Spring for Heart Repair. ACS Nano 2019, 13, 14122–14137. [Google Scholar] [CrossRef]
- Cui, H.; Liu, C.; Esworthy, T.; Huang, Y.; Yu, Z.-x.; Zhou, X.; San, H.; Lee, S.-j.; Hann, S.Y.; Boehm, M.; et al. 4D physiologically adaptable cardiac patch: A 4-month in vivo study for the treatment of myocardial infarction. Sci. Adv. 2020, 6, eabb5067. [Google Scholar] [CrossRef]
- Xu, T.; Baicu, C.; Aho, M.; Zile, M.; Boland, T. Fabrication and characterization of bio-engineered cardiac pseudo tissues. Biofabrication 2009, 1, 035001. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Su, J.; Li, Y.; Zhang, H.; Shi, Y.; Yan, C.; Lu, J. 3D/4D printed bio-piezoelectric smart scaffolds for next-generation bone tissue engineering. Int. J. Extrem Manuf. 2023, 5, 32007. [Google Scholar] [CrossRef]
- Mancuso, E.; Shah, L.; Jindal, S.; Serenelli, C.; Tsikriteas, Z.M.; Khanbareh, H.; Tirella, A. Additively manufactured BaTiO3 composite scaffolds: A novel strategy for load bearing bone tissue engineering applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 126, 112192. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, X.; Mei, X.; Zeng, D.; Ying, B.; Yu, Z.; Liu, S.; Li, R.; Qin, Y. 3D-printed porous PEI/TCP composite scaffolds loaded with graphdiyne oxide on the surface for bone defect repair and near-infrared light-responsive antibacterial. Mater. Des. 2024, 237, 112569. [Google Scholar] [CrossRef]
- Liu, X.; Miller, A.L., II; Park, S.; George, M.N.; Waletzki, B.E.; Xu, H.; Terzic, A.; Lu, L. Two-Dimensional Black Phosphorus and Graphene Oxide Nanosheets Synergistically Enhance Cell Proliferation and Osteogenesis on 3D Printed Scaffolds. ACS Appl. Mater. Interfaces 2019, 11, 23558–23572. [Google Scholar] [CrossRef]
- Leppik, L.; Oliveira, K.M.C.; Bhavsar, M.B.; Barker, J.H. Electrical stimulation in bone tissue engineering treatments. Eur. J. Trauma Emerg. Surg. 2020, 46, 231–244. [Google Scholar] [CrossRef]
- Wang, C.; Yue, H.; Liu, J.; Zhao, Q.; He, Z.; Li, K.; Lu, B.; Huang, W.; Wei, Y.; Tang, Y.; et al. Advanced reconfigurable scaffolds fabricated by 4D printing for treating critical-size bone defects of irregular shapes. Biofabrication 2020, 12, 045025. [Google Scholar] [CrossRef]
- Kalogeropoulou, M.; Díaz-Payno, P.J.; Mirzaali, M.J.; van Osch, G.J.V.M.; Fratila-Apachitei, L.E.; Zadpoor, A.A. 4D printed shape-shifting biomaterials for tissue engineering and regenerative medicine applications. Biofabrication 2024, 16, 22002. [Google Scholar] [CrossRef]
- Soleymani, S.; Naghib, S.M. 3D and 4D printing hydroxyapatite-based scaffolds for bone tissue engineering and regeneration. Heliyon 2023, 9, e19363. [Google Scholar] [CrossRef]
- Kalali, A.; Rezaie, H.; Hesaraki, S.; Khodaei, M.; Teimoory, F.; Saboori, A. 3D printing of composite scaffolds based on polycaprolactone matrix reinforced with monticellite and akermanite for bone repair, mechanical and biological properties. Materialia 2024, 34, 102057. [Google Scholar] [CrossRef]
- Lai, J.; Wang, C.; Liu, J.; Chen, S.; Liu, C.; Huang, X.; Wu, J.; Pan, Y.; Xie, Y.; Wang, M. Low temperature hybrid 3D printing of hierarchically porous bone tissue engineering scaffolds with in situ delivery of osteogenic peptide and mesenchymal stem cells. Biofabrication 2022, 14, 045006. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Zhou, C.; Fan, H.; Zhang, B.; Pei, X.; Fan, Y.; Jiang, Q.; Bao, R.; Yang, Q.; Dong, Z.; et al. Novel 3D porous biocomposite scaffolds fabricated by fused deposition modeling and gas foaming combined technology. Compos. Part B Eng. 2018, 152, 151–159. [Google Scholar] [CrossRef]
- Li, N.; Cui, J.; Chi, M.; Thieringer, F.M.; Sharma, N. Building a better bone: The synergy of 2D nanomaterials and 3D printing for bone tissue engineering. Mater. Des. 2023, 234, 112362. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, S.; Yu, J.; Gu, Z.; Zhang, Y. Advances in Translational 3D Printing for Cartilage, Bone, and Osteochondral Tissue Engineering. Small 2022, 18, e2201869. [Google Scholar] [CrossRef]
- Xu, J.; Ji, J.; Jiao, J.; Zheng, L.; Hong, Q.; Tang, H.; Zhang, S.; Qu, X.; Yue, B. 3D Printing for Bone-Cartilage Interface Regeneration. Front. Bioeng. Biotechnol. 2022, 10, 828921. [Google Scholar] [CrossRef]
- Qu, M.; Wang, C.; Zhou, X.; Libanori, A.; Jiang, X.; Xu, W.; Zhu, S.; Chen, Q.; Sun, W.; Khademhosseini, A. Multi-Dimensional Printing for Bone Tissue Engineering. Adv. Healthc. Mater. 2021, 10, 2001986. [Google Scholar] [CrossRef]
- Maroulakos, M.; Kamperos, G.; Tayebi, L.; Halazonetis, D.; Ren, Y. Applications of 3D printing on craniofacial bone repair: A systematic review. J. Dent. 2019, 80, 1–14. [Google Scholar] [CrossRef]
- Vyas, J.; Shah, I.; Singh, S.; Prajapati, B.G. Biomaterials-based additive manufacturing for customized bioengineering in management of otolaryngology: A comprehensive review. Front. Bioeng. Biotechnol. 2023, 11, 1234340. [Google Scholar] [CrossRef]
- Koons, G.L.; Diba, M.; Mikos, A.G. Materials design for bone-tissue engineering. Nat. Rev. Mater. 2020, 5, 584–603. [Google Scholar] [CrossRef]
- Chen, A.Y.; Pegg, E.; Chen, A.; Jin, Z.; Gu, G.X. 4D Printing of Electroactive Materials. Adv. Intell. Syst. 2021, 3, 2100019. [Google Scholar] [CrossRef]
- Asim, S.; Tabish, T.A.; Liaqat, U.; Ozbolat, I.T.; Rizwan, M. Advances in Gelatin Bioinks to Optimize Bioprinted Cell Functions. Adv. Healthc. Mater. 2023, 12, 2203148. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Liu, Y.; Wan, W.; Hu, C.; Liu, Z.; Wong, C.T.; Fukuda, T.; Shen, Y. Hybrid 3D printing and electrodeposition approach for controllable 3D alginate hydrogel formation. Biofabrication 2017, 9, 025032. [Google Scholar] [CrossRef] [PubMed]
- Handral, H.K.; Natu, V.P.; Cao, T.; Fuh, J.Y.H.; Sriram, G.; Lu, W.F. Emerging trends and prospects of electroconductive bioinks for cell-laden and functional 3D bioprinting. Bio-Des. Manuf. 2022, 5, 396–411. [Google Scholar] [CrossRef]
- Muller, J.; Huaux, F.; Moreau, N.; Misson, P.; Heilier, J.-F.; Delos, M.; Arras, M.; Fonseca, A.; Nagy, J.B.; Lison, D. Respiratory toxicity of multi-wall carbon nanotubes. Toxicol. Appl. Pharmacol. 2005, 207, 221–231. [Google Scholar] [CrossRef]
- Shanmugam, D.K.; Anitha, S.C.; Souresh, V.; Madhavan, Y.; Sampath, S.; Venugopal, D.C.; Saravanan, M. Current advancements in the development of bionic organs using regenerative medicine and 3D tissue engineering. Int. J. Bioprint 2023, 38, 2242732. [Google Scholar] [CrossRef]
- Barcena, A.J.R.; Ravi, P.; Kundu, S.; Tappa, K. Emerging Biomedical and Clinical Applications of 3D-Printed Poly(Lactic Acid)-Based Devices and Delivery Systems. Bioengineering 2024, 11, 705. [Google Scholar] [CrossRef]
- Hahn, A.; Brandes, G.; Wagener, P.; Barcikowski, S. Metal ion release kinetics from nanoparticle silicone composites. J. Control. Release 2011, 154, 164–170. [Google Scholar] [CrossRef]
- Ananth, K.P.; Jayram, N.D. A comprehensive review of 3D printing techniques for biomaterial-based scaffold fabrication in bone tissue engineering. Ann. 3D Print. Med. 2024, 13, 100141. [Google Scholar] [CrossRef]
- Xie, Z.-T.; Zeng, J.; Miyagawa, S.; Sawa, Y.; Matsusaki, M. 3D puzzle-inspired construction of large and complex organ structures for tissue engineering. Mater. Today Bio 2023, 21, 100726. [Google Scholar] [CrossRef]
- Tavares-Negrete, J.A.; Babayigit, C.; Najafikoshnoo, S.; Lee, S.W.; Boyraz, O.; Esfandyarpour, R. A Novel 3D-Bioprinting Technology of Orderly Extruded Multi-Materials via Photopolymerization. Adv. Mater. Technol. 2023, 8, 2201926. [Google Scholar] [CrossRef]
- Shamma, R.N.; Sayed, R.H.; Madry, H.; EL Sayed, N.S.; Cucchiarini, M. Triblock Copolymer Bioinks in Hydrogel Three-Dimensional Printing for Regenerative Medicine: A Focus on Pluronic F127. Tissue Eng. Part B Rev. 2022, 28, 451–463. [Google Scholar] [CrossRef] [PubMed]
- Puistola, P.; Miettinen, S.; Skottman, H.; Mörö, A. Novel strategy for multi-material 3D bioprinting of human stem cell based corneal stroma with heterogenous design. Mater. Today Bio 2024, 24, 100924. [Google Scholar] [CrossRef] [PubMed]
- Marovič, N.; Ban, I.; Maver, U.; Maver, T. Magnetic nanoparticles in 3D-printed scaffolds for biomedical applications. Nanotechnol. Rev. 2023, 12, 20220570. [Google Scholar] [CrossRef]
- Chauhan, N.; Saxena, K.; Rawal, R.; Jain, U. Advanced Bioink Materials for Tissue Engineering Applications. Curr. Tissue Microenviron. Rep. 2024, 5, 13–23. [Google Scholar] [CrossRef]
- Moroni, L.; Boland, T.; Burdick, J.A.; De Maria, C.; Derby, B.; Forgacs, G.; Groll, J.; Li, Q.; Malda, J.; Mironov, V.A.; et al. Biofabrication: A Guide to Technology and Terminology. Trends Biotechnol. 2018, 36, 384–402. [Google Scholar] [CrossRef]
- Morrison, R.J.; Kashlan, K.N.; Flanangan, C.L.; Wright, J.K.; Green, G.E.; Hollister, S.J.; Weatherwax, K.J. Regulatory Considerations in the Design and Manufacturing of Implantable 3D-Printed Medical Devices. Clin. Transl. Sci. 2015, 8, 594–600. [Google Scholar] [CrossRef]
- European Union. Regulation (EU) 2017/745 of the European Parliament and of the Council. Available online: https://eur-lex.europa.eu/eli/reg/2017/745/oj/eng (accessed on 1 January 2025).
- Xia, D.; Chen, J.; Zhang, Z.; Dong, M. Emerging polymeric biomaterials and manufacturing techniques in regenerative medicine. Aggregate 2022, 3, e176. [Google Scholar] [CrossRef]
- Knowlton, S.; Yenilmez, B.; Tasoglu, S. Towards Single-Step Biofabrication of Organs on a Chip via 3D Printing. Trends Biotechnol. 2016, 34, 685–688. [Google Scholar] [CrossRef]
- Osouli-Bostanabad, K.; Masalehdan, T.; Kapsa, R.M.I.; Quigley, A.; Lalatsa, A.; Bruggeman, K.F.; Franks, S.J.; Williams, R.J.; Nisbet, D.R. Traction of 3D and 4D Printing in the Healthcare Industry: From Drug Delivery and Analysis to Regenerative Medicine. ACS Biomater. Sci. Eng. 2022, 8, 2764–2797. [Google Scholar] [CrossRef]
- Bosmans, C.; Rodriguez, N.G.; Karperien, M.; Malda, J.; Teixeira, L.M.; Levato, R.; Leijten, J. Towards single-cell bioprinting: Micropatterning tools for organ-on-chip development. Trends Biotechnol. 2024, 42, 739–759. [Google Scholar] [CrossRef]
- Huang, G.; Zhao, Y.; Chen, D.; Wei, L.; Hu, Z.; Li, J.; Zhou, X.; Yang, B.; Chen, Z. Applications, advancements, and challenges of 3D bioprinting in organ transplantation. Biomater. Sci. 2024, 12, 1425–1448. [Google Scholar] [CrossRef]









| Material System | Conductivity Range | Elastic Modulus Range | Charge Storage Capacity/Impedance | Primary Tissue Target | Key Functional Outcomes | Reference |
|---|---|---|---|---|---|---|
| PEDOT:PSS (pure/hydrogel) | 102–105 S/cm | 26–35 MPa (compressive) | CSC: up to 127 mC cm−2; Low impedance (<1 kΩ at 1 kHz) | Neural, Cardiac | Enhances neuronal differentiation, supports synchronous cardiomyocyte beating | [50,52,56] |
| PANI (doped) | 10−4–103 S/cm | – | – | Cardiac, Skeletal muscle | Promotes cardiomyocyte cluster synchronization, supports aligned myotube formation | [57,58,59] |
| PPy (composite) | 10−2–102 S/cm | 0.059–1.08 MPa | – | Neural, Peripheral nerve | Facilitates Schwann cell proliferation, axonal regeneration under ES | [45,60] |
| Graphene/GO/rGO composites | 10−3–103 S/m | 0.1–22.6 kPa (compressive) | Reduced impedance in EIS | Neural, Cardiac, Bone | Promotes neurogenesis, enhances cardiomyocyte maturation, supports osteogenesis | [61] |
| CNTs (MWCNT/SWCNT) | >106 S/cm (intrinsic) | 84 MPa (composite) | CSC: 2.21 mC cm−2 (0.1% in PEGDA) | Neural, Bone | Enhances NSC differentiation, osteogenic differentiation under ES | [62,63] |
| MXene (Ti3C2) nanosheets | ~0.072 S/m (in HA/Alg) | Increased with loading | – | Neural, Cardiac | Supports high cell viability, enables high-resolution bioprinting | [64] |
| Au nanoparticles/nanowires | ~0.13 S/m (in CS hydrogel) | 5.4 MPa (composite) | – | Cardiac, Muscle | Improves electrical coupling between cardiomyocytes, promotes myoblast alignment | [65] |
| Ag nanowires/particles | – | – | Resistance increase <20% after 6 days in fluid | Skin, Bone | Antibacterial, enhances wound healing, supports ES | [66] |
| Method | Material Composition | Electrical Properties | Mechanical Properties | Resolution | Application | Effects | Ref. |
|---|---|---|---|---|---|---|---|
| SLA | PEDOT:PSS-PEGDA resin | 8.2–13.8 mS cm−1 | E = 26.3 ± 4.2 MPa | ≤10 µm | Neural | >95% DRG viability, ↑ neurite outgrowth | [56] |
| SLA | PEGDA + MWCNT-NH2 | 2.21 mC cm−2 (CSC) | 1.1 MPa (↑ 189%) | 190 ± 50 µm voxel | Nerve | ↑ neurite length 113%, ↑ TUJ1/GFAP with ES | [99] |
| SLA | PPF scaffold + ssDNA@CNT coating | >200 S/m | N/A | 101.6 µm layer | Bone | ↑ ALP, OCN, RUNX2 under ES | [101] |
| SLA + ES | PPy/silk fibroin | 0.114 mS/mm | 0.059 MPa | 427 nm layer | Nerve | ↑ axon diameter 1.5× | [60] |
| SLA + electrospinning | PPy/SF + SF nanofiber | 1.13 × 10−3 S/cm | N/A | 80–180 µm channel | Nerve | Optimal Schwann cell proliferation at 80/500 µm | [46] |
| DLP | GelMA + PEDOT:PSS microgrooves | Impedance ↓ 2× vs. control | 45.2 ± 3.1 kPa (P4) | 200 µm groove | Skin | 7-day complete regeneration with ES | [188] |
| DLP | GelMA/CS + PEDOT shell | 0.18 S m−1 (100× ↑) | E = 604.7 kPa (↑ 16×) | 50 µm layer | Nerve | ↑ axon length 2× vs. control | [189] |
| DLP | PGSA-PVP | 1.63 × 10−4 S/cm | Elastic, 27% swell | 100 µm groove | Nerve | 250 µm/day healing rate with ES | [111] |
| DLP | PEGDA + graphene | 1.85 × 10−4 S/m | 189% modulus ↑ | 25–100 µm groove | Myocardium ↑ | ↑ CM maturation | [190] |
| Extrusion | PVA/κ-Carrageenan + PEDOT:PSS + CmZnO | 4.95–5.32 mS cm−1 | 743 kPa tensile, 394 kPa compressive | 100 µm pore | Infected wound | 95% closure by day 12, ↑ angiogenesis | [191] |
| Extrusion | GelMA + PEDOT:PSS bioink | 10−2–10−1 S cm−1 | G′ ≈ 1 kPa (4 °C) | 120–140 µm | Cardiac/Neural | >95% C2C12 viability, maintains contractility | [42] |
| Extrusion | PCL + DBM | 4.77 × 105 Ω/sq (20% coat) | E = 107.2 ± 6.1 MPa | 410 µm nozzle | Bone | N/A | [192] |
| Extrusion | Gelatin:HA:PPy-NP | 4.3 µS cm−1 | E = 1.08 ± 0.26 MPa | ~200 µm filament | Spinal cord | >84% NSC/MSC viability, aligned growth | [193] |
| Extrusion | ADA-GEL + PPy:PSS | 1.0–1.4 S m−1 | E ↑ 10-fold (≈1.3 MPa) | 250–900 µm strut | Cartilage | ↑ z-seeding depth, cytocompatible | [194] |
| Extrusion | Alg-Gel + CNF | 4.1 × 10−4 S/cm | 534.7 ± 2.7 kPa | 410–450 µm | Cardiac/Neural | >88% fibroblast viability | [195] |
| Extrusion | PCL + MWCNT | 1 × 10−2 S cm−1 (3 wt%) | 84 MPa (↑ 70%) | 366–397 µm pore | Bone | ↑ new bone area 2× with ES | [196] |
| Extrusion | PCL + PANI microparticles | 2.5 × 10−4 S/cm (1 wt%) | 68.4 MPa | 375 µm fiber | Bone | 88% viability (0.1 wt% PANI) | [197] |
| Extrusion | CNF/SWCNT ink | 3.8 × 10−1 S cm−1 | Yield > 20 Pa, thixotropic | 1 mm guide | Neural | ↑ neurite length 100–200 µm | [21] |
| Extrusion | HA/Alg + MXene | 5.5 mS cm−1 | Modulus ↑ with MXene | 660 µm nozzle | Neural | >95% viability, physiological conductivity | [64] |
| Extrusion | GelMA + AuNP/MXene | 0.82–0.94 S m−1 | 0.54–0.61 kPa | 215–220 µm filament | Skeletal muscle | Fusion index ↑ 2.1× (AuNP), 97% viability | [87] |
| Inkjet | AgNP microelectrodes | ~30 kΩ @ 1 kHz | N/A | Sub-µm | Locust nerve | Minimally invasive cuff interface | [198] |
| Dual-ink plotting | GelMA/CS + in situ PPy IPN | 7.97 × 102 S cm−1 | 59.2 kPa (↑ 3.3×) | 200–250 µm filament | Peripheral nerve | N/A | [199] |
| EHD-jet | PCL + rGO or PPy-b-PCL fibers | 10−3–10−2 S cm−1 | E = 16.8 ± 3 MPa | 5 µm fiber | Nerve guide | ↑ PC12 neurite, Schwann-cell migration | [200] |
| EHD-jet | PCL/PPy-b-PCL | 0.09 µS cm−1 (PCL) | 204 ± 6.7 MPa | 30–44 µm fiber | NGC | ↑ proliferation 2.5× (0.5% PPy) | [23] |
| Aerosol jet | MXene-PEG hydrogel | 1.1 × 104 S/m | 7–145 kPa (tunable) | 1–5 µm droplet | Cardiac | ↑ MYH7 2.3× vs. glass | [201] |
| Hybrid EHD | PEDOT:PSS-PEO/PCL fibers | 1.72 × 103 S/m | 13 MPa | 2.5–9.5 µm | Cardiac | Layer-specific cell alignment | [202] |
| DIW | GelMA-PPy dynamic semi-IPN | 12.8 mS cm−1 (wet) | E ≈ 3.8 MPa, toughness 324 kJ m−3 | 400 µm nozzle | Bone | 93% hBMSC viability, ↑ mineralization & M2 macrophage | [203] |
| DIW | PVA + PEDOT:PSS | 125–894 mS m−1 | E = 125–894 mS m−1 (matched) | 600 µm | Cardiac patch | 77.7% CM sensitivity, Ca2+ fidelity | [204] |
| MEW | PCL/Collagen | (4.3 ± 2.4) × 10−4 S m−1 | retains 84.1% of initial stress after 100 cycles (60% strain) | 574 ± 14 nm | Nerve | ↑ neurite length 4.4× (7 d) | [205] |
| MEW | PCL + Au-sputter sinusoidal mesh | 1.16/0.75 S m−1 (anisotropic) | 1.1 MPa (long) | 11.5 µm fiber | Cardiac | Synchronous beating, no fibrosis | [206] |
| NFEP | PLCL + rGO microfibers | 0.95 S cm−1 | Elastic PLCL core | 15–150 µm | Neural | 3D neuronal networks under ES | [169] |
| M3DP | rGO microfibers | ~500 Ω·cm | Scaffolds maintain structural integrity post-hydrothermal reduction. | 100–200 µm fiber | Neural/Cardiac | >90% SH-SY5Y viability, aligned growth | [170] |
| FDM | PCL + 0.5 wt% AuNP | 0.8 S/m (↑ 25%) | 43.4 ± 5.2 MPa | 300 µm layer | Cardiac patch | Non-cytotoxic, enhanced integration | [207] |
| Electrospinning | Collagen/PCL + graphene | 0.39 S/m (0.5 wt%) | 39.5 MPa | 280 nm fiber | Nerve | NCV ↑ to 73% of normal | [77] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhang, K.; Fang, L.; Xu, C.; Zhou, W.; Deng, X.; Shan, C.; Zhang, Q.; Pan, L. Advanced 3D/4D Bioprinting of Flexible Conductive Materials for Regenerative Medicine: From Bioinspired Design to Intelligent Regeneration. Micro 2026, 6, 8. https://doi.org/10.3390/micro6010008
Zhang K, Fang L, Xu C, Zhou W, Deng X, Shan C, Zhang Q, Pan L. Advanced 3D/4D Bioprinting of Flexible Conductive Materials for Regenerative Medicine: From Bioinspired Design to Intelligent Regeneration. Micro. 2026; 6(1):8. https://doi.org/10.3390/micro6010008
Chicago/Turabian StyleZhang, Kuikui, Lezhou Fang, Can Xu, Weiwei Zhou, Xiaoqiu Deng, Chenkun Shan, Quanling Zhang, and Lijia Pan. 2026. "Advanced 3D/4D Bioprinting of Flexible Conductive Materials for Regenerative Medicine: From Bioinspired Design to Intelligent Regeneration" Micro 6, no. 1: 8. https://doi.org/10.3390/micro6010008
APA StyleZhang, K., Fang, L., Xu, C., Zhou, W., Deng, X., Shan, C., Zhang, Q., & Pan, L. (2026). Advanced 3D/4D Bioprinting of Flexible Conductive Materials for Regenerative Medicine: From Bioinspired Design to Intelligent Regeneration. Micro, 6(1), 8. https://doi.org/10.3390/micro6010008






