Polyphenol–Macromolecule Interactions by Isothermal Titration Calorimetry
Abstract
1. Introduction
2. Practical Aspects Related to ITC
2.1. Careful Experimental Design and Setup
2.2. Data Analysis
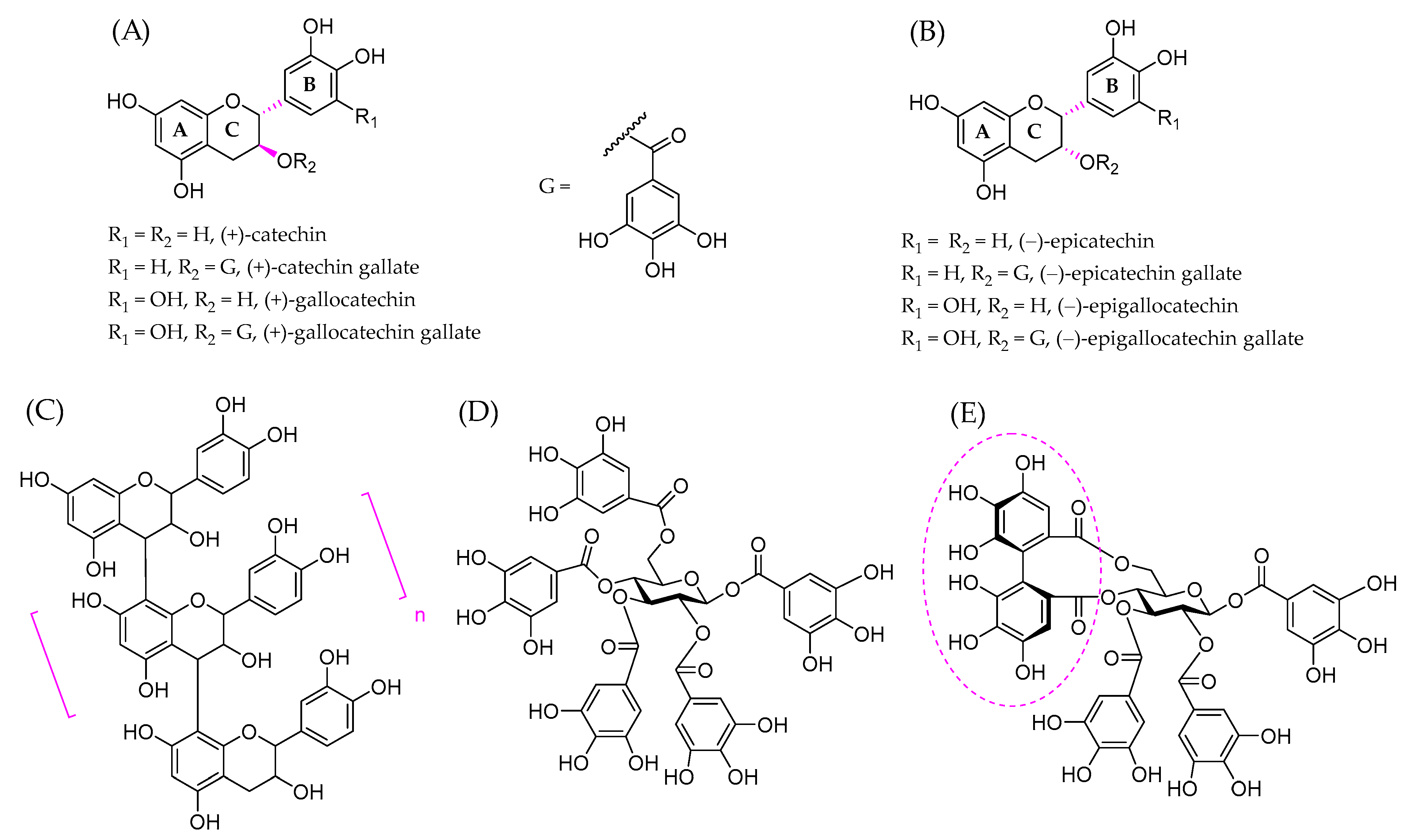
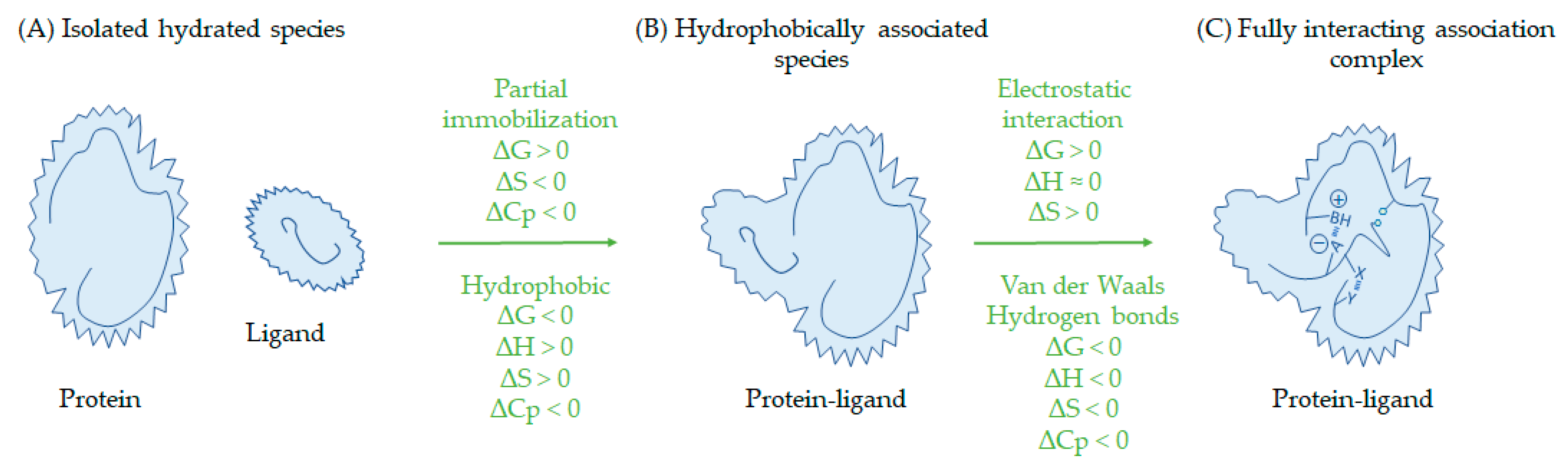
3. Polyphenols and Proteins
4. Polyphenols and Polysaccharides
5. Polyphenols and Lipids
6. Impact on Bioavailability, Food, and Other Applications
7. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant Polyphenols: Chemical Properties, Biological Activities, and Synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, A.E. Fifty Years of Polyphenol–Protein Complexes. In Recent Advances in Polyphenol Research, Volume 3; Cheynier, V., Sarni-Manchado, P., Quideau, S., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2012; pp. 71–97. [Google Scholar]
- Le Bourvellec, C.; Renard, C.M.G.C. Interactions between Polyphenols and Macromolecules: Quantification Methods and Mechanisms. Crit. Rev. Food Sci. Nutr. 2012, 52, 213–248. [Google Scholar] [CrossRef] [PubMed]
- Le Bourvellec, C.; Renard, C.M.G.C. Non-Covalent Interaction between Procyanidins and Apple Cell Wall Material. Part II: Quantification and Impact of Cell Wall Drying. Biochim. Biophys. Acta 2005, 1725, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Padayachee, A.; Netzel, G.; Netzel, M.; Day, L.; Zabaras, D.; Mikkelsen, D.; Gidley, M.J. Binding of Polyphenols to Plant Cell Wall Analogues—Part 1: Anthocyanins. Food Chem. 2012, 134, 155–161. [Google Scholar] [CrossRef]
- Le Bourvellec, C.; Renard, C.M.G.C. Interactions between Polyphenols and Macromolecules: Effect of Tannin Structure. Encycl. Food Chem. 2019, 2, 515–521. [Google Scholar] [CrossRef]
- Virtanen, V.; Green, R.J.; Karonen, M. Interactions between Hydrolysable Tannins and Lipid Vesicles from Escherichia coli with Isothermal Titration Calorimetry. Molecules 2022, 27, 3204. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, V.; Puljula, E.; Räikkönen, S.; Karonen, M. Ellagitannin-Lipid Interaction by HR-MAS NMR. Molecules 2021, 26, 373. [Google Scholar] [CrossRef] [PubMed]
- Scheidt, H.A.; Huster, D. The Interaction of Small Molecules with Phospholipid Membranes Studied by 1H NOESY NMR under Magic-Angle Spinning. Acta Pharmacol. Sin. 2008, 29, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Scheidt, H.A.; Pampel, A.; Nissler, L.; Gebhardt, R.; Huster, D. Investigation of the Membrane Localization and Distribution of Flavonoids by High-Resolution Magic Angle Spinning NMR Spectroscopy. Biochim. Biophys. Acta 2004, 1663, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Kakiuchi, N.; Wang, X.; Hattori, M.; Okuda, T.; Namba, T. Circular Dichroism Studies on the Ellagitannins-Nucleic Acids Interaction. Chem. Pharm. Bull. 1987, 35, 2875–2879. [Google Scholar] [CrossRef][Green Version]
- Srivastava, V.K.; Yadav, R. Chapter 9—Isothermal titration calorimetry. In Data Processing Handbook for Complex Biological Data Sources; Misra, G., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 125–137. ISBN 9780128165485. Available online: https://www.sciencedirect.com/science/article/pii/B9780128165485000095 (accessed on 5 September 2024). [CrossRef]
- Wilcox, D.E. Isothermal Titration Calorimetry of Metal Ions Binding to Proteins: An Overview of Recent Studies. Inorganica Chim. Acta 2008, 361, 857–867. [Google Scholar] [CrossRef]
- Grossoehme, N.E.; Spuches, A.M.; Wilcox, D.E. Application of Isothermal Titration Calorimetry in Bioinorganic Chemistry. J. Biol. Inorg. Chem. 2010, 15, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Bastos, M.; Velazquez-Campoy, A. Isothermal Titration Calorimetry (ITC): A Standard Operating Procedure (SOP). Eur. Biophys. J. 2021, 50, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Archer, W.R.; Schulz, M.D. Isothermal Titration Calorimetry: Practical Approaches and Current Applications in Soft Matter. Soft Matter 2020, 16, 8760–8774. [Google Scholar] [CrossRef] [PubMed]
- Bastos, M.; Briggner, L.-E.; Shehatta, I.; Wadsö, I. The Binding of Alkane-α,ω-Diols to α-Cyclodextrin. A Microcalorimetric Study. J. Chem. Thermodyn. 1990, 22, 1181–1190. [Google Scholar] [CrossRef]
- Deaville, E.R.; Green, R.J.; Mueller-Harvey, I.; Willoughby, I.; Frazier, R.A. Hydrolyzable Tannin Structures Influence Relative Globular and Random Coil Protein Binding Strengths. J. Agric. Food Chem. 2007, 55, 4554–4561. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.A.; Murphy, K.P. Isothermal Titration Calorimetry. In Methods in Molecular Biology: Protein-Ligand Interactions: Methods and Applications; Humana Press: Totowa, NJ, USA, 2005; Volume 305, pp. 1–15. ISBN 9788578110796. [Google Scholar]
- PEAQ-ITC Systems. Available online: https://www.malvernpanalytical.com/en/assets/malvern_panalytical_microcal_peaq_itc_brochure_pn12309_tcm50-23553.pdf (accessed on 21 August 2024).
- Microcalorimetry: TA Instruments RS-DSC, DSC & ITC. Available online: https://www.tainstruments.com/pdf/brochure/TA-Instruments-RSDSC-DSC-ITC-Brochure-EN.pdf (accessed on 21 August 2024).
- Lopez, M.M.; Makhatadze, G.I. Isothermal Titration Calorimetry. Methods Mol. Biol. 2004, 173, 121–126. [Google Scholar] [CrossRef]
- Wiseman, T.; Williston, S.; Brandts, J.F.; Lin, L.-N. Rapid Measurement of Binding Constants and Heats of Binding Using a New Titration Calorimeter. Anal. Biochem. 1989, 179, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Biswas, T.; Tsodikov, O.V. An Easy-to-Use Tool for Planning and Modeling a Calorimetric Titration. Anal. Biochem. 2010, 406, 91–93. [Google Scholar] [CrossRef]
- Broecker, J.; Vargas, C.; Keller, S. Revisiting the Optimal c Value for Isothermal Titration Calorimetry. Anal. Biochem. 2011, 418, 307–309. [Google Scholar] [CrossRef] [PubMed]
- Frazier, R.A.; Papadopoulou, A.; Green, R.J. Isothermal Titration Calorimetry Study of Epicatechin Binding to Serum Albumin. J. Pharm. Biomed. Anal. 2006, 41, 1602–1605. [Google Scholar] [CrossRef] [PubMed]
- Frazier, R.A.; Deaville, E.R.; Green, R.J.; Stringano, E.; Willoughby, I.; Plant, J.; Mueller-Harvey, I. Interactions of Tea Tannins and Condensed Tannins with Proteins. J. Pharm. Biomed. Anal. 2010, 51, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Karonen, M.; Oraviita, M.; Mueller-Harvey, I.; Salminen, J.-P.; Green, R.J. Binding of an Oligomeric Ellagitannin Series to Bovine Serum Albumin (BSA): Analysis by Isothermal Titration Calorimetry (ITC). J. Agric. Food Chem. 2015, 63, 10647–10654. [Google Scholar] [CrossRef] [PubMed]
- Karonen, M.; Oraviita, M.; Mueller-Harvey, I.; Salminen, J.-P.; Green, R.J. Ellagitannins with Glucopyranose Cores Have Higher Affinities to Proteins than Acyclic Ellagitannins by Isothermal Titration Calorimetry. J. Agric. Food Chem. 2019, 67, 12730–12740. [Google Scholar] [CrossRef]
- Kilmister, R.L.; Faulkner, P.; Downey, M.O.; Darby, S.J.; Falconer, R.J. The Complexity of Condensed Tannin Binding to Bovine Serum Albumin—An Isothermal Titration Calorimetry Study. Food Chem. 2016, 190, 173–178. [Google Scholar] [CrossRef]
- Poncet-Legrand, C.; Gautier, C.; Cheynier, V.; Imberty, A. Interactions between Flavan-3-Ols and Poly(L-Proline) Studied by Isothermal Titration Calorimetry: Effect of the Tannin Structure. J. Agric. Food Chem. 2007, 55, 9235–9240. [Google Scholar] [CrossRef]
- Zhu, W.; Wang, R.; Khalifa, I.; Li, C. Understanding toward the Biophysical Interaction of Polymeric Proanthocyanidins (Persimmon Condensed Tannins) with Biomembranes: Relevance for Biological Effects. J. Agric. Food Chem. 2019, 67, 11044–11052. [Google Scholar] [CrossRef] [PubMed]
- Watrelot, A.A.; Le Bourvellec, C.; Imberty, A.; Renard, C.M.G.C. Neutral Sugar Side Chains of Pectins Limit Interactions with Procyanidins. Carbohydr. Polym. 2014, 99, 527–536. [Google Scholar] [CrossRef]
- Engström, M.T.; Karonen, M.; Ahern, J.R.; Baert, N.; Payré, B.; Hoste, H.; Salminen, J.-P. Chemical Structures of Plant Hydrolyzable Tannins Reveal Their in Vitro Activity against Egg Hatching and Motility of Haemonchus contortus Nematodes. J. Agric. Food Chem. 2016, 64, 840–851. [Google Scholar] [CrossRef]
- Wang, C.C.; Chen, H.F.; Wu, J.Y.; Chen, L.G. Stability of Principal Hydrolysable Tannins from Trapa Taiwanensis Hulls. Molecules 2019, 24, 365. [Google Scholar] [CrossRef]
- Leppä, M.M.; Karonen, M.; Tähtinen, P.; Engström, M.T.; Salminen, J.-P. Isolation of Chemically Well-Defined Semipreparative Liquid Chromatography Fractions from Complex Mixtures of Proanthocyanidin Oligomers and Polymers. J. Chromatogr. A 2018, 1576, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Guyot, S.; Marnet, N.; Sanoner, P.; Drilleau, J.-F. Direct Thiolysis on Crude Apple Materials for High-Performance Liquid Chromatography Characterization and Quantification of Polyphenols in Cider Apple Tissues and Juices. Methods Enzymol. 2001, 335, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Guyot, S.; Marnet, N.; Laraba, D.; Sanoner, P.; Drilleau, J.-F. Reversed-Phase HPLC Following Thiolysis for Quantitative Estimation and Characterization of the Four Main Classes of Phenolic Compounds in Different Tissue Zones of a French Cider Apple Variety (Malus domestica Var. Kermerrien). J. Agric. Food Chem. 1998, 46, 1698–1705. [Google Scholar] [CrossRef]
- Engström, M.T.; Pälijärvi, M.; Fryganas, C.; Grabber, J.H.; Mueller-Harvey, I.; Salminen, J.-P. Rapid Qualitative and Quantitative Analyses of Proanthocyanidin Oligomers and Polymers by UPLC-MS/MS. J. Agric. Food Chem. 2014, 62, 3390–3399. [Google Scholar] [CrossRef]
- Ràfols, C.; Bosch, E.; Barbas, R.; Prohens, R. The Ca2+-EDTA Chelation as Standard Reaction to Validate Isothermal Titration Calorimeter Measurements (ITC). Talanta 2016, 154, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Sigurskjold, B.W. Exact Analysis of Competition Ligand Binding by Displacement Isothermal Titration Calorimetry. Anal. Biochem. 2000, 277, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Swamy, M.J.; Sankhala, R.S. Probing the Thermodynamics of Protein–Lipid Interactions by Isothermal Titration Calorimetry. In Lipid-Protein Interactions; Kleinschmidt, J., Ed.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2013; Volume 974, pp. 37–53. ISBN 9781627032759. [Google Scholar] [CrossRef]
- Giri, P.; Pal, C. An Overview on the Thermodynamic Techniques Used in Food Chemistry. Mod. Chem. Appl. 2014, 2, 100142. [Google Scholar] [CrossRef]
- Prigent, S.V.E.; Voragen, A.G.J.; van Koningsveld, G.A.; Baron, A.; Renard, C.M.G.C.; Gruppen, H. Interactions between Globular Proteins and Procyanidins of Different Degrees of Polymerization. J. Dairy. Sci. 2009, 92, 5843–5853. [Google Scholar] [CrossRef] [PubMed]
- Le, V.H.; Buscaglia, R.; Chaires, J.B.; Lewis, E.A. Modeling Complex Equilibria in ITC Experiments: Thermodynamic Parameters Estimation for a Three Binding Site Model. Anal. Biochem. 2013, 434, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Freire, E.; Schön, A.; Velazquez-Campoy, A. Isothermal Titration Calorimetry: General Formalism Using Binding Polynomials. Methods Enzymol. 2009, 455, 127–155. [Google Scholar] [PubMed]
- Freire, E.; Mayorga, O.L.; Straume, M. Isothermal Titration Calorimetry. Anal. Chem. 1990, 62, 950A–959A. [Google Scholar] [CrossRef]
- Brautigam, C.A. Fitting Two- and Three-Site Binding Models to Isothermal Titration Calorimetric Data. Methods 2015, 76, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Menéndez, M. Isothermal Titration Calorimetry: Principles and Applications. eLS 2020, 1, 113–127. [Google Scholar] [CrossRef]
- Freyer, M.W.; Lewis, E.A. Isothermal Titration Calorimetry: Experimental Design, Data Analysis, and Probing Macromolecule/Ligand Binding and Kinetic Interactions. Methods Cell Biol. 2008, 84, 79–113. [Google Scholar] [CrossRef]
- Velazquez-Campoy, A.; Leavitt, S.A.; Freire, E. Characterization of Protein-Protein interactions by Isothermal Titration Calorimetry. In Methods in Molecular Biology: 18 19 20 Protein-Protein Interactions: Methods and Protocols; Meyerkord, C.L., Haian, F., Eds.; Springer Science+Business Media: New York, NY, USA, 2015; pp. 183–204. [Google Scholar]
- Herrera, I.; Winnik, M.A. Differential Binding Models for Isothermal Titration Calorimetry: Moving beyond the Wiseman Isotherm. J. Phys. Chem. B 2013, 117, 8659–8672. [Google Scholar] [CrossRef]
- Freiburger, L.A.; Auclair, K.; Mittermaier, A.K. Elucidating Protein Binding Mechanisms by Variable-c ITC. ChemBioChem 2009, 10, 2871–2873. [Google Scholar] [CrossRef] [PubMed]
- Tellinghuisen, J. Statistical Error in Isothermal Titration Calorimetry. Methods Enzymol. 2004, 383, 245–281. [Google Scholar] [PubMed]
- Pethica, B.A. Misuse of Thermodynamics in the Interpretation of Isothermal Titration Calorimetry Data for Ligand Binding to Proteins. Anal. Biochem. 2015, 472, 21–29. [Google Scholar] [CrossRef]
- Karefyllakis, D.; Altunkaya, S.; Berton-Carabin, C.C.; van der Goot, A.J.; Nikiforidis, C.V. Physical Bonding between Sunflower Proteins and Phenols: Impact on Interfacial Properties. Food Hydrocoll. 2017, 73, 326–334. [Google Scholar] [CrossRef]
- Liu, X.; Le Bourvellec, C.; Renard, C.M.G.C. Interactions between Cell Wall Polysaccharides and Polyphenols: Effect of Molecular Internal Structure. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3574–3617. [Google Scholar] [CrossRef]
- Falconer, R.J. Applications of Isothermal Titration Calorimetry—The Research and Technical Developments from 2011 to 2015. J. Mol. Recognit. 2016, 29, 504–515. [Google Scholar] [CrossRef]
- Dobreva, M.A.; Frazier, R.A.; Mueller-Harvey, I.; Clifton, L.A.; Gea, A.; Green, R.J. Binding of Pentagalloyl Glucose to Two Globular Proteins Occurs via Multiple Surface Sites. Biomacromolecules 2011, 12, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Frazier, R.A.; Papadopoulou, A.; Mueller-Harvey, I.; Kissoon, D.; Green, R.J. Probing Protein-Tannin Interactions by Isothermal Titration Microcalorimetry. J. Agric. Food Chem. 2003, 51, 5189–5195. [Google Scholar] [CrossRef]
- You, Y.; Yang, L.; Chen, H.; Xiong, L.; Yang, F. Effects of (-)-Epigallocatechin-3-Gallate on the Functional and Structural Properties of Soybean Protein Isolate. J. Agric. Food Chem. 2021, 69, 2306–2315. [Google Scholar] [CrossRef] [PubMed]
- Spencer, C.M.; Cai, Y.; Martin, R.; Gaffney, S.H.; Goulding, P.N.; Magnolato, D.; Lilley, T.H.; Haslam, E. Polyphenol Complexation-Some Thoughts and Observations. Phytochemistry 1988, 27, 2397–2409. [Google Scholar] [CrossRef]
- Dobreva, M.A.; Green, R.J.; Mueller-Harvey, I.; Salminen, J.-P.; Howlin, B.J.; Frazier, R.A. Size and Molecular Flexibility Affect the Binding of Ellagitannins to Bovine Serum Albumin. J. Agric. Food Chem. 2014, 62, 9186–9194. [Google Scholar] [CrossRef] [PubMed]
- Thao, S.; Escalante-Semerena, J.C. Biochemical and Thermodynamic Analyses of Salmonella enterica Pat, a Multidomain, Multimeric N-Lysine Acetyltransferase Involved in Carbon and Energy Metabolism. mBio 2011, 2, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ross, P.D.; Subramanian, S. Thermodynamics of Protein Association Reactions: Forces Contributing to Stability. Biochemistry 1981, 20, 3096–3102. [Google Scholar] [CrossRef] [PubMed]
- Swamy, M.J.; Sankhala, R.S.; Singh, B.P. Thermodynamic Analysis of Protein-Lipid Interactions by Isothermal Titration Calorimetry. In Lipid-Protein Interactions: Methods and Protocols, Methods in Molecular Biology; Kleinschmidt, J.H., Ed.; Springer Science+Business Media: New York, NY, USA, 2019; Volume 2003, pp. 71–89. ISBN 9781493990658. [Google Scholar]
- Gabriel, G.J.; Pool, J.G.; Som, A.; Dabkowski, J.M.; Coughlin, E.B.; Muthukumar, M.; Tew, G.N. Interactions between Antimicrobial Polynorbornenes and Phospholipid Vesicles Monitored by Light Scattering and Microcalorimetry. Langmuir 2008, 24, 12489–12495. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.; Oliveira, J.; Fonseca, F.; Ferreira-da-Silva, F.; Mateus, N.; Vincken, J.P.; de Freitas, V. Molecular Binding between Anthocyanins and Pectic Polysaccharides—Unveiling the Role of Pectic Polysaccharides Structure. Food Hydrocoll. 2020, 102, 105625. [Google Scholar] [CrossRef]
- Fernandes, P.A.R.; Le Bourvellec, C.; Renard, C.M.G.C.; Wessel, D.F.; Cardoso, S.M.; Coimbra, M.A. Interactions of Arabinan-Rich Pectic Polysaccharides with Polyphenols. Carbohydr. Polym. 2020, 230, 115644. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Li, T.; Li, R.; Zhou, H.; Liu, C.; Chen, J.; McClements, D.J. Utilization of Plant-Based Protein-Polyphenol Complexes to Form and Stabilize Emulsions: Pea Proteins and Grape Seed Proanthocyanidins. Food Chem. 2020, 329, 127219. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Dai, T.; Li, T.; Huang, K.; Li, Y.; Liu, C.; Chen, J. Investigation on the Binding Interaction between Rice Glutelin and Epigallocatechin-3-Gallate Using Spectroscopic and Molecular Docking Simulation. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2019, 217, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Yan, X.; Li, Q.; Li, T.; Liu, C.; McClements, D.J.; Chen, J. Characterization of Binding Interaction between Rice Glutelin and Gallic Acid: Multi-Spectroscopic Analyses and Computational Docking Simulation. Food Res. Int. 2017, 102, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.; Lian, Z.; Qi, W.; Chen, Y.; Tong, X.; Tian, T.; Lyu, B.; Wang, M.; Wang, H.; Jiang, L. Non-Covalent Interaction of Soy Protein Isolate and Catechin: Mechanism and Effects on Protein Conformation. Food Chem. 2022, 384, 132507. [Google Scholar] [CrossRef]
- McRae, J.M.; Falconer, R.J.; Kennedy, J.A. Thermodynamics of Grape and Wine Tannin Interaction with Polyproline: Implications for Red Wine Astringency. J. Agric. Food Chem. 2010, 58, 12510–12518. [Google Scholar] [CrossRef] [PubMed]
- de Freitas, V.A.P.; Glories, Y.; Bourgeois, G.; Vitry, C. Characterisation of Oligomeric and Polymeric Procyanidins from Grape Seeds by Liquid Secondary Ion Mass Spectrometry. Phytochemistry 1998, 49, 1435–1441. [Google Scholar] [CrossRef]
- de Freitas, V.; Mateus, N. Structural Features of Procyanidin Interactions with Salivary Proteins. J. Agric. Food Chem. 2001, 49, 940–945. [Google Scholar] [CrossRef]
- Cala, O.; Pinaud, N.; Simon, C.; Fouquet, E.; Laguerre, M.; Dufourc, E.J.; Pianet, I. NMR and Molecular Modeling of Wine Tannins Binding to Saliva Proteins: Revisiting Astringency from Molecular and Colloidal Prospects. FASEB J. 2010, 24, 4281–4290. [Google Scholar] [CrossRef]
- Tarascou, I.; Barathieu, K.; Simon, C.; Ducasse, M.A.; André, Y.; Fouquet, E.; Dufourc, E.J.; De Freitas, V.; Laguerre, M.; Pianet, I. A 3D Structural and Conformational Study of Procyanidin Dimers in Water and Hydro-Alcoholic Media as Viewed by NMR and Molecular Modeling. Magn. Reson. Chem. 2006, 44, 868–880. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, A.E.; Rice, M.E.; Ritchard, N.T. Mechanisms of Protein Precipitation for Two Tannins, Pentagalloyl Glucose and Epicatechin16 (4-8) Catechin (Procyanidin). J. Agric. Food Chem. 1998, 46, 2590–2595. [Google Scholar] [CrossRef]
- Murray, N.; Williamson, M.; Lilley, T.; Haslam, E. Study of the Interaction between Salivary Proline-Rich Proteins and a Polyphenol by 1H-NMR Spectroscopy. Eur. J. Biochem. 1994, 219, 923–935. [Google Scholar] [CrossRef]
- Luck, G.; Liao, H.; Murray, N.J.; Grimmer, H.R.; Warminski, E.E.; Williamson, M.P.; Lilley, T.H.; Haslam, E. Polyphenols, Astringency and Proline-Rich Proteins. Phytochemistry 1994, 37, 357–371. [Google Scholar] [CrossRef]
- Gaffney, S.H.; Martin, R.; Lilley, T.H.; Haslam, E.; Magnolato, D. The Association of Polyphenols with Caffeine and α- and β-Cyclodextrin in Aqueous Media. J. Chem. Soc. Chem. Commun. 1986, 107–109. [Google Scholar] [CrossRef]
- Kríž, Z.; Koča, J.; Imberty, A.; Charlot, A.; Auzély-Velty, R. Investigation of the Complexation of (+)-Catechin by β-Cyclodextrin by a Combination of NMR, Microcalorimetry and Molecular Modeling Techniques. Org. Biomol. Chem. 2003, 1, 2590–2595. [Google Scholar] [CrossRef] [PubMed]
- Watrelot, A.A.; Le Bourvellec, C.; Imberty, A.; Renard, C.M.G.C. Interactions between Pectic Compounds and Procyanidins Are Influenced by Methylation Degree and Chain Length. Biomacromolecules 2013, 14, 709–718. [Google Scholar] [CrossRef]
- Mamet, T.; Ge, Z.-Z.; Zhang, Y.; Li, C.-M. Interactions between Highly Galloylated Persimmon Tannins and Pectins. Int. J. Biol. Macromol. 2018, 106, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Seijen-ten-Hoorn, J.; Velikov, K.P. Colloidal Complexes from Associated Water Soluble Cellulose Derivative (Methylcellulose) and Green Tea Polyphenol (Epigallocatechin Gallate). J. Colloid. Interface Sci. 2011, 364, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Li, J.; Li, B. Multiple Steps and Critical Behaviors of the Binding of Tannic Acid to Wheat Starch: Effect of the Concentration of Wheat Starch and the Mass Ratio of Tannic Acid to Wheat Starch. Food Hydrocoll. 2019, 94, 174–182. [Google Scholar] [CrossRef]
- Salminen, J.-P.; Karonen, M. Chemical Ecology of Tannins and Other Phenolics: We Need a Change in Approach. Funct. Ecol. 2011, 25, 325–338. [Google Scholar] [CrossRef]
- Pina, F.; Oliveira, J.; De Freitas, V. Anthocyanins and Derivatives Are More than Flavylium Cations. Tetrahedron 2015, 71, 3107–3114. [Google Scholar] [CrossRef]
- Koh, J.; Xu, Z.; Wicker, L. Binding Kinetics of Blueberry Pectin-Anthocyanins and Stabilization by Non-Covalent Interactions. Food Hydrocoll. 2020, 99, 105354. [Google Scholar] [CrossRef]
- He, Y.; Wang, S.; Li, J.; Liang, H.; Wei, X.; Peng, D.; Jiang, Z.; Li, B. Interaction between Konjac Glucomannan and Tannic Acid: Effect of Molecular Weight, PH and Temperature. Food Hydrocoll. 2019, 94, 451–458. [Google Scholar] [CrossRef]
- Brahem, M.; Renard, C.M.G.C.; Bureau, S.; Watrelot, A.A.; Le Bourvellec, C. Pear Ripeness and Tissue Type Impact Procyanidin-Cell Wall Interactions. Food Chem. 2019, 275, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Le Bourvellec, C.; Watrelot, A.A.; Ginies, C.; Imberty, A.; Renard, C.M.G.C. Impact of Processing on the Noncovalent Interactions between Procyanidin and Apple Cell Wall. J. Agric. Food Chem. 2012, 60, 9484–9494. [Google Scholar] [CrossRef]
- Patel, A.R.; Seijen Ten-Hoorn, J.; Hazekamp, J.; Blijdenstein, T.B.J.; Velikov, K.P. Colloidal Complexation of a Macromolecule with a Small Molecular Weight Natural Polyphenol: Implications in Modulating Polymer Functionalities. Soft Matter 2013, 9, 1428–1436. [Google Scholar] [CrossRef]
- Da Silva, V.M.; Sato, J.A.P.; Araujo, J.N.; Squina, F.M.; Muniz, J.R.C.; Riske, K.A.; Garcia, W. Systematic Studies of the Interactions between a Model Polyphenol Compound and Microbial β-Glucosidases. PLoS ONE 2017, 12, e0181629. [Google Scholar] [CrossRef]
- Olsen, S.N.; Bohlin, C.; Murphy, L.; Borch, K.; McFarland, K.C.; Sweeny, M.D.; Westh, P. Effects of Non-Ionic Surfactants on the Interactions between Cellulases and Tannic Acid: A Model System for Cellulase-Poly-Phenol Interactions. Enzyme Microb. Technol. 2011, 49, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.Z.; Chen, W.Y.; Tasi, L.M.; Yang, S.P. Microcalorimetric and Shear Studies on the Effects of Cholesterol on the Physical Stability of Lipid Vesicles. Colloids Surf. A Physicochem. Eng. Asp. 2000, 172, 57–67. [Google Scholar] [CrossRef]
- Carneiro, F.A.; Bianconi, M.L.; Weissmüller, G.; Stauffer, F.; Da Poian, A.T. Membrane Recognition by Vesicular Stomatitis Virus Involves Enthalpy-Driven Protein-Lipid Interactions. J. Virol. 2002, 76, 3756–3764. [Google Scholar] [CrossRef] [PubMed]
- Rovere, M.; Sanderson, J.B.; Fonseca-Ornelas, L.; Patel, D.S.; Bartels, T. Refolding of Helical Soluble α-Synuclein through Transient Interaction with Lipid Interfaces. FEBS Lett. 2018, 592, 1464–1472. [Google Scholar] [CrossRef]
- Wang, C.K.; Wacklin, H.P.; Craik, D.J. Cyclotides Insert into Lipid Bilayers to Form Membrane Pores and Destabilize the Membrane through Hydrophobic and Phosphoethanolamine-Specific Interactions. J. Biol. Chem. 2012, 287, 43884–43898. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, F.A.; Lapido-Loureiro, P.A.; Cordo, S.M.; Stauffer, F.; Weissmüller, G.; Bianconi, M.L.; Juliano, M.A.; Juliano, L.; Bisch, P.M.; Da Poian, A.T. Probing the Interaction between Vesicular Stomatitis Virus and Phosphatidylserine. Eur. Biophys. J. 2006, 35, 145–154. [Google Scholar] [CrossRef]
- Andrushchenko, V.V.; Aarabi, M.H.; Nguyen, L.T.; Prenner, E.J.; Vogel, H.J. Thermodynamics of the Interactions of Tryptophan-Rich Cathelicidin Antimicrobial Peptides with Model and Natural Membranes. Biochim. Biophys. Acta 2008, 1778, 1004–1014. [Google Scholar] [CrossRef] [PubMed]
- Dehsorkhi, A.; Castelletto, V.; Hamley, I.W.; Seitsonen, J.; Ruokolainen, J. Interaction between a Cationic Surfactant-like Peptide and Lipid Vesicles and Its Relationship to Antimicrobial Activity. Langmuir 2013, 29, 14246–14253. [Google Scholar] [CrossRef]
- Sauder, R.; Seelig, J.; Ziegler, A. Thermodynamics of Lipid Interactions with Cell-Penetrating Peptides. In Cell-Penetrating Peptides: Methods and Protocols, Methods in Molecular Biology; Langel, Ü., Ed.; Springer Science+Business Media: New York, NY, USA, 2011; Volume 683, pp. 129–155. ISBN 9781607619192. [Google Scholar]
- Kerek, E.; Hassanin, M.; Zhang, W.; Prenner, E.J. Preferential Binding of Inorganic Mercury to Specific Lipid Classes and Its Competition with Cadmium. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Klasczyk, B.; Knecht, V.; Lipowsky, R.; Dimova, R. Interactions of Alkali Metal Chlorides with Phosphatidylcholine Vesicles. Langmuir 2010, 26, 18951–18958. [Google Scholar] [CrossRef]
- Krylova, O.O.; Jahnke, N.; Keller, S. Membrane Solubilisation and Reconstitution by Octylglucoside: Comparison of Synthetic Lipid and Natural Lipid Extract by Isothermal Titration Calorimetry. Biophys. Chem. 2010, 150, 105–111. [Google Scholar] [CrossRef]
- Pruchnik, H.; Bonarska-Kujawa, D.; Żyłka, R.; Oszmiański, J.; Kleszczyńska, H. Application of the DSC and Spectroscopy Methods in the Analysis of the Protective Effect of Extracts from the Blueberry Fruit of the Genus Vaccinium in Relation to the Lipid Membrane. J. Therm. Anal. Calorim. 2018, 134, 679–689. [Google Scholar] [CrossRef]
- Huh, N.-W.W.; Porter, N.A.A.; McIntosh, T.J.J.; Simon, S.A.A. The Interaction of Polyphenols with Bilayers: Conditions for Increasing Bilayer Adhesion. Biophys. J. 1996, 71, 3261–3277. [Google Scholar] [CrossRef] [PubMed]
- Caturla, N.; Pérez-Fons, L.; Estepa, A.; Micol, V. Differential Effects of Oleuropein, a Biophenol from Olea europaea, on Anionic and Zwiterionic Phospholipid Model Membranes. Chem. Phys. Lipids 2005, 137, 2–17. [Google Scholar] [CrossRef] [PubMed]
- Ulrih, N.P.; Maričić, M.; Ota, A.; Šentjurc, M.; Abram, V. Kaempferol and Quercetin Interactions with Model Lipid Membranes. Food Res. Int. 2015, 71, 146–154. [Google Scholar] [CrossRef]
- Selvaraj, S.; Krishnaswamy, S.; Devashya, V.; Sethuraman, S.; Krishnan, U.M. Influence of Membrane Lipid Composition on Flavonoid-Membrane Interactions: Implications on Their Biological Activity. Prog. Lipid Res. 2015, 58, 1–13. [Google Scholar] [CrossRef]
- Malekar, S.A.; Sarode, A.L.; Bach, A.C.; Worthen, D.R. The Localization of Phenolic Compounds in Liposomal Bilayers and Their Effects on Surface Characteristics and Colloidal Stability. AAPS PharmSciTech 2016, 17, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Altunayar-Unsalan, C.; Unsalan, O.; Mavromoustakos, T. Insights into Molecular Mechanism of Action of Citrus Flavonoids Hesperidin and Naringin on Lipid Bilayers Using Spectroscopic, Calorimetric, Microscopic and Theoretical Studies. J. Mol. Liq. 2022, 347, 118411. [Google Scholar] [CrossRef]
- Coones, R.T.; Karonen, M.; Green, R.J.; Frazier, R. Interactions of Galloylated Polyphenols with a Simple Gram-Negative Bacterial Membrane Lipid Model. Membranes 2024, 14, 47. [Google Scholar] [CrossRef] [PubMed]
- Chiu, M.; Prenner, E. Differential Scanning Calorimetry: An Invaluable Tool for a Detailed Thermodynamic Characterization of Macromolecules and Their Interactions. J. Pharm. Bioallied Sci. 2011, 3, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Jelesarov, I.; Bosshard, H.R. Isothermal Titration Calorimetry and Differential Scanning Calorimetry as Complementary Tools to Investigate the Energetics of Biomolecular Recognition. J. Mol. Recognit. 1999, 12, 3–18. [Google Scholar] [CrossRef]
- Huang, C. Structural Organization and Properties of Membrane Lipids. In Cell Physiology Source Book; Sperelakis, N., Ed.; Academic Press: San Diego, CA, USA, 2001; pp. 43–63. [Google Scholar]
- Šturm, L.; Ulrih, N.P. Basic Methods for Preparation of Liposomes and Studying Their Interactions with Different Compounds, with the Emphasis on Polyphenols. Int. J. Mol. Sci. 2021, 22, 6547. [Google Scholar] [CrossRef]
- Patil, Y.P.; Jadhav, S. Novel Methods for Liposome Preparation. Chem. Phys. Lipids 2014, 177, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Vitkova, V.; Hazarosova, R.; Valkova, I.; Momchilova, A.; Staneva, G. Glycerophospholipid Polyunsaturation Modulates Resveratrol Action on Biomimetic Membranes. Colloids Surf. B Biointerfaces 2024, 238, 113922. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Jiang, H.; Pu, Y.; Cao, J.; Jiang, W. Inhibitory Effect of Condensed Tannins from Banana Pulp on Cholesterol Esterase and Mechanisms of Interaction. J. Agric. Food Chem. 2019, 67, 14066–14073. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food Sources and Bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Jakobek, L. Interactions of Polyphenols with Carbohydrates, Lipids and Proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef]
- Pripp, A.H.; Vreeker, R.; van Duynhoven, J. Binding of Olive Oil Phenolics to Food Proteins. J. Sci. Food Agric. 2005, 85, 354–362. [Google Scholar] [CrossRef]
- Lombardi, L.; Stellato, M.I.; Oliva, R.; Falanga, A.; Galdiero, M.; Petraccone, L.; D’Errico, G.; De Santis, A.; Galdiero, S.; Del Vecchio, P. Antimicrobial Peptides at Work: Interaction of Myxinidin and Its Mutant WMR with Lipid Bilayers Mimicking the P. aeruginosa and E. coli Membranes. Sci. Rep. 2017, 7, srep44425. [Google Scholar] [CrossRef]
- Sikorska, E.; Dawgul, M.; Greber, K.; Iłowska, E.; Pogorzelska, A.; Kamysz, W. Self-Assembly and Interactions of Short Antimicrobial Cationic Lipopeptides with Membrane Lipids: ITC, FTIR and Molecular Dynamics Studies. Biochim. Biophys. Acta 2014, 1838, 2625–2634. [Google Scholar] [CrossRef] [PubMed]
- Santos-Buelga, C.; Scalbert, A. Proanthocyanidins and Tannin-like Compounds—Nature, Occurrence, Dietary Intake and Effects on Nutrition and Health. J. Sci. Food Agric. 2000, 80, 1094–1117. [Google Scholar] [CrossRef]
- Obreque-Slier, E.; Peña-Neira, Á.; López-Solís, R. Interactions of Enological Tannins with the Protein Fraction of Saliva and Astringency Perception Are Affected by PH. LWT-Food Sci. Technol. 2012, 45, 88–93. [Google Scholar] [CrossRef]
- Shpigelman, A.; Israeli, G.; Livney, Y.D. Thermally-Induced Protein-Polyphenol Co-Assemblies: Beta Lactoglobulin-Based Nanocomplexes as Protective Nanovehicles for EGCG. Food Hydrocoll. 2010, 24, 735–743. [Google Scholar] [CrossRef]
- Stojadinovic, M.; Radosavljevic, J.; Ognjenovic, J.; Vesic, J.; Prodic, I.; Stanic-Vucinic, D.; Cirkovic Velickovic, T. Binding Affinity between Dietary Polyphenols and β-Lactoglobulin Negatively Correlates with the Protein Susceptibility to Digestion and Total Antioxidant Activity of Complexes Formed. Food Chem. 2013, 136, 1263–1271. [Google Scholar] [CrossRef]
- Gonçalves, R.; Mateus, N.; De Freitas, V. Influence of Carbohydrates on the Interaction of Procyanidin B3 with Trypsin. J. Agric. Food Chem. 2011, 59, 11794–11802. [Google Scholar] [CrossRef]
- Jung, M.H.; Seong, P.N.; Kim, M.H.; Myong, N.H.; Chang, M.J. Effect of Green Tea Extract Microencapsulation on Hypertriglyceridemia and Cardiovascular Tissues in High Fructose-Fed Rats. Nutr. Res. Pract. 2013, 7, 366–372. [Google Scholar] [CrossRef][Green Version]
- Ozawa, T.; Lilley, H. Terence; Haslam Edwin Polyphenol Interactions: Astrigency and the Loss of Astrigency in Ripening Fruit. Phytochemistry 1987, 26, 2937–2942. [Google Scholar] [CrossRef]
- Taira, S.; Ono, M.; Matsumoto, N. Reduction of Persimmon Astringency by Complex Formation between Pectin and Tannins. Postharvest Biol. Technol. 1997, 12, 265–271. [Google Scholar] [CrossRef]
- Houtman, J.C.D.; Brown, P.H.; Bowden, B.; Yamaguchi, H.; Appella, E.; Samelson, L.E.; Schuck, P. Studying Multisite Binary and Ternary Protein Interactions by Global Analysis of Isothermal Titration Calorimetry Data in SEDPHAT: Application to Adaptor Protein Complexes in Cell Signaling. Protein Sci. 2007, 16, 30–42. [Google Scholar] [CrossRef]
- Cotrina, E.Y.; Gimeno, A.; Llop, J.; Jiménez-Barbero, J.; Quintana, J.; Valencia, G.; Cardoso, I.; Prohens, R.; Arsequell, G. Calorimetric Studies of Binary and Ternary Molecular Interactions between Transthyretin, Aβ Peptides, and Small-Molecule Chaperones toward an Alternative Strategy for Alzheimer’s Disease Drug Discovery. J. Med. Chem. 2020, 63, 3205–3214. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.W.; Armaou, A.; Rioux, R.M. Continuous Injection Isothermal Titration Calorimetry for in Situ Evaluation of Thermodynamic Binding Properties of Ligand-Receptor Binding Models. J. Phys. Chem. B 2021, 125, 8075–8087. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.W.; Mu, Y.; Armaou, A.; Rioux, R.M. Direct Determination of High-Affinity Binding Constants by Continuous Injection Isothermal Titration Calorimetry. J. Phys. Chem. B 2023, 127, 10833–10842. [Google Scholar] [CrossRef]
- Johannes Gutenberg University Mainz, I. of P. and B.S. ITC-Calculator. Available online: https://www.pharmazie.uni-mainz.de/ (accessed on 10 December 2024).
- Zheng, F.; Jiang, X.; Wen, Y.; Yang, Y.; Li, M. Systematic Investigation of Machine Learning on Limited Data: A Study on Predicting Protein-Protein Binding Strength. Comput. Struct. Biotechnol. J. 2024, 23, 460–472. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Huang, R.; Xia, M.; Patterson, T.A.; Hong, H. Fingerprinting Interactions between Proteins and Ligands for Facilitating Machine Learning in Drug Discovery. Biomolecules 2024, 14, 72. [Google Scholar] [CrossRef]
- Wang, D.D.; Ou-Yang, L.; Xie, H.; Zhu, M.; Yan, H. Predicting the Impacts of Mutations on Protein-Ligand Binding Affinity Based on Molecular Dynamics Simulations and Machine Learning Methods. Comput. Struct. Biotechnol. J. 2020, 18, 439–454. [Google Scholar] [CrossRef] [PubMed]

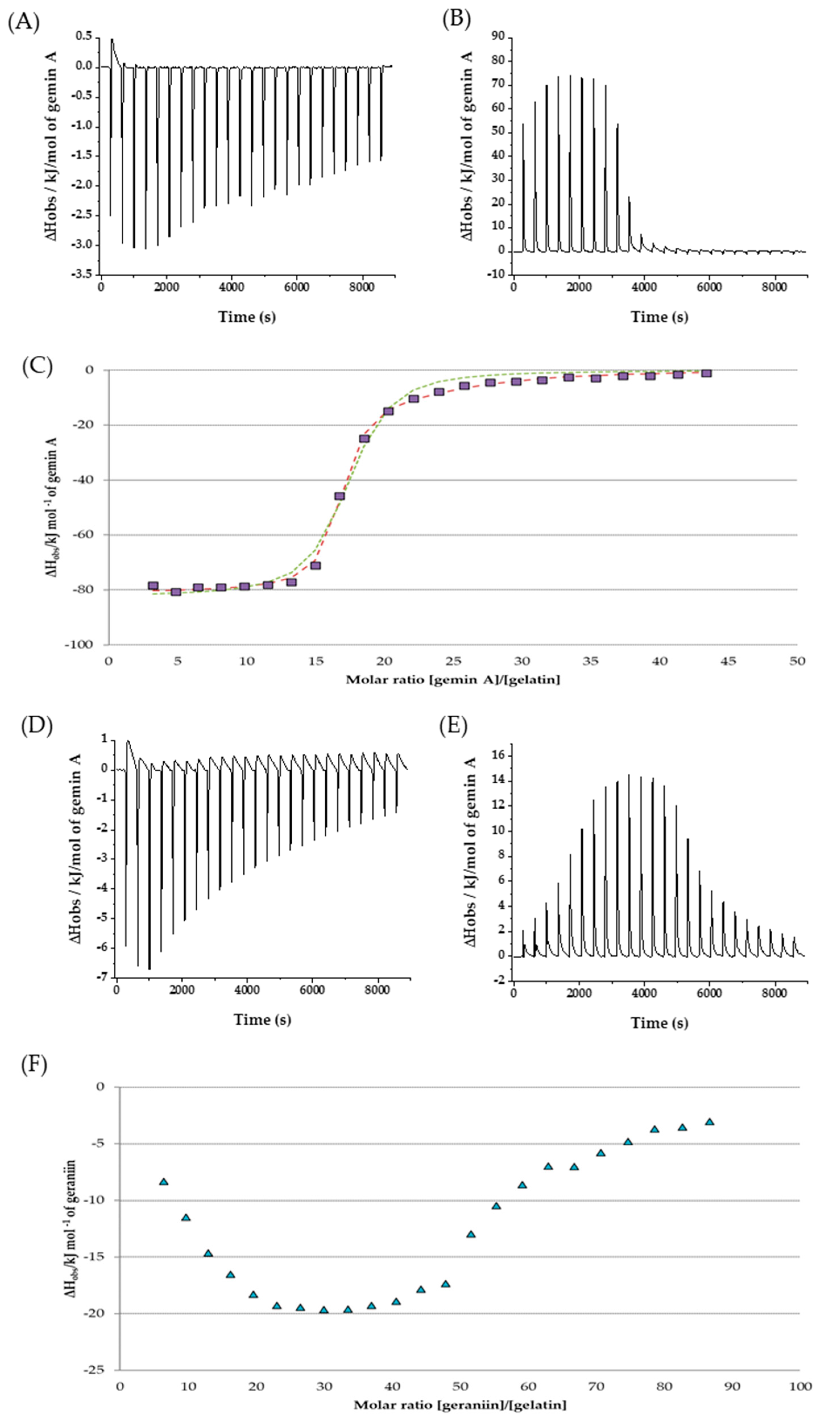


| Interacting Species | n1 | K1 (M–1) | ΔH1 (kJ mol−1) | n2 | K2 (M–1) | ΔH2 (kJ mol−1) | Ref. |
|---|---|---|---|---|---|---|---|
| Agrimoniin–BSA (one-site model) | 17 | 1.7 × 104 | −24 | [29] | |||
| Agrimoniin–BSA (two-site model) | 16 | 3.6 × 104 | −18 | 46 | 6.9 × 102 | −8 | [29] |
| Agrimoniin–gelatin (one-site model) | 20 | 7.4 × 104 | −76 | [29] | |||
| Agrimoniin–gelatin (two-site model) | 18 | 1.7 × 105 | −65 | 52 | 1.1 × 104 | −10 | [29] |
| Chestnut tannins–BSA (one-site model) | 18 | 9.0 × 102 | −40 | [18] | |||
| Chestnut tannins–gelatin (two-site model) | 46 | 1.5 × 106 | −22 | 32 | 1.1 × 104 | −27 | [18] |
| Epicatechin–BSA 1 (one-site model) | 4 | 2.9 × 102 | −37 | [26] | |||
| Epicatechingallate–poly(L-proline) (one-site model) | 7 | 8.1 × 104 | −33 | [31] | |||
| Epigallocatechingallate– poly(L-proline) (one-site model) | 9 | 3.7 × 104 | −25 | [31] | |||
| Epicatechin tetramer–poly(L-proline) (one-site model) | 6 | 3.4 × 105 | −11 | [31] | |||
| Gemin A–BSA (one-site model) | 14 | 1.1 × 104 | −45 | [29] | |||
| Gemin A–BSA (two-site model) | 14 | 1.6 × 104 | −37 | 30 | 7.8 × 102 | −8 | [29] |
| Gemin A–BSA (two-site model) | 13 | 1.8 × 106 | −47 | 25 | 6.3 × 104 | −11 | [63] |
| Gemin A–gelatin (one-site model) | 16 | 4.2 × 105 | −81 | [29] | |||
| Gemin A–gelatin (two-site model) | 15 | 1.3 × 106 | −64 | 21 | 4.2 × 104 | −17 | [29] |
| Grape seed tannins–BSA (one-site model) | 7 | 1.5 × 103 | −102 | [27] | |||
| Grape seed tannins–gelatin (one-site model) | 35 | 3.3 × 105 | −38 | [27] | |||
| Green tea polyhenols–BSA (one-site model) | 19 | 1.4 × 102 | −154 | [27] | |||
| Green tea polyphenols–gelatin (one-site model) | 71 | 9.8 × 103 | −46 | [27] | |||
| Lambertianin C–BSA (one-site model) | 11 | 1.1 × 105 | −28 | [29] | |||
| Lambertianin C–BSA (two-site model) | 11 | 1.6 × 105 | −25 | 37 | 3.1 × 103 | −3 | [29] |
| Lambertianin C–gelatin (one-site model) | 11 | 1.5 × 106 | −93 | [29] | |||
| Lambertianin C–gelatin (two-site model) | 11 | 1.9 × 106 | −91 | 15 | 2.5 × 102 | −22 | [29] |
| Macrocyclic tetrameric ET–BSA (one-site model) | 9 | 2.7 × 104 | −69 | [28] | |||
| Macrocyclic tetrameric ET–BSA (two-site model) | 9 | 7.3 × 104 | −52 | 90 | 1.8 × 102 | −19 | [28] |
| Macrocyclic pentameric ET–BSA (one-site model) | 9 | 2.3 × 104 | −80 | [28] | |||
| Macrocyclic pentameric ET–BSA (two-site model) | 9 | 7.7 × 104 | −56 | 35 | 2.6 × 102 | −44 | [28] |
| Macrocyclic hexameric ET–BSA (one-site model) | 10 | 1.7 × 104 | −88 | [28] | |||
| Macrocyclic hexameric ET–BSA (two-site model) | 9 | 5.6 × 104 | −63 | 59 | 1.2 × 102 | −59 | [28] |
| Macrocyclic heptameric ET–BSA (one-site model) | 10 | 1.7 × 104 | −93 | [28] | |||
| Macrocyclic heptameric ET–BSA (two-site model) | 9 | 3.5 × 104 | −73 | 84 | 1.7 × 102 | −27 | [28] |
| Mimosa tannins–BSA (one-site model) | 39 | 5.9 × 103 | −10 | [27] | |||
| Mimosa tannins–gelatin (one-site model) | 34 | 8.6 × 104 | −35 | [27] | |||
| Myrabolan tannins–BSA | 178 | [60] | |||||
| Myrabolan tannins–BSA (one-site model) | 22 | 7.0 × 102 | −58 | [18] | |||
| Myrabolan tannins–gelatin (two-site model) | 36 | 2.2 × 106 | −28 | 39 | 8.3 × 103 | −31 | [18] |
| Oenothein B–BSA (one-site model) | 12 | 5.7 × 103 | −14 | [28] | |||
| Oenothein B–BSA (two-site model) | 6 | 9.8 × 103 | −14 | 36 | 1.1 × 103 | −7 | [28] |
| Oenothein B–BSA (two-site model) | 4 | 6.5 × 105 | −21 | 20 | 3.3 × 104 | −10 | [63] |
| Oenothein A–BSA (one-site model) | 11 | 7.6 × 103 | −45 | [28] | |||
| Oenothein A–BSA (two-site model) | 11 | 1.2 × 104 | −33 | 32 | 3.5 × 102 | −9 | [28] |
| Pedunculagin–BSA (two-site model) | 2 | 4.2 × 104 | −11 | 30 | 1.1 × 103 | −5 | [63] |
| Pentagalloylglucose–BSA (two-site model) | 17 | 2.2 × 105 | −38 | 67 | 6.0 × 102 | −49 | [18] |
| Pentagalloylglucose–BSA (two-site model) | 26 | 1.8 × 105 | −29 | 26 | 8.0 × 102 | −29 | [59] |
| Pentagalloylglucose–BSA (one-site model) | 26 | 2.3 × 104 | −40 | [59] | |||
| Pentagalloylglucose–gelatin (two-site model) | 31 | 2.8 × 105 | −47 | 60 | 7.5 × 102 | −44 | [18] |
| Pentagalloylglucose–rubisco (two-site model) | 204 | 1.2 × 106 | −35 | 406 | 3.1 ×104 | −20 | [59] |
| Pentagalloylglucose–rubisco (one-site model) | 466 | 1.3 × 104 | −37 | [59] | |||
| Procyanidin tetramer–BSA (one-site model) | 53 | 6.3 × 103 | −11 | [30] | |||
| Procyanidin pentamer–BSA (one-site model) | 34 | 1.0 × 104 | −13 | [30] | |||
| Procyanidin hexamer–BSA (one-site model) | 21 | 1.7 × 104 | −21 | [30] | |||
| Procyanidin heptamer–BSA (one-site model) | 18 | 1.1 × 104 | −27 | [30] | |||
| Procyanidin octamer–BSA (one-site model) | 18 | 1.4 × 104 | −24 | [30] | |||
| Roburin A–BSA (two-site model) | 2 | 2.3 × 105 | −15 | 16 | 1.8 × 104 | −8 | [63] |
| Roshenin C–gelatin (one-site model) | 30 | 1.7 × 104 | −57 | [29] | |||
| Roshenin C–gelatin (two-site model) | 24 | 1.5 × 105 | −30 | 30 | 4.2 × 104 | −57 | [29] |
| Sanguiin H-6–BSA (one-site model) | 19 | 1.3 × 104 | −26 | [29] | |||
| Sanguiin H-6–BSA (two-site model) | 17 | 3.5 × 104 | −18 | 94 | 1.1 × 103 | −6 | [29] |
| Sanguiin H-6–gelatin (one-site model) | 17 | 7.3 × 104 | −78 | [29] | |||
| Sanguiin H-6–gelatin (two-site model) | 16 | 2.2 × 105 | −63 | 38 | 4.2 × 104 | −12 | [29] |
| Sorghum tannins–BSA (one-site model) | 11 | 3.3 × 104 | −21 | [27] | |||
| Sorghum tannins–gelatin (one-site model) | 16 | 2.0 × 106 | −60 | [27] | |||
| Sumac tannins–BSA (two-site model) | 9 | 1.7 × 105 | −30 | 24 | 2.2 × 103 | −30 | [18] |
| Sumac tannins–gelatin (two-site model) | 35 | 6.9 × 105 | −38 | 21 | 4.2 × 102 | −49 | [18] |
| Tara tannins–BSA | 48 | [60] | |||||
| Tara tannins–BSA (two-site model) | 3 | 1.0× 104 | −33 | 15 | 6.8 × 102 | −25 | [18] |
| Tara tannins–gelatin (two-site model) | 53 | 8.0 × 103 | −21 | 58 | 1.5 × 102 | −21 | [18] |
| Tellimagrandin I–BSA (one-site model) | 6 | 1.8 × 104 | −24 | [28] | |||
| Tellimagrandin I–BSA (two-site model) | 6 | 2.2 × 104 | −20 | 4 | 1.8 × 103 | −10 | [28] |
| Tellimagrandin I–gelatin (one-site model) | 67 | 7.6 × 103 | −39 | [29] | |||
| Tellimagrandin I–gelatin (two-site model) | 52 | 1.8 × 105 | −14 | 120 | 8.9 × 104 | −11 | [29] |
| Tellimagrandin II–BSA (one-site model) | 30 | 7.3 × 103 | −37 | [29] | |||
| Tellimagrandin II–BSA (two-site model) | 29 | 8.3 × 103 | −33 | 110 | 3.6 × 101 | −28 | [29] |
| Tellimagrandin II–gelatin (one-site model) | 31 | 7.6 × 104 | −59 | [29] | |||
| Tellimagrandin II–gelatin (two-site model) | 31 | 8.4 × 104 | −56 | 59 | 7.1 × 103 | −3 | [29] |
| Vescalagin–BSA (two-site model) | 3 | 5.2 × 104 | −8 | 30 | 1.1 × 103 | −7 | [63] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karonen, M. Polyphenol–Macromolecule Interactions by Isothermal Titration Calorimetry. Macromol 2025, 5, 2. https://doi.org/10.3390/macromol5010002
Karonen M. Polyphenol–Macromolecule Interactions by Isothermal Titration Calorimetry. Macromol. 2025; 5(1):2. https://doi.org/10.3390/macromol5010002
Chicago/Turabian StyleKaronen, Maarit. 2025. "Polyphenol–Macromolecule Interactions by Isothermal Titration Calorimetry" Macromol 5, no. 1: 2. https://doi.org/10.3390/macromol5010002
APA StyleKaronen, M. (2025). Polyphenol–Macromolecule Interactions by Isothermal Titration Calorimetry. Macromol, 5(1), 2. https://doi.org/10.3390/macromol5010002






