Lamination of Cast Hemp Paper with Bio-Based Plastics for Sustainable Packaging: Structure-Thermomechanical Properties Relationship and Biodegradation Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
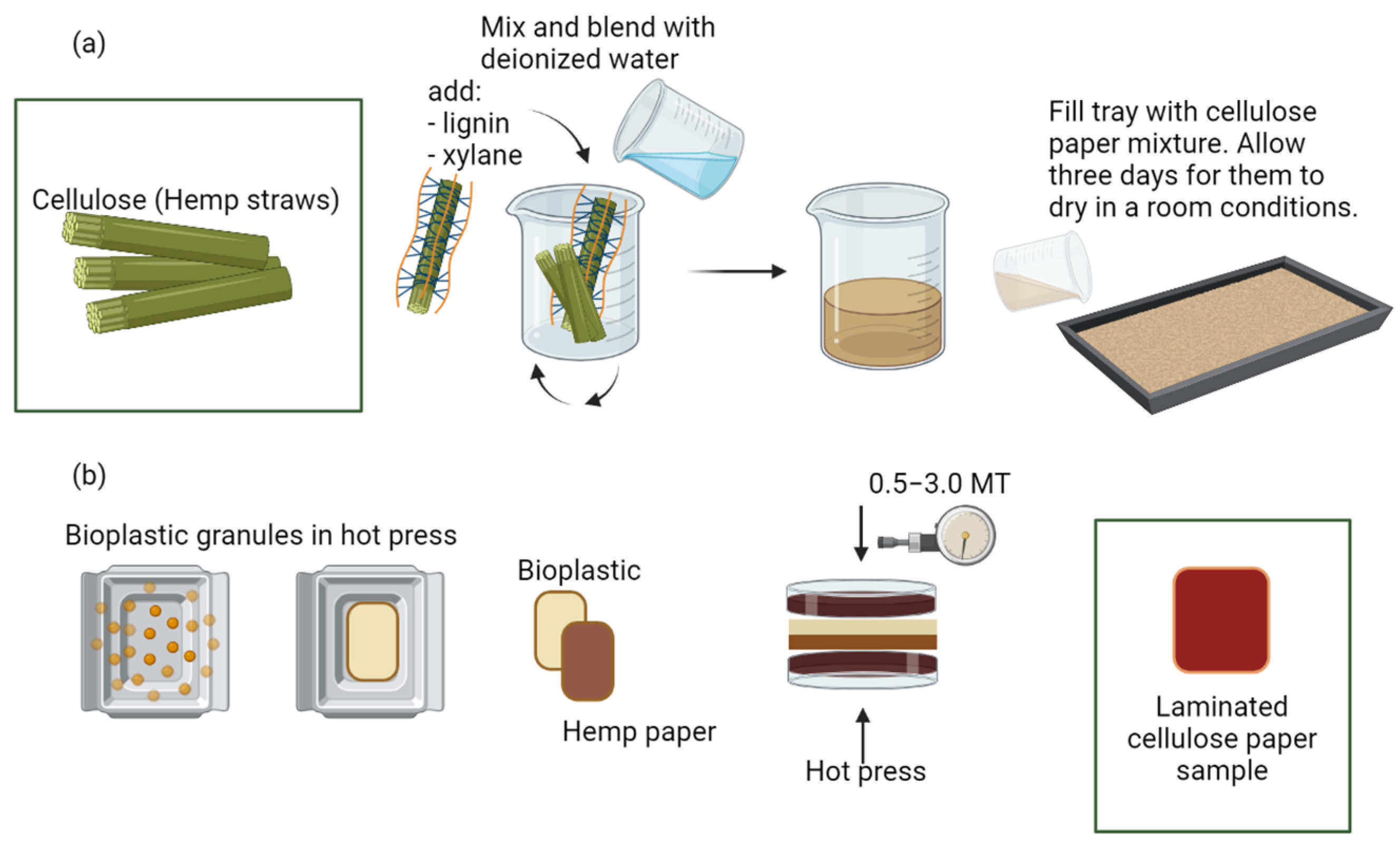
2.2. Hemp Paper Preparation
2.3. Cellulose Paper Lamination
2.4. Characterization Methods
3. Results and Discussion
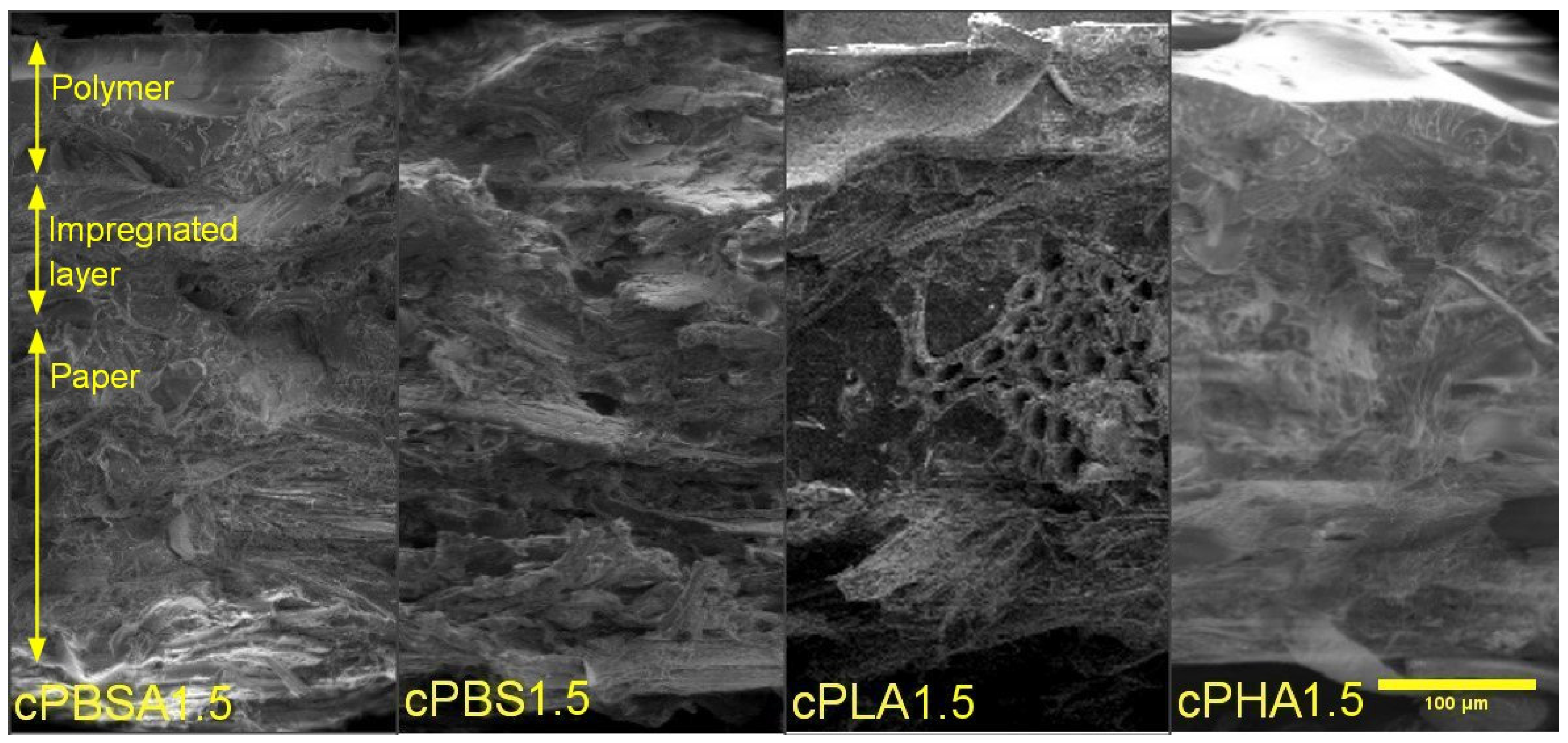
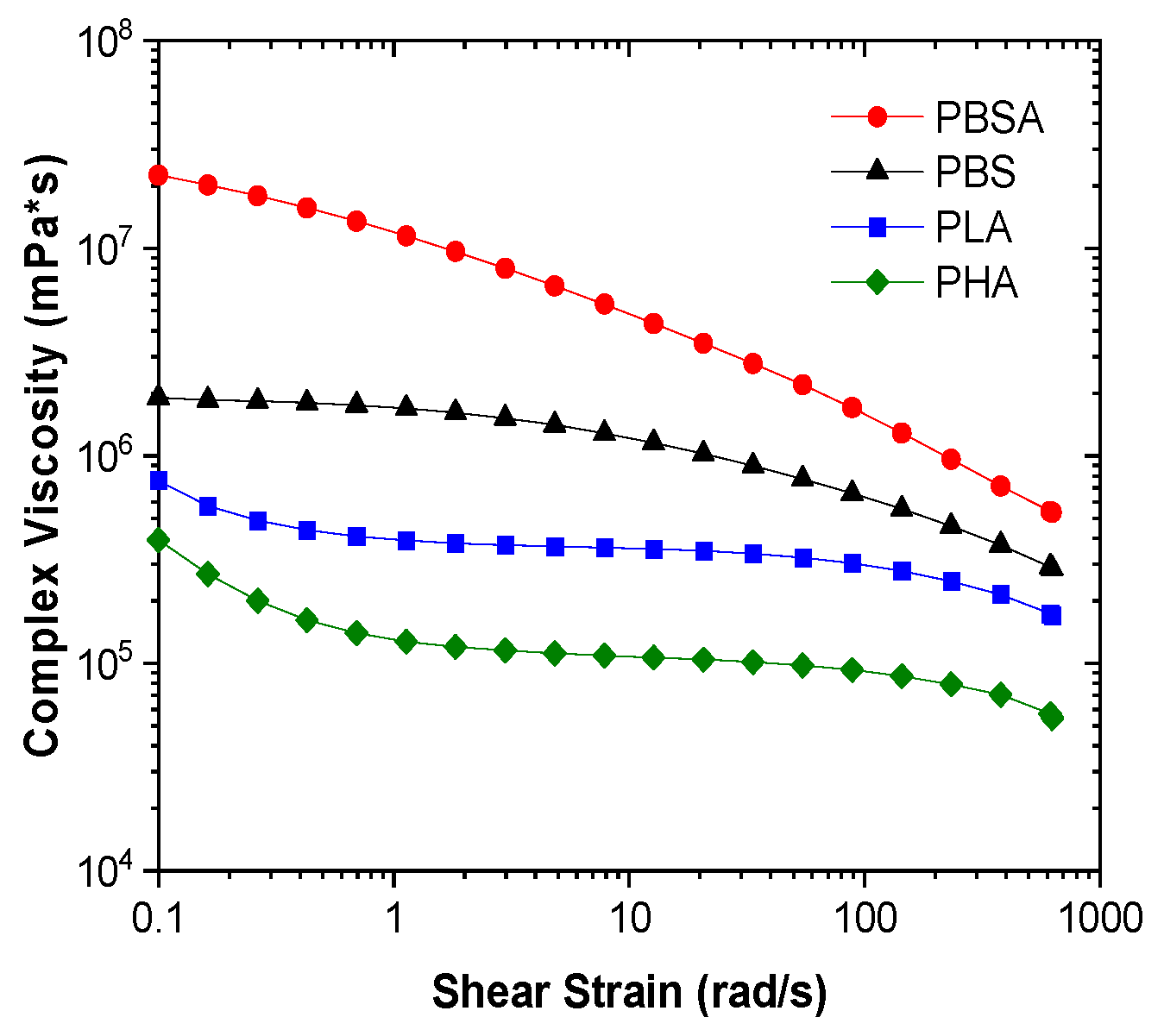
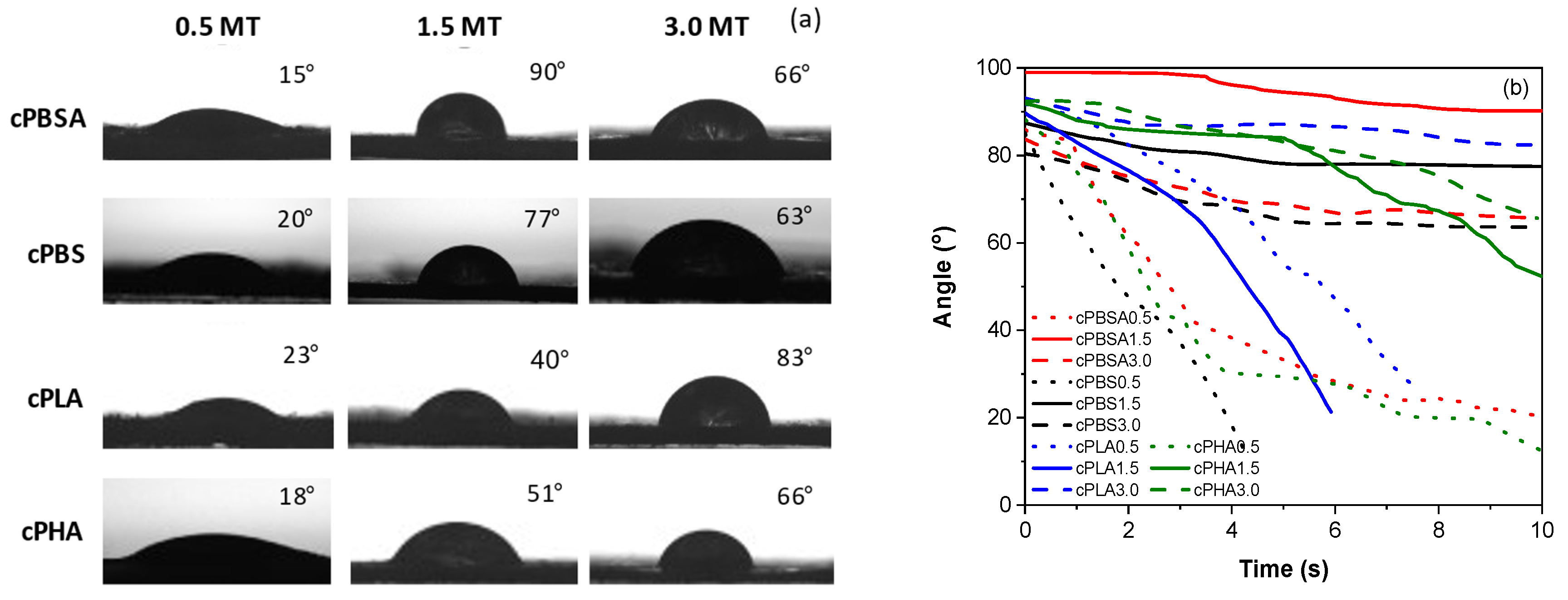
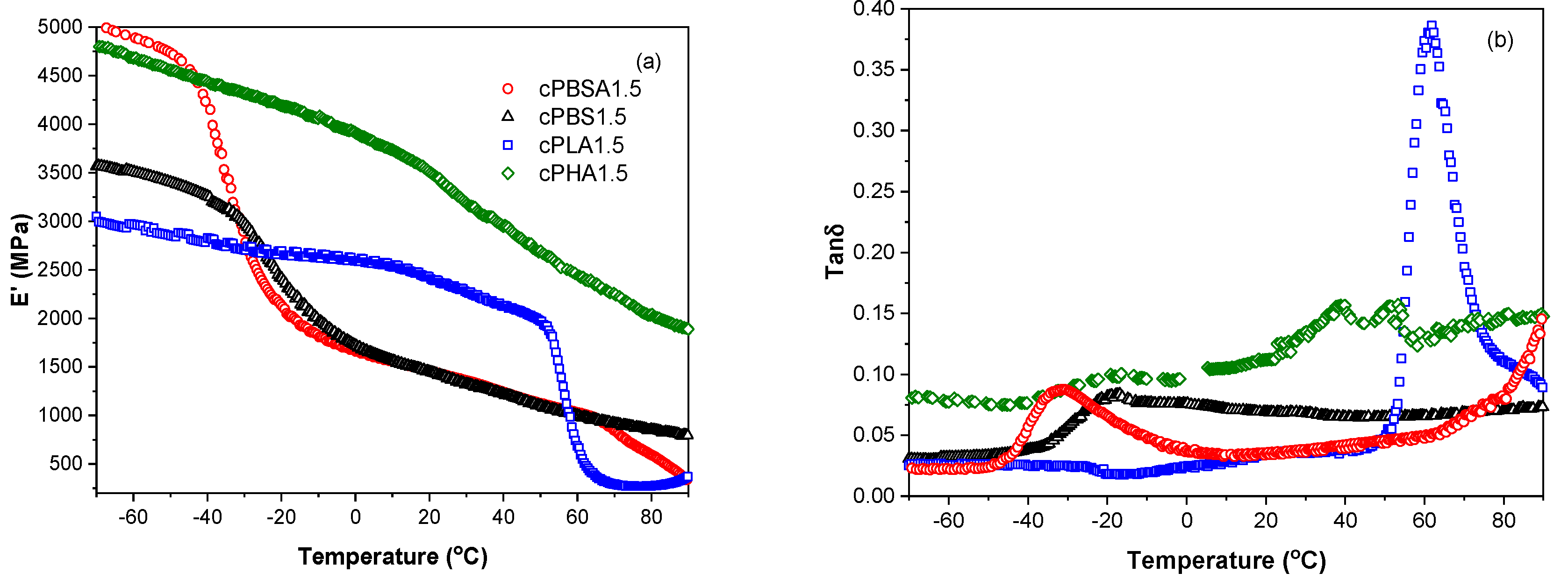
3.1. Structure Analysis
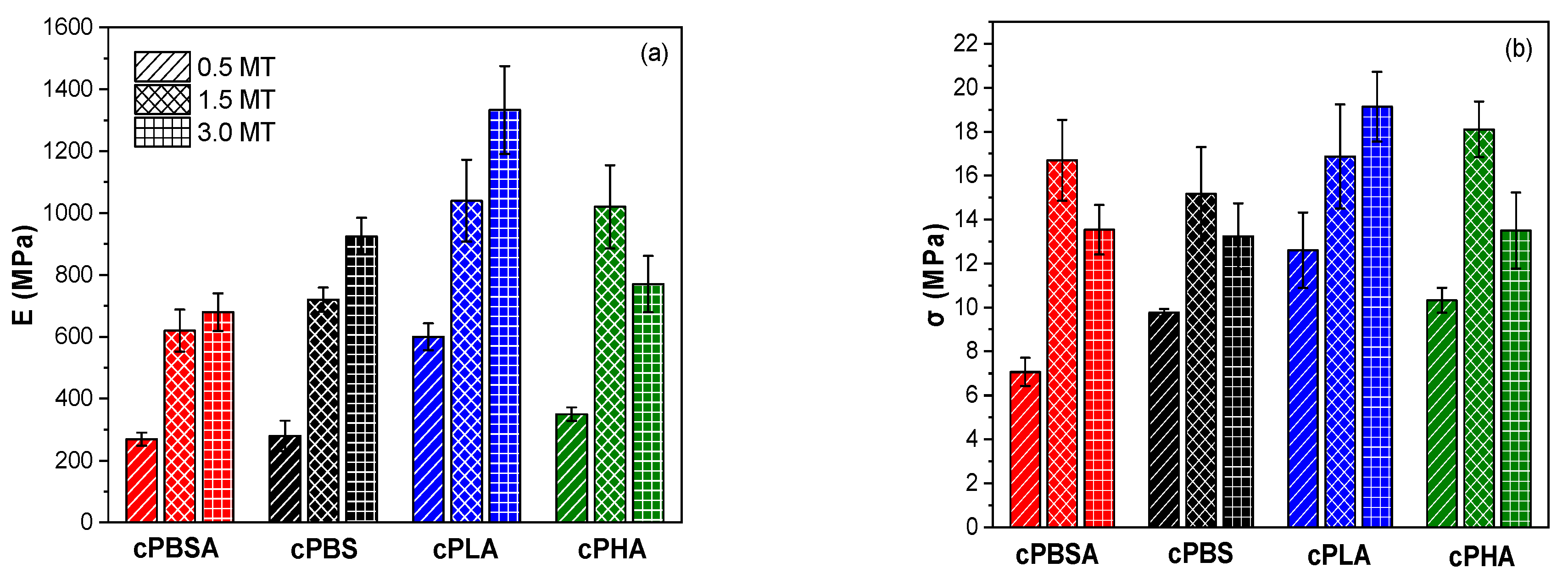
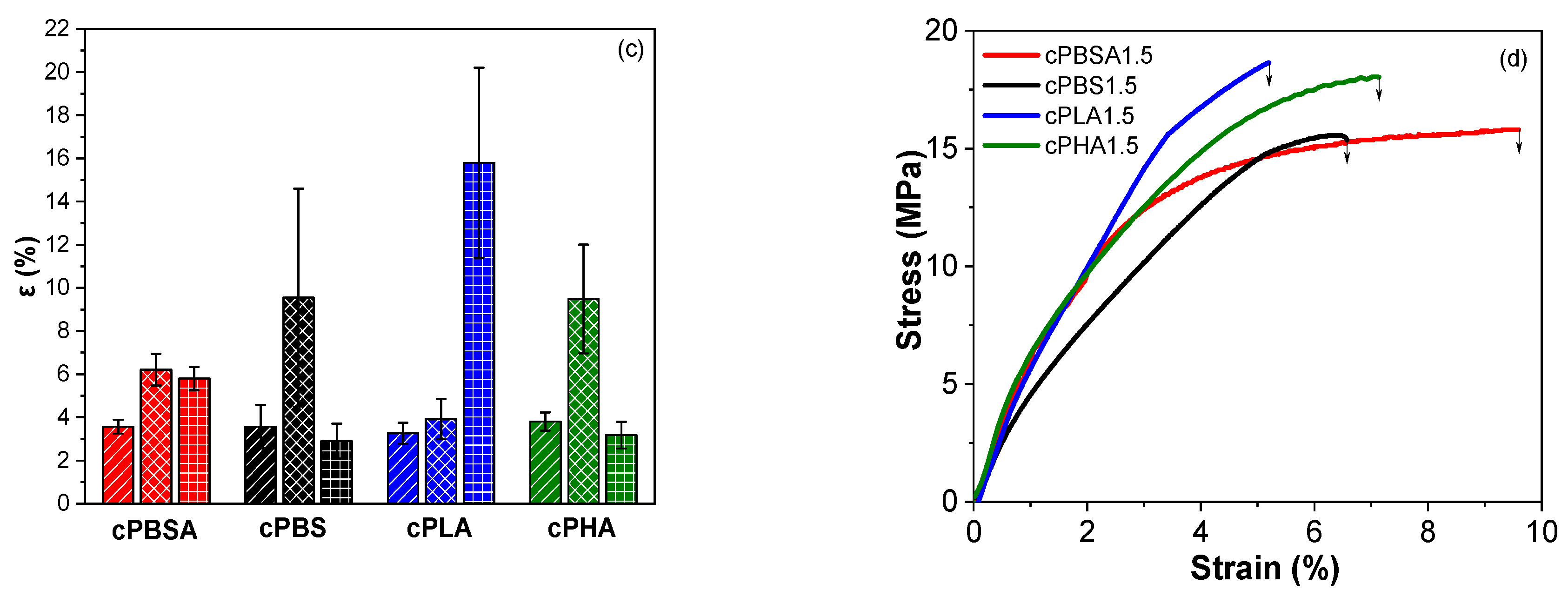
3.2. Mechanical Properties
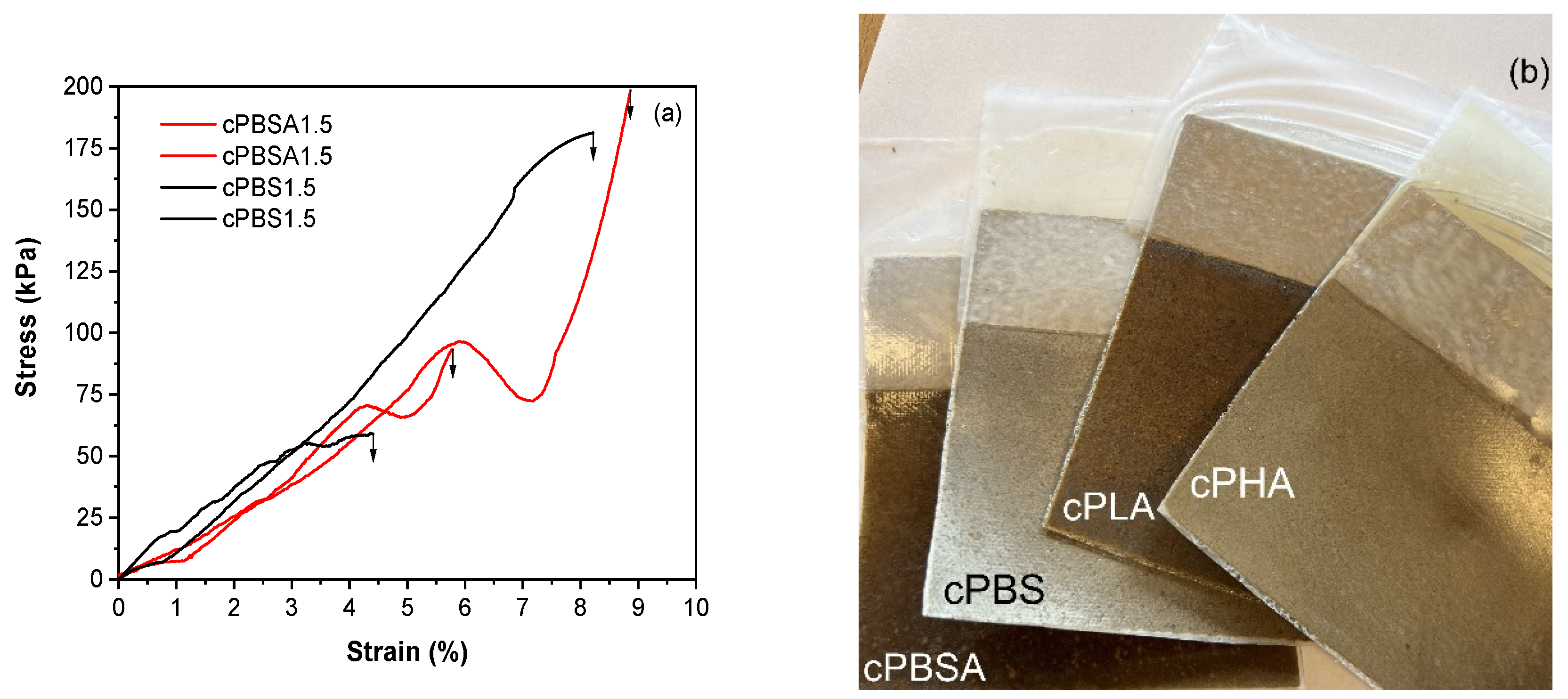
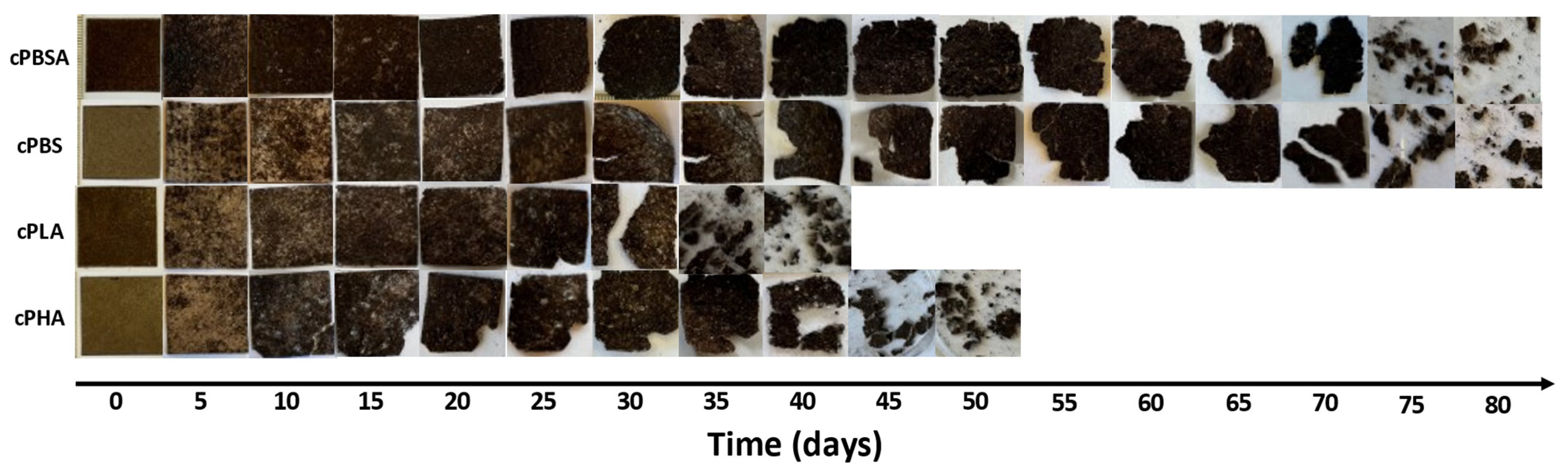
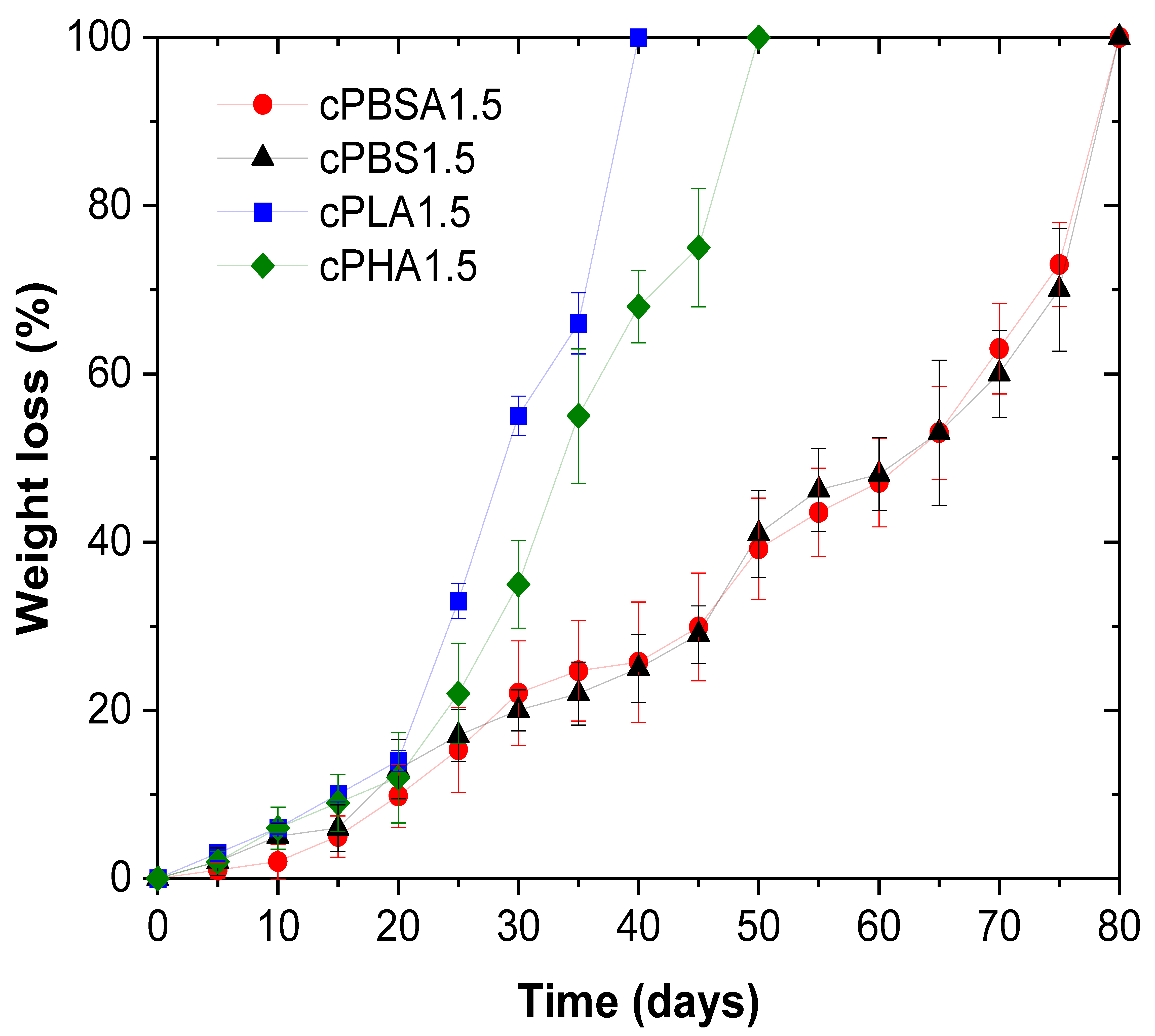
3.3. Biodegradation in Composting Conditions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thurber, H.; Curtzwiler, G.W. Suitability of poly(butylene succinate) as a coating for paperboard convenience food packaging. Int. J. Biobased Plast. 2020, 2, 1–12. [Google Scholar] [CrossRef]
- Reichert, C.L.; Bugnicourt, E.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, A.; Canesi, I.; Braca, F.; Martínez, B.M.; Alonso, R.; Agostinis, L.; et al. Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability. Polymers 2020, 12, 1558. [Google Scholar] [CrossRef] [PubMed]
- Puchalski, M.; Szparaga, G.; Biela, T.; Gutowska, A.; Sztajnowski, S.; Krucińska, I. Molecular and Supramolecular Changes in Polybutylene Succinate (PBS) and Polybutylene Succinate Adipate (PBSA) Copolymer during Degradation in Various Environmental Conditions. Polymers 2018, 10, 251. [Google Scholar] [CrossRef]
- Qasim, U.; Osman, A.I.; Al-Muhtaseb, A.a.H.; Farrell, C.; Al-Abri, M.; Ali, M.; Vo, D.-V.N.; Jamil, F.; Rooney, D.W. Renewable cellulosic nanocomposites for food packaging to avoid fossil fuel plastic pollution: A review. Environ. Chem. Lett. 2021, 19, 613–641. [Google Scholar] [CrossRef]
- Rani, M.; Marchesi, C.; Federici, S.; Rovelli, G.; Alessandri, I.; Vassalini, I.; Ducoli, S.; Borgese, L.; Zacco, A.; Bilo, F.; et al. Miniaturized Near-Infrared (MicroNIR) Spectrometer in Plastic Waste Sorting. Materials 2019, 12, 2740. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef]
- European Commission. Environmental Impact of Waste Management—Revision of EU Waste Framework. Available online: https://ec.europa.eu/info/law/better-regulation/have-your-say/initiatives/13225-Environmental-impact-of-waste-management-revision-of-EU-waste-framework_en (accessed on 17 August 2022).
- European Bioplastics. Revision of the Packaging and Packaging Waste Directive. Available online: https://www.european-bioplastics.org/policy/revision-of-the-packaging-and-packaging-waste-directive/ (accessed on 17 August 2022).
- Abedi, E.; Hashemi, S.M.B. Lactic acid production—Producing microorganisms and substrates sources-state of art. Heliyon 2020, 6, e04974. [Google Scholar] [CrossRef]
- Roshafima, R.A.; Rahman, W.A.W.A.; Rafiziana, M.K.; Ibrahim, N. Starch Based Biofilms For Green Packaging. Int. J. Mater. Metall. Eng. 2012, 6, 937–941. [Google Scholar] [CrossRef]
- Song, J.H.; Murphy, R.J.; Narayan, R.; Davies, G.B.H. Biodegradable and compostable alternatives to conventional plastics. Phil. Trans. R. Soc. B 2009, 364, 2127–2139. [Google Scholar] [CrossRef]
- Selvamurugan, M.; Sivakumar, P. Bioplastics—An Eco-friendly Alternative to Petrochemical Plastics. Curr. World Environ. 2019, 14, 49–59. [Google Scholar] [CrossRef]
- Glaskova-Kuzmina, T.; Starkova, O.; Gaidukovs, S.; Platnieks, O.; Gaidukova, G. Durability of Biodegradable Polymer Nanocomposites. Polymers 2021, 13, 3375. [Google Scholar] [CrossRef] [PubMed]
- Vilarinho, F.; Sanches Silva, A.; Vaz, M.F.; Farinha, J.P. Nanocellulose in green food packaging. Crit. Rev. Food Sci. Nutr. 2018, 58, 1526–1537. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Garcia, M.D.; Lopez-Rubio, A.; Lagaron, J.M. Natural micro and nanobiocomposites with enhanced barrier properties and novel functionalities for food biopackaging applications. Trends Food Sci. Technol. 2010, 21, 528–536. [Google Scholar] [CrossRef]
- Samantaray, P.K.; Ellingford, C.; Farris, S.; O’Sullivan, D.; Tan, B.; Sun, Z.; McNally, T.; Wan, C. Electron Beam-Mediated Cross-Linking of Blown Film-Extruded Biodegradable PGA/PBAT Blends toward High Toughness and Low Oxygen Permeation. ACS Sustain. Chem. Eng. 2022, 10, 1267–1276. [Google Scholar] [CrossRef]
- Aliotta, L.; Seggiani, M.; Lazzeri, A.; Gigante, V.; Cinelli, P. A Brief Review of Poly (Butylene Succinate) (PBS) and Its Main Copolymers: Synthesis, Blends, Composites, Biodegradability, and Applications. Polymers 2022, 14, 844. [Google Scholar] [CrossRef]
- Jambunathan, P.; Zhang, K. Engineered biosynthesis of biodegradable polymers. J. Ind. Microbiol. Biotechnol. 2016, 43, 1037–1058. [Google Scholar] [CrossRef]
- Rafiqah, S.A.; Khalina, A.; Harmaen, A.S.; Tawakkal, I.A.; Zaman, K.; Asim, M.; Nurrazi, M.N.; Lee, C.H. A Review on Properties and Application of Bio-Based Poly(Butylene Succinate). Polymers 2021, 13, 1436. [Google Scholar] [CrossRef]
- Prendiz, J.; Vega-Baudrit, J.; Mena, M. Polylactic Acid (PLA) As A Bioplastic And Its Possible Applications In The Food Industry. Food Sci. Nutr. 2019, 5, 048. [Google Scholar] [CrossRef]
- European Environment Agency. Bio-Waste in Europe: Turning Challenges into Opportunities; Publications Office of the European Union: Luxembourg, 2020; Available online: https://www.eea.europa.eu/publications/bio-waste-in-europe (accessed on 17 August 2022).
- Filiciotto, L.; Rothenberg, G. Biodegradable Plastics: Standards, Policies, and Impacts. ChemSusChem 2021, 14, 56–72. [Google Scholar] [CrossRef]
- Lee, S.Y. Plastic bacteria? Progress and prospects for polyhydroxyalkanoate production in bacteria. Trends Biotechnol. 1996, 14, 431–438. [Google Scholar] [CrossRef]
- Dilkes-Hoffman, L.S.; Lant, P.A.; Laycock, B.; Pratt, S. The rate of biodegradation of PHA bioplastics in the marine environment: A meta-study. Mar. Pollut. Bull. 2019, 142, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Meereboer, K.W.; Misra, M.; Mohanty, A.K. Review of recent advances in the biodegradability of polyhydroxyalkanoate (PHA) bioplastics and their composites. Green Chem. 2020, 22, 5519–5558. [Google Scholar] [CrossRef]
- Pabortsava, K.; Lampitt, R.S. High concentrations of plastic hidden beneath the surface of the Atlantic Ocean. Nat. Commun. 2020, 11, 4073. [Google Scholar] [CrossRef] [PubMed]
- Platnieks, O.; Gaidukovs, S.; Kumar Thakur, V.; Barkane, A.; Beluns, S. Bio-based poly (butylene succinate): Recent progress, challenges and future opportunities. Eur. Polym. J. 2021, 161, 110855. [Google Scholar] [CrossRef]
- Su, S.; Kopitzky, R.; Tolga, S.; Kabasci, S. Polylactide (PLA) and Its Blends with Poly(butylene succinate) (PBS): A Brief Review. Polymers 2019, 11, 1193. [Google Scholar] [CrossRef]
- Puekpoonpoal, N.; Phattarateera, S.; Kerddonfag, N.; Aht-Ong, D. Morphology development of PLAs with different stereo-regularities in ternary blend PBSA/PBS/PLA films. Polym. Plast. Technol. Mat. 2021, 60, 1672–1685. [Google Scholar] [CrossRef]
- Kedzierski, M.; Frère, D.; Le Maguer, G.; Bruzaud, S. Why is there plastic packaging in the natural environment? Understanding the roots of our individual plastic waste management behaviours. Sci. Total Environ. 2020, 740, 139985. [Google Scholar] [CrossRef]
- Hervy, M.; Blaker, J.J.; Braz, A.L.; Lee, K.-Y. Mechanical response of multi-layer bacterial cellulose nanopaper reinforced polylactide laminated composites. Compos. Part A Appl. Sci. 2018, 107, 155–163. [Google Scholar] [CrossRef]
- Hervy, M.; Bock, F.; Lee, K.-Y. Thinner and better: (Ultra-)low grammage bacterial cellulose nanopaper-reinforced polylactide composite laminates. Compos. Sci. Technol. 2018, 167, 126–133. [Google Scholar] [CrossRef]
- Dilkes-Hoffman, L.S.; Pratt, S.; Lant, P.A.; Levett, I.; Laycock, B. Polyhydroxyalkanoate coatings restrict moisture uptake and associated loss of barrier properties of thermoplastic starch films. J. Appl. Polym. Sci. 2018, 135, 46379. [Google Scholar] [CrossRef]
- Melendez-Rodriguez, B.; Torres-Giner, S.; Angulo, I.; Pardo-Figuerez, M.; Hilliou, L.; Escuin, J.M.; Cabedo, L.; Nevo, Y.; Prieto, C.; Lagaron, J.M. High-Oxygen-Barrier Multilayer Films Based on Polyhydroxyalkanoates and Cellulose Nanocrystals. Nanomaterials 2021, 11, 1443. [Google Scholar] [CrossRef] [PubMed]
- Beluns, S.; Gaidukovs, S.; Platnieks, O.; Barkane, A.; Gaidukova, G.; Grase, L.; Nabels-Sneiders, M.; Kovalovs, A.; Thakur, V.K. Clean manufacturing of cellulose nanopapers by incorporating lignin and xylan as sustainable additives. Carbohydr. Polym. Technol. Appl. 2022, 3, 100207. [Google Scholar] [CrossRef]
- Borch, J. Thermodynamics of polymer-paper adhesion: A review. J. Adhes. Sci. Technol. 1991, 5, 523–541. [Google Scholar] [CrossRef]
- Mokhena, T.C.; Sadiku, E.R.; Mochane, M.J.; Ray, S.S.; John, M.J.; Mtibe, A. Mechanical properties of cellulose nanofibril papers and their bionanocomposites: A review. Carbohydr. Polym. 2021, 273, 118507. [Google Scholar] [CrossRef] [PubMed]
- Haslach, H.W. The Moisture and Rate-Dependent Mechanical Properties of Paper: A Review. Mech. Time-Depend. Mater. 2000, 4, 169–210. [Google Scholar] [CrossRef]
- Boopathy, R. Factors limiting bioremediation technologies. Bioresour. Technol. 2000, 74, 63–67. [Google Scholar] [CrossRef]
- Da Luz, J.M.R.; Paes, S.A.; Nunes, M.D.; da Silva, M.d.C.S.; Kasuya, M.C.M. Degradation of Oxo-Biodegradable Plastic by Pleurotus ostreatus. PLoS ONE 2013, 8, e69386. [Google Scholar] [CrossRef]
- Ishigaki, T.; Sugano, W.; Nakanishi, A.; Tateda, M.; Ike, M.; Fujita, M. The degradability of biodegradable plastics in aerobic and anaerobic waste landfill model reactors. Chemosphere 2004, 54, 225–233. [Google Scholar] [CrossRef]
- Gutierrez-Wing, M.T.; Stevens, B.E.; Theegala, C.S.; Negulescu, I.I.; Rusch, K.A. Aerobic Biodegradation of Polyhydroxybutyrate in Compost. Environ. Eng. Sci. 2011, 28, 477–488. [Google Scholar] [CrossRef]
- Kunioka, M.; Ninomiya, F.; Funabashi, M. Biodegradation of poly(lactic acid) powders proposed as the reference test materials for the international standard of biodegradation evaluation methods. Polym. Degrad. Stab. 2006, 91, 1919–1928. [Google Scholar] [CrossRef]
- Kunioka, M.; Ninomiya, F.; Funabashi, M. Biodegradation of Poly(butylene succinate) Powder in a Controlled Compost at 58 °C Evaluated by Naturally-Occurring Carbon 14 Amounts in Evolved CO2 Based on the ISO 14855-2 Method. Int. J. Mol. Sci. 2009, 10, 4267–4283. [Google Scholar] [CrossRef] [PubMed]
- Platnieks, O.; Sereda, A.; Gaidukovs, S.; Thakur, V.K.; Barkane, A.; Gaidukova, G.; Filipova, I.; Ogurcovs, A.; Fridrihsone, V. Adding value to poly (butylene succinate) and nanofibrillated cellulose-based sustainable nanocomposites by applying masterbatch process. Ind. Crops Prod. 2021, 169, 113669. [Google Scholar] [CrossRef]
- Samantaray, P.K.; Little, A.; Wemyss, A.M.; Iacovidou, E.; Wan, C. Design and Control of Compostability in Synthetic Biopolyesters. ACS Sustain. Chem. Eng. 2021, 9, 9151–9164. [Google Scholar] [CrossRef]
| Sample | Lamination Pressure, MT |
|---|---|
| cPBSA0.5 | 0.5 |
| cPBSA1.5 | 1.5 |
| cPBSA3.0 | 3.0 |
| cPBS0.5 | 0.5 |
| cPBS1.5 | 1.5 |
| cPBS3.0 | 3.0 |
| cPLA0.5 | 0.5 |
| cPLA1.5 | 1.5 |
| cPLA3.0 | 3.0 |
| cPHA0.5 | 0.5 |
| cPHA1.5 | 1.5 |
| cPHA3.0 | 3.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nabels-Sneiders, M.; Platnieks, O.; Grase, L.; Gaidukovs, S. Lamination of Cast Hemp Paper with Bio-Based Plastics for Sustainable Packaging: Structure-Thermomechanical Properties Relationship and Biodegradation Studies. J. Compos. Sci. 2022, 6, 246. https://doi.org/10.3390/jcs6090246
Nabels-Sneiders M, Platnieks O, Grase L, Gaidukovs S. Lamination of Cast Hemp Paper with Bio-Based Plastics for Sustainable Packaging: Structure-Thermomechanical Properties Relationship and Biodegradation Studies. Journal of Composites Science. 2022; 6(9):246. https://doi.org/10.3390/jcs6090246
Chicago/Turabian StyleNabels-Sneiders, Martins, Oskars Platnieks, Liga Grase, and Sergejs Gaidukovs. 2022. "Lamination of Cast Hemp Paper with Bio-Based Plastics for Sustainable Packaging: Structure-Thermomechanical Properties Relationship and Biodegradation Studies" Journal of Composites Science 6, no. 9: 246. https://doi.org/10.3390/jcs6090246
APA StyleNabels-Sneiders, M., Platnieks, O., Grase, L., & Gaidukovs, S. (2022). Lamination of Cast Hemp Paper with Bio-Based Plastics for Sustainable Packaging: Structure-Thermomechanical Properties Relationship and Biodegradation Studies. Journal of Composites Science, 6(9), 246. https://doi.org/10.3390/jcs6090246








