Advances in Manufacturing Composite Carbon Nanofiber-Based Aerogels
Abstract
1. Introduction
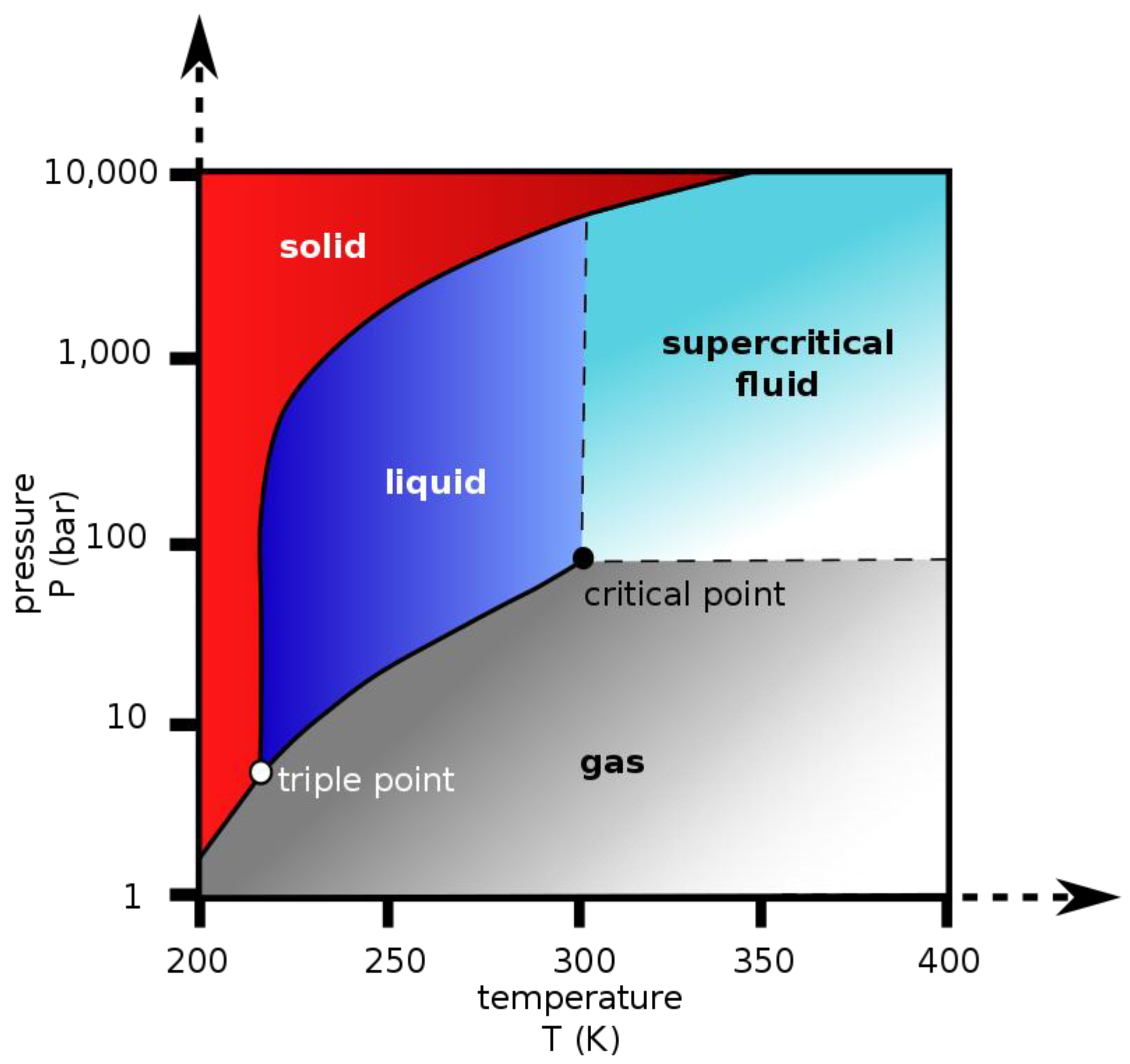
2. Aerogel Processing and Manufacturing Technology
3. Materials and Manufacturing Processes for Composite Carbon Nanofiber Aerogels
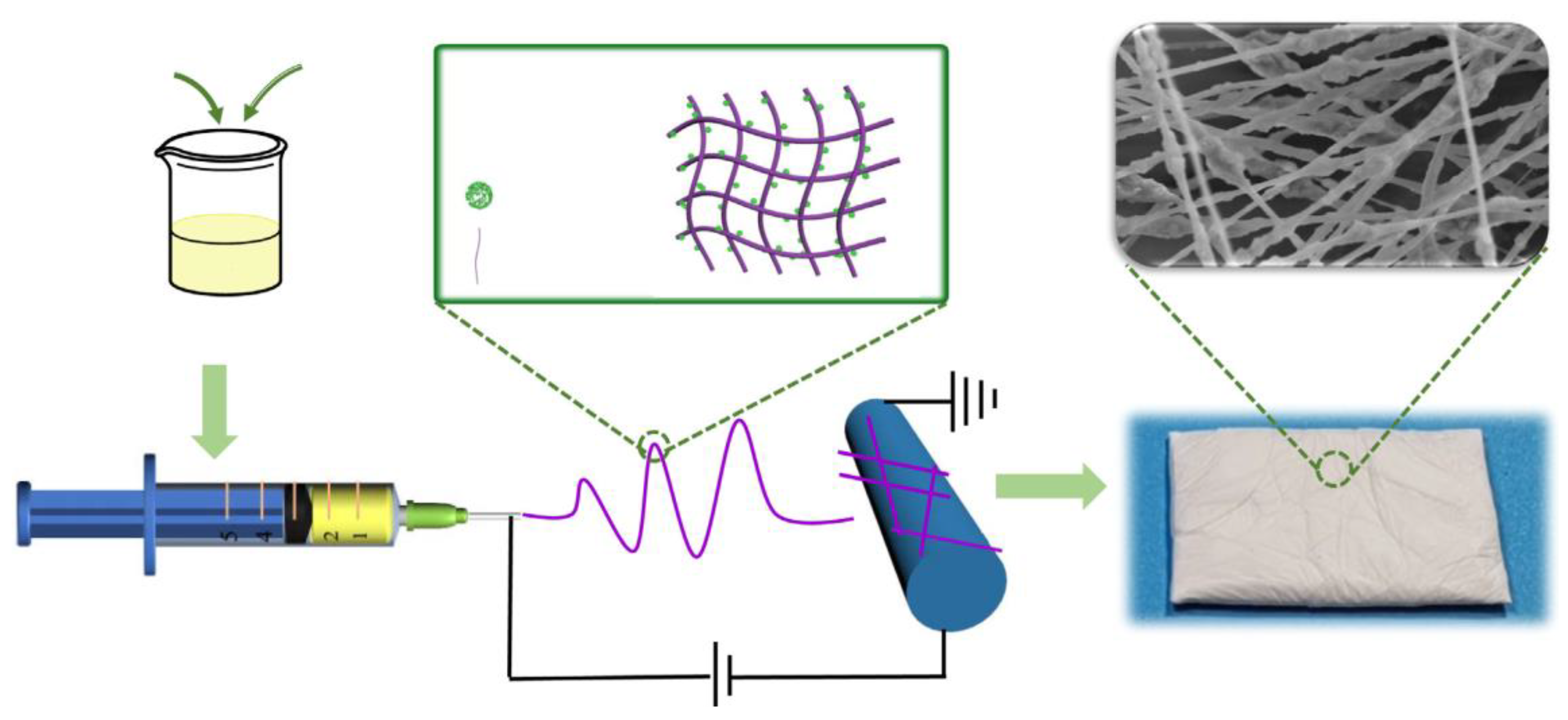
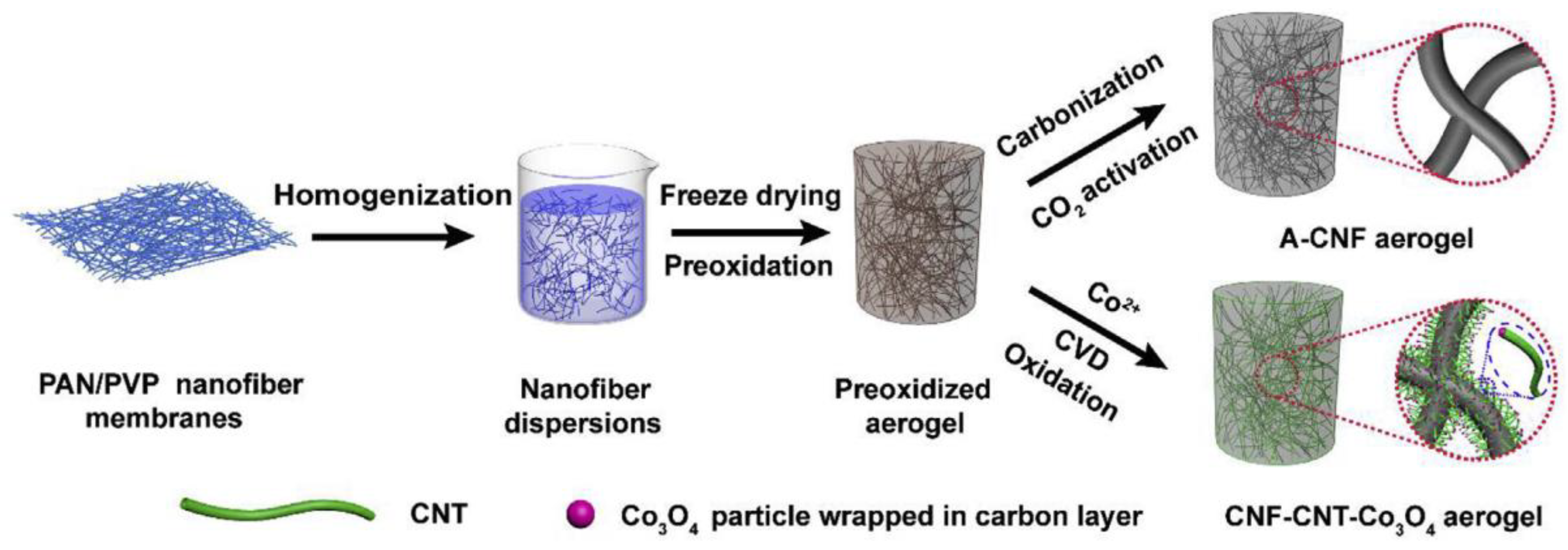
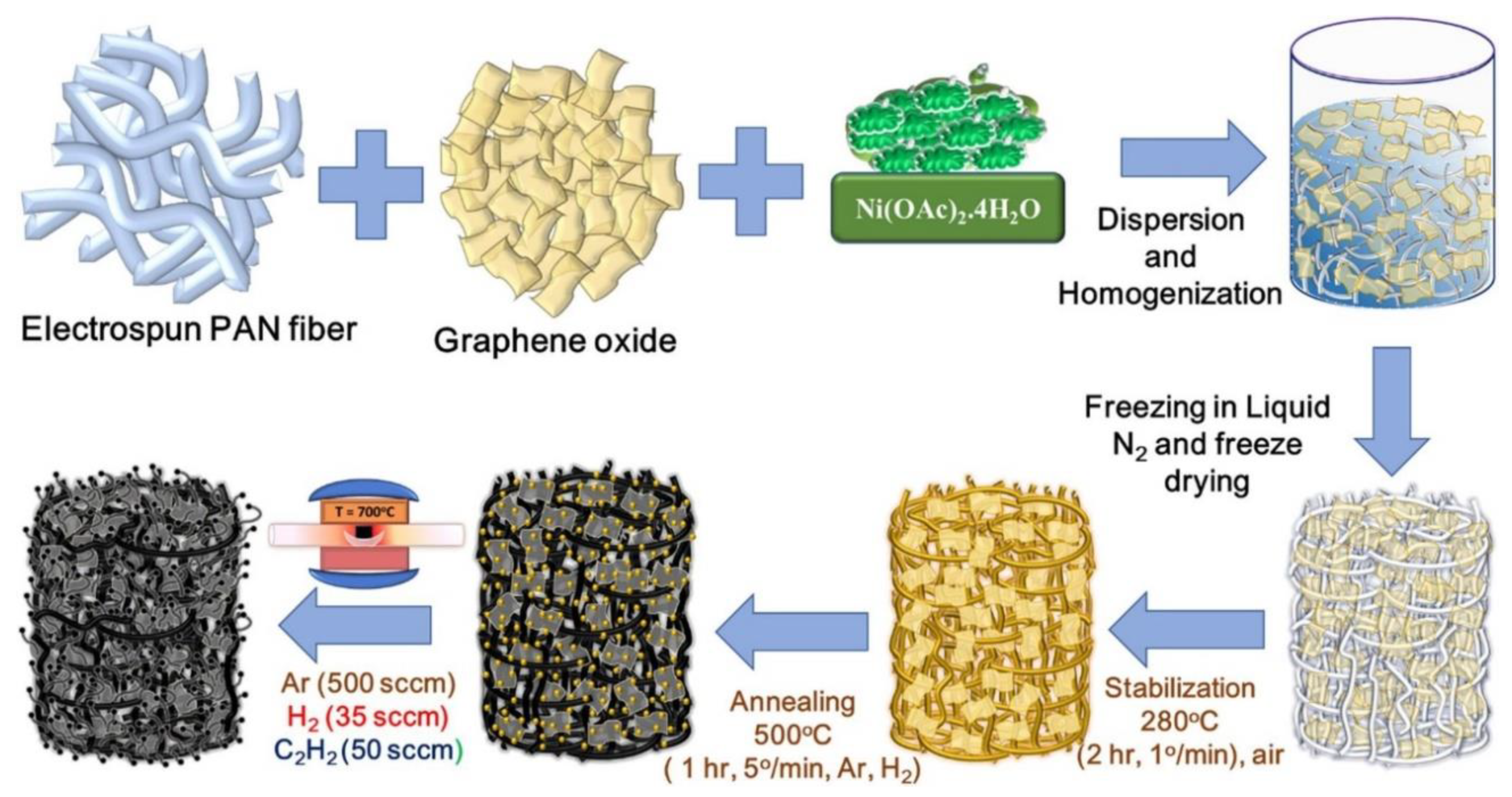
3.1. Polyacrylonitrile (PAN) Derived Composite Carbon Nanofiber Aerogels
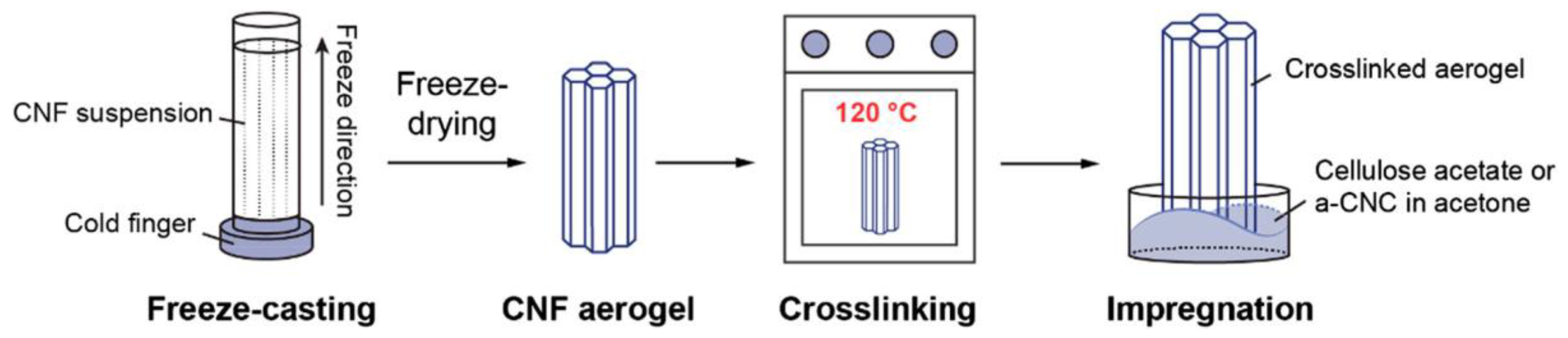
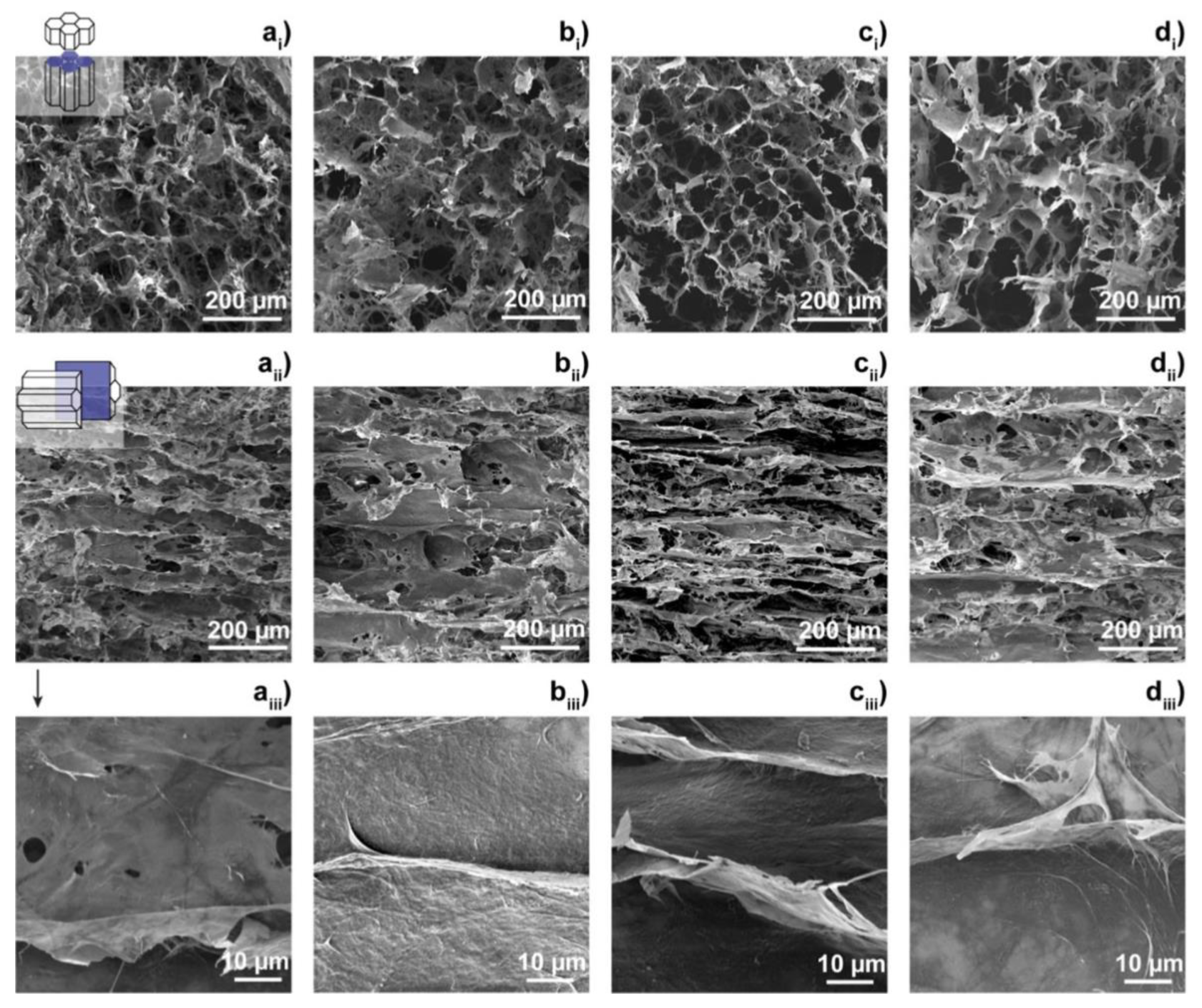
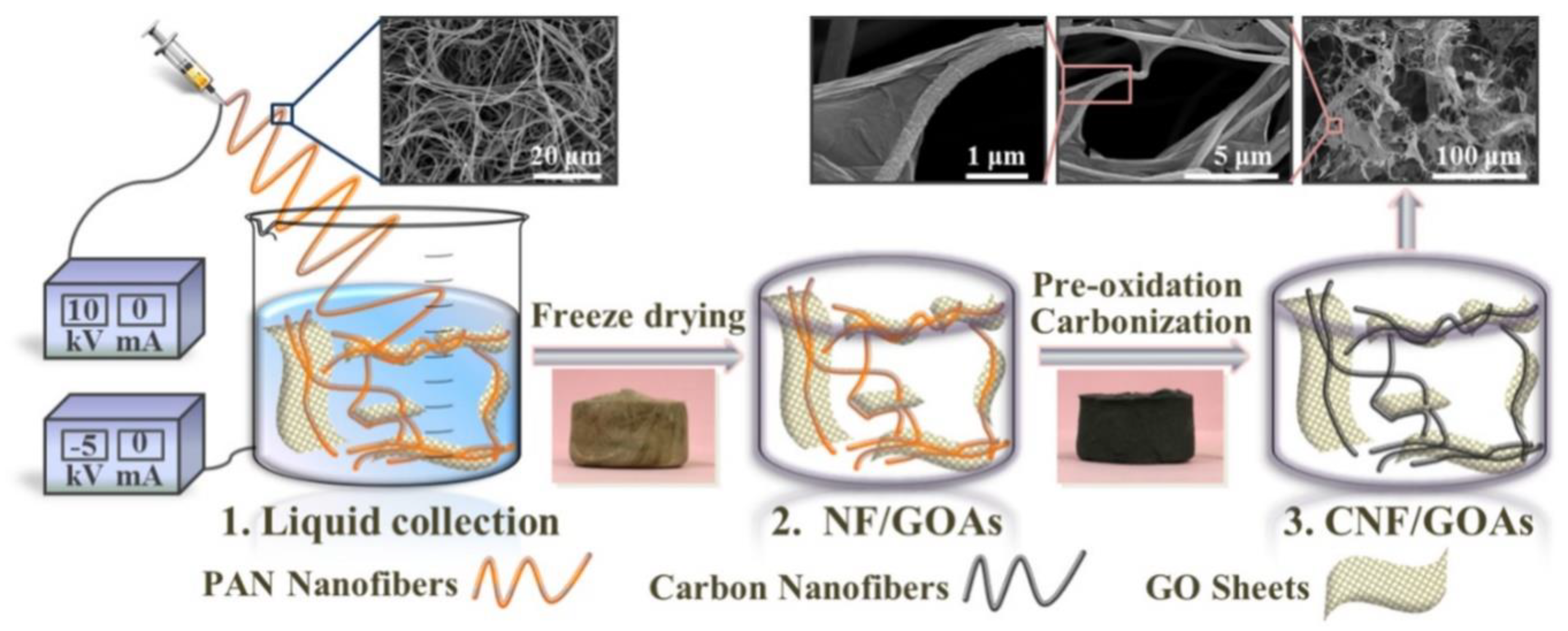
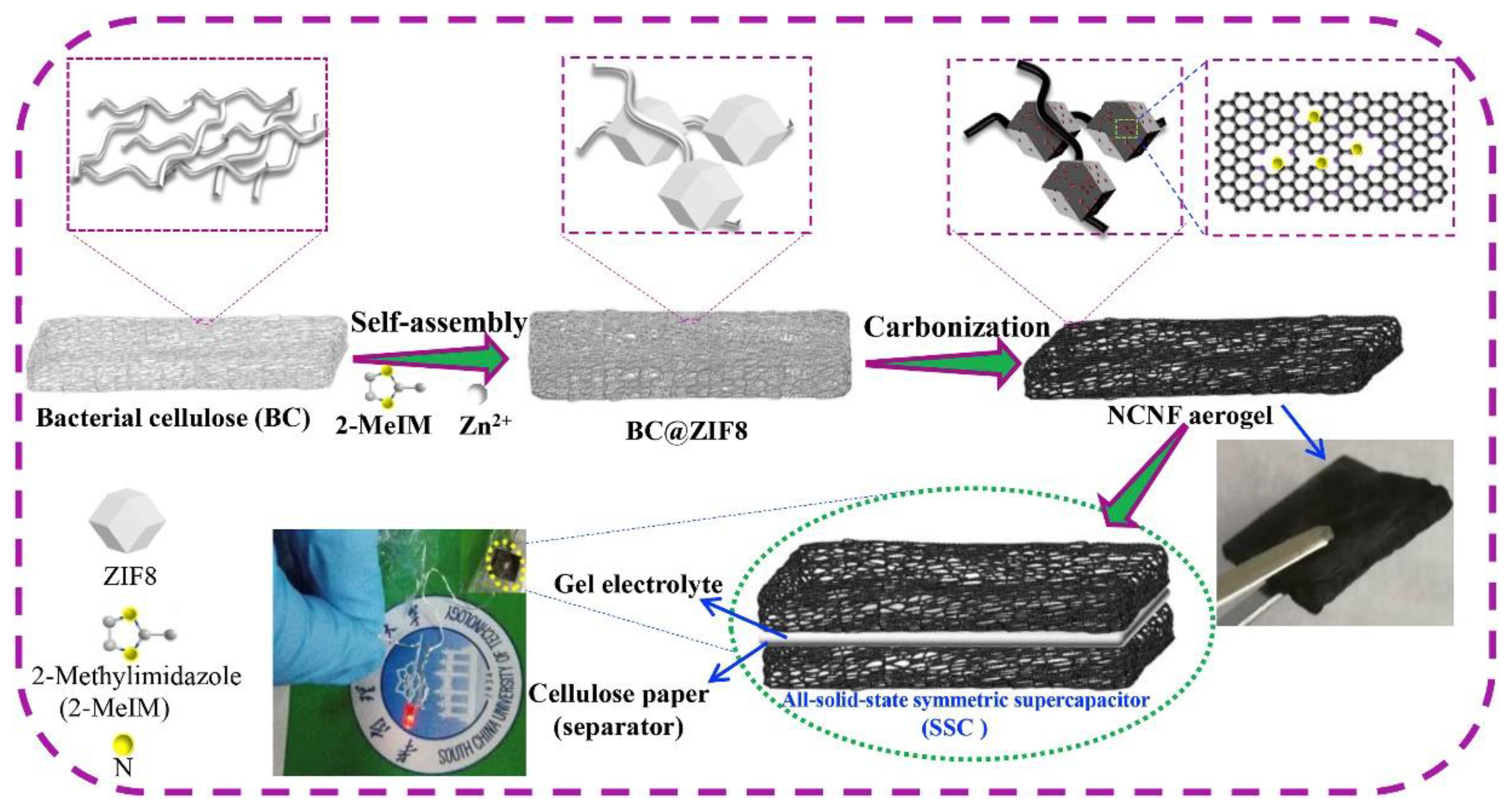
3.2. Cellulose (CE) Based Composite Carbon Nanofiber Aerogels
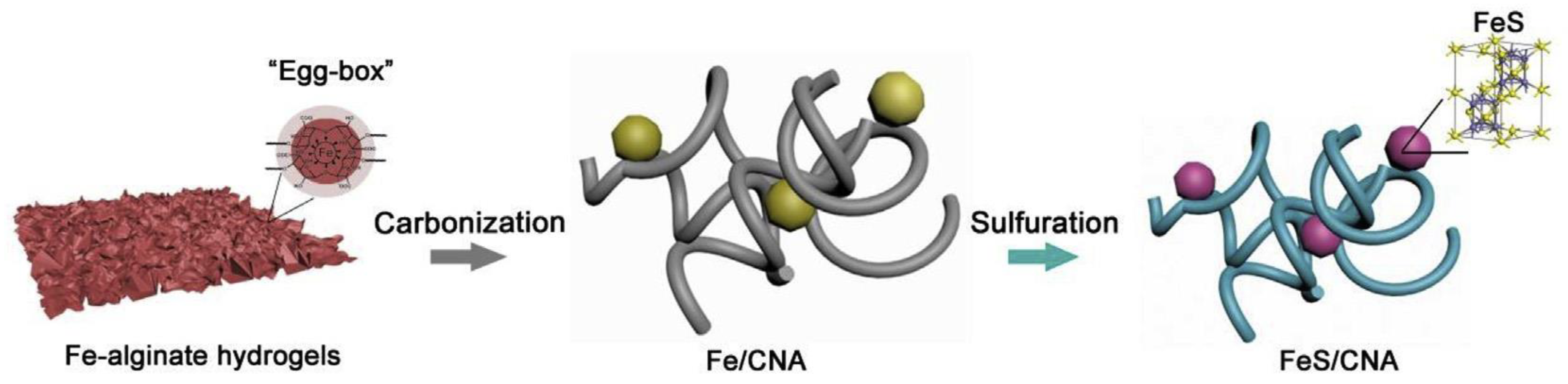
3.3. Alginate Derived Composite Carbon Nanofiber Aerogels
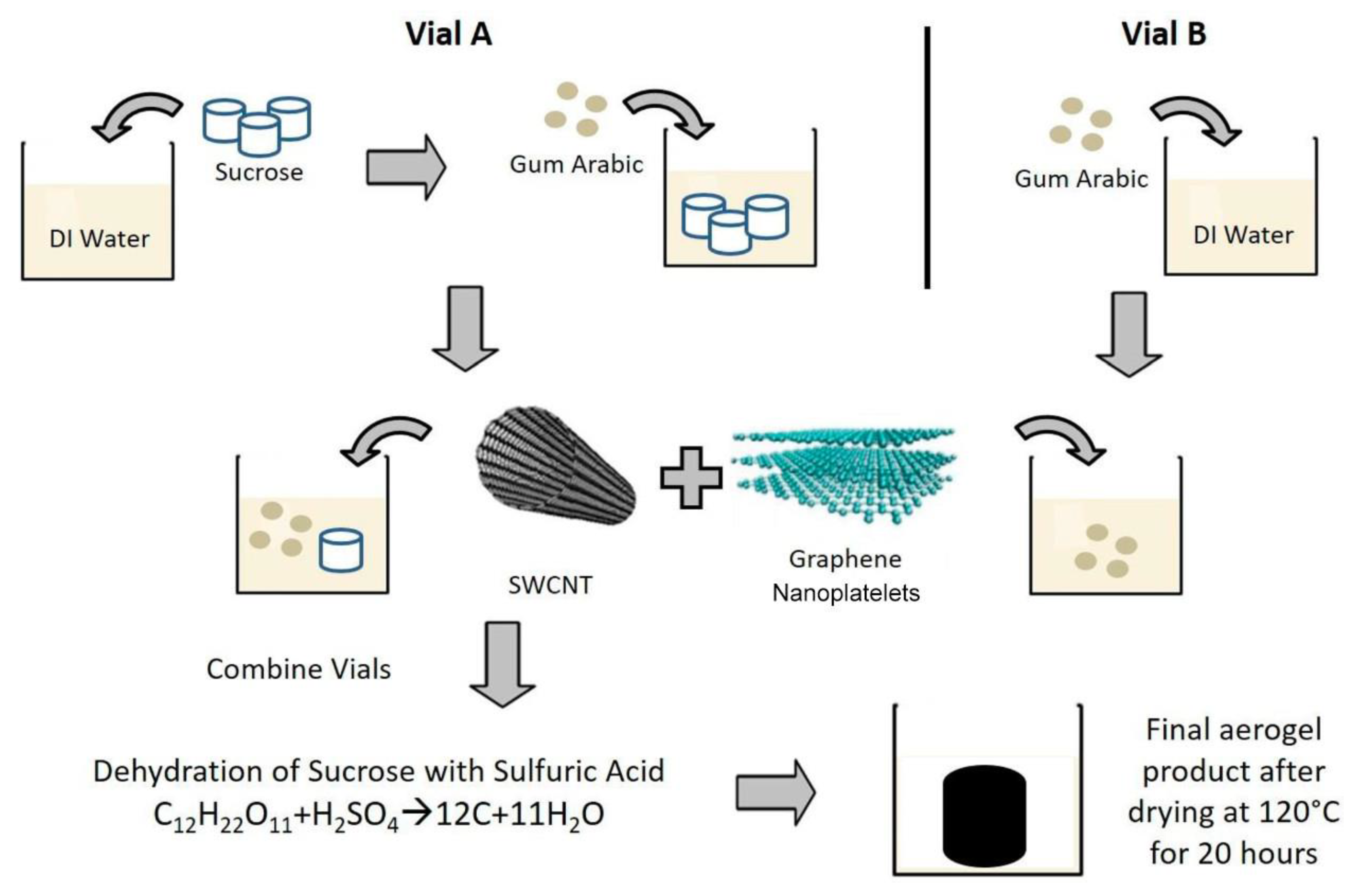
3.4. Biomass/Renewable Resources Derived Composite Carbon Nanofiber Aerogels
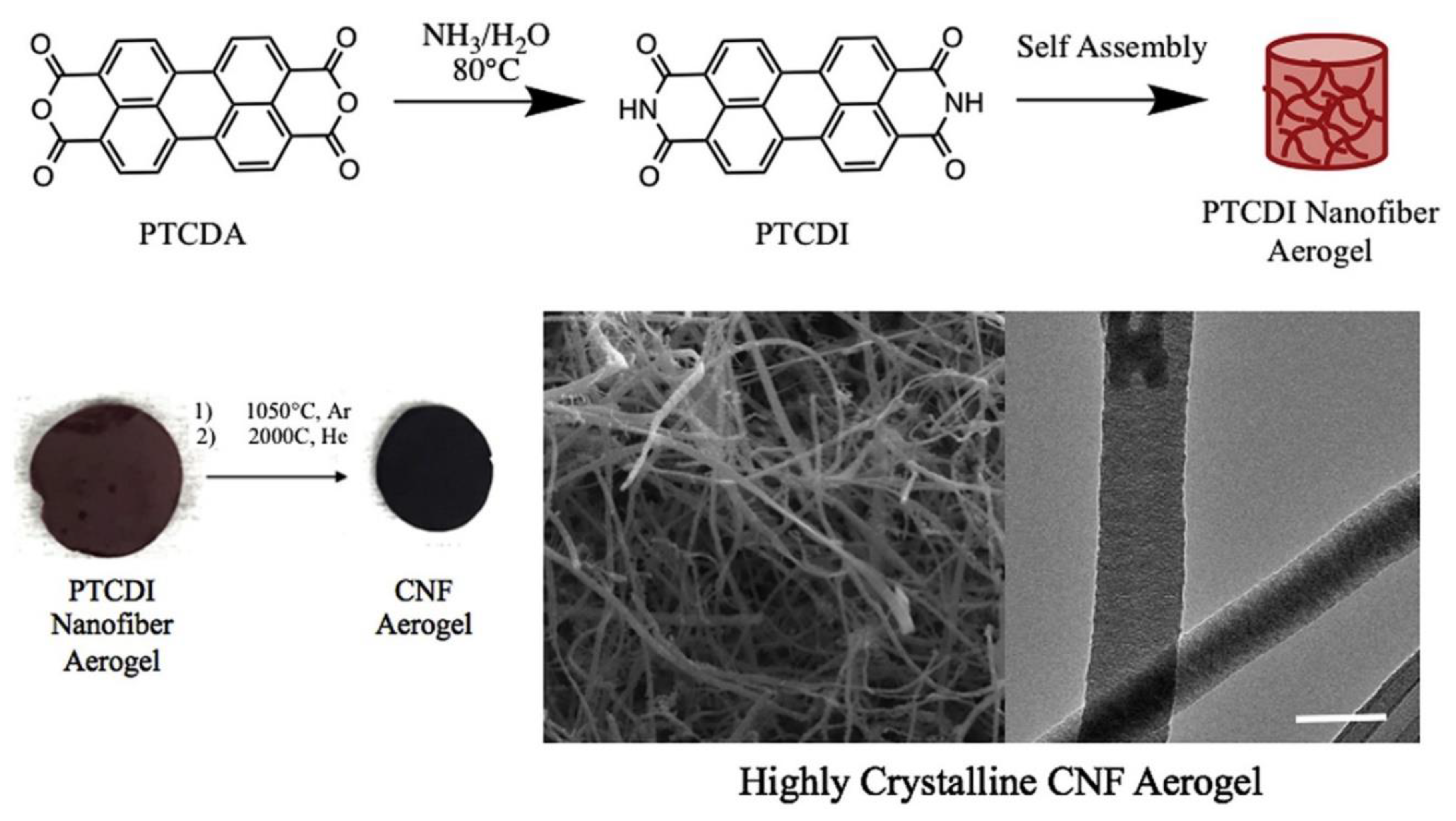
3.5. Template Derived Composite Carbon Nanofiber Aerogels
4. Modeling and Characterization
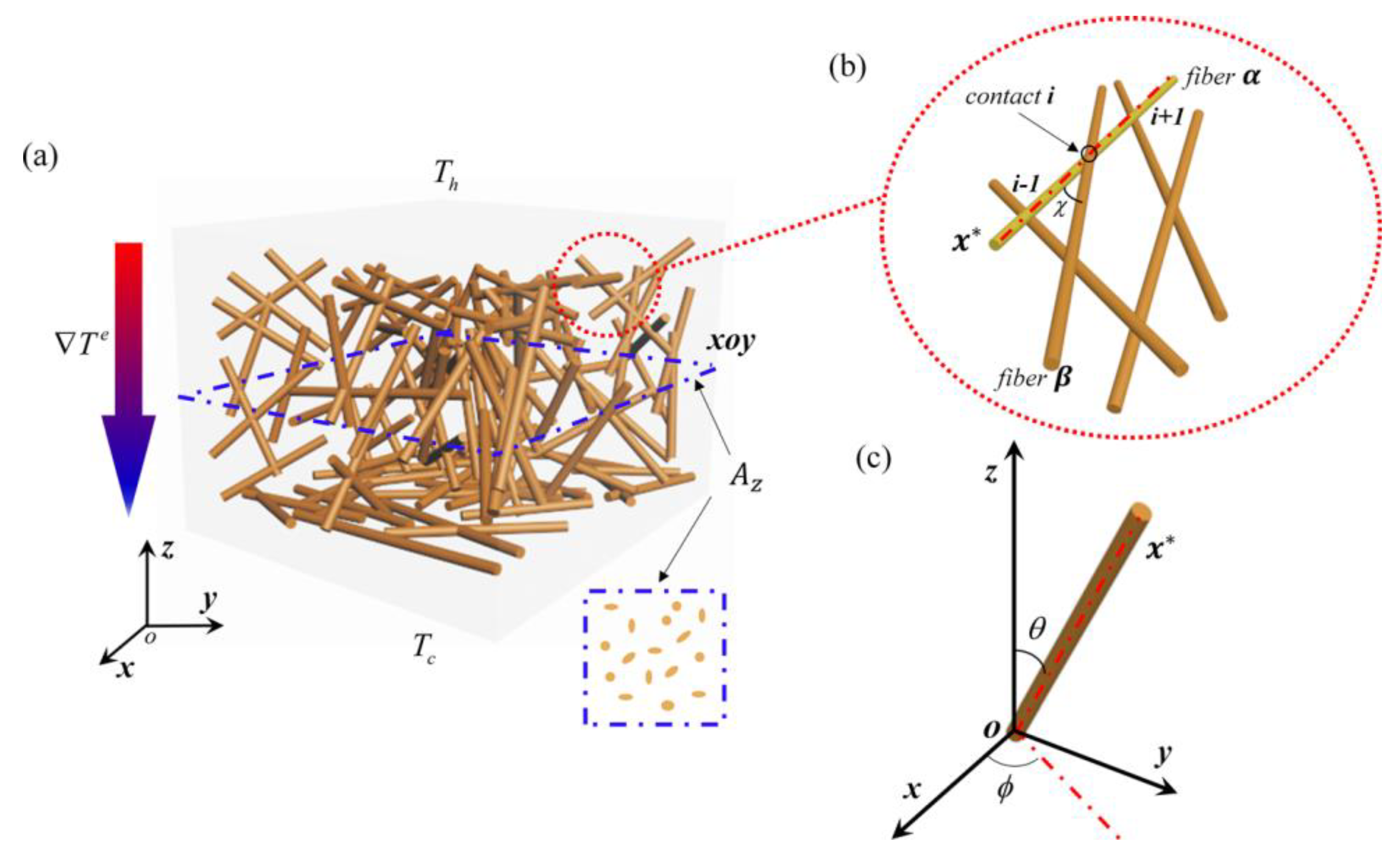
4.1. Modeling the Distribution of Particles in Composite Carbon Nanofibers
4.2. Modeling the Transport Behavior of the Composite Nanofiber Aerogels
4.3. Thermoelectric Property Measurement and Transport Behavior Characterization
4.4. Electrical and Thermal Conductivity Measurement
4.5. Effect of Particle on the Carbonization Kinetics of Carbon Nanofibers
5. Applications
5.1. Supercapacitors
5.2. Secondary Batteries
5.3. Water Purification
5.4. Catalysis
6. Perspectives to Challenges, Opportunities, and Future Directions
7. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Kistler, S.S. Coherent expanded aerogels and jellies. Nature 1931, 127, 741. [Google Scholar] [CrossRef]
- Kistler, S.S. Coherent expanded aerogels. J. Phys. Chem. 1932, 36, 52–64. [Google Scholar] [CrossRef]
- Khalily, M.A.; Eren, H.; Akbayrak, S.; Susapto, H.H.; Biyikli, N.; Ozkar, S.; Guler, M.O. Facile synthesis of three-dimensional Pt-TiO2 nano-networks: A highly active catalyst for the hydrolytic dehydrogenation of ammonia-borane. Angew. Chem. Int. Ed. 2016, 55, 12257–12261. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, J.S.; Liu, Z.K.; Xu, H.Y.; Zheng, Y.H.; Zhong, J.S.; Yang, Q.L.; Tian, H.F.; Shi, Z.Q.; Yao, J.L.; et al. Facile fabrication of Fe3O4 nanoparticle/carbon nanofiber aerogel from Fe-ion cross-linked cellulose nanofibrils as anode for lithium-ion battery with superhigh capacity. J. Alloys Compd. 2020, 829, 154541. [Google Scholar] [CrossRef]
- Finney, B.; Jacobs, M. Commons, Image: Carbon Dioxide Pressure-Temperature Phase Diagram.jpg, CC0. Available online: https://commons.wikimedia.org/w/index.php?curid=4315735 (accessed on 12 May 2019).
- Barrios, E.; Fox, D.; Sip, Y.Y.L.; Catarata, R.; Calderon, J.E.; Azim, N.; Afrin, S.; Zhang, Z.Y.; Zhai, L. Nanomaterials in advanced, high-performance aerogel composites: A review. Polymers 2019, 11, 726. [Google Scholar] [CrossRef] [PubMed]
- Beckman, E.J. Supercritical or near-critical CO2 in green chemical synthesis and processing. J. Supercrit. Fluids 2004, 28, 121–191. [Google Scholar] [CrossRef]
- Joung, S.N.; Yoo, C.W.; Shin, H.Y.; Kim, S.Y.; Yoo, K.-P.; Lee, C.S.; Huh, W.S. Measurements and correlation of high-pressure VLE of binary CO2–alcohol systems (methanol, ethanol, 2-methoxyethanol and 2-ethoxyethanol). Fluid Phase Equilib. 2001, 185, 219–230. [Google Scholar] [CrossRef]
- Matsuyama, K.; Morotomi, K.; Inoue, S.; Nakashima, M.; Nakashima, H.; Okuyama, T.; Kato, T.; Muto, H.; Sugiyama, H. Antibacterial and antifungal properties of Ag nanoparticle-loaded cellulose nanofiber aerogels prepared by supercritical CO2 drying. J. Supercrit. Fluids 2019, 143, 1–7. [Google Scholar] [CrossRef]
- Wang, D.; Peng, H.Y.; Yu, B.; Zhou, K.Q.; Pan, H.F.; Zhang, L.P.; Li, M.; Liu, M.M.; Tian, A.L.; Fu, S.H. Biomimetic structural cellulose nanofiber aerogels with exceptional mechanical, flame-retardant and thermal-insulating properties. Chem. Eng. J. 2020, 389, 124449. [Google Scholar] [CrossRef]
- Yu, Y.X.; Ma, Q.Y.; Zhang, J.B.; Liu, G.B. Electrospun SiO2 aerogel/polyacrylonitrile composited nanofibers with enhanced adsorption performance of volatile organic compounds. Appl. Surf. Sci. 2020, 512, 145697. [Google Scholar] [CrossRef]
- Qiu, J.L.; Zheng, W.T.; Yuan, R.R.; Yue, C.L.; Li, D.W.; Liu, F.Q.; Zhu, J.J. A novel 3D nanofibrous aerogel-based MoS2@Co3S4 heterojunction photocatalyst for water remediation and hydrogen evolution under simulated solar irradiation. Appl. Catal. B-Environ. 2020, 264, 118514. [Google Scholar] [CrossRef]
- Wei, J.Y.; Geng, S.Y.; Hedlund, J.; Oksman, K. Lightweight, flexible, and multifunctional anisotropic nanocellulose-based aerogels for CO2 adsorption. Cellulose 2020, 27, 2695–2707. [Google Scholar] [CrossRef]
- Deville, S.; Saiz, E.; Nalla, R.K.; Tomsia, A.P. Freezing as a path to build complex composites. Science 2006, 311, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.X.; Yu, C.; Panahi, N.; Gan, J.B.; Cheng, W. Processing iron oxide nanoparticle-loaded composite carbon fiber and the photosensitivity characterization. Fibers 2019, 7, 25. [Google Scholar] [CrossRef]
- Zhang, M.; Yang, D.Z.; Zhang, S.Y.; Xu, T.; Shi, Y.Z.; Liu, Y.X.; Chang, W.; Yu, Z.Z. Elastic and hierarchical carbon nanofiber aerogels and their hybrids with carbon nanotubes and cobalt oxide nanoparticles for high-performance asymmetric supercapacitors. Carbon 2020, 158, 873–884. [Google Scholar] [CrossRef]
- Cipriani, E.; Zanetti, M.; Bracco, P.; Brunella, V.; Luda, M.P.; Costa, L. Crosslinking and carbonization processes in PAN films and nanofibers. Polym. Degrad. Stab. 2016, 123, 178–188. [Google Scholar] [CrossRef]
- Schierholz, R.; Kroger, D.; Weinrich, H.; Gehring, M.; Tempel, H.; Kungl, H.; Mayer, J.; Eichel, R.A. The carbonization of polyacrylonitrile-derived electrospun carbon nanofibers studied by in situ transmission electron microscopy. RSC Adv. 2019, 9, 6267–6277. [Google Scholar] [CrossRef]
- Alarifi, I.M.; Khan, W.S.; Asmatulu, R. Synthesis of electrospun polyacrylonitrile-derived carbon fibers and comparison of properties with bulk form. PLoS ONE 2018, 13, e0201345. [Google Scholar] [CrossRef] [PubMed]
- Alarifi, I.M.; Alharbi, A.; Khan, W.S.; Rahman, A.S.; Asmatulu, R. Mechanical and thermal properties of carbonized PAN nanofibers cohesively attached to surface of carbon fiber reinforced composites. Macromol. Symp. 2016, 365, 140–150. [Google Scholar] [CrossRef]
- Ghorpade, R.V.; Cho, D.W.; Hong, S.C. Effect of controlled tacticity of polyacrylonitrile (co)polymers on their thermal oxidative stabilization behaviors and the properties of resulting carbon films. Carbon 2017, 121, 502–511. [Google Scholar] [CrossRef]
- Hameed, N.; Sharp, J.; Nunna, S.; Creighton, C.; Magniez, K.; Jyotishkumar, P.; Salim, N.V.; Fox, B. Structural transformation of polyacrylonitrile fibers during stabilization and low temperature carbonization. Polym. Degrad. Stab. 2016, 128, 39–45. [Google Scholar] [CrossRef]
- Jing, M.; Wang, C.G.; Wang, Q.; Bai, Y.J.; Zhu, B. Chemical structure evolution and mechanism during pre-carbonization of PAN-based stabilized fiber in the temperature range of 350–600 °C. Polym. Degrad. Stab. 2007, 92, 1737–1742. [Google Scholar] [CrossRef]
- Zhang, Y.; Tajaddod, N.; Song, K.; Minus, M.L. Low temperature graphitization of interphase polyacrylonitrile (PAN). Carbon 2015, 91, 479–493. [Google Scholar] [CrossRef]
- Chai, X.; Mi, H.; Zhu, C.; He, C.; Xu, J.; Zhou, X.; Liu, J. Low-temperature thermal stabilization of polyacrylontrile-based precursor fibers towards efficient preparation of carbon fibers with improved mechanical properties. Polymer 2015, 76, 131–139. [Google Scholar] [CrossRef]
- Korobeinyk, A.V.; Whitby, R.L.D.; Mikhalovsky, S.V. High temperature oxidative resistance of polyacrylonitrile-methylmethacrylate copolymer powder converting to a carbonized monolith. Europ. Polym. J. 2012, 48, 97–104. [Google Scholar] [CrossRef]
- Joo, S.; Park, J.Y.; Seo, S.; Moon, S.; Lee, K.J.; An, J.; Lee, C.S.; Bae, J. Study on peculiar carbon pattern formation from polymer blend thin films under electric fields. Thin Solid Film. 2018, 660, 846–851. [Google Scholar] [CrossRef]
- Sinha-Ray, S.; Yarin, A.L.; Pourdeyhimi, B. The production of 100/400 nm inner/outer diameter carbon tubes by solution blowing and carbonization of core–shell nanofibers. Carbon 2010, 48, 3575–3578. [Google Scholar] [CrossRef]
- Yang, G.Z.; Xu, R.S.; Chen, M.; Wang, X.; Ling, L.C.; Zhang, R.; Yang, J.H. Hollow carbon nanospheres prepared by carbonizing polymethylmethacrylate/polyacrylonitrile core/shell polymer particles. New Carbon Mater. 2008, 23, 205–208. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, J.; Zhu, P.; Yan, C.; Jia, H.; Kiyak, Y.; Zang, J.; He, J.; Dirican, M.; Zhang, X. Glass fiber separator coated by porous carbon nanofiber derived from immiscible PAN/PMMA for high-performance lithium-sulfur batteries. J. Memb. Sci. 2018, 552, 31–42. [Google Scholar] [CrossRef]
- Gong, J.; Chen, X.; Tang, T. Recent progress in controlled carbonization of (waste) polymers. Progr. Polym. Sci. 2019, 94, 1–32. [Google Scholar] [CrossRef]
- Rao, M.; Geng, X.; Liao, Y.; Hu, S.; Li, W. Preparation and performance of gel polymer electrolyte based on electrospun polymer membrane and ionic liquid for lithium ion battery. J. Memb. Sci 2012, 399–400, 37–42. [Google Scholar] [CrossRef]
- Nataraj, S.K.; Yang, K.S.; Aminabhavi, T.M. Polyacrylonitrile-based nanofibers-A state-of-the-art review. Progr. Polym. Sci. 2012, 37, 487–513. [Google Scholar] [CrossRef]
- Liu, W.; Wang, W.; Xing, Z.; Qi, Y.; Wu, G. Radiation-induced crosslinking of polyacrylonitrile fibers and the subsequent regulative effect on the peroxidation process. Rad. Phys. Chem. 2012, 81, 622–627. [Google Scholar] [CrossRef]
- Arshad, S.N.; Naraghi, M.; Chasiotis, I. Strong carbon nanofibers from electrospun polyacrylonitrile. Carbon 2011, 49, 1710–1719. [Google Scholar] [CrossRef]
- Lin, S.; Cai, Q.; Ji, J.; Sui, G.; Yu, Y.; Yang, X.; Ma, Q.; Wei, Y.; Deng, X. Electrospun nanofiber reinforced and toughened composites through in situ nano-interface formation. Comp. Sci. Technol. 2008, 68, 3322–3329. [Google Scholar] [CrossRef]
- Xu, T.; Nguyen, A.; Rosas, N.; Flores, I.; Chen, C.; Gan, J.B.; Hamdan, A.S.; Gan, Y.X. Effect of pyrolysis temperature on the electrical property and photosensitivity of a PAN-PMMA derived carbon fiber. ChemEngineering 2019, 3, 86. [Google Scholar] [CrossRef]
- Kshetri, T.; Tran, D.T.; Nguyen, D.C.; Kim, N.H.; Lau, K.T.; Lee, J.H. Ternary graphene-carbon nanofibers-carbon nanotubes structure for hybrid supercapacitor. Chem. Eng. J. 2020, 380, 122543. [Google Scholar] [CrossRef]
- Kshetri, T.; Thanh, T.D.; Singh, S.B.; Kim, N.H.; Lee, J.H. Hierarchical material of carbon nanotubes grown on carbon nanofibers for high performance electrochemical capacitor. Chem. Eng. J. 2018, 345, 39–47. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Rahaman, M.S.A.; Ismail, A.F.; Mustafa, A. A review of heat treatment on polyacrylonitrile fiber. Polym. Degrad. Stab. 2007, 92, 1421–1432. [Google Scholar] [CrossRef]
- Dalton, S.; Heatley, F.; Budd, P.M. Thermal stabilization of polyacrylonitrile fibres. Polymer 1999, 40, 5531–5543. [Google Scholar] [CrossRef]
- Kumar, M.; Ando, Y. Chemical vapor deposition of carbon nanotubes: A review on growth mechanism and mass production. J. Nanosci. Nanotechnol. 2010, 10, 3739–3758. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.Z.; Zhong, L.B.; Dou, S.; Shao, Z.D.; Liu, Q.; Zheng, Y.M. Facile synthesis of electrospun carbon nanofiber/graphene oxide composite aerogels for high efficiency oils absorption. Environ. Int. 2019, 128, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Sriplai, N.; Mongkolthanaruk, W.; Eichhorn, S.J.; Pinitsoontorn, S. Magnetic bacterial cellulose and carbon nanofiber aerogel by simple immersion and pyrolysis. J. Mater. Sci. 2020, 55, 4113–4126. [Google Scholar] [CrossRef]
- Fauziyah, M.; Widiyastuti, W.; Balgis, R.; Setyawan, H. Production of cellulose aerogels from coir fibers via an alkali-urea method for sorption applications. Cellulose 2019, 26, 9583–9598. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, L.; Qing, Y.; Lu, X.H.; Wu, Y.Q.; Yan, N.; Yang, W. Manipulation of nanoplate structures in carbonized cellulose nanofibril aerogel for high-performance supercapacitor. J. Phys. Chem. C 2019, 123, 23374–23381. [Google Scholar] [CrossRef]
- Chen, H.; Liu, T.; Mau, J.R.; Zhang, W.J.; Jiang, Z.J.; Liu, J.; Huang, J.L.; Liu, M.L. Free-standing N-self-doped carbon nanofiber aerogels for high-performance all-solid-state supercapacitors. Nano Energy 2019, 63, 103836. [Google Scholar] [CrossRef]
- Yang, Q.L.; Yang, J.W.; Gao, Z.D.F.; Li, B.; Xiong, C.X. Carbonized cellulose nanofibril/graphene oxide composite aerogels for high-performance supercapacitors. ACS Appl. Energy Mater. 2020, 3, 1145–1151. [Google Scholar] [CrossRef]
- Liu, H.L.; Lv, C.X.; Chen, S.; Song, X.Y.; Liu, B.H.; Sun, J.; Zhang, H.W.; Yang, D.J.; She, X.L.; Zhao, X.L. Fe-alginate biomass-derived FeS/3D interconnected carbon nanofiber aerogels as anodes for high performance sodium-ion batteries. J. Alloys Compd. 2019, 795, 54–59. [Google Scholar] [CrossRef]
- Lv, C.X.; Xu, W.J.; Liu, H.L.; Zhang, L.X.; Chen, S.; Yang, X.F.; Xu, X.J.; Yang, D.J. 3D sulfur and nitrogen codoped carbon nanofiber aerogels with optimized electronic structure and enlarged interlayer spacing boost potassium-ion storage. Small 2019, 15, 1900816. [Google Scholar] [CrossRef]
- Litts, B.S.; Eddy, M.K.; Zaretzky, P.M.; Ferguson, N.N.; Dichiara, A.B.; Rogers, R.E. Construction of a carbon nanomaterial-based nanocomposite aerogel for the removal of organic compounds from water. ACS Appl. Nano Mater. 2018, 1, 4127–4134. [Google Scholar] [CrossRef]
- Takeshita, S.; Akasaka, S.; Yoda, S. Structural and acoustic properties of transparent chitosan aerogel. Mater. Lett. 2019, 254, 258–261. [Google Scholar] [CrossRef]
- Takeshita, S.; Yoda, S. Chitosan aerogels: Transparent, flexible thermal insulators. Chem. Mater. 2015, 27, 7569–7572. [Google Scholar] [CrossRef]
- Kharissova, O.V.; Torres, C.E.I.; Gonzalez, L.T.; Kharisov, B.I. All-carbon hybrid aerogels: Synthesis, properties, and applications. Ind. Eng. Chem. Res. 2019, 58, 16258–16286. [Google Scholar] [CrossRef]
- Liu, X.; Roberts, A.; Ahmed, A.; Wang, Z.; Li, X.; Zhang, H. Carbon nanofibers by pyrolysis of self-assembled perylene diimide derivative gels as supercapacitor electrode materials. J. Mater. Chem. A 2015, 3, 15513–15522. [Google Scholar] [CrossRef]
- Turner, S.; Shevitski, B.; Long, H.; Lorenzo, M.; Marquez, J.; Aloni, S.; Altoe, V.; Worsley, M.A.; Zettl, A. Self-assembly and metal-directed assembly of organic semiconductor aerogels and conductive carbon nanofiber aerogels with controllable nanoscale morphologies. Carbon 2019, 153, 648–656. [Google Scholar] [CrossRef]
- Xia, B.; Lenggoro, I.W.; Okuyama, K. Preparation of Ni particles by ultrasonic spray pyrolysis of NiCl2∙6H2O precursor containing ammonia. J. Mater. Sci. 2001, 36, 1701–1705. [Google Scholar] [CrossRef]
- Obraztsov, A.N. Making graphene on a large scale. Nat. Nanotechnol. 2009, 4, 212–213. [Google Scholar] [CrossRef]
- Ortiz-Medina, J.; Wang, Z.; Cruz-Silva, R.; Morelos-Gomez, A.; Wang, F.; Yao, X.; Terrones, M.; Endo, M. Defect engineering and surface functionalization of nanocarbons for metal-free catalysis. Adv. Mater 2019, 1805717. [Google Scholar] [CrossRef]
- Jiang, Y.; Yang, L.; Sun, T.; Zhao, J.; Lyu, Z.; Zhuo, O.; Wang, X.; Wu, Q.; Ma, J.; Hu, Z. Significant contribution of intrinsic carbon defects to oxygen reduction activity. ACS Catal. 2015, 5, 6707–6712. [Google Scholar] [CrossRef]
- Gao, H.; Liu, C.; Jeong, H.E.; Yang, P. Plasmon-enhanced photocatalytic activity of iron oxide on gold nanopillars. ACS Nano 2012, 6, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, S.W.; Noh, H.; Brudvig, G.W.; Cao, H.; Schmuttenmaer, C.A. Plasmonic enhancement of dye-sensitized solar cells using coreeshelleshell nanostructures. J. Phys. Chem. C 2013, 117, 927–934. [Google Scholar] [CrossRef]
- Huang, J.S.; Wang, D.W.; Hou, H.Q.; You, T.Y. Electrospun palladium nanoparticle-loaded carbon nanofibers and their electrocatalytic activities towards hydrogen peroxide and NADH. Adv. Funct. Mater. 2008, 18, 441–448. [Google Scholar] [CrossRef]
- Guo, Q.H.; Huang, J.S.; You, T.Y. Electrospun palladium nanoparticle-loaded carbon nanofiber for methanol electro-oxidation. Chin. J. Anal. Chem. 2013, 41, 210–214. [Google Scholar] [CrossRef]
- Oh, H.-S.; Kim, H. Efficient synthesis of Pt nanoparticles supported on hydrophobic graphitized carbon nanofibers for electrocatalysts using noncovalent functionalization. Adv. Funct. Mater. 2011, 21, 3954–3960. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, H.B.; Hoster, H.E.; Lou, X.W. Strongly coupled carbon nanofiber-metal oxide coaxial nanocables with enhanced lithium storage properties. Energy Environ. Sci. 2014, 7, 302–305. [Google Scholar] [CrossRef]
- Yang, D.-J.; Kamienchick, I.; Youn, D.Y.; Rothschild, A.; Kim, I.-D. Ultrasensitive and highly selective gas sensors based on electrospun SnO2 nanofibers modified by Pd loading. Adv. Funct. Mater. 2010, 20, 4258–4264. [Google Scholar] [CrossRef]
- Janjua, M.; Nudurupati, S.; Singh, P.; Aubry, N. Electric field-induced self-assembly of micro- and nanoparticles of various shapes at two-fluid interfaces. Electrophoresis 2011, 32, 518–526. [Google Scholar] [CrossRef]
- Hwang, K.; Singh, P.; Aubry, N. Destabilization of Pickering emulsions using external electric fields. Electrophoresis 2010, 31, 850–859. [Google Scholar] [CrossRef]
- Kadaksham, J.; Singh, P.; Aubry, N. Manipulation of particles using dielectrophoresis. Mech. Res. Comm. 2006, 33, 108–122. [Google Scholar] [CrossRef]
- Kadaksham, A.T.J.; Singh, P.; Aubry, N. Dielectrophoresis of nanoparticles. Electrophoresis 2004, 25, 3625–3632. [Google Scholar] [CrossRef] [PubMed]
- Aubry, N.; Singh, P. Influence of particle-particle interactions and particles rotational motion in traveling wave dielectrophoresis. Electrophoresis 2006, 27, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.B. Electromechanics of Particles; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Zhao, X.P.; Huang, C.L.; Liu, Q.K.; Smalyukh, I.I.; Yang, R.G. Thermal conductivity model for nanofiber networks. J. Appl. Phys. 2018, 123, 085103. [Google Scholar] [CrossRef]
- Pan, N. A modified analysis of the microstructural characteristics of general fiber assemblies. Text. Res. J. 1993, 63, 336–345. [Google Scholar] [CrossRef]
- Pan, N.; Gibson, P. Thermal and Moisture Transport in Fibrous Materials; CRC Press: Boca Raton, FL, USA; Woodhead Publishing: Cambridge, UK, 2006; pp. 3–41. [Google Scholar]
- Mott, N.F. Conduction in Non-Crystalline Materials; Clarendon: Oxford, UK, 1987; p. 53. [Google Scholar]
- Gan, Y.X.; Chen, A.D.; Gan, R.N.; Hamdan, A.S. Energy conversion behaviors of antimony telluride particle loaded partially carbonized nanofiber composite mat manufactured by electrohydrodynamic casting. Microelectron. Eng. 2017, 181, 16–21. [Google Scholar] [CrossRef]
- Yuan, L.; Wei, X.; Martinez, J.P.; Yu, C.; Panahi, N.; Gan, J.B.; Zhang, Y.; Gan, Y.X. Reaction spinning titanium dioxide particle-coated carbon fiber for photoelectric energy conversion. Fibers 2019, 7, 49. [Google Scholar] [CrossRef]
- Kansara, S.; Patel, S.; Gan, Y.X.; Jaimes, G.; Gan, J.B. Dye adsorption and electrical property of oxide-loaded carbon fiber made by electrospinning and hydrothermal treatment. Fibers 2019, 7, 74. [Google Scholar] [CrossRef]
- Arabkhani, P.; Asfaram, A. Development of a novel three-dimensional magnetic polymer aerogel as an efficient adsorbent for malachite green removal. J. Hazard. Mater. 2020, 384, 121394. [Google Scholar] [CrossRef]
- Wu, H.Y.; Wang, Z.M.; Kumagai, A.; Endo, T. Amphiphilic cellulose nanofiber-interwoven graphene aerogel monolith for dyes and silicon oil removal. Comp. Sci. Technol. 2019, 171, 190–198. [Google Scholar] [CrossRef]
- Zhou, L.J.; Zhai, S.C.; Chen, Y.M.; Xu, Z.Y. Anisotropic cellulose nanofibers/polyvinyl alcohol/graphene aerogels fabricated by directional freeze-drying as effective oil adsorbents. Polymers 2019, 11, 712. [Google Scholar] [CrossRef]
- He, X.; Cheng, L.; Wang, Y.R.; Zhao, J.Q.; Zhang, W.; Lu, C.H. Aerogels from quaternary ammonium-functionalized cellulose nanofibers for rapid removal of Cr(VI) from water. Carbohydr. Polym. 2014, 111, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Baig, N.; Alghunaimi, F.I.; Saleh, T.A. Hydrophobic and oleophilic carbon nanofiber impregnated styrofoam for oil and water separation: A green technology. Chem. Eng. J. 2019, 360, 1613–1622. [Google Scholar] [CrossRef]
- Xiao, J.L.; Lv, W.Y.; Song, Y.H.; Zheng, Q. Graphene/nanofiber aerogels: Performance regulation towards multiple applications in dye adsorption and oil/water separation. Chem. Eng. J. 2018, 338, 202–210. [Google Scholar] [CrossRef]
- Fu, Q.X.; Liu, L.F.; Si, Y.; Yu, J.Y.; Ding, B. Shapeable, underwater superelastic, and highly phosphorylated nanofibrous aerogels for large-capacity and high-throughput protein separation. ACS Appl. Mater. Int. 2019, 11, 44874–44885. [Google Scholar] [CrossRef]
- Zhan, Z.Y.; Song, Q.C.; Zhou, Z.H.; Lu, C.H. Ultrastrong and conductive MXene/cellulose nanofiber films enhanced by hierarchical nano-architecture and interfacial interaction for flexible electromagnetic interference shielding. J. Mater. Chem. C 2019, 7, 9820–9829. [Google Scholar] [CrossRef]
- Pitkanen, O.; Tolvanen, J.; Szenti, I.; Kukovecz, A.; Hannu, J.; Jantunen, H.; Kordas, K. Lightweight hierarchical carbon nanocomposites with highly efficient and tunable electromagnetic interference shielding properties. ACS Appl. Mater. Interf. 2019, 11, 19331–19338. [Google Scholar] [CrossRef]
- Lv, J.; Gu, W.H.; Cui, X.Q.; Dai, S.S.; Zhang, B.S.; Ji, G.B. Nanofiber network with adjustable nanostructure controlled by PVP content for an excellent microwave absorption. Sci. Rep. 2019, 9, 4271. [Google Scholar] [CrossRef]
- Hong, X.H.; Chung, D.D.L. Carbon nanofiber mats for electromagnetic interference shielding. Carbon 2017, 111, 529–537. [Google Scholar] [CrossRef]
- Xu, W.Z.; Xing, Y.; Liu, J.; Wu, H.P.; Cuo, Y.; Li, D.W.; Guo, D.Y.; Li, C.R.; Liu, A.P.; Bai, H. Efficient water transport and solar steam generation via radially, hierarchically structured aerogels. ACS Nano 2019, 13, 7930–7938. [Google Scholar] [CrossRef]
- Jiang, F.; Liu, H.; Li, Y.; Kuang, Y.D.; Xu, X.; Chen, C.J.; Huang, H.; Jia, C.; Zhao, X.P.; Hitz, E.; et al. Lightweight, mesoporous, and highly absorptive all-nanofiber aerogel for efficient solar steam generation. ACS Appl. Mater. Interf. 2018, 10, 1104–1112. [Google Scholar] [CrossRef]
- Chen, Z.R.; Wei, C.; Gong, Y.Y.; Lv, J.; Xu, Z.H.; Hu, J.Y.; Du, L.L. Preparation and electrochemical performances of cellulose nanofiber/graphene nanosheet/polyaniline composite film via in-situ polymerization. Int. J. Electrochem. Sci. 2017, 12, 6662–6675. [Google Scholar] [CrossRef]
- Isogai, A. Structural characterization and modifications of surface-oxidized cellulose nanofiber. J. Jpn. Pet. Inst. 2015, 58, 365–375. [Google Scholar] [CrossRef]
- Liu, Q.Z.; Jing, S.S.; Wang, S.; Zhuo, H.; Zhong, L.X.; Peng, X.W.; Sun, R.C. Flexible nanocomposites with ultrahigh specific area capacitance and tunable properties based on a cellulose derived nanofiber-carbon sheet framework coated with polyaniline. J. Mater. Chem. A 2016, 4, 13352–13362. [Google Scholar] [CrossRef]
- Boday, D.J.; Muriithi, B.; Stover, R.J.; Loy, D.A. Polyaniline nanofiber-silica composite aerogels. J. Non-Cryst. Solids 2012, 358, 1575–1580. [Google Scholar] [CrossRef]
- Dourani, A.; Haghgoo, M.; Hamadanian, M. Multi-walled carbon nanotube and carbon nanofiber/polyacrylonitrile aerogel scaffolds for enhanced epoxy resins. Comp. Part B Eng. 2019, 176, 107299. [Google Scholar] [CrossRef]
- Li, C.; Ding, Y.W.; Hu, B.C.; Wu, Z.Y.; Gao, H.L.; Liang, H.W.; Chen, J.F.; Yu, S.H. Temperature-invariant superelastic and fatigue resistant carbon nanofiber aerogels. Adv. Mater. 2020, 32, 1904331. [Google Scholar] [CrossRef]
- Wang, C.; Pan, Z.Z.; Lv, W.; Liu, B.L.; Wei, J.; Lv, X.H.; Luo, Y.; Nishihara, H.; Yang, Q.H. A directional strain sensor based on anisotropic microhoneycomb cellulose nanofiber-carbon nanotube hybrid aerogels prepared by unidirectional freeze drying. Small 2019, 15, 1805363. [Google Scholar] [CrossRef]
- Yao, Q.F.; Fan, B.T.; Xiong, Y.; Wang, C.; Wang, H.W.; Jin, C.D.; Sun, Q.F. Stress sensitive electricity based on Ag/cellulose nanofiber aerogel for self-reporting. Carbohydr. Polym. 2017, 168, 265–273. [Google Scholar] [CrossRef]
- Wang, M.; Anoshkin, I.V.; Nasibulin, A.G.; Korhonen, J.T.; Seitsonen, J.; Pere, J.; Kauppinen, E.I.; Ras, R.H.A.; Ikkala, O. Modifying native nanocellulose aerogels with carbon nanotubes for mechanoresponsive conductivity and pressure sensing. Adv. Mater. 2013, 25, 2428–2432. [Google Scholar] [CrossRef]
- Hu, P.Y.; Lyu, J.; Fu, C.; Gong, W.B.; Liao, J.H.; Lu, W.B.; Chen, Y.P.; Zhang, X.T. Multifunctional aramid nanofiber/carbon nanotube hybrid aerogel films. ACS Nano 2020, 14, 688–697. [Google Scholar] [CrossRef]
- Wu, Z.Y.; Liang, H.W.; Chen, L.F.; Hu, B.C.; Yu, S.H. Bacterial cellulose: A robust platform for design of three dimensional carbon-based functional nanomaterials. Accounts Chem. Res. 2016, 49, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wu, Z.Y.; Liang, H.W.; Chen, J.F.; Yu, S.H. Ultralight multifunctional carbon-based aerogels by combining graphene oxide and bacterial cellulose. Small 2017, 13, 1700453. [Google Scholar] [CrossRef] [PubMed]
- Lai, F.L. Electrospun carbon nanofiber aerogel meets polyimide-derived carbon sheets: A rational structural design for environmental protection and energy storage. Abstr. Pap. Am. Chem. Soc. 2016, 252, 493. [Google Scholar]
- Yang, X.; Kong, L.Y.; Ma, J.F.; Liu, X.G. Facile construction of hierarchically porous carbon nanofiber aerogel for high-performance supercapacitor. J. Appl. Electrochem. 2019, 49, 241–250. [Google Scholar] [CrossRef]
- Li, W.Y.; Islam, N.; Ren, G.F.; Li, S.Q.; Fan, Z.Y. AC-filtering supercapacitors based on edge oriented vertical graphene and cross-linked carbon nanofiber. Materials 2019, 12, 604. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.C.; Jiao, Y.; Liang, D.X.; Wu, Y.Q.; Li, J. A high-performance, all-textile and spirally wound asymmetric supercapacitors based on core-sheath structured MnO2 nanoribbons and cotton-derived carbon cloth. Electrochim. Acta 2018, 285, 262–271. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.C.; Jiang, L. Freestanding carbon aerogels produced from bacterial cellulose and its Ni/MnO2/Ni(OH)2 decoration for supercapacitor electrodes. J. Appl. Electrochem. 2018, 48, 495–507. [Google Scholar] [CrossRef]
- Islam, N.; Hoque, M.N.F.; Zu, Y.J.; Wang, S.; Fan, Z.Y. Carbon nanofiber aerogel converted from bacterial cellulose for kilohertz AC-supercapacitors. MRS Adv. 2018, 3, 855–860. [Google Scholar] [CrossRef]
- Islam, N.; Li, S.Q.; Ren, G.F.; Zu, Y.J.; Warzywoda, J.; Wang, S.; Fan, Z.Y. High-frequency electrochemical capacitors based on plasma pyrolyzed bacterial cellulose aerogel for current ripple filtering and pulse energy storage. Nano Energy 2017, 40, 107–114. [Google Scholar] [CrossRef]
- Li, L.; Zhang, M.Y.; Zhang, X.T.; Zhang, Z.G. New Ti3C2 aerogel as promising negative electrode materials for asymmetric supercapacitors. J. Power Sources 2017, 364, 234–241. [Google Scholar] [CrossRef]
- Lai, F.L.; Huang, Y.P.; Zuo, L.Z.; Gu, H.H.; Miao, Y.E.; Liu, T.X. Electrospun nanofiber-supported carbon aerogel as a versatile platform toward asymmetric supercapacitors. J. Mater. Chem. A 2016, 4, 15861–15869. [Google Scholar] [CrossRef]
- Guo, C.X.; Yilmaz, G.; Chen, S.C.; Chen, S.F.; Lu, X.M. Hierarchical nanocomposite composed of layered V2O5/PEDOT/MnO2 nanosheets for high-performance asymmetric supercapacitors. Nano Energy 2015, 12, 76–87. [Google Scholar] [CrossRef]
- Yang, F.; Xu, M.W.; Bao, S.J.; Wei, H.; Chai, H. Self-assembled hierarchical graphene/polyaniline hybrid aerogels for electrochemical capacitive energy storage. Electrochim. Acta 2014, 137, 381–387. [Google Scholar] [CrossRef]
- Gao, K.Z.; Shao, Z.Q.; Li, J.; Wang, X.; Peng, X.Q.; Wang, W.J.; Wang, F.J. Cellulose nanofiber-graphene all solid-state flexible supercapacitors. J. Mater. Chem. A 2013, 1, 63–67. [Google Scholar] [CrossRef]
- Li, Q.Q.; Liu, F.; Zhang, L.; Nelson, B.J.; Zhang, S.H.; Ma, C.; Tao, X.Y.; Cheng, J.P.; Zhang, X.B. In situ construction of potato starch based carbon nanofiber/activated carbon hybrid structure for high-performance electrical double layer capacitor. J. Power Sources 2012, 207, 199–204. [Google Scholar] [CrossRef]
- Ghosh, A.; Ra, E.J.; Jin, M.; Jeong, H.K.; Kim, T.H.; Biswas, C.; Lee, Y.H. High pseudocapacitance from ultrathin V2O5 films electrodeposited on self-standing carbon-nanofiber paper. Adv. Funct. Mater. 2011, 21, 2541–2547. [Google Scholar] [CrossRef]
- Wu, P.; Hu, H.Y.; Xie, N.; Wang, C.; Wu, F.; Pan, M.; Li, H.F.; Wang, X.D.; Zeng, Z.L.; Deng, S.G.; et al. A N-doped graphene-cobalt nickel sulfide aerogel as a sulfur host for lithium-sulfur batteries. RSC Adv. 2019, 9, 32247–32257. [Google Scholar] [CrossRef]
- Zhu, L.; Jiang, H.T.; Ran, W.X.; You, L.J.; Yao, S.S.; Shen, X.Q.; Tu, F.Y. Turning biomass waste to a valuable nitrogen and boron dual-doped carbon aerogel for high performance lithium-sulfur batteries. Appl. Surf. Sci. 2019, 489, 154–164. [Google Scholar] [CrossRef]
- Zhang, M.; Huang, X.X.; Xin, H.L.; Li, D.Z.; Zhao, Y.; Shi, L.D.; Lin, Y.M.; Yu, J.L.; Yu, Z.Q.; Zhu, C.Z.; et al. Coaxial electrospinning synthesis hollow Mo2C@C core-shell nanofibers for high-performance and long-term lithium-ion batteries. Appl. Surf. Sci. 2019, 473, 352–358. [Google Scholar] [CrossRef]
- Cao, D.X.; Xing, Y.J.; Tantratian, K.; Wang, X.; Ma, Y.; Mukhopadhyay, A.; Cheng, Z.; Zhang, Q.; Jiao, Y.C.; Chen, L.; et al. 3D printed high-performance lithium metal microbatteries enabled by nanocellulose. Adv. Mater. 2019, 31, 1807313. [Google Scholar] [CrossRef]
- Lv, C.X.; Liu, H.L.; Li, D.H.; Chen, S.; Zhang, H.W.; She, X.L.; Guo, X.X.; Yang, D.J. Ultrafine FeSe nanoparticles embedded into 3D carbon nanofiber aerogels with FeSe/Carbon interface for efficient and long-life sodium storage. Carbon 2019, 143, 106–115. [Google Scholar] [CrossRef]
- Yang, Z.Y.; Zhang, P.; Wang, J.; Yan, Y.; Yu, Y.; Wang, Q.H.; Liu, M.K. Hierarchical carbon@SnS2 aerogel with “skeleton/skin” architectures as a high-capacity, high-rate capability and long cycle life anode for sodium ion storage. ACS Appl. Mater. Interfaces 2018, 10, 37434–37444. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.T.; Yu, C.; Li, S.F.; Guo, W.; Zhao, Y.; Dong, Q.; Lin, X.T.; Song, Z.X.; Tan, X.Y.; Wang, C.H.; et al. Ultrahigh-capacity and long-life lithium-metal batteries enabled by engineering carbon nanofiber-stabilized graphene aerogel film host. Small 2018, 14, 1803310. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.; Chen, D.J. Fabrication of nano-sized attapulgite-based aerogels as anode material for lithium ion batteries. J. Mater. Sci. 2018, 53, 2054–2064. [Google Scholar] [CrossRef]
- Zhu, L.; You, L.J.; Zhu, P.H.; Shen, X.Q.; Yang, L.Z.; Xiao, K.S. High performance lithium-sulfur batteries with a sustainable and environmentally friendly carbon aerogel modified separator. ACS Sustain. Chem. Eng. 2018, 6, 248–257. [Google Scholar] [CrossRef]
- Kong, X.L.; Lu, Y.; Ye, G.C.; Li, D.H.; Sun, J.; Yang, D.J.; Yin, Y.F. Nanofibrillated cellulose derived hierarchical porous carbon aerogels: Efficient anode material for lithium ion battery. Chem. J. Chin. Univ. Chin. 2017, 38, 1941–1946. [Google Scholar] [CrossRef]
- Li, S.Q.; Warzywoda, J.; Wang, S.; Ren, G.F.; Fan, Z.Y. Bacterial cellulose derived carbon nanofiber aerogel with lithium polysulfide catholyte for lithium-sulfur batteries. Carbon 2017, 124, 212–218. [Google Scholar] [CrossRef]
- Cui, J.; Yao, S.S.; Huang, J.Q.; Qin, L.; Chong, W.G.; Sadighi, Z.; Huang, J.Q.; Wang, Z.Y.; Kim, J.K. Sb-doped SnO2/graphene-CNT aerogels for high performance Li-ion and Na-ion battery anodes. Energy Storage Mater. 2017, 9, 85–95. [Google Scholar] [CrossRef]
- Wutthiprom, J.; Phattharasupakun, N.; Khuntilo, J.; Maihom, T.; Limtrakul, J.; Sawangphruk, M. Collaborative design of Li-S batteries using 3D N-doped graphene aerogel as a sulfur host and graphitic carbon nitride paper as an interlayer. Sus. Energy Fuels 2017, 1, 1759–1765. [Google Scholar] [CrossRef]
- Wan, Y.Z.; Yang, Z.W.; Xiong, G.Y.; Guo, R.S.; Liu, Z.; Luo, H.L. Anchoring Fe3O4 nanoparticles on three-dimensional carbon nanofibers toward flexible high-performance anodes for lithium-ion batteries. J. Power Sources 2015, 294, 414–419. [Google Scholar] [CrossRef]
- Wan, Y.Z.; Yang, Z.W.; Xiong, G.Y.; Luo, H.L. A general strategy of decorating 3D carbon nanofiber aerogels derived from bacterial cellulose with nano-Fe3O4 for high-performance flexible and binder-free lithium-ion battery anodes. J. Mater. Chem. A 2015, 3, 15386–15393. [Google Scholar] [CrossRef]
- Liang, H.W.; Wu, Z.Y.; Chen, L.F.; Li, C.; Yu, S.H. Bacterial cellulose derived nitrogen-doped carbon nanofiber aerogel: An efficient metal-free oxygen reduction electrocatalyst for zinc-air battery. Nano Energy 2015, 11, 366–376. [Google Scholar] [CrossRef]
- Garakani, M.A.; Abouali, S.; Zhang, B.; Takagi, C.A.; Xu, Z.L.; Huang, J.Q.; Huang, J.Q.; Kim, J.K. Cobalt carbonate/and cobalt oxide/graphene aerogel composite anodes for high performance Li-ion batteries. ACS Appl. Mater. Interf. 2014, 6, 18971–18980. [Google Scholar] [CrossRef] [PubMed]
- Sides, C.R.; Li, N.C.; Patrissi, C.J.; Scrosati, B.; Martin, C.R. Nanoscale materials for lithium-ion batteries. MRS Bull. 2002, 27, 604–607. [Google Scholar] [CrossRef]
- Ye, G.C.; Zhu, X.Y.; Chen, S.; Li, D.H.; Yin, Y.F.; Lu, Y.; Komarneni, S.; Yang, D.J. Nanoscale engineering of nitrogen-doped carbon nanofiber aerogels for enhanced lithium ion storage. J. Mater. Chem. A 2017, 5, 8247–8254. [Google Scholar] [CrossRef]
- Xu, C.Y.; Dai, J.C.; Teng, X.G.; Zhu, Y.M. Preparation of a new carbon nanofiber as a high-capacity air electrode for nonaqueous lithium-oxygen batteries. Chemcatchem 2016, 8, 3725–3731. [Google Scholar] [CrossRef]
- Bian, Z.H.; Xu, Y.; Yuan, T.; Peng, C.X.; Pang, Y.P.; Yang, J.H.; Zheng, S.Y. Hierarchically designed CNF/S-Cu/CNF nonwoven electrode as free-standing cathode for lithium-sulfur batteries. Batter. Supercaps 2019, 2, 560–567. [Google Scholar] [CrossRef]
- Li, D.H.; Wang, Y.; Sun, Y.Y.; Lu, Y.; Chen, S.; Wang, B.B.; Zhang, H.W.; Xia, Y.Z.; Yang, D.J. Turning gelidium amansii residue into nitrogen-doped carbon nanofiber aerogel for enhanced multiple energy storage. Carbon 2018, 137, 31–40. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Wang, Y.Y.; Peng, B.; Yu, W.T.; Wang, H.Y.; Wang, T.; Deng, B.W.; Chai, L.Y.; Zhang, K.; Wang, J.X. Preparation of a macroscopic, robust carbon-fiber monolith from filamentous fungi and its application in Li-S batteries. Green Chem. 2014, 16, 3926–3934. [Google Scholar] [CrossRef]
- Li, S.Q.; Mou, T.; Ren, G.F.; Warzywoda, J.; Wei, Z.D.; Wang, B.; Fan, Z.Y. Gel based sulfur cathodes with a high sulfur content and large mass loading for high-performance lithium-sulfur batteries. J. Mater. Chem. A 2017, 5, 1650–1657. [Google Scholar] [CrossRef]
- Ding, M.; Bannuru, K.K.R.; Wang, Y.; Guo, L.; Baji, A.; Yang, H.Y. Free-standing electrodes derived from metal-organic frameworks/nanofibers hybrids for membrane capacitive deionization. Adv. Mater. Technol. 2018, 3, 1800135. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, H.Y.; Xu, H.F.; Zhang, L. Enhanced capacitive deionization by nitrogen-doped porous carbon nanofiber aerogel derived from bacterial-cellulose. J. Electroanalyt. Chem. 2018, 822, 81–88. [Google Scholar] [CrossRef]
- Luo, G.M.; Wang, Y.Z.; Gao, L.X.; Zhang, D.Q.; Lin, T. Graphene bonded carbon nanofiber aerogels with high capacitive deionization capability. Electrochim. Acta 2018, 260, 656–663. [Google Scholar] [CrossRef]
- Liu, X.; Chen, T.; Qiao, W.C.; Wang, Z.; Yu, L. Fabrication of graphene/activated carbon nanofiber composites for high performance capacitive deionization. J. Taiwan Inst. Chem. Eng. 2017, 72, 213–219. [Google Scholar] [CrossRef]
- Li, Y.Z.; Jiang, Y.P.; Wang, T.J.; Zhang, C.; Wang, H.F. Performance of fluoride electrosorption using micropore-dominant activated carbon as an electrode. Separat. Purif. Technol. 2017, 172, 415–421. [Google Scholar] [CrossRef]
- Kohli, D.K.; Bhartiya, S.; Singh, A.; Singh, R.; Singh, M.K.; Gupta, P.K. Capacitive deionization of ground water using carbon aerogel based electrodes. Desalinat. Water Treat. 2016, 57, 26871–26879. [Google Scholar] [CrossRef]
- Wang, G.; Dong, Q.; Wu, T.T.; Zhan, F.; Zhou, M.; Qiu, J.S. Ultrasound-assisted preparation of electrospun carbon fiber/graphene electrodes for capacitive deionization: Importance and unique role of electrical conductivity. Carbon 2016, 103, 311–317. [Google Scholar] [CrossRef]
- Dong, Q.; Wang, G.; Qian, B.Q.; Hu, C.; Wang, Y.W.; Qiu, J.S. Electrospun composites made of reduced graphene oxide and activated carbon nanofibers for capacitive deionization. Electrochim. Acta 2014, 137, 388–394. [Google Scholar] [CrossRef]
- Liu, J.Y.; Wang, S.P.; Yang, J.M.; Liao, J.J.; Lu, M.; Pan, H.J.; An, L. ZnCl2 activated electrospun carbon nanofiber for capacitive desalination. Desalination 2014, 344, 446–453. [Google Scholar] [CrossRef]
- Meng, F.L.; Li, L.; Wu, Z.; Zhong, H.X.; Li, J.C.; Yan, J.M. Facile preparation of N-doped carbon nanofiber aerogels from bacterial cellulose as an efficient oxygen reduction reaction electrocatalyst. Chin. J. Catal. 2014, 35, 877–883. [Google Scholar] [CrossRef]
- El-Deen, A.G.; Barakat, N.A.M.; Khalil, K.A.; Kim, H.Y. Development of multi-channel carbon nanofibers as effective electrosorptive electrodes for a capacitive deionization process. J. Mater. Chem. A 2013, 1, 11001–11010. [Google Scholar] [CrossRef]
- Nie, C.Y.; Zhan, Y.K.; Pan, L.K.; Li, H.B.; Sun, Z. Electrosorption of different cations and anions with membrane capacitive deionization based on carbon nanotube/nanofiber electrodes and ion-exchange membranes. Desalinat. Water Treat. 2011, 30, 266–271. [Google Scholar] [CrossRef]
- Wang, X.Z.; Pan, L.K.; Li, M.G.; Chen, Y.W.; Cheng, R.M.; Huang, S.M.; Sun, Z. Effect of acetylene flow rate on electrosorption capacity of carbon nanotube and nanofiber composite film electrodes. Surf. Rev. Lett. 2007, 14, 135–139. [Google Scholar] [CrossRef]
- Gao, R.N.; Lu, Y.; Xiao, S.L.; Li, J. Facile fabrication of nanofibrillated chitin/Ag2O heterostructured aerogels with high iodine capture efficiency. Sci. Rep. 2017, 7, 4303. [Google Scholar] [CrossRef]
- Xu, X.T.; Liu, Y.; Lu, T.; Sun, Z.; Chua, D.H.C.; Pan, L.K. Rational design and fabrication of graphene/carbon nanotubes hybrid sponge for high-performance capacitive deionization. J. Mater. Chem. A 2015, 3, 13418–13425. [Google Scholar] [CrossRef]
- Wang, G.; Dong, Q.; Ling, Z.; Pan, C.; Yu, C.; Qiu, J.S. Hierarchical activated carbon nanofiber webs with tuned structure fabricated by electrospinning for capacitive deionization. J. Mater. Chem. 2012, 22, 21819–21823. [Google Scholar] [CrossRef]
- Liu, W.J.; Zhu, H.; Ying, L.R.; Zhu, Z.F.; Li, H.N.; Lu, S.L.; Duan, F.; Du, M.L. In situ synthesis of small Pt nanoparticles on chitin aerogel derived N doped ultra-thin carbon nanofibers for superior hydrogen evolution catalysis. New J. Chem. 2019, 43, 16490–16496. [Google Scholar] [CrossRef]
- Zhou, Y.; Cheng, Q.Q.; Huang, Q.H.; Zou, Z.Q.; Yan, L.M.; Yang, H. Highly dispersed cobalt-nitrogen co-doped carbon nanofiber as oxygen reduction reaction catalyst. Acta Phys. Chim. Sin. 2017, 33, 1429–1435. [Google Scholar] [CrossRef]
- Li, R.C.; Shao, X.F.; Li, S.S.; Cheng, P.P.; Hu, Z.X.; Yuan, D.S. Metal-free N-doped carbon nanofibers as an efficient catalyst for oxygen reduction reactions in alkaline and acid media. Nanotechnology 2016, 27, 505402. [Google Scholar] [CrossRef]
- Kolla, P.; Lai, C.L.; Mishra, S.; Fong, H.; Rhine, W.; Smirnova, A. CVD grown CNTs within iron modified and graphitized carbon aerogel as durable oxygen reduction catalysts in acidic medium. Carbon 2014, 79, 518–528. [Google Scholar] [CrossRef]
- Pajarito, B.B.; Llorens, C.; Tsuzuki, T. Effects of ammonium chloride on the yield of carbon nanofiber aerogels derived from cellulose nanofibrils. Cellulose 2019, 26, 7727–7740. [Google Scholar] [CrossRef]
- Simon-Herrero, C.; Caminero-Huertas, S.; Romero, A.; Valverde, J.L.; Sanchez-Silva, L. Effects of freeze-drying conditions on aerogel properties. J. Mater. Sci. 2016, 51, 8977–8985. [Google Scholar] [CrossRef]
- Sufiani, O.; Elisadiki, J.; Machunda, R.L.; Jande, Y.A.C. Modification strategies to enhance electrosorption performance of activated carbon electrodes for capacitive deionization applications. J. Electroanalyt. Chem. 2019, 848, 113328. [Google Scholar] [CrossRef]
- White, R.J.; Brun, N.; Budarin, V.L.; Clark, J.H.; Titirici, M.M. Always look on the ‘light’ side of life: Sustainable carbon aerogels. Chemsuschem 2014, 7, 670–689. [Google Scholar] [CrossRef] [PubMed]
- Vivod, S.L.; Meador, M.A.B.; Cakmak, M.; Guo, H.Q. Carbon nanofiber/polyimide aerogel thin films. Abstr. Paper Am. Chem. Soc. 2011, 242, 338-POLY. [Google Scholar]
- Korhonen, J.T.; Hiekkataipale, P.; Malm, J.; Karppinen, M.; Ikkala, O.; Ras, R.H.A. Inorganic hollow nanotube aerogels by atomic layer deposition onto native nanocellulose templates. ACS Nano 2011, 5, 1967–1974. [Google Scholar] [CrossRef]
- Lu, W.J.; Steigerwalt, E.S.; Moore, J.T.; Sullivan, L.M.; Collins, W.E.; Lukehart, C.M. Carbothermal transformation of a graphitic carbon nanofiber/silica aerogel composite to a SiC/silica nanocomposite. J. Nanosci. Nanotechnol. 2004, 4, 803–808. [Google Scholar] [CrossRef]
- Gan, Y.X. Structural assessment of nanocomposites. Micron 2012, 43, 782–817. [Google Scholar] [CrossRef]







© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gan, Y.X.; Gan, J.B. Advances in Manufacturing Composite Carbon Nanofiber-Based Aerogels. J. Compos. Sci. 2020, 4, 73. https://doi.org/10.3390/jcs4020073
Gan YX, Gan JB. Advances in Manufacturing Composite Carbon Nanofiber-Based Aerogels. Journal of Composites Science. 2020; 4(2):73. https://doi.org/10.3390/jcs4020073
Chicago/Turabian StyleGan, Yong X., and Jeremy B. Gan. 2020. "Advances in Manufacturing Composite Carbon Nanofiber-Based Aerogels" Journal of Composites Science 4, no. 2: 73. https://doi.org/10.3390/jcs4020073
APA StyleGan, Y. X., & Gan, J. B. (2020). Advances in Manufacturing Composite Carbon Nanofiber-Based Aerogels. Journal of Composites Science, 4(2), 73. https://doi.org/10.3390/jcs4020073




